Abstract
Objective:
The impact of preoperative gabapentin on tramadol consumption using patient-controlled analgesia (PCA) and postoperative pain was assessed in patients undergoing radical retropubic prostatectomy (RRP).
Methods:
In this prospective, randomized trial, 51 patients undergoing RRP were randomized into two groups: the gabapentin group received 900 mg gabapentin orally 2 h before surgery; the control group did not receive gabapentin. Postoperative analgesia was provided by tramadol PCA. Pain was assessed using a visual analogue scale for 24 h, postoperatively.
Results:
Mean cumulative tramadol consumption at 24 h was comparable in the two groups. Pain scores at 45 min, 60 min and 2 h postoperatively, and the number of patients who required rescue analgesia, were significantly lower in the gabapentin group than in the control group. Side-effects were similar in the two groups.
Conclusions:
Preoperative administration of 900 mg gabapentin did not decrease tramadol consumption, but was associated with lower pain scores in the early postoperative phase and a reduced need for rescue analgesia, compared with controls, in patients undergoing RRP.
Keywords
Introduction
Gabapentin, first introduced for the treatment of epilepsy in the early 1990s, has shown promising results for the treatment of chronic neuropathic pain. It has a high affinity to the α2δ subunit of presynaptic voltage-dependent calcium channels, which inhibits calcium influx and the subsequent release of excitatory neurotransmitters in the pain pathways. 1 There is considerable overlap between the pathophysiology of postoperative pain and that of neuropathic pain. Allodynia and hyperalgesia, which are the cardinal signs and symptoms of neuropathic pain, are also present after trauma and surgery. 1 Gabapentin reduces central sensitization by decreasing the hyperexcitability of dorsal horn neurons induced by tissue injury. 2 These antiallodynic and antihyperalgesic properties may therefore also be beneficial in acute postoperative pain. 3 A number of randomized trials have indicated that gabapentin is effective as a postoperative analgesic.3 – 7
A procedure-specific systematic review published in 2007 showed that perioperative use of gabapentin has a significant opioid-sparing effect and improves pain scores in both abdominal hysterectomy and spinal surgery. 8 Radical retropubic prostatectomy (RRP) has been the most common surgical approach for localized prostate cancer for the past two decades. 9 This procedure is performed under general anaesthesia and causes moderate to severe postoperative pain. 10 The effects of gabapentin on postoperative pain after RRP have not been reported in the literature. The present prospective, randomized study tested the hypothesis that a single preoperative 900 mg oral dose of gabapentin can decrease postoperative tramadol consumption using patient-controlled analgesia (PCA), after RRP under general anaesthesia.
Patients and methods
Patients
Patients aged between 30 and 80 years with American Society of Anesthesiologists (ASA) physical status class 1 – 3, undergoing elective RRP under general anaesthesia at the Department of Urology, Faculty of Medicine, Ege University, Izmir, Turkey, between March 2010 and October 2010, were recruited. Patients with allergy to any of the study medications, an inability to use the PCA device, coagulation abnormalities, or a history of liver disease, renal disease, gastroduodenal ulcer, chronic pain conditions or chronic opioid medication were excluded from the study.
Written informed consent was obtained from all patients and the study protocol was approved by the Ethics Committee of Ege University, Izmir, Turkey.
Anaesthesia and Surgical Procedure
Before the operation, all patients were taught to use the PCA device (Pain Management Provider, Abbott Laboratories, North Chicago, IL, USA) and a visual analogue scale (VAS) for pain assessment, where 0 indicated no pain and 10 indicated the worst possible pain.
Patients were randomly allocated to one of two groups using a computer-generated list of random numbers. Patients in the gabapentin group received 900 mg gabapentin orally with sips of water 2 h before surgery; patients in the control group did not receive any drugs preoperatively. No premedication was given to any of the patients.
After routine monitoring and intravenous access were established, each patient received the same anaesthesia and surgical technique. Anaesthesia was induced with intravenous 0.5 mg/kg atropine, 2 mg/kg propofol, 1 μg/kg remifentanil and 0.65 mg/kg rocuronium, and maintained with 2 – 2.5% sevoflurane in oxygen and air (50 : 50) and an intravenous infusion of remifentanil (0.25 – 1.0 μg/kg per min). All patients were intubated and ventilated mechanically. Heart rate, arterial blood pressure and peripheral oxygen saturation were all recorded at 5-min intervals throughout the surgical procedure. At the end of the anaesthesia period, neuromuscular block was reversed using standard doses of atropine and neostigmine.
Postoperative Analgesia
Postoperative analgesia consisted of intravenous tramadol PCA, (bolus dose of 50 mg followed by 20 mg on demand; lockout interval of 15 min) starting on arrival in the postanaesthesia care unit. Rescue analgesia consisted of 1 g paracetamol intravenously, with 75 mg diclofenac sodium, administered intramuscularly if paracetamol proved to be inadequate.
Patient Assessment
Postoperative pain at rest was assessed by a blind observer using the VAS at 0, 15, 30, 45 and 60 min, and at 2, 4, 6, 12 and 24 h. Total tramadol consumption and whether rescue analgesia was required were recorded for each patient. The presence or absence of the following adverse effects was also recorded: postoperative nausea and/or vomiting; dizziness; light-headedness; headache; pruritus; respiratory depression (respiratory rate < 8 breaths/min). Patient satisfaction was rated as excellent, good, mild or poor.
Statistical Analyses
The primary outcome measure was PCA tramadol consumption. Secondary endpoints included pain scores, need for rescue analgesia and presence of adverse effects.
Assuming a difference of 30% total tramadol consumption between the groups, 24 patients were required in each group with 80% power and a significance level of 0.05. Data were presented as mean ± SD or numbers of patients. The χ2-test was used to compare ASA class, duration of surgery, presence of adverse effects and patient satisfaction in the two groups; other data were compared using the Student's t-test. A P-value of < 0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS® software version 16.0 (SPSS Inc., Chicago, IL, USA).
Results
A total of 51 patients undergoing elective RRP were enrolled in the study. All patients completed the protocol. No significant differences in age, weight, ASA class or duration of surgery were observed between the groups (Table 1). PCA tramadol consumption and other postoperative data are summarized in Table 2. Postoperative pain scores are presented in Fig. 1.
Mean postoperative pain scores in patients undergoing radical retropubic prostatectomy under general anaesthesia with (gabapentin group, n = 25) or without (control group, n = 26) preoperative gabapentin. Patients scored pain on a visual analogue scale: scores ranged from 0 (no pain) to 10 (worst possible pain) *Statistically significant between-group difference (P < 0.05); independent samples test
Demographic data and duration of surgery for 51 patients undergoing radical retropubic prostatectomy under general anaesthesia with (gabapentin group) or without (control group) preoperative gabapentin
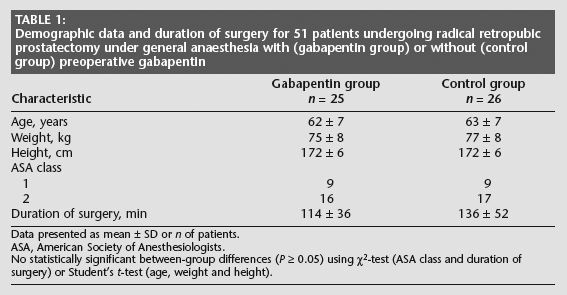
Data presented as mean ± SD or n of patients.
ASA, American Society of Anesthesiologists.
No statistically significant between-group differences (P ≥ 0.05) using χ2-test (ASA class and duration of surgery) or Student's t-test (age, weight and height).
Postoperative patient-controlled analgesia tramadol consumption, rescue analgesia and side-effects, together with patient satisfaction scores, in 51 patients undergoing radical retropubic prostatectomy under general anaesthesia with (gabapentin group) or without (control group) preoperative gabapentin

χ2-test (postoperative nausea and patient satisfaction) or Student's t-test (tramadol consumption and rescue analgesia).
Data presented as mean ± SD or n of patients.
NS, no statistically significant between-group differences (P ≥ 0.05).
The mean cumulative tramadol consumption at 24 h was comparable in the two groups (Table 2). Pain scores were significantly lower in the gabapentin group compared with controls at 45 min, 60 min and 2 h postoperatively (P < 0.05) (Fig. 1). The number of patients who required rescue analgesia was lower in the gabapentin group than in the control group (P < 0.05) (Table 2). Although more patients reported postoperative nausea in the control group than in the gabapentin group, the difference did not reach statistical significance (Table 2). No other adverse effects were reported in either group. Patient satisfaction was similarly high in both groups (Table 2).
Discussion
The results of the present study demonstrated that preoperative oral administration of 900 mg gabapentin resulted in lower pain scores in the early postoperative phase (at 45 min, 60 min and 2 h) compared with controls. Preoperative gabapentin also reduced the need for rescue analgesia without serious side-effects. However, it did not decrease overall tramadol consumption.
Gabapentin has previously been reported to be useful for early postoperative pain. A systematic review of the procedure-specific effects of gabapentin on postoperative pain analysed combined data from similar procedures by calculating the weighted mean difference (WMD) of 24-h cumulative opioid requirements and VAS pain scores in the early (6-h) and late (24-h) postoperative periods between the study groups. 8 In four trials including 112 patients undergoing abdominal hysterectomy,11 – 14 the WMD in early VAS pain scores at rest between the treatment groups was significantly in favour of gabapentin, whereas the WMD in late VAS pain scores between the treatment groups was not significant. 8 This is consistent with the findings of the present study, that gabapentin given preoperatively was effective in decreasing early, but not late, postoperative pain. The authors of the systematic review put forward a number of explanations for this effect. 8 Most of the analysed studies used a single dose of preoperative gabapentin. The maximum plasma concentration of gabapentin occurs 2 – 3 h after administration, so the plasma level of gabapentin is therefore highest in the early postoperative phase. In addition, postoperative pain scores are generally higher in the early postoperative phase than in the late phase, and so any effect on pain scores produced by gabapentin could be relatively large in this early phase. 8
It has been shown in animal models that administration of gabapentin before the injection of formalin is more effective and longer lasting than administration of gabapentin after the injection of formalin. 15 In a rat model of postoperative pain, pretreatment with a single dose of gabapentin blocked the development of hyperalgesia and tactile allodynia for up to 2 days, whereas giving gabapentin 1 h after the incision reduced symptoms for 3 h. 16 In addition, Yoon and Yaksh15,17 reported that intrathecal gabapentin attenuated pain behaviour when given prior to the injection of formalin into the rat hind paw, but did not attenuate pain behaviour when given after the formalin injection.
In most studies that have evaluated gabapentin as a postoperative analgesic, the drug was administered 1 – 2 h preoperatively as a single dose.4– 8,18– 20 The peak plasma level of gabapentin is achieved 2 – 3 h after ingestion; it does not undergo metabolism and is eliminated unchanged in the urine, with an elimination half-life of 5 – 9 h. 21 The absence of hepatic metabolism and the low level of protein binding of gabapentin contribute to its lack of clinically relevant drug interactions. 21 In the present study, gabapentin was given 2 h preoperatively so that the blood level would be optimum at the start of the tissue injury.
Although gabapentin has been given preoperatively in most of the published clinical studies, two reports compared the efficacy of pre- versus postincision administration of gabapentin. In these studies, similar doses of gabapentin provided the same postoperative analgesia whether administered pre- or postincision.22,23 Therefore, future work is needed to determine the optimal timing of gabapentin administration for postoperative pain control.
In studies evaluating a single dose of gabapentin, the doses administered varied between 300 mg and 1200 mg.4 – 7 The recommended starting dose of gabapentin for neuropathic pain is 300 mg on day 1, 300 mg twice daily on day 2, then 300 mg three times daily thereafter. 1 It is essential to use this dose titration when starting gabapentin therapy for neuropathic pain because of the potential for side-effects such as dizziness and drowsiness. In the present study, 900 mg gabapentin was given as a single dose because this is the lowest therapeutic daily dose for ongoing treatment for neuropathic pain. Administering a higher dose before anaesthesia and surgery may be associated with dizziness and drowsiness. In a study of the optimal dose of pre-/postincision gabapentin for pain relief following lumbar laminectomy, it was shown that in the first 12 h morphine consumption and pain scores were lower, and the time to the first demand for analgesia was longer, in groups receiving either 900 mg or 1200 mg gabapentin, either pre- or postincision, compared with placebo or 600 mg gabapentin. 22 Thus, 900 mg seems to be the lowest effective dose for gabapentin.
In some studies gabapentin was given as multiple (repeat) doses initiated the day before surgery. A multiple dosing regimen was not used in the present study because the central nervous system side-effects of gabapentin (i.e., sedation and dizziness) can be troublesome to patients in the postoperative period, especially during ambulation. Using higher doses of gabapentin for a prolonged period increased the incidence of such side-effects. 19 In another systematic review, multiple dosing with gabapentin given preoperatively and continued postoperatively did not appear to reduce VAS pain scores, compared with single dosing. 4
In the present study, there was no significant difference in tramadol consumption between the two groups. However, the need for rescue analgesia was significantly higher in the control group compared with the gabapentin group. Most of the rescue analgesia in both groups was given in the first 60 min postoperatively. Srivastava et al. 24 studied the effect of preoperative gabapentin on postoperative pain after minilap open cholecystectomy using a similar tramadol PCA regimen, and reported a significant decrease in tramadol consumption with gabapentin compared with placebo. Their patients received a bolus dose of 50 mg tramadol followed by 20 mg on demand, with a lockout interval of 20 min (maximum dose, 240 mg in 4 h). However, they allowed a 30-mg increase in tramadol at any time if analgesia was inadequate and they did not use rescue analgesia. The difference in effect on tramadol consumption between their study and the present study may be due to the different methods used for inadequate pain relief. The different surgical interventions may also have resulted in different levels of tramadol consumption, as RRP is more painful than minilap open cholecystectomy. Gabapentin may not show as great an opioid-sparing effect after very painful surgeries compared with those that are less painful.
Fewer patients reported nausea in the gabapentin group than in the control group in the present study, but this difference did not reach statistical significance. In the systematic review by Mathiesen et al., 8 analysis of side-effects showed a significantly lower incidence of nausea in favour of gabapentin for patients undergoing abdominal hysterectomy. They did not find any reports of clinically limiting side-effects (i.e., sedation and dizziness) with gabapentin. This is consistent with the results of the present study.
Gabapentin has been studied extensively in surgical populations, including gynaecology (hysterectomy), orthopaedics and neurosurgery (lumbar disc surgery). Nine meta-analyses evaluating these studies have demonstrated that gabapentin displays an effect on both postoperative pain scores and opioid usage in most of the studies.3,4 – 8,18 – 20 Tiippana et al. 3 reported that the opioid-sparing effect during the first 24 h after a single dose of 300 – 1200 mg gabapentin administered 1 – 2 h preoperatively ranged between 20 and 62%. Clivatti et al. 19 reported that pain scores were lower in 75% of patients who received a single preoperative dose of gabapentin, and in 55.6% of patients who received pre- and postoperative gabapentin. Opioid consumption was reduced in 82.4% of patients receiving a single preoperative dose of gabapentin, and in 77.8% of patients who received pre- and postoperative doses. 19 In another meta-analysis, gabapentin provided better postoperative analgesia and rescue analgesia sparing than placebo in six of the 10 randomized controlled trials that administered only pre-emptive analgesia. 20 However, a limitation of these systematic reviews is the wide variability in the gabapentin doses, dosing regimens, types of postoperative rescue analgesia and, most importantly, types of surgery. In these meta-analyses, data from studies with different surgical interventions are combined; therefore, the effect of gabapentin in a particular surgical intervention is difficult to predict. 4
The use of gabapentin as an analgesic has also been studied in more extensive surgeries. A single dose of gabapentin given pre- or postoperatively within a multimodal analgesia regimen did not reduce morphine consumption or pain scores in hospital after hip arthroplasty. 25 Preoperative use of gabapentin, followed by postoperative dosing for 2 days, did not significantly affect postoperative pain or opioid consumption in patients undergoing cardiac surgery. 26 In addition, preoperative gabapentin did not reduce pain scores or opioid consumption following elective thoracotomy, and did not confer any analgesic benefit in the setting of effective multimodal analgesia that included thoracic epidural infusion. 27 Minimally invasive surgery (such as laparoscopic cholecystectomy or arthroscopic surgery) does not cause such severe postoperative pain as abdominal surgery or other major procedures. As the analgesic effect of gabapentin appears to be related to the surgical procedure undertaken, a more procedure-specific evaluation is needed for different types of surgery. 28 Future studies should examine the analgesic efficacy of gabapentin in major or painful surgeries, to evaluate in more detail its opioid-sparing effect for the treatment of postoperative pain.
In conclusion, in the present study, preoperative administration of 900 mg gabapentin resulted in lower early postoperative pain scores and reduced the need for rescue analgesia in patients undergoing RRP, but it did not decrease overall tramadol consumption. Although gabapentin seems to be effective as a postoperative analgesic, further studies within different surgical subspecialties are necessary to find the optimum dose, timing and treatment duration, in order to include gabapentin definitively in postoperative pain treatment plans.
Footnotes
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
