Abstract
Objectives
To investigate the effects of glutamate receptor, ionotropic, alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionate (AMPA) 2 (GRIA2) rs4260586 and glutamate receptor, ionotropic, AMPA 4 (GRIA4) rs10736648 single nucleotide polymorphisms (SNPs) on response to antidepressants in Korean patients with major depressive disorder (MDD), and to ascertain whether epistatic interactions might exist between these SNPs.
Methods
In this retrospective analysis, patients were assessed at hospital admission and discharge using the Montgomery–Åsberg depression rating scale (MADRS). A multiple regression model was employed to investigate the effects of the two SNP variants on clinical/sociodemographic outcomes relating to MDD.
Results
Out of 145 Korean patients, the presence of both GRIA2 rs4260586 and GRIA4 rs10736648 polymorphisms had no significant association with MADRS improvement scores or other clinical/sociodemographic variables.
Conclusions
These data potentially suggest a lack of epistatic interaction between GRIA2 and GRIA4 variants, regarding clinical outcomes in patients with MDD. The study was limited by small sample size, use of different antidepressants and incomplete coverage of genes under investigation. Future research should include larger patient samples treated with different antidepressants, analysis of different SNPs and/or investigation of different gene–gene interactions within the glutamatergic system.
Keywords
Introduction
Major depressive disorder (MDD) is a common mental disorder, with a lifetime US incidence of 12% in men and 20% in women. 1 Evidence suggests that MDD has a strong genetic aetiology. 2 Several genetic variants – including single nucleotide polymorphisms (SNPs) within genes encoding the serotonin transporter and several serotonin receptors – significantly modulate the response to antidepressants. 3
Pharmacogenetic studies of psychotropic drug response that focus on one only SNP have shown inconsistent results or relatively small effects. 4 Consequently, there is increasing emphasis on investigating SNPs within the genes encoding transporters and receptors that are unrelated to the serotonergic system. These include investigations of the glutamatergic system, 5 or of epistatic interactions between genetic polymorphisms in candidate genes that may influence treatment outcomes in complex diseases, such as psychiatric disorders.6,7
Research investigating possible associations between the glutamatergic system and antidepressant response has gained attention, with Maeng and Zarate hypothesizing that the efficacy of antidepressant drugs might be explained (at least in part) by direct or indirect actions of these drugs on the glutamatergic system. 5 Consistent with this hypothesis, there is evidence that antidepressants can modulate glutamatergic transmission, 8 and that variations within some glutamatergic genes can modulate antidepressant response. 9
Epistasis is defined as a functional interaction between genes, encompassing various events including promoter activity control, epigenetic control, chromatin remodelling, and many other molecular reactions. 10 These events can impact cell life-cycles and complex traits, and are orchestrated through genetically driven complex, yet flexible, activities. 10 Thus, research into epistatic interactions between different candidate genes could be relevant in understanding the biological diversity that could influence antidepressant response.
Interactions between trace amine-associated receptor 6 and heat shock protein-70 gene polymorphisms have been found to affect the development of schizophrenia and mood disorders.11,12 The present retrospective analysis of a published dataset 13 investigated the relationship between genetic variants of glutamate receptor, ionotropic, alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionate (AMPA) 2 (GRIA2) and glutamate receptor, ionotropic, AMPA 4 (GRIA4), in a sample of 145 Korean MDD patients treated with different antidepressants; such variants may influence antidepressant response. These two genes, which code for two different subunits of the glutamatergic AMPA-sensitive receptors, have been investigated individually; 13 no association between the GRIA2 and GRIA4 polymorphisms investigated and clinical outcomes was found.
Patients and methods
Study population
The present retrospective study was conducted at Bucheon St Mary’s Hospital, The Catholic University of Korea College of Medicine, Bucheon, Kyounggi-Do, Republic of Korea, in October 2012. Details of the study population have been reported. 13 Briefly, consecutive inpatients diagnosed with MDD according to the Diagnostic and Statistical Manual of Mental Disorders fourth edition (DSM-IV) criteria 14 were included and underwent clinical interview using the Mini International Neuropsychiatric Interview (MINI). 15 There was no restriction regarding the type of antidepressant treatment administered. Patients were excluded if they had: current severe (or unstable) medical or neurological conditions; current treatment with long-acting antipsychotic drugs; concomitant alcohol and substance abuse disorders. Patients were also excluded if they were not of Korean ethnicity. 13 All patients admitted to the hospital – including those in the present study – are routinely assessed for severity of illness at admission and at discharge, by means of the Montgomery–Åsberg Depression Rating Scale (MADRS). 16 Response was defined a priori as a ≥50% reduction in symptoms between admission and discharge; remission was defined as a MADRS score ≤7 at discharge, both as described previously. 17 The following additional clinical and sociodemographic variables were recorded: total MADRS scores at admission and discharge; MDD subtypes; age; sex; age at MDD onset; family history of MDD; past history of suicide attempt; type of antidepressant and concomitant medication used.
The study protocol was approved by the Institutional Review Board of Bucheon St Mary’s Hospital, The Catholic University of Korea College of Medicine (approval number, HC10TISI0031). All patients included in the study provided written informed consent.
Outcome measures
The main outcome measure was the relationship between GRIA2 rs4260586 and GRIA4 rs10736648 SNPs and improvements in depressive symptoms, as measured by the change in total MADRS scores between admission and discharge. Secondary outcome measures included: presence of both GRIA2 rs4260586 and GRIA4 rs10736648 polymorphisms and total MADRS scores at admission and discharge; any response to treatment; remission; MDD subtypes; age; sex; age at MDD onset; family history of MDD; past history of suicide attempt; duration of admission; type of antidepressant used; concomitant anxiolytic therapy.
Genotyping of GRIA2 rs4260586 and GRIA4 rs10736648 SNPs
The protocol used to genotype the patient population has been described previously. 13 To cover the highest possible proportion of the gene, the GRIA2 rs4260586 and GRIA4 rs10736648 SNPs were selected from SNPs within the original dataset. 13 One SNP was selected for each gene that tagged the largest number of SNPs within the same gene. This approach ensured that ∼14% of GRIA2 variations and ∼1% of GRIA4 variations were covered.
Statistical analyses
Statistical analyses were performed using the STATISTICA software package, version 5 (StatSoft Italia, Vigonza, Padua, Italy) for Windows®. A multiple regression model was used to investigate the relationship between the two genotypes and clinical and sociodemographic variables. All P-values were 2-tailed and statistical significance was set at the P ≤ 0.004 level, after correction of multiple comparisons. These parameters gave sufficient power (0.80) to detect a medium–large (d = 0.29) effect size for patients carrying the TT rs4260586 genotype, compared with those carrying the AT genotype. 18 Such an effect size corresponded with the possibility of detecting four-point differences in final MADRS scores.
Results
Influence of GRIA2 rs4260586 and GRIA4 rs10736648 single nucleotide polymorphisms on clinical and sociodemographic outcomes, in Korean patients with major depressive disorders (MDD).
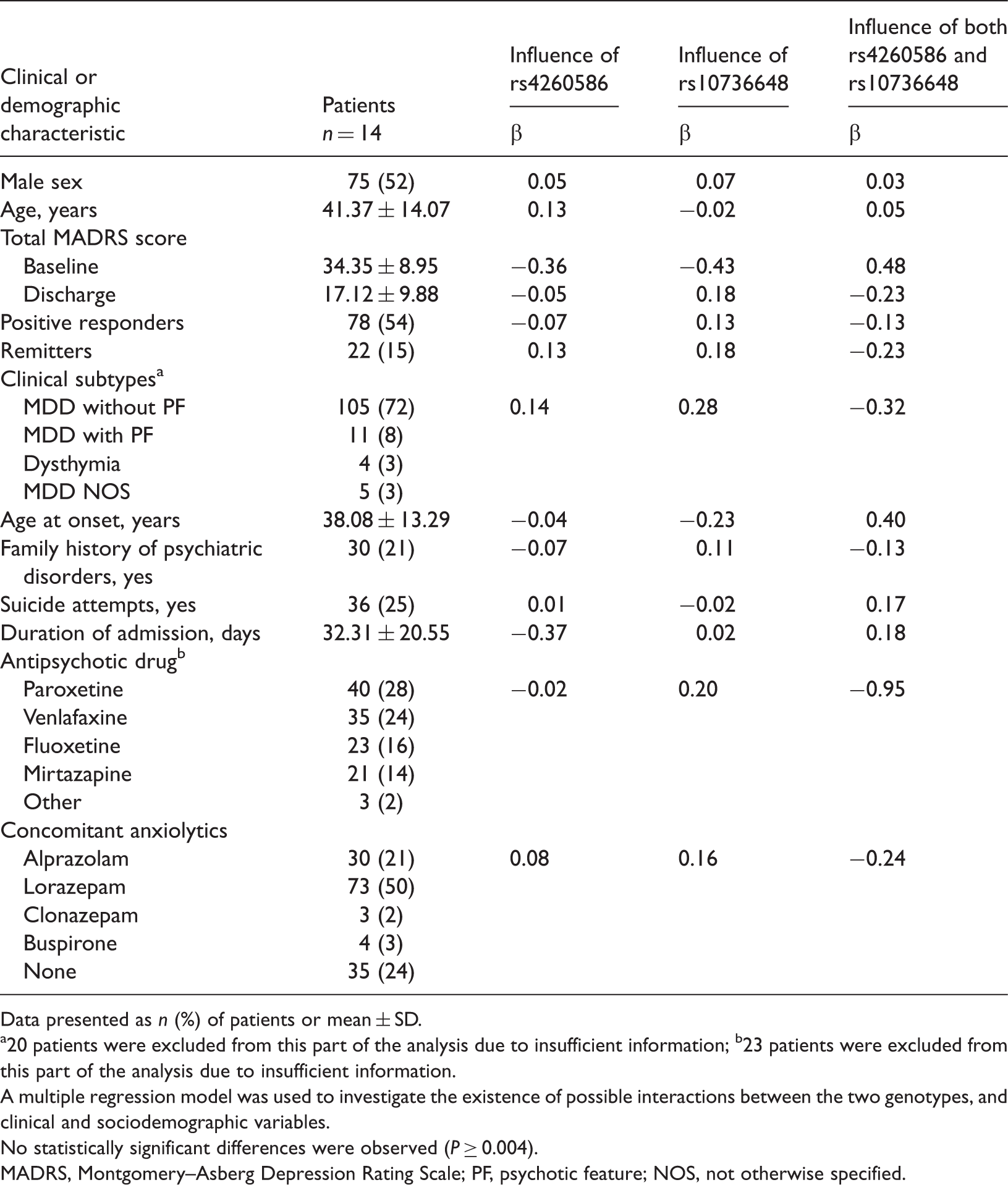
Data presented as n (%) of patients or mean ± SD.
20 patients were excluded from this part of the analysis due to insufficient information; b23 patients were excluded from this part of the analysis due to insufficient information.
A multiple regression model was used to investigate the existence of possible interactions between the two genotypes, and clinical and sociodemographic variables.
No statistically significant differences were observed (P ≥ 0.004).
MADRS, Montgomery–Asberg Depression Rating Scale; PF, psychotic feature; NOS, not otherwise specified.
Discussion
In an attempt to understand whether potential epistatic interactions exist, the present study explored whether the presence of both the GRIA2 rs4260586 and GRIA4 rs10736648 SNPs was associated with improved MADRS scores and other clinical variables, in a sample of patients with MDD who were being treated with various antidepressants. There was no significant correlation between the presence of the two SNPs and improvement in MADRS scores, which potentially suggests that there is no epistatic interaction between the GRIA2 rs4260586 and GRIA4 rs10736648 polymorphisms, in terms of MDD. A possible explanation for this negative finding could be that the sample size was too small to detect subtle differences that are usually associated with single genes or gene–gene interactions, in complex disorders. 19 Another possibility is that the results reflect a lack of epistatic interaction between the two SNPs investigated and clinical improvement, in terms of MADRS scores. In another study that focused on single SNPs within GRIA2 and GRIA4, no significant association with MADRS scores was observed. 13 There is, however, some evidence to suggest that epistatic interactions can be observed, even when single SNPs considered on an individual basis do not lead to positive findings.20,21 Such a possibility raises substantial computational and theoretical difficulties, when considering all possible gene–gene interactions and other forms of gene-expression control. 22
No statistically significant effects were observed regarding any of the secondary outcome measures in the presence of both GRIA2 rs4260586 and GRIA4 rs10736648 polymorphisms, further suggesting the possibility that no relevant epistatic interactions exist between the two SNPs under investigation. These data support results from a study that investigated the presence of these polymorphisms on an individual basis. 13
Several limitations of the present study should be considered. First, the lack of associations observed could be simply due to the lack of statistical power that, in turn, could obscure small effects exerted by single SNPs. This issue is of particular concern if one considers that, even among pharmacogenetic studies with large sample sizes, results tend to remain conflicting.23,24 A further limitation is the use of antidepressants with different mechanisms of action; this prevents conclusions from being drawn, with regard to the influence of these SNPs on specific drugs or classes of drugs. The decision to include patients who were being treated with different drugs could, however, have the advantage of being closer to ‘real-world’ clinical practice. The duration of hospitalization in the present study may be considered insufficient to ascertain a lack of response and remission; however, this time frame is consistent with common clinical practice.25,26 Finally, this study was limited by the incomplete coverage of the genes under investigation, due to the tagging approach used.
The analysis of epistasis between different genes is particularly relevant to genetic-association studies, as it increases knowledge of the mechanism of action of the so-called reactome.11,12 The analysis of both gene epistasis and the impact of single genotypes and haplotypes should be standard practice in future association studies.11,12
The preliminary findings that are reported as a consequence of the present study suggest that it is likely that no epistatic interactions exist between GRIA2 and GRIA4, in terms of clinical outcomes, in patients with MDD treated with different antidepressants. Further research should include a larger sample size of patients with MDD and treated with different antidepressants, or include different SNPs and/or different gene–gene interactions within other genes that are known to be involved in the glutamatergic system.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Footnotes
Funding
This study was supported by a grant of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A120004).
