Abstract
This case report describes a rare instance of heart failure temporally associated with the administration of mydriatic eye drops during routine retinopathy of prematurity screening. A 36-week-old preterm infant developed acute tachypnea, tachycardia, and hepatomegaly shortly after receiving 0.5% tropicamide and 0.5% phenylephrine, and was subsequently diagnosed with heart failure, which responded well to intravenous deslanoside. Cardiac imaging later revealed multiple congenital anomalies, including an atrial septal defect, persistent left superior vena cava, and horseshoe lung, among others. No similar cardiovascular symptoms were observed during a subsequent screening using tropicamide alone, suggesting that phenylephrine may have contributed to the observed hemodynamic instability. This case illustrates the potential for severe cardiovascular complications associated with mydriatic agents in neonates with undiagnosed congenital heart defects. It underscores the need for individualized risk assessment, vigilant monitoring, and cautious application of mydriatic protocols to ensure patient safety in high-risk preterm populations.
Keywords
Introduction
Retinopathy of prematurity (ROP) is a severe complication in preterm infants that can lead to vision impairment and blindness if untreated. Mydriatic eye drops, such as cyclopentolate, tropicamide, and phenylephrine, are used before ophthalmic examinations to visualize the peripheral fundus. 1 These agents can be absorbed through the conjunctiva or nasal mucosa, bypassing first-pass metabolism and entering systemic circulation, leading to potential side effects. While side effects such as gastrointestinal, respiratory issues, and cardiovascular symptoms, including hypertension and tachycardia, following the use of mydriatic agents in neonates have been reported, heart failure has not been previously documented.2,3 In China, ROP screening is recommended for infants born at ⩽32 weeks of gestation or with a birth weight ⩽2000 g, with the initial examination typically conducted at 4–6 weeks of chronological age or at 31–33 weeks postmenstrual age, whichever is later. Infants outside these criteria who are considered high-risk may also undergo screening at the clinician’s discretion. 4 Preterm infants are particularly susceptible to systemic effects due to their immature physiological systems. Enhanced drug absorption, reduced metabolism, and limited drug elimination increase the risk of prolonged exposure and adverse reactions. In particular, phenylephrine poses concern due to its strong vasoconstrictive and cardiac stimulatory properties, which can significantly increase the risk of hypertension, arrhythmia, and cardiac overload in neonates with immature autonomic regulation. Mydriatic agents, such as phenylephrine, bind to alpha-1 receptors, causing vasoconstriction and pupil dilation. The onset of action occurs within 15–30 min, with peak effects at 60–90 min, and a duration of 3–6 h. Due to immature metabolism and clearance in neonates, systemic exposure is prolonged, increasing the risk of adverse reactions. This report presents a rare case of heart failure temporally associated with the use of mydriatic eye drops in a premature infant with congenital heart disease during ROP screening. In addition, we review the uncommon yet potentially serious systemic side effects associated with ROP examinations in high-risk neonates.
Case report
This neonate, a 36-week-old twin B, was born to a 27-year-old mother during her first pregnancy via cesarean section. Antenatal ultrasound had suggested a potential interrupted ascending aorta, but no other anomalies were detected. Twin A was normal. The neonate, classified as small-for-gestational-age with a birth weight of 1900 g, had Apgar scores of 9 and 10 at 1 and 5 min, respectively. Due to low birth weight, the infant was admitted to the local neonatal intensive care unit for observation. No assisted ventilation or oxygen therapy was required, as her condition remained stable. On day 4 of life, she was referred to our hospital for prematurity and suspected cardiac abnormalities. Initial echocardiography revealed an atrial septal defect (ASD) measuring 5.2 mm, a persistent left superior vena cava (PLSVC), and coronary sinus enlargement. Despite these findings, her circulatory system was stable, with normal hemodynamics: heart rate 130–150 bpm, respiratory rate 40 bpm, blood pressure within normal range, oxygen saturation 90%–95% on room air, and normal cardiac function per echocardiography. No vasoactive medications were required.
At 12 days of age, during her first ophthalmological screening for ROP, she received one drop each of 0.5% tropicamide and 0.5% phenylephrine in each eye, repeated three times at 5-min intervals. Two hours post-administration, she developed tachypnea (respiratory rate 50–70 bpm) and tachycardia (heart rate 150–180 bpm), while her blood pressure (62/40 mmHg) and oxygen saturation (90%–95%) remained within the normal range. Initially, infection was suspected, and intravenous antibiotics were administered. However, the infant remained afebrile, and blood tests revealed normal white blood cell counts and C-reactive protein levels. The following day, her symptoms persisted, with hepatomegaly extending 5 cm below the costal margin. The diagnosis of neonatal heart failure in this patient was made based on the presence of the following clinical indicators: tachypnea (respiratory rate >60 bpm), tachycardia (heart rate >150 bpm), hepatomegaly (liver palpable >3 cm below the costal margin), and an increased cardiothoracic ratio >0.6. These findings are consistent with established diagnostic criteria for heart failure in neonates. Intravenous deslanoside was administered at a dose of 10 μg/kg. Echocardiography revealed mildly reduced left ventricular systolic function. The patient received continuous monitoring of vital signs and electrocardiography. Supportive therapy included short-term use of furosemide and head-box oxygen therapy. Laboratory parameters—including liver and renal function tests and serum electrolyte measurements—remained within normal limits during treatment. Deslanoside was administered twice during the course. The patient exhibited significant clinical improvement within 12 h, with resolution of tachypnea and a reduction of hepatomegaly to 2 cm, enabling discontinuation of oxygen therapy. Deslanoside was discontinued after stabilization, and no adverse effects were observed. Following stabilization, cardiac CT revealed a left superior vena cava draining into the coronary sinus, along with a meandering right pulmonary vein, scimitar sign, collateral vessels, horseshoe lung, and pulmonary sequestration (Figure 1).
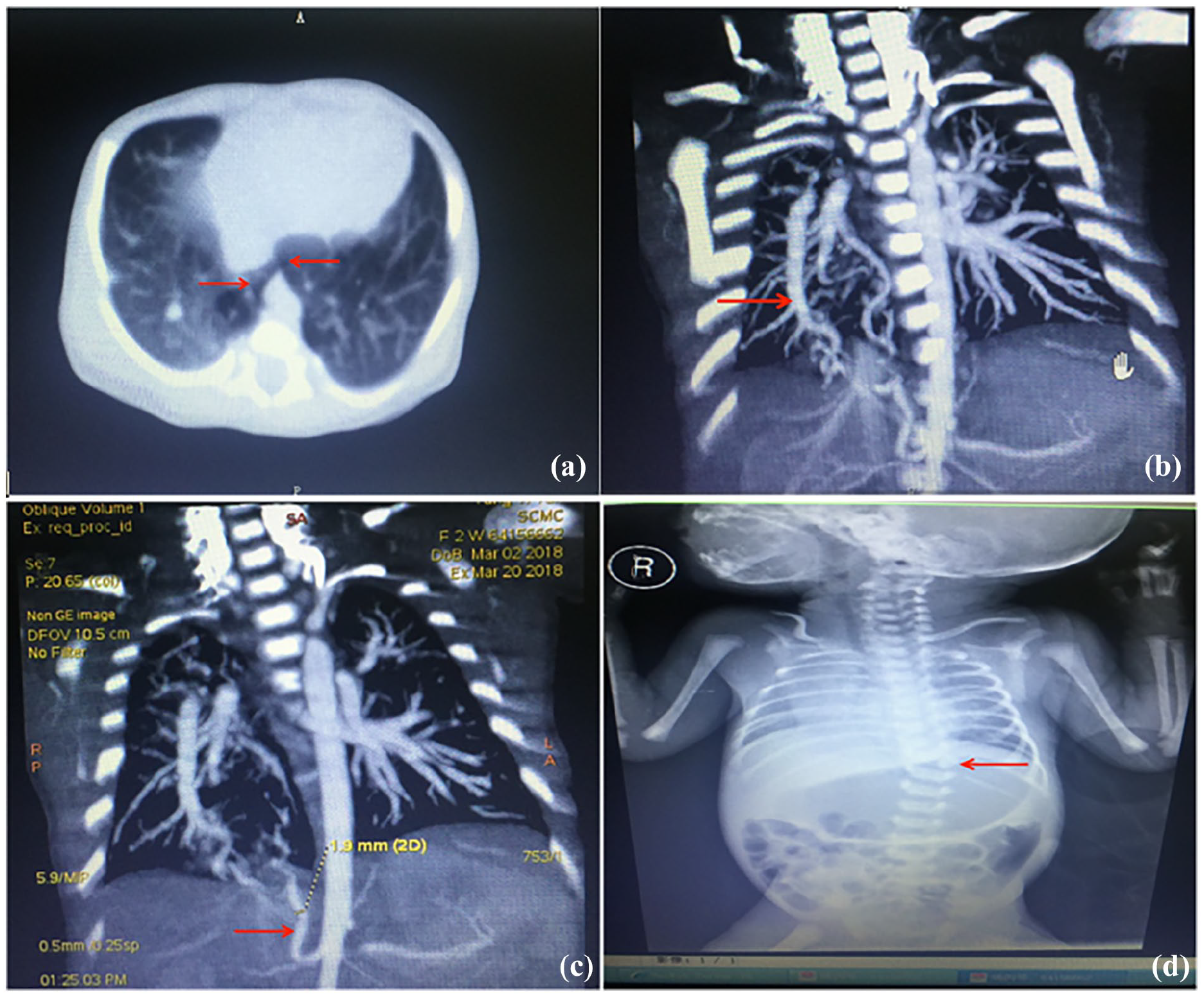
Enhanced chest computed tomography scan of this infant. Red arrows indicated: (a) horseshoe lung: the posterior basal segment of the right lower lobe and the left lower lobe are connected to the posterior part of the heart. (b) Scimitar sign: the right lower pulmonary vein converges upward, similar to a scimitar that converges with the right upper and middle pulmonary veins and enters the left atrium. (c) Pulmonary sequestration: the abdominal aorta displays a diameter of about 1.9 mm of collateral vessels supplying the right lower lung field. (d) Scoliosis.
In a subsequent ROP screening, only tropicamide was used. The infant experienced mild transient tachypnea, which resolved spontaneously without complications. Six days after the heart failure episode (on day 18 of life), follow-up echocardiography demonstrated an ASD measuring 6.4 mm, associated with collateral vessel formation and horseshoe lung. The patient was discharged on day 20 based on the cardiologist’s recommendation for outpatient follow-up and elective surgical repair. No signs of heart failure were observed during the outpatient monitoring period. At 8 months of age, repeat echocardiography revealed a reduction in the size of the ASD (3.7 mm), mild crossover of the pulmonary artery branches, right ventricular wall hypertrophy, and persistence of the left superior vena cava. The patient subsequently underwent surgical correction, with an uneventful postoperative recovery. During more than 2 years of follow-up, cardiac function remained stable, and no recurrence of heart failure was documented.
Discussion
ROP is a condition that primarily affects preterm infants and is a leading cause of childhood blindness worldwide. Effective management of ROP involves regular ophthalmologic examinations for at-risk preterm infants, which require the use of eye drops to dilate the pupils. 1 There are two main types of eye drops used for this purpose: parasympathetic blockers and sympathetic stimulants. Sympathetic stimulants, such as phenylephrine, directly or indirectly stimulate contraction of the iris radial muscle, causing pupil dilation. On the other hand, parasympathetic blockers, such as tropicamide, cyclopentolate, and atropine, act by inhibiting the contraction of the iris sphincter, resulting in pupil dilation. 2
Due to the incomplete development of the pupillary dilator muscle in neonates, multiple instillations are necessary for adequate pupil dilation. While several guidelines describe ROP screening, none specify the type of eye drops, combinations, or intervals of administration. Consequently, pupil dilation protocols may vary across hospitals within the same country. Common formulations for neonatal mydriasis include 0.5%–1.0% tropicamide, 0.5%–10% phenylephrine, and 0.2%–1.0% cyclopentolate. 2 Research indicates varying mydriatic effects depending on the doses and combinations used. 3 Phenylephrine and tropicamide eye drops are generally considered safe. Tropicamide, an anticholinergic, is preferred over cyclopentolate for low-birth-weight infants due to its shorter onset and faster recovery time. 5 However, in vulnerable preterm infants, particularly those weighing <1500 g, topical ophthalmic medications bypass gastrointestinal and hepatic metabolism, entering systemic circulation directly. This makes premature infants with immature cardiovascular and nervous systems potentially more susceptible to drug-related adverse effects. Rosales et al. administered 2.5% phenylephrine and 0.5% tropicamide to 10 low-birth-weight infants (<1600 g) every 5 min for three doses. 6 They found that systolic blood pressure increased by more than 20% in eight infants, and by more than 50% in three of these eight infants. Lux et al. conducted a study involving 512 preterm infants (birth weight ⩽1500 g and/or gestational age ⩽30 weeks), administering two drops of 5% phenylephrine and 0.5% tropicamide every 5 min. In this cohort, encompassing 1033 ocular fundus examinations, the retrospective analysis found no instances of mortality associated with the use of these eye drops. 7 In these prospective studies, all participating preterm infants were clinically stable, resulting in a low incidence and severity of eye drop side effects. However, serious complications during ROP screening have frequently been reported in preterm infants with underlying conditions, including necrotizing enterocolitis, bronchospasm, cardiac arrest, and even death. Although direct causality has not been established, the temporal association and physiological mechanisms suggest a potential contribution of mydriatic agents to these adverse events in vulnerable infants. A summary of adverse reactions in preterm infants with underlying conditions undergoing ROP screening with tropicamide (or atropine) and phenylephrine-containing mydriatic agents is provided in Table 1.5,8–14
Summary of adverse reactions in preterm infants undergoing ROP screening with mydriatic agents.

ROP: retinopathy of prematurity; NM: no mention; NEC: necrotizing enterocolitis; RDS: respiratory distress syndrome; BPD: bronchopulmonary dysplasia; IVH: intraventricular hemorrhage.
This report highlights the potential cardiovascular risks associated with the use of phenylephrine-containing eye drops in preterm infants with complex congenital heart defects. We conducted fundus screening for the patient in strict accordance with the Chinese guidelines for ROP screening. 4 A temporal relationship was observed, as tachycardia, tachypnea, and hepatomegaly developed 2 h after phenylephrine administration. Although systemic side effects of phenylephrine are typically rapid, research suggests that adverse effects may occur over a longer timeframe due to cumulative systemic absorption and the immature drug clearance pathways in neonates. Phenylephrine is an alpha-1 adrenergic agonist that induces vasoconstriction by binding to alpha-1 receptors on smooth muscle cells, resulting in an increase in systemic vascular resistance and cardiac afterload. In neonates with preexisting cardiac anomalies, such as ASD, PLSVC, and pulmonary venous abnormalities, this additional hemodynamic stress may overwhelm impaired compensatory mechanisms, potentially contributing to heart failure. The immature physiological systems in neonates, including underdeveloped autonomic regulation and limited drug metabolism and elimination capabilities, may amplify the systemic effects of phenylephrine, thereby increasing the risk of adverse outcomes such as cardiac decompensation. Previous studies have demonstrated that phenylephrine, even at low doses, can have significant systemic effects in vulnerable populations like preterm infants. Alpay et al. 12 and Shinomiya et al. 10 describe systemic vasoconstriction, reflex bradycardia, and renal complications following phenylephrine use in neonates, with these effects being more pronounced in individuals with underlying vulnerabilities. In addition, the absence of adverse events during a subsequent retinal screening using tropicamide alone supports the possibility that phenylephrine may have contributed to the observed complications. In the process of ROP screening, preterm infants with underlying heart conditions typically exhibit symptoms such as periorbital pallor, abdominal distension, or feeding intolerance, without signs of heart failure.10,11,13 To our knowledge, this is the first reported case in which heart failure occurred temporally after ROP screening, raising concern about a possible link between phenylephrine use and cardiac decompensation in vulnerable neonates. Nevertheless, we acknowledge that causality cannot be established based on a single case. While the infant’s congenital heart defects likely increased her susceptibility to hemodynamic stress, phenylephrine’s vasoconstrictive properties may have played a contributing role in the development of heart failure. This case underscores the importance of cautious drug selection and vigilant monitoring during procedures such as ROP screening in high-risk neonates.
To minimize the side effects of mydriatic eye drops, clinicians should evaluate premature infants before dilation, ensuring that vital signs—particularly peripheral oxygen saturation (SpO2), heart rate, and blood pressure—are stable. The administration of eye drops should adhere to the suggestions by Shinomiya et al.: (1) use the drops cautiously, monitoring pupil response to avoid overdosing; (2) apply pressure to the lacrimal sac after instillation to prevent drainage into the nasolacrimal duct; (3) monitor the infant’s vital signs closely for 12 h post-procedure to detect any adverse reactions. 10 Recent advancements in non-mydriatic imaging technologies, such as ultra-widefield fundus imaging, have enhanced the screening process for ROP in preterm infants. These methods reduce the need for pharmacological pupil dilation, minimizing potential systemic risks associated with mydriatic agents. In addition, microdosing techniques are being explored to decrease drug exposure during necessary dilation. These innovations offer a forward-looking perspective, emphasizing the development of safer screening methods for high-risk neonates, providing alternative approaches to ROP screening with reduced systemic risks. 15
Several limitations of this report should be acknowledged. First, as a single case study, the findings may not be generalizable to all preterm infants with congenital heart disease. Second, systemic drug levels were not measured, limiting our ability to assess the extent of phenylephrine absorption. Third, the infant’s preexisting cardiac anomalies may have independently contributed to hemodynamic instability, irrespective of phenylephrine exposure. Further research is needed to clarify the potential interactions between phenylephrine and specific congenital cardiac conditions.
Conclusion
We report a case of a premature infant with complex congenital heart disease and horseshoe lung who developed acute heart failure temporally associated with routine ROP screening with mydriatic eye drops. Prompt symptomatic treatment led to rapid clinical improvement. To our knowledge, this is the first reported case in which heart failure occurred shortly after the use of mydriatic agents during ROP screening. This case underscores the need for individualized risk assessment, close post-administration monitoring, and cautious use of mydriatic protocols in neonates with congenital heart disease. Further research is warranted to elucidate the potential association between mydriatic agents and cardiovascular complications, and to provide stronger evidence for their safe application in high-risk preterm populations.
Footnotes
Acknowledgements
The authors would like to thank the patient and his parents for allowing us to publish this case report.
Ethical Considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent for Publication
Written consent was obtained from parents for participants under 18 years old. Written consent was obtained from parents for the publication of their child’s personal or clinical details in this study.
Author Contributions
The authors’ contributions were as follows: X.L.: manuscript writing, conducting the study, and data collection. X.C.: designed and supervised the study. J.B. and J.S.: data collection and analysis. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The costs of the publication and molecular analyses of this research were supported by Research Project (Task) Assignment of Shanghai Science and Technology Commission (project ID: 20Z11900600). The funding bodies played no role in the design of the study, the collection, analysis, and interpretation of the data, or the writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the ICMJE uniform disclosure form. The authors have no conflicts of interest to declare.
