Abstract
Objectives
As an infectious agent might play a role in rheumatoid arthritis (RA) development, this study investigated effects of Ureaplasma urealyticum lipid-associated membrane proteins (UuLAMPs) on RA synovial fibroblast (RASF) proliferation, and tumour necrosis factor (TNF)-α and interleukin (IL)-1β production by THP-1 macrophages. Possible immunogenic proteins in UuLAMPs were identified.
Methods
RASFs were cultured from synovial tissue from donors with RA. Serum samples from donors with/without RA and with/without U. urealyticum infection were used for immunogenicity analyses. THP-1 macrophages served as a model for synovial macrophages. TNF-α and IL-1β mRNA levels were assessed using reverse transcription–polymerase chain reaction; protein levels were estimated using enzyme-linked immunosorbent assay. UuLAMPs underwent separation and Western blot analyses.
Results
UuLAMPs (0.025–0.4 µg/ml) stimulated RASF proliferation in a dose- and time-dependent manner, and increased TNF-α and IL-1β levels in THP-1 macrophages. Several immunogenic UuLAMPs were identified, but antibodies to a 25 kDa protein were only found in RA patients with U. urealyticum infection.
Conclusions
UuLAMPs might induce RASF proliferation and proinflammatory cytokine secretion in synovium from RA patients. A 25 kDa U. urealyticum protein might act as a cross-reactive antigen.
Keywords
Introduction
Rheumatoid arthritis (RA) is a disease characterized by chronic inflammation that causes polyarthritis, which is associated with very high morbidity rates.1,2 RA is characterized by joint stiffness with varying degrees of deformity and dysfunction; its pathology is characterized by synovial fibroblast proliferation and proinflammatory cytokine production.3,4
Although much research has been carried out on the aetiology and pathogenesis of RA, the underlying cause of the disease remains unclear. 3 Ureaplasma urealyticum is a bacterium that belongs to the family Mycoplasmataceae; U. urealyticum, which does not have a cell wall, is the smallest self-propagating prokaryote. U. urealyticum lipid-associated membrane proteins (UuLAMPs) have been shown to influence the development of some autoimmune diseases.5,6 Consequently, the relationship between different U. urealyticum proteins and the pathogenesis of RA is of considerable interest.
The present study investigated the effect of UuLAMPs on the proliferation of RA synovial fibroblasts (RASFs) from human donors and the production of proinflammatory cytokines by the human acute monocytic leukaemia THP-1 cell line, in vitro. Preliminary investigations into whether any fractions of the UuLAMPs were immunogenic proteins were also conducted.
Patients and methods
Study population
For the RASF experiments, patients with RA who were undergoing total knee arthroplasty or arthroscopic synovectomy provided samples for analysis in this study. For Western blot analysis of serological immunogenicity, participants were divided into five groups, depending on the results of the analysis: patients with U. urealyticum infection alone; patients with reactive arthritis (ReA) and U. urealyticum infection; patients with RA and U. urealyticum infection; patients with RA without U. urealyticum infection; healthy control subjects.
All patients with RA had been admitted to the Provincial Hospital affiliated to Shandong University, Jinan, China between January 2008 and December 2010. These patients were recruited from the Department of Rheumatology at this institute, during the study period, and had been diagnosed with RA according to the clinical criteria of the American College of Rheumatology. 7 Patients infected with U. urealyticum were recruited from the Reproductive Medicine Centre at this institute: in these patients, cervical samples had been cultured by standard means (using a test kit from Zhuhai Livzon Diagnostics, Guangdon, China), and were shown to be positive for U. urealyticum infection.
Patients with any other autoimmune disease, any other Mycoplasma infection or Chlamydia trachomatis infection were excluded. Healthy donors were recruited from people attending the Examination Centre (Provincial Hospital affiliated to Shandong University) who did not have any autoimmune diseases, Mycoplasma infections or C. trachomatis infection.
Blood samples (5 ml) were collected routinely from the antecubital vein of each participant, then centrifuged at 1500
The study protocol was approved by the Human Ethics Review Committee of the Provincial Hospital affiliated to Shandong University. A signed consent form was obtained from each patient or subject, prior to study participation.
Preparation of Uu lamps
The U. urealyticum strain serovar4 (ATCC27816: American Type Culture Collection, Manassas, VA, USA) was cultured in self-prepared liquid medium containing 20% horse serum (Gibco BRL, Life Technologies, Gaithersburg, USA), 25% yeast extract (Gibco BRL, Life Technologies), 1.5 ml 40% urephil (Haibo, Qingdao, China), 0.5 ml 0.4% phenol red and 5.0 ml 10 000 IU/ml penicillin (per 100 ml). Preparation of LAMPs and the aqueous phase (used as a control) followed the methods described by You et al.
8
Briefly, U. urealyticum was cultured in 500 ml of medium to the stationary phase. After centrifugation (10 000
Rasf culture
Synovial tissue samples measuring approximately 5 × 5 × 5 mm were collected from RA patients during total knee replacement; these samples were finely minced into 2–3-mm pieces under aseptic conditions and incubated in Dulbecco’s modified Eagle’s medium (DMEM; Sigma-Aldrich, St Louis, MO, USA) containing 0.25% trypsin (AMRESCO, Solon, OH, USA) at 37℃, in a 5% CO2 incubator for 1.5 h. Trypsin digestion was stopped by addition of complete culture medium (DMEM containing 10% heat-inactivated fetal bovine serum [FBS], 100 IU/ml penicillin and 100 µg/ml streptomycin). Cells were centrifuged 1000
Rasf identification
Subcultured RASFs were observed and identified by inverted phase-contrast microscopy at a magnification of ×200. Protein levels of vimentin were examined by immunohistochemistry using the avidin–biotin–peroxidase complex method with an ABC kit (Beijing Zhongshan Jinqiao Biotechnology, Beijing, China). Briefly, the cells were cultured on glass slides for 72 h. After air drying, cells were fixed in 4% paraformaldehyde, dried, washed and preincubated with blocking solution from the ABC kit. Rabbit antihuman vimentin antibodies (1 : 250 dilution; Bioss, Beiing, China) were added to the slides at 4℃ overnight. The slides were then washed three times with 10 mM phosphate-buffered saline (PBS), pH 7.2, and incubated with biotinylated antirabbit antibody (1 : 5000 dilution; Beijing Zhongshan Jinqiao Biotechnology). Next, the slides were washed three times with 10 mM PBS (pH 7.2) and incubated with streptavidin–horseradish peroxidase using a LASB kit (Beijing Zhongshan Jinqiao Biotechnology), according to the manufacturer’s instructions. The signals were finally developed with diaminobenzidine (Boster Biological Technology, Wuhan, China). Positive cells were counted at ×400 magnification, in 10 randomly chosen fields, by two observers (M.F. and G.X.) in a blinded manner.
Rasf proliferation assay
The proliferation of RASFs was evaluated with the thiazolyl blue tetrazolium bromide assay (MTT; AMRESCO). Cells (5 × 103 cells/well) were seeded into 96-well plates in complete medium, and cultured at 37℃ and 5% CO2 for 24 h. After centrifugation at 1000
Culture and treatment of THP-1 cells
To investigate the effects of UuLAMPs on synovial cells, THP-1 macrophages were used as a cell model for synovial macrophages. The human acute monocytic leukaemia THP-1 cell line was cultured, as described previously. 9 Cells were seeded into six-well plates at 1 × 106 cells/ml and 50 ng/ml of phorbol 12-myristate 13-acetate was added to the cell cultures to induce differentiation. 10 After 72 h, nonadherent cells were aspirated and washed three times with 10 mM PBS, pH 7.2. Based on pilot experiments, THP-1 macrophages were stimulated with different concentrations of UuLAMPs (0, 1 µg/ml, 2 µg/ml and 4 µg/ml) for 24 h. Culture supernatant and cells were collected separately for subsequent experiments.
Measurment of TNF-α and IL-1β M RNA By RT–PCR
Primer sequences used for reverse transcription–polymerase chain reaction to amplify β-actin, tumour necrosis factor (TNF)-α and interleukin (IL)-1β) mRNA.

Total RNA was extracted from THP-1 macrophages using TRIzol® (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. The quality and yield of the RNA was verified by spectrophotometric assessments using the 260/280 nm ratio. The cDNA was synthesized using a reverse transcription reagent (Takara Biotechnology [Dalian], Dalian, China) and the PCR reactions were performed using a PCR kit (TransGen China Corporation, Beijing, China) according to the manufacturer’s instructions, using a thermal cycler (TG, Biometra GmbH, Göttingen, Germany). The thermal cycling conditions involved an initial denaturation step at 94℃ for 5 min, followed by 33 cycles of denaturation at 94℃ for 30 s, annealing at 55℃ for 30 s, and an extension at 72℃ for 1 min. A final step at 72℃ for 7 min was added for termination of DNA synthesis. PCR products were separated by 2% agarose gel electrophoresis, and analysed by ethidium bromide staining and image digitization. The gel was photographed and analysed using the JEDA 801 GEL imaging system (Jiangsu Jieda Technology Development, Jiangsu, Nanjing, China). The relative content of each sample was calculated by measuring the mean OD of each band and comparing it with the internal control gene, β-actin.
Measurement of TNF-α and IL-1β protein
Supernatants were collected from the cultures of THP-1 macrophages following incubation with 0, 1 µg/ml, 2 µg/ml and 4 µg/ml concentrations of UuLAMPs for 24 h. Supernatants were stored at −80℃. TNF-α and IL-1β protein levels were estimated using enzyme-linked immunosorbent assays (ELISA; ADL Systems, Sino–US Joint Venture Company, Wuhan, China) with monoclonal antibodies against TNF-α and IL-1β (both from ADL Systems). All samples were measured in duplicate.
Uu lamps separation and western blot analyses
A sample (10 µg) of UuLAMPs was mixed in Laemmli loading buffer (pH 6.8), boiled for 10 min, and separated by 10–15% sodium dodecyl sulphate–polyacrilamide gel electrophoresis (SDS–PAGE) using the Laemmli method. 11 The gels were run at 30 V for 60 min in the first stage of electrophoresis, then at 100 V during the second stage, in which the run time was determined by the molecular markers (Fermentas Canada, Burlington, ON, Canada) and the molecular weights of the proteins. The molecular weights of the proteins on the gels and blotting membranes were calculated according to the molecular markers and Quantity One software (Bio-Rad Laboratories, Hercules, CA, USA).
Two gels were prepared during each experiment: one for Coomassie Brilliant Blue staining 12 (to characterize the UuLAMPs); one for Western blot analysis (to identify immunoreactive proteins from the UuLAMP preparations that cross-reacted with immunoglobulins [Igs] from the serum samples from participants in each of the five study groups).
The UuLAMPs separated by SDS–PAGE were transferred to 0.22-nm nitrocellulose membranes (Pall, Port Washington, NY, USA). Membrane strips were washed under shaking conditions in TBS (pH 7.4) containing 0.1% Tween 20 (TBST) and blocked with 5% skimmed milk in TBST for 2 h. Serum samples from patients were diluted at 1 : 100 in 1% skimmed milk in TBST and incubated overnight at 4℃ with the membrane strips. After washing three times as described above, membrane strips were incubated with specific antibody conjugates: peroxidase-labelled rabbit antihuman-IgG and goat antihuman-IgM antibodies (Boster Biological Technology), diluted at 1 : 8000 and 1 : 4000, respectively. After 10 min, membrane strips were washed six times with distilled water, developed with an EZ–ECL Chemiluminescence Detection Kit for horseradish peroxidase (Invitrogen), and analysed using a chemiluminescent image reader LAS3000 mini (Fujifilm, Tokyo, Japan).
In every experiment, the conditions were kept the same (i.e. electrophoretic solution, voltage, and run time as determined by molecular weight marker electrophoresis) so that the results of each trial could be analysed under the same conditions.
Statistical analyses
Data were expressed as mean ± SD. Each experiment was repeated at least three times to assess reproducibility. Data were analysed using the SPSS® software package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. One-way analysis of variance was used to compare more than two groups. Differences between groups were assessed using the Student–Newman–Keuls test if variances were equal and Kruskal–Wallis H-test followed by Tamhane’s T2 multiple comparison test if variances were unequal. A P-value < 0.05 was considered to be statistically significant.
Results
Clinical characteristics of patients with reactive arthritis (ReA) or rheumatoid arthritis (RA), with or without Ureaplasma urealyticum infection, and healthy controls included in a study where serum samples were used for immunogenicity analyses.

Data presented as n or mean ± SD.
Adherent RASFs exhibited vimentin immunoreactivity, which presented as a yellow cell membrane and cytoplasm, but no yellow staining in the nucleus (Figure 1). The rate of vimentin-positive cells was >98%, and RASFs between passage 4 and 6 were used in subsequent experiments.
The identification of rheumatoid arthritis synovial fibroblasts (RASFs) by immunocytochemistry. Detection of vimentin protein in RASFs was performed using specific antivimentin antibodies. RASFs were shown to be vimentin-positive under a light microscope (which resulted in yellow staining). Original magnification × 400. The colour version of this figure is available at: http://imr.sagepub.com.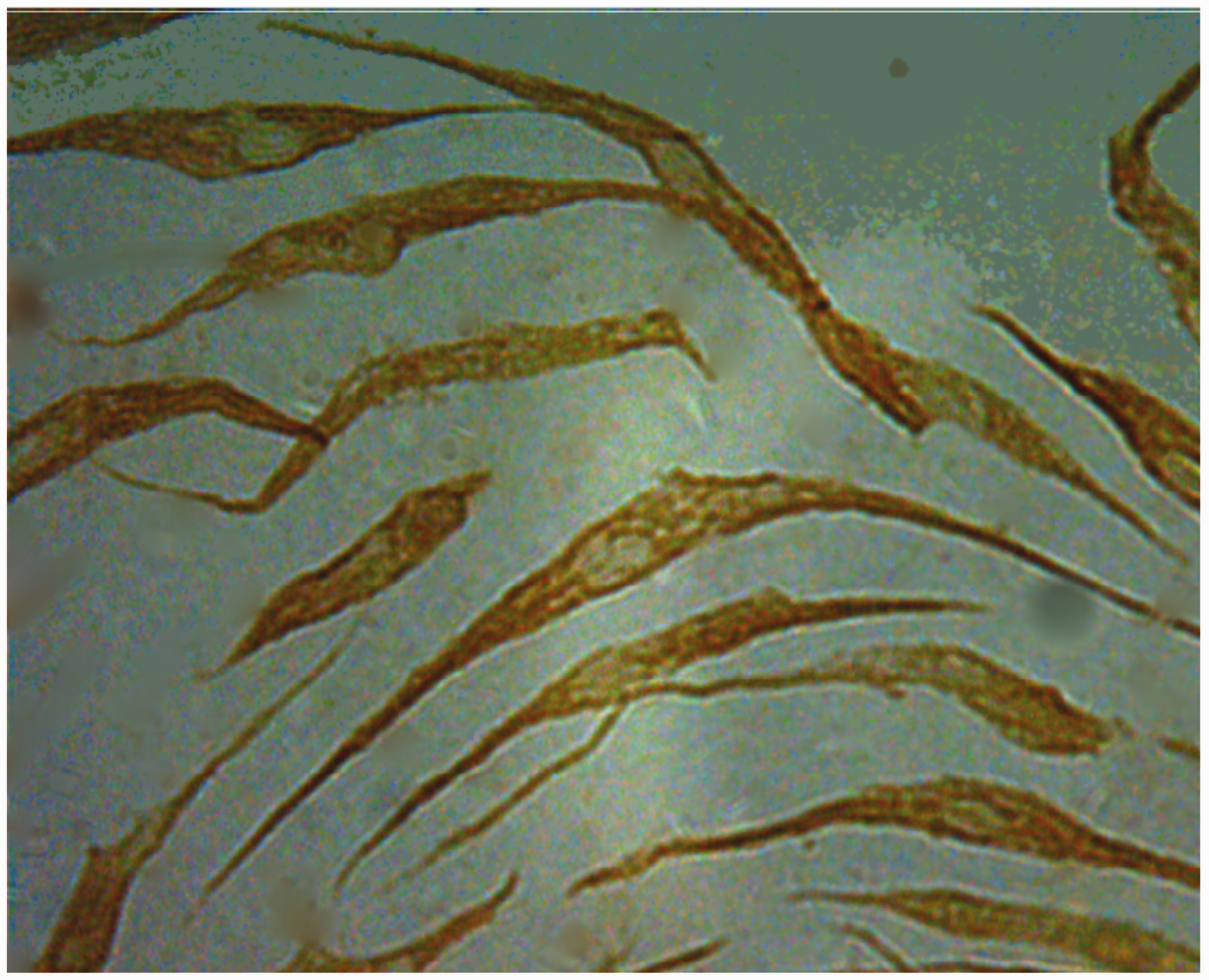
Compared with cells treated with aqueous phase (negative controls), RASF proliferation was significantly increased following incubation with different doses (0.025–0.4 µg/ml) of UuLAMPs for 24 h (P < 0.05) (Figure 2A). Although still significantly greater than the control groups, proliferation was decreased when the concentration of UuLAMPs reached 0.4 µg/ml. Treatment of RASFs with 0.05 µg/ml of UuLAMPs for 24–72 h resulted in a significant increase in proliferation compared with cells treated with aqueous phase (negative controls) (P < 0.05) (Figure 2B). Maximum RASF proliferation occurred after 72 h of treatment with 0.05 µg/ml of UuLAMPs. Thus, UuLAMPs, at concentrations of 0.025–0.4 µg/ml, stimulated the proliferation of RASFs in a dose- and time-dependent manner.
Effect of Ureaplasma urealyticum lipid-associated membrane proteins (UuLAMPs) on proliferation of rheumatoid arthritis synovial fibroblast (RASFs) in vitro. (a) Compared with the aqueous phase group (negative controls), cells of the experimental groups (0.025–0.4 µg/ml UuLAMPs) demonstrated significantly greater proliferation (aP < 0.05); significantly greater proliferation was observed at 0.2 µg/ml versus 0.4 µg/ml UuLAMPs (bP < 0.05) and (b) Compared with the respective aqueous phase (negative controls), proliferation of cells in the experimental groups was significantly higher following treatment with 0.05 µg/ml at each time point (P < 0.05) and increased significantly over time (aP < 0.05). Data presented are the mean ± SD of the optical density (OD) value (i.e. the OD value of the experimental well minus that of the blank control well) from triplicate samples. Experiments were repeated at least three times; a representative data set is presented. Student–Newman–Keuls test was used if variances were equal; Kruskal-Wallis H-test followed by Tamhane’s T2 multiple comparison test was used if variances were unequal.
The RT–PCR results (Figure 3) demonstrated that treatment of THP-1 macrophages with 1 µg/ml UuLAMPs resulted in a moderate but significant elevation in TNF-α and IL-1β mRNA, compared with results in control samples (P < 0.05). Treatment with 4 µg/ml UuLAMPs resulted in a moderate but significant reduction in TNF-α and a moderate but significant elevation IL-1β mRNA, compared with results in control samples (P < 0.05). Treatment with 2 µg/ml UuLAMPs resulted in the largest significant increase in TNF-α and IL-1β mRNA compared with the control samples (P < 0.05). Following treatment with 2 µg/ml UuLAMPs, TNF-α and IL-1β mRNA levels were significantly higher than following treatment with 1 µg/ml or 4 µg/ml UuLAMPs (P < 0.05 for both). TNF-α and IL-1β protein levels were significantly higher following treatment with each concentration of UuLAMPs compared with control samples (P < 0.05) (Figure 4). The maximum stimulatory effect for both proteins was observed with 2 µg/ml UuLAMPs.
Levels of tumour necrosis factor (TNF)-α and interleukin (IL)-1β mRNA in THP-1 macrophages, measured using reverse transcription–polymerase chain reaction (RT–PCR). Macrophages had been cultured for 24 h with increasing concentrations of Ureaplasma urealyticum lipid-associated membrane proteins (UuLAMPs) (0, 1, 2 and 4 µg/ml). (a) Representative results from RT–PCR analysis of TNF-α and IL-1β mRNA are shown in increasing order of dose of UuLAMPs (0, 1, 2 and 4 µg/ml, respectively): Control group, β-actin (200 base pairs [bp]) in lanes 1, 4, 7 and 10; TNF-α (171 bp) in lanes 2, 5, 8 and 11; IL-1β (192 bp) in lanes 3, 6, 9 and 12 and (b) Corresponding densitometric measurements are shown. Data presented as mean ± SD, from three independent experiments. *P < 0.05 compared with control group (0 µg/ml UuLAMPs); **P < 0.05 compared with 1 and 4 µg/ml UuLAMPs; there was no significant difference between 1 and 4 µg/ml UuLAMPs; Student–Newman–Keuls test was used if variances were equal; Kruskal-Wallis H-test followed by Tamhane’s T2 multiple comparison test was used if variances were unequal. Levels of tumour necrosis factor (TNF)-α and interleukin (IL)-1β protein in THP-1 macrophages, measured using an enzyme-linked immunosorbent assay. THP-1 macrophages were cultured for 24 h with increasing concentrations of Ureaplasma urealyticum lipid-associated membrane proteins (UuLAMPs) (0, 1, 2 and 4 µg/ml). Representative densitometric results are shown. Data presented as mean ± SD from three independent experiments. *P < 0.05 compared with controls; #P < 0.05 compared with 1 and 4 µg/ml UuLAMPs; there was no significant difference between 1 and 4 µg/ml UuLAMPs; Student–Newman–Keuls test was used if variances were equal; Kruskal-Wallis H-test followed by Tamhane’s T2 multiple comparison test was used if variances were unequal.

Four major proteins, p78, p67, p61 and p27, were found following SDS–PAGE analyses (Figure 5). In addition, proteins of 24, 25, 37, 43, 52, 54, 115, 140 and 175 kDa were observed. The p25 and p43 bands identified in this study were described in the literature as being specific for U. urealyticum.
13
Electrophoresis protein profile following 10–15% sodium dodecyl sulphate–polyacrilamide gel electrophoresis of Ureaplasma urealyticum lipid-associated membrane protein (UuLAMP) preparations stained with Coomassie Brilliant Blue. Left-hand lane shows the molecular weight markers; right-and land shows UuLAMPs. Arrows indicate the 175, 140, 115, 78, 67, 61, 54, 52, 43, 37, 27, 25 and 24 kDa UuLAMPs. The colour version of this figure is available at: http://imr.sagepub.com.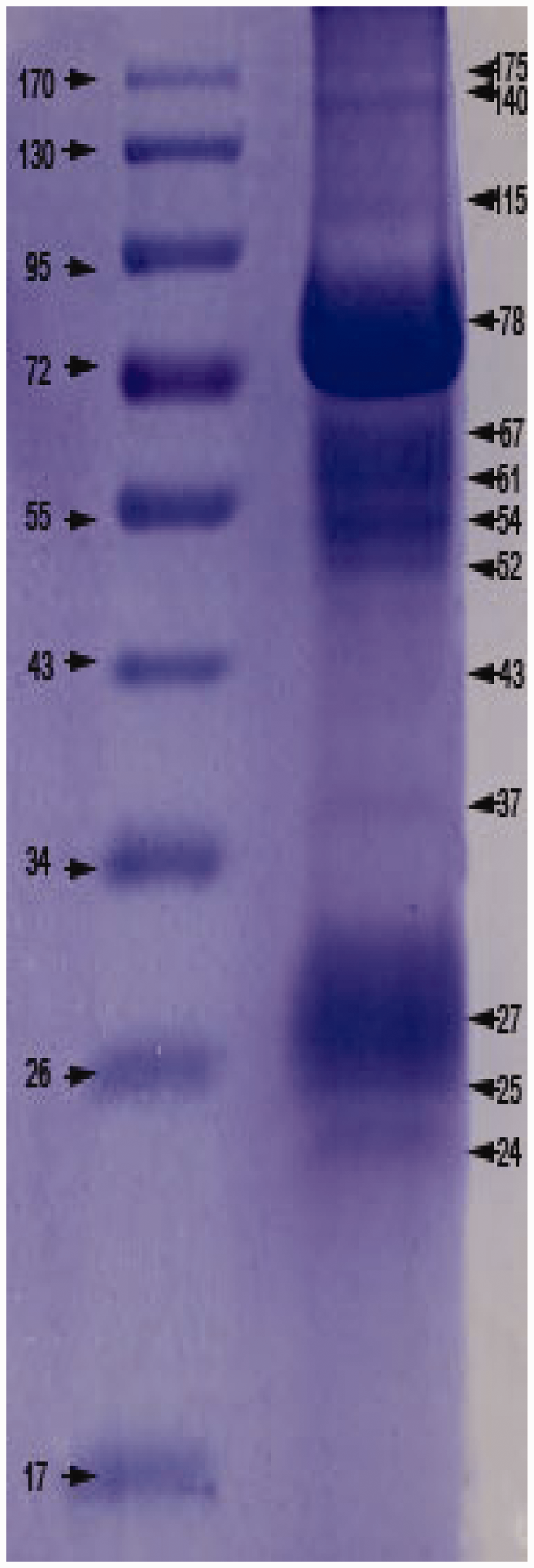
Immunoglobulin M antibodies against U. urealyticum were detected in the five study groups (i.e., patients with U. urealyticum infection, ReA patients with U. urealyticum infection, RA patients with or without U. urealyticum infection, healthy control subjects) at the following rates, respectively: 88.2% (15/17); 77.8% (14/18); 100% (16/16); 58.8% (10/17); 0% (none of 22). Western blot analysis showed IgM antibody reactivity for the following: 78, 67, 54 and 37 kDa proteins in patients with U. urealyticum infection; 78, 61, 52 and 37 kDa proteins in ReA patients with U. urealyticum infection; 61 and 25 kDa proteins in RA patients with U. urealyticum infection; 78 and 37 kDa proteins in RA patients without U. urealyticum infection; no proteins in the healthy controls (Figure 6A). It was noteworthy that IgM antibody for a 25 kDa protein was found only in RA patients with U. urealyticum infection, whereas the other proteins were found in two or more groups.
Western blot analysis of Ureaplasma urealyticum lipid-associated membrane protein (UuLAMP) preparations. Immunoreactivity profiles for UuLAMPs in serum samples from patients with U. urealyticum infection (lane A), patients with reactive arthritis and U. urealyticum infection (lane B), rheumatoid arthritis patients with U. urealyticum infection (lane C), rheumatoid arthritis patients without U. urealyticum infection (lane D) and healthy control subjects (lane E). Left-hand lane (lane M) shows the molecular weight markers. Immunoglobulin M (IgM) and IgG antibodies are shown in (a) and (b), respectively.
Quantitative analysis of Western blot experiments to investigate cross-reactivity of serum samples from study participants with or without Ureaplasma urealyticum infection, with U. urealyticum lipid-associated membrane proteins.

Ig, immunoglobulin.
Discussion
Infectious agents such as bacteria, viruses and Mycoplasma spp. might be implicated in the initiation and perpetuation of RA disease progression.14–16 Many types of bacteria parasitize humans and may be essential factors in certain pathological disorders; they may cause disease through several different pathways. Among the numerous potential infectious agents, Mycoplasma spp. seem to be likely candidates to become involved in the aetiology of RA. There is evidence that some Mycoplasma spp. can lead to acute or chronic arthritis in animal models.17,18 For example, M. hyosynoviae causes arthritis in pigs aged >10 years and M. arthritidis induces arthritis in rats (indeed, the latter has been widely used as an animal model of RA). 19 In human RA, however, the fact that inflammation is present in the urogenital system, which houses these bacteria, is often ignored. Enhanced mucosal permeability may play a crucial role in maintaining a chronic inflammatory response, due to a genetic predisposition or direct contact with bacteria or their products. 20 Mycoplasma spp. (especially M. fermentans) have been detected many times using different test methods.21–23
Pathological analysis shows that the hyperplasia of RASFs is noteworthy in synovium sections taken from patients with RA. RASFs play important roles in the pathogenesis and progression of RA.24,25 UuLAMPs stimulated RASF proliferation in a dose- and time-dependent manner within the concentration range studied, in the present analysis. Geiler et al. 26 developed an animal model of RA to investigate several aspects of the affected human joint under controlled conditions in vivo. Briefly, human RASFs and fresh human cartilage were co-implanted, together with an inert sponge, under the kidney capsule of severe combined immunodeficient (SCID) mice. 26 The SCID mice had severe defects both in their cellular and humoral immune responses, and were unable to reject these implants. When the implants were removed after about 60 days, the molecular mechanisms of the interaction of the synovial fibroblasts and human cartilage were studied, and cartilage destruction was assessed histologically. 26 Pannus was observed in the cartilage, and synovial fibroblasts had adhered to (and destroyed) the cartilage. 26 Other studies have confirmed these findings;27–30 some showed that the pathology was similar to the synovium of RA patients.29,30 From these studies,26–30 it could be concluded that in the model of human-SCID mice arthritis, human synovium implants and RASFs induced destructive arthritis in SCID mice. Sack et al. 31 injected cultured LS48 cells into the knees of SCID mice, causing rapidly progressive cartilage destruction in 10 days; in addition, the LS48 cells showed similar features to activated RASFs. RASFs can grow very fast over multiple passages in vitro. Cell proliferation is considered to be the major mechanism for the hyperplastic growth of the RA synovium, 32 and the role played by RASFs in the occurrence and development of RA has become a focus for current investigations.
The present study showed that, at the mRNA and protein level, UuLAMPs promoted the secretion of TNF-α and IL-1β in THP-1 macrophages: a finding that was similar to other research. 33 Experiments have mainly focused on the relationship between Mycoplasma and macrophages. For example, one study showed that the LAMPs of M. fermentans and M. salivarium could induce the apoptosis of monocytes and lymphocytes in a process that was mediated by Toll-like receptor-2. 34 Another report demonstrated that Mycoplasma infection inhibited cell apoptosis. 35 Mycoplasma can interfere with immune-cell function.34,36 The conflicting results observed could be due to different Mycoplasma spp. or their proteins having different effects on cells. We suggest that the effects of Mycoplasma infection depend on the unique interaction between Mycoplasma and the host cells.
Several studies have shown an association between Mycoplasma infection and RA. For example, Stuckey et al. 37 reported on a 10-year-old male patient with congenital agammaglobulinaemia and polyarthritis. A synovium puncture was positive for U. urealyticum, from which the authors hypothesized that U. urealyticum may induce arthritis. 37 In another study, M. pneumoniae was detected by nested PCR in 19/24 (79%) RA patients compared with eight of 10 (80%) osteoarthritis patients, six of six (100%) patients with inflammatory arthritis other than RA, none of nine (0%) patients with post-traumatic osteoarthritis, and none of four normal controls. 38 Furthermore, it was found that 30/55 (54.5%) RA patients tested were U. urealyticum positive: 39 significant differences were found in symptom morbidity between the RA group and the control group. 39 The prevalence of Mycoplasma infection in RA patients is not well known, though many studies have suggested that the rate of such infection in RA patients is higher than that in healthy subjects or patients with other types of arthritis.38–40 An investigation serum samples from 106 human immunodeficiency virus-infected patients using ELISA demonstrated seropositivity rates of 25.5%, 9.1% and 15.1% for anti-M. penetrans IgG, IgM and IgA, respectively; 40 in 110 patients with sexually transmitted disease seropositivity, this study also found rates of 17.3%, 9.1% and 17.3% for anti-M. penetrans IgG, IgM and IgA, respectively. 40 Serological testing is a very efficient method for assessing Mycoplasma infection and establishing the distribution of these micro-organisms in different clinical conditions. UuLAMPs were separated into several protein fractions in the present study. To screen out protein fractions that might participate in the RA disease process, Western blot analysis was used to detect anti-UuLAMPs IgM and IgG in serum samples from five study groups. Western blot analysis of UuLAMPs revealed IgG reactivity for p115, p78, p67, p43, p37, p32 and p25. These proteins were considered to be evidence of previous infection with U. urealyticum, except for proteins that only occurred in healthy controls (namely p115, p43 and p32). IgG antibody to p25 was found only in RA patients with U. urealyticum infection, however, which suggests that the immune system of these patients had been in contact with U. urealyticum proteins or other similar proteins. IgM reactivity was observed for p78, p67, p61, p54, p52, p37 and p25 in four patient groups but not in healthy controls. IgM antibodies against a 25 kDa protein were only detected in RA patients with U. urealyticum infection, which suggests that this protein might be a cross-reacting antigen, and this might be a candidate pathway for RA pathogenesis.
In conclusion, the present study demonstrated that UuLAMPs stimulated the proliferation of RASFs in a dose-related and time-dependent manner in vitro, which appeared to be similar to the pathological hyperplastic changes that happen to RASFs during RA. UuLAMPs increased TNF-α and IL-1β mRNA and protein levels in THP-1 macrophages within a specified concentration range. Our research also demonstrated that some of the UuLAMP fractions were immunogenic, and that a 25 kDa protein might participate in the pathogenesis of RA by acting as a cross-reactive antigen. Thus, UuLAMPs could be a major contributor to the RA disease process.
Footnotes
Acknowledgements
The authors thank Yaoran Zhao, Laicheng Wang and Jie Zhang of the Provincial Hospital affiliated to Shandong University for their technical support.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
