Abstract
Objective
To test the proof-of-principle that genetically-engineered mesenchymal stem cells (MSCs) transfected with the human hyperpolarization-activated cyclic nucleotide-gated channel 1 (hHCN1) gene can be modified to become cardiac pacemaker cells.
Methods
MSCs were transfected with the hHCN1 gene using lentiviral-based transfection. The expressed pacemaker current (If) in hHCN1-transfected MSCs was recorded using whole-cell patch-clamp analysis. The effect of the hHCN1-transfected MSCs on cardiomyocyte excitability was determined by coculturing the MSCs with neonatal rabbit ventricular myocytes (NRVM). The spontaneous action potentials of the NRVM were recorded by whole-cell current-clamp analysis.
Results
A high level time- and voltage-dependent inward hyperpolarization current that was inhibited by 4 mM caesium chloride was detected in hHCN1-transfected MSCs, suggesting that the HCN1 proteins acted as If channels in MSCs. The mean ± SE beating frequency in NRVMs cocultured with control MSCs transfected with the pcDNA3 plasmid control was 82 ± 8 beats/min (n = 5) compared with 129 ± 11 beats/min (n = 5) in NRVMs cocultured with hHCN1-transfected MSCs.
Conclusions
Genetically-engineered MSCs transfected with the hHCN1 gene can be modified to become cardiac pacemaker cells.
Introduction
Research has suggested that application of the hyperpolarization-activated cyclic nucleotide-gated channel (HCN) gene may hold great promise in the development of biological pacemakers. 1 The evidence for this hypothesis includes: (i) the expression of four HCN genes varies among different cardiac cells and the main difference between pacemaker cells and common cardiomyocytes depends on which HCN genes are fully expressed; (ii) animals lacking HCN channels show a pronounced cardiac phenotype, which is due to dysfunction of the sinoatrial node (SAN); (iii) it is suggested that primary defects in HCN channels might underlie certain cardiac arrhythmias in several families with hereditary sinus node dysfunction.2–4
In a previous study by this research team, cardiac excitability was modified by genetically-engineered mesenchymal stem cells (MSCs) transfected with the human HCN4 gene using lentiviral vector (LentiV). 5 With this in mind, this present study tested the proof-of-principle that genetically-engineered MSCs transfected with the human HCN1 (hHCN1) gene can be modified to become cardiac pacemaker cells.
Materials and methods
Animals
This study protocol was reviewed and approved by the Animal Care and Use Committee for Teaching and Research of Soochow University, Suzhou, Jiangsu Province, China (Permit Number: 12-1156). New Zealand neonatal rabbits (1–3 days old; weight, 50–60 g) and adult New Zealand rabbits (90–100 days old; weight, 2500–3000 g) were purchased from the Laboratory Animal Research Centre of Soochow University. The neonatal rabbits were used immediately on receipt from the supplier. The adults were kept under standard animal husbandry conditions (12-h light/12-h dark cycle) and had free access to food and water.
MSC preparation
The MSCs were isolated from adult New Zealand rabbits as previously described. 5 The MSCs were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS) at 37℃ in a humidified atmosphere of 5% CO2. Cells from passages 2–4 were used for the hHCN1 gene transfection studies.
Isolation and culture of NRVM
A standard trypsin dissociation method was used to isolate neonatal rabbit ventricular myocytes (NRVM) from neonatal New Zealand rabbits as described previously. 6 The cells were plated at a final density of 1 × 105−2 × 105 cells/ml. For the first 72 h of cell culture, 10 µmol/l 5-bromo-2-deoxyuridine (Sigma-Aldrich, St Louis, MO, USA) was applied to inhibit fibroblast proliferation. Prior to the coculture experiments, NRVM were labelled with 50 µmol/l fluorescent dye 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (Roche, Basel, Switzerland) for 24 h at 37℃, to distinguish them from the MSCs when they were viewed under a fluorescence microscope (CFM-500; Changfang Group, Shanghai, China).
LentiV-mediated stable genetic modification of MSCs
For stable genetic modification of the MSCs with the GFP-hHCN1 gene or the pcDNA3 plasmid control, self-inactivating HIV-based LentiV was used for transgene delivery, as previously described. 5 The LentiV-green fluorescent protein (GFP) was constructed with the plasmid hHCN1/pcDNA3 by Telebio Biomedical Company (Shanghai, China). The MSCs underwent stable transfection and colony selection, as previously described. 5
RT–PCR analysis
The levels of hHCN1 mRNA in MSCs transfected with the hHCN1 gene and control MSCs transfected with pcDNA3 plasmid control were determined using reverse transcription–polymerase chain reaction (RT–PCR). Total RNA was extracted from 1 × 106 MSCs using TRIzol® reagent according to the manufacturer’s instructions (Life Technologies, Grand Island, NY, USA). Total RNA (3 µg) was reverse transcribed to cDNA in a volume of 20 µl using a ThermoScript™ One-Step RT–PCR System for Long Templates Kit (Telebio Biomedical Company); spectrophotometry was used to quantify cDNA levels according to the manufacturer’s instructions. PCR was performed using a PCR kit (CloneEZ® PCR Cloning Kit; Telebio Biomedical Company) according to the manufacturer’s instructions. The PCR primers for hHCN1 were: 5′-CGCCTGATCCGCTACATCCAT-3′ (sense) and 5′-AGTGCGAAGGAGTACAGTTCACT-3′ (antisense) (Telebio Biomedical Company). All PCR cycles were performed in a Mastercycler® gradient thermal cycler (Eppendorf, Hamburg, Germany).The cycling programme involved preliminary denaturation at 95℃ for 5 min, followed by 40 cycles of denaturation at 94℃ for 30 s, annealing at 60℃ for 40 s, and elongation at 72℃ for 60 s, followed by a final elongation step at 72℃ for 5 min. The PCR products were separated on 1.5% agarose gels and visualized using ethidium bromide staining and ultraviolet light. The size of the hHCN1 PCR product was 3672 base pairs.
Electrophysiological recording of cardiac pacemaker current expressed in MSCs and spontaneous action potentials in NRVM
Whole-cell patch-clamp analysis was used to study membrane currents in the MSCs transfected with the pcDNA3 plasmid control and those transfected with the hHCN1 gene. Expressed cardiac pacemaker current If (i.e. IHCN1) was measured as described previously. 5 After the If was recorded, cells were superfused with extracellular solution containing 4 mM caesium chloride and the currents were measured accordingly.
Action potentials were recorded using the whole-cell current-clamp configuration in spontaneously beating NRVM using an Axopatch-700B amplifier and pClamp® 9.0 software, version 9.2.1.8 (Axon Instruments [Molecular Devices], Sunnyvale, CA, USA), as described previously. 6
MSC coculture with NRVM
To test whether the MSCs coupled electrically with cardiac myocytes, 5 × 105 MSCs transfected with the hHCN1 gene, or MSCs transfected with the pcDNA3 plasmid control, were cocultured with 5 × 105
NRVM. The cells were cocultured in DMEM containing 10% FBS at 37℃ in a humidified atmosphere of 5% CO2. The beating rate of the NRVM after 3 days’ coculture with the MSCs was determined from the action potential recordings, as described previously. 5
Statistical analyses
All statistical analyses were performed using the SPSS® statistical software package, version 13.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Data were presented as mean ± SE. Analysis of variance was used for multiple comparisons. A P-value < 0.05 was considered statistically significant.
Results
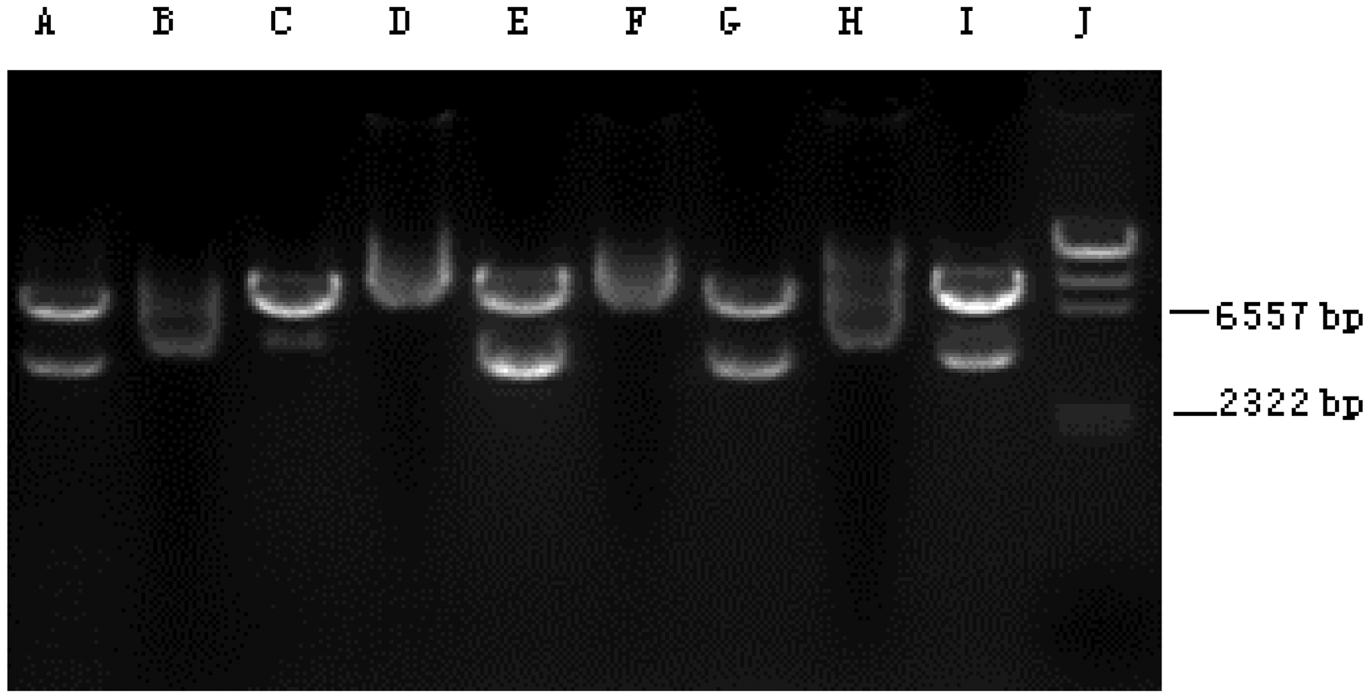
A GFP immunofluorescence image, indicating that the MSCs had been successfully transfected with the GFP-hHCN1 gene, is shown in Figure 1. Control MSCs transfected with the control pcDNA3 plasmid were negative for GFP immunofluorescence (image not shown). Using RT–PCR analysis, the level of hHCN1 mRNA was shown to be similar in different clones of MSCs transfected with the hHCN1 gene, suggesting a similar degree of efficiency of the cell transcription/translation machinery for the individual MSC clones; MSCs transfected with the control pcDNA3 plasmid demonstrated no hHCN1 mRNA (Figure 2).
Green fluorescent protein (GFP) immunofluorescence image, indicating the successful transfection of mesenchymal stem cells with the GFP-hHCN1 gene. The colour version of this figure is available at: http://imr.sagepub.com. Reverse transcription–polymerase chain reaction products from individual clones of mesenchymal stem cells transfected with either the hHCN1 gene (the 3672 base pair [bp] band in lanes A, E, G and I is the hHCN1 mRNA) or the control pcDNA3 plasmid (lanes B, C, D, F, and H). Lane J: molecular weight markers.
An important characteristic of a cardiac pacemaking myocyte is the expression of an inward If.
7
Using of the whole-cell configuration of the patch-clamp technique, If was elicited in hHCN1-tranfected MSCs using hyperpolarizing steps in 10-mV increments from −40 mV to −140 mV from a holding potential of −40 mV and was voltage-dependent. The voltage threshold for activation of If was approximately −80 mV in hHCN1-tranfected MSCs (Figure 3A). All of the hHCN1-tranfected MSCs exhibited a large caesium-sensitive If that was inhibited by 4 mM caesium chloride (Figure 3B), thereby confirming that this current was the If.7,8 An If was not elicited in control MSCs transfected with the control pcDNA3 plasmid (Figure 3C).
Cardiac pacemaker current (If) versus time traces recorded for mesenchymal stem cells (MSCs) transfected with the hHCN1 gene are shown to depict If currents during hyperpolarization. (A) If was elicited in MSCs transfected with the hHCN1 gene from −40 mV to −140 mV, from a holding potential of −40 mV, and was voltage-dependent. The threshold voltage for activation of If was approximately −80 mV. (B) In all of the hHCN1-tranfected cells, the If was inhibited by 4 mM caesium chloride. (C) An If was not elicited in control MSCs transfected with the control pcDNA3 plasmid.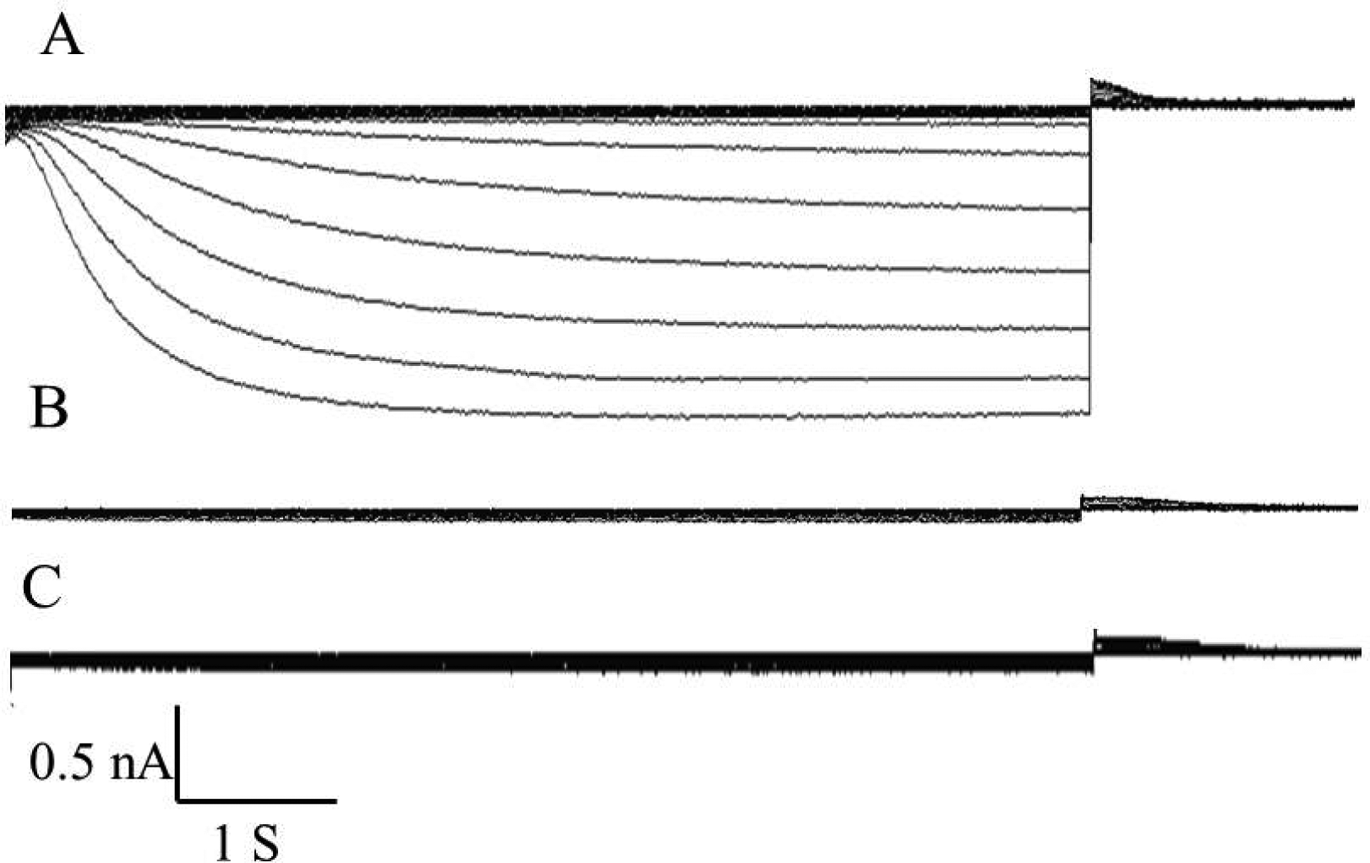
It was hypothesized that hHCN1-tranfected MSCs could influence the excitability of coupled NRVM. The whole-cell current-clamp configuration was used to measure the action potentials of NRVM after 3 days’ coculture with MSCs. NRVM cocultured with MSCs transfected with the control pcDNA3 plasmid beat spontaneously with a mean ± SE rate of 82 ± 8 beats/min (n = 5); the cycle length tended to vary from beat to beat (Figure 4A). The mean ± SE maximal diastolic potential (MDP) was−87 ± 4 mV. In NRVM cocultured with hHCN1-tranfected MSCs, a more regular rhythm and more rapid spontaneously beating frequency emerged (mean ± SE 129 ± 11 beats/min; n = 5; Figure 4B). Correspondingly, the mean ± SE MDP was reduced to −66 ± 6 mV. There were significant differences between the spontaneous beating rate and MDP between the NRVM cocultured with hHCN1-tranfected MSCs and those cocultured with the MSCs transfected with the control pcDNA3 plasmid (P < 0.05 for both comparisons).
Representative action potential traces for neonatal rabbit ventricular myocytes (NRVM), cocultured with mesenchymal stem cells (MSCs) transfected with the control pcDNA3 plasmid (A) or NRVM cocultured with hHCN1-transfected MSCs (B).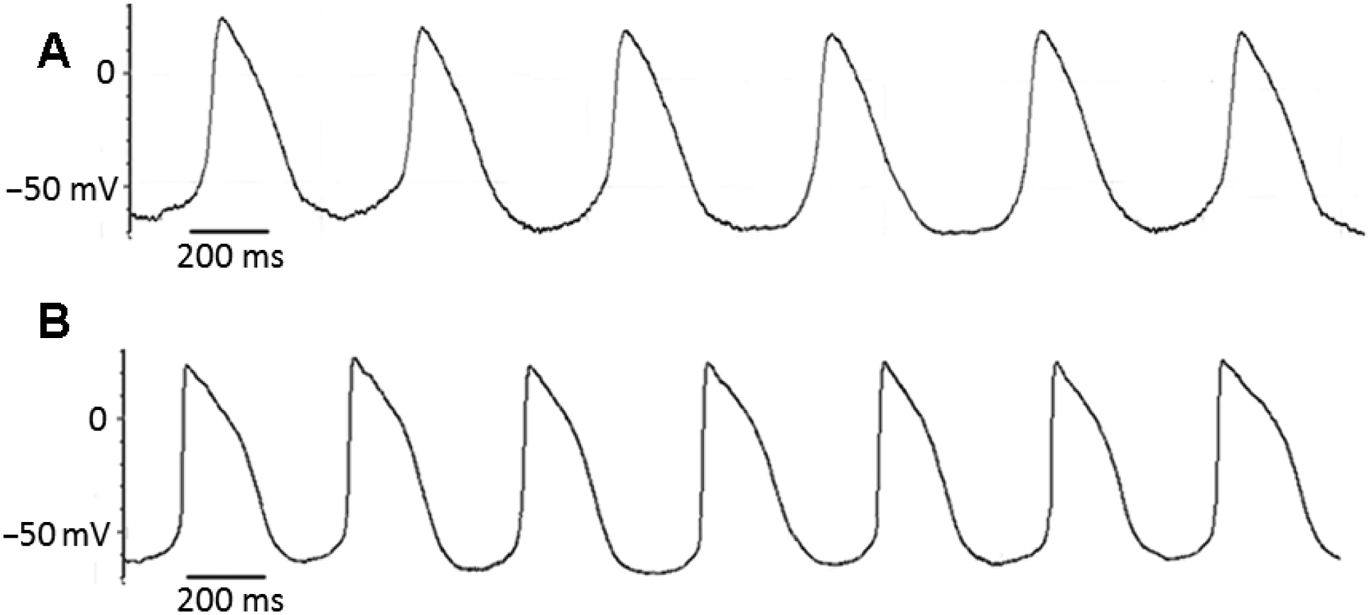
Discussion
Pacemaker cells differ from common cardiomyocytes due to the presence of a spontaneous depolarization process, which progressively reduces the membrane potential during the diastolic phase of the cardiac cycle. 1 When the reduction of the membrane potential reaches a critical threshold value, the sodium channels open and the action potential ensues. 3 The spontaneous diastolic depolarization of pacemaker cells is due to the expression of HCN genes, which code for specific HCN proteins, providing the presence of an inward current named If. 3 Therefore, the main difference between pacemaker cells and other cardiomyocytes depends only on which HCN genes are fully expressed.6–8
Expression of HCN protein isoforms varies somewhat among different cardiac cell types. 2 The HCN1, HCN2, and HCN4 proteins are the main isoforms in the heart. 2 HCN3 protein levels are low in embryonic pacemaker cells.2,7 The normal pacemaking region of the heart, the SAN, shows a significant amount of HCN2 protein and exhibits both the largest and most positively activating pacemaker current.9,10 The most predominant HCN isoform in the rat SAN is HCN2, which accounts for 81% of the total HCN mRNA.2,3 The remaining HCN mRNA transcripts are made up of HCN4, with only very low levels of HCN1 found. 3 In rats, HCN2 is the predominant ventricular isoform, especially in adult animals.11,12 Although the biophysical properties of heterologously expressed HCN isoforms cannot fully account for the observed variation in the native If, there is some correlation between which isoforms are expressed in a specific region and the voltage dependence of the native pacemaker current; regions with the most negative activation (e.g. the ventricle) tend to express HCN2 predominantly.1–3 In contrast, regions with more positive voltage ranges of activation (e.g. the SAN) mainly express HCN4.2,3 This present study investigated the ability of genetically-engineered MSCs transfected with the hHCN1 gene to modify the activity of cardiac pacemaker cells.
Mesenchymal stem cells are an attractive cellular vehicle for gene delivery applications. They can be obtained in relatively large numbers through a standard clinical procedure, are easily expanded in culture and are capable of long-term transgene expression.13,14 Their administration can be autologous or via banked stores, giving evidence that they may be immunoprivileged.15,16 The long-term function of such a pacemaker would be based on the prolonged expression of hHCN1, which in turn, requires integration into the genome of the MSCs. The hHCN1-transfected MSCs in the present study expressed an anticipated high level of hHCN1 mRNA, produced an If-like current and were capable of increasing the spontaneous beating rate of cocultured NRVMs. MSCs transfected with the control pcDNA3 plasmid, and not the hHCN1 gene, did not exert these effects. Thus, the electrical effects of the hHCN1-transfected MSCs were similar to the effects of overexpression of the same gene in myocytes studied in an in vitro system.2,3,10 These present findings suggest that MSCs may serve as an alternative approach for the delivery of pacemaker genes for cardiac implantation. These hHCN1-transfected MSCs appeared to be able to generate a depolarizing current that spread to coupled cardiac myocytes, driving the myocytes to the threshold voltage.17,18 In sinus node myocytes, the HCN gene generates an inward current necessary for cardiac excitation.2,10 It is hypothesized that, as long as the MSCs contain the pacemaker gene and couple to cardiac myocytes via gap junctions, they will function as cardiac pacemakers in an analogous manner to the normal primary pacemaker in the SAN.
This present study had a number of limitations. The work was only carried out in vitro which, because of the complexity of the body, cannot be fully representative of the body itself. Further research is necessary to determine whether the pacemaker function is enhanced in vivo following transplantation of hHCN1-transfected MSCs.
In conclusion, this present study demonstrated that MSCs transfected with the hHCN1 gene using lentiviral vector transfection were confirmed to have high levels of hHCN1 mRNA by RT–PCR analysis, produced an If-like current and were capable of increasing the spontaneous beating rate of cocultured NRVMs. The findings of this present study suggest that genetically-engineered MSCs transfected with the hHCN1 gene could be modified to become cardiac pacemaker cells in vitro.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by grants from the National Natural Science Foundation of China (no. 81170174), the Natural Scientific Fund of Jiangsu Province (no. BK2011304) and Jiangsu Province’s Key Provincial Talents Programme (no. RC2011111).
