Abstract
OBJECTIVES:
This study investigated the pharmacokinetic and pharmaco -dynamic profile of tolvaptan, and verified its efficacy and safety in patients with liver cirrhosis-associated ascites, with insufficient response to conventional diuretic treatment.
Methods:
This multicentre, doubleblind, parallel-group study allocated patients with cirrhosis to receive either 3.75 or 7.5 mg/day tolvaptan orally, once daily, for 7 days. Pharmacokinetic, pharmacodynamic and efficacy variables were measured.
Results:
Tolvaptan was shown to have high plasma concentrations, and prolonged duration of maximum concentration and half life, in these patients with impaired hepatic function. Tolvaptan resulted in dose-dependent decreases in body weight and ascites volume, and increases in urine output. There were no effects on urinary or serum electrolytes. Tolvaptan was well tolerated, with a good safety profile.
Conclusions:
Tolvaptan at 3.75 mg/day exerts some effects due to the pharmacokinetic profile in patients with liver cirrhosis. Tolvaptan at 7.5 mg/day is a clinically useful option for treating patients who do not respond well to conventional diuretics.
Introduction
Hepatic function plays a central role in the metabolism kinetics of most drugs, with the result that patients with hepatic dysfunction are more sensitive to drug effects compared with those with no hepatic dysfunction. Dose adjustment in patients with hepatic dysfunction may be essential to avoid excessive drug accumulation and serious adverse reactions. 1 Ascites is the most common complication of liver cirrhosis, 2 but ascites management in patients with liver cirrhosis is complicated by their poor response to conventional diuretics. 3 High-dose loop diuretic treatment may have undesirable adverse effects, including activation of the renin–angiotensin and sympathetic nervous systems, electrolyte disturbances, and worsening of renal function.4 – 7 making it difficult to perform dose-escalating adjustments (for optimal diuretic effects) in patients with hepatic dysfunction.8 – 10
Renal vasopressin V2 receptor antagonists increase solute-free water excretion and serum sodium concentrations.11,12 Tolvaptan (developed by Otsuka Pharmaceutical, Osaka, Japan) is an oral nonpeptide selective vasopressin V2 receptor antagonist for the treatment of clinically significant hypervolaemic or euvolaemic hyponatraemia (serum sodium < 125 mEq/l) or less-marked hyponatraemia, in patients with heart failure, cirrhosis or syndrome of inappropriate antidiuretic hormone (SIADH). 13 Tolvaptan selectively binds the vasopressin V2 receptor and inhibits vasopressin-mediated reabsorption of water in renal collecting ducts, resulting in excretion of electrolyte-free water without increasing electrolyte excretion.14 – 17
Tolvaptan at ≥ 15 mg/day, administered orally, has been shown to improve serum sodium levels effectively in patients with SIADH, cardiac failure or cirrhosis.18,19 In addition, tolvaptan treatment resulted in decreased body weight and abdominal circumference, and improvements in ascites and oedema, in cirrhotic patients with persistent ascites and/or lower limb oedema. 20 A multiple-dose study to assess the pharmacokinetics, pharmacodynamics and safety of tolvaptan in healthy Japanese subjects found dose-dependent aquaretic effects and good tolerability at all doses (≥ 15 mg/day). 21 Tolvaptan in combination with furosemide exerted diuretic effects and decreased body weight in Japanese patients with heart failure, with 15 mg/day tolvaptan providing better results than 7.5 mg/day. 22
Patients with cirrhosis have impaired liver function, therefore there is an urgent need to evaluate the pharmacokinetics and pharmacodynamics of tolvaptan at lower doses, such as 7.5 mg/day, in these patients.
The present study investigated the pharmacokinetics and pharmacodynamics of 3.75 or 7.5 mg/day tolvaptan, administered orally for 7 days, and verified treatment efficacy and safety in patients with ascites associated with liver cirrhosis.
Patients and methods
Study Population
This multicentre, double-blind, parallel-group, phase III study was conducted between February 2010 and August 2011, at 33 study sites in Japan. Male or female inpatients with liver cirrhosis who fulfilled the following criteria were eligible for inclusion in the study: (i) aged 20 – 80 years; (ii) persistent ascites despite conventional diuretic treatment; (iii) previous imaging diagnosis of liver cirrhosis; (iv) patients whose dose of conventional diuretics could not be increased due to adverse drug reactions or risk of adverse drug reactions, or where efficacy was insufficient; (v) patients who had been receiving one of the following combination therapies (loop diuretic and oral antialdosterone agent): (a) loop diuretic dose equivalent to ≥ 40 mg/day furosemide and ≥ 25 mg/day spironolactone or (b) loop diuretic dose equivalent to ≥ 20 mg/day furosemide and ≥ 50 mg/day spironolactone; (vi) inpatients or patients who could be admitted to the study site; (vii) adequate contraception.
Exclusion criteria were: (i) hepatic encephalopathy (Inuyama classification 23 ≥ grade II), vascular invasive hepatocellular carcinoma, oesophageal or gastric varices requiring new therapy, repeated haemorrhoidal bleeding due to rectal varicose veins, heart failure, anuria or impaired urination; (ii) history of cerebrovascular disorder; (iii) history of hypersensitivity or idiosyncratic reaction to benzazepine derivatives; (iv) body mass index > 35; (v) systolic blood pressure < 90 mmHg; (vi) haemoglobin < 8.0 g/dl, total bilirubin > 4.0 mg/dl, serum creatinine > 2.0 mg/dl, serum sodium > 147 mEq/l or serum potassium > 5.5 mEq/l; (vii) inability to take oral medication; (viii) pregnancy; (ix) history of treatment with albumin products or blood products containing albumins; (x) previous investigational drug treatment; (xi) previous tolvaptan treatment; (xii) patients otherwise judged by the investigator to be inappropriate for inclusion in the study.
All patients provided written informed consent to participate in the study, and the study was approved by the institutional review board at each study site. The study was conducted in compliance with the Declaration of Helsinki according to Good Clinical Practice. 24
Treatment Design
The study comprised a screening period, a 3-day pretreatment observation period, a 7-day treatment period and a post-treatment observation period (Fig. 1). After providing informed consent, patients underwent screening tests and were enrolled in the 3-day pretreatment observation period. Patients with ascites ≥ 1000 ml (assessed via computed tomography) and increase or decrease in body weight of ≤ 1.0 kg between days 2 – 3 (or ≤ 2.0 kg between days 1 – 3) of the pretreatment observation period were eligible for treatment. Patients were allocated to receive either 3.75 or 7.5 mg/day tolvaptan in a double-blind manner, taken orally, once daily, after breakfast for 7 days. Concomitant conventional diuretic treatment was fixed from time of patient consent until final tolvaptan dose. Patients were hospitalized from the day before pretreatment observation until the completion of post-treatment observation. Final safety and efficacy evaluations were performed on day 7 following initiation of tolvaptan administration.
Design of a study to evaluate the efficacy, safety, pharmacokinetics and pharmacodynamics of two different doses of tolvaptan in patients with liver cirrhosis-associated ascites, refractory to conventional diuretic treatment
Pharmacokinetics and Pharmacodynamics
Pharmacodynamic parameters were recorded using data from the pretreatment observation period as the baseline. Parameters included daily urine volume, urine sodium excretion, urine potassium excretion, serum sodium concentration and serum potassium concentration. Urine volume and urinary electrolytes were determined from samples collected at baseline and daily during the treatment period. Blood samples for serum sodium and potassium determination were collected at baseline, 4 – 8 and 22 – 24 h after dosing on day 1, 22 – 24 h after dosing on days 2, 3 or 4, and 22 – 24 h after dosing on day 7.
Pharmacokinetic variables of tolvaptan included maximum plasma concentration (Cmax), area under the curve at 24 h (AUC24h), maximum drug concentration time (tmax, h) and half-life (t½, h). Blood samples were collected in sterile tubes containing heparin sodium at 2, 4, 6, 8, 12 and 24 h postdose on days 1 and 7 of the treatment period. Plasma tolvaptan was quantified via liquid chromatography/mass spectrometry (API 3000; MDSSciex, Toronto, Canada), with a limit of detection of 2 ng/ml.
Efficacy Evaluation
Efficacy was evaluated using data from the pretreatment observation period as baseline. Criteria included changes in body weight, ascites volume and abdominal circumference, and were used as a reflection of overall volume status. Body weight was measured before breakfast and after urination at baseline and on days 1 – 7 of treatment. Ascites volume was measured via computed tomography at baseline and on day 7. Abdominal circumference was measured before breakfast at baseline and on day 7.
Safety Assessment
Safety evaluation was based on the assessment of physical signs and symptoms, laboratory tests (haematology, clinical chemistry and urinalysis), vital signs (blood pressure, pulse rate and body temperature), 12-lead electrocardiography, and pregnancy test (in women of childbearing age).
Statistical Analyses
The study used separate analysis sets for pharmacodynamics, pharmacokinetics, efficacy and safety analyses. Demographic and other baseline characteristics were calculated by treatment group. Descriptive statistics of plasma concentrations and pharmacokinetic parameters of tolvaptan at each timepoint were calculated by group and treatment day. Pharmacokinetic parameters were calculated using pharmacokinetic analysis software (WinNonlin®, version 5.3; Pharsight Corporation, Mountain View, CA, USA). Descriptive statistics were calculated for each treatment group, for measured values and changes from baseline in pharmacodynamic parameters, body weight, ascites volume and abdominal circumference.
Statistical analyses were performed using SAS® software, version 9.1.3 (SAS Institute, Cary, NC, USA). Paired Student's t-test was used to examine differences in variables on outcome measures at baseline and treatment assessments. A two-tailed P-value < 0.05 was considered statistically significant.
Results
In total, 65 patients were enrolled in the study, 40 of whom fulfilled inclusion and screening criteria, and underwent treatment. A flowchart indicating study recruitment, group allocation and treatment compliance is shown in Fig. 2. In the 3.75 mg/day group, three of 19 patients withdrew from the study (gastrointestinal haemorrhage n = 1, protocol violation n = 2), and four of 21 patients in the 7.5 mg/day group discontinued treatment (hepatic encephalopathy n = 1, withdrawal of consent n = 1, investigators' decisions n = 2). In total 33/40 (82.5%) patients completed treatment. Baseline demographic and clinical characteristics of the study groups are shown in Table 1.
Flow chart indicating the recruitment and randomization of patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment, included in a study to evaluate the efficacy, safety, pharmacokinetics and pharmacodynamics of two different doses of tolvaptan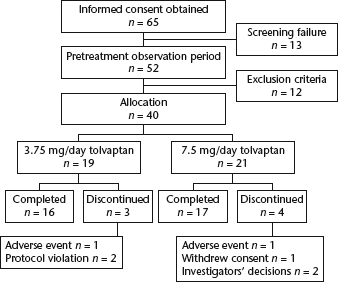
Baseline demographic and clinical characteristics of patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment, included in a study to evaluate the efficacy, safety, pharmacokinetics and pharmacodynamics of two different doses of oral tolvaptan (n = 40)

Data presented as mean ± SD or n (%) of patients.
No statistically significant between-group differences (P ≥ 0.05; paired Student's t-test).
Pharmacodynamic and efficacy datasets included 16/19 patients in the 3.75 mg/day group (n = 3 exclusions for protocol violations) and 20/21 patients in the 7.5 mg/day group (n = 1 exclusion for deviation from inclusion criteria). Pharmacokinetic datasets included 17/19 patients in the 3.75 mg/day group (n = 2 exclusions for protocol violations) and 20/21 patients in the 7.5 mg/day group (n = 1 exclusion for deviation from inclusion criteria). In the 3.75 mg/day group, one patient was judged to be eligible for pharmacokinetic evaluation despite protocol violation. In the 7.5 mg/day group, four patients who withdrew during the treatment period were excluded from pharmacokinetic, pharmacodynamic and efficacy evaluations thereafter. Patients who received ≥ 1 dose of tolvaptan were included in the safety evaluation dataset.
Pharmacokinetic parameters of tolvaptan are shown in Table 2. The Cmax and AUC24h were higher on day 7 than on day 1 in both treatment groups, and higher in the 7.5 mg/day group than the 3.75 mg/day group on both days 1 and 7, but none of these differences was statistically significant.
Pharmacokinetic parameters of oral tolvaptan in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment

Data presented as mean ± SD.
Cmax, maximum plasma concentration; AUC24h, area under the curve at 24 h; tmax, duration of maximum concentration; t½, half life.
No statistically significant differences (P ≥ 0.05; paired Student's t-test, within group comparisons; unpaired Student's t-test, between-group comparisons).
Figure 3 presents the 24-h timecourse of plasma tolvaptan concentrations on days 1 and 7 in both groups. Plasma tolvaptan concentrations remained at detectable levels at 24 h after administration on day 1 in both treatment groups. Tolvaptan concentrations before administration on day 7 were not higher than those 24 h after administration on day 1.
Timecourse of plasma concentration of tolvaptan in the 24-h period following administration of (A) 3.75 or (B) 7.5 mg doses orally, in patients with persistent ascites associated with liver cirrhosis, on days 1 and 7 of treatment
Changes in daily urine volume from baseline and during the treatment period are shown in Table 3. Daily urine volumes were significantly higher than baseline in both treatment groups throughout the treatment period (P < 0.05 for all comparisons).
Daily urine volume and change from baseline in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment, following daily oral administration of 3.75 or 7.5 mg tolvaptan
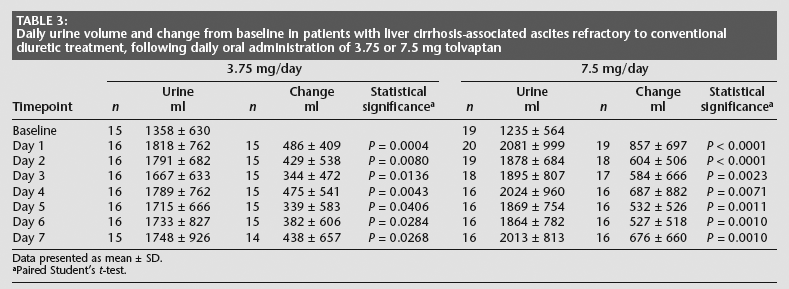
Data presented as mean ± SD.
Paired Student's t-test.
Table 4 presents data regarding total daily urinary sodium excretion. There were no significant changes from baseline in sodium excretion in either treatment group at day 1 or day 7. In addition, there were no significant changes from baseline in daily urinary potassium excretion in either group at day 1 or day 7 (data not shown).
Urinary sodium excretion and change from baseline in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment, following daily oral administration of 3.75 or 7.5 mg tolvaptan

Data presented as mean ± SD.
No statistically significant differences (P ≥ 0.05; paired Student's t-test).
Treatment with 3.75 mg/day tolvaptan had no significant effect on serum sodium concentrations (Table 5). In the 7.5 mg/day group, however, serum sodium concentrations were significantly higher than baseline at 24 h, days 2 – 4 and day 7 (P < 0.05 for all comparisons; Table 5), although these values remained within normal limits (135 – 144 mEq/l).27,28 There were no statistically significant changes in serum potassium concentrations at any timepoint in either group (data not shown).
Serum sodium concentration and change from baseline in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment, following daily oral administration of 3.75 or 7.5 mg tolvaptan

Data presented as mean ± SD.
Paired Student's t-test.
NS, not statistically significant (P ≥ 0.05).
Tolvaptan treatment induced significant decreases in body weight from baseline, starting from day 1, in both groups (P < 0.05 for all comparisons; Table 6). The decrease in body weight reached a plateau on day 4 in the 3.75 mg/day group, but patients in the 7.5 mg/day group continued to lose weight throughout the treatment period. Significant decreases in ascites volume on day 7 compared with baseline occurred in both treatment groups (P < 0.05 for both comparisons; Table 7). Abdominal circumference was smaller on day 7 than baseline in both groups, but this was only statistically significant in the 3.75 mg/day group (P = 0.028; Table 7).
Body weight and change from baseline in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment, following daily oral administration of 3.75 or 7.5 mg tolvaptan
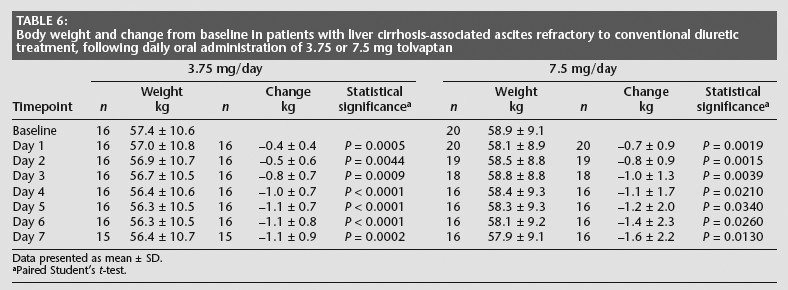
Data presented as mean ± SD.
Paired Student's t-test.
Ascites volume and abdominal circumference at baseline and after treatment with 3.75 or 7.5 mg/day tolvaptan, administered orally, for 7 days in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment

Data presented as mean ± SD.
Paired Student's t-test.
NS, not statistically significant (P ≥ 0.05).
There were no deaths in the present study. Table 8 lists adverse events experienced by patients included in the safety evaluation dataset. Serious adverse events included hepatic encephalopathy (one patient at 3.75 mg/day and three patients at 7.5 mg/day). Adverse events were reported in 13/19 patients (68.4%) in the 3.75 mg/day group and 15/21 (71.4%) patients in the 7.5 mg/day group.
Adverse events following daily oral administration of 3.75 or 7.5 mg tolvaptan for 7 days in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment

Data presented as n (%) of patients.
Adverse events observed with an incidence ≥ 10% or leading to discontinuation are presented; some patients experienced > 1 adverse event.
Patients who received at least one dose of tolvaptan were included in the safety evaluation.
Discussion
The present study investigated the pharmacokinetic and pharmacodynamic properties, and verified the efficacy and safety profiles of 3.75 and 7.5 mg/day tolvaptan, administered orally in patients with liver cirrhosis-associated ascites refractory to conventional diuretic treatment.
Research in patients with heart failure has reported pharmacokinetic parameters for the 7.5 mg/day tolvaptan that were considerably lower than those observed in the 7.5 mg/day group in the present study. 22 In addition, tolvaptan was present at detectable levels in plasma 24 h after administration in the present study, in contrast to the findings in patients with heart failure. 22 Tolvaptan plasma concentrations before administration on day 7 were not higher than those observed 24 h after administration on day 1 in the present study, suggesting no accumulation of plasma tolvaptan concentration. The findings of the present study are consistent with drug metabolism being dependent on hepatic function. 1 Tolvaptan is mainly metabolized by cytochrome P450 3A4 (CYP3A4), a member of the human hepatic microsomal cytochrome P450 superfamily. 29 It is possible that the extended AUC24h, tmax and t½ of tolvaptan in the present study are associated with diminished CYP3A4 activity, in patients with impaired hepatic function.
Administration of tolvaptan resulted in dose-dependent reductions in body weight, ascites volume and abdominal circumference, and an increase in urine volume, in the present study. There were no changes in urinary electrolyte (sodium and potassium) excretion after administration of 3.75 or 7.5 mg/day tolvaptan, demonstrating the aquaretic effect of this agent. 30 In addition, tolvaptan had minimal effects on serum electrolyte concentrations. The effects of 3.75 mg/day tolvaptan on body weight, urine volume, ascites volume and abdominal circumference may be explained by the high AUC24h and prolonged tmax and t1/2 in patients with impaired hepatic function due to cirrhosis. 31 Tolvaptan dose adjustment may be necessary in patients with liver cirrhosis, to find the optimal treatment approach for each patient. Further studies are required to determine a definitive factor or marker for optimal dose adjustment. Although administration of 3.75 mg/day tolvaptan demonstrated significant effects in the present study, 7.5 mg/day may be more beneficial, based on the continuous and consistent decrease in body weight and marked improvement in ascites volume observed at this dose.
In conclusion, 7.5 mg/day tolvaptan is a clinically useful option for the treatment of patients with liver cirrhosis who do not respond well to conventional diuretics, without inducing serum electrolyte abnormalities.
Footnotes
Conflicts of interest: This study was funded by Otsuka Pharmaceutical, Osaka, Japan. Dr Isao Sakaida received research grants from Otsuka Pharmaceutical, MSD, Chugai Pharmaceutical, and Takeda Pharmaceutical, and served as a consultant for Otsuka Pharmaceutical with payment of consultancy fee to Yamaguchi University Graduate School of Medicine. Mitsuru Okada is an employee of Otsuka Pharmaceutical. No other author has any conflict of interest to declare in relation to this article.
