Abstract
Background
Acute ischemic lesions are challenging to detect by conventional computed tomography (CT). Virtual monoenergetic images may improve detection rates by increased tissue contrast.
Purpose
To compare the ability to detect ischemic lesions of virtual monoenergetic with conventional images in patients with acute stroke.
Material and Methods
We included consecutive patients at our center that underwent brain CT in a spectral scanner for suspicion of acute stroke, onset <12 h, with or without (negative controls) a confirmed cortical ischemic lesion in the initial scan or a follow-up CT or magnetic resonance imaging. Attenuation was measured in predefined areas in ischemic gray (guided by follow-up exams), normal gray, and white matter in conventional images and retrieved in spectral diagrams for the same locations in monoenergetic series at 40–200 keV. Signal-to-noise ratio (SNR) and contrast-to-noise ratio (CNR) were calculated. Visual assessment of diagnostic measures was performed by independent review by two neuroradiologists blinded to reconstruction details.
Results
In total, 29 patients were included (January 2018 to July 2019). SNR was higher in virtual monoenergetic compared to conventional images, significantly at 60–150 keV. CNR between ischemic gray and normal white matter was higher in monoenergetic images at 40–70 keV compared to conventional images. Virtual monoenergetic images received higher scores in overall image quality. The sensitivity for diagnosing acute ischemia was 93% and 97%, respectively, for the reviewers, compared to 55% of the original report based on conventional images.
Conclusion
Virtual monoenergetic reconstructions of spectral CIs may improve image quality and diagnostic ability in stroke assessment.
Keywords
Introduction
Making an early and accurate diagnosis by identifying the ischemic lesion is essential for the treatment and outcome of stroke (1,2). Computed tomography (CT) is one of the most widely used imaging modalities for diagnosing acute brain infarction (3,4). However, the appearance of early ischemic changes is known to be subtle and difficult to detect on conventional polyenergetic CIs (1,5) since signs of ischemia such as gyral effacement and the reduction of gray-white matter differentiation may be very discreet in the initial stage. Image interpretation may be further limited by beam hardening artifacts caused by adjacent dense material (6). The role of the initial CT is to rule out hemorrhage, and to identify the size and the location of any early ischemic lesions that may affect treatment, level of care, and further stroke work-up (7). Improved rates of detection of ischemic lesions in the patient with acute stroke would therefore be of value.
Several advancements in CT technology, such as multidetector-row CT (8,9), wide detector CT (10), iterative reconstruction techniques (11), and recently artificial intelligence applications (12,13) have improved diagnostic ability. Another landmark innovation is dual-energy CT (DECT) (14) with reconstruction of virtual monoenergetic images (VMIs) using the specific attenuation properties of different materials at different energy levels. The use of VMIs have been shown to reduce image artifacts and improve soft tissue contrast in both healthy brains and established brain infarcts compared to conventional CIs (CIs) (15–20). The use of material decomposition images generated from DECT has been shown to exhibit improved sensitivity and specificity for detecting early ischemic lesions compared to CIs (21).
A novel DECT solution based on a dual-layer detector (DLCT), with a top and bottom layer sensitive to high and low energy photons, respectively, may provide several advantages compared to other DECT technologies. These advantages include reduced sensitivity to patient motion, access to a wide spectrum of virtual monoenergetic images at postprocessing from a standard acquisition protocol, requires no preplanning, and a reduction in beam hardening artifacts without increasing radiation dose or examination time. The technology of DECT and DLCT and the potential advantages of DLCT have been described, for example by McCollough et al. (22) and Große Hokamp et al. (23).
Therefore, the aim of the present study was to quantitatively and qualitatively evaluate whether the ability to detect acute cortical ischemic lesions is improved in DLCT virtual monoenergetic image reconstructions compared to conventional polyenergetic CIs.
Material and Methods
Study population
We performed a retrospective search in our Picture and Archiving Communication System (PACS) for patients who had undergone non-contrast brain CT using an IQon Spectral CT (Philips Healthcare, The Netherlands) between 1 January 2018 and 27 July 2019. The following criteria needed to be met for inclusion in the study: (i) clinical suspicion of stroke; (ii) last seen well <12 h previously; (iii) finding of an acute, cortical ischemic lesion with an area measuring ≥70 mm2 on either the initial CT or on a follow-up CT or magnetic resonance imaging (MRI); (iv) available spectral data files from the initial CT; and (v) age ≥18 years.
Negative control patients were included for the same period and criteria except that no acute ischemic lesions nor any other identified pathology were to be present in either the initial CT or the follow-up CT or MRI.
National Institute of Health Stroke Scale (NIHSS) score at admission was noted for all patients with ischemic lesions. For patients with no NIHSS score recorded in the patient file, we assessed the score as >5 or ≥5 based on the described neurological status. For all patients with an ischemic lesion, it was noted in which hemisphere and lobe the lesion was located. In addition, the affected vessel (if known) and the maximum diameter of the ischemic lesion (measured in centimeters and rounded off to the closest 0.5 value) in axial plane images of the follow-up exam or the initial scan (only if there was no follow-up exam) were noted. The present study was approved by the Swedish Ethical Review Authority (reference no. 2019-02225) and the requirement for informed consent was waived.
Image acquisition and reconstruction parameters
All scans were performed in the same IQon Spectral CT (Philips Healthcare, The Netherlands) and according to the same scan parameters: tube voltage = 120 kV; tube current modulation (Dose Right, Z-DOM, Dose Right Index 37); collimation = 64 × 0.625; pitch = 0.36; and rotation time = 0.33 s. All images were then saved in PACS. For the purpose of this study, the images were retrieved from PACS, and dose length product (DLP), milliampere-seconds (mAs), and computed tomography dose index (CTDI)) were noted. The effective dose was calculated by multiplying DLP with a conversion factor of 0.0024 mSv/mGycm (24). The images were then transferred to ISP v. 10.1.4.21403 software (IntelliSpace Portal, Philips Healthcare, The Netherlands), where images with a slice thickness of 3 mm were reconstructed and all quantitative data retrieval was performed.
Quantitative analysis
Regions of interest (ROIs) were then manually drawn in the locations below, one ROI per location, using the conventional images of the initial CT exam (Fig. 1).
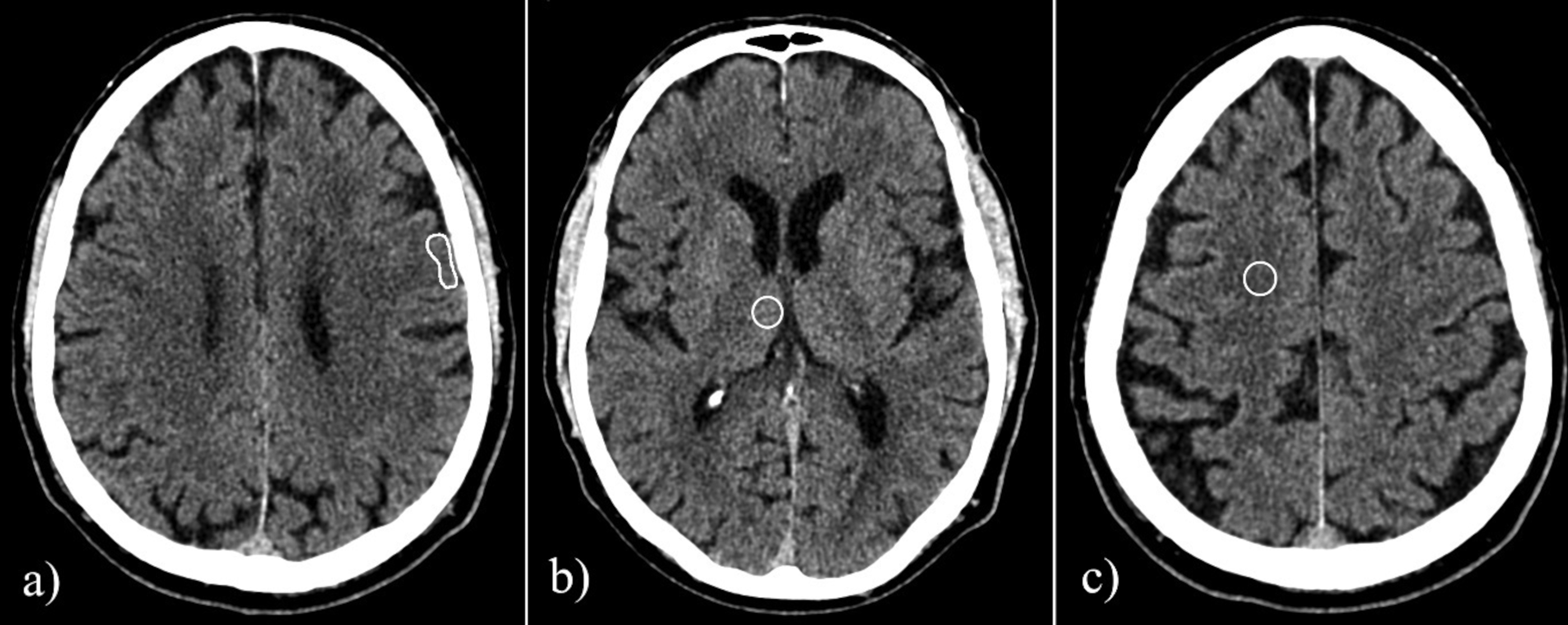
Example of placement of regions of interest in (a) ischemic gray matter, (b) normal gray matter in the thalamus, and (c) in normal white matter. The follow-up CT image and a set of virtual monoenergetic images of the same patient are shown in Fig. 2. CT, computed tomography.
Ischemic gray matter (IGM): The ROI was manually drawn in one image slice guided by findings on the follow-up CT or MRI and shaped to include the ischemic cortex only and to a size of 80 ± 10 mm2 (ROI area calculated by ISP software).
Normal gray matter (NGM): A circular ROI measuring 10 ± 1 mm in diameter was drawn in the thalamus on either side. We chose this placement in the thalamus instead of, for example, in contralateral cortical gray matter in order to make the ROI of a standardized and fair size without the risk of partial volume effect.
Normal white matter (NWM): A circular ROI measuring 10 ± 1 mm in diameter was drawn between the vertex and the lateral ventricle in either hemisphere.
The ROI measurements were performed by a resident in radiology; in any case, with uncertainty of the measurement placement the case was discussed with a senior neuroradiologist. The mean and standard deviation (SD) attenuation measured in Hounsfield units (HU) were noted for each conventional image ROI and then retrieved through spectral diagrams using ISP software for VMIs at 40–200 keV with intervals of 10 keV.
Signal-to-noise ratio (SNR) was calculated as HU/SD, where HU indicates the mean attenuation of the tissue (measured in Hounsfield units), and SD the standard deviation of the tissue, within the ROI.
Contrast-to-noise ratio (CNR) was calculated for IGM-NWM, NGM-IGM, and NGM-NWM as (HU1– HU2)/√(SD1 + SD2), where HU1 indicates the mean of tissue 1 (in Hounsfield units), HU2 the mean of tissue 2 (in Hounsfield units), SD1 the standard deviation of tissue 1, and SD2 the standard deviation of tissue 2, within the ROI, in line with previous studies (6,20). The differences in attenuation values (ΔHU) were calculated for IGM-NWM, NGM-IGM, and NGM-NWM as HU1 – HU2.
Qualitative analysis
Qualitative analysis was performed by visual grading of image quality by independent reviewers, blinded to patient data, follow-up imaging, and to information regarding the type of image reconstruction. All images were displayed on standard diagnostic monitors (Coronis® Fusion MDCC-6430 6MP; Barco, Kortrijk, Belgium). CIs and VMIs at 50, 60, and 70 keV were included in the qualitative analysis based on previous work (17,19,20). The qualitative analysis was performed in two steps:
First, CIs and VMIs at 50–70 keV were assessed for a general impression of image quality using Viewdex (Viewer for Digital Evaluation of X-ray images) version 2.272 software (25,26), which allows visualization in an axial CT stack with standard window settings (width = 65, level = 35) and the liberty to adjust window settings. CIs and VMIs were randomly presented to the reviewers for grading using the Viewdex software. No clinical details regarding symptomatology were provided to the reviewers. Grading was performed by two independent neuroradiologists (JW and MD) with >5 years of experience. Before the evaluation, the reviewers were given example cases to assess in order to guide them on grading scale requirements. These example cases were not included in the cohort of the present study. The following image quality parameters were assessed: (i) overall impression of image quality; and (ii) impression of image ability of diagnosing acute ischemia. These parameters were rated on a 5-point Likert scale (1 = non diagnostic, 2 = poor, 3 = fair, 4 = good, 5 = excellent) in line with previous studies (19,27,28).
For the second part, control cases without ischemic lesions, and no other identified pathology, in follow-up imaging were added to the analysis. The grading was performed by one radiology resident and one experienced neuroradiologist (AH and PU). All image series (CIs and VMIs at 50, 60, and 70 keV) for each patient, both the original cohort and negative controls, were assessed by each reviewer who was asked to identify: the presence of ischemic lesion (yes or no); infarct location (if an ischemic lesion was assessed as present); and which reconstruction could best visualize ischemic lesions. For each patient, the reviewers had access to axial stacks of all four reconstructions (CIs and VMIs at 50, 60, and 70 keV) placed side-by-side and a moderator (an experienced interventional neurologist) provided clinical information on the original clinical referral. The reviewers were still blinded to image reconstruction type. The moderator noted the reviewer's replies and assessed the reviewer's identified infarct location as correct or incorrect by comparing the reviewer-identified infarct location to the location of the ischemia on the follow-up MRI or CT (Fig. 2).
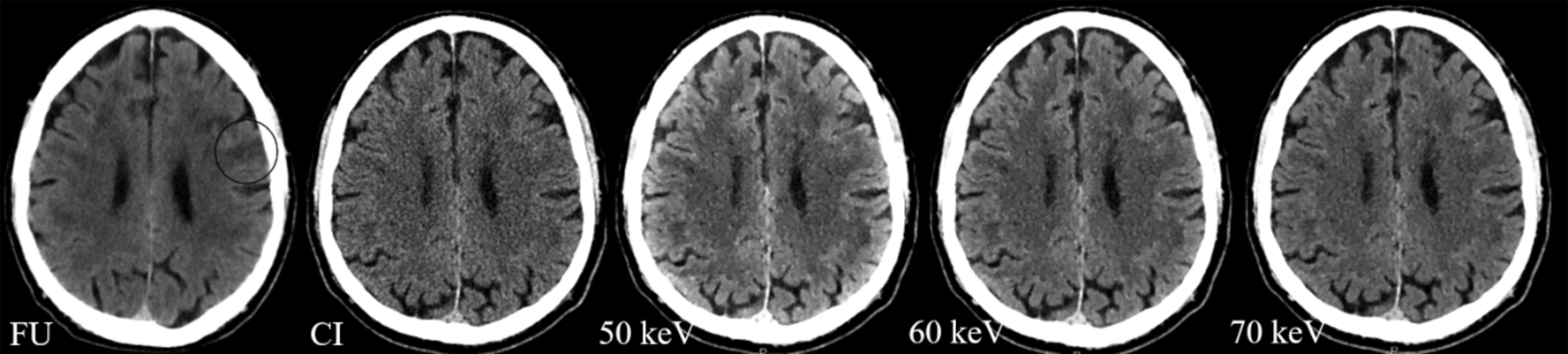
Example images of a patient with a left-sided frontal ischemic lesion (circle) in the follow-up examination one day after onset (FU), followed by an acute exam conventional image and virtual monoenergetic reconstructions at 50, 60, and 70 keV.
The infarct location stated in the original radiology report (based on only the CIs, all assessed or reassessed by a neuroradiologist) was also compared to the final ischemic lesion on the follow-up MRI or CT and noted as correct or incorrect.
Two-by-two tables were created to calculate diagnostic measurements of CIs alone (based on the original report) and for CIs in combination with VMIs at 50, 60, and 70 keV.
Statistical analysis
Quantitative data are presented as median (interquartile range [IQR]), and qualitative data as median ± (95% confidence interval [CI]) or quantity (%). A Wilcoxon signed rank test was used to compare non-parametric dependent continuous and ordinal data, a chi-square test was used to compare nominal non-parametric data, and a Mann–Whitney U test was performed to compare independent continuous non-parametric data. P values ≤0.05 were considered statistically significant and the statistical calculations were performed using SPSS Statistics for Windows version 25.0 (IBM Corp., Armonk, NY, USA). Inter-rater agreement was calculated as a weighted Cohen's kappa coefficient using MedCalc v. 19.3.1 software (MedCalc, Ostend, Belgium) and interpreted as suggested by Altman (29).
Results
Study population characteristics
Patient characteristics are presented in Table 1. A total of 29 patients with acute ischemic lesions and 23 negative control cases were included in the study. All ischemic lesions were supratentorial but involving different vascular territories (Table 1). There were significant differences in sex and effective dose but not in age between the studied cohort and the controls (Table 1).
Patient and lesion characteristics in 29 patients presenting with sudden onset of focal neurologic deficits and ischemic lesions with cortical involvement on follow-up brain imaging, and 23 negative control patients with sudden onset of focal neurological deficits but with no ischemic lesions on neither the initial CT or the follow-up CT or MRI.
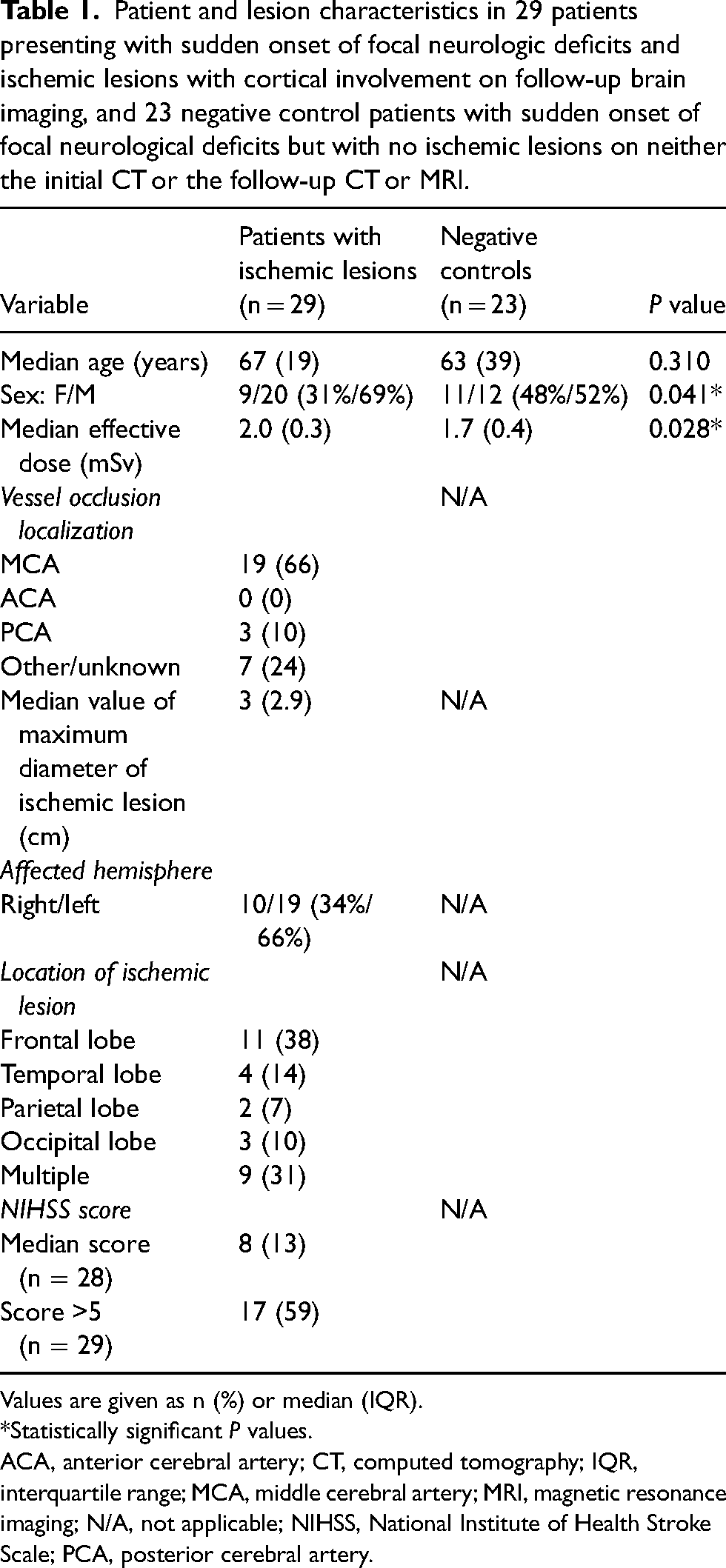
Values are given as n (%) or median (IQR).
*Statistically significant P values.
ACA, anterior cerebral artery; CT, computed tomography; IQR, interquartile range; MCA, middle cerebral artery; MRI, magnetic resonance imaging; N/A, not applicable; NIHSS, National Institute of Health Stroke Scale; PCA, posterior cerebral artery.
Quantitative analysis
Line graphs of median attenuation values, SNR, and differences in attenuation for conventional images and monochromatic reconstructions can be found in the Supplemental Material (Supplemental Figure 1).
Attenuation values in predefined brain locations
Attenuation values for all ROIs are shown as boxplots in Fig. 3. The median attenuation values of all three ROIs generally decreased with higher energy level VMIs. The median attenuation values of IGM compared to NGM were significantly lower in CIs and in VMIs at 60–200 keV. The ΔHU of NGM-IGM was lower in VMIs at 40–80 keV compared to CIs, significantly so in 40–70 keV reconstructions (all P values <0.05).
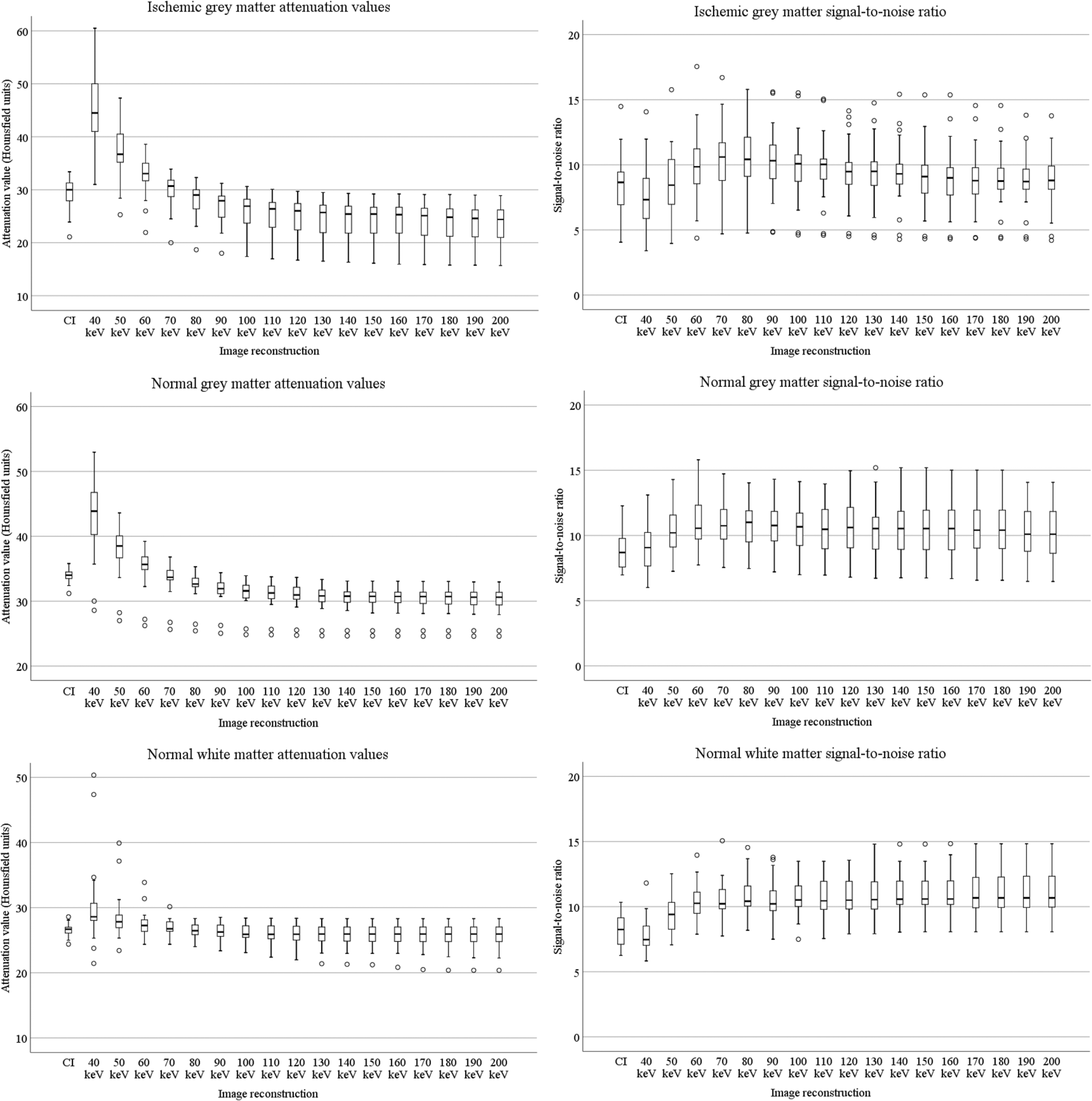
Boxplots of attenuation values (HU) and signal-to-noise ratio for ischemic gray matter, normal gray matter, and normal white matter for conventional images and monochromatic reconstructions at 40–200 keV.
SNR in predefined brain locations
SNR for all ROIs are shown as boxplots in Fig. 3. For IGM, the SNR was highest in VMIs at 70 keV (10.6, IQR = 3.2) and was significantly higher in VMIs at 60–150 keV than in the CIs. For NGM, the SNR was highest in VMIs at 80 keV (11.0 (IQR = 2.8)) and was significantly higher in VMIs at 50–200 keV compared to CIs. In normal white matter, SNR was highest in VMIs at 170–200 keV (10.7 (IQR 2.4, 2.4, 2.5, and 2.5, respectively) and was significantly higher in VMIs at 50–200 keV compared to CIs (Fig. 3).
CNR in predefined brain locations
Boxplots for CNR values are shown in Fig. 4. For IGM-NWM, CNR was greatest in VMIs at 40 keV (4.9 (IQR = 3.3)) and was significantly higher in VMIs at 40–70 keV than in CIs. For VMIs at energy levels >70 keV, the CNR values were significantly lower compared to the conventional images.
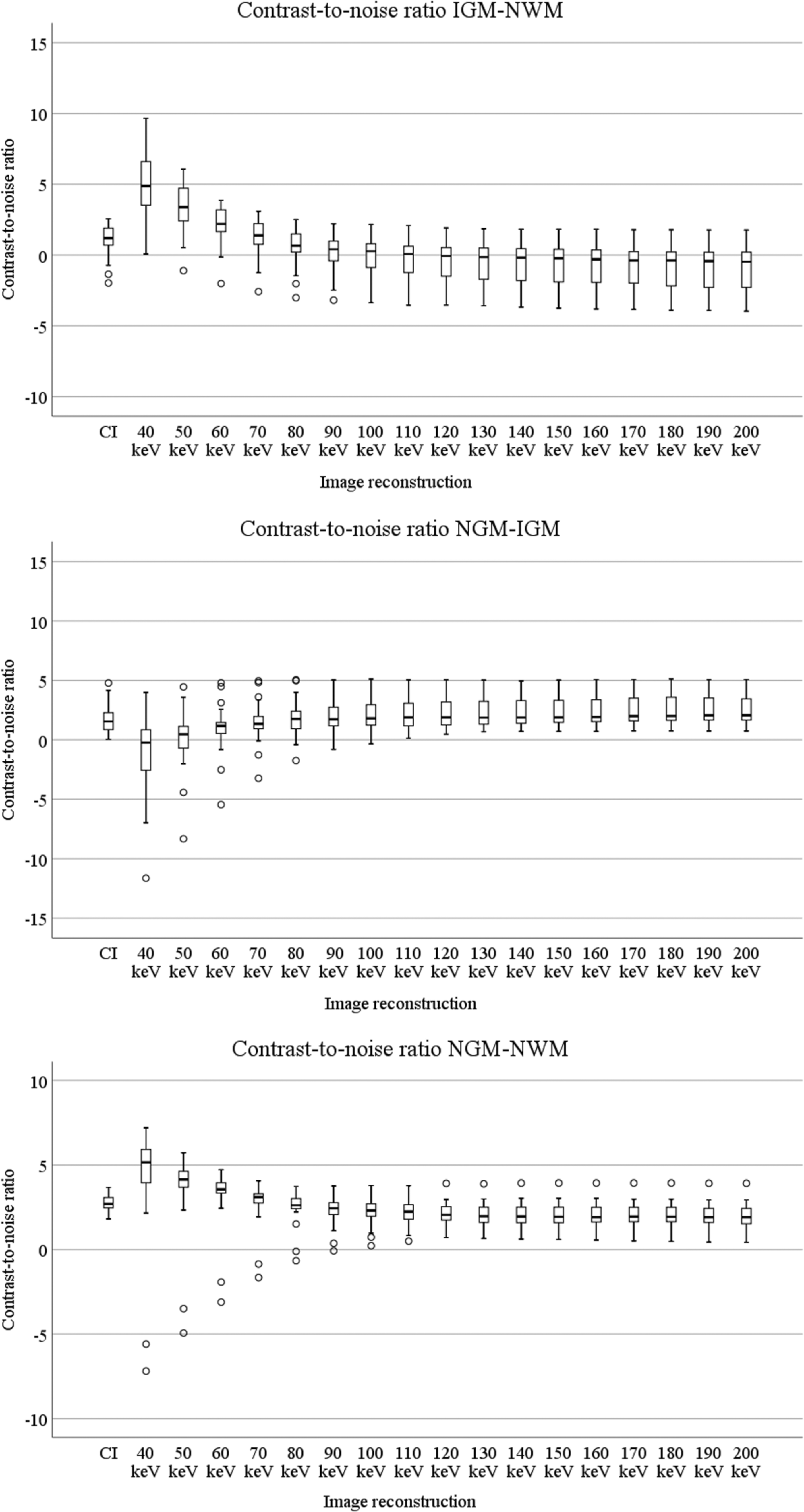
Contrast-to-noise ratio for IGM- NWM, NGM-IGM, and NGM-NWM for conventional images and virtual monoenergetic reconstructions at 40–200 keV. IGM, ischemic gray matter; NGM, normal gray matter; NWM, normal white matter.
For NGM-IGM, the CNR was highest in VMIs at 200 keV (2.1, IQR = 2.1). The CNR in VMIs at 40, 50, and 60 keV was significantly lower than in CIs but was significantly greater in VMIs at 80–200 keV than in CIs.
The CNR values for NGM-NWM were highest for VMIs at 40 keV (5.2, IQR = 2.1). The CNR was significantly higher in VMIs at 40, 50, 60, and 70 keV than in CIs, yet significantly lower in VMIs at 90–200 keV than in CIs.
Qualitative analysis
Overall impression of image quality
Reviewer 1 rated the VMIs at 50 keV and 60 keV the highest in overall image quality (4, 95% CI = 3.8–4.5 and 4, 95% CI = 3.7–4.4, respectively). Reviewer 2 rated the VMI at 60 keV the highest (4, 95% CI = 3.3–4.9). The scores were significantly higher in all VMIs compared to CIs (P < 0.001 for all) for both reviewers. The inter-rater agreement was moderate (Cohen's kappa coefficient = 0.43).
Impression of image ability of diagnosing acute ischemia
The highest diagnostic ability median rating score was 5 (95% CI = 4.2–4.8) at 50 keV and 4 (95% CI = 3.5–3.9) at 60 keV for reviewers 1 and 2, respectively. All VMIs received significantly higher scores compared to conventional images (P < 0.001 for all) for both reviewers. The inter-rater agreement was moderate (Cohen's kappa coefficient = 0.49).
Assessment of location of ischemia and optimal reconstruction
In total, 52 patients (29 cases with an ischemic lesion and 23 cases without) were included in this analysis. The number of correctly localized ischemic areas was 27/29 (93%) and 28/29 (97%) for reviewers 1 and 2, respectively. The number of correctly diagnosed ischemic lesions from the original radiology reports (based on the CIs alone) was 16/29 (55%). The sensitivity was 0.93 (95% CI = 0.77–0.99) and 0.97 (95% CI = 0.82–0.99) for reviewers 1 and 2, compared to 0.55 (95% CI = 0.36–0.74) for the original report. The specificity was, however, generally lower for the reviewers, at 0.82 (95% CI = 0.61–0.95) and 0.91 (95% CI = 0.72–0.99), compared to the original assessment based on only conventional images that had a specificity of 1.0 (95% CI = 0.85–1.0). The inter-rater agreement was very good (Cohen's kappa coefficient = 0.8). All diagnostic measurements are shown in Table 2.
Diagnostic performance of assessment of acute ischemic lesions in 29 patients with an acute ischemic lesion and 23 negative control cases with no acute ischemic lesions on follow-up imaging (total cases = 52).
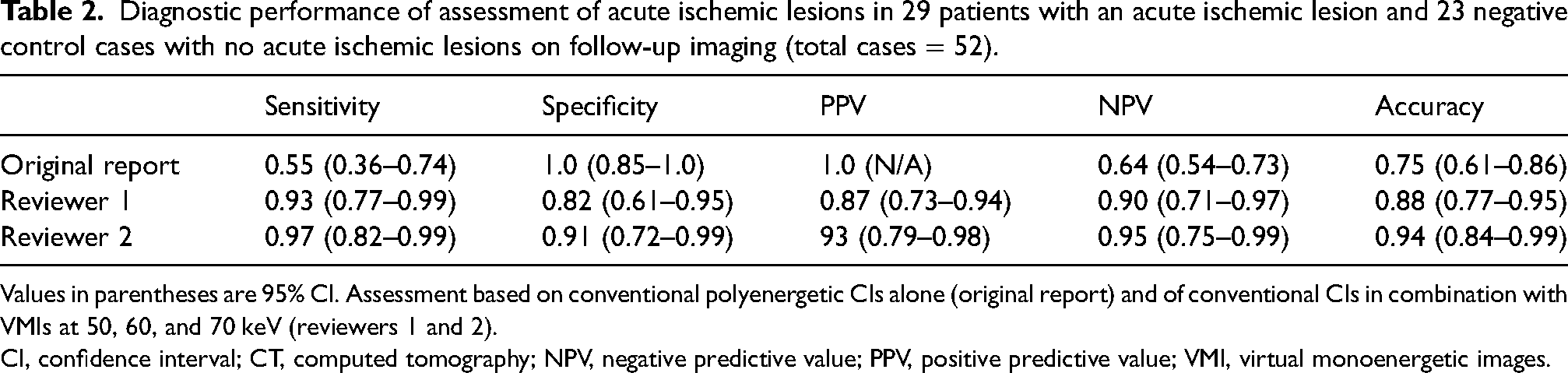
Values in parentheses are 95% CI. Assessment based on conventional polyenergetic CIs alone (original report) and of conventional CIs in combination with VMIs at 50, 60, and 70 keV (reviewers 1 and 2).
CI, confidence interval; CT, computed tomography; NPV, negative predictive value; PPV, positive predictive value; VMI, virtual monoenergetic images.
Reviewer 1 rated the reconstructions at 60 keV in 28 cases and the reconstructions at 50 keV in 19 cases as the optimal reconstruction. Reviewer 2 rated the conventional images as the optimal in 35 cases, and the reconstructions at 60 keV in nine cases (see Supplemental Figure 2 for a bar chart of assessed optimal reconstruction for both reviewers).
Discussion
In this retrospective study, we assessed whether acute cortical ischemic lesions could be more easily detected using a set of spectral virtual monoenergetic image reconstructions compared to conventional images.
Quantitatively, we found that SNR values were generally higher in VMIs compared to CIs, and that the CNR between IGM-NWM and NGM-NWM was significantly higher in VMIs at 40–70 keV compared to CIs. The higher SNR and CNR should theoretically be a result of decreased image noise and/or higher attenuation values, possibly due to a reduction in artifacts caused by the beam hardening effect and Compton scatter. These results are in accordance with previous studies by Neuhaus et al. and Pomerantz et al. (19, 30), who found generally higher SNR in all monoenergetic reconstructions and higher CNR in mainly lower energy reconstructions. The methods used to calculate the CNR tend to differ in the calculation of the noise value. For example, the standard deviation of the tissue, the sum of standard deviations, or, as used in the present study, the square root of the sum of the standard deviations have all been widely used in the literature (17,19,20,30,31). This is an important consideration when comparing CNR values between studies. Therefore, we chose the most commonly used method reported in previous CT imaging studies.
Qualitatively, we found that all VMIs received higher scores than CIs in overall image quality and subjective diagnostic ability of acute ischemia. The results also showed a higher ability to correctly diagnose acute ischemic lesions when grading CIs in combination with a set of monoenergetic reconstructions in the lower energy range (reconstructions at 50, 60, and 70 keV) compared to assessing CIs alone. The inter-rater agreement of assessing the presence of acute ischemic lesions was very good when using a set of CIs and VMIs together. We did not find one specific monoenergetic reconstruction to be superior compared to other VMIs.
At our comprehensive stroke center with a catchment area of 1.5 million people, all CT brain examinations performed off-hours are retrospectively reassessed by a neuroradiologist who may then alter the report. Hence, the improved rate of correctly diagnosed ischemic lesions cannot be explained by a difference in clinical experience, but an actual improvement in diagnostic properties of the images. The bias of a more thorough reading for this study compared to the reassessment of the original report cannot be entirely excluded. However, in a clinical setting, the radiologist has sagittal and coronal plane reconstructions available in addition to the axial plane, which could provide a better diagnostic potential than was available to the reviewers in this study. One of the reviewers, an experienced neuroradiologist, rated the CIs as the optimal for assessment of ischemic lesions in 35 of 52 (67%) cases, which may be an indication that VMIs should be used rather as a complement to CIs. This could, however, also, at least in part, be due to the fact that the practice of assessment of ischemic lesions is presently based upon evaluation of CIs, the type of images that radiologists are accustomed to evaluating. Nevertheless, the two reviewers, were able to correctly diagnose a higher proportion of acute ischemic lesions (sensitivity 93% and 97% for reviewers 1 and 2, respectively) using only axial plane CIs and monoenergetic reconstructions compared to the original radiology reports based on CIs alone (sensitivity = 55%). The specificity was lower in the assessment of CIs and VMIs (82% and 91% for reviewers 1 and 2, respectively), compared to the 100% specificity of the original report. This may at least partly be explained by the fact that the reviewers did not have access to previous scans or to sagittal or coronal reconstructions, resulting in the misperception of a chronic lesion for an acute.
The use of spectral CT technology in neuroradiology is currently being extensively investigated. So far, reports have shown or indicated improved gray/white matter contrast in lower VMI energy levels, and decreased metal artifacts in monoenergetic reconstructions at higher energy levels (17,19,32–34). Another benefit is the possibility of reconstructing virtual non-contrast images by selectively suppressing the attenuation by iodine (31,35). Regarding ischemia, our hypothesis is that increased gray/white matter contrast should be beneficial for identifying subtle early ischemic changes. This is one of the first studies to evaluate the use of spectral unenhanced CIs in the initial assessment of early ischemia. Van Ommen et al. recently evaluated the diagnostic ability of unenhanced spectral brain CT in patients with stroke and found that VMIs of 80 and 90 keV marginally improved infarct detection and location (36). However, Van Ommen et al. used dual-source DECT and in the present study we evaluated dual-layer detector DECT. Another difference is that we evaluated the sensitivity and specificity for assessment of early ischemia using a set of three VMIs together instead of evaluating them separately. In a study comparing different DECT technologies in abdominal imaging, Sellerer et al. (37) found that in dual-source DECT the image noise increases in lower keV reconstructions but for dual-layer detector DECT the noise remained low even in the low-energy reconstructions. This might explain our results of a higher diagnostic ability in lower keV-reconstructions compared to Van Ommen et al., since in dual-layer detector DECT the CNR is not decreased by high noise in low-energy reconstructions. The strengths of this study include using both quantitative and qualitative methods of assessment. Ståhl et al. assessed VMIs in early ischemic lesions in the anterior circulation using an automated ASPECTS scoring system and found VMIs at 70 keV provided the highest sensitivity, accuracy, and negative predictive value (38). However, the 70-keV reconstruction is the one corresponding most to CIs, which is what the ASPECTS algorithm is trained for. This might affect the findings of Ståhl et al.
The present study has some limitations. These include include a small cohort size, although the number was large enough to find statistically significant results. Other limitations were that the exact time from the onset of symptoms until CT exam was not available, which is important with respect to radiological signs of ischemia. All lesions were supratentorial involving cortical tissue, which is not entirely comparable to the day-to-day clinic. Lesions may, for example, also be located in the deep nuclei. We made this restraint since we hypothesized that the increased contrast between gray and white matter would be important. For the qualitative analysis, as CIs and three VMIs were assessed side-by-side, each reconstruction may have affected the interpretation of the others. However, in the clinical setting it is also possible to review more than one reconstruction at a time to gain more information and evaluate eventual findings. A confounding factor of subjective analysis is the possibility of a more experienced reader or a more thorough reading for the purpose of this study, compared to the reading of the original radiology report. Only the axial plane images were used in the present study, whereas sagittal and coronal plane images are widely used in the routine clinical setting and may contribute to better diagnostic capability. Thus, it is possible that the clinical diagnostic ability of acute ischemia could be even further improved with the addition of VMIs in the coronal and sagittal planes. The lesions evaluated were heterogenous in size and location, but all cortical and supratentorial. Therefore, future studies should aim for larger sample sizes, and allow for stratification by lesion size or location. In addition, a prospective approach may allow for more detailed clinical information, including the exact time of onset.
In conclusion, we found that monoenergetic reconstructions of spectral CIs may improve the diagnostic ability by enhancing the tissue contrast, and we believe that this can improve the radiological accuracy in acute stroke assessments. However, these results need to be confirmed in other cohorts.
Supplemental Material
sj-docx-1-acr-10.1177_02841851221130612 - Supplemental material for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke
Supplemental material, sj-docx-1-acr-10.1177_02841851221130612 for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke by Helena Mellander, Pär Bengtsson, Veronica Fransson, Birgitta Ramgren, Per Undrén, Mattias Drake, Kristina Ydström, Jimmy Lätt, Amir Hilal, Johan Wassélius and Teresa Ullberg in Acta Radiologica
Supplemental Material
sj-xls-2-acr-10.1177_02841851221130612 - Supplemental material for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke
Supplemental material, sj-xls-2-acr-10.1177_02841851221130612 for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke by Helena Mellander, Pär Bengtsson, Veronica Fransson, Birgitta Ramgren, Per Undrén, Mattias Drake, Kristina Ydström, Jimmy Lätt, Amir Hilal, Johan Wassélius and Teresa Ullberg in Acta Radiologica
Supplemental Material
sj-xls-3-acr-10.1177_02841851221130612 - Supplemental material for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke
Supplemental material, sj-xls-3-acr-10.1177_02841851221130612 for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke by Helena Mellander, Pär Bengtsson, Veronica Fransson, Birgitta Ramgren, Per Undrén, Mattias Drake, Kristina Ydström, Jimmy Lätt, Amir Hilal, Johan Wassélius and Teresa Ullberg in Acta Radiologica
Supplemental Material
sj-docx-4-acr-10.1177_02841851221130612 - Supplemental material for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke
Supplemental material, sj-docx-4-acr-10.1177_02841851221130612 for Virtual monoenergetic images by spectral detector computed tomography may improve image quality and diagnostic ability for ischemic lesions in acute ischemic stroke by Helena Mellander, Pär Bengtsson, Veronica Fransson, Birgitta Ramgren, Per Undrén, Mattias Drake, Kristina Ydström, Jimmy Lätt, Amir Hilal, Johan Wassélius and Teresa Ullberg in Acta Radiologica
Footnotes
Acknowledgements
Lee Nolan is gratefully acknowledged for professional language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and material
Requests to access an anonymized dataset supporting the conclusions of this article may be sent to the corresponding author after obtaining the appropriate ethics approval.
Code availability
Requests to access an anonymized dataset/code supporting the conclusions of this article may be sent to the corresponding author after obtaining the appropriate ethics approval.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Crafoord Foundation (grant no. 20180610), STINT (The Swedish Foundation for International Cooperation in Research and Higher Education, grant no. IB2018-7532), The Swedish Stroke Association, Skåne University Hospital and Region Skåne (grant nos. I-ALF 47447 and YF-ALF 43435) to JW.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
