Abstract
The positron emission tomography radioligand [11C]UCB-J binds to synaptic vesicle glycoprotein 2 A (SV2A), a regulator of vesicle release. Increased neuronal firing could potentially affect tracer concentrations if binding site availability is altered during vesicle exocytosis. This study assessed whether physiological brain activation induces changes in [11C]UCB-J tissue influx (
Introduction
Synaptic density can be assessed in vivo using the positron emission tomography (PET) radioligand [11C]UCB-J, which binds to the synaptic vesicle glycoprotein 2 A (SV2A). 1 Despite a growing body of literature demonstrating the utility of [11C]UCB-J to assess synaptic loss or dysfunction in clinical populations,1–5 questions remain about the interpretation of signals from SV2A-binding radiotracers. SV2A is expressed ubiquitously at synapses throughout the mammalian brain and plays a crucial role in normal synaptic function.6–8 [11C]UCB-J in vivo binding measures show excellent agreement with in vitro quantification of SV2A and synaptophysin, 1 another common synaptic marker protein. However, several factors in addition to the number of synapses in a given brain region could influence [11C]UCB-J binding. For example, while SV2A is expressed at a consistent level of approximately five copies per vesicle, 9 variation in the number of vesicles per synapse could affect tracer binding in the absence of differences in synaptic density itself. Number of synaptic vesicles is closely linked to capacity for neurotransmitter release, 10 suggesting that signals from SV2A-bindingra diotracers may reflect, to some degree, the functional capacity of synapses as well as their density. The potential effects of dynamic vesicle release processes on tracer binding remain to be determined.
Neurotransmitter release through synaptic vesicle exocytosis is an active and tightly regulated process involving numerous biochemical mediators of vesicle trafficking, filling, priming, docking at the plasma membrane, fusion, release, and recycling. 11 , 12 Loss of SV2A leads to impairments in neurotransmission, 13 but its precise functional role in these processes is not yet clear. SV2A appears to be important in regulating vesicle exocytosis triggered by Ca2+ influx, in part through interactions with synaptotagmin or regulation of presynaptic Ca2+ levels. 7 , 14 , 15 Accordingly, SV2A can be presumed to have varying binding partners, 16 physical conformations, 17 or electrostatic properties 18 at different stages of the vesicle release process. Each of these could affect accessibility or affinity of the [11C]UCB-J binding site, which would result in variation in radiotracer binding measures related to the level of synaptic activity during scanning.
Regional changes in cerebral blood flow associated with heightened neuronal activity underlie the use of [15O]water PET and functional magnetic resonance imaging (fMRI) to measure brain response to stimuli including sensory, motor, or cognitive challenges. In PET studies assessing protein targets, differential effects of tracer delivery and binding can be accurately separated using kinetic modeling. Previous PET studies using neuroreceptor-binding radiotracers have demonstrated that changes in blood flow only are reflected in the tissue influx constant,
Stability of [11C]UCB-J binding is a crucial consideration in research and clinical applications. Activity-related changes in binding would mean that differences in brain state at the time of scan could induce an effect on [11C]UCB-J binding measurements in the absence of true differences in synaptic density. This would introduce a potential confound or source of variability, making it more difficult to identify true differences in synaptic density. The objective of this study was to assess whether physiological brain activation alters [11C]UCB-J binding measures. We hypothesized that during a visual activation task, increased neuronal activity would lead to higher
Materials and methods
Effects of brain stimulation on [11C]UCB-J tissue delivery and binding were compared in a two-scan baseline and activation paradigm using 8 Hz checkerboard visual stimulation to induce spatially restricted neuronal activation.
22
Previous studies using fMRI blood oxygen level dependent (BOLD) signal, cerebral blood flow measures, and EEG have found that neuronal and vascular responses to activation in the visual cortex habituate over time23–26 but that this effect is typically less pronounced with repeated intermittent blocks of stimulation compared to sustained stimulation in blocks longer than 2–5 min.24–26 Therefore, to achieve predictable, sustained responses across the 60-min scan length, visual stimulation was presented in 12 repeating 5-min blocks, each comprising a stimulation and a rest period. Given this intermittent stimulation, simulations were performed to evaluate sensitivity to time-varying changes in
PET simulations
[11C]UCB-J data, under steady state conditions, have been shown to be well-described by the one-tissue compartment model (1TCM).
27
To assess the impact of non-steady state conditions, PET data with time-varying rate constants were simulated and estimates of
Using these time-varying
Experimental study design
Seven healthy participants (three men and four women; mean age 37.6 ± 10.8 years) completed two PET scans and one MRI session with adequate compliance (see below). PET scans were completed on the same day separated by at least 4 h with scan order counter-balanced across subjects. MRI was acquired on a separate day before (
The visual stimulus consisted of an 8 Hz alternating black and white radial checkerboard filling the field of view with a static fixation cross at the center. This was alternated with off blocks consisting of a white fixation cross on black background for fMRI and with rest blocks comprising a static, low-contrast grey and black rectangular checkerboard for PET.
MRI
Visual activation
Stimulus was presented on a screen visible in the scanner via a mirror. Two fMRI runs were acquired with the checkerboard stimulus presented according to two timing patterns. First, to allow comparison between PET results and typical fMRI visual activation responses, one run was acquired using blocks of 30-s off, 30-s on for 6 min. To specifically assess the time course of the BOLD response during the sustained activation used in the PET study, a second scan was acquired using the chosen on-off pattern based on the simulation results, i.e. three blocks of 180-s on, 120-s off for a total of 15 min. Participants were instructed to press a key when the fixation cross in the center of the field of view changed color during each activation block to ensure they were watching the presentation on the screen. The two task sessions were separated by 12 min during which participants rested in the scanner with a black screen presented.
Acquisition
Structural and functional MRI scans were acquired on a 3.0 T Prisma Fit scanner (Siemens Medical Solutions). A high resolution, three-dimensional magnetization prepared rapid acquisition gradient echo (MPRAGE) T1-weighted sequence was first acquired for co-registration with PET images (TR = 2530 ms, TE = 2.81 ms, FOV = 256 × 256 mm2, matrix = 256 × 256 mm2, slice thickness = 1.0 mm without gap, 176 slices, voxel size 1.0 × 1.0 × 1.0 mm3). Functional runs included 360 or 900 whole-brain volumes acquired using a multiband echo-planar imaging sequence with the following parameters: TR = 1 s, TE = 29.6 ms, flip angle = 60°, matrix = 84 × 84, in-plane resolution = 2.5 mm2, 60 axial-oblique slices parallel to the ac–pc line, slice thickness = 2 mm, multiband = 4, acceleration factor = 2.
Analysis
Anatomical images were registered to the MNI152 template using non-linear transformations and segmented into grey and white matter with CAT12 (http://www.neuro.uni-jena.de/cat/). fMRI data were pre-processed and analyzed using SPM12 (Wellcome Trust Centre for Neuroimaging, http://www.fil.ion.ucl.ac.uk/spm/software/spm12/). The first 15 volumes of each functional run were discarded to allow magnetization to reach a steady state. Images were then motion-corrected, linearly registered to subject’s anatomical image, then registered to the template and spatially smoothed with an 8 mm FWHM Gaussian kernel. A temporal high-pass filter with cutoff frequency equal to twice the block length was applied to remove low-frequency drifts. A general linear model was then produced for each subject with one task regressor convolved with the hemodynamic response function. Individual time course and percent signal change during activation blocks was extracted using MarsBaR (http://marsbar.sourceforge.net/) from V1 (Brodmann area 17) as defined in the Talaraich Daemon Brodmann area atlas in PickAtlas software (http://fmri.wfubmc.edu/software/PickAtlas). To identify voxels where the BOLD signal increased during activation blocks, activation/rest contrast images for each subject were compared in a one-sample
PET
Visual activation
Stimuli were presented on video display glasses (z920VGHD, Zetronix, Boston MA) worn by the participant throughout both PET scans. During the activation scan, stimulation was initiated 30 s before the start of radiotracer injection, beginning with an ‘on’ block and continuing through the 60-min scan for a total of 12.1 5-min presentation cycles. As in fMRI, participants were instructed to press a key on a handheld keypad when the fixation cross in the center of the field of view changed color, which occurred at pseudorandom intervals of 3–6 min throughout the scan (15 occurrences total). One participant responded to less than 75% of these prompts and so was excluded from the analysis. The remaining seven participants responded to >90% of prompts. In the baseline PET scan, a static white cross on black background was shown throughout.
Acquisition
[11C]UCB-J was synthesized as previously described. 29 After positioning the video glasses, a 6-min transmission scan was performed for attenuation correction. [11C]UCB-J was then administered as a 1-min intravenous infusion. Dynamic PET data were acquired for 60 min on a high resolution research tomograph (HRRT; CTI/Siemens, Knoxville TN). Head motion was tracked using an optical detector (Vicra, NDI Systems, Waterloo, Ontario, Canada). Blood samples were drawn at intervals throughout the scan via an arterial cannula placed in the wrist of the subject’s nondominant hand. Radiometabolite analyses were performed using an automatic column-switching HPLC system as previously described. 29 The unmetabolized parent fraction was determined as the ratio of the radioactivity corresponding to the parent to the total amount of radioactivity collected. The parent fraction curve was fitted with an inverted integrated gamma function. Arterial plasma input function was calculated as the product of the total plasma activity, parent fraction curve, and extraction efficiency curve, which was determined from the ratio of filtered to original plasma concentrations fitted with an exponential function.
Analysis
Dynamic PET data were reconstructed with corrections for attenuation, normalization, scatter, randoms, deadtime and motion using the MOLAR algorithm
30
into 21 frames (6 × 30 s, 3 × 1 min, 2 × 2 min, 10 × 5 min). TACs were extracted from each ROI in the subject’s PET space to determine
Results
PET simulations
Three stimulation timing paradigms within the 5-min blocks were assessed. In all cases, model fits were good using the standard 1TCM (i.e. assuming constant
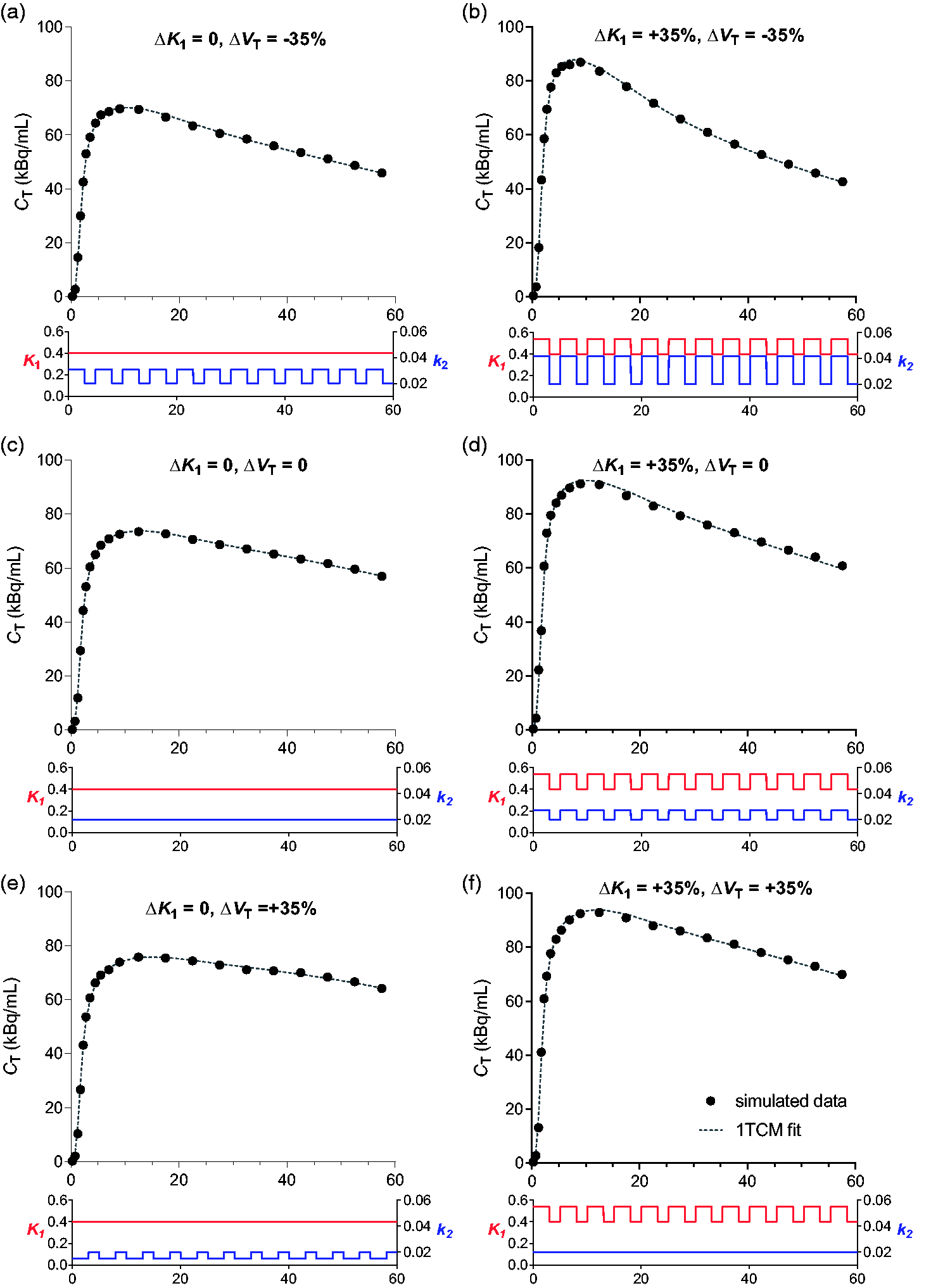
Simulated [11C]UCB-J time-activity curves with intermittent changes in
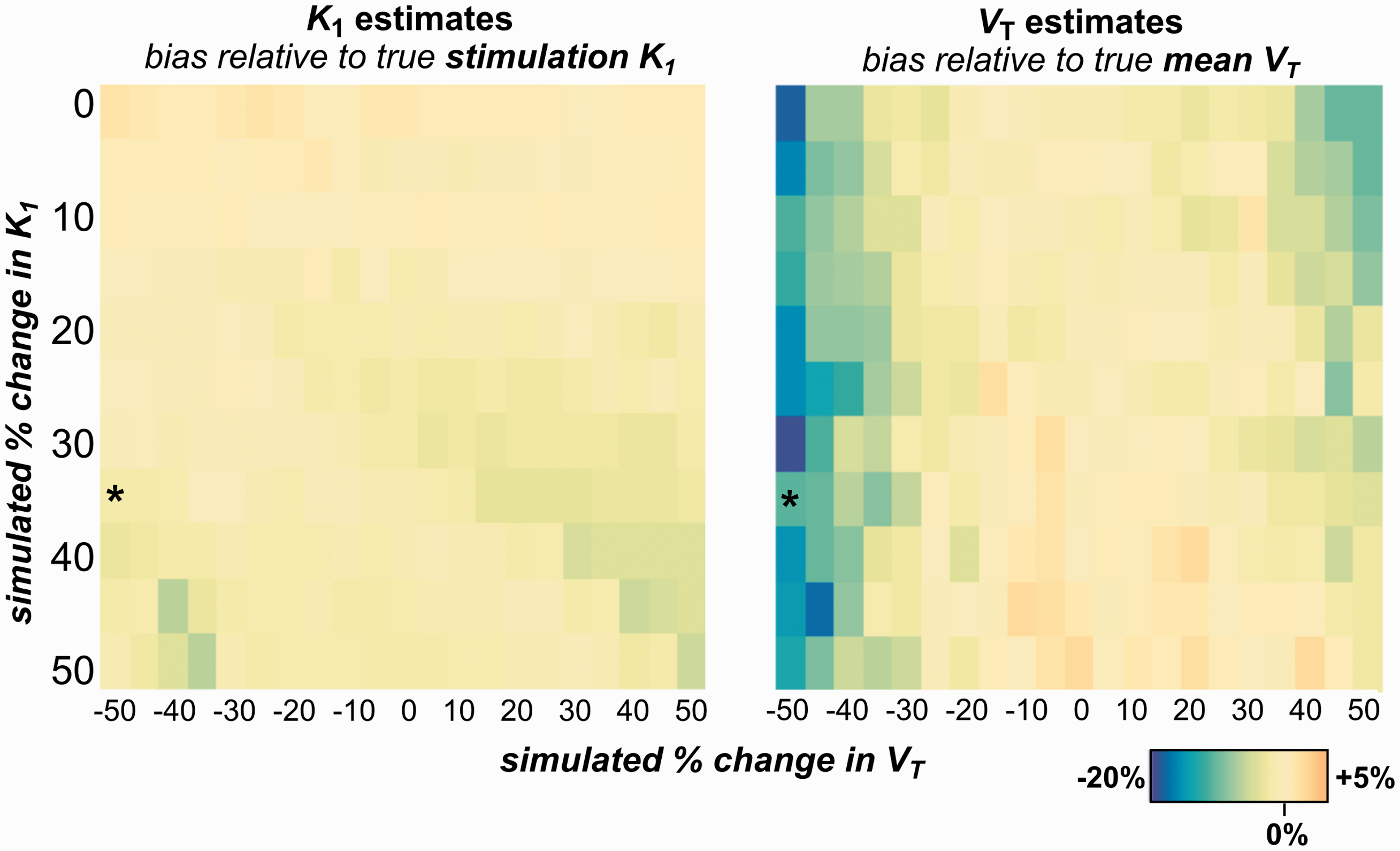
Estimates of
Overall, the simulations suggest that model estimates of
Experimental results
fMRI
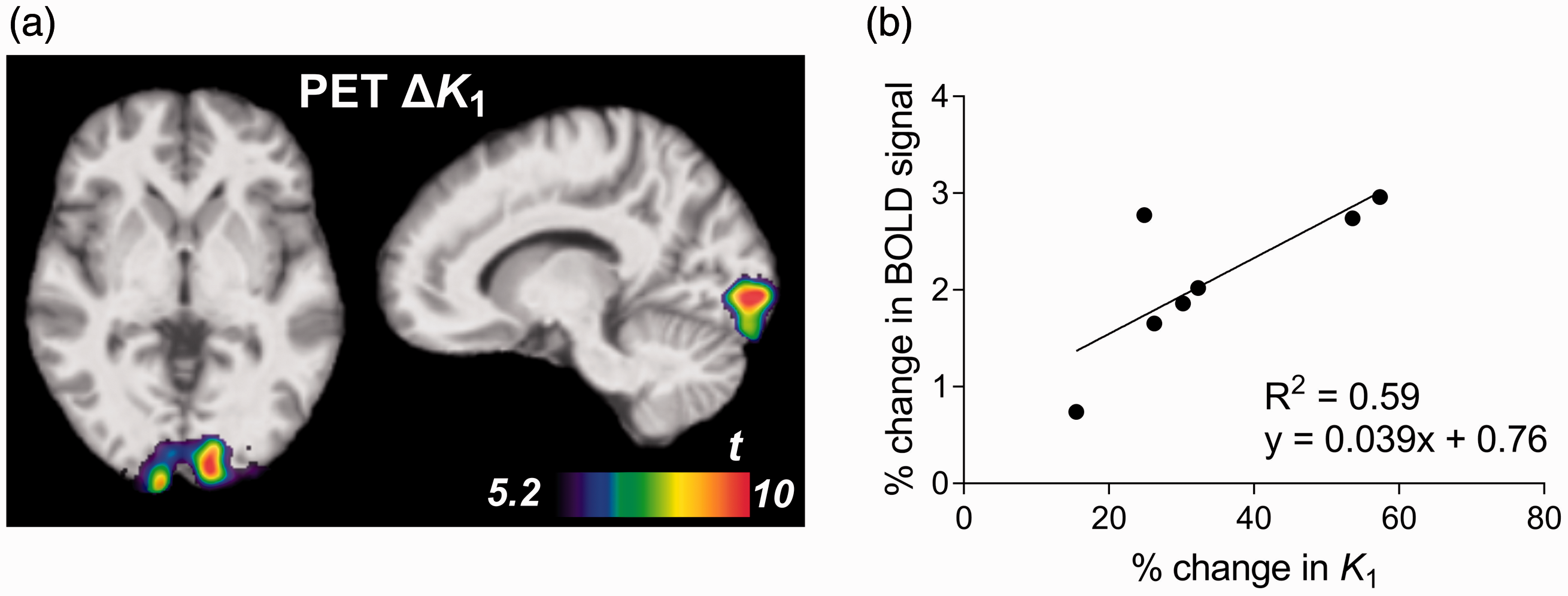
Checkerboard visual stimulation increased fMRI BOLD signal in the occipital cortex (peak MNI coordinates x = −26, y = −74, z = −20; cluster size 4497 voxels; peak T = 16.3, pFWE < 0.001; Figure 3(a)) as well as in the right and left lateral geniculate nucleus (LGN) of the thalamus (x = 22, y = −18, z = −2; 181 voxels; peak T = 14.9, pFWE = 0.001; x = −12, y = −26, z = 0; 262 voxels; peak T = 14.8, pFWE < 0.001). Figure 3(b) and (c) show time courses of the BOLD signal within V1 during short (6 × 30 s) and long (3 × 180 s) stimulation periods. Average percent signal change was 2.11% ± 0.79% during the 30-s blocks, consistent with changes of 1–3% reported in previous studies using similar visual stimuli. 35 , 36 Percent change during the 180-s blocks was 0.91% ± 0.31%, reflecting a slight decrease in the response after an initial peak within the longer blocks (Figure 3(c)). Change from baseline appeared to be sustained across blocks in both cases, suggesting that habituation of the BOLD response was minimal within these 6- and 15-min scans.
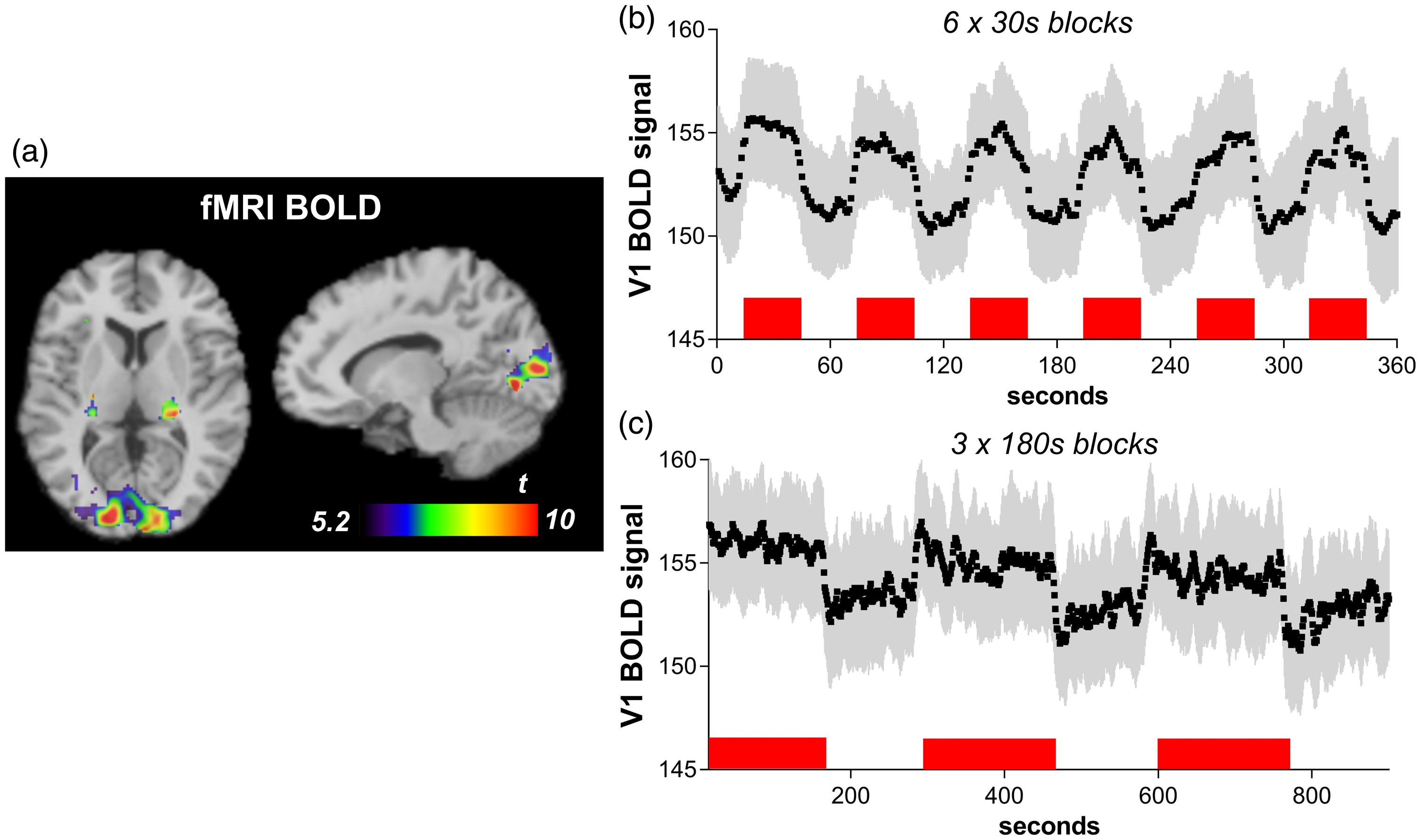
fMRI BOLD response to visual stimulation. (a) Increase in BOLD signal during 30-s stimulation blocks compared to rest (pFWE<0.05) in occipital cortex and lateral geniculate nucleus. (b–c) Time course of BOLD signal (mean ± SD) within V1 for 30-s (b) and 180-s (c) stimulation periods, shown in red.
PET
There were no differences between the baseline and activation scans in injected dose (baseline 649 ± 90.2 MBq, activation 660 ± 114 MBq,
K
1
Visual stimulation induced a robust increase in
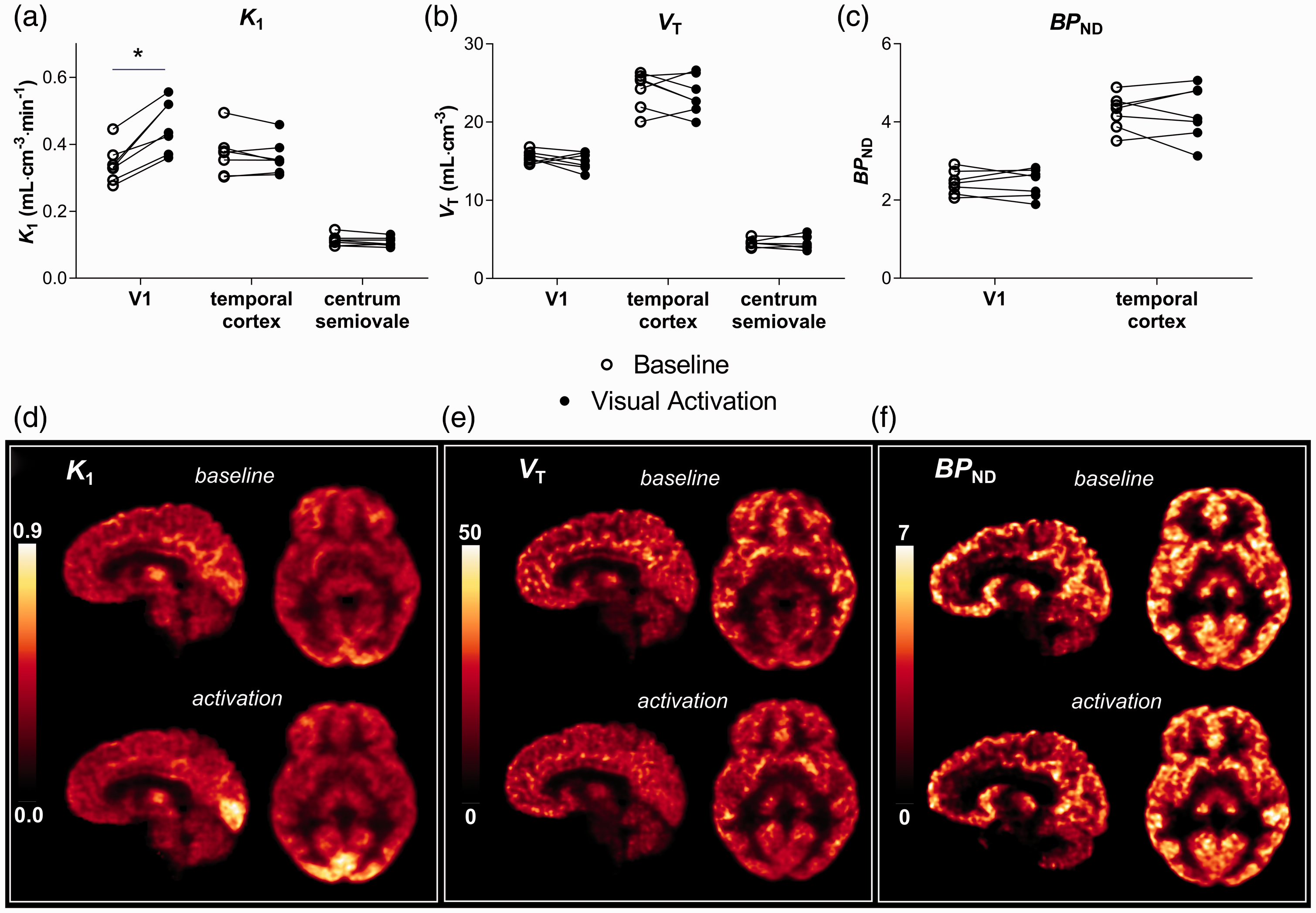
[11C]UCB-J tissue delivery and binding during visual stimulation. (a–c)
Regional changes in
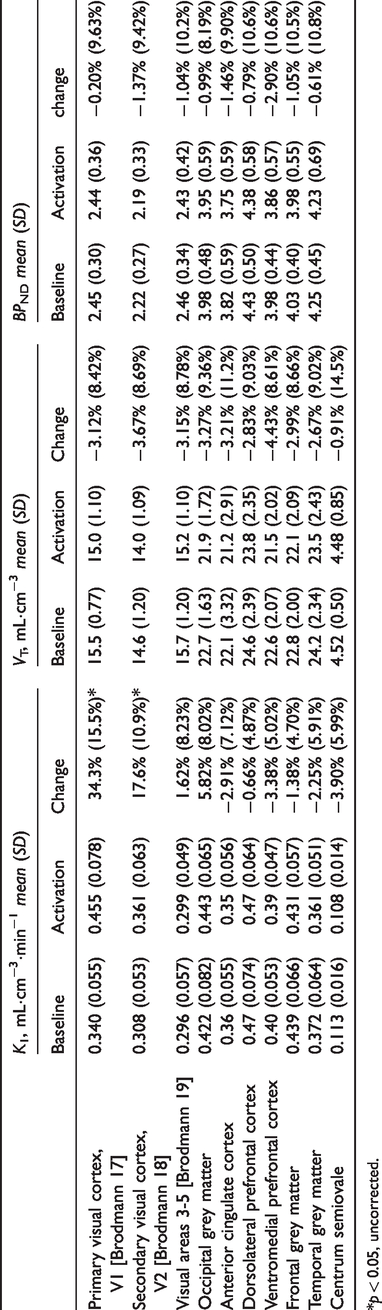
*
Change in [11C]UCB-J
Binding measures
Discussion
This study assessed binding stability of the SV2A radiotracer [11C]UCB-J under conditions of elevated neuronal activity. Despite robust, regionally specific increases in tracer delivery to the primary visual cortex during the presentation of a visual stimulus, there was no apparent change in total or specific binding of [11C]UCB-J in this or any other brain region. This suggests that [11C]UCB-J
Mean increase in [11C]UCB-J
Change in fMRI BOLD signal during visual activation within V1 was in line with previous studies using checkerboard visual stimulation.
24
,
35
,
36
In addition to activation in the visual cortex, BOLD signal increased in LGN (Figure 2(a)), an upstream relay nucleus for visual information.
46
,
47
Given this effect, we assessed PET
Increases in cerebral blood flow and fMRI BOLD signals are correlated with synaptic activity,
48
reflecting the increased energy demands of vesicle-mediated neurotransmission. Previous studies have demonstrated that measures of specific binding from neuroreceptor-targeting radiotracers using kinetic modeling outcome measures are not sensitive to changes in blood flow.
19
,
20
,
48
Here, it was hypothesized that the nature of the SV2A target would lead to changes in binding parameters due to specific changes in tracer-target interactions. Contrary to this prediction, there was no significant change in
Consistent with previous work exploring time-varying system kinetics,
34
model-derived
Altogether, results from the present study suggest that binding properties of [11C]UCB-J are not affected by increased vesicle release despite the crucial functional role of SV2A in these processes. There are several possible explanations for this. It may be the case that despite apparent interactions with other proteins or with Ca2+, the radiotracer binding site remains unaffected during vesicle trafficking and exocytosis. Alternately, studies in several model systems suggest that approximately 80–90% of vesicles are not actively recycled during stimulation and comprise instead a reserve pool whose physiological function and recruitment mechanisms are not yet well understood. 12 , 49 , 50 If vesicles involved in the release cycle are a small proportion of total SV2A-expressing vesicles, overall binding may not be measurably affected even if binding site availability is altered during these processes. It is also possible that binding changes may occur that were not captured by this study. There is some evidence that SV2A expression at excitatory and inhibitory terminals varies by brain region or developmental stage 51 or that the release mechanisms involving SV2A differ between glutamatergic and gamma-aminobutyric acid (GABA)-releasing neurons. 14 If this is the case, SV2A binding behavior (to radiotracer or other relevant proteins) could conceivably vary across neurochemical systems. Neuronal transmission activated by visual stimulation is largely but not entirely glutamatergic,52–54 with evidence for elevated release of GABA and modulatory transmitters including acetylcholine as well.55–57 We do not rule out the possibility that stimuli activating different systems or circuits could alter [11C]UCB-J binding properties. However, given the robust stimulus, ubiquity of SV2A expression across the brain, 6 and highly conserved nature of vesicle release machinery overall, 10 , 11 we conclude that [11C]UCB-J binding is unlikely to be strongly influenced by physiological variation in brain activity.
[11C]UCB-J has demonstrated utility in the assessment of a range of neurological 2 , 4 , 33 and psychiatric 3 , 5 conditions. The present data provide support for a promising advantage of the radiotracer in clinical and research applications, in that they suggest that comparisons between clinical groups or time points will not be confounded by differences in brain state at the time of scan. This is a notable distinction from [18F]FDG, which provides a measure of cerebral metabolic rate of glucose (CMRGlc) and can identify regional impairments in brain activity. [18F]FDG is used to diagnose and track progression of Alzheimer’s disease and other neurological conditions. 58 This is despite its parallel use in applications assessing brain activation, specifically exploiting its sensitivity to transient neural responses. [18F]FDG measurements of CMRGlc are sensitive to sensory stimulation at the time of scan, including the stimulus type used here, 21 , 59 among other physiological and technical factors. 60 , 61 Studies have reported intrasubject variability of up to 25% in test–retest comparisons even in healthy people scanned at close points in time under well-controlled research conditions. 62 As a result, [18F]FDG PET requires careful control of scanning conditions (for example, patient fasting) and its sensitivity is limited to detect only relatively large changes or group differences, approximately 25% or greater. 62 In contrast, our present results and previous test–retest data with [11C]UCB-J PET suggest that changes in synaptic density as small as 10% can be identified, which could allow for the detection of earlier or subtler disease effects. The data presented here support the assertion that in vivo [11C]UCB-J binding can be interpreted as a static measure of SV2A protein density with negligible influence of variability in brain function.
The large, consistent, and highly specific changes in
In summary, binding parameters of the SV2A-targeting radiotracer [11C]UCB-J were unaffected by a robust visual activation stimulus in healthy volunteers. Changes in [11C]UCB-J
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20946198 - Supplemental material for Binding of the synaptic vesicle radiotracer [11C]UCB-J is unchanged during functional brain activation using a visual stimulation task
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20946198 for Binding of the synaptic vesicle radiotracer [11C]UCB-J is unchanged during functional brain activation using a visual stimulation task by Kelly Smart, Heather Liu, David Matuskey, Ming-Kai Chen, Kristen Torres, Nabeel Nabulsi, David Labaree, Jim Ropchan, Ansel T Hillmer, Yiyun Huang and Richard E Carson in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NINDS R01NS094253 and NIAAA K01 AA024788.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
KS designed the study, analyzed data, and drafted the manuscript. HL performed data analysis. DM and M-KC carried out medical screening and supervision. KT oversaw participant recruitment and scanning. NN, DL, and JR performed PET experiments. ATH designed the study and assisted with interpretation of findings. YH designed and performed PET experiments. REC conceived and designed the study. All authors critically revised the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
