Abstract
Objectives
This randomized, double-blind, placebo-controlled trial evaluated intraperitoneal (IP) lidocaine administration and intravenous (IV) lidocaine infusion for postoperative pain control after laparoscopic cholecystectomy (LC).
Methods
Patients who underwent LC were randomized to either group IV (intravenous lidocaine infusion), group IP (intraperitoneal lidocaine administration), or group C (control, IP and IV saline). Outcome measures were total postoperative pain severity (TPPS), total fentanyl consumption (TFC), frequency of administering patient-controlled analgesia (FPB), and a pain control satisfaction score (PCSS).
Results
Significantly reduced TPPS, TFC and FPB scores were observed in groups IP (
Conclusions
The IP administration of lidocaine and IV lidocaine infusion significantly reduced postoperative pain and opioid consumption in LC patients, compared with control infusions. For convenience, IV lidocaine could be used for pain reduction following LC; IP administration places additional burden on the surgeon.
Introduction
The first reported laparoscopic cholecystectomy (LC) was performed by a French surgeon, Phillipe Mouret, in 1987. 1 Since then, LC has been the recommended procedure for the management of symptomatic gallstones, 1 and LC has achieved outcomes superior to those of conventional open procedures in terms of recovery time, postoperative pain, cosmesis and morbidity.1–3 However, patients who have undergone LC still complain of postoperative pain, which can be superficial incisional wound pain, deep visceral pain and/or postlaparoscopy shoulder pain, all of which may require systemic opioid analgesia and result in delayed hospital discharge. 4
Various treatment modalities have been used to relieve pain following LC, including nonsteroidal anti-inflammatory drugs, opioids and local anaesthetics, but none has demonstrated consistent efficacy.5,6 Intraperitoneal (IP) administration of a local anaesthetic has been shown to reduce postoperative pain and analgesic consumption following laparoscopic surgery. 7 However, spraying lidocaine during a laparoscopic procedure to reduce postoperative pain places an additional burden on the surgeon, especially when the operation is complicated. 8 One alternative would be intravenous (IV) lidocaine, which has analgesic, 9 anti-inflammatory 10 and antihyperalgesic properties. 11 It has been reported to reduce postoperative pain6,12 and seems to be effective for pain management, especially after abdominal surgery. 12
The aim of the present study was to compare the analgesic efficacy of IP lidocaine administration with IV lidocaine infusion in LC patients.
Patients and methods
The study protocol was approved by the Ethics Committee of Chung-Ang University, Seoul, Republic of Korea, and registered at Clinical trial.gov (NCT01608373). The study was carried out according to the principles of the Declaration of Helsinki (2000), and following CONSORT guidelines. Written informed consent was obtained from each participant before their inclusion in the trial.
Study population
During the period between May 2011 and May 2012, consecutive patients fulfilling American Society of Anesthesiologists (ASA) physical status I and II, 13 undergoing elective LC at the College of Medicine, Chung-Ang University, Seoul, Republic of Korea, were recruited into this prospective, randomized, double-blind, placebo-controlled study. The exclusion criteria disqualified patients with any of the following: in receipt of analgesics or sedatives 24 h before scheduled surgery; spillage or cholelithiasis with known common bile duct pathology; body weight <45 kg or >100 kg; underlying severe systemic disease; history of abdominal surgery, a chronic pain disorder other than gallbladder disease or allergy to lidocaine. The decision to enrol or exclude patients was made by a single investigator (Y.H.J.) who did not otherwise participate in the study.
Study design and anaesthesia protocol
All patients were transferred to the operating room without premedication. Anaesthesia was induced with IV thiopental (5 mg/kg). Orotracheal intubation was facilitated with rocuronium (0.6 mg/kg). All patients were mechanically ventilated using a volume-controlled model ventilator (Aestiva/5, Datex-Ohmeda, Helsinki, Finland) and ventilation was adjusted to keep the end-tidal CO2 at 35–40 mmHg. Anaesthesia was maintained with oxygen (FIO2 = 0.5), air and sevoflurane (2–3 % vol). Arterial blood pressure, electrocardiography and pulse oximetry were monitored continuously. Lactated Ringer’s solution (3–6 ml/kg per h) was infused throughout surgery. No additional IV analgesics, including opioids, were injected. At the end of the operation, residual neuromuscular blockade was antagonized with pyridostigmine (0.2 mg/kg) and glycolpyrrolate (0.005 mg/kg).
Randomization of patients into one of the three study groups was performed using Excel® software (Microsoft, Redmond, WA, USA) random-number generation function. Details of the series, which were generated by a statistician who did not otherwise participate in this study (C.W.B.), were unknown to the investigators or patients. The numbers determining group assignment were written on cards within a set of sealed envelopes, and the appropriate numbered envelope was opened by an investigator prior to surgery. This investigator prepared syringes with 0.55 ml/kg of 1% lidocaine (or normal saline of the same volume) for IV injection, to be used by the anaesthesiologist, and 1.75 ml/kg of 0.2% lidocaine (3.5 mg/kg) or the same volume of normal saline, for IP administration by the surgeon. This ensured the surgeon and the anaesthesiologist were blind to the patient’s group.
Patients assigned to the IP administration group received IP injections of 3.5 mg/kg lidocaine and IV normal saline injection. Those in the IV group were given an IV bolus injection of 1.5 mg/kg lidocaine followed by a continuous IV infusion of 2 mg/kg/h lidocaine and IP normal saline. For the patients who were assigned to group C (the placebo control group), IV and IP saline were administered.
The IP administration of lidocaine solution (total dose, 3.5 mg/kg) or normal saline solution (the same volume as the lidocaine solution) was undertaken as follows: immediately after creation of the pneumoperitoneum, the surgeon sprayed one-quarter of the total solution on the upper surface of the liver under the right subdiaphragmatic space, and another quarter of the total solution under the left subdiaphragmatic space. In order to allow the sprayed solution to diffuse under the diaphragmatic space, the Trendelenberg position was maintained for ∼2 min. After the Trendelenberg position was relaxed, an additional half of the total solution was sprayed around the cholecystectomy site. This was performed using catheters inserted into the subumbilical area and the right upper quadrant trocars under direct laparoscopic control. These procedures were completed 10 min before beginning the surgery.
The IV group patients received an IV bolus injection of lidocaine (1.5 mg/kg) 2 min before orotracheal intubation. This was followed by a continuous IV lidocaine infusion at 2 mg/kg per h during the operation. Patients in groups C and IP received the same amount of normal saline IV as the volume of lidocaine given intravenously to those in the group IV.
To control the severity of postoperative pain, IV fentanyl (15 µg/kg, mixed with normal saline, total 100 ml) contained in a computerized IV patient-controlled analgesia (PCA) system (Automed 3300TM, ACE Medical, Seoul, Republic of Korea) was used. The mode of PCA was set to a bolus of 0.1 µg/kg, a lock-out interval of 15 min and a continuous infusion of 0.1 µg/kg per h. Patients were taught to push the button of the PCA system to receive a bolus of drug at the first onset of pain. A 10-point visual analogue scale (VAS) of pain severity was used to assess pain levels in patients, with 0 denoting the patient was pain free, and 10 denoting that the patient was in intolerable pain. If the VAS score was >3 despite the bolus, an additional 50 µg of fentanyl was administered IV until the pain was below a VAS score of 3.
One research nurse, who was blinded to the details of the study, collected the postoperative data.
Surgical technique
All the surgical procedures were performed by the same surgical team using the same technical principles and the 3-trocar technique. The patients were placed in the reverse Trendelenburg position (angled at ∼30°), with the table tilted downward to the patient’s left. The Veress needle (Tyco Healthcare, Dublin, Ireland) was introduced blind at the supra-umbilical site; this was followed by insertion of a 1-mm trocar. Pneumoperitoneum was created at 15 mmHg and maintained by insufflation with CO2 gas. The other two ports were inserted under direct laparoscopic visual guidance. The epigastric port was created at the right border of the falciform ligament using an 11-mm trocar, and the other (5-mm) trocar was placed in the right upper abdomen two finger breadths below the right costal margin, between the midclavicular and the midaxillary line. Clipping and transection were delayed until Calot’s triangle was exposed by electrocauterization and blunt dissection. The gallbladder was dissected from the liver bed using a Hook bovie (Covidien, Dublin, Ireland), and was extracted through the umbilical port site. At the end of surgery, CO2 was carefully evacuated by manual compression of the abdomen with open trocars.
Study assessments
For each patient, age, sex, ASA physical status, duration of anaesthesia (from injection of thiopental to extubation) and duration of operation (from skin incision to closure) were recorded.
The primary outcome measure of the study was total postoperative pain severity (TPPS, defined as superficial incisional pain plus deep visceral pain plus postlaparoscopy shoulder pain). TPPS was assessed at 2, 4, 8, 12, 24 and 48 h after surgery using the VAS, which was measured by the research nurse who was blind to the rest of the study. The frequency with which patients pushed the bolus button (FPB) of the PCA system and the total fentanyl consumption (TFC: the sum of the fentanyl delivered by the PCA system and additional IV fentanyl administration) were measured at similar time intervals (encompassing 0–2 h, 2–4 h, 4–8 h, 8–12 h, 12–24 h and 24–48 h) and compared between groups.
Satisfaction scores with regard to postoperative pain control (PCSS) were obtained using a numeric rating scale (NRS; 0, ‘very dissatisfied’, 10, ‘very satisfied’) on discharge. After surgery, patients were observed closely for fentanyl- and lidocaine-related side-effects.
The time to bowel sounds, starting a regular diet and duration of hospital stay were also noted. Solid food was provided as soon as the patients could tolerate it and when bowel function was deemed adequate. The decision to discharge a patient was made solely by the surgeon in charge (Y.S.C.) based on the following criteria: adequate pain relief at rest (VAS < 4 with oral analgesic administration only); oral intake without nausea and vomiting; adequate ambulation; no surgical complications. None of the other trial investigators was involved with this decision.
Statistical analyses
To estimate the sample size, a pilot study was conducted to measure the TPPS using the VAS 2 h after surgery in 10 patients who received normal saline (IP and IV). The SD of VAS scores in this group was 2.1. For the power calculations, equal SD were assumed in the IP and IV groups. In order to demonstrate a two-fold difference in the VAS 2 h after surgery among the groups, with a power of 80% and an α-value of 0.05, 22 patients were needed per group. Assuming a compliance rate of 80%, 83 patients were required for this study.
An intention-to-treat strategy was used for this study – that is, all participants were included in the analysis, irrespective of whether they had completed the study. Missing data were entered using a last-observation-carried forwards analysis. The association between NRS, FPB and fentanyl consumption was analysed using an as-treated strategy.
For intergroup comparisons, the distribution of the data was first evaluated for normality using the Shapiro–Wilk test. Normally distributed data were presented as mean ± SD, and groups were compared using an analysis of variance and a post hoc Tukey test. Non-normally distributed data were expressed as a median value (quartile 1–3), then analysed using the Kruskal–Wallis test with Bonferroni’s correction. Lidocaine consumption was compared between the IV and IP groups using Student’s
Descriptive variables were subjected to χ2 analysis or Fisher’s exact test, as appropriate;
Results
Among the 83 patients who were eligible for the study (age range, 18–65 years), five refused to participate and six were excluded as they had morbid obesity, cardiac, pulmonary and/or hepatic disease, or allergy to anaesthetics. Of the remaining 72 patients, 24 were randomized to group C, 22 to group IP and 26 to group IV. The flow diagram for patient allocation and follow-up is shown in Figure 1.There were no statistically significant differences among the groups in terms of age, sex, height, weight, BMI, ASA class, duration of anaesthesia or operation time (Table 1). There was no statistically significant difference in the quantity of lidocaine used between the IP and IV groups (Table 1).
CONSORT statement flow diagram for patients undergoing laparoscopic cholecystectomy, who received: intraperitoneal (IP) 3.5 mg/kg lidocaine on initiation of the pneumoperitoneum and intravenous (IV) normal saline injection (group IP); IV bolus injection of 1.5 mg/kg lidocaine before orotracheal intubation followed by continuous IV infusion of 2 mg/kg per h lidocaine and IP normal saline (group IV); or IV and IP saline (group C). Demographic and clinical characteristics of patients undergoing laparoscopic cholecystectomy, who received: intraperitoneal (IP) 3.5 mg/kg lidocaine on initiation of the pneumoperitoneum and intravenous (IV) normal saline injection (group IP); IV bolus injection of 1.5 mg/kg lidocaine before orotracheal intubation followed by continuous IV infusion of 2 mg/kg per h lidocaine and IP normal saline (group IV); or IV and IP saline (group C). Data presented as mean ± SD or median (quartile 1–3) except for ASA class and sex, which are expressed as There were no significant differences between the groups ( Data analysed using: aKruskal–Wallis test; banalysis of variance; cStudent’s BMI, body mass index; ASA, American Society of Anaesthesiologists physical status score.
13


During the postoperative follow-up, three patients in group C and one each in groups IP and IV were treated with other analgesics because of postoperative nausea and vomiting that was unresponsive to standard antiemetic treatment (IV ramosetron 0.3 mg once a day) and likely induced by fentanyl injection. One patient in group IV required meperidine due to postoperative shivering. None of the patients in the IP or IV groups experienced any lidocaine-related complications. No patient was withdrawn from the study.
The fentanyl-related side-effects noted were nausea, vomiting, pruritus, urinary retention and respiratory depression. The lidocaine-related side-effects noted were blurred vision, hearing problems, peripheral paraesthesia, dizziness, uncontrolled muscle contraction, convulsions, hypotension, bradycardia, headache and itching.
The TPPS findings are presented in Figure 2. Despite the administration of fentanyl through a PCA system, the VAS score in group C was >3 until 12 h after surgery. TPSS were significantly lower between 2 and 24 h in group IP compared with group C ( Total postoperative pain severity (TPPS) findings for patients undergoing laparoscopic cholecystectomy, who received: intraperitoneal (IP) 3.5 mg/kg lidocaine on initiation of the pneumoperitoneum and intravenous (IV) normal saline injection (group IP); IV bolus injection of 1.5 mg/kg lidocaine before orotracheal intubation followed by continuous IV infusion of 2 mg/kg per h lidocaine and IP normal saline (group IV); or IV and IP saline (group C). TPPS was defined as superficial incisional pain plus deep visceral pain plus postlaparoscopy shoulder pain, and was assessed using a 10-point visual analogue scale (VAS). Data presented as mean ± SD. a
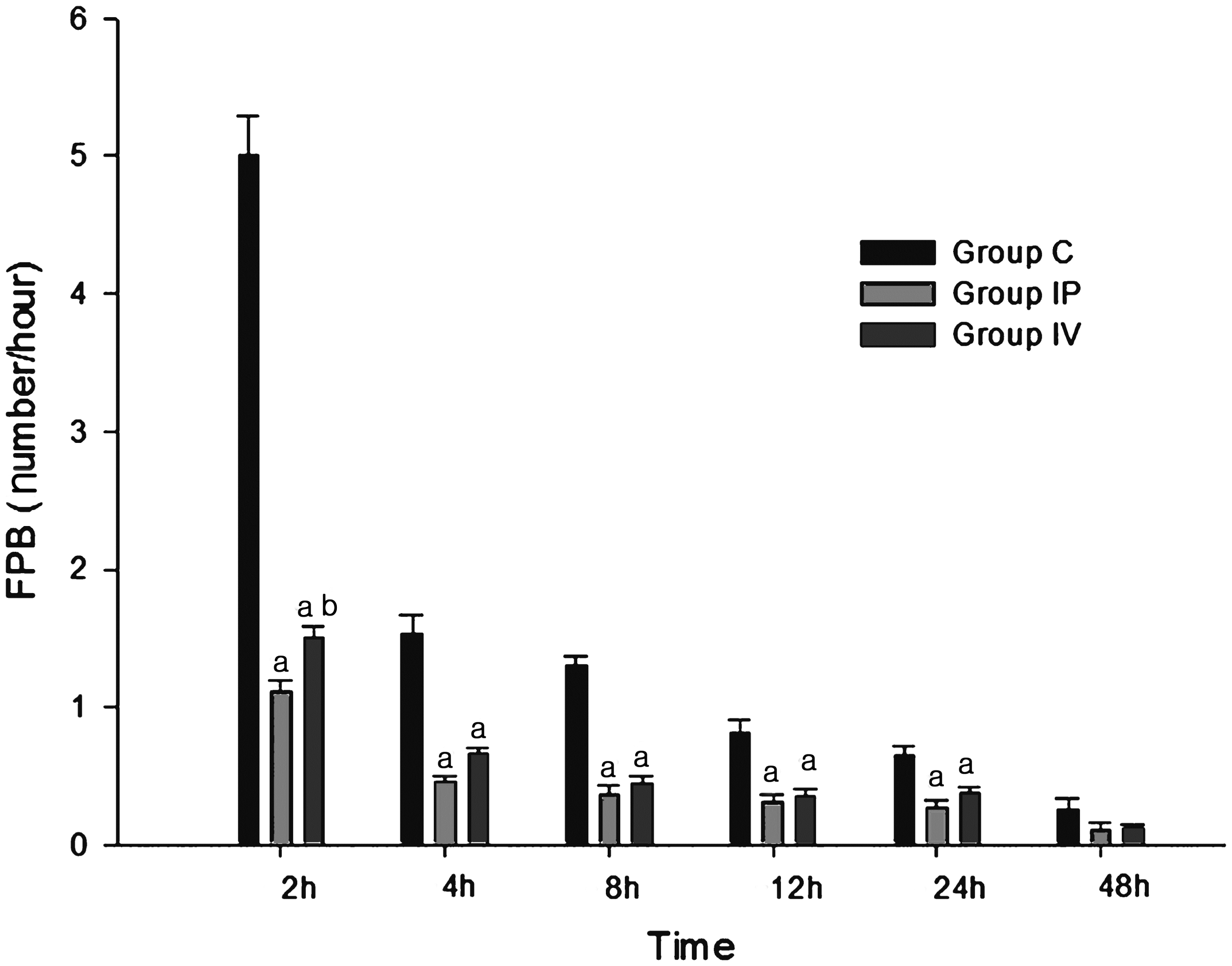
At each timepoint measured, TFC and FPB were significantly higher in group C than in groups IP and IV, until 24 h after surgery (Figures 3 and 4). The difference between the IP and IV groups for TFC was not significant (Figure 3). The FPB was significantly lower in group IP than group IV at the 0–2 h interval (Figure 4). There was a positive correlation between TPPS and TFC (ρ = 0.524, Total fentanyl consumption for patients undergoing laparoscopic cholecystectomy, who received: intraperitoneal (IP) 3.5 mg/kg lidocaine on initiation of the pneumoperitoneum and intravenous (IV) normal saline injection (group IP); IV bolus injection of 1.5 mg/kg lidocaine before orotracheal intubation followed by continuous IV infusion of 2 mg/kg per h lidocaine and IP normal saline (group IV); or IV and IP saline (group C). Data presented as mean ± SD. * Patient controlled anaesthesia (FPB; no. of button pushes per h) for patients undergoing laparoscopic cholecystectomy, who received: intraperitoneal (IP) 3.5 mg/kg lidocaine on initiation of the pneumoperitoneum and intravenous (IV) normal saline injection (group IP); IV bolus injection of 1.5 mg/kg lidocaine before orotracheal intubation followed by continuous IV infusion of 2 mg/kg per h lidocaine and IP normal saline (group IV); or IV and IP saline (group C). Data presented as mean ± SD. a
Incidence of side-effects and postoperative pain control satisfaction scores (PCSS) for patients undergoing laparoscopic cholecystectomy, who received: intraperitoneal (IP) 3.5 mg/kg lidocaine on initiation of the pneumoperitoneum and intravenous (IV) normal saline injection before orotracheal intubation (group IP); IV bolus injection of 1.5 mg/kg lidocaine before orotracheal intubation followed by continuous IV infusion of 2 mg/kg per h lidocaine and IP normal saline (group IV); or IV and IP saline (group C).

Data presented as
a
Time to various key outcome measures in patients undergoing laparoscopic cholecystectomy, who received: intraperitoneal (IP) 3.5 mg/kg lidocaine on initiation of the pneumoperitoneum and intravenous (IV) normal saline injection (group IP); IV bolus injection of 1.5 mg/kg lidocaine before orotracheal intubation followed by continuous IV infusion of 2 mg/kg per h lidocaine and IP normal saline (group IV); or IV and IP saline (group C).

Data presented as mean ± SD.
There were no significant differences between the groups (
Discussion
In this prospective, randomized, double-blind, placebo-controlled study, IP and IV lidocaine significantly reduced postoperative pain and fentanyl consumption compared with control treatment in LC patients. There was a positive correlation between pain and fentanyl consumption.
Laparoscopic cholecystectomy boasts many advantages compared with open cholecystectomy.1–3 However, the patient generally experiences moderate pain after LC, which can delay hospital discharge and may necessitate the use of opioid analgesics. 4 In the present study, despite the use of postoperative PCA and rescue analgesia, the mean VAS pain score in the control group was >3 at 12 h after surgery, which reflects notable postoperative pain.
Three types of pain are reported after LC surgery. 14 The first is parietal (somatic) pain, caused by the holes made in the abdominal wall for the trocars. 15 The second is visceral pain, caused by surgical handling and diaphragmatic irritation by dissolved CO2. 16 The third is shoulder tip pain, which is caused by rapid distension of the peritoneum (associated with the tearing of blood vessels, traumatic traction of the nerves and the release of inflammatory mediators) and excitation of the phrenic nerve. 17 Visceral pain is predominant during the first 24 h postoperatively, is short-lived, is unaffected by mobilization and is increased by coughing. Shoulder pain may occur in as many as 63% or as few as 35% of patients, 18 is associated with persistent pneumoperitoneum and can sometimes last for 3 days. 17
As pain after LC is multifactorial, postoperative pain control typically involves a multimodal approach. 6 Lidocaine has been administered intravenously since 1960. 19 The indications for IV lidocaine are regional blocks, antiarrhythmia, 20 analgesia for neuropathic and central pain 21 and postoperative pain. 12 Lidocaine IV suppresses neuronal excitability in dorsal horn neurons and depresses spike activity, amplitude and conduction time in both myelinated A-δ and unmyelinated C fibres. 22 This treatment also decreases the neural response to postoperative pain by blocking or inhibiting nerve conduction, 23 suppressing central sensitization, inhibiting spinal visceromotor neurons, 24 and reducing inflammation. 10 The patient therefore experiences less pain with, than without, IV lidocaine. 23
The mechanism of action for systemic analgesia is still poorly understood and is unlikely to be explained solely on the basis of the well-known sodium channel blockade effect of lidocaine. 9 Other potential factors are direct or indirect interaction with different receptors and nociceptive transmission pathways (e.g., muscarinic antagonists, glycine inhibitors, reduced production of excitatory amino acids, thromboxane A2, release of endogenous opioids, reduced neurokinin levels). 9
Despite an incomplete understanding of the mechanisms of action of systemic analgesia, the evidence supporting perioperative lidocaine infusions as part of a multimodal analgesic regimen for the management of postoperative pain is accumulating. Groudine
Research has validated the effect of a topical wash (liver and gallbladder surface wash).
27
Additionally, Roberts
Therefore, in the current study, LA was sprayed on the upper surface of the liver, under the right subdiaphragmatic space, under the left subdiaphragmatic space and around the cholecystectomy site. Another study concluded that the peritrocal infiltration of LA and the IP instillation of LA significantly lowered the intensity of postoperative pain in a synergistic fashion. 29 It should be noted that IP lidocaine administration, especially subperitoneal diaphragmatic injection, may result in complications (e.g., temporary loss of diaphragmatic function, pneumothorax or haematoma). 28
Both IV lidocaine infusion and IP lidocaine administration have been documented to be useful options for postoperative pain control after LC in several studies.4,25,29 However, in a patient with an intra-abdominal infection (such as panperitonitis or loculated peritoneal fluid collection), IP administration involves the risk of turning a localized infection into generalized peritonitis. IV lidocaine, which is a convenient technique to use and has a good safety profile, would be a preferable alternative to IP lidocaine for those with serious intra-abdominal inflammatory conditions.
In the current study, overall VAS pain scores, frequency of PCA button pushes and TFC were lower in groups IP and IV than in group C. Opioid consumption is not only a reflection of pain intensity, but is also profoundly influenced by various psychological factors including anxiety level, mood and expectation of recovery. In this regard, the frequency of PCA button pushes and fentanyl consumption could be a reflection of the overall level of satisfaction of patients, rather than a measure of pain severity alone. Measured satisfaction of patients in groups IP and IV was higher than in group C, as expected.
Similar results were reported in a study that compared IV lidocaine with IP lidocaine in patients who had undergone laparoscopic appendectomy. 8 However, in contrast to the previous report, in the current study, the TPPS values were lower in group IP than group IV at 2 h and FPB was lower in group IP than group IV at the 0–2 h interval. This may be due to differences in pain patterns among various types of laparoscopic procedures. The pain immediately following LC was somewhat intense and was characterized primarily as visceral pain. 14 Another potential explanation for these discrepancies may be the duration of lidocaine administration. In the current study, group IV received lidocaine until the end of surgery, while patients in group IP received lidocaine before surgery began. As the total quantities of lidocaine administered were similar in groups IV and IP, it appears that the amount of lidocaine administered for the purpose of achieving pre-emptive analgesia was relatively insufficient in group IV. 30 However, TPPS, FPB and TFC were lower in groups IP and IV than in group C. PCSS values were higher in groups IP and IV compared with group C. There was also a nonsignificant trend towards reduced times to bowel movement, initiation of a regular diet and discharge from hospital in group IV: effects which have been reported in other clinical studies examining the effect of IV lidocaine on postoperative pain.12,23,25 This suggests that IV lidocaine could be used as part of a multimodal approach to the treatment of pain after LC, particularly when IP lidocaine administration is not feasible, as in cases of panperitonitis or intra-abdominal infection.
One limitation of the current study was that the nature of postoperative pain and the VAS score that corresponded to each type of pain was not characterized. As the pain experienced post-LC is multifactorial, a more nuanced analysis could elucidate the mechanisms underlying IV versus IP lidocaine analgesia. In addition, although no lidocaine-related complications were noted in either group receiving the drug, this may have been due to the relatively small study population. A large-scale investigation might be required in order to determine the complication rates for each delivery method more definitively; such information could guide the choice between IP and IV administration. Serum levels of lidocaine were not measured, because the minimal toxicity from commonly studied lidocaine doses is well documented.21,31
In conclusion, both IV infusion and IP administration of lidocaine reduced postoperative pain in LC patients, compared with control (saline) infusions. Although IP is effective, IV lidocaine infusion is a more convenient method that is simpler to undertake and has a more favourable safety profile, compared with the IP method. We conclude that IV lidocaine is therefore a viable alternative to IP lidocaine administration when seeking to alleviate postoperative pain among patients undergoing LC.
Declaration of conflicting interest
The authors had no conflicts of interest to declare in relation to this article.
Footnotes
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012R1A1A1003700).
Acknowledgement
The authors would like to thank Song Hee Han, research nurse, for collecting the postoperative data from patients.
