Abstract
Objective:
Change in thoracic fluid content (TFC) derived via a bioreactance technique with a noninvasive cardiac output monitoring device (NICOM) reportedly shows a good correlation with the amount of fluid removed. The present study prospectively evaluated the utility and clinical application of TFC in the intraoperative fluid management of paediatric patients with congenital heart disease, undergoing cardiac surgery with bioreactance-based noninvasive monitoring.
Methods:
Haemodynamic parameters, patient body weight and parameters derived from the NICOM device (including cardiac output, cardiac index, TFC, percentage change in TFC compared with baseline [TFCd0%] and stroke volume variation) were recorded after anaesthesia induction but before surgical incision, and just before departure from the operating room to the intensive care unit.
Results:
In the 80 paediatric patients included in this study, linear regression analyses demonstrated good correlations between body weight gain and TFCd0%, between body weight gain % and TFCd0%, and between intra -operative fluid balance and TFCd0%.
Conclusion:
TFCd0% may be a useful indicator for intraoperative fluid management in paediatric patients with congenital heart disease, undergoing cardiac surgery.
Keywords
Introduction
Fluid management during cardiac surgery is a critical factor in stabilizing haemodynamic status that can have an important effect on patient outcomes. Excessive fluid administration can lead to a risk of volume overload and pulmonary oedema and/or ventricular dysfunction, and an increase in cardiac work; inadequate fluid administration can induce unstable haemodynamic status, and organ hypoperfusion and dysfunction.1 –3 In paediatric cardiac surgery, fluid management is even more important because of the large size of the fluid shifts compared with the small body surface area and body weight of the patient. Inflammatory reactions induced by cardiopulmonary bypass (CPB) can aggravate the patient's unstable haemodynamic state.4–6
A widely used method to evaluate patient volume status in adult cardiac surgery is pulmonary artery catheterization with measurement of central venous pressure (CVP) and pulmonary capillary wedge pressure. Insertion of a catheter into the pulmonary artery in paediatric patients is, however, a complex procedure due to the patient's small size or aberrant anatomy, and it carries a greater risk of complications than the equivalent procedure in adults.7,8
Although CVP has been widely used in paediatric cardiac surgery to estimate intravascular volume status, its use is inappropriate as a marker of exact volume status. This is because many factors (such as venous capacitance, cardiac chamber compliance, valve competence and pulmonary arterial pressure) can affect the accuracy of the pressure measurements.9 – 11 Noninvasive or less invasive methods, such as bioreactance techniques (using a noninvasive cardiac output monitoring [NICOM] device) and femoral artery catheterization with a thermodilution technique (the pulse-induced continuous cardiac output system), which can all be used in paediatric patients, have been introduced.12 – 14
Noninvasive cardiac output monitoring devices have been used in several clinical fields. It has been demonstrated that there is a good correlation between changes in thoracic fluid content (TFC) derived via a bioreactance technique with a NICOM device and the amount of fluid removal, and this technique was shown to be appropriate in patients undergoing haemodialysis. 15 The utility of TFC in paediatric cardiac surgery, however, has yet to be established.
The aim of the present study was to evaluate the utility and clinical application of TFC in intraoperative fluid management of paediatric patients with congenital heart disease undergoing cardiac surgery with bioreactance-based noninvasive monitoring. Correlations between the values of TFC, changes in TFC, intraoperative fluid balance, and changes in body weight were evaluated.
Patients and methods
Study Population
Paediatric patients undergoing cardiac surgery due to congenital heart disease, between November 2011 and May 2012, were studied prospectively at the Department of Anaesthesiology and Pain Medicine, Konkuk University Hospital, Konkuk University Medical Centre, Konkuk University School of Medicine, Seoul, Republic of Korea. Exclusion criteria were: urgent or emergency cases; patients with dermatological disease that interfered with attachment of the NICOM patch; patients with a body weight of > 20 kg; cases of thoracotomy to correct congenital cardiac anomaly. Cardiac surgery procedures were performed by a single paediatric cardiac surgery team (H.J.S. and D.M.S.); CPB was performed by a single perfusionist on the cardiac surgery team. Nurses and perfusionists were blinded to the study design, and cardiac surgeons were blinded to the NICOM device-derived parameters, to ensure that surgical procedures were conducted according to standard protocols.
Ethical approval (No. KUH1160033; 3 November 2011) for the study design was obtained from the Institutional Review Board of Konkuk University Medical Centre. Written informed consent was obtained from the parents of participating patients.
Bioreactance-Based Noninvasive Monitoring
After anaesthesia induction was achieved according to the standard regimen of the Department of Anaesthesiology and Pain Medicine, Konkuk University Hospital, the NICOM electrode strips were placed on the patient's chest and connected to the NICOM controller (The Cheetah NICOM®; Cheetah Medical, Vancouver, WA, USA). Each electrode sensor strip consisted of two contact points. The upper thoracic electrode strips were placed at the midsubclavian region; the lower electrode strips were placed in the middle region of the lower costal margin. After initial calibration of the NICOM system, continuous cardiac output (CO), cardiac index (CI), stroke volume (SV) and stroke volume variation (SVV) were monitored.
The NICOM system's signal processing unit determines the relative phase shift (Φ) between input and output signals, which reflects changes in blood volume in the aorta. SV determined by the NICOM system can be estimated using the equation: SV = C × VET × dφ/dtmax, where C is a constant of proportionality, VET is ventricular ejection time, and dφ/dtmax is the peak rate of change of Φ. 16 The value of C has been optimized in prior studies, accounting for patient age, gender and body size. 17 Maximal and minimal values of beat-to-beat SV were determined over a single respiratory cycle. SVV was calculated as SVV (%) = (SVVmax – SVVmin)/(SVVmax + SVVmin/2) × 100.
The water content of the body, whether in the blood (intravascular) or outside the blood (extravascular), contains high concentrations of various electrolytes such as sodium, chloride, potassium and calcium. These electrolytes are good conductors of electricity. The more fluid a patient has in his/her chest, the more electrolytes are available for electrical conductance, and vice versa. TFC is calculated as TFC = 1/Z0, where Z0 is basic thoracic impedance in Ω, moving in the opposite direction to the water content. TFC provides a relative measure of changes in thoracic fluid. Specifically, higher TFC values compared with baseline indicate that the water content of the patient's chest is increased, and vice versa. TFCd0% is the percentage change in TFC compared with baseline. Parameters measured by the NICOM device were mean values during 1 min, with a 1-min interval between measurements.
Determination of Patient Body Weight
After anaesthesia induction with routine invasive arterial blood pressure and noninvasive patient monitoring (pulse oximetry, electrocardiography), a central venous catheter (inserted under sonographic guidance) and a urinary catheter were inserted into each patient. The electrode strips of the NICOM were attached. Patient body weight was measured with portable scales (SW-30H; CAS Corporation, Yangju-si, Republic of Korea) while the monitoring equipment was attached.
Measurement of Haemodynamic Parameters, Weight Gain and TFC
Patient body weight (kg), haemodynamic parameters including mean arterial blood pressure (MAP, mmHg), heart rate (HR, beats/min) and CVP (mmHg), and parameters derived from the NICOM device, including CO (l/min), CI (l/min/m2), TFC (Ω–1), TFCd0% (%) and SVV (%) were recorded at two points: after anaesthesia induction but before surgical incision (T0) and immediately before departure from the operating room to the intensive care unit (T1). Pressure transducers (PX600F; Edwards Lifesciences, Irvine, CA, USA) for MAP and CVP were placed on the midaxillary line with guidance from a laser leveller (PhysioTrac™; Edwards Lifesciences) and fixed to the operating table, to keep the transducer at atrial level during the entire protocol. The body weight gain of patients and the difference in TFC (ΔTFC) were calculated as the difference between the values at T1 and T0. The percentage body weight gain was calculated as follows: body weight gain % = 100 × (body weight at T1 – body weight at T0)/body weight at T0. The intraoperative fluid balance (input/output) was estimated by calculation of intraoperative fluid and blood product administration, urinary output and the fluid balance of the CPB.
Statistical Analyses
All statistical analyses were performed using the SigmaStat software package, version 3.1 (Systat® Software, Chicago, IL, USA). A sample size of 80 patients was considered suitable for the present study, based on the minimal and maximal patient numbers enrolled in previous analyses.15,16 Continuous variables were analysed using the paired t-test or Wilcoxon signed–rank test to compare values between the two measurement points. A linear regression analysis was used to determine the possible correlation between variables. An additional regression analysis, excluding patients who underwent a Fontan procedure or bidirectional cavopulmonary shunt (BCPS), was also performed because measures of CVP can be inaccurate in these patients. Data were expressed as mean ± SD or median (25th – 75th percentile) and numbers of patients. A P-value < 0.05 was considered to be statistically significant.
Results
During the study, a total of 95 paediatric patients underwent congenital cardiac surgery. Fifteen of these patients were excluded: six because the NICOM device was not used, five due to parental refusal of consent, and four due to instrument error. A total of 80 patients were studied and included in the final analysis. The study ended when the number of patients met the predetermined sample size (i.e., 80 patients), and no further evaluation or follow-up was performed after the end of surgery. No unexpected events as a result of the procedure occurred during the study and no patients experienced an adverse event.
The baseline demographic characteristics and preoperative diagnoses are summarized in Table 1. Fontan procedures or BCPS were carried out in 12 patients. There were no significant differences in MAP and HR between T0 and T1 (Table 2). CVP was significantly higher and CO and CI were significantly lower at T1 than at T0 (P < 0.001 for all comparisons). Patient body weight and TFC values were significantly greater at T1 than at T0 (P < 0.001 for both comparisons). The values of SVV did not differ significantly between T0 and T1. In an analysis excluding the 12 patients in whom the Fontan procedure or BCPS was performed, CVP was significantly higher at T1 than at T0 (median [25th – 75th percentile], 6.0 [5.0 – 7.0] mmHg at T0 versus 8.0 [6.5 – 10.0] mmHg at T1; P < 0.001). Perioperative parameters for the included patients are shown in Table 3.
Baseline demographic characteristics and preoperative diagnoses for paediatric patients who underwent cardiac surgery for congenital heart disease (n = 80)

Comparisons of haemodynamic parameters and body weight between T0 and T1 in paediatric patients who underwent cardiac surgery for congenital heart disease (n = 80)

Data presented as mean ± SD or median (25th – 75th percentile).
Statistical analyses undertaken using the paired t-test or Wilcoxon signed–rank test to compare values between T0 and T1.
T0, after anaesthesia induction but before surgical incision; T1, just before departure from the operating room to the intensive care unit; MAP, mean arterial blood pressure; HR, heart rate; CVP, central venous pressure; CO, cardiac output; CI, cardiac index; TFC, thoracic fluid content; SVV, stroke volume variation; NS, not statistically significant (P ≥ 0.05).
Perioperative parameters for paediatric patients who underwent cardiac surgery for congenital heart disease (n = 80)

ΔTFC, change of thoracic fluid content compared with baseline value; TFCd0%, percentage of change compared with TFC at baseline; SVV, stroke volume variation.
Linear regression analyses revealed good correlations between body weight gain and TFCd0%, between body weight gain % and TFCd0%, and between intraoperative fluid balance and TFCd0% (P < 0.001 for all comparisons, Table 4). In addition, good correlations were determined between body weight gain or body weight gain % and intraoperative fluid balance (P < 0.001). Nonsignificant correlations were observed between body weight gain and change in CVP and between intraoperative fluid balance and change in CVP (Table 4). The linear regression analyses also showed nonsignificant correlations between body weight gain, body weight gain % or intraoperative fluid balance and SVV at T1.
Linear regression analyses comparing body weight gain, body weight gain % and intraoperative fluid balance with other haemodynamic parameters in paediatric patients who underwent cardiac surgery for congenital heart disease (n = 80)
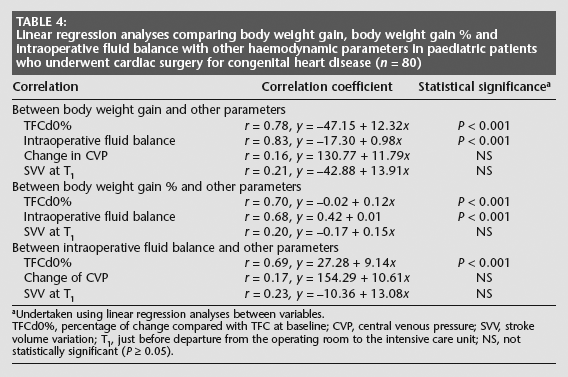
Undertaken using linear regression analyses between variables.
TFCd0%, percentage of change compared with TFC at baseline; CVP, central venous pressure; SVV, stroke volume variation; T1, just before departure from the operating room to the intensive care unit; NS, not statistically significant (P ≥ 0.05).
In additional linear regression analyses that excluded patients who underwent a Fontan procedure or BCPS, good correlations were determined between body weight gain or body weight gain % with TFCd0%, and between intraoperative fluid balance and TFCd0% (P < 0.001 for all comparisons, Table 5). Correlations between body weight gain and change in CVP, and between intraoperative fluid balance and change in CVP, remained nonsignificant.
Linear regression analyses comparing body weight gain, body weight gain % and intraoperative fluid balance with other haemodynamic parameters in paediatric patient who underwent cardiac surgery for congenital heart disease excluding those patients who underwent a Fontan procedure or bidirectional cavopulmonary shunt (n = 68)

Undertaken using linear regression analyses between variables.
TFCd0%, percentage of change compared with TFC at baseline; CVP, central venous pressure; NS, not statistically significant (P ≥ 0.05).
Discussion
Results from the present study demonstrated good correlations between body weight gain and TFCd0%, between body weight gain % and TFCd0%, and between intraoperative fluid balance and TFCd0%. These findings suggest that TFCd0% is an appropriate parameter to measure intraoperative fluid management in paediatric patients undergoing cardiac surgery.
In the resuscitation of haemodynamically unstable patients, the first step is the accurate assessment of the patient's volume status. Several studies have found that cardiac filling pressures such as CVP, which have traditionally been used to guide fluid management, are inappropriate for evaluating a patient's actual volume status.18 – 22 This problem is associated with the changes in venous tone, intrathoracic pressure, left ventricle (LV) and right ventricle (RV) compliance, and geometry that occur in critically ill patients.1,23 In paediatric patients undergoing cardiac surgery due to congenital heart disease, CVP is also inappropriate for estimating a patient's exact volume status. 11 This may be related to the intracardiac shunt (left to right or right to left), altered pulmonary vascular resistance, altered LV and RV compliance and complex anatomy encountered in congenital heart disease. In 16 paediatric patients undergoing cardiac surgery, Mahajan et al. 24 reported that CVP was not indicative of a patient's actual volume status, even after the congenital cardiac anomaly was corrected. This may be associated with a suboptimal cardiac state such as cardiac compliance, pulmonary vascular resistance and valvular regurgitation. Data from the present study corresponded well with other published reports suggesting that CVP is not an adequate indicator of volume status after cardiac surgery in paediatric patients.11,24
In the present study, 12 patients who underwent a Fontan procedure or BCPS due to a functional single ventricle were included in the regression analyses. The values of CVP in these patients did not accurately reflect atrial pressures, but rather reflected pulmonary arterial pressures. Pulmonary vascular resistance might affect the measured values of CVP, such that CVP measurements in these patients may have differed from those in the other patients. Correlations between body weight gain or intraoperative fluid balance and change in CVP in the present study may therefore be inaccurate, so the data were analysed again after excluding these 12 patients. The correlations of body weight gain or body weight gain % with TFCd0% decreased slightly following exclusion of this patient subgroup, but still demonstrated a good relationship. Similarly, correlations between body weight gain or intraoperative fluid balance with change in CVP were slightly improved but still remained nonsignificant. These findings have several implications. First, even if patients undergoing a Fontan procedure or BCPS were excluded, CVP was still an inappropriate indicator of intraoperative fluid management, as reflected by low correlations. Secondly, the higher value of the correlation between body weight gain or body weight gain % with TFCd0% when the 12 patients were included, compared with the correlation after excluding them, indicated that TFCd0% could be a useful parameter for assessing volume status during paediatric cardiac surgery, regardless of cardiac anatomical structure. Additional larger prospective studies are needed to confirm the utility of TFC and TFCd0% in patients undergoing a Fontan procedure or BCPS.
The absolute values of CO and CI at each measured point in this study were lower than the normal ranges. Although no investigation to evaluate the accuracy and validity of NICOM-derived CO and CI was performed in the current study, it has already been reported that measurements of these parameters by bioreactance techniques tend to be underestimated.12,25
When parameters were compared between T0 and T1 in the present study, CO and CI were significantly lower and CVP was significantly higher at T1 than at T0. SVV was slightly higher at T1 than at T0, but the difference was not statistically significant. In the analyses excluding the 12 patients who underwent the Fontan procedure or BCPS, CVP was still significantly higher at T1 than at T0. Regarding overall haemodynamic parameters, if decreased CO and CI and increased SVV indicate the condition of low intravascular volume at T1, it is clear that CVP does not accurately reflect the patient's actual volume status.
The correlations of body weight gain or body weight gain % with intraoperative fluid balance determined in the present study were comparable with those of body weight gain or body weight gain % with TFCd0%. These results indicate that intraoperative fluid balance could be a reliable indicator for intraoperative fluid management. Considering that intraoperative fluid balance is the major factor affecting changes in body weight, these results are not surprising. It can, however, be difficult to estimate fluid balance accurately during the postoperative phase, especially after the CPB weaning period, due to bleeding in the surgical field, surgical bleeding absorbed by gauzes and insensible loss of body fluid. An accurate process for estimating the intraoperative fluid balance would be time consuming. Furthermore, paediatric patients have a lower cardiac reservoir than adult patients, resulting in greater haemodynamic variation due to surgical bleeding or surgical manipulation in the operating room. In this regard, continuous monitoring of fluid management would be necessary, but to achieve this by calculation of fluid balance might be difficult, as would be its practical clinical application. Theoretically, TFCd0% reflects changes in intra- and extravascular volume status, and cannot distinguish intravascular from extravascular volume change. If excessive fluid is administered and tissue oedema occurs in the thorax, TFCd0% can increase without any increase in intravascular volume. TFCd0% and simultaneous use of the parameters induced by respiratory variation (such as SVV, which might reflect the fluid responsiveness) would be more useful in intraoperative fluid management.
In conclusion, TFCd0% derived by a bioreactance technique correlates well with body weight gain, body weight gain % and intraoperative fluid balance. These data suggest that TFCd0% is a useful indicator for intraoperative fluid management in paediatric patients undergoing cardiac surgery due to congenital heart disease.
Footnotes
Acknowledgement
This research was supported by a 2013 research grant from Konkuk University, Seoul, Republic of Korea.
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
