Abstract
Objective:
To evaluate the impact of multiple-dose PB-119 injection (a pegylated exenatide formulation) on the pharmacokinetics of single-dose Digoxin or Warfarin sodium and on the pharmacodynamics of Warfarin sodium in healthy Chinese adults.
Methods:
This study is a single-centre, open-label, randomized, two-period, two-sequence, parallel-group design phase I clinical trial of drug interactions, aiming to evaluate the pharmacokinetic and pharmacodynamic effects of multiple administrations of PB-119 injection (a pegylated exenatide formulation) on single-dose oral Digoxin and Warfarin sodium in healthy Chinese adults. Sixteen healthy subjects were enrolled in each of two cohorts: Digoxin (Digoxin 0.5 mg versus Digoxin 0.5 mg + PB-119 150 μg) and Warfarin sodium (Warfarin 5 mg versus Warfarin 5 mg + PB-119 150 μg). Treatments were administered in a fixed sequence. The total duration of the study for each subject was 33 days, consisting of a 14-day screening phase (day −14 to day −1), a 9-day inpatient treatment phase (day 1 to day 9 for the Digoxin cohort; day 1 to day 16 for the Warfarin cohort to allow international normalized ratio follow-up), and a 10-day post-treatment safety follow-up (day 10 or day 17 to day 23 or day 30, respectively).
Statistical analysis:
Pharmacokinetic parameters for Digoxin and Warfarin sodium, as well as pharmacodynamic parameters (INRAUC0–last and INRmax) for Warfarin, were calculated using non-compartmental analysis with SAS® 9.4 (SAS Institute, Inc., Cary, NC, United States). Bioequivalence and safety were assessed in both cohorts.
Results:
The rate and extent of absorption of Digoxin or Warfarin sodium administered alone were comparable to those observed when co-administered with PB-119. The 90% confidence intervals of the geometric mean ratios for the primary pharmacokinetic parameters (
Conclusions:
Multiple-dose PB-119 injection does neither alter the pharmacokinetic characteristics of single-dose Digoxin or Warfarin sodium, nor does it influence the pharmacodynamic characteristics of Warfarin sodium, in healthy Chinese subjects.
Background
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder characterized by persistent hyperglycaemia resulting from impaired insulin secretion and/or insulin action, accounting for approximately 95% of all diabetes cases worldwide. 1 In China, nearly 10% of the adult population—about 110 million individuals—have diabetes, while almost 50% (≈500 million) are in a pre-diabetic state. Without urgent public-health interventions targeting modifiable risk factors, the number of people with diabetes in China is projected to reach 150 million by 2040. 2 Consequently, the development of novel antihyperglycaemic agents that achieve robust and durable glycaemic control and ideally attenuate diabetes-related complications—remains an important therapeutic goal.
Glucagon-like peptide-1 (GLP-1) is an incretin hormone released from intestinal L-cells. Binding of GLP-1 to its G-protein-coupled receptor (GLP-1R) potentiates glucose-dependent insulin secretion, suppresses glucagon release, and exerts β-cell-protective effects. In addition, GLP-1 slows gastric emptying and intestinal motility, increases satiety, and reduces food intake.2–4 Compared with conventional antihyperglycaemic agents, GLP-1 receptor agonists (GLP-1RAs) offer several advantages: they stimulate insulin secretion only under hyperglycaemic conditions, improve β-cell function, delay gastric emptying, promote satiety, facilitate body-weight reduction, and slow the progression of diabetic kidney disease—all with a low risk of hypoglycaemia.5–8 Byetta® (Exenatide), the first GLP-1RA approved for T2DM, received FDA clearance in 2005. 9 Its short half-life necessitates twice-daily injections, which compromises long-term adherence and is associated with dose-dependent gastrointestinal adverse events such as nausea and vomiting. 10
In the present study, we employed PB-119 injection, a long-acting GLP-1 receptor agonist engineered through a site-specific polyethylene glycol (PEG)ylation platform. Covalent attachment of branched PEG chains increases the apparent molecular weight, thereby reducing renal filtration and prolonging circulatory half-life. 11 In addition, the hydrated PEG shell creates a steric barrier that shields the peptide backbone from proteolytic enzymes, further decreasing degradation and extending pharmacological activity. 12 Pre-clinical studies (manuscript in preparation) demonstrate that PB-119 exhibits superior GLP-1 receptor potency, favourable pharmacokinetics (PK), and an expanded dosing interval compared with native exenatide, while maintaining a lower incidence of gastrointestinal adverse events.
GLP-1RA typically has the effect of delaying gastric emptying and gastrointestinal motility. For patients who need oral drugs to be rapidly absorbed through the gastrointestinal tract, the increased release caused by the prolonged gastric retention time may slightly increase drug exposure. Diabetes patients often have comorbidities such as hypertension, hyperlipidaemia, and cardiovascular diseases, and often use multiple drugs. Therefore, the selection of Digoxin or Warfarin sodium is based on their wide application in diabetic patients. Warfarin sodium is an oral anticoagulant of the coumarin class, which is a mixture of two different active racemic isomers, R and S isomers. The two different racemic isomers have different metabolic pathways in the liver. This drug is mainly used for the prevention and treatment of thromboembolic diseases. Digoxin is a moderately potent cardiac glycoside purified from
This study evaluated the effects of multiple subcutaneous injections of PB-119 injection on the PK characteristics of a single oral dose of Digoxin/Warfarin sodium and the pharmacodynamic (PD) characteristics of Warfarin sodium in healthy Chinese subjects through the assessment of Digoxin and Warfarin sodium, using a single-dose administration design.
Materials and objects
Research materials
PB-119 injection (0.15 mg/0.3 mL, lot 21112661; Hangzhou Asia-Pacific Biotechnology Co., Ltd., Hangzhou, China); Digoxin (0.25 mg/tablet, lot no. BHG0746; Sanofi (Hangzhou) Pharmaceutical Co., Ltd., Hangzhou, China); and Warfarin sodium (2.5 mg/tablet, lot no. 1M0281DC8; Qilu Pharma-ceutical Co., Ltd., Jinan, China) are research materials. The above-mentioned drugs were all provided by PAG Biopharmaceuticals (Suzhou) Co., Ltd. (Suzhou, China).
Sample size and dose selection
This study adopted a two-cycle crossover design. The equivalence criterion was set as the 90% confidence interval completely falling within the range of 80%–125%. Based on the coefficient of variation within subjects of Digoxin (8.5%) and Warfarin (4.9%), 15 each group required 12 evaluable subjects. Considering a 25% dropout rate, ultimately, 16 subjects were enrolled in each group, totalling 32 subjects. This number was sufficient to meet the equivalence test requirements for the absence of clinically significant interactions between PB-119 and Digoxin or Warfarin at an 80% efficacy level.
Based on the animal data that has been obtained (not disclosed), we conducted preclinical measurements of the acute and long-term toxic effects of PB-119 in animal models to assess the potential safety risks and side effects of PB-119 in clinical applications. The results indicated that PB-119 exhibited good safety and tolerance at dose levels ⩽200 μg. Similarly, Liu et al.
16
demonstrated that the median
Inclusion criteria
Inclusion criteria include: (1) the subjects can communicate effectively with the researchers and understand and comply with all the requirements of this study; (2) healthy Chinese male subjects aged 18 to 55 years (inclusive) at the time of signing the informed consent form (ICF); (3) body mass index (BMI) ranging from 19.0 to 28.0 kg m−2 (inclusive); and weight ⩾50.0 kg; and (4) there are diseases with abnormal clinical manifestations in the past and at present that need to be excluded (judged by the researchers), including but not limited to diseases of the nervous system, cardiovascular system, blood and lymphatic system, immune system, kidneys, liver, gastrointestinal tract, respiratory system, metabolism, bones, etc.
Exclusion criteria
Exclusion criteria include: (1) those with a history of active ulcers or significant bleeding in the gastrointestinal, genitourinary or respiratory tracts, central nervous system, haemorrhage, or anaemia; (2) those with a history of drug abuse or drug use within the 6 months prior to screening, or with a positive urine drug screen result; and (3) subjects or their partners who have plans to conceive during the entire study period and within 90 days after the last administration of the study drug, or who are unwilling to take effective contraceptive measures.
Scheme design
This study was a phase I, open-label, fixed-sequence clinical trial conducted from June 7, 2022 to August 2022 at the First Affiliated Hospital of Bengbu Medical University. The ethical review conclusion was “Agree,” and the ethical approval number was: [2022]065. This study was conducted in accordance with the Declaration of Helsinki (2024 revision) and the International Council for Harmonisation Good Clinical Practice (ICH-GCP) guidelines. 18 The informed consent process was strictly conducted in accordance with GCP requirements. After the investigators or their designated personnel fully explained the purpose, methods, expected benefits, and potential risks of the study to the subjects, the subjects confirmed their voluntary participation in the clinical trial, filled out the questionnaire and signed the ICF.
Dosing regimen
This study plans to include 2 parallel groups, with 16 subjects planned to be enrolled in each group. Subjects in the first group will receive Digoxin tablets and PB-119 injection in a fixed sequence, while subjects in the second group will receive Warfarin sodium tablets and PB-119 injection in a fixed sequence.
Before taking the Warfarin sodium/Digoxin tablets/PB-119, the subjects should fast overnight for at least 10 h. The Warfarin sodium/Digoxin tablets/PB-119 can be administered at approximately 8:00 a.m. (±2 h) in an empty stomach. After the administration, they should fast for at least 1 h before receiving breakfast (e.g., about 9:00 a.m. to eat), then receive the standard lunch about 4 h after the administration, and have the dinner as the standard meal. In the Digoxin group, blood samples were collected continuously from day D1 to D17 until 168 h (7 days) post-administration. The subjects D1 to D8 and D17 to D24 had to be hospitalized. In the Warfarin group, blood samples were collected continuously from D1 to D24 until 168 h. The subjects D1 to D8 and D24 to D31 had to be hospitalized.
During each outpatient visit and phone follow-up, inquire about the use of all medications by all subjects and record “prescription drugs, over-the-counter drugs, vaccines, dietary supplements or Chinese herbal medicines,” etc. During the follow-up (including phone follow-up), actively ask if any non-study drugs are being used concurrently; if any inducers/inhibitors that may affect PK are found, they need to be recorded and evaluated to determine if they deviate from the protocol. The dosing regimens are shown in Figures 1 and 2.

Study administration process of Digoxin group.

Administration process of Warfarin group.
Blood sample collection and processing
PK blood sample collection
For the Digoxin group and Warfarin sodium group, single doses of Digoxin tablets and Warfarin sodium tablets were administered on D1 and D17, and on D1 and D24, respectively. Blood samples were collected within 1 h before administration and at 0.25 h ± 1 min, 0.5 h ± 2 min, 1 h ± 2 min, 1.5 h ± 2 min, 2 h ± 2 min, 2.5 h ± 2 min, 3 h ± 2 min, 4 h ± 2 min, 6 h ± 2 min, 8 h ± 2 min, 12 h ± 5 min, 24 h ± 5 min, 36 h ± 5 min, 48 h ± 5 min, 72 h ± 5 min, 96 h ± 5 min, 144 h ± 5 min, and 168 h ± 5 min after administration for the assessment of PK parameters.
PD blood sample collection
In the Warfarin sodium group, single doses of Warfarin sodium tablets were administered on D1 and D24. PD blood samples were collected within 1 h before administration and at 12 h ± 5 min, 24 h ± 5 min, 36 h ± 5 min, 48 h ± 5 min, 60 h ± 5 min, 72 h ± 5 min, 96 h ± 5 min, 120 h ± 5 min, 144 h ± 5 min, and 168 h ± 5 min after administration for the assessment of PD parameters.
Blood sample processing
For each subject in this trial, one tube of blood was collected at each PK time point, with a total volume of 4 mL. The collection tubes were purple-cap 4 mL K2-EDTA plastic tubes containing EDTA-K2 anticoagulant. After collection, the tubes were centrifuged at 1300
PK, PD parameters, and statistical analysis
The plasma concentration data were analysed using the non-compartmental analysis method with Phoenix WinNonlin software (Certara USA, Inc., Princeton, NJ, United States) version 8.2. The main PK parameters of the study drug, including
Safety
The safety evaluation indicators of this study include: adverse events, 12-lead electrocardiogram, physical examination, vital signs, blood glucose levels, blood routine, blood biochemistry, coagulation function, urine routine, etc. After the subjects sign the ICF, if any adverse medical event (AE) occurs due to the procedures specified in the trial protocol, it should also be recorded as AE.
Results
Demographic characteristics
Baseline characteristics are presented separately for the two cohorts (Digoxin cohort and Warfarin cohort). The age of the Digoxin group and the Warfarin sodium group was (30.0 ± 9.25) and (30.0 ± 8.22) years old, the height was (172.31 ± 7.234) and (174.47 ± 6.236) cm, the weight was (65.96 ± 7.458) and (70.31 ± 8.242) kg, and the BMI was (22.199 ± 1.964) and (23.104 ± 2.576) kg m−2, respectively. The specific demographic characteristics are shown in Tables 1 and 2.
Demographic and baseline characteristics of the Digoxin group subjects (

BMI: body mass index; SD: standard deviation; CV: coefficient of variation.
Demographic and baseline characteristics of the Warfarin sodium group subjects (
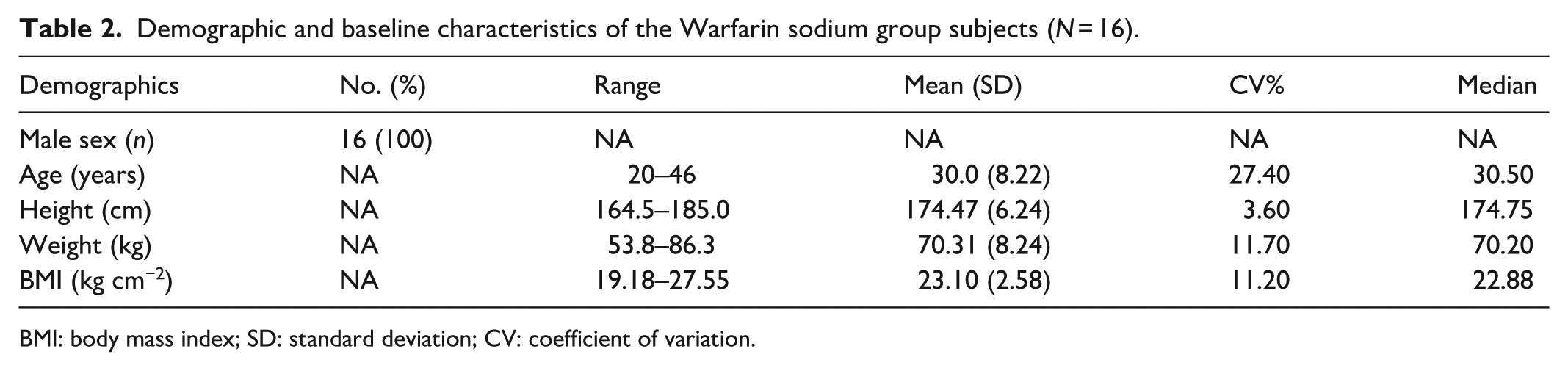
BMI: body mass index; SD: standard deviation; CV: coefficient of variation.
PK parameters
The
Pharmacokinetic parameters of Digoxin tablets/Digoxin tablets + PB-119 injection in healthy subjects.

PK: pharmacokinetics; AUC: area under the curve; CI: confidence interval.
One subject discontinued the subsequent trial medication and withdrew from the trial due to a Serious Adverse Event (SAE).
Geometric mean ratio. PB-119 dosage: 150 μg, subcutaneous injection.
Pharmacokinetic parameters of Warfarin sodium tablets/Warfarin sodium tablets + PB-119 injection in healthy subjects.

PB-119 dosage: 150 μg, subcutaneous injection. PK: pharmacokinetics; AUC: area under the curve; CI: confidence interval.

Average plasma Digoxin concentration–time curve

Mean plasma concentration–time curve of S-Warfarin.

Mean plasma concentration–time curve of R-Warfarin.
PD parameters
The INRAUC0–last for Warfarin alone was 166.72 ± 8.076 h, while for PB-119 + Warfarin, it was approximately 174.52 ± 7.12 h, indicating an increase of about 4.7% in INR AUC. However, the confidence interval was entirely within the 80%–125% range, suggesting no clinically significant interaction. Both INRmax and INR
Pharmacodynamic parameters of Warfarin sodium tablets/Warfarin sodium tablets + PB-119 injection in healthy subjects.

PD: pharmacodynamics; AUC: area under the curve; INR: international normalized ratio.
Safety assessment
In the Digoxin group and the Warfarin sodium group, 16 subjects were randomly assigned to each group. In the Digoxin group, 15 subjects (93.8%) experienced a total of 51 treatment-emergent adverse events (TEAEs) related to PB-119 injection and 57 TEAEs related to Digoxin tablets. Only one subject (6.3%) withdrew from the trial due to a serious adverse event (haemorrhagic erosive gastritis) after the first injection of PB-119 injection. The severity of the remaining subjects was mild to moderate. According to the MedDRA system organ classification, the TEAEs were mainly systemic diseases, various neurological diseases, metabolic and nutritional diseases, and various examinations. By prothrombin time classification, the TEAEs occurred in the following order: nausea (13 cases, 81.3%), abdominal distension (9 cases, 56.3%), vomiting (6 cases, 37.5%), taste disorder (4 cases, 25.0%), etc.
In the Warfarin sodium group, 16 subjects (100%) experienced a total of 67 TEAEs related to PB-119 injection, and 15 subjects (93.8%) experienced a total of 62 TEAEs related to Warfarin sodium tablets. All TEAEs were of mild to moderate severity. According to the MedDRA system organ classification, the TEAEs were mainly gastrointestinal diseases, metabolic and nutritional diseases, various neurological diseases, etc. The TEAEs occurred in the following order: abdominal distension (13 cases, 81.3%), hypoglycaemia (9 cases, 56.3%), nausea (5 cases, 31.3%), vomiting (4 cases, 25.0%), etc.
In this study, the adverse reactions of the subjects in the Digoxin group and the Warfarin sodium group after a single fasting oral administration of Digoxin tablets (0.25 mg)/Warfarin sodium tablets (2.5 mg) and PB-119 injection (150 μg) were all within the adverse reactions described in the instructions. This indicates that the safety and tolerability of multiple subcutaneous injections of PB-119 injection combined with a single oral administration of Digoxin tablets/Warfarin sodium tablets are good.
Discussion
According to the 2021 statistics of the International Diabetes Federation, diabetes has become one of the more serious and widespread chronic diseases in the current era. It is estimated that by the end of 2045, the prevalence of diabetes among Chinese adults aged 20–79 will rise to 12.5%.
19
Bydureon, compared with endogenous GLP-1 (7–37), is less likely to be rapidly degraded by dipeptidyl peptidase-4 inhibitors (DPP-IV) in the body, but its half-life is still relatively short (
Digoxin is a commonly used drug for treating heart failure and other diseases. The main influencing factor of Digoxin metabolism in the body is the level of drug transport protein (P-glycoprotein (P-gP)). 20 When Digoxin is co-administered with other drugs, it is excreted through the kidneys mediated by P-gP, and at the same time, it promotes the absorption process in the small intestine. When Digoxin is used in combination with P-gP inhibitors or inducers and other drugs, the expression activity of P-gP in the intestine may change, thereby affecting the bioavailability of Digoxin.13,21 Therefore, when Digoxin is used in combination with other drugs, its PK and PD parameters are easily disturbed, and the blood concentration of Digoxin needs to be monitored in a timely manner. Warfarin sodium is a major anticoagulant drug in clinical practice, and its metabolism in the liver mainly depends on liver microsomal enzymes, including CYP2C9 (mainly metabolizing S-Warfarin sodium), CYP1A2, and CYP3A (mainly metabolizing R-Warfarin sodium), ultimately converting Warfarin sodium into inactive metabolites. Therefore, drugs involving vitamin K epoxide reductase complex 1 (VKORC1) and liver drug-metabolizing enzymes, including CYP2C9, will affect the anticoagulant effect of Warfarin sodium. 22 In summary, diabetic patients with heart failure or those requiring Warfarin sodium for anticoagulation need to consider the interaction when using exenatide injection in combination with Digoxin/Warfarin sodium preparations.
Soon et al. studied the pharmacokinetic and pharmacodynamic effects of exenatide (25 mg) on Warfarin sodium tablets in healthy Asian men.
23
The results showed that all PK parameters were higher than those in the Warfarin sodium group of this study, possibly due to the higher dose used in that study; however, formal evidence for dose-proportional PK of Exenatide within this range is lacking, and a contribution of non-linear clearance cannot be excluded. Other teams also studied the pharmacokinetic effects of GLP-1RA, including semaglutide (1.0 mg),
24
albiglutide (50 mg),
25
and dulaglutide (50 mg),
26
on Digoxin and Warfarin sodium in healthy subjects. The results showed that the
Although this study did not detect a significant effect of PB-119 on the PK/PD parameters of Warfarin, the potential pharmacological sensitivity warrants in-depth discussion. Since the research mainly relied on biomarkers and clinical observation indicators, these objective indicators were not pre-tested through traditional questionnaires either. Warfarin’s anticoagulant response exhibits considerable interindividual variability, primarily influenced by polymorphisms in the CYP2C9 and VKORC1 genes. 18 Alleles such as CYP2C9*2 and CYP2C9*3 result in reduced enzyme activity, thereby slowing the clearance of S-warfarin.27,28 Meanwhile, the VKORC1 −1639G>A polymorphism affects the expression level of VKORC1. 28 Patients carrying these variant alleles exhibit heightened sensitivity to warfarin and typically require lower doses to achieve the same anticoagulant effect. Although the population-level averages in this study revealed no significant interaction, individual patients may possess different susceptibility profiles. Moreover, GLP-1RAs may indirectly influence warfarin sensitivity through multiple mechanisms. Firstly, GLP-1RAs have been shown to improve vascular endothelial function, which could modulate sensitivity to anticoagulants by affecting tissue factor expression and coagulation factor synthesis.13,29 Secondly, GLP-1RAs possess anti-inflammatory properties30,31 and may influence the coagulation–inflammation interplay by reducing systemic inflammation, thereby indirectly altering Warfarin’s anticoagulant efficacy. Thirdly, delayed gastric emptying induced by GLP-1RAs 32 may alter gut microbiota composition, thereby affecting endogenous vitamin K synthesis and intestinal absorption, potentially leading to changes in warfarin sensitivity. Similarly, Digoxin has a narrow therapeutic index, meaning that even minor fluctuations in plasma concentration may lead to toxicity or therapeutic failure. GLP-1RAs have been reported to increase heart rate, 33 an effect potentially mediated by direct activation of GLP-1 receptors in the sinoatrial node or via neuroendocrine pathways. In contrast, Digoxin exerts negative chronotropic and dromotropic effects. The potential interaction between these two agents in heart rate regulation warrants careful monitoring in clinical practice, particularly in patients with pre-existing arrhythmias or conduction system disorders. Digoxin is primarily eliminated via renal excretion, while PB-119 is also cleared renally. Although this study did not reveal significant pharmacokinetic interactions, clearance of both drugs may be reduced in patients with renal impairment, necessitating close monitoring of Digoxin plasma concentrations.
These mechanisms were not evident in the short-term setting of the present trial, they may carry clinical relevance in patients receiving long-term PB-119 therapy. Therefore, even in the absence of significant population-level changes in PK/PD parameters, individualized monitoring for alterations in warfarin or Digoxin sensitivity remains essential in subsequent clinical practice.
However, a major limitation of most studies on the PK and PD parameters of GLP-1RA in combination with Digoxin/Warfarin sodium is that the majority of the enrolled subjects are healthy male subjects, while the main target population for GLP-1RA is diabetic and obese patients, who may have comorbidities that affect PK results.
34
There are relatively few research teams focusing on this area. There are also studies indicating that Exenatide prolonged the gastric half-emptying time in all diabetic patients without gastroparesis (
Conclusion
Based on the main pharmacokinetic and pharmacodynamic parameters, it can be concluded that the absorption rate and extent of Digoxin tablets/Warfarin sodium tablets alone in the body are comparable to those when combined with PB-119 injection. The 90% CIs of the geometric mean ratios of the main pharmacokinetic parameters (
Supplemental Material
sj-docx-1-smo-10.1177_20503121261424728 – Supplemental material for A study to evaluate the impact of PB-119 injection (a pegylated exenatide formulation) on the pharmacokinetic profiles of Digoxin and Warfarin sodium in healthy subjects
Supplemental material, sj-docx-1-smo-10.1177_20503121261424728 for A study to evaluate the impact of PB-119 injection (a pegylated exenatide formulation) on the pharmacokinetic profiles of Digoxin and Warfarin sodium in healthy subjects by Yang Yu, Wang Hu, Tonghao Zhang, Yu Peng, Jiaxiang Ding, Xiaoni Wang, Hongwei He, Daolei Zhou, Dongshuang Han, Jie Cao, Ning Cheng, Jinmei Zhou and Huan Zhou in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to express our gratitude to the subjects who participated in this trial, as well as the clinical, laboratory and pharmacy staff of the First Affiliated Hospital of Bengbu Medical University. We are grateful for their outstanding support.
Ethical considerations
The questionnaire and methodology for this study was approved by the Human Research Ethics committee of the University of Bengbu Medical University First Affiliated Hospital.
Consent to participate
The study was performed in accordance with the ethical standards as laid down in the Declaration of Helsinki and its later amendments or comparable ethical standards. All subjects involved filled out the written informed consent form. All protocols, amendments, and consent forms were reviewed by the Institutional Ethics Committee (EC). The clinical trial registration number is: CTR20221249.
Consent for publication
The informed consent form has informed the subjects that all data will support the publication of research results, and research information will be legally authorized, but the confidentiality of personal information will not be violated.
Author contributions
Yang Yu, Wang Hu, Tonghao Zhang, and Yu Peng contributed equally as co-first authors and were responsible for study conduct, data collection, and drafting of the manuscript. Jiaxiang Ding and Xiaoni Wang performed the pharmacokinetic and statistical analyses. Hongwei He, Daolei Zhou, Dongshuang Han, Jie Cao, Ning Cheng, and Jinmei Zhou supervised the clinical trial, ensured regulatory compliance, and critically reviewed the manuscript. Huan Zhou conceived and designed the study, secured funding, and served as the principal investigator; she also revised and finalized the manuscript for intellectual content. All authors read and approved the final version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Project of Natural Science Research in Universities of Anhui Province, China (under Grant number 2023AH040406); 2024 Key Project of Natural Science Research in Universities of Anhui Province, China (under Grant number 2024AH051141); and Longhu Talent Project of Bengbu Medical University of Anhui Province, China (under Grant number LH250101002).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
