Abstract
Objectives:
To investigate the presence of aquaporin (AQP) 1 and AQP2 in kidneys of cirrhotic rats with ascites, and to determine the effect of terlipressin on AQP1 and AQP2 levels and its therapeutic efficacy in ascites treatment.
Methods:
Eighteen rats were randomly divided into normal noncirrhotic rats treated with saline, cirrhotic rats treated with saline and cirrhotic rats treated with terlipressin (
Results:
Terlipressin resolved ascites in all animals in the terlipressintreated group, and significantly increased the 24-h net fluid-excretion volume and decreased portal vein pressure compared with saline treatment. AQP1 and AQP2 were significantly upregulated in cirrhotic rat kidneys compared with normal control rat kidneys. Terlipressin administration significantly down regulated AQP2 in rat kidneys but did not affect AQP1.
Conclusions:
AQP1 and AQP2 are important factors in ascites induction. Terlipressin appears to be an effective drug for the treatment of ascites due to liver cirrhosis in a rat model, possibly due to AQP2 reduction in kidneys.
Introduction
Ascites is a complication of cirrhosis that results from disruption in fluid regulation throughout the body. 1 In patients with cirrhosis, the presence of ascites complicates treatment, indicates disease severity and increases the mortality rate. 2 Despite its importance as an indicator of disease progression, the pathological mechanism of ascites in liver cirrhosis remains to be elucidated. Major factors involved in the pathogenesis of ascites are portal and sinusoidal hypertension, arterial vasodilatation and neurohumoral activation. 2 It is generally accepted that arginine vasopressin (AVP) plays an important role in these processes. AVP acts on two receptors: the V1 and V2 receptors. The V1 receptor is primarily related to vasoconstriction, whereas the V2 receptor, which acts on aquaporin (AQP) 2, controls the antidiuretic properties of AVP.3–5 AQP water channels are present in collecting ducts of kidneys, serving as the final channels that help to regulate water excretion in patients with cirrhosis. AQP levels in the kidney vary significantly between different models of liver cirrhosis,6,7 and the role played by AQPs in the pathogenesis of cirrhosis remains to be determined.
Terlipressin, a vasopressin analogue that was developed as a vessel constrictor, is widely used in the treatment of bleeding oesophageal varices and hepatorenal syndrome.8–10 Adding terlipressin to albumin was effective for treating patients with refractory ascites. 10 Because terlipressin interacts with both V1 and V2 receptors and successfully treats ascites, it was hypothesized that terlipressin may play a role in diuresis through the AQP system. Thus, the present study investigated AQP levels in the kidneys, using a rat model of cirrhosis; the effects of terlipressin on ascites and AQP following treatment were also investigated. The rat model of cirrhosis was developed using carbon tetrachloride, which is rapidly metabolized into the trichloromethyl radical by cytochrome P450 (CYP450) 2E1: the reactive intermediate attacks membrane lipids, resulting in the formation of lipid peroxide molecules and necrosis of hepatocytes. 11 Phenobarbitone can increase the sensitivity of the liver to carbon tetrachloride by increasing CYP450 activity. 12
Materials and methods
Liver Cirrhosis Rat Model
Eight-week-old male Sprague–Dawley rats (body weight 200 – 250 g) were obtained from the Laboratory Animal Research Centre, Fudan University, Shanghai, China. Animals were housed in a temperature- and moisture-controlled room with a 12-h light/dark cycle, with free access to water and food. All experimental procedures in this study were approved by the Institutional Ethics Committee of Fudan University and were conducted according to guidelines for experimental animals, developed by Fudan University.
Eighteen rats were randomly divided into three equal groups according to a computer-generated randomization schedule: (1) normal control group, normal noncirrhotic rats treated with saline; (2) saline-treated group, cirrhotic rats treated with saline; (3) terlipressin-treated group, cirrhotic rats treated with terlipressin. There were two randomization steps. First, rats were randomly divided into the normal control and ascites groups. To induce liver cirrhosis and ascites, 350 mg/l phenobarbital was administered orally in drinking water every day for the first week of the induction period. Rats then received 0.8 g/kg carbon tetrachloride in olive oil subcutaneously twice a week for 8 weeks. These injections decreased to three times every 2 weeks for the subsequent 6 weeks. Abdominal circumference measurements were performed weekly and a diagnostic abdominal paracentesis was performed during the last week (week 15), according to standard procedures, to confirm whether ascites had formed. The normal control group received no similar or placebo treatment. Once ascites was confirmed, rats with ascites were randomly assigned to the saline- and terlipressin-treated groups.
Study Treatment
Before treatment commenced, rats were kept in a metabolic cage for 24 h to record the total net fluid-excretion volume (defined as the urine volume minus water volume intake over a 24-h period) and urine was collected for osmotic pressure tests. Saline-treated rats received 2 ml saline via caudal vein injection daily for 5 days. A bolus injection of 0.5 – 2.0 mg terlipressin every 6 h is used in managing hepatorenal syndrome, 8 which equates to 0.04 – 0.16 mg/kg of terlipressin per day for a patient weighing 50 kg. The terlipressin-treated group in the present study was, therefore, treated with 0.05 mg/kg terlipressin (Ferring GmbH, Kiel, Germany), dissolved in saline via caudal vein injection, daily for 5 days. On the fifth day, all rats were again kept in the metabolic cage for 24 h to record the total net fluid-excretion volume and 24-h urine volume. It was predicted that terlipressin would be a therapeutically successful treatment of cirrhosis-related ascites. Thus, following treatment, rats' abdomens were exposed under anaesthesia to see whether there was any ascitic fluid.
Blood and Tissue Sampling
At the end of the treatment period, rats' abdomens were exposed under ketamine anaesthesia and portal vein pressure was measured by the catheter insertion method.
13
Blood (6 ml) was taken from the inferior vena cava, 3 ml for plasma separation and 3 ml for serum serparation. Half of the blood sample was treated with 0.1 mg/ml heparin for 1 h at room temperature; plasma was separated by centrifugation at 2300
Detection of Serum Parameters
Rat serum parameters, including blood sodium, serum alanine aminotransferase (ALT), total bilirubin and albumin levels, together with creatinine and urea nitrogen levels, were measured using standard assays on a Hitachi 7600 automatic analyser (Hitachi, Tokyo, Japan).
Plasma AVP Detection
Plasma AVP levels were detected using a commercially available diagnostic radioimmunoassay AVP test (Diagnostic Systems Laboratory, Webster, TX, USA), according to the manufacturer's instructions.
RT–PCR Analysis of AQP Mrna
Reverse transcription–polymerase chain reaction (RT–PCR) was conducted on rat tissue samples, obtained as described above. Total RNA was extracted from 50 – 100 mg of frozen tissue using TRIzol® reagent (Invitrogen, Gaithersburg, MD, USA) according to the manufacturer's instructions. Table 1 shows the primer sequences used in these analyses. The PCR was performed using a one-step RT–PCR kit (Access RT–PCR System; Promega, Madison, WI, USA). Final concentrations of the components included in each 50-μl reaction mix were: avian myeloblastosis virus/thermus flavus (AMV/Tfl) 1× reaction buffer; 0.2 mM deoxyribonucleoside triphosphate mix; 1 mM magnesium sulphate; 1 μM of each primer; 0.1 μg/μl AMV reverse transcriptase; 0.1 μg/μl Tfl DNA polymerase; 0.1 μg/μl RNA sample. The PCR was performed using a GeneAmp™ PCR System 9600 (PerkinElmer, Foster City, CA, USA) as follows: one cycle at 48 °C for 45 min and inactivation of AMV reverse transcriptase at 94 °C for 2 min, then 40 cycles of denaturation at 94 °C for 30 s, annealing at 50°C for 1 min and elongation at 68 °C for 2 min, followed by one cycle of final elongation for 7 min at 68 °C. The PCR products were analysed using 2.5% agarose gel electrophoresis, ethidium bromide staining and ultraviolet light; mRNA levels were quantified by measuring band density with a UVP GDS-8000 Image Analysis System (UVP, Upland, CA, USA).
Primer sequences used in the reverse transcription–polymerase chain reaction analyses of aquaporin (AQP) 1 and 2 in liver, kidney and abdominal wall tissues of normal control rats (noncirrhotic), saline-treated cirrhotic rats and terlipressin-treated cirrhotic rats

GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Immunohistochemical Analysis of AQP Protein
Sections of paraffin-embedded tissues (5-μm thickness, obtained as described above) were used for immunostaining. Tissue sections were deparaffinized in xylene and rehydrated through a graded series of alcohols. Endogenous peroxidases were inactivated by immersing sections in 0.6% hydrogen peroxide in methanol at room temperature for 30 min. Antigen retrieval was performed in 0.01 mmol/l citric acid buffer (pH 6.0) for 10 min in a 700 W microwave oven at 95 °C. Sections were incubated with primary antibodies, rabbit anti-AQP1 antibody (1:80 dilution) or rabbit anti-AQP2 antibody (1:200 dilution) (both from Calbiochem–Novabiochem, San Diego, CA, USA) overnight at 4 °C, then with horseradish peroxidase conjugated goat antirabbit immunoglobulin G (1:50 dilution; ImmuClub Labs, Sunnyvale, CA, USA) for 1 h at 37 °C. All antibodies were diluted in 0.01 M phosphate-buffered saline (PBS, pH 7.4). Both incubation steps were followed by washing three times with 0.01 M PBS, pH 7.4. Finally, the sections were developed with 3-amino-9-ethylcarbazole and counterstained with haematoxylin. Quantification of AQP1 or AQP2 protein in immunohistochemical stained specimens was evaluated by measuring the positive intensity using KS 400 image analysis software (Carl Zeiss, Oberkochen, Germany). Positive intensity was defined as the mean grey scale of the positive area times the percentage of the positive stained area.
Statistical Analyses
Statistical analyses were carried out using the SPSS® statistical package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Data were presented as mean ± SD and were analysed using an unpaired Student's
Results
All rats in the saline- and terlipressin-treated groups developed ascites. Terlipressin treatment resolved ascites in all six rats (100%) in the terlipressin-treated group compared with none of the six rats (0%) in the saline-treated group (
The 24-h net fluid-excretion volumes at baseline and after 5 days' treatment are shown in Fig. 1A. At baseline, both groups of cirrhotic rats had significantly decreased fluid-excretion volumes compared with the normal controls (
(A) The mean 24-h net fluid-excretion volumes in normal control noncirrhotic rats, saline-treated cirrhotic rats and terlipressin-treated cirrhotic rats (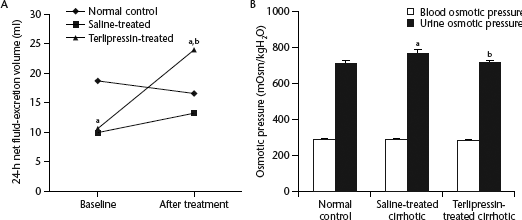
Levels of serum alanine aminotransferase (ALT), total bilirubin, albumin, creatinine and blood urea nitrogen (BUN) in normal control rats (noncirrhotic), saline-treated cirrhotic rats, and terlipressin-treated cirrhotic rats after 5 days' treatment

Data presented as mean ± SD.
After 5 days' treatment, the mean plasma AVP level was significantly higher in saline-treated cirrhotic rats than normal noncirrhotic controls (
Plasma arginine vasopressin (AVP) levels and portal vein pressures after 5 days' treatment in normal control noncirrhotic rats, saline-treated cirrhotic rats and terlipressin-treated cirrhotic rats. (A) The plasma AVP level was significantly higher in the saline-treated cirrhotic group than in normal controls; terlipressin further elevated the AVP level. (B) The portal vein pressure increased in saline-treated cirrhotic rats but was significantly attenuated by terlipressin (mean ± SD; a
The AQP1 mRNA levels in the renal cortex and medulla were significantly higher in the saline- and terlipressin treated cirrhotic rats than in the normal noncirrhotic controls (
Aquaporin (AQP) 1 and AQP2 mRNA and protein levels in kidneys of normal control noncirrhotic rats, saline-treated cirrhotic rats and terlipressin-treated cirrhotic rats. (A) Representative image of reverse transcription–polymerase chain reaction of AQP1 and AQP2 mRNA in kidneys from all three groups after 5 days' treatment. AQP1 and AQP2 mRNA was upregulated in saline-treated cirrhotic rats; terlipressin downregulated AQP2 mRNA, but showed no effect on AQP1 mRNA. (B) Positive intensity of AQP1 in the kidneys by immunohistochemical staining. AQP1 was upregulated in saline-treated cirrhotic rats compared with controls but there was no difference between the saline- and terlipressin-treated cirrhotic groups. (C) Positive intensity of AQP2 in the kidneys by immunohistochemical staining. AQP2 was elevated in saline-treated cirrhotic rats compared with controls and was downregulated by terlipressin in both the kidney cortex and medulla (mean ± SD; a
The level of AQP2 mRNA was increased in the kidneys of cirrhotic rats (saline-treated group) and was significantly greater than in the normal noncirrhotic controls (
Discussion
Ascites and water retention are the most common complications of cirrhosis; both are associated with increased mortality rates. 14 It has been reported that terlipressin increases water excretion during a water load test in cirrhotic patients without hyponatremia. 15 Another study showed a synergistic effect for diuresis, when terlipressin was administered (together with albumin and diuretics) in patients with refractory ascites. 10 In the present study, terlipressin resolved the ascites in cirrhotic rats without hepatorenal syndrome, which confirmed previous observations.16,17 Research has reported side-effects of hyponatraemia during terlipressin therapy; 18 however, this effect was not observed in the present study. Additionally, terlipressin did not affect indices of liver and renal function including serum ALT, total bilirubin, albumin, creatinine and blood urea nitrogen. Thus, these data indicated that terlipressin has the potential to be an effective drug treatment for ascites caused by cirrhosis.
One important factor contributing to ascites in patients with portal hypertension is splanchnic vasodilatation.19,20 Development of systemic and splanchnic vasodilatation results in a decrease in the effective arterial blood volume and a hyperdynamic circulation, 21 followed by portal and sinusoidal hypertension. Terlipressin selectively stimulates V1 receptors and increases mean arterial pressure and systemic vascular resistance, while decreasing portal venous blood flow. 22 Treatment of ascites in the present study was approached with the understanding that terlipressin decreases portal pressure. Reduction in portal pressure ameliorates the hyperdynamic circulation, thereby improving the effective circulatory volume and renal perfusion pressure,23,24 and increasing the glomerular filtration rate and renal function. 17
Another possible mechanism making terlipressin a potentially successful treatment for ascites is the observed downregulation of AQP2 in the kidney. AQP1 is abundant in the renal proximal tubule and descending thin limb, but AQP2 is abundant in the collecting duct principal cells. 25 Levels and excretion of AQP2 are reportedly increased in cirrhotic patients.6,26 The present study found increased levels of AQP1 and AQP2 mRNA and protein in the renal tubules of cirrhotic rats. Elevated AQP1 and AQP2 levels would facilitate reabsorption of water in the kidney and promote ascites development.
The present study showed that AQP2 was downregulated in cirrhotic rats after terlipressin treatment, accompanied by an increase in urine output. These data suggest that the decrease in AQP2 resulted in a decrease of renal water reabsorption and, consequently, resolution of ascites. The classic AQP2 pathway is that vasopressin stimulates the V2 receptor by the Gs adenylyl cyclase system and finally promotes membrane accumulation of AQP2 by dual effects on AQP2 exocytosis and endocytosis. 27 Furthermore, terlipressin has been found to increase urine excretion of AQP2 in patients with ascites. 28 Interestingly, in the present study, terlipressin decreased AQP2 but increased AVP. Because terlipressin mainly acts through V1 receptors, which are mediated by calcium, 29 other AVP-independent signalling pathways might also affect AQP2 levels. For example, AQP2 has been reported to be regulated by prostaglandin E2 or nitric oxide synthetase. 30 The AQP1 level did not change after terlipressin administration in the present study, indicating that AQP1 may not be involved in the therapeutic mechanisms of terlipressin.
In conclusion, the present study demonstrated that AQP1 and AQP2 contribute to the generation of ascites. Terlipressin treatment resolved cirrhosis-induced ascites in the current rat model, probably by reducing the AQP2 level and improving renal function. Further studies, where terlipressin is used to treat ascites due to cirrhosis in humans, are needed to demonstrate whether this drug can successfully reverse ascites, which would represent a measurable benefit to patient health.
Footnotes
The authors had no conflicts of interest to declare in relation to this article.
