Abstract
Objective:
To evaluate the relationship between serum total homocysteine levels and retinopathy of prematurity (ROP).
Methods:
This prospective case-control study involved premature infants diagnosed with ROP 4 weeks after birth (cases); controls were premature infants not developing ROP during follow-up. Fasting serum total homocysteine concentrations were determined in all participants 4 weeks after birth, using high performance liquid chromatography.
Results:
A total of 45 and 35 infants were included in the case and control groups, respectively. The mean ± SD (range) serum total homocysteine levels were 10.36 ± 1.72 μmol/l (7.45 - 14.84) in infants with ROP and 8.41 ± 2.12 μmol/l (5.56 - 13.90) in controls. This difference was statistically significant. Mean ± SD total homocysteine levels were higher in infants with more severe ROP (11.45 ± 1.76 μmol/l) compared with mild ROP (9.92 ± 1.56 μmol/l).
Conclusions:
Elevated serum total homocysteine levels are associated with the development of ROP in premature infants. Further studies with larger patient populations are required, to improve understanding of the relationship between homocysteine and ROP development.
Introduction
Retinopathy of prematurity (ROP) is a major cause of blindness in children, particularly in those born in less developed countries.1,2 Although ablation of the retina with indirect diode laser photocoagulation and/or cryotherapy reduces the incidence of blindness,3,4 ROP continues to be an important problem in both the developing and the developed world.5–7 Current ROP therapies are limited by long-term adverse events, despite successful treatment. 8 There is an unmet medical need for preventive therapy for ROP, which can only be developed if there is a better understanding of the pathophysiology of this disease. 9
High levels of postnatal oxygenation, prolonged mechanical ventilation, multiple transfusions, multiple gestation, low birth weight and gestational age at birth have all been shown to increase the risk of ROP development. 9 In addition, major roles for vascular endothelial growth factor (VEGF) and insulin-like growth factor-1 in the development of ROP have been demonstrated.10,11 Factors that affect vascular development in premature infants continue to be actively researched.
Homocysteine is a sulphur-containing amino acid formed by the hydrolysis of S-adenosyl-homocysteine in the methionine metabolic cycle. 12 Hyperhomocysteinaemia, which results from impaired methionine metabolism, is a major independent risk factor for vascular diseases in adults, 13 and numerous studies have investigated the relationship between this condition and eye diseases. It has been demonstrated that hyperhomocysteinaemia is associated with central retinal vein occlusion, glaucoma and age-related macular degeneration in adults.14–16 Plasma homocysteine levels are also elevated in vitreoretinal diseases such as proliferative diabetic retinopathy. 17 In newborn infants and children, hyperhomocysteinaemia was found to be a risk factor for stroke,18,19 although no association between total homocysteine levels and intraventricular haemorrhage was reported in preterm infants. 20
There are no published data describing the relationship between homocysteine levels and ROP, which is a condition associated with neovascularization. The present study assessed serum total homocysteine levels in premature infants with or without ROP 4 weeks after birth, and analysed the possible relationship between total homocysteine levels and ROP.
Patients and methods
Study Population
This prospective case–control study included consecutive premature infants with a gestational age ≤ 34 weeks, diagnosed with ROP of varying degrees after routine ROP screening at 4 weeks after birth. Patients were screened at the Department of Ophthalmology, Cerrahpasa Medical Faculty, Istanbul University, Istanbul, Turkey, between September 2011 and January 2012. ROP diagnosis and staging were carried out using a standard indirect ophthalmoscope after application of 1% tropicamide and 2.5% phenylephrine eye drops. Control subjects were premature infants, gestational age ≤ 34 weeks, undergoing routine ROP screening 4 weeks after birth within the same department, who did not develop ROP. Infants were excluded if they had impaired renal function or had received anticonvulsant therapy that could interfere with the measurement of total homo cysteine levels.21,22
The Ethics Committee of the Cerrahpasa Medical Faculty approved the study protocol (No. 1802). Written informed consent was obtained from the parents of all participants, after a detailed explanation of the purpose and methods of the study was given. The study adhered to the Declaration of Helsinki for human participants.
ROP Classification
Retinopathy of prematurity was classified according to the International Classification 23 and subdivided into the following stages: Stage 1, demarcation line; Stage 2, ridge; Stage 3, ridge with extraretinal fibrovascular proliferations; Stage 4, subtotal retinal detachment; Stage 5, total retinal detachment. Each infant was classified according to the most advanced ROP stage observed. Infants with ROP were further grouped into mild (Stage 1 and 2) and severe (Stages 3 – 5) categories.
Serum Total Homocysteine Measurement
To eliminate the effects of diurnal variation, fasting blood samples were drawn early in the morning 4 weeks after birth by venipuncture, or obtained using the heel-stick procedure. Blood samples were immediately placed on ice, centrifuged at 3000
Statistical Analyses
All statistical analyses were performed using SPSS® statistical software, version 16.0 (SPSS Inc. Chicago, IL, USA) for Windows®. Data were presented as mean ± SD. Student's t-test, Pearson's correlation coefficient and analysis of covariance (ANCOVA) were used for data analysis. A P-value of < 0.05 was considered to be statistically significant.
Results
This study involved 45 premature infants with ROP (cases) and 35 without ROP (controls). There were no significant between-group differences with respect to gender, mean birth weight and mean gestational age (Table 1). Among the 45 infants in the case group, 24 (53%) had Stage 1 ROP, 8 (18%) had Stage 2 ROP, six (13%) had Stage 3 ROP, four (9%) had Stage 4 ROP and three (7%) had Stage 5 ROP.
Characteristics of premature infants diagnosed with retinopathy of prematurity (ROP, cases) and infants who did not develop ROP (controls), at routine ROP screening 4 weeks after birth
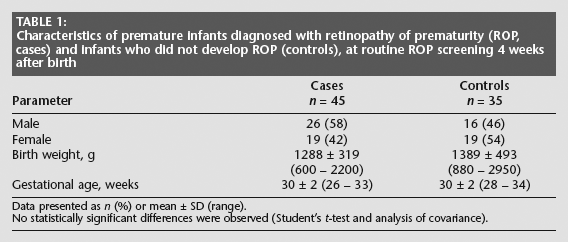
Data presented as n (%) or mean ± SD (range).
No statistically significant differences were observed (Student's t-test and analysis of covariance).
The overall mean ± SD serum total homocysteine level in all participants was 9.50 ± 2.13 μmol/l (range 5.56 – 14.84). Infants with ROP had significantly higher total homocysteine levels compared with control infants (mean ± SD 10.36 ± 1.72 μmol/l [range 7.45 – 14.84] versus 8.41 ± 2.12 μmol/l [range 5.56 – 13.90]; P < 0.001 [95% CI 1.10, 2.81]).
Total homocysteine levels were significantly higher in infants with more severe ROP, Stages 3 – 5 (mean ± SD 11.45 ± 1.76 μmol/l [range 8.42 – 14.84]) compared with mild ROP, Stages 1 and 2 (mean ± SD 9.92 ± 1.56 μmol/l [range 7.45 – 13.35]; P = 0.0054 [95% CI –2.59, –0.48).
Evaluation of all study participants revealed no significant correlation between birth weight and total homocysteine level. A weak positive correlation between gestational age and total homocysteine level was observed (r = 0.20; P = 0.028). Mean adjusted total homocysteine levels, as determined by ANCOVA controlling for gestational age and birth weight, were higher in cases compared with controls (F [1, 77] = 22.150, [P = 0.001] and F [1, 77] = 23.845 [P = 0.001], respectively).
Discussion
The present study demonstrated a statistically significant relationship between elevated serum total homocysteine levels and ROP. This observation is consistent with published data that indicate a higher risk for vascular disease in adults with hyperhomocysteinaemia.13,17
Hyperhomocysteinaemia is an independent risk factor for vascular disease. The underlying mechanisms leading to vascular dysfunction in individuals with this condition are unclear, although endothelial injury and/or dysfunction have been proposed as major causes.24,25 Homocysteine has been shown to decrease nitric oxide levels in vitro by a glutathione peroxidase mechanism. 26 This study also found that homocysteine generated hydrogen peroxide and reduced hydrogen peroxide detoxification, and indicated a role for homocysteine in inducing endothelial cytotoxicity though the production of other reactive oxygen species. 26 In addition, it has been reported that homocysteine-induced endoplasmic reticulum stress and growth arrest in human vascular endothelial cells involve changes in gene expression specific for these effects; 27 the decrease in antioxidant enzymes suggests that homocysteine could indirectly enhance the effects of agents or conditions known to cause oxidative stress.
The role of homocysteine in retinal diseases that have a vascular component has also been extensively studied in adults. In vitro, hyperhomocysteinaemia has been shown to contribute to the development of retinal neovascularization through stimulation of VEGF in retinal cells. 28 Plasma homocysteine could be a useful biomarker and/or a risk factor for increased risk of diabetic retinopathy in patients with type 2 diabetes: 29 elevated vitreous and plasma levels of homocysteine have been reported in those with proliferative diabetic retinopathy.17,30 It has been suggested that the role of homocysteine in diabetic retinopathy might be explained by a stimulatory effect on VEGF upregulation. 17 Taking all these findings together with observations from the present study suggests that homocysteine may also be a useful biomarker for ROP development. In the case of ROP, high oxygen concentrations may cause the production of reactive oxygen species that, under conditions of hyperhomocysteinaemia, may cause vascular damage. Hyperhomocysteinaemia in ROP could also cause growth arrest in vascular endothelial cells by inducing endoplasmic reticulum stress and delaying vascularization originating centrifugally from the optic disc. Furthermore, increases in VEGF that occur in hyperhomocysteinaemia may have an important role in ROP. The development of ROP is biphasic: 9 the first (ischaemic) phase begins after premature birth, during which higher oxygen concentrations suppress VEGF, causing delayed retinal vascular development and degeneration of existing blood vessels (vaso-obliteration); in the second (vasoproliferative) phase, the hypoxic retina causes a compensatory release of VEGF, leading to neovascularization and abnormal vessel development, which is responsible for many of the sight-threatening complications.
There are no definitive published data on normal levels of serum total homocysteine in premature infants. As part of a routine screening programme in newborns, the median (5th – 95th percentile) serum total homocysteine concentration in 4992 capillary blood samples was found to be 6.8 μmol/l (4.2 – 12.8). 31 A study of 28 preterm infants (< 34 weeks' gestation) reported that serum total homocysteine levels in infants between 1 and 4 weeks of age ranged from 0.8 to 12.2 μmol/l: levels were lower with greater prematurity but no further age-related changes were observed after birth. 32 Characteristics of this study population, 32 such as mean gestational age (29 weeks [range 24 – 33 weeks]) and mean birth weight (1372 g [range 685 – 2230 g]), were similar to those recorded in the present study. Overall, mean serum total homocysteine levels were higher in the present study compared with the previous report (9.50 μmol/l versus 6.80 μmol/l). 32 These differences may be reflective of the different populations studied (Turkish versus British) or the different sampling and measurement methods used. Although the previous report did not analyse serum vitamin B6 or B12 status, higher levels of serum total homocysteine were observed in those who had received preterm formula milk compared with those who had not. 32 Increases in total homocysteine with postnatal age have been suggested to be due to parenteral amino acid infusions. 33 There was no evidence of a correlation between birth weight and serum total homocysteine levels in the present study. A weak positive relationship between gestational age and homocysteine levels was observed, however, although there were no significant differences between the case and control groups. Analysis of the contributing effects of birth weight and gestational age indicated that adjusted mean serum total homocysteine levels were higher in the case group compared with the controls. Furthermore, statistical analyses within the ROP categories demonstrated that infants with more severe ROP had higher homocysteine levels than those with mild ROP.
Data from 5883 women demonstrated a strong association between elevated serum total homocysteine and adverse pregnancy outcomes (such as prematurity, low birth weight and growth retardation). 34 Two other studies also reported correlations between maternal and neonatal total homocysteine levels.35,36 Although maternal serum total homocysteine levels were not analysed in the present study, taking the present results together with previously published data suggests that elevated homocysteine concentrations in premature infants could result from elevated maternal homocysteine levels.
The mean birth weight and gestational age of infants in the present study were both higher than those reported in other studies undertaken in developed countries. One Turkish study reported that all infants with a gestational age < 34 weeks or birth weight of < 2000 g were at risk for ROP. 37 The present study included premature infants of a higher body weight [range 600 – 2950 g] and greater gestational age [range 26 – 34 weeks], since a relatively high incidence of ROP – particularly in its severe form – has been reported in more mature infants in developing countries. 37 One limitation of the present study was that other variables (such as parenteral nutrition, feeding behaviour, serum folate or vitamin B12 levels) were not recorded. In addition, the findings are only a snapshot of total homocysteine levels in a small study population at one particular time point. The main aim of the present study was, however, to evaluate any association between serum total homocysteine and ROP, not to investigate the causes of elevated homocysteine. Future studies that analyse a broader range of variables will provide more data regarding the relationship between homocysteine and ROP.
In conclusion, despite some limitations, the present study is the first to report an association between elevated serum total homocysteine levels and ROP. Determination of the exact role of homocysteine in ROP may facilitate the development of new strategies (such as addition of folic acid, vitamin B6 and vitamin B12 to infant formulae), to reduce homocysteine levels and help prevent more severe forms of ROP. More studies, including larger patient populations, are required to improve our understanding of the relationship between homocysteine and the development of ROP.
Footnotes
Acknowledgement
The authors wish to thank Gunay Can, MD, Department of Public Health, Cerrahpasa Faculty of Medicine, Istanbul University, Istanbul, Turkey, for assistance with the statistical analyses.
The authors had no conflicts of interest to declare in relation to this article.
