Abstract
Objective
The present study determined the role of DEP domain containing mTOR-interacting protein (DEPTOR) in the proliferation, apoptosis and chemosensitivity of RPMI-8226 multiple myeloma cells, using small hairpin RNA (shRNA) to knock down DEPTOR gene expression in vitro.
Methods
DEPTOR mRNA and protein levels in RPMI-8226 cells treated with DEPTOR-specific shRNA were evaluated by reverse transcription–polymerase chain reaction and Western blotting. Expression of apoptosis-associated proteins (including cleaved caspase-3 and cleaved poly-ADP ribose polymerase [PARP]) and activation of the phosphatidylinositol 3-kinase (PI3K)/v-akt murine thymoma viral oncogene homologue 1 (AKT) signalling pathway were detected by Western blotting.
Results
Transfection of DEPTOR-specific shRNA successfully knocked down DEPTOR gene expression in transfected RPMI-8226 cells. These transfected cells, together with control RPMI-8226 cells, were treated with 20 µmol/l melphalan for 24 h. Knockdown of DEPTOR exacerbated melphalan-induced growth inhibition and apoptosis, increased levels of cleaved caspase-3 and cleaved PARP, and reduced levels of phosphor-AKT.
Conclusion
Downregulation of DEPTOR inhibited proliferation and increased chemosensitivity to melphalan in human multiple myeloma RPMI-8226 cells via inhibiting the PI3K/AKT pathway.
Keywords
Introduction
Multiple myeloma is the second most common haematological malignancy; it is characterized by the synthesis of excess immunoglobulin, which forms endoplasmic reticulum-localized unfolded or misfolded proteins that are potentially toxic to myeloma cells. 1 Few patients with multiple myeloma experience long-term remission,1,2 so new therapeutic strategies need to be explored.
The DEP domain containing mTOR-interacting (DEPTOR) protein is a mammalian target of rapamycin (mTOR) binding protein that normally functions to inhibit the mTOR complex (mTORC) 1 and 2 pathways. 3 Phosphatidylinositol 3-kinase/v-akt murine thymoma viral oncogene homologue 1 (PI3K/AKT) signalling is frequently activated in many human tumours and hence represents a major cell-survival pathway. 4 Downstream effectors of the PI3K/AKT pathway include caspase-3. 5 Activation of the PI3K/AKT pathway has long been associated with proliferation and apoptosis. 6 The level of expression of the DEPTOR gene has been studied in many cancers and has been found to be low. 3 However, the DEPTOR gene is overexpressed in human multiple myeloma cells, which results in the promotion of in vitro cell viability via mediation of the PI3K/AKT pathway.7–9
This current study aimed to determine the role of DEPTOR in the proliferation and apoptosis of the multiple myeloma cell line RPMI-8226, which produces high levels of DEPTOR protein. The study also aimed to elucidate the mechanisms by which DEPTOR contributes to the chemosensitivity of RPMI-8226 cells in response to melphalan treatment. This study used small hairpin RNA (shRNA, which is an effective and efficient technique for downregulating specific gene expression) 10 to investigate the role of DEPTOR using RNA interference (RNAi) technology in vitro.
Materials and methods
Cell culture
The RPMI-8226 cells were obtained from Wuhan University (Wuhan, Hubei Province, China) and cultured in RPMI 1640 medium (Gibco, Carlsbad, CA, USA), supplemented with 10% fetal calf serum (Gibco) at 37°C in a humidified atmosphere containing 5% carbon dioxide.
Lentivirus-mediated DEPTOR gene knockdown
The vector hU6-MCS-CMV-EGFP(GV115) (Genechem, Shanghai, China) was used to generate shRNA specific for the DEPTOR gene. The region of the DEPTOR mRNA (GenBank® accession number NM_022783.2) was selected as the RNAi target site. The sequence was as follows: 5′-CATGACAATCGGAAATCTA-3′. shRNA-control encoding a nonspecific shRNA was synthesized and used as the negative control. The negative control sequences have been used previously.11,12 These oligonucleotides were annealed and subcloned into the AgeI and EcoRI sites of the hU6-MCS-CMV-EGFP(GV115) vector according to the manufacturer's instructions. Transfections of shRNA were carried out using Lipofectamine™ 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
pcDNA3.1/AKT plasmid and transfection
The pcDNA3.1/AKT plasmid was kindly provided by Dr Tianci Yao (Xiamen University, Xiamen, Fujian Province, China). The plasmid was transfected into RPMI-8226 cells using the Amaxa® Cell Line Optimization Nucleofector® Kit (Lonza Group, Basel, Switzerland) following the manufacturer’s instructions. In brief, 2 × 106 RPMI-8226 cells were resuspended in Nucleofector® V solution. Then, 100 µl of cell suspension was mixed with 2 µg of pcDNA3.1/AKT and transferred to a cuvette, to undergo nucleofection using a Nucleofector™ II Device (Lonza Group). Cells were transfected using the G-016 Nucleofector® Program (Lonza Group). Cells were transferred to culture plates and cultured at a density of 5 × 105 cells/ml for 2 h before incubation with melphalan (described below).
Cell proliferation assay
Cell proliferation was assessed using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) cell-viability assay (Sigma-Aldrich, St Louis, MO, USA). Briefly, RPMI-8226 cells were seeded into 96-well plates at a density of 2 × 103 cells/well in 200 µl of RPMI 1640 medium, then cultured in 1 µmol/l – l00 µmol/l melphalan (Sigma-Aldrich) for 24 h. Control cultures contained no melphalan. Subsequently, 20 µl of 5 mg/ml MTT solution was added to each well and incubated for 4 h. The supernatant was carefully removed and 200 µl dimethyl sulphoxide was added to each well. Optical density was measured at 490 nm with a multimodal microplate reader (SpectraMax M5, Molecular Devices, Sunnyvale, CA, USA). The concentration of melphalan required to inhibit the growth of RPMI-8226 cells by 50% (IC50) was calculated. All experiments were repeated in triplicate.
Flow cytometric analysis of apoptosis
The RPMI-8226 cells were incubated with or without 20 µmol/l melphalan (Sigma-Aldrich) for 24 h at 37°C in the dark, then pelleted by centrifugation at 1200
RT–PCR analysis
The silencing effects of DEPTOR-specific shRNA in RPMI-8226 cells were evaluated using reverse transcription–polymerase chain reaction (RT–PCR). From control shRNA-treated RPMI-8226 cells, parental RPMI-8226 cells and DEPTOR shRNA-treated RPMI-8226 cells, total RNA from 1 × 106 cells was extracted using TRIzol® reagent (Invitrogen) according to the manufacturer’s instructions and the method of Lu et al. 13 Total RNA (4 µg) was reverse transcribed to cDNA using the ThermoScript™ RT–PCR System Kit (Gibco) according to the manufacturer’s instructions. Primer sequences for RT–PCR for the human DEPTOR were 5′-CCTACCCAAACTGTTTTGTCGC-3′ (sense) and 5′-CGGTCTGCTAATTTCTGCATGAG-3′ (antisense). Primers for the control glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were 5′-TGACTTCAACAGCGACACCCA-3′ (sense) and 5′-CACCCTGTTGCTGTAGCCAAA-3′ (antisense). All primers were supplied by Genechem, Shanghai, China. All PCR cycles were performed in a Mastercycler® gradient thermal cycler (Eppendorf, Hamburg, Germany). The cycling programme involved preliminary denaturation at 95°C for 3 min, followed by 35 cycles of denaturation at 95°C for 15s, annealing at 60°C for 30s, and elongation at 72°C for 45s, followed by a final elongation step at 72°C for 10 min. The transcript levels were normalized according to the GAPDH transcripts. PCR products were separated on 1.5% agarose gels and visualized using ethidium bromide staining and ultraviolet light. Sizes of the PCR amplification products were as follows: GAPDH 121 base pairs (bp); DEPTOR 109 bp. Quantitative estimation was done by densitometric analysis of the PCR products using Scion Image Software (version Beta 4.03; Scion, Frederick, MD, USA).
Western blotting
Western blotting was undertaken to detect the levels of cleaved caspase-3 and cleaved PARP proteins, which signify the activation of apoptosis; it was also undertaken to clarify the role of PI3K/AKT signalling in DEPTOR-mediated chemosensitivity. RPMI-8226 cells were treated with or without 20 µmol/l melphalan for 24 h. Protein was extracted from 1 × 106 RPMI-8226 cells and the protein concentration was determined according to the method of Zhang et al. 14 The protein (80 µg) was separated by 6–15% sodium dodecyl sulphate–polyacrylamide gel electrophoresis at 100 V for 70 min. Proteins separated in the gel were transferred onto polyvinylidene fluoride membranes (Sigma-Aldrich) using electroblot apparatus at 70 V for 60 min (Bio-Rad, Hercules, CA, USA). The membranes were blocked with Tris-buffered saline–Tween 20 (TBST; pH 7.5; 20 mmol/l Tris–HCl, 150 mmol/l sodium chloride, 0.1% Tween-20) containing 5% nonfat milk at 4°C overnight. The membranes were incubated with the following antibodies for 2 h at 4°C: rabbit anticleaved caspase-3 (1 : 400 dilution in TBST; Asp175), rabbit anticleaved poly-adenosine diphosphate ribose polymerase (PARP) (1 : 400 dilution in TBST; Asp124), rabbit anti-AKT (1 : 400 dilution in TBST), rabbit antiphosphor-AKT (p-AKT) (1 : 400 dilution in TBST; Ser473), rabbit antiphosphor-70-kDa ribosomal S6 kinase 1 (p-P70S6K) (1 : 400 dilution in TBST; Thr421/Ser424), rabbit antiphosphor-eIF4E-binding protein 1 (p-4Ebp-1) (1 : 400 dilution in TBST; Thr70) (all from Cell Signaling Technology®, Danvers, MA, USA); rabbit anti-DEPTOR (1 : 400 dilution in TBST; Millipore, Billerica, MA, USA); rabbit anti-GAPDH (1 : 400 dilution in TBST; Santa Cruz Biotechnology, Santa Cruz, CA, USA). After washing three times with TBST (pH 7.5), membranes were incubated with horseradish peroxidase-conjugated goat antirabbit secondary antibody (1 : 1000 dilution in TBST; Beijing Biosynthesis Biotechnology, Beijing, China) for 1 h at room temperature. Membranes were washed three times with TBST (pH 7.5). Blots were detected using an enhanced chemiluminescence reagent kit (Beyotime Biotech, Haimen, China). Protein bands were quantified by scanning densitometry using Scion Image Software (version Beta 4.03; Scion).
Statistical analyses
Data were expressed as the mean ± SD. All statistical analyses were performed using the SPSS® statistical software package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Student’s t-test was used to determine the significance of differences in multiple comparisons. A P-value < 0.05 was considered statistically significant.
Results
Results of the silencing-effects analyses (namely, RT–PCR and Western blot) revealed that the mean ± SD ratio of DEPTOR/GAPDH mRNA in the DEPTOR shRNA-treated RPMI-8226 cells was 39.8 ± 1.6%. This was significantly lower than that observed in the control shRNA-treated RPMI-8226 cells or the parental RPMI-8226 cells (P < 0.05; Figure 1A). Western blot analysis revealed that the mean ± SD ratio of DEPTOR/GAPDH protein in the DEPTOR shRNA-treated RPMI-8226 cells was 34.3 ± 2.1%, which was significantly lower than that in the control shRNA-treated RPMI-8226 cells or the parental RPMI-8226 cells (P < 0.05; Figure 1B). There was no significant difference between the control shRNA-treated RPMI-8226 cells and the parental RPMI-8226 cells.
Effect of DEP domain containing mTOR-interacting protein (DEPTOR) small hairpin RNA (shRNA) on levels of DEPTOR mRNA and DEPTOR protein in RPMI-8226 cells. (A) Relative mRNA levels were analysed by reverse transcription–polymerase chain reaction using glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the loading control. (B) DEPTOR protein levels were measured using Western blot; GAPDH was used as the loading control. *P < 0.05 versus control shRNA-treated or parental RPMI-8226 cells; Student’s t-test. Lane/column numbers: 1, parental RPMI-8226 cells; 2, control shRNA-treated RPMI-8226 cells; 3, DEPTOR shRNA-treated RPMI-8226 cells.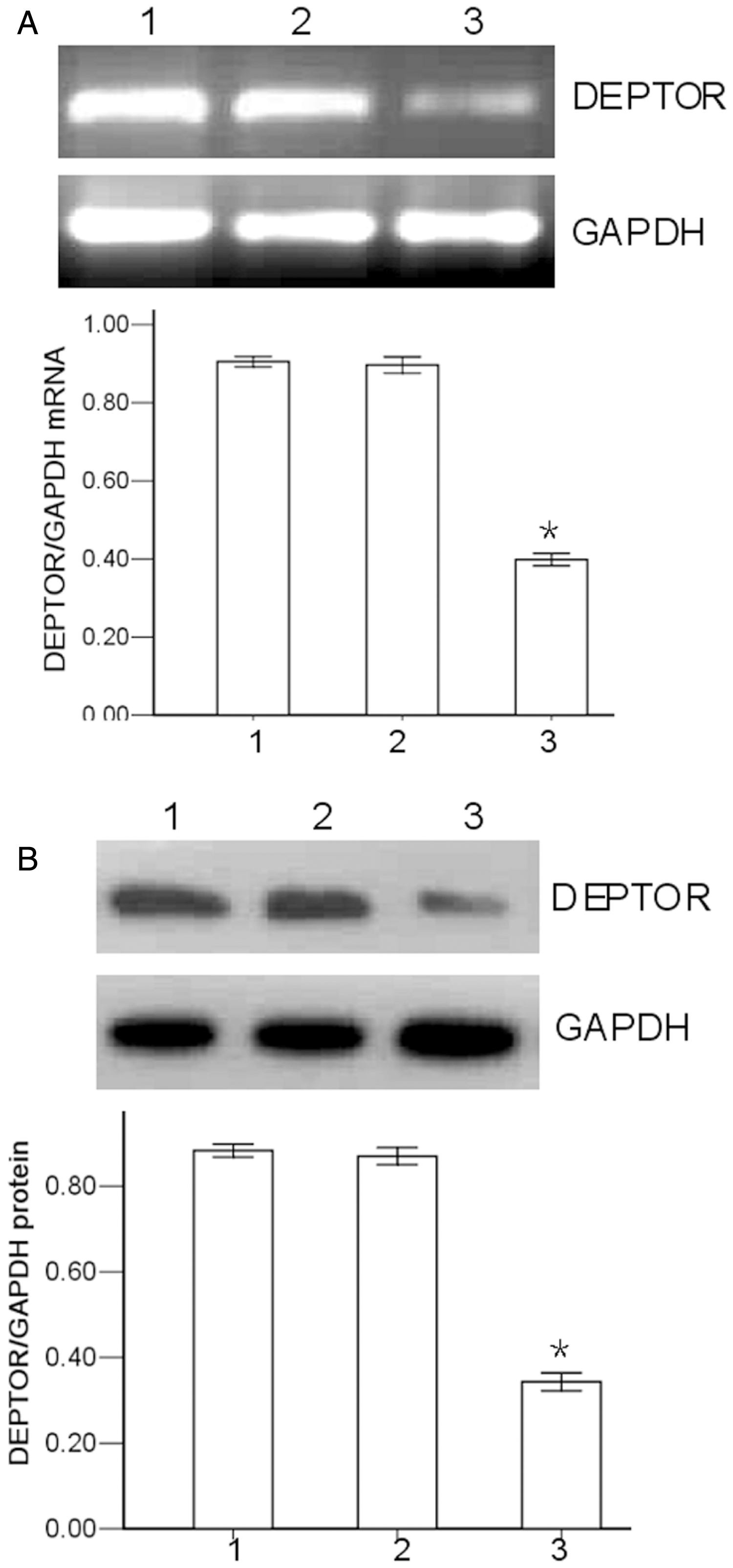
When the RPMI-8226 cells were exposed to 1 µmol/l – l00 µmol/l melphalan for 24 h, the IC50 (calculated based on data from the MTT assay) showed that DEPTOR knockdown enhanced the sensitivity of RPMI-8226 cells to melphalan (Figure 2). DEPTOR knockdown decreased the melphalan IC50: the mean ± SD IC50 of melphalan to RPMI-8226 cells decreased from 23.3 ± 1.3 µmol/l (parental RPMI-8226 cells) to 8.5 ± 0.6 µmol/l (DEPTOR shRNA-treated RPMI-8226 cells).
Effect of DEP domain containing mTOR-interacting protein (DEPTOR) knockdown on melphalan-induced cytotoxicity in RPMI-8226 cells. The melphalan IC50 was determined using the 3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide assay after exposure to 1 µmol/l – l00 µmol/l melphalan for 24 h. DEPTOR knockdown decreased the melphalan IC50. *P < 0.05 versus control shRNA-treated RPMI-8226 cells; Student’s t-test. IC50, concentration required to inhibit proliferation by 50%. Column numbers: 1, parental RPMI-8226 cells; 2, control shRNA-treated RPMI-8226 cells; 3, DEPTOR shRNA-treated RPMI-8226 cells; 4, DEPTOR shRNA-treated RPMI-8226 cells + pcDNA3.1/AKT. AKT, v-akt murine thymoma viral oncogene homologue 1.
Further investigations showed that the inhibition rate in the DEPTOR shRNA-treated RPMI-8226 cells treated with 20 µmol/l melphalan (for 24 h) was significantly higher than that in the control shRNA-treated RPMI-8226 cells treated with 20 µmol/l melphalan (P < 0.05; Figure 3). There was no significant difference between the control shRNA-treated RPMI-8226 cells and the parental RPMI-8226 cells, in terms of percentage inhibition following treatment with 20 µmol/l melphalan.
Effect of DEP domain containing mTOR-interacting protein (DEPTOR) knockdown on melphalan-induced inhibition of proliferation in RPMI-8226 cells. RPMI-8226 cells were treated with or without 20 µmol/l melphalan for 24 h. Cell growth inhibition was determined using the 3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) assay. *P < 0.05 versus control shRNA-treated RPMI-8226 cells; #P < 0.05 versus control shRNA-treated RPMI-8226 cells + melphalan; Student’s t-test. Column numbers: 1, parental RPMI-8226 cells; 2, control shRNA-treated RPMI-8226 cells; 3, DEPTOR shRNA-treated RPMI-8226 cells; 4, parental RPMI-8226 cells + melphalan; 5, control shRNA-treated RPMI-8226 cells + melphalan; 6, DEPTOR shRNA-treated RPMI-8226 cells + melphalan; 7 – DEPTOR shRNA-treated RPMI-8226 cells + melphalan + pcDNA3.1/AKT. AKT, v-akt murine thymoma viral oncogene homologue 1.
Flow cytometry showed that, following exposure to 20 µmol/l melphalan for 24 h, apoptosis was significantly increased in DEPTOR shRNA-treated RPMI-8226 cells compared with control shRNA-treated RPMI-8226 cells (P < 0.05) (Figure 4).
Effect of DEP domain containing mTOR-interacting protein (DEPTOR) knockdown on melphalan-induced apoptosis in RPMI-8226 cells. The apoptotic fraction of RPMI-8226 cells (treated with or without 20 µmol/l melphalan for 24 h) was quantified by flow cytometry. *P < 0.05 versus control shRNA-treated RPMI-8226 cells; #P < 0.05 versus control shRNA-treated RPMI-8226 cells + melphalan; Student’s t-test. Column numbers: 1, parental RPMI-8226 cells; 2, control shRNA-treated RPMI-8226 cells; 3, DEPTOR shRNA-treated RPMI-8226 cells; 4, parental RPMI-8226 cells + melphalan; 5, control shRNA-treated RPMI-8226 cells + melphalan; 6, DEPTOR shRNA-treated RPMI-8226 cells + melphalan; 7, DEPTOR shRNA-treated RPMI-8226 cells + melphalan + pcDNA3.1/AKT. AKT, v-akt murine thymoma viral oncogene homologue 1.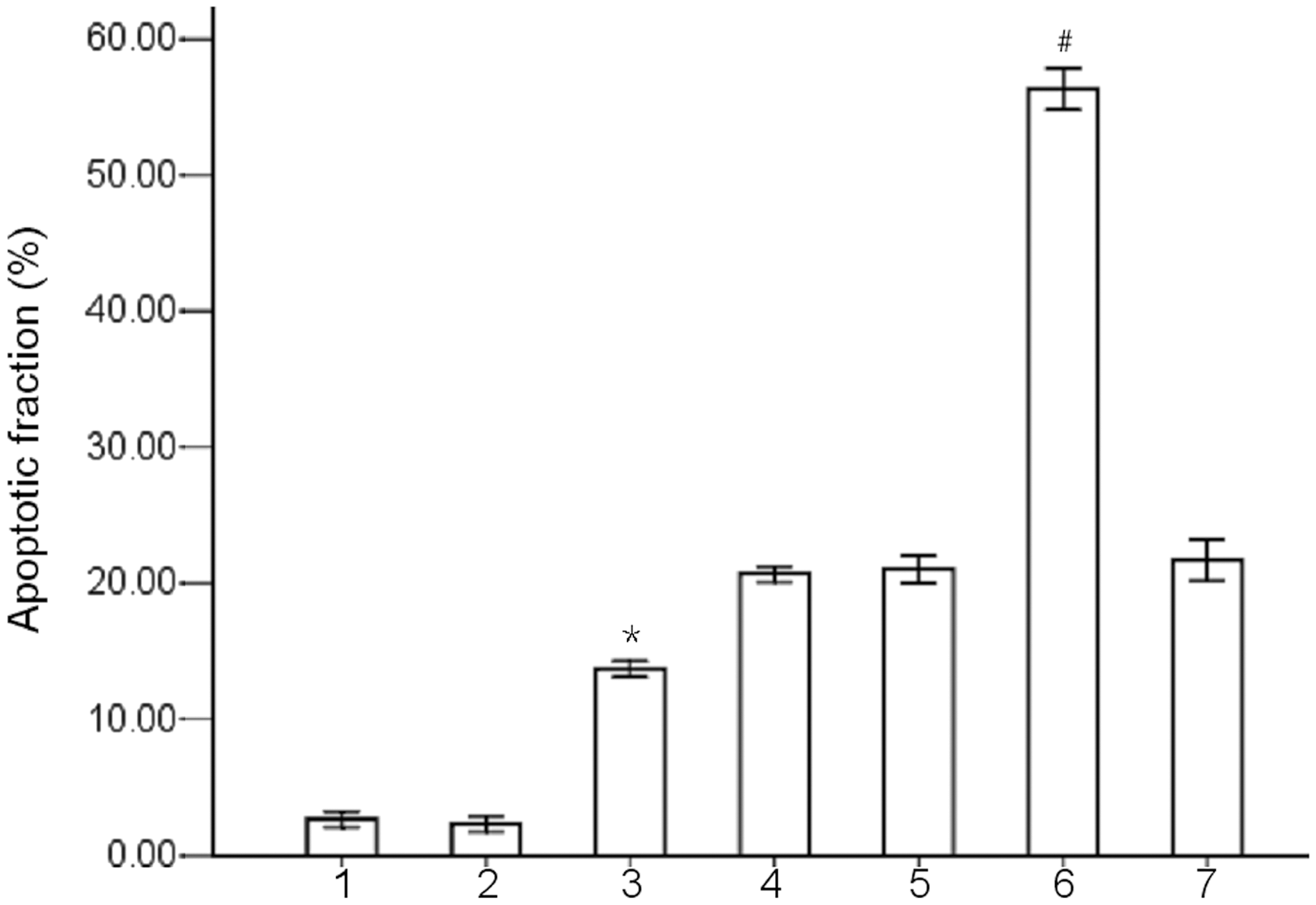
After exposure to 20 µmol/l melphalan for 24 h, protein levels of cleaved caspase-3 and cleaved PARP were significantly increased in DEPTOR shRNA-treated RPMI-8226 cells, compared with control shRNA-treated RPMI-8226 cells (P < 0.05 for both comparisons) (Figure 5).
Effect of DEP domain containing mTOR-interacting protein (DEPTOR) knockdown on levels of apoptotic-associated proteins in RPMI-8226 cells. RPMI-8226 cells were exposed to 20 µmol/l melphalan for 24 h, then subjected to Western blot analysis. (A) Representative blots showing protein levels of cleaved caspase-3 and cleaved poly-ADP ribose polymerase (PARP) in three cell groups; glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the loading control. (B) Densitometric analysis of the results shown in A. Results presented in relation to GAPDH levels. *P < 0.05 versus control shRNA-treated RPMI-8226 cells; Student’s t-test. Lane/column numbers: 1, parental RPMI-8226 cells; 2, control shRNA-treated RPMI-8226 cells; 3, DEPTOR shRNA-treated RPMI-8226 cells.
The level of p-AKT protein was significantly reduced in DEPTOR shRNA-treated RPMI-8226 cells compared with control shRNA-treated RPMI-8226 cells (P < 0.05), whereas the level of total AKT remained very similar among the three groups of RPMI-8226 cells (Figure 6). In addition, the levels of p-P70S6K and p-4Ebp-1 proteins were significantly increased in DEPTOR shRNA-treated RPMI-8226 cells compared with control shRNA-treated RPMI-8226 cells (P < 0.05).
Effect of DEP domain containing mTOR-interacting protein (DEPTOR) knockdown on the activation of the phosphatidylinositol 3-kinase/v-akt murine thymoma viral oncogene homologue1 (PI3K/AKT) signalling pathway in RPMI-8226 cells. RPMI-8226 cells were exposed to 20 µmol/l melphalan for 24 h and then subjected to Western blot analysis. (A) Representative blots showing the protein levels of total AKT (T-AKT), phosphor-AKT (p-AKT), phosphor-70-kDa ribosomal S6 kinase 1 (p-P70S6K), and phosphor-eIF4E-binding protein 1 (p-4Ebp-1) in three cell groups. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the loading control. (B) Densitometric analysis of the results shown in A. Results presented in relation to GAPDH levels. *P < 0.05 versus control shRNA-treated RPMI-8226 cells; Student’s t-test. Lane/column numbers: 1 – parental RPMI-8226 cells; 2 – control shRNA-treated RPMI-8226 cells; 3 – DEPTOR shRNA-treated RPMI-8226 cells.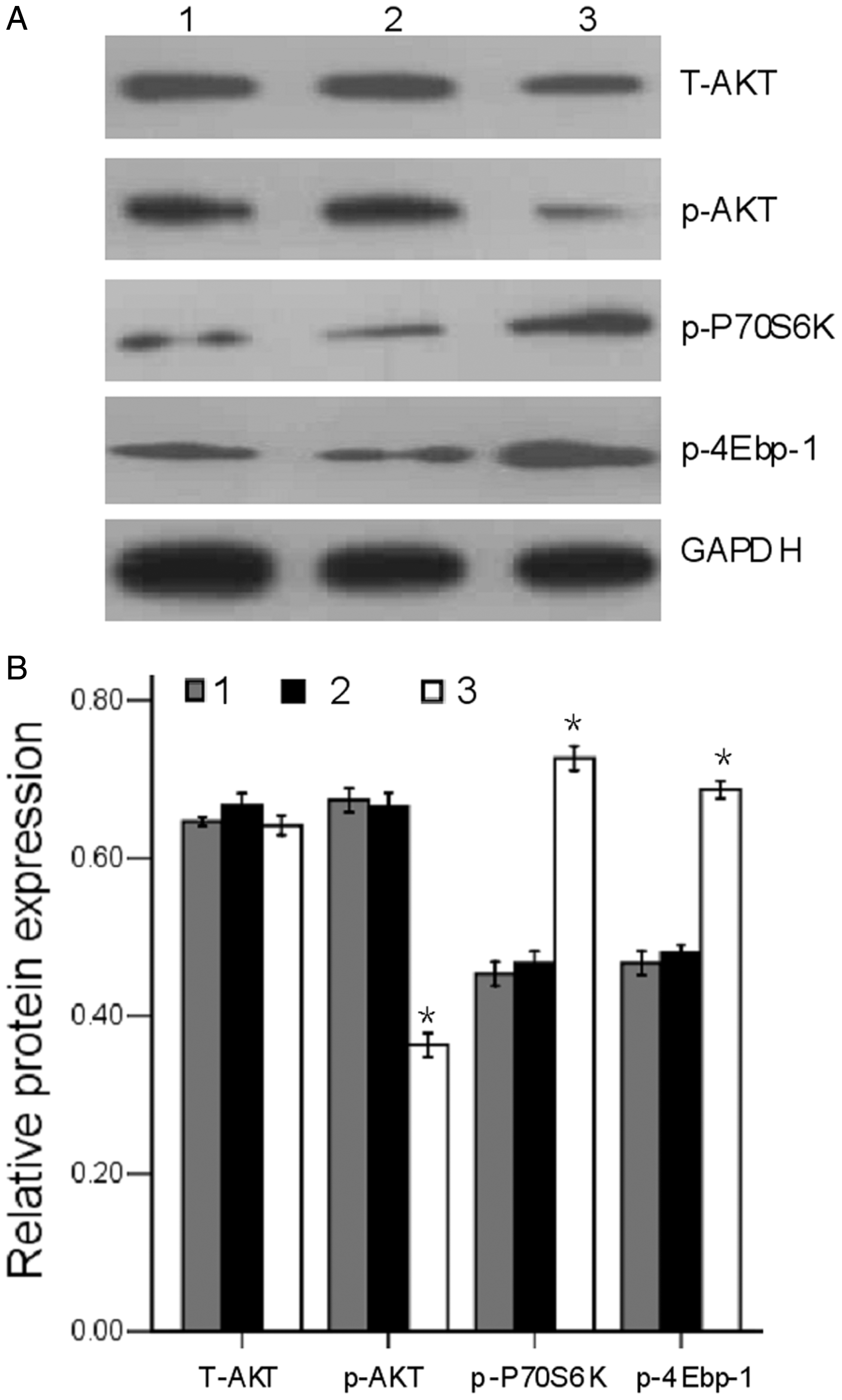
To further demonstrate that DEPTOR contributes to the chemoresistance of RPMI-8226 cells via the activation of PI3K/AKT signalling, when AKT protein was overexpressed in DEPTOR shRNA-treated RPMI-8226 cells, an increase in the melphalan IC50 was observed (Figure 2). In addition, the overexpression of AKT protein suppressed melphalan-induced cell growth inhibition and apoptosis in DEPTOR shRNA-treated RPMI-8226 cells (Figures 3 and 4, respectively).
Discussion
In this current study, shRNAs specifically targeting the DEPTOR gene were successfully transfected into the human multiple myeloma cell line, RPMI-8226, and DEPTOR gene expression was effectively inhibited at both the mRNA and protein levels. Results of this current study showed that DEPTOR knockdown promoted melphalan-induced growth inhibition and apoptosis, and increased the chemosensitivity of RPMI-8226 cells to melphalan. In addition, the p-AKT protein level in RPMI-8226 cells treated with melphalan was downregulated on DEPTOR knockdown. These data suggest that targeting DEPTOR gene expression might be able to be exploited, in the treatment of multiple myeloma.
Research has demonstrated that DEPTOR results in the inhibition of proliferation and induction of apoptosis. 7 The level of DEPTOR gene expression appears to be important for the survival of myeloma cells. 7 In addition, high levels of DEPTOR protein are predictive of a response to thalidomide in myeloma, 15 which indicates that the level of DEPTOR gene expression is important for the survival of myeloma patients.
Our current study investigated the effects of DEPTOR inhibition on the anticancer potency of the DNA-damaging drug melphalan, which is the front-line chemotherapeutic drug for patients with multiple myeloma. The recommended dose of melphalan is 0.18 mg/kg, administered for 4 consecutive days every 4–6 weeks in myeloma patients. 16 In the present study, melphalan induced inhibition of cell proliferation with an IC50 of 23.3 µmol/l in RPMI-8226 cells at 24 h. In contrast, the IC50 of melphalan was 8.5 µmol/l in DEPTOR shRNA-treated RPMI-8226 cells. These results clearly demonstrated that DEPTOR depletion could significantly enhance the sensitivity of RPMI-8226 cells to melphalan. Collectively, these data suggest that DEPTOR knockdown promoted melphalan-induced growth inhibition and apoptosis, and increased the chemosensitivity to melphalan in multiple myeloma cells in vitro. Therefore, these data provide a rationale for the clinical evaluation of DEPTOR inhibition, in combination with DNA-damaging chemotherapy, in patients with multiple myeloma.
Active caspases play a vital role in the induction of apoptosis. 17 In the present study, DEPTOR knockdown led to increased levels of cleaved caspase-3 and cleaved PARP. These results indicate that the DEPTOR knockdown by shRNA is sufficient to trigger caspase-dependent apoptosis, which could be the reason for the decreased cell viability.
This current study dissected the signalling pathway by which DEPTOR modulates the chemotherapeutic response of multiple myeloma cells to melphalan. The PI3K/AKT signalling pathway plays an important role in cellular proliferation, motility, apoptosis, drug resistance and survival.6,15,18,19 Constitutive activation of the PI3K/AKT pathway is considered to be an important molecular event that contributes to the malignant phenotype of the tumour cell in multiple myeloma. 20 The major upstream regulator of AKT is PI3K, which is activated by a variety of transmembrane receptors. 21 Under certain circumstances, DEPTOR acts as an oncogene by relieving the feedback inhibition from p-P70S6K to PI3K, thus activating AKT.22,23 AKT is a critical downstream factor of the PI3K signalling pathway and is important in promoting cell survival and inhibiting apoptosis. 24 AKT is a major mediator of cell survival by acting directly to inhibit proapoptotic proteins (such as caspase-9 and BCL2-associated agonist of cell death [known as Bad]). 25 Activated AKT modulates the function of many substrates involved in cell-cycle progression, cellular growth, and regulation of cell survival.26,27
Our current findings demonstrated that DEPTOR shRNA reduced the levels of p-AKT protein in RPMI-8226 cells, suggesting the involvement of PI3K/AKT signalling downstream of DEPTOR. Our results showed that the level of p-AKT protein was markedly reduced in DEPTOR shRNA-treated RPMI-8226 cells compared with control shRNA-treated RPMI-8226 cells. In addition, AKT controls cell growth through its effects on the mTOR/P70S6K pathway.6,20,28 mTORC1 regulates cell growth in part by phosphorylating P70S6K and 4Ebp-1. 29 In the present study, levels of p-P70S6K and p-4Ebp-1 protein were markedly increased in DEPTOR shRNA-treated RPMI-8226 cells compared with control shRNA-treated RPMI-8226 cells. Moreover, DEPTOR knockdown RPMI-8226 cells recovered chemoresistance on the overexpression of p-AKT protein. Taken together, these data suggest that the suppression of PI3K/AKT activity following DEPTOR knockdown is responsible for the increased sensitivity of RPMI-8226 cells to melphalan.
In conclusion, our current study findings demonstrate that DEPTOR knockdown inhibits the PI3K/AKT pathway and leads to changes in the levels of apoptosis-related proteins. This may explain the observation that knockdown of DEPTOR enhanced cytotoxicity and caspase-mediated apoptosis in human multiple myeloma RPMI-8226 cells. after melphalan treatment. Our current results provide evidence that DEPTOR is an important target for the treatment of multiple myeloma and raise the possibility that inhibitors of DEPTOR could enhance the effectiveness of melphalan, in the treatment of multiple myeloma.
Footnotes
Funding
This work was supported in part by the National Natural Science Foundation of China (No. 30871111), the Provincial Education Department of Fujian Program (No. JB11056) and the National Natural Science Foundation of China (No. 81272628).
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
