Abstract
Objective:
To investigate the effects of a phosphatase and tensin homologue (PTEN) antisense oligonucleotide on PTEN and mammalian target of rapamycin (mTOR) mRNA and protein, cell proliferation and apoptosis in oesophageal squamous cell carcinoma (OCSS) cell lines.
Methods:
EC9706 and EC1 cells were transfected with PTEN antisense oligonucleotide, sense oligo -nucleotide or nonsense oligonucleotide. Cell proliferation and apoptosis were quantified. Immuno cyto chemistry and in situ hybridization were used to determine PTEN and mTOR protein and mRNA levels, respectively.
Results:
Transfection with PTEN antisense oligonucleotide dose-and time-dependently enhanced cell proliferation and inhibited apoptosis in both EC9706 and EC1 cells. PTEN mRNA and protein were significantly downregulated, and mTOR protein and mRNA were significantly upregulated.
Conclusion:
These data suggest that PTEN is an important tumour suppressor gene in the development of OSCC.
Introduction
Oesophageal cancer is the sixth most common cause of cancer-related death worldwide. 1 Oesophageal squamous cell carcinoma (OSCC) is the dominant histological type, 2 and is one of the most frequently diagnosed cancers in the developing world, particularly in China and other Asian countries. 3 The aetiology and mode of carcinogenesis of this disease are not fully understood, however. 4 The prognosis of patients with OSCC remains poor despite improvements in therapeutic strategies, with the 5-year survival rate being 5 – 10%. 5 Novel molecular markers for the diagnosis and treatment of OSCC are therefore required.
The proline-regulated serine/threonine protein kinase, mammalian target of rapamycin (mTOR), is a component of the phosphatidylinositol-4,5-bisphosphate 3-kinase (PI3K)/protein kinase B (Akt)/mTOR signalling pathway. 6 Studies have demonstrated that mTOR is involved in the regulation of many biological processes including protein synthesis, cell proliferation, cell-cycle progression and apoptosis,7,8 and that the mTOR pathway is activated in many human tumours, including OSCC.9 – 11 The phosphatase and tensin homologue (PTEN) gene has tumour suppressive activity12,13 and acts as a molecular switch in the PI3K/Akt/mTOR signalling pathway. 14 PTEN dephosphorylates 3,4,5-phosphatidyl inositol triphosphate (PIP3), 15 reducing the activation of Akt, mTOR complex 1, and ribosomal protein S6 kinase. PTEN deficiency increases cell growth, proliferation and survival. 16 Mutation of PTEN occurs in a number of human cancers such as glioblastoma, endometrial carcinoma, melanoma and advanced prostate adenocarcinoma.17 – 22 Decreased PTEN expression occurs in OSCC and its levels are correlated with tumour differentiation.23,24 Studies have demonstrated that inactivation of PTEN and activation of the downstream mTOR signalling pathway are closely related to the proliferation, apoptosis, invasiveness and metastasis of tumour cells.25 – 27
Antisense oligonucleotides are useful in the study of gene function because of their ability to inhibit the expression of target genes, specifically.28,29 The aim of the present study was to determine the effects of a PTEN antisense oligonucleotide on PTEN and mTOR mRNA and protein, cell proliferation and apoptosis in the human OSCC cell lines EC9706 and EC1.
Materials and methods
Cell Culture
The EC9706 cell line was provided by the State Key Laboratory of Molecular Oncology, Chinese Academy of Medical Sciences, Beijing, China; EC1 was provided by Dr Shihua Cao, Department of Pathology, Hong Kong University, Hong Kong. Cells were cultured in RPMI 1640 medium (Sijiqing Company, Beijing, China) supplemented with 10% fetal bovine serum (Hyclone Laboratories, Logan, UT, USA), 100 IU/ml penicillin and 100 μg/ml streptomycin, at 37°C, in 5% CO2 in air.
Transfection
Antisense, sense and nonsense oligonucleotides were designed with reference to the PTEN mRNA sequence (available at: http://www.ncbi.nlm.nih.gov/nuccore/NM_001243157.1) using oligo -nucleotide design software (TaKaRa Bio, Dalian, China) and synthesized by Beijing AuGCT DNA-SYN Biotechnology (Beijing, China). EC9706 and EC1 cells were seeded in 24-well plates (5 × 105 cells/well) and incubated for 24 h until approximately 90% confluent. Cells were then incubated with 33 μl Lipofectamine™ 2000 (Invitrogen, Carlsbad, CA, USA) together with 3, 6, or 12 μmol/l PTEN antisense oligonucleotide (5′-GTTCGTCCCTTTCCAGCTTTACA-3′), 12 mol/l PTEN nonsense oligonucleotide (5′-GTCTATACTACCAGACAGCTTGAGT-3′), or 12 μmol/l PTEN sense oligonucleotide (5′-GCCCTAGATTTCTATGGGGAAGT-3′) for 24, 48 or 72 h, according to the manufacturer's instructions. Parallel negative control cultures were incubated with Lipofectamine™ 2000 alone. Positive control cultures were transfected with 12 μmol/l fluorescein isothiocyanate-labelled PTEN antisense oligonucleotide (Beijing AuGCT DNA-SYN Biotechnology) for 24 h. Labelled cells were observed with a fluorescence microscope at an excitation wavelength of 520 – 530 nm, in order to determine transfection efficiency.
Immunocytochemistry
Immunocytochemistry was performed using the PV2000 immunohistochemistry kit (Zhongshan Golden Bridge Biotechnology, Beijing, China), according to the manufacturer's instructions. Cells were plated on sterile glass slides (105 cells/slide) and cultured for 24 h. The slides were then rinsed three times with phosphate buffered saline for 3 min each wash (PBS; 0.01 M, pH 7.4), fixed in 4% formaldehyde for 10 min at room temperature, rinsed with PBS and incubated with 3% hydrogen peroxide for 10 min. Nonspecific binding was blocked by incubation with 5% normal goat serum for 30 min at room temperature followed by three washes with PBS (3 min each wash). Slides were incubated with rabbit antihuman PTEN or mTOR antibody (1 : 100 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA) or PBS (negative control) overnight at 4°C, then washed three times with PBS (3 min each wash). The slides were incubated with goat antirabbit immunoglobulin G secondary antibody (Zhongshan Golden Bridge Biotechnology) for 30 min at 37 °C and staining was visualised using 0.03% 3,3′ diamino benzidine. Cells were immediately examined via light microscopy and staining was evaluated in 10 high power fields (× 400 magnification). Extent of staining (percentage of positive cells) was scored as 0 (< 1%), 1 (1 – 25%), 2 (26 – 50%), 3 (51 – 75%), 4 (76 – 100%). Staining intensity was scored as 0 (no staining), 1 (pale yellow), 2 (brown to yellow), 3 (tan). The final score was calculated as the product of the two individual scores.
In Situ Hybridization
Cells were plated on sterile glass slides (105 cells/slide), cultured for 24 h and washed twice with PBS (5 min each wash). Cells were dehydrated in 70 % and 50% ethanol for 5 min each, followed by incubation in 3% hydrogen peroxide and 0.3% Triton X-100™ in PBS at room temperature for 20 min. Slides were heated at 60°C for 1 h in 3% citrate buffer (pH 7.0), then incubated in 25% proteinase K for 2 – 3 min at room temperature. After proteinase K treatment, slides were incubated in prehybridization buffer (Boster Biological Technology, Wuhan, China) containing 0.2 N hydrochloric acid, 0.1 M triethanolamine and 0.02 M acetic anhydride for 2 – 4 h at 42 °C. Slides were then incubated with 20 μl hybridization buffer (Boster Biological Technology) containing 6 – 12 ng of oligonucleotide probe against PTEN mRNA
Cells were examined via light microscopy and staining was evaluated in 10 high power fields (× 400 magnification). Extent of staining (percentage of positive cells) was scored as 0 (< 1%), 1 (1 – 25%), 2 (26 – 50%), 3 (51 – 75%), 4 (76 – 100%). Staining intensity was scored as 0 (no staining), 1 (light blue), 2 (blue), 3 (blue/violet). The final score was calculated as the product of the two individual scores.
Cell Proliferation
Cell proliferation was quantified via MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetra -zolium bromide) assay. Cells were seeded into 96-well plates at 5 × 103 cells in 100 μl per well and cultured for 24 h, after which the culture medium was replaced with an equal volume of serum-free medium. After a further 42 h, 10 μl of the culture medium was replaced with an equal volume of fresh medium containing 5 g/l MTT and 150 μl dimethyl sulphoxide. Cultures were incubated for a further 6 h then terminated by the addition of 20% sodium dodecyl sulphate (100 μl/well). The absorbance at 570 nm was determined using a microplate spectrophotometer, and cell proliferation was calculated as optical densityexperimental group/ optical densitycontrol group.
Apoptosis Analysis
Cells were plated on sterile glass slides (105 cells/slide) and cultured for 4 h. Apoptosis was determined using a terminal deoxynucleotidyl transferase-mediated biotinylated dUTP nick end-labelling kit (Roche, Basel, Switzerland), according to the manufacturer's instructions. Slides were immediately examined via light microscopy and 200 cells were evaluated per slide. Apoptotic index was calculated as (number of apoptotic cells/200) × 100%.
Statistical Analyses
Data were presented as mean ± SD, and between-group comparisons were made using one-way analysis of variance or Student's t-test. Statistical analyses were performed using SPSS® version 13.0 (SPSS Inc., Chicago, IL, USA) for Windows®. A two-sided P-value < 0.05 was considered statistically significant.
Results
Transfection of EC9706 and EC1 cells with fluorescent-labelled PTEN antisense oligo -nucleotide was successful, as shown in Fig. 1.
Fluorescence photomicrographs of human oesophageal squamous cell carcinoma cell lines (A) EC9706 and (B) EC1, transfected with 12 μmol/l fluorescein isothiocyanate-labelled phosphatase and tensin homologue (PTEN) antisense oligonucleotide for 24 h, indicating effective transfection. Unlabelled nontransfected cells are visible (arrow). Original magnification × 400
Transfection of EC9706 and EC1 cells with PTEN antisense oligonucleotide significantly dose- and time-dependently increased cell proliferation (P < 0.05 for all comparisons; Table 1) and decreased apoptosis (P < 0.05 for all comparisons; Table 2) compared with nontransfected cells. Transfection with sense or nonsense oligonucleotide had no effect on cell proliferation or apoptosis. In addition, transfection of EC9706 and ECl cells with PTEN antisense oligonucleotide significantly decreased PTEN and increased mTOR protein (as assessed by immunocytochemical staining; P < 0.05 for all comparisons, Tables 3 and 4) and decreased PTEN and increased MTOR mRNA levels (as assessed by in situ hybridization), in a dose- and time-dependent manner (P < 0.05 for all comparisons; Tables 5 and 6).
Effect of transfection with phosphatase and tensin homologue (PTEN) antisense oligonucleotide, sense oligonucleotide or nonsense oligonucleotide for 24, 48 or 72 h on cell proliferation in human oesophageal squamous cell carcinoma cell lines EC9706 and EC1
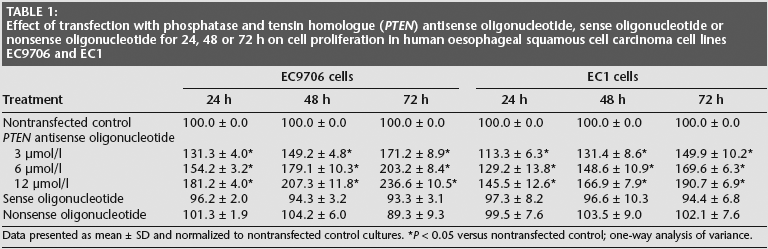
Data presented as mean ± SD and normalized to nontransfected control cultures.
P < 0.05 versus nontransfected control; one-way analysis of variance.
Effect of transfection with phosphatase and tensin homologue (PTEN) antisense oligonucleotide, sense oligonucleotide or nonsense oligonucleotide for 24, 48 or 72 h on apoptosis in human oesophageal squamous cell carcinoma cell lines EC9706 and EC1

Data presented as mean ± SD.
P < 0.05 versus nontransfected control; one-way analysis of variance.
Effect of transfection with phosphatase and tensin homologue (PTEN) antisense oligonucleotide, sense oligonucleotide or nonsense oligonucleotide for 24, 48 or 72 h on immunocytochemical staining a for PTEN protein in human oesophageal squamous cell carcinoma cell lines EC9706 and EC1

Data presented as mean ± SD.
Staining was evaluated in 10 light microscopy fields (x 400 magnification). Extent of staining (% of positive cells) scored as 0 (< 1%), 1 (1 - 25%), 2 (26 - 50%), 3 (51 - 75%), 4 (76 - 100%). Staining intensity scored as 0 (no staining), 1 (pale yellow), 2 (brown to yellow), 3 (tan). Final score was calculated as the product of the two individual scores.
P < 0.05 versus nontransfected control; one-way analysis of variance.
Effect of transfection with phosphatase and tensin homologue (PTEN) antisense oligonucleotide, sense oligonucleotide or nonsense oligonucleotide for 24, 48 or 72 h on immunocytochemical staining a for mechanistic target of rapamycin (mTOR) protein in human oesophageal squamous cell carcinoma cell lines EC9706 and EC1

Data presented as mean ± SD.
Staining was evaluated in 10 light microscopy fields (x 400 magnification). Extent of staining (% of positive cells) scored as 0 (< 1%), 1 (1 - 25%), 2 (26 - 50%), 3 (51 - 75%), 4 (76 - 100%). Staining intensity scored as 0 (no staining), 1 (pale yellow), 2 (brown to yellow), 3 (tan). Final score was calculated as the product of the two individual scores.
P < 0.05 versus nontransfected control; one-way analysis of variance.
Effect of transfection with phosphatase and tensin homologue (PTEN) antisense oligonucleotide, sense oligonucleotide or nonsense oligonucleotide for 24, 48 or 72 h on PTEN mRNA a levels in human oesophageal squamous cell carcinoma cell lines EC9706 and EC1

Data presented as mean ± SD.
Assessed via in situ hybridization. Staining was evaluated in 10 light microscopy fields (x 400 magnification). Extent of staining (% of positive cells) scored as 0 (< 1 %), 1 (1 - 25%), 2 (26 - 50%), 3 (51 - 75%), 4 (76 - 100%). Staining intensity scored as 0 (no staining), 1 (light blue), 2 (blue) or 3 (blue/violet). Final score was calculated as the product of the two individual scores.
P < 0.05 versus nontransfected control; one-way analysis of variance.
Effect of transfection with phosphatase and tensin homologue (PTEN) antisense oligonucleotide, sense oligonucleotide or nonsense oligonucleotide for 24, 48 or 72 h on mechanistic target of rapamycin (mTOR) mRNA a levels in human oesophageal squamous cell carcinoma cell lines EC9706 and EC1

Data presented as mean ± SD.
Assessed via in situ hybridization. Staining evaluated in 10 light microscopy fields (x 400 magnification). Extent of staining (% of positive cells) scored as 0 (< 1 %), 1 (1 - 25%), 2 (26 - 50%), 3 (51 - 75%), 4 (76 - 100%). Staining intensity scored as 0 (no staining), 1 (light blue), 2 (blue), 3 (blue/violet). Final score was calculated as the product of the two individual scores.
P < 0.05 versus nontransfected control; one-way analysis of variance.
Discussion
The P13K/Akt/mTOR signalling pathway is involved in cell growth, apoptosis, angiogenesis and cell-cycle regulation.6,30 The aberrant activation of this pathway plays an important role in tumour development, invasion and metastasis.31–33 Abnormalities in PTEN expression are implicated in tumour development.34,35 PI3K is activated after cell stimulation by growth-and other factors, leading to phosphorylation of phosphatidylinositol 3,4-bisphosphate (PIP2) and the subsequent activation of Akt and mTOR. 36 Activation of the proline-regulated serine/threonine protein kinase, mTOR, indirectly initiates mRNA translation via phosphorylation of eukaryotic initiation factor 4E binding protein and the ribosomal protein P70S6K. 6 PTEN is a negative regulator of the PIK3/Akt pathway, 37 and mutated PTEN enhances PIK3/Akt activity, leading to increased downstream mTOR activity. 6 In addition, PTEN dephosphorylates PIP3 to PIP2, lowering PIP3 concentrations and negatively regulating PI3K/Akt signal transduction. 6 Research has shown that a PTEN antisense oligonucleotide effectively silenced PTEN gene expression, thereby increasing PI3K and activating Akt, resulting in upregulation of mTOR and abnormal cell proliferation. 38
In the present study, transfection of the OSCC cell lines EC9706 and EC1 with PTEN antisense oligonucleotide downregulated PTEN protein and mRNA, increased cell proliferation, inhibited apoptosis and upregulated mTOR protein and mRNA. These findings suggest that PTEN is an important tumour suppressor gene in the development of OSCC.
Footnotes
Conflicts of interest: The authors had no conflict of interest to declare in relation to this article.
