Abstract
Objective:
These studies evaluated the feasibility of using oral prolonged-release oxycodone/naloxone (OXN PR) for the management of acute postoperative pain.
Methods:
Three studies were undertaken: (i) the analgesic efficacy of OXN PR was compared with prolonged-release oxycodone (OXY PR) in patients with knee arthroplasty in an immediate postoperative period (IPOP) study; (ii) OXN PR treatment was compared with other opioids during rehabilitation after knee arthroplasty in a noninterventional study (NIS); and (iii) surgical patients on other opioids were switched to OXN PR postoperatively during a quality improvement programme (QIP).
Results:
In the IPOP study, the pain intensity at rest score decreased by a similar amount in the OXN PR and OXY PR groups, indicating similar analgesic efficacies. In the NIS, patient assessments indicated enhanced efficacy and tolerability for OXN PR compared with other opioids. The QIP indicated significant improvements in bowel function and less difficulty passing urine at the end of OXN PR treatment compared with baseline. No safety concerns were raised.
Conclusions:
The analgesic efficacies of OXN PR and OXY PR were similar in postoperative pain settings. OXN PR reduced the degree of restriction in relation to patients carrying out physio - therapy compared with other opioids, and improved bowel and bladder function.
Keywords
Some of results of the immediate postoperative period study were given as an oral presentation at the 31st Congress of the Scandinavian Society of Anaesthesiology and Intensive Care Medicine (SSAI), Bergen, Norway, 15 – 17 June 2011 (Abstract No. 0087).
Noninterventional study data were presented at the Sueddeutschen Orthopaedenkongress, Baden-Baden, Germany, 28 April – 1 March 2011.
Introduction
Postoperative pain after major orthopaedic surgery is often severe and significant pain may continue for several weeks. Severe acute pain is thought to be a risk factor for persistent postoperative pain and should always be treated effectively to prevent long-term physical and psychological problems.1,2 Nonsteroidal anti-inflammatory drugs are highly effective in relieving bone pain and are usually sufficient to manage mild to moderate pain;3,4 however, they are not adequate in patients with severe pain and are contraindicated in some patients. In a multimodal pain management programme, nonopioid analgesics were combined with opioid analgesics to enhance analgesic efficacy, reduce adverse effects and improve patient satisfaction. 5 Oxycodone is a μ-opioid agonist that is increasingly used in acute postoperative pain management. 6 Studies have demonstrated that oxycodone is well tolerated and highly potent in the management of moderate to severe pain.7,8 The elimination half-life of oxycodone is, however, relatively short (2 – 3 h), so controlled-release enteral preparations are preferred in conditions where pain is expected to be prolonged.
Controlled-release opioids were introduced to the management of postoperative pain in the 1990s, 9 and regular dosing with prolonged-release oxycodone (OXY PR) has been shown to be effective in improving pain control after major orthopaedic surgery. 10 OXY PR rapidly achieves and maintains effective blood concentrations of oxycodone over 12 h, which enables patients to be mobile for longer periods and to sleep through the night. 8
Naloxone is a narcotic antagonist that is given parenterally in the treatment of opioid overdose. When naloxone was administered orally to patients with opioid-induced constipation, bowel movements were significantly increased and laxative use was decreased. 11 When oxycodone was coadministered with naloxone in a combined prolonged-release tablet (OXN PR) to patients with chronic pain, the analgesic efficacy of oxycodone was sustained while opioid-induced bowel dysfunction was reduced.12–14
The OXN PR combination has been investigated in a number of different clinical trial settings, particularly in the management of chronic pain; however the efficacy of OXN PR has not been established in postoperative pain relief. In perioperative settings, it is theoretically possible that naloxone could antagonize the analgesic efficacy of opioids used for pain relief. It has been shown, however, that low systemic exposure to naloxone due to low bioavailability 15 may alleviate some of the adverse effects of opioids without reducing therapeutic efficacy.16–19
Since it has been shown that OXY PR provides effective analgesia following knee arthroplasty, 10 an immediate postoperative period (IPOP) study was designed to investigate whether OXN PR would be as effective as OXY PR in this situation. Since significant pain after knee arthroplasty may last for several weeks during rehabilitation, a concurrent, noninterventional study (NIS) investigated pain management after discharge using OXN PR or other opioids. In addition, as part of a quality improvement programme (QIP), the outcomes from usual clinical practice of patients treated with OXN PR after cervical spine or major orthopaedic joint surgery were observed.
Patients and methods
Study Designs
IPOP Study
The IPOP study was a randomized, double-blind, double-dummy, parallel-group, multicentre phase IV study to demonstrate noninferiority of the analgesic efficacy of OXN PR compared with OXY PR in patients with postoperative pain after unilateral primary knee arthroplasty. It was conducted between 25 March 2010 (first patient enrolled) and 17 October 2010 (last patient's last visit) at five tertiary hospitals in Finland (Turku University Hospital; Central Hospital of Pori; Coxa Hospital for Joint Replacement, Tampere; Hospital of Oulaskangas, Oulainen; Kuopio University Hospital). The study was approved by the Research Ethics Committee of the Hospital District of Northern Savo, Kuopio, Finland (dnro 133/2009), the Finnish Medicines Agency was notified of the study protocol (KLnro 175/2009) and it was registered with EudraCT (2009-016957-17) and ClinicalTrials.gov (NCT01083485). Written informed consent was obtained from all patients.
NIS
The NIS was an open-label, prospective study conducted at two hospitals in Germany (SRH Karlsbad-Langensteinbach Hospital, Karlsbad; Orthopädische Praxis Bad Camberg, Bad Camberg) to collect data on the efficacy, tolerability, functionality and effects on health-related quality of life (QoL) of OXN PR compared with other opioids (control group) in patients with severe pain after total knee arthroplasty. It was conducted between 28 September 2009 (first patient enrolled) and 15 December 2010 (last patient's last visit in the extension phase). The study was approved by the research ethics committee Ethikkommission Medizinische Fakultät, Heidelberg, Germany. The Federal Institute for Drugs and Medical Devices was notified of the study protocol and it was registered with the German Association of Research-based Pharmaceutical Companies database. Written informed consent was obtained from all patients.
QIP
The QIP was an open-label, exploratory, prospective follow-up survey conducted at two hospitals in Finland (Kuopio University Hospital, Kuopio; Harjula Hospital, Kuopio) with the aim of improving the care of patients who presented for cervical spine surgery or who were in a rehabilitation unit after major orthopaedic joint surgery. Patient observations were made between 7 December 2010 and 15 April 2011.
As this was a quality improvement project, approval of the local research ethics committee was not required (personal communication Kirsi Luoto, Head of Research Unit, Kuopio University Hospital, Kuopio, Finland).
Patients
IPOP Study
Male and female patients aged 18 – 75 years who were scheduled to have unilateral primary knee arthroplasty due to osteoarthritis, had not been given opioids in the 3 months prior to the study and who had a body mass index of 18 – 35 kg/m2 were recruited. Patients who were pregnant or lactating were excluded. All patients were to receive planned postoperative epidural analgesia for approximately 48 h and had an anticipated requirement for daily opioid treatment after epidural analgesia of 60 h.
NIS
Patients scheduled for knee arthroplasty surgery who had no contraindications to the use of OXN PR or other opioid therapy were eligible for inclusion in the NIS.
QIP
Patients scheduled for cervical spine surgery or receiving rehabilitation after major orthopaedic joint surgery and using regular opioid analgesics to control their pain were eligible for the QIP. All patients were anticipated to require daily opioid treatment for at least 7 days during recovery.
Patient Assessment
IPOP Study
The IPOP study was conducted in two phases. The first was a prerandomization phase, comprising a screening period (days –16 to –2), and a run-in period comprising the day of surgery (day –2) and the first 48 h after surgery (days –2 to 1). The second, double-blind phase comprised the 60-h treatment period (days 1, 2 and 3); the first day of dosing with study medication was day 1.
On the morning of day 1 (48 h after surgery), the patients were randomized in a 1 : 1 ratio to receive either oral OXN PR or OXY PR. Randomization was stratified by study site and by age (< 65 years and 65 years, according to local guidelines) using a block size of four and was performed by the sponsor using a validated system that automated the random assignment of treatment groups to random numbers.
Five doses of study medication were given: mornings and evenings on days 1 and 2 and in the morning on day 3. The first dose was given 1 h before the end of postoperative epidural analgesia. The dose of study medication was determined by the age of the patient: patients aged < 65 years received 20/10 mg OXN PR or 20 mg OXY PR, whereas patients aged 65 years received 10/5 mg OXN PR or 10 mg OXY PR. A double-dummy technique was used to maintain blinding.
For background analgesia, patients received paracetamol and meloxicam. For rescue analgesia, immediate-release 5 mg oxycodone capsules were given as required if the pain score at rest was 4/10 or higher on an 11-point numerical rating scale (NRS) of 0 – 10 (0, no pain; 10, most severe pain imaginable).
Prohibited concomitant medications were opioids other than the study medication and corticosteroids other than dexamethasone, which was allowed during the run-in period for the prevention of postoperative nausea and vomiting for patients at risk.
During the double-blind phase, patients rated their pain intensity on the 11-point NRS immediately before the first dose of study medication and then 1 h after each dose for all five doses. At each assessment, patients were asked to rate their average and worst pain intensity during the previous 24 h, and the pain right now, giving scores for both pain at rest and during movement (dynamic pain).
Vital signs, use of rescue medication, adverse effects and concomitant medications were recorded throughout the double-blind phase by the investigator. The primary efficacy outcome variable was the 24-h average pain intensity score at rest. The secondary efficacy outcome variables were the 24-h average dynamic pain intensity scores, the current rest and dynamic pain intensity scores, the 24-h worst rest and dynamic pain intensity scores and the use of rescue analgesia.
NIS
The NIS comprised three phases: screening, core, and follow-up. In the first phase, patients who gave informed consent were screened for their suitability to be included in the study. The day of surgery was designated day 1. The core phase of the study commenced on day 3 and was considered in three periods: the inpatient treatment period, the outpatient or rehabilitation period, and an initial follow-up period. The planned observation times were preoperative, 3 days after surgery, 6 days after surgery or at discharge from the hospital, 3 weeks after surgery or in the rehabilitation centre, and 4 – 5 weeks after surgery or at discharge from the rehabilitation centre. Patients who completed the core phase could enter the follow-up phase, which comprised an optional assessment 6 months after surgery.
As a noninterventional study, treatment for pain was given according to the attending physician's medical judgement. The dose and administration route of OXN PR were in accordance with the summary of product characteristics for Targin® (also marketed as Targinact® and Targiniq® available at http://www.medicines.ie/medicine/14383/SPC/Targin+10mg+5mg+and+20mg+10mg+prolonged+release+tablets/), with an initial dose of 10/5 mg OXN PR. Further titration and administration of a maintenance daily dose was based on each patient's individual need for analgesia and tolerability. Patients treated with other opioid analgesics comprised the control group. All medication for patients treated in the NIS was obtained from commercial sources.
Pain was evaluated using the Brief Pain Inventory–Short Form (BPI–SF), 20 which consists of seven pain interference items scored on an 11-point scale (0, does not interfere; 10, completely interferes). Overall efficacy and tolerability were scored by physicians and patients on a five-item scale (ranging from ‘very good' to ‘very bad'). Overall patient compliance was also scored by physicians in the same way. The degree of restriction in the ability to carry out physiotherapy was rated on a five-item scale (ranging from ‘without restriction' to ‘not able'). Patients were also assessed according to their rescue analgesic use, patient assessment of constipation using the Bowel Function Index (BFI), 21 the Hospital for Special Surgery (HSS) score (orthopaedic score), 22 the Larson knee score23,24 and a QoL questionnaire (Short Form [SF]-36). 25
Safety was assessed by monitoring patients for adverse drug reactions (ADRs).
QIP
The QIP was conducted in two parallel cohorts: patients receiving cervical spine surgery, and those receiving orthopaedic joint surgery. In both cohorts, the patients were interviewed using a structured questionnaire at baseline, at the end of OXN PR treatment and 14 days after the end of OXN PR treatment. At baseline, patients were questioned about bowel function and any adverse effects of the prescribed opioid analgesic. Patients rated their symptoms using an 11-point NRS and also by answering binomial questions (yes/no).
Patients scheduled for cervical spine surgery were switched from the opioid in use to OXN PR in the evening before surgery and were then given OXN PR twice daily for 7 days, i.e. a total of 14 doses of OXN PR. Other postoperative pain treatment was prescribed according to the medical judgement of the attending physician. After 7 days, the patients were switched back to the initial opioid that had been in use before surgery.
Patients in the rehabilitation unit following orthopaedic joint surgery were switched from the opioid in use to OXN PR twice daily for 14 days, i.e. a total of 28 doses of OXN PR. These patients were allowed to use concomitant nonopioid analgesics during the observation period but other opioid preparations were prohibited. After 14 days, the patients were switched back to the initial opioid that had previously been in use.
For each cohort, at the end of OXN PR treatment and at 14 days after the end of OXN PR treatment, the patients were asked to rate their satisfaction with the treatment on a five-point Likert scale (1, significantly worse; 5, much better). All medications for patients treated in the QIP were obtained from commercial sources.
Statistical Analyses
IPOP Study
The sample size for the IPOP study was calculated for a significance level of 2.5% (one-sided) with 90% power. The assumptions were within a SD of ± 1.7, an expected treatment difference of 0 and a noninferiority margin of –1.0 (for OXN PR versus OXY PR). Hence, a
For the primary efficacy outcome variable, absolute changes from baseline were analysed on the per protocol data using a mixed-model repeated measures analysis of covariance (RMANCOVA). The covariates used were site, age group, time point, treatment group and pain results immediately before the first dose. The noninferiority boundary was set to 1.0, i.e. noninferiority could be concluded if the upper 95% confidence limit of the treatment difference (OXN PR versus OXY PR) was ≤ 1.0. Supportive analysis of the full analysis population was also performed. For the secondary efficacy outcome variables of pain intensity scores, a similar statistical model was applied as for the primary efficacy outcome variable but the focus was on estimation of the treatment differences.
All statistical analyses were performed using SAS software version 9.1.3 (SAS Institute Inc., Cary, NC, USA).
NIS
Power calculations for the NIS were based on a two-sample
The treatment groups were compared by Fisher's exact test at each visit using the data available. All statistical tests were performed on an exploratory basis with a two-sided level of significance (α = 5%). The BPI–SF data were analysed by a RMANCOVA model with fixed factors of treatment, visit and treatment–visit interaction, and with the preoperative value as a covariate. The SF-36 items were analysed using the same methods as used for the BPI–SF. Summary statistics were produced for each of the efficacy and safety variables.
All statistical analyses were performed using SAS software version 9.1.3 (SAS Institute Inc.).
QIP
No formal sample size calculation was performed for the QIP. Samples consisting of 24 patients who were scheduled for cervical spine surgery and 20 patients in the rehabilitation unit were considered to provide sufficient information for clinical decision making. Ordinal data were tested with the Friedman two-way analysis of variance, and the Friedman/Cochran
Results
Patients
The numbers of patients enrolled, randomized and analysed in all three studies are summarized in Fig. 1, and Table 1 summarizes their demographic and clinical characteristics. The mean age of the patients who underwent spinal surgery was lower than that of the patients in the other treatment groups, otherwise the groups appeared reasonably well matched (not analysed statistically).
Flow charts of (A) the immediate postoperative period (IPOP) study, (B) the noninterventional study (NIS), and (C) the quality improvement programme (QIP) showing the number of patients enrolled and analysed (OXN PR, prolonged-release oxycodone/naloxone; OXY PR, prolonged-release oxycodone; FA, full analysis; PP, per protocol)
Baseline demographic and clinical characteristics of the patients (full analysis populations) who were enrolled in the immediate postoperative period (IPOP) study, the noninterventional study (NIS) and the quality improvement programme (QIP) investigating pain management using prolonged-release oxycodone/naloxone (OXN PR) and prolonged-release oxycodone (OXY PR)
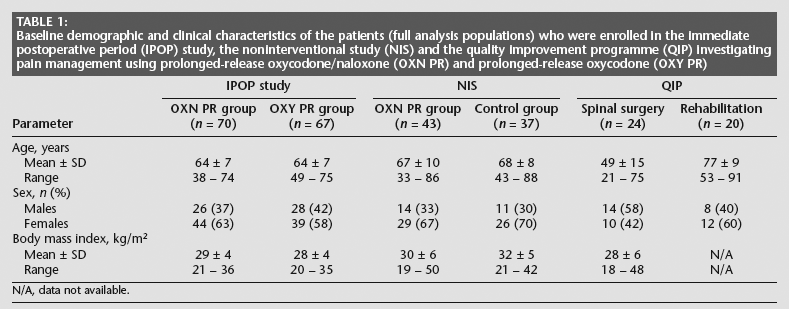
N/AN/A, data not available.
Due to the observational nature of the NIS, the times for patient assessment showed some variation. Observation was conducted preoperatively and at 3 days after surgery in all patients. The third assessment (6 days after surgery or at discharge from the hospital) took place a minimum of 4 days and a maximum of 8 days after surgery, the fourth assessment (3 weeks after surgery or in the rehabilitation centre) took place a minimum of 2.5 weeks and a maximum of 5.5 weeks after surgery, the fifth assessment (4 – 5 weeks after surgery or at discharge from the rehabilitation centre) took place a minimum of 4 weeks and a maximum of 10 weeks after surgery, and the final assessment (6 months after surgery) took place a minimum of 5 months and a maximum of 7 months after surgery.
Efficacy
24-h average pain intensity at rest score (IPOP study)
There were no clinically relevant differences between the two groups in the primary efficacy outcome parameter of 24-h average pain intensity score at rest in the per protocol population (Fig. 2; not analysed statistically). At baseline, the mean 24-h average pain intensity at rest score was slightly higher in the OXN PR group (3.4 points) than in the OXY PR group (3.1 points). Overall, the 24-h average pain intensity at rest score decreased by a mean of 1.2 (95% confidence interval [CI] –1.5, –0.9) in the OXN PR group and by a mean of 1.1 (95% CI –1.4, –0.8) in the OXY PR group. The mean difference between the groups in the change from baseline in 24-h average pain intensity at rest score was –0.1 (95% CI –0.5, 0.3). A sensitivity analysis on the full analysis population supported these results in the per protocol population (Table 2). These data confirm the noninferiority of OXN PR compared with OXY PR.
The 24-h average pain intensity scores at rest over time for patients in the immediate postoperative period study (per protocol population) who were treated with either prolonged-release oxycodone/naloxone (OXN PR) or prolonged-release oxycodone (OXY PR) (data presented as mean ± SD; time points were the same for the two groups but the data are displayed offset for clarity)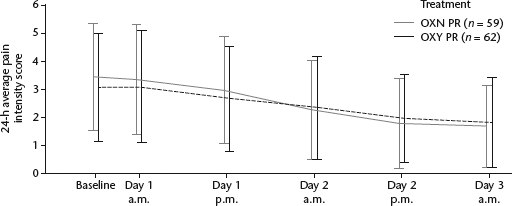
Baseline pain intensities, estimated changes from baseline in pain intensities and estimated differences between treatment groups for patients in the immediate postoperative period study (full analysis population) who were treated with either prolonged-release oxycodone/naloxone (OXN PR) or prolonged-release oxycodone (OXY PR)

CI, confidence interval.
Immediate-release oxycodone was taken during the treatment phase as rescue medication by 55 out of 70 patients (79%) in the OXN PR group and by 49 out of 67 patients (73%) in the OXY PR group.
Additional pain intensity scores (IPOP study)
As for the 24-h average pain intensity at rest scores, there were also no differences between the two groups in the 24-h average dynamic pain intensity scores. In addition, there were no differences between the groups in the secondary pain intensity measurements of current pain intensity and worst pain intensity scores at rest and on movement (Table 2).
BPI–SF (NIS)
The results of the composite of the seven pain interference items of the BPI–SF (‘patient pain experience') are shown in Fig. 3. Mean overall pain scores preoperatively were 4.1 in the OXN PR group and 4.3 in the control group. On day 3 postoperatively, overall pain experience scores decreased in both groups: to 3.9 in the OXN PR group and to 4.2 in the control group. At discharge from hospital (or at day 6 postoperatively), overall pain experience scores were 3.2 in the OXN PR group and 3.4 in the control group. Overall pain experience scores continued to fall throughout the follow-up period in both groups. Overall, there was no statistically significant effect of treatment group on the BPI–SF patient pain experience score.
‘Pain experience’ scores for patients in the noninterventional study (full analysis population) who were treated with either prolonged-release oxycodone/naloxone (OXN PR) or other opioid analgesics (control) (data presented as the mean ± SD of the composite scores of the Brief Pain Inventory–Short Form
20
of seven pain interference items scored on an 11-point scale [0, does not interfere 10, completely interferes]; time points were the same for the two groups but the data are displayed offset for clarity)
Results for each of the seven individual pain interference items from the BPI–SF are given in Table 3. At week 3, mean scores in the OXN PR group appeared to be slightly better than in the control group for mood, normal work, relations with others, sleep, and enjoyment of life, whereas mean scores in the control group appeared to be slightly better than in the OXN PR group for general activity and were the same in both groups for walking ability (data not compared statistically at each time point). In both groups pain interference scores improved from preoperative values over the period of the study. The effects of baseline value and visit on pain interference were statistically significant (both
Brief Pain Inventory-Short Form pain interference scores a for patients in the noninterventional study (full analysis population) who were treated with either prolonged-release oxycodone/naloxone (OXN PR) or other opioid analgesics (control)

Data presented as mean ± SD.
Scored on an 11-point scale: 0, does not interfere 10, completely interferes.
Pain interference scores improved from preoperative values over the period of the study in both groups. The effects of baseline value and visit on pain interference were statistically significant (both
The most frequently prescribed rescue opioid was immediate-release morphine sulphate, which was given to 17 out of 43 patients (40%) in the OXN PR group and 32 out of 37 (87%) patients in the control group.
Ability to carry out physiotherapy (NIS)
Patients in the OXN PR group were less restricted in their ability to carry out physiotherapy at the follow-up visit at weeks 4 – 5 than patients taking other opioid analgesics (control group) (Table 4). No patient in either group was rated as ‘with severe restrictions' or ‘not able' to carry out physiotherapy.
Comparison of treatment with prolonged-release oxycodone/naloxone (OXN PR group) or other opioid analgesics (control group) on the degree of restriction in carrying out physiotherapy (noninterventional study; full analysis population)

Data presented as number (%) of patients.
Overall assessment of efficacy, tolerability and compliance (NIS)
At the end of the core phase (4 – 5 weeks after surgery), efficacy and tolerability ratings scored by physicians and patients appeared to be generally better in the OXN PR group than in the control group (Table 5; not analysed statistically). There were no efficacy or tolerability ratings of ‘bad' or ‘very bad' in the physician assessments 4 – 5 weeks after surgery; furthermore no patient rated efficacy as ‘bad' or ‘very bad'. At 6 months after surgery, both assessments by physicians and patients indicated better efficacy in the OXN PR group compared with the control group (Table 5; not analysed statistically).
Overall assessment of efficacy, tolerability and compliance for patients in the noninterventional study (full analysis population) who were treated with either prolonged-release oxycodone/naloxone (OXN PR) or other opioid analgesics (control)

Data presented as number (%) of patients.
Data not available for all patients.
At both time points, physicians tended to rate compliance in the OXN PR group as better than in the control group (Table 5; not analysed statistically). No patient was assessed as having ‘bad' or ‘very bad' compliance.
Bowel function (NIS)
The BFI scores were not significantly different between the groups at any time (data not shown). Laxative use was, however, more common and they were taken for longer in the control group than in the OXN PR group: laxatives were taken by nine out of 43 patients (21%) in the OXN PR group and in 12 out of 37 patients (32%) in the control group.
Other assessments (NIS)
Overall improvements compared with baseline appeared to occur for both patient groups over time in HSS scores, Larson knee scores and QoL measures (data not shown; not analysed statistically). QoL assessment as measured by SF-36 scores appeared to improve in line with the decrease in patients' pain scores (data not shown; not analysed statistically).
Experience from the QIP
Of the 24 patients undergoing cervical spine surgery in the QIP, three received 5/2.5 mg, 13 received 10/5 mg and eight received 20/10 mg OXN PR. Of the 20 patients undergoing rehabilitation following orthopaedic joint surgery, five received 5/2.5 mg, seven received 10/5 mg and eight received 20/10 mg OXN PR.
Most patients had symptoms of bowel dysfunction at baseline, however significant improvements in bowel function were observed after switching to OXN PR (Table 6). The effects were sustained 14 days after treatment with OXN PR, indicating the possibility of a carryover effect. Moreover, OXN PR improved not only bowel function but also bladder function. At baseline, nine out of 44 patients (20%) on opioid analgesics reported voiding difficulties but, at the end of OXN PR treatment, only two out of 36 patients (6%) still had difficulties. The beneficial effect on bladder function was not sustained for as long as the change in bowel function; within 14 days after switching back to an opioid without naloxone, voiding difficulties recurred in two additional patients (9 out of 44 patients; 20%).
Based on patient satisfaction rated on a five-point Likert scale, the analgesic efficacy of OXN PR was graded as not inferior compared with the initial opioid compound (data not shown). When patients were questioned about bowel function and any adverse effects of the prescribed opioid analgesic rated using an 11-point NRS, the mean ± SD for patients' satisfaction was also similar compared with the initial opioid compound (6.7 ± 1.8 at baseline before switching to OXN PR and 7.0 ± 1.7 at the end of OXN PR treatment).
Bowel and bladder function data from the quality improvement programme evaluation of pain management using prolonged-release oxycodone/naloxone (OXN PR) in patients following cervical spine or orthopaedic joint surgery

Data presented as mean ± SD or number (%) of patients/available data.
Comprises 19 patients who had undergone cervical spine surgery treated with OXN PR for 7 days and 19 patients who had undergone orthopaedic joint surgery treated with OXN PR for 14 days.
Comprises 22 patients who had undergone cervical spine surgery and 17 patients who had undergone orthopaedic joint surgery.
Numerical rating scale variables were analysed using Friedman's two-way analysis of variance; binary variables were analysed using Friedman's/Cochran's
Scored on an 11-point scale.
Proportion of patients who answered ‘yes’.
Safety
There were no serious ADRs related to the medications in any of the three studies. All of the ADRs recorded were mild or moderate.
During the double-blind phase of the IPOP study, there was no difference between the two groups in the incidence or severity of ADRs (i.e. unlikely, possibly, probably or definitely related adverse events): 36 out of 70 patients (51%) in the OXN PR group and 34 out of 67 patients (51%) in the OXY PR had ADRs. The most common ADRs in both groups were constipation, nausea, vomiting, dizziness and insomnia. Three patients were withdrawn from the study due to adverse effects: two from the OXN PR group (dizziness and vomiting) and one from the OXY PR group (fatigue).
In the NIS, 10 out of 43 patients (23%) in the OXN PR group and 14 out of 37 patients (38%) in the control group had ADRs. Two patients in the control group (5%) discontinued opioid analgesics permanently due to moderate constipation. The most frequent ADRs in both treatment groups were gastrointestinal disorders (constipation and nausea). Constipation was recorded as an ADR in eight out of 43 patients (19%) in the OXN PR group and in 11 out of 37 patients (30%) in the control group. Nausea was recorded as an ADR in three out of 43 patients (7%) and four out of 37 patients (11%) in the OXN PR and control groups, respectively.
At baseline in the QIP, before switching to OXN PR, 22 out of 44 patients (50%) reported ADRs related to opioid analgesia. The most common ADRs were constipation, dizziness and somnolence. Four patients discontinued OXN PR in the QIP due to ADRs: two due to dizziness and two due to nausea. At the end of the OXN PR treatment period, 11 out of 38 patients (29%) reported ADRs, with insomnia and mild dizziness being the most common symptoms. Within the first 14 days after switching back to the initial opioid, 12 out of 39 patients (31%) reported ADRs, the most common being neurological and muscle symptoms.
Discussion
Both the short-term randomized controlled trial (IPOP) and the longer-term NIS and QIP data confirmed that the OXN PR combination provides comparable analgesic efficacy to OXY PR alone and that the naloxone component does not impair analgesic efficacy. In these studies the analgesic efficacy of OXN PR was not inferior to OXY PR in the management of postoperative pain during the first days after surgery in hospital or later after discharge during the rehabilitation period. These findings are consistent with the results of other studies that have compared OXN PR with OXY PR in the treatment of chronic cancer pain and noncancer pain.12–14,26,27
In the IPOP study, the primary efficacy outcome measure of mean 24-h pain intensity score at rest at baseline in the full analysis population was higher in the OXN PR group compared with the OXY PR group (3.7 points versus 3.1 points, respectively). Since the mean estimated change from baseline was slightly greater in the OXN PR group compared with the OXY PR (1.2 points versus 1.1 points, respectively), the mean 24-h pain intensity scores at rest at the end of treatment were similar in the two groups. All the secondary efficacy outcome variables of pain intensity scores also decreased in both groups. Therefore, the findings from the secondary efficacy analysis support those of the primary efficacy analysis in that there was no clinically meaningful difference between OXN PR and OXY PR in terms of pain relief. The present data are consistent with a previous study that showed that a fixed-dose, scheduled oral opioid treatment as part of multimodal pain management is a feasible approach to postoperative pain management in total joint arthroplasty. 28
Opioid-induced constipation is the most common and burdensome adverse effect of opioid treatment, often leading to discontinuation of opioid analgesics despite the patient's need for effective pain relief.18,19 Results from the NIS and QIP showed that the incidence and severity of constipation (QIP) as well as the need for additional laxatives (NIS) were lower in patients receiving OXN PR treatment, suggesting that OXN PR has better gastrointestinal tolerability than other opioids.
The present NIS and QIP data, which were collected under usual clinical practice conditions, showed that OXN PR provided effective analgesia and offered several long term benefits. Due to effective pain control, patients' ability to undertake physiotherapy improved. It is likely that more physiotherapy would result in an improved early functional result in patients with postoperative pain after knee arthroplasty surgery. The beneficial effects were still perceptible 6 months after surgery. Long-term observations from clinical practice in the QIP showed that OXN PR may provide beneficial effects, not only on bowel function but there may also be a decreased incidence of other opioid-related peripheral adverse effects. In the QIP, patients reported a lower incidence of bladder dysfunction at the end of OXN PR treatment than during the treatment periods with other opioid analgesics. The present data have suggested that OXN PR can have a positive carryover effect on bowel function in patients with a long-term need for opioid analgesia and there were indications that bowel function remained improved compared with baseline up to 14 days after stopping treatment with OXN PR.
In our opinion, the present findings are clinically relevant. The data from the IPOP study showed that a combination of oxycodone and naloxone in prolonged-release tablets provided the same analgesic efficacy as oxycodone alone, which mitigates against the theoretical possibility of naloxone antagonizing the analgesic efficacy of opioids in this setting. The combination treatment provided effective oral analgesia after weaning off epidural analgesia, thereby facilitating patient mobility and recovery. Moreover, patients on opioid analgesia often exhibit opioid-related ADRs that may negate the beneficial effects of pain relief. The present data support the view that switching to OXN PR may relieve some of the opioid-induced ADRs and, thus, allow patients to have effective analgesia during recovery from surgery.
While all patients in the IPOP study were opioid naïve, patients in the NIS and QIP were not. The safety findings in all three studies were consistent with the known adverse effects of opioids and no new unexpected safety concerns were raised. This indicates that a fixed-dose oxycodone regimen could be used as a part of multimodal pain management regimen in patients with arthroplasty. This approach could also potentially reduce hospital costs and lead to reduced use of healthcare resources, as has been shown by de Beer
In conclusion, OXN PR (oxycodone/ naloxone 10/5 mg and 20/10 mg) was as effective as OXY PR (10 mg and 20 mg) or other opioids at reducing pain for all the pain intensity score measures used in these studies. The results of the IPOP study were supported by the findings from data collected from usual clinical practice in the NIS and QIP, which showed that the analgesic efficacy of OXN PR is sustained over a long period of time, leading to improved recovery both in terms of the ability to carry out physiotherapy and fewer adverse effects such as opioid-induced constipation. There may also be additional benefits of OXN PR, such as less difficulty passing urine. Overall, the use of prolonged-release OXN PR was shown to be an effective analgesic for the management of acute postoperative pain following knee arthroplasty.
Footnotes
Acknowledgements
We thank the coinvestigators and study nurses for their clinical support in performing this study. We thank Tiina Hakonen MSc and Wendy Kingdom PhD of Encorium Ltd, Espoo, Finland, for providing statistical analysis and medical writing services, respectively, on behalf of Mundipharma Oy, Vantaa, Finland. The contributions of Mrs Hakonen and Dr Kingdom were funded at commercial rates by Mundipharma Oy.
We also thank Mundipharma Oy for scientific discussions of the data. In particular, we thank Jaana Siljander who was employed by Mundipharma Oy at the time the study was conducted. The corresponding author takes responsibility for the integrity and accuracy of the data analysis and also had final responsibility for the decision to submit to the publication.
Copyright for the BFI is owned by Mundipharma GmbH, 2002; the BFI is the subject of European Patent Application Publication No. EP 1 702 558 A1 and corresponding patents and applications in other countries.
The IPOP study (protocol number OXN4505) was designed with Mundipharma Oy and was conducted by qualified investigators under the full sponsorship of Mundipharma Oy. Data were gathered by the sponsor and evaluated jointly by the authors and the sponsor. The NIS study was designed with Mundipharma GmbH, Limburg an der Lahn, Germany, and was conducted by qualified investigators under the full sponsorship of Mundipharma GmbH. The observations from daily clinical practice for QIP were not funded externally. Writing assistance and publication fees for this manuscript were funded by Mundipharma Oy. Note: Targin®, Targinact®, and Targiniq® are registered trademarks.
