Abstract
Objectives
To investigate the ability of cytokeratin (CK)-19, galectin-3, human bone marrow endothelial cell (HBME)-1 and tyrosine kinase-type cell surface receptor HER2 (HER-2/neu) to differentiate papillary thyroid carcinoma (PTC) from benign thyroid nodules.
Methods
Archival specimens of PTC and benign thyroid nodules were included in this retrospective immunohistochemical study. CK-19, galectin-3, HBME-1, and HER-2/neu protein levels were analysed immunohistochemically using routinely prepared tissue sections.
Results
Specimens from 331 patients with PTC and 664 patients with benign thyroid nodules were reviewed. For all four protein markers, the percentage of samples with cells that demonstrated positive immunostaining was significantly higher in PTC specimens than in benign thyroid nodules. As a single protein marker, CK-19 was the most sensitive (92.7%) and HBME-1 was the most specific (97.9%). The combination of CK-19 and galectin-3 was the most sensitive (82.8%), and the combinations of HBME-1 with CK-19 or galectin-3 or HER-2/neu were the most specific (98.3%).
Conclusions
The combination of HBME-1 and either CK19 or galectin-3 or HER-2/neu can improve the specificity of the diagnosis of PTC.
Keywords
Introduction
Papillary thyroid carcinoma (PTC) is the most common malignant neoplasm of the neck and the incidence of thyroid cancer has increased dramatically. 1 Between 1996 and 2008, an increasing incidence of PTC was reported in Denmark, with age-standardized incidence rates increasing from 1.43 per 100 000 per year in 1996 to 2.16 per 100 000 per year in 2008. 2 The pathological diagnosis of PTC in both cytological and histological specimens is based upon the demonstration of typical nuclear morphology. 3 Using morphological criteria, most PTCs can be diagnosed with ease, except cases in which there is a paucity of diagnostic nuclear features.3,4 Immunohistochemical markers can help distinguish PTCs from benign thyroid lesions.5,6 Studies have shown that immunohistochemical staining for various cytokeratins can be applied as an ancillary technique for the diagnosis of PTC using histological or cytological specimens.4,7 However, the use of combinations of several markers in order to increase the accuracy of PTC diagnosis has been controversial.8–10 Nga et al. 9 reported that the sensitivity and specificity of the combination of human bone marrow endothelial cell (HBME)-1 and cytokeratin (CK)-19 in the cytological diagnosis of PTC were both 100%. However, another study showed that the sensitivity and specificity of the combination of HBME-1 and CK-19 were 62% and 95%, respectively. 10 Saleh et al. 11 reported that the combined detection of two or more immunohistochemical markers did not significantly improve the sensitivity and specificity for differentiating benign from malignant follicular-derived thyroid nodules.
This current retrospective study used immunohistochemical analysis to compare the protein levels of CK-19, galectin-3, HBME-1, and tyrosine kinase-type cell surface receptor HER2 (HER-2/neu), in PTC and benign thyroid nodule specimens in order to: (i) determine the significance of the CK-19, galectin-3, HBME-1, and HER-2/neu protein levels in the differential diagnosis of PTC; (ii) identify a useful panel of these four markers for more accurate diagnosis.
Patients and methods
Patient population
This retrospective study analysed surgical specimens excised from consecutive patients with surgically and histologically confirmed PTC and consecutive patients with benign thyroid nodules, who underwent these procedures between April 2009 and June 2010 in the Department of General Surgery, Huashan Hospital, Fudan University, Shanghai, China. Surgical specimens were retrieved from the archive of the Department of Pathology, Huashan Hospital.
The study complied with the Declaration of Helsinki and was approved by the Ethics Committee of Huashan Hospital, Fudan University (no. 2013M-002). As only routine procedures were used on archival surgical specimens and no treatment interventions were investigated, patients providing samples for the study were not required to provide written or verbal informed consent.
Immunohistochemical analysis
Tissue samples were fixed in 30% formalin, dehydrated in ethanol, and embedded in paraffin wax. For routine pathological examination, all specimens were sliced continuously into 4 -µm sections, stained with haematoxylin and eosin, and examined by two independent pathologists (Z.Z. and F.T.) with experience in thyroid pathology. These examinations were conducted blindly and independently; the pathologists did not know the original histological diagnosis. All specimens were classified according to published diagnostic criteria for thyroid tumours. 12
Immunohistochemical staining was performed using the streptavidin-peroxidase method and 4 -µm sections, prepared as described above.13,14 After the sections had been deparaffinized and rehydrated in descending alcohol dilutions, they were heated in boiling 0.01 mol/l citrate buffer (pH 6.0) for 20 min for antigen retrieval. After cooling to room temperature, the sections were treated with 3% hydrogen peroxide for 5 min. After blocking with goat serum (1 : 10 dilution in 0.1 M phosphate-buffered saline [PBS; pH 7.4]; Long Island Biotech, Shanghai, China) for 10 min at room temperature, the sections were incubated with primary antibodies to CK-19, galectin-3, HBME-1, or HER-2/neu (all were mouse antihuman antibodies, used at 1 : 100 dilution; Long Island Biotech) for 20 min at room temperature. The slides were then washed three times using 0.1 M PBS (pH 7.4) and incubated with a biotinylated horseradish peroxidase goat antimouse secondary antibody (1 : 500 dilution; Long Island Biotech) for 60 min at 37℃. Next, the slides were washed three times after incubation with the secondary antibody using 0.1 M PBS (pH 7.4). Immunolabelling was visualized with 0.05% diaminobenzidine (Long Island Biotech) in 0.01 M PBS (pH 7.4) for 5 min at room temperature, followed by rinsing for 2 min under running tap water. Haematoxylin was used for counterstaining; after drying, the sections were sealed and covered with glass coverslips.
The immunohistochemical staining was examined and photographed at ×100 and ×400 magnifications using an Olympus BX51T light microscope (Olympus Optical, Tokyo, Japan). The percentage of positive cells was evaluated semiquantitatively by counting the number of labelled cells in 10 high-power fields for each specimen. Samples of PTC that stained positively for CK-19, galectin-3, HBME-1, and HER-2/neu were provided by the Department of Pathology, Huashan Hospital and were used as the positive controls for immunohistochemical staining. For negative control slides, the primary antibody was replaced with 0.1 M PBS (pH 7.4). Galectin-3 displayed cytoplasmic and nuclear staining, HBME-1 staining was cytoplasmic with membranous and luminal accentuation, CK-19 staining was diffusely cytoplasmic, and HER-2/neu staining was cytoplasmic and/or membranous. An immunohistochemical score (IHS) was calculated for CK-19, galectin-3, HBME-1, and HER-2/neu for each specimen. This IHS took into account the percentage of cells (0–100%) as well as the staining intensity category (1–3). The scoring system used for CK-19, galectin-3, HBME-1, and HER-2/neu proteins was similar to published methods.15–17 The extent of positively stained cells was estimated and classified on a five-point scale as follows: grade 0, <10%; grade 1, ≥10% and ≤25%; grade 2, >25% and ≤50%; grade 3, >50% and ≤75%; grade 4, >75%. Positive staining intensity was categorized into three groups: weak (1); moderate (2); strong (3). A final IHS value was obtained by multiplying the score for the extent and the score for intensity as follows: 0, negative (−); 1–4, weakly positive (+); 5–8, moderately positive (++); 9–12, strongly positive (+++).
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 11.5 (SPSS Inc., Chicago, IL, USA) for Windows®. Fisher's exact test was used to determine the significance of any differences between the IHS scores for the two groups of tissue specimens. A P-value < 0.05 was considered statistically significant.
Results
Archival tissue samples were retrieved from 331 patients with PTC (82 men and 249 women; age range 23–75 years) and 664 patients with benign thyroid nodules (173 men and 491 women; age range 17–78 years). There was no statistically significant difference in the sex distribution between patients with PTC and those with benign thyroid nodules. The distribution of pathologically diagnosed benign thyroid disorders was as follows: 389 nodular goitres; 245 thyroid adenomas; 30 cases of Hashimoto's thyroiditis.
Immunohistochemical scores for the staining of four protein markers in surgical specimens of thyroid cancer from patients with papillary thyroid carcinoma (PTC; n = 331) compared with samples from patients with benign thyroid nodules (Benign; n = 664).

Data presented as n specimens with the corresponding immunohistochemical score: −, negative; + to +++, weakly positive to strongly positive.
PTC tissue versus benign nodules; χ2 = 628.4, Fisher’s exact test.
PTC tissue versus benign nodules; χ2 = 555.5, Fisher’s exact test.
PTC tissue versus benign nodules; χ2 = 654.2, Fisher’s exact test.
PTC tissue versus benign nodules; χ2 = 444.3, Fisher's exact test.
CK-19, cytokeratin-19; HBME-1, human bone marrow endothelial cell-1; HER-2/neu, tyrosine kinase-type cell surface receptor HER2.
Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of each of the four protein markers to diagnose papillary thyroid carcinoma (PTC), based on extent of immunostaining of each protein marker in surgical specimens of thyroid cancer from patients with PTC (n = 331) compared with samples of benign nodules (n = 664).

CK-19, cytokeratin-19; HBME-1, human bone marrow endothelial cell-1; HER-2/neu, tyrosine kinase-type cell surface receptor HER2.
Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of different combinations of each of the four protein markers to diagnose papillary thyroid carcinoma (PTC), based on the extent of immunostaining of each protein marker in surgical specimens of thyroid cancer from patients with PTC (n = 331) compared with samples of benign nodules (n = 664).
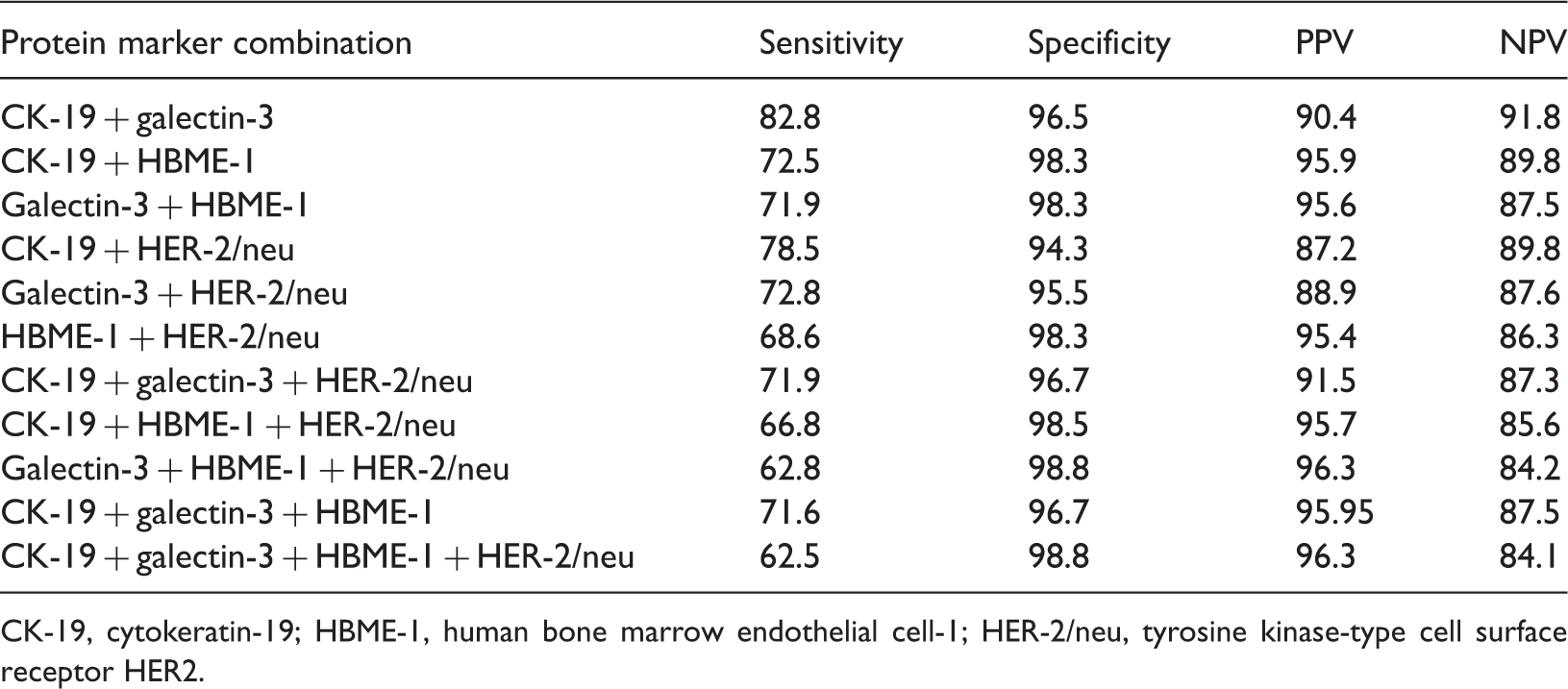
CK-19, cytokeratin-19; HBME-1, human bone marrow endothelial cell-1; HER-2/neu, tyrosine kinase-type cell surface receptor HER2.
Discussions
The incidence of malignant tumour of the thyroid (MTT) has increased substantially and it is now the most common malignant tumour of the neck.18,19 PTC is the most frequently observed MTT, accounting for ∼94.8% of all MTT cases. 20 The accurate diagnosis of thyroid carcinoma is critical for initiation of the correct clinical management. Currently, the gold standard for the diagnosis of thyroid lesions, particularly PTC, is histology. Using morphological criteria, most papillary cancers can be diagnosed with ease, except cases in which there is a paucity of diagnostic nuclear features. 3 Some ancillary approaches (such as immunohistochemistry and molecular techniques) have been explored. 8
Cytokeratin-19 is a low molecular weight cytokeratin that is expressed in simple stratified epithelium. 6 CK-19 has been demonstrated to discriminate PTC from multinodular goitres exhibiting papillary formations and papillary carcinoma-like changes in Graves’ disease.21,22 However, some studies have demonstrated that CK-19 expression is mostly focal and weak in benign hyperplastic nodules.21,22 The results of our current study suggest that, as a single marker, there was a significant difference in the extent of immunostaining for CK-19 between PTC specimens and benign thyroid nodules (P < 0.001). CK-19 immunostaining also had the highest sensitivity (92.7%) to detect PTC among the four protein markers, but there were 72 benign nodules that stained positive for CK-19 among the 664 benign thyroid nodules, yielding a specificity of 89.2%. This current result is in agreement with most other studies, which showed that CK-19 immunostaining was strong and diffuse in PTC specimens, and that there was either none or only weak immunostaining in benign lesions.7,23–25
Galectin-3 is a 31-kDa member of the β-galactoside-binding protein family. 17 This lectin is both intracellular (where it is located in the nucleus), and extracellular (where it is involved in cell-to-cell and cell-to-matrix interactions). 26 Galectin-3 appears to play a role in cell growth and differentiation, apoptosis, tumour progression and metastasis.26–28 Galectin-3 is expressed in a high proportion of PTC specimens and most likely plays a significant role in the malignant transformation of thyroid follicular cells.29,30 Our current study found that the sensitivity of galectin-3 to detect PTC was 81.9%, just below that of CK-19, whereas the specificity was 92.3%, just below that of HBME-1.
A component of the microvilli on the surface of mesothelial cells, HBME-1 has been used for the diagnosis of tumours originating from mesothelial cells.31,32 This marker has also been used for the diagnosis of thyroid carcinoma. 6 HBME-1 expression has been observed in thyroid malignancies, especially PTCs, whereas benign lesions have been found to be negative for HBME-1 expression.30,33,34 Casey et al. 35 observed CK-19 and galectin-3 positivity in benign thyroid lesions, whereas HBME-1 positivity was uncommon. Our present study showed that HBME-1 was the most specific of the four protein markers (97.9%) to detect PTC, as immunostaining was either negative or only weakly positive in a small number of benign nodules. In contrast, 79.2% of the PTC specimens demonstrated positive immunostaining for HBME-1. Our results indicate that HBME-1 could be used as a marker for differentiating benign and malignant thyroid lesions, especially for PTC, which was consistent with the findings of other studies.8,9,34
The HER-2/neu (also known as proto-oncogene c-ErbB-2) protein is encoded by a gene also known as ERBB2, NEU, NGL, and TKR1.36,37 The ERBB2 gene encodes for a 185-kDa protein that shows 50% homology with the epidermal growth factor receptor and is a member of the tyrosine kinase receptor family.36–38 Following the identification of HER2/neu, much of the original work on this protein was performed in the context of breast, ovarian and bladder cancer.39,40 Seçkin et al. 41 reported that, in their study of 23 PTC cases, 56.5% were stained strongly for HER-2/neu. In our current study, there was a significant difference in HER-2/neu immunostaining between PTC specimens and benign thyroid nodules (P < 0.001); and the sensitivity of HER-2/neu to detect PTC was 79.5%, whereas the specificity was 88.0%.
The combined use of several markers for the purpose of increasing the accuracy of PTC diagnosis has been controversial.8–10 Nga et al. 9 reported that the sensitivity, specificity and positive predictive value of the combined use of HBME-1 and CK-19 in the cytological diagnosis of PTC were all 100%. However, another study showed that the sensitivity and specificity of the combination of HBME-1 and CK-19 were 62% and 95%, respectively. 10 Saleh et al. 11 reported that the combined detection of two or more protein markers, such as galectin-3 + HBME-1, galectin-3 + CK-19, HBME-1 + CK-19, or galectin-3 + HBME-1 + CK-19, did not significantly improve the sensitivity and specificity. Our current study demonstrated that the combined detection of CK-19 + galectin-3, CK-19 + HBME-1 or galectin-3 + HBME-1 did not significantly increase the sensitivity to detect PTC. However, the combination of two protein markers significantly increased the specificity (P < 0.01), especially when the combination included HBME-1, relative to the specificity associated achieved with the use of single markers. The combination of HBME-1 and either CK-19 or galectin-3 or HER-2/neu was the most specific (98.3%), followed by the combination of CK-19 and galectin-3 (96.5%). We therefore recommend the use of HBME-1 in combination with CK-19 or galectin-3 or HER-2/neu to increase the reliability of PTC diagnosis.
Our current study demonstrated that the combined use of three or four protein markers did not further improve the diagnostic sensitivity or specificity to detect PTC. Therefore, we suggest that the immunohistochemical analysis of two protein markers is sufficient for the diagnosis of PTC. In addition, the use of combinations of two protein markers is recommended because it might help avoid technical or operational bias. 11
In conclusion, CK-19 was the most sensitive protein marker and HBME-1 was the most specific protein marker for the diagnosis of PTC when the protein markers were used individually. Galectin-3 was the best single marker for differentiating PTC from benign thyroid nodules. The combination of HBME-1 and either CK19 or galectin-3 or HER-2/neu improved the specificity of the diagnosis of PTC. The combined use of three or four markers did not further improve the diagnostic sensitivity or specificity. These findings suggest that the immunohistochemical analysis of two protein markers is sufficient for the diagnosis of PTC.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
