Abstract
We investigate whether common circle of Willis (CoW) variants relate to cerebral blood flow (CBF) characteristics among aging adults. Vanderbilt Memory and Aging Project participants free of clinical stroke (n = 390, 71 ± 8 years, 55% male) completed magnetic resonance angiography to assess cerebral artery variants and pseudocontinuous arterial spin labeling to quantify resting CBF and spatial coefficient of variation (sCoV), a marker of perfusion heterogeneity or stability. Linear regression models related categorical variants to regional CBF and sCoV, adjusting for age, sex, race/ethnicity, education, cognitive status, Framingham Stroke Risk Profile, and APOE-ε4 status. Absence of at least one posterior communicating artery (PcoA) related to lower occipital lobe CBF (p = 0.001) and higher global (p = 0.003), temporal (p = 0.001), and occipital lobe sCoV (p = 0.01). Bilateral PcoA absence related to lower global (p = 0.01), parietal (p = 0.01), and occipital lobe CBF (p = 0.0001) and higher global (p < 0.0001), temporal (p < 0.0001), and occipital lobe sCoV (p = 0.0003). Hypoplastic and fetal-type variants did not relate to outcomes (p > 0.22). Findings demonstrate that missing PcoAs influence both perfusion magnitude and efficiency, suggesting that common CoW variants may produce regionally coherent hemodynamic effects in heart-healthy aging adults and represent markers of early cerebrovascular vulnerability.
Introduction
The circle of Willis (CoW) plays critical roles in maintaining stable perfusion, including collateral circulation,1–3 dynamic flow regulation, 4 hemodynamic stress buffering, 5 and long-term adaptation to chronic vascular changes.6,7 Structural variations are observed in approximately 70% of adults. 8 Most prior work has focused on individuals with clinically evident cerebrovascular disease, revealing that posterior (PcoA) and anterior (AcoA) communicating artery variants increase risk, particularly under conditions requiring compensation (e.g. stenosis, occlusion).9–11 There has been comparatively less emphasis on how variants affect dynamic cerebrovascular indicators. Emerging work suggests posterior CoW variants may contribute to perfusion asymmetries,12,13 and missing multiple segments may contribute to cognitive deficits, 14 suggesting threshold effects. Investigating hemodynamic correlates of CoW variation in a healthy aging cohort may clarify how subtle differences in arterial anatomy influence cerebral perfusion prior to the development of overt vascular disease.
Common CoW variants may exacerbate vulnerability to cerebrovascular dysregulation, even in otherwise healthy adults. While research on cerebral perfusion has traditionally emphasized the microcirculation and local autoregulation mechanisms, experimental evidence also highlights the influence of large cerebral arteries,15–17 which play increasingly important roles in aging and cerebrovascular disease. 18 The spatial coefficient of variation (sCoV) 19 offers a valuable complement to cerebral blood flow (CBF) magnetic resonance imaging (MRI) measures, capturing clinically significant perfusion heterogeneity 20 that can signal early or subtle hemodynamic stress. Although single post-labeling delay (PLD) arterial spin labeling (ASL) MRI remains sensitive to both true physiological variation and arterial transit time (ATT) effects,12,13 integrated CBF and sCoV analyses can improve differentiation of physiological and measurement contributions. Novel signatures of emerging hemodynamic insufficiency can be identified, including preserved CBF alongside elevated sCoV (reflecting active compensation and early perfusion instability), reduced CBF alongside preserved sCoV (reflecting uniform hypoperfusion or moderate risk of ATT bias), or reduced CBF alongside elevated sCoV (reflecting heterogenous vascular compromise or high risk of ATT bias).
The Vanderbilt Memory and Aging Project provides a unique opportunity to isolate the effects of common CoW variants on early hemodynamic changes in a cardiovascular-healthy and stroke-free cohort, minimizing confounding from overt disease. We hypothesize that absent or hypoplastic CoW segments, particularly PcoAs, may lead to localized hemodynamic disturbances in anatomically vulnerable regions, providing insight into subclinical pathways linking vascular anatomy to cerebrovascular and cognitive risk.
Methods
Study cohort
The Vanderbilt Memory and Aging Project 21 (VMAP) is a longitudinal study investigating vascular health and brain aging. Between 2012 and 2014, participants were recruited into the “Legacy Cohort” and required to be ⩾60 years of age, have English proficiency, have adequate auditory and visual acuity, have a reliable study partner, and be cognitively unimpaired (CU) or meet diagnostic criteria for early mild cognitive impairment (MCI) 22 or MCI. 23 Starting in 2021, the cohort was expanded (“Expansion Cohort”) to include participants ⩾50 years of age who were required to have English proficiency, have adequate auditory and visual acuity, have a reliable study partner, and be CU. Exclusion criteria for all participants included MRI contraindication; a history of neurological disease (e.g. clinical dementia, clinical stroke, head injury with loss of consciousness >5 min); heart failure; major psychiatric illness; or a systemic or terminal illness affecting follow-up examination participation. At enrollment, participants completed a comprehensive evaluation, including (but not limited to) fasting blood draw, physical examination, clinical interview, medication review, neuropsychological assessment, and multimodal brain MRI. The current study excluded participants for missing predictor, covariate, or outcome data. The protocol, guided by the Belmont Report, was approved by the Vanderbilt University Medical Center Institutional Review Board, and written informed consent was obtained prior to data collection. Data, analytic methods, and study materials can be obtained by contacting the corresponding author.
Brain MRI—CoW variants
Between 2012 and 2017, participants were scanned at the Vanderbilt University Institute of Imaging Science on a 3 T MRI system (Philips Achieva, Best, the Netherlands) with an eight-channel phased-array sensitivity encoding (SENSE) receiver head coil. In 2017, the system was upgraded to a 32-channel dStream head coil. Differences in scanner head coils were evaluated and accounted for as needed (see harmonization methods below). Time-of-flight (TOF) magnetic resonance angiography (MRA) images (TR = 6.8 ms, TE = 3.8 ms, spatial resolution = 0.35 × 0.35 × 0.50 mm3) were acquired as part of the larger multimodal neuroimaging protocol. 2D source MRA images were used to assess the patency of the CoW due to their ability to better preserve signal intensity, vessel continuity, and small or low-flow vessel visibility compared to 3D reconstructions. 24 All images were reviewed on two separate occasions by a board-certified neuroradiologist (LTD) blinded to clinical information. CoW communicating artery (AcoA, left PcoA, right PcoA), P1 (left P1, right P1), and A1 (left A1, right A1) segments were coded based on manual measurements of single image slices as normal (⩾0.8 mm), hypoplastic (<0.8 mm), or aplastic (invisible on MRA). All CoW segments that were coded differently between the two measurements were further assessed by a separate reviewer (CWB) using 3D MRA reconstructed images in OsiriX (OsiriX, Geneva, Switzerland) for the Legacy Cohort, which offered complementary spatial information for secondary evaluations of how vessels connect or branch. The 3D reconstructed images were used to make a final decision on whether segments with discrepancies were normal, hypoplastic, or aplastic (i.e. missing). The entire CoW was then classified as normal across all segments, hypoplastic in at least one segment, or missing in at least one segment for descriptive purposes. Six CoW variant categories were created for analytical purposes, including missing communicating arteries (AcoA missing, unilateral or bilateral PcoAs missing, AcoA and unilateral or bilateral PcoAs missing) and the reference group of otherwise patent communicating arteries (AcoA and bilateral PcoAs normal or hypoplastic); hypoplastic communicating arteries (AcoA hypoplastic, unilateral or bilateral PcoA hypoplastic, AcoA and unilateral or bilateral PcoA hypoplastic) and the reference group of otherwise normal communicating arteries (AcoA and bilateral PcoAs normal); full fetal-type posterior cerebral artery (FTP) variant (unilateral or bilateral P1 missing) and the reference group of otherwise patent P1s (bilateral P1s normal or hypoplastic); and partial FTP (unilateral or bilateral P1 hypoplasia and normal ipsilateral PcoA) and the reference group of otherwise normal P1s (bilateral P1s normal). Variants were further subdivided by unilateral and bilateral characteristics (i.e. “lateralized subgroups”) when sample sizes were sufficient (e.g. missing unilateral PcoA only, missing bilateral PcoA only, missing AcoA + unilateral PcoA, missing AcoA + bilateral PcoA).
Brain MRI—CBF and CBF sCoV
In the VMAP Legacy Cohort, T1-weighted MPRAGE images were acquired using the following parameters: TR = 8.9 ms, TE = 4.6 ms, flip angle = 5°, and spatial resolution = 1 × 1 × 1 mm3. The inversion time (TI) was session-specific, with the “shortest” viable TI selected, ranging from 916.44–978.81 ms (mean = 937.04 ms). In the VMAP Expansion Cohort, T1-weighted MPRAGE images were acquired using the following parameters: TR = 6.5 ms, TE = 2.9 ms, TI = 900 ms, FA = 9°, and spatial resolution = 1 × 1 × 1 mm3. Differences in the T1-weighted MPRAGE sequence were evaluated and accounted for as described in the harmonization methods below. T1-weighted MPRAGE images were post-processed using a NiChart_DLMUSE pipeline (DLMUSE) with parcellation of five gray matter regions of interest (ROI), including whole brain and frontal, temporal, parietal, and occipital lobes.
Standard pseudo-continuous ASL MRI with a 2D EPI readout (label duration = 1.65 s; post-labeling delay = 1.525 s; spatial resolution = 3 × 3 × 7.5 mm3; TR = 3900 ms, TE = 13 ms) assessed CBF (ml of blood/100 g tissue/min) at rest using a reproducible protocol,21,25 enabling reliable measures of CBF and sCoV and enhanced sensitivity to subtle perfusion differences between groups. An equilibrium magnetization (M0) image was acquired separately with identical geometry and TR = 20,000 ms. Images were corrected for motion using the 3dvolreg tool in AFNI version 23.1.10. 26 Skull stripping was performed using SynthStrip. 27 Distortion correction of M0 and ASL images was performed using a field map-less method. 28 Images were slice-time-corrected, normalized by the equilibrium magnetization (M0), and converted to absolute CBF units using recommended guidelines and participant-specific T1b (calculated from participant-specific hematocrit). 29 Individual control-label pairs with poor quality, including large ATT-related signal fluctuations, were excluded using ENhancement of Automated Blood fLow Estimates (ENABLE), an automated method that down-weights or removes image pairs exhibiting excessive motion, signal dropout, temporal variance, and slice-wise artifacts. 30 For our PLD, which fell within the range of the predefined ENABLE PLD index, relative weights for these quality measures were linearly interpolated. Sessions with mean framewise-displacement above 1 mm were excluded due to excessive motion. 31 Final CBF maps were visually inspected and excluded for motion or labeling error artifacts, and vascular artifacts were flagged for exclusion in sensitivity analyses. CBF maps were co-registered to the anatomic T1-weighted map and standard Montreal Neurological Institute template. 32 Transformation matrices were applied to CBF maps. Mean resting CBF was calculated within the gray matter ROIs described above.
Following absolute CBF calculations, the CBF spatial coefficient of variation (sCoV) was assessed as a measure of perfusion heterogeneity or stability. 19 sCoV was calculated by dividing the standard deviation of the CBF signal within a given gray matter ROI by the corresponding mean CBF signal (i.e. σ (CBFROI)/μ (CBFROI) × 100). We additionally calculated a nuisance covariate, pseudo sCoV, within each ROI using a pseudo CBF image created from smoothed gray matter and white matter segmentations to adjust for partial volume-related signal variability, as previously described. 33 Partial volume corrected (PVC) CBF and pseudo sCoV maps were generated using DLMUSE segmentations to account for tissue-specific signal variability.
Brain MRI—harmonization
The present study combined baseline data across the VMAP Legacy Cohort and Expansion Cohort. To account for batch variable effects (e.g. differences in scanner coil, scanner software, T1-sequence parameters), ASL-based outputs (CBF, sCoV, PVC–CBF, pseudo sCoV) were harmonized using longitudinal ComBat 34 across the combined VMAP Legacy and Expansion Cohorts. T1 sequence was included as a batch variable in CBF harmonization to account for scanner-specific differences, while MPRAGE images themselves were not directly harmonized prior to ASL analysis.
Harmonization outputs were evaluated primarily via visual inspection of spaghetti plots to check for outliers, epoch-wise issues, and general patterns. This approach ensures consistent population-level adjustment for scanner and sequence effects while preserving physiologically interpretable measures for CBF and sCoV analyses. Following longitudinal harmonization, data were subset for the present cross-sectional analysis.
Analytical plan
Covariates were identified a priori for their potential to confound the analytical models, including age,35–37 sex, 38 education, 39 race/ethnicity,40,41 cognitive status, 42 Framingham Stroke Risk Profile (FSRP) excluding points assigned for age,43,44 and apolipoprotein E ε4 (APOE-ε4) carrier status (defined as positive (ε2/ε4, ε3/ε4, ε4/ε4) or negative (ε2/ε2, ε2/ε3, ε3/ε3)). 45 FSRP assigned points by sex for age, systolic blood pressure (accounting for antihypertensive medication use), diabetes mellitus, cigarette smoking, left ventricular hypertrophy, cardiovascular disease, and atrial fibrillation.46,47
Linear regression models using ordinary least square estimates related categorical CoW variants (i.e. missing communicating arteries; missing communicating arteries by lateralized subgroups; hypoplastic communicating arteries; hypoplastic communicating arteries by lateralized subgroups; full FTP; partial FTP) to region-specific mean resting CBF (one test per model) or log-transformed sCoV (one test per model). Each model yielded a global F-test from analysis of variance (ANOVA) to assess whether CoW variant status collectively explained a significant proportion of variance in CBF or sCoV, as well as level-specific estimates comparing each CoW variant to the reference category. Models were adjusted for age, sex, race/ethnicity, education, cognitive status, FSRP (excluding points assigned for age), and APOE-ε4 status. Sensitivity analyses were performed excluding statistical outliers (defined as ⩾4 standard deviations from the sample mean) to test if these conditions accounted for significant results. Additional sensitivity analyses assessed whether partial-volume related signal variability (e.g. tissue atrophy) confounded significant results by re-running CBF models using PVC CBF outcomes, and separately, adding pseudo sCoV covariates to sCoV models. To account for multiple comparisons, the Benjamini-Hochberg procedure was used to control the false discovery rate (FDR) at a threshold of Q = 0.05. 48 FDR was chosen for its ability to balance error control and sensitivity in datasets with expected correlation among ROI measures. The number of comparisons was set to m = 10 outcomes per predictor, consistent with hypotheses (i.e. variant effect profiles across regional perfusion outcomes). This overall approach supports hypothesis-driven inference while accounting for dependence among related measures. Final sensitivity analyses applied a global FDR correction across all tests (m = 60) to assess whether observed results reflect stable, reproducible effects. All analyses were conducted using R version 3.2.3. 47
Results
Participant characteristics
The cohort was composed of 390 adults (71 ± 8 years, 55% male, 88% self-identified as non-Hispanic White), including 245 CU participants, 121 participants with MCI, and 24 with early MCI. Mean CBF was 48 ± 10 ml/100 g/min and mean sCoV was 41% ± 9% across whole brain gray matter. Sex differences existed across missing communicating artery subgroups (p = 0.002). See Table 1 for more details.
Participant characteristics by communicating artery variant.
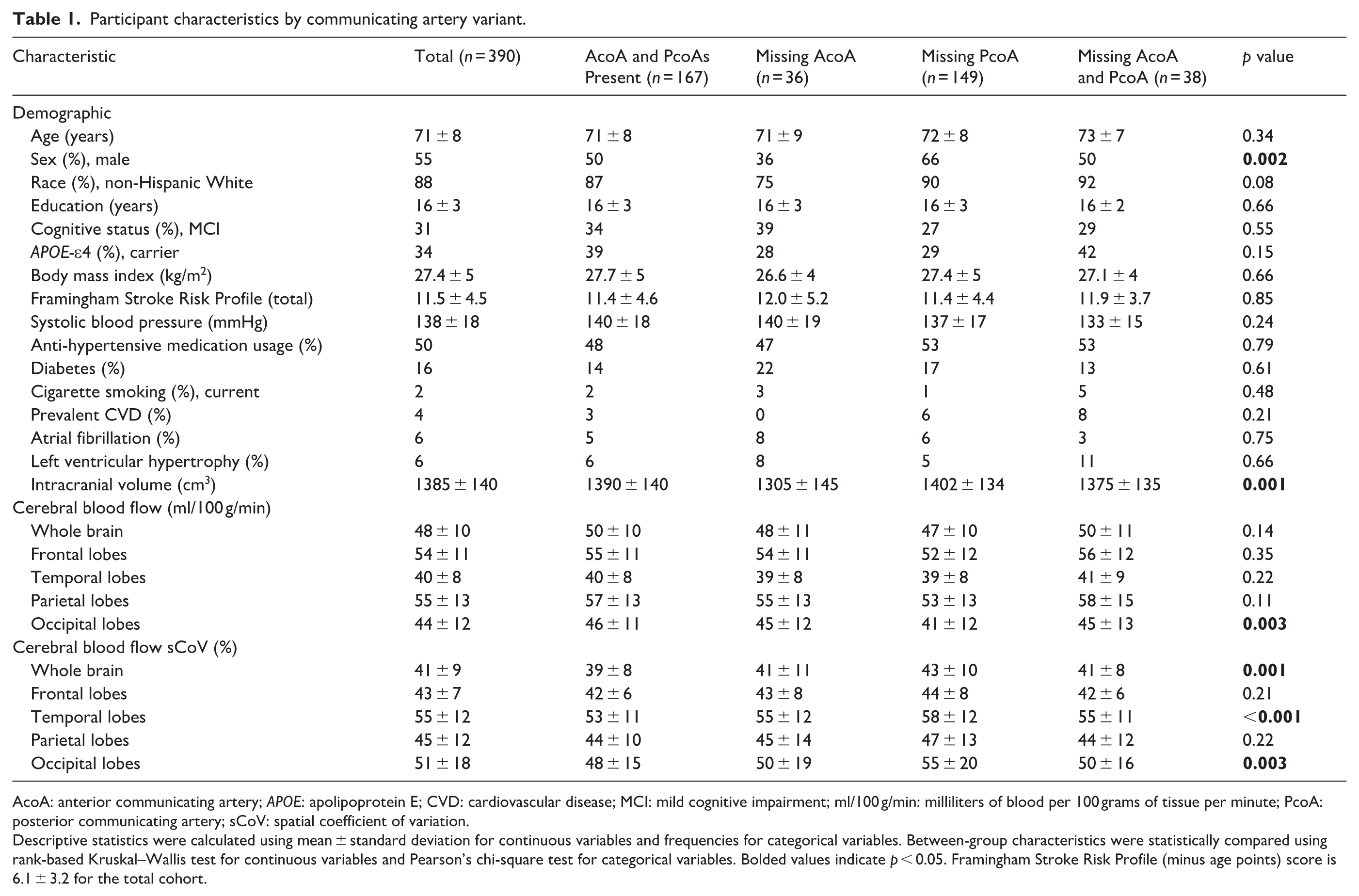
AcoA: anterior communicating artery; APOE: apolipoprotein E; CVD: cardiovascular disease; MCI: mild cognitive impairment; ml/100 g/min: milliliters of blood per 100 grams of tissue per minute; PcoA: posterior communicating artery; sCoV: spatial coefficient of variation.
Descriptive statistics were calculated using mean ± standard deviation for continuous variables and frequencies for categorical variables. Between-group characteristics were statistically compared using rank-based Kruskal–Wallis test for continuous variables and Pearson’s chi-square test for categorical variables. Bolded values indicate p < 0.05. Framingham Stroke Risk Profile (minus age points) score is 6.1 ± 3.2 for the total cohort.
CoW variant prevalence
Variant CoW structures were observed among 96.2% of participants at baseline. Variant communicating arteries were observed among 91.3% of participants, including 34.1% with at least one hypoplastic communicating artery and 57.2% with at least one missing communicating artery. Among individual arteries, A1 variants were observed among 9.7% of participants (including 6.9% hypoplastic and 2.8% missing), AcoA variants among 59.3% of participants (including 40.3% hypoplastic and 19.0% missing), P1 variants among 22.1% of participants (including 14.9% hypoplastic and 8.5% missing), and PcoA variants among 77.4% of participants (including 38.2% hypoplastic and 47.9% missing). Unilateral variants were observed among 9.7% of participants for A1 segments (including 6.9% hypoplastic and 2.8% missing), 21.3% of participants for P1 segments (including 13.1% hypoplastic and 8.2% missing), and 51.5% of participants for PcoA segments (including 25.9% hypoplastic and 25.6% missing). Bilateral variants were observed among 2.1% of participants for P1 segments (including 1.8% hypoplastic and 0.3% missing) and 34.6% of participants for PcoA segments (including 12.3% hypoplastic and 22.3% missing); no bilateral A1 variants were observed. The prevalence of full FTP variants was 8.5% and partial FTP variants was 13.6%. See Table 2 for further details.
Prevalence of CoW variants.
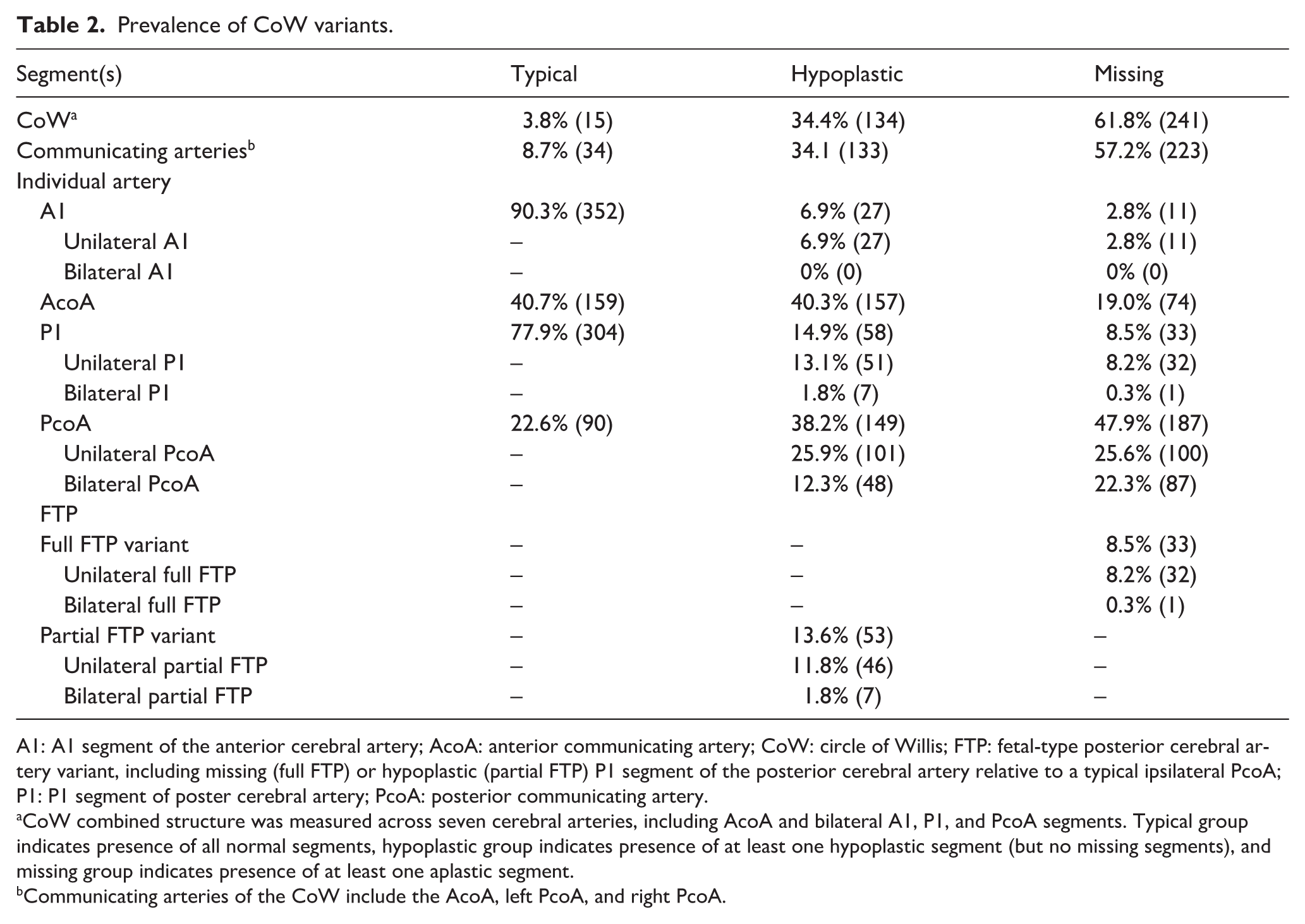
A1: A1 segment of the anterior cerebral artery; AcoA: anterior communicating artery; CoW: circle of Willis; FTP: fetal-type posterior cerebral artery variant, including missing (full FTP) or hypoplastic (partial FTP) P1 segment of the posterior cerebral artery relative to a typical ipsilateral PcoA; P1: P1 segment of poster cerebral artery; PcoA: posterior communicating artery.
CoW combined structure was measured across seven cerebral arteries, including AcoA and bilateral A1, P1, and PcoA segments. Typical group indicates presence of all normal segments, hypoplastic group indicates presence of at least one hypoplastic segment (but no missing segments), and missing group indicates presence of at least one aplastic segment.
Communicating arteries of the CoW include the AcoA, left PcoA, and right PcoA.
CoW variants and CBF
Missing communicating arteries related to differences in occipital lobe CBF (p = 0.01). The association survived outlier exclusion (p = 0.01), partial volume correction (p = 0.03), and vascular artifact exclusion (p = 0.02) but was attenuated with FDR correction (p = 0.07). Level-specific estimates indicated that missing PcoAs related to lower occipital lobe CBF (β = −4.15 ml/100 g/min, p = 0.001), and other variant levels were not related (p-values > 0.43). Missing communicating arteries by lateralized subgroup related to differences in whole brain (p = 0.04), parietal (p = 0.02), and occipital lobe CBF (p = 0.001). Associations survived outlier exclusion (p-values < 0.04) and vascular artifact exclusion (p-values < 0.04); while parietal and occipital lobe CBF associations survived FDR correction (FDR-adjusted p-values < 0.05) and partial volume correction (p-values < 0.04), whole brain CBF associations were attenuated with FDR correction (p = 0.07) and partial volume correction (p = 0.09). Level-specific estimates indicated that missing bilateral PcoAs related to lower whole brain (β = −3.93 ml/100 g/min, p = 0.01), parietal (β = −4.87 ml/100 g/min, p = 0.01), and occipital lobe CBF (β = −6.54 ml/100 g/min, p = 0.0001); missing AcoA and bilateral PcoAs related to higher parietal lobe CBF (β = 6.47 ml/100 g/min, p < 0.05); and other variant levels were not related (p-values > 0.21). Neither hypoplastic communicating arteries (p-values > 0.40), hypoplastic communicating arteries by lateralized subgroup (p-values > 0.32), full FTP (p-values > 0.70), nor partial FTP variants (p-values > 0.22) related to any of the CBF ROIs. In sensitivity analyses using a global FDR correction, missing communicating arteries by lateralized subgroup remained related to differences in occipital lobe CBF (p = 0.03). See Table 3 for details and Figure 1 for illustrations.
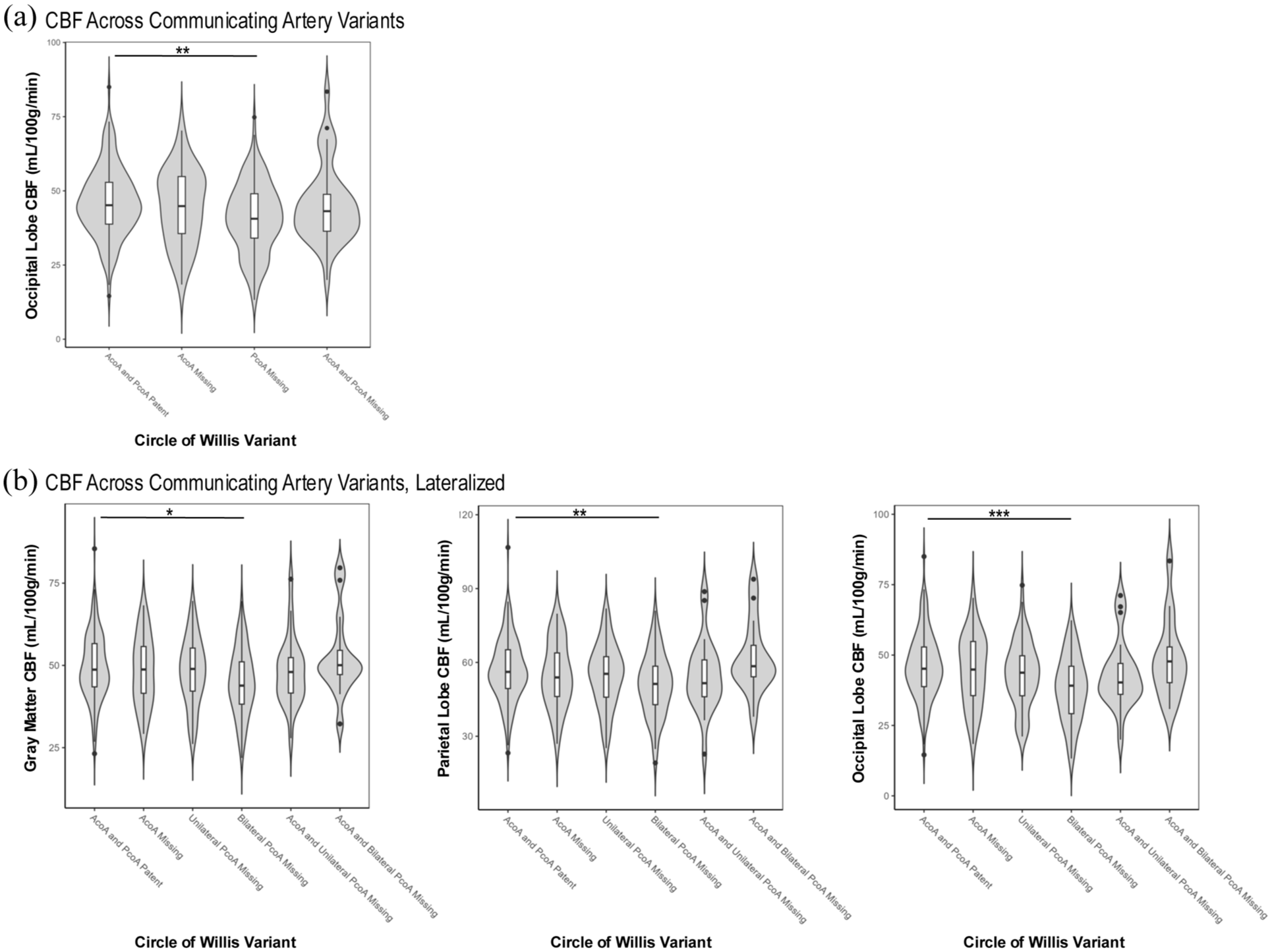
Resting CBF across missing communicating artery variants. Occipital lobe CBF differed significantly across missing communicating artery variants (a), such that missing PcoAs related to lower occipital lobe CBF compared to the patent AcoA and PcoA reference level, indicating hypoperfusion. Whole brain, parietal, and occipital lobe CBF differed significantly across missing communicating artery variants when examined by unilateral or bilateral characteristics (b), such that missing bilateral PcoAs related to lower CBF levels compared to the patent AcoA and PcoA reference level. In addition, within parietal lobe CBF models, concurrently missing AcoA and bilateral PcoAs marginally related to higher CBF compared to the patent AcoA and PcoA reference level.
CoW variants and resting CBF associations.
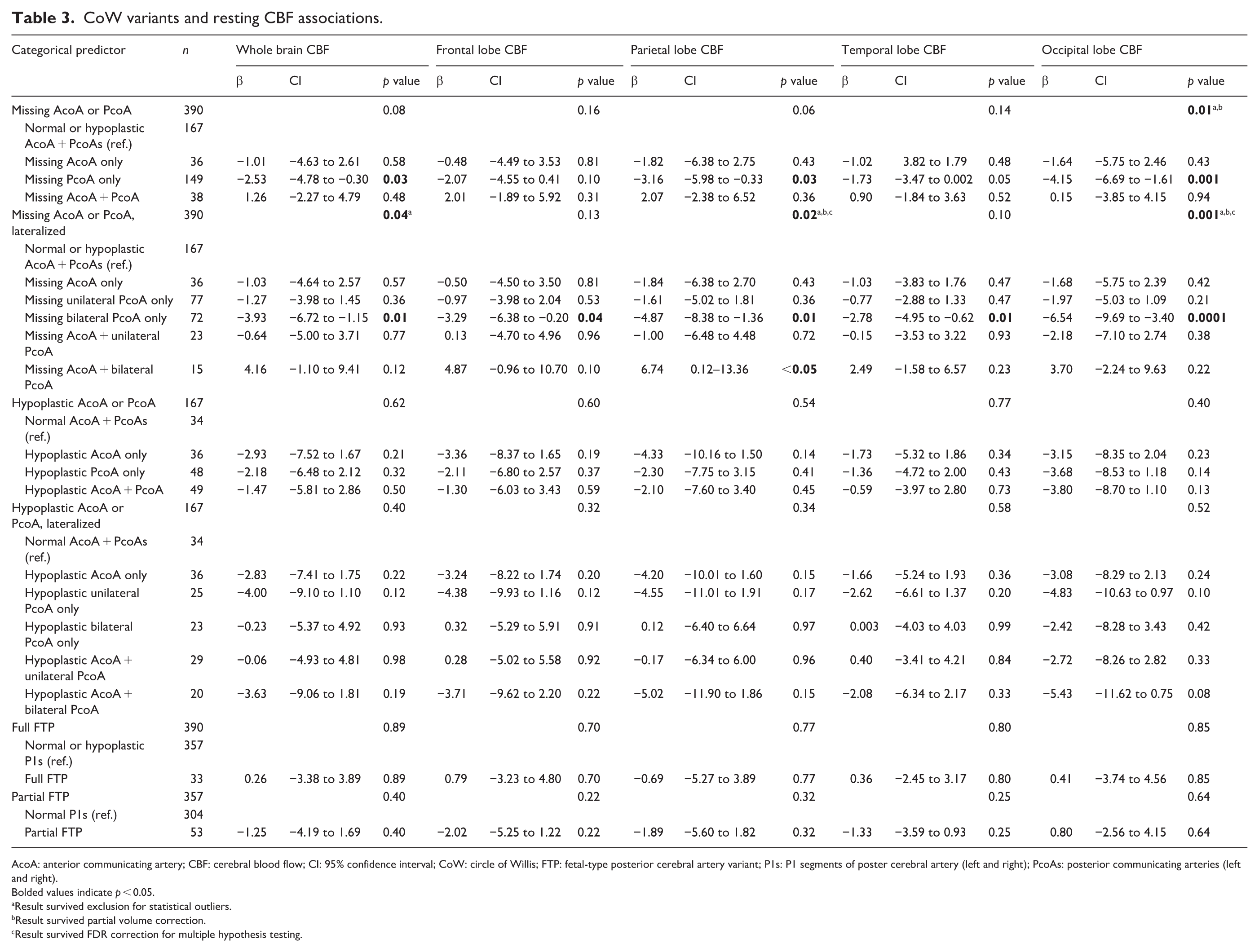
AcoA: anterior communicating artery; CBF: cerebral blood flow; CI: 95% confidence interval; CoW: circle of Willis; FTP: fetal-type posterior cerebral artery variant; P1s: P1 segments of poster cerebral artery (left and right); PcoAs: posterior communicating arteries (left and right).
Bolded values indicate p < 0.05.
Result survived exclusion for statistical outliers.
Result survived partial volume correction.
Result survived FDR correction for multiple hypothesis testing.
CoW variants and CBF sCoV
Missing communicating arteries related to differences in whole brain (p = 0.03), temporal (p = 0.01), and occipital lobe sCoV (p = 0.04). Associations survived outlier exclusion (p-values < 0.05) but were attenuated with FDR correction (p-values > 0.06); temporal and occipital lobe sCoV associations survived partial volume correction (p-values < 0.05) but whole brain sCoV did not (p = 0.05); and temporal lobe associations survived vascular artifact exclusion (p = 0.03) but whole brain and occipital lobe sCoV did not (p-values > 0.07). Level-specific estimates indicated that missing PcoAs related to higher whole brain (p = 0.002), temporal (p = 0.001), and occipital lobe sCoV (p = 0.01), and other variant levels were not related (p-values > 0.17). Missing communicating arteries by lateralized subgroup related to differences in whole brain (p = 0.002), temporal (p = 0.001), and occipital lobe sCoV (p = 0.01). Associations survived FDR correction (p-values < 0.01), outlier exclusion (p-values < 0.01), partial volume correction (p-values < 0.005), and vascular artifact exclusion (p-values < 0.01). Level-specific estimates indicated that missing bilateral PcoAs related to higher whole brain (p < 0.0001), temporal (p < 0.0001), and occipital lobe sCoV (p = 0.0003). Neither hypoplastic communicating arteries (p-values > 0.22), hypoplastic communicating arteries by lateralized subgroup (p-values > 0.32), full FTP (p-values > 0.60), nor partial FTP variants (p-values > 0.82) related to sCoV ROIs. In sensitivity analyses using a global FDR correction, missing communicating arteries by lateralized subgroup remained related to differences in whole brain (p = 0.03) and temporal lobe sCoV (p = 0.03). See Table 4 for details and Figure 2 for illustrations.
CoW variants and CBF sCoV associations.
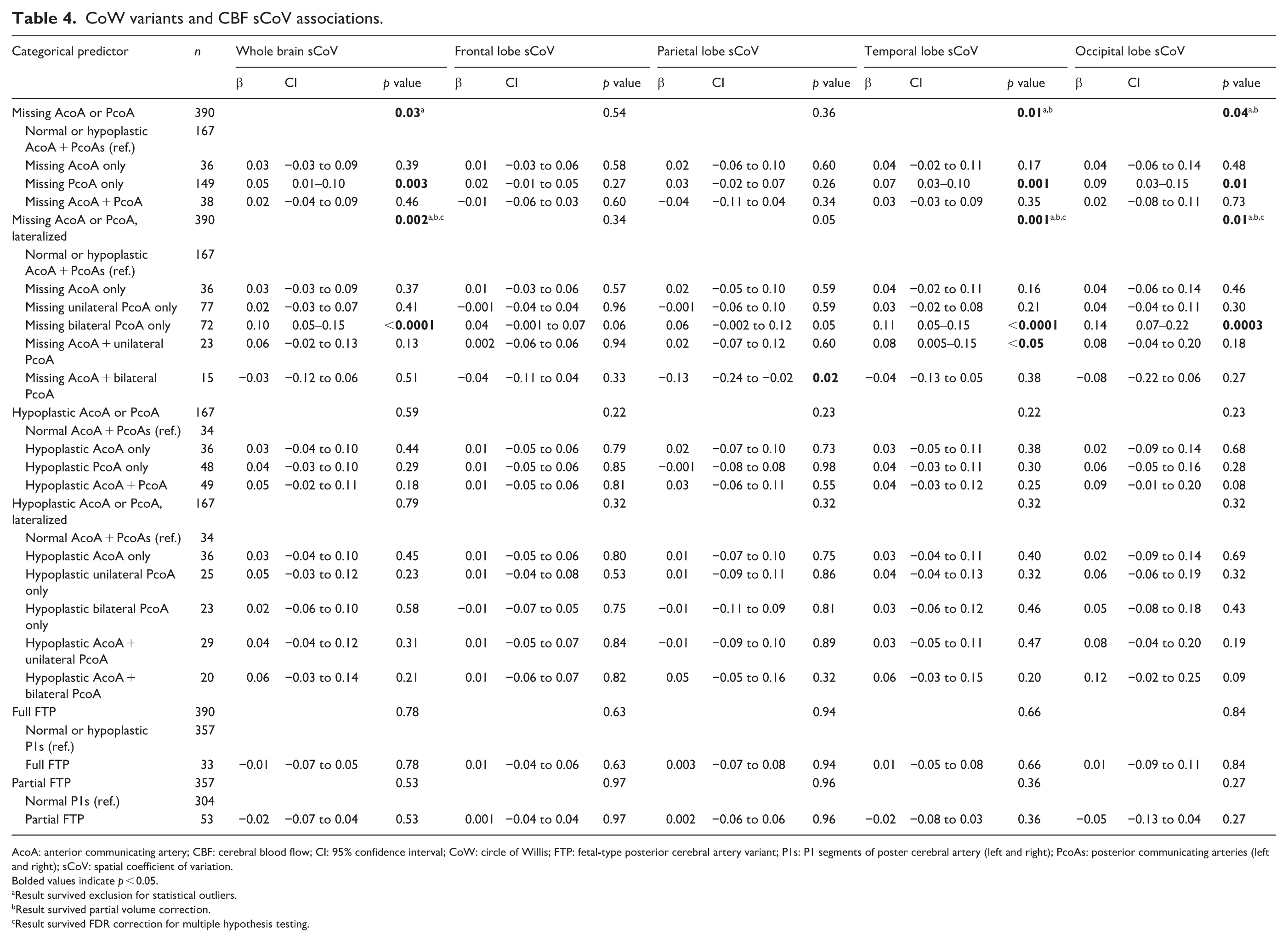
AcoA: anterior communicating artery; CBF: cerebral blood flow; CI: 95% confidence interval; CoW: circle of Willis; FTP: fetal-type posterior cerebral artery variant; P1s: P1 segments of poster cerebral artery (left and right); PcoAs: posterior communicating arteries (left and right); sCoV: spatial coefficient of variation.
Bolded values indicate p < 0.05.
Result survived exclusion for statistical outliers.
Result survived partial volume correction.
Result survived FDR correction for multiple hypothesis testing.
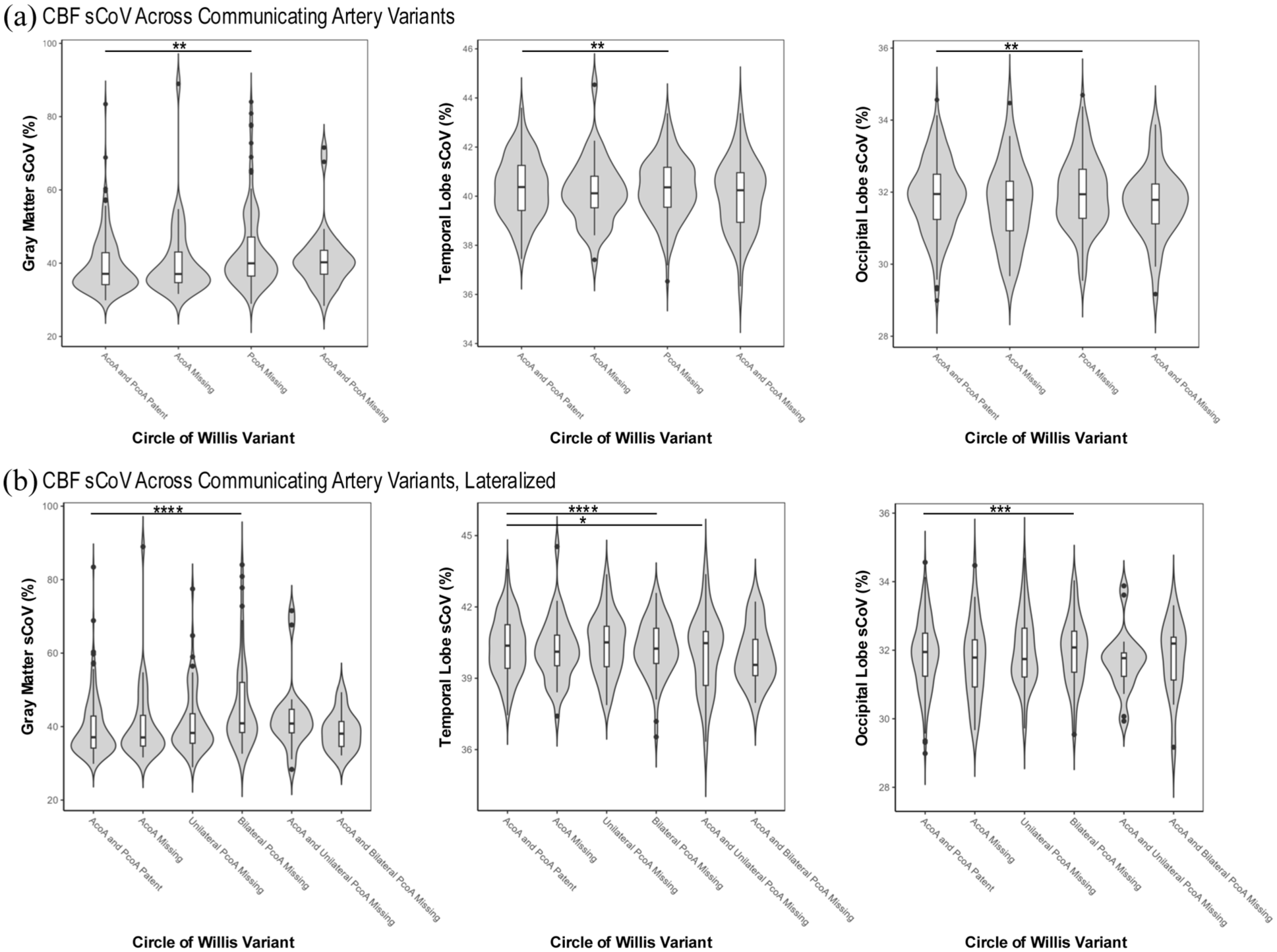
CBF sCoV across missing communicating artery variants. Whole brain, temporal, and occipital lobe sCoV differed significantly across missing communicating artery variants (a), such that the missing PcoAs related to higher sCoV compared to the patent AcoA and PcoA reference level, indicating higher perfusion instability. The same sCoV regions differed significantly across missing communicating artery variants when examined by unilateral or bilateral characteristics (b), such that missing bilateral PcoAs related to higher whole brain, temporal, and occipital lobe sCoV compared to the patent AcoA and PcoA reference level. In addition, within temporal lobe sCoV models, concurrently missing AcoA and unilateral PcoA marginally related to higher temporal lobe sCoV compared to the patent AcoA and PcoA reference level.
Discussion
Among community-dwelling middle-aged and older adults free of clinical stroke at baseline, structural variants of the CoW communicating arteries related to greater cerebral perfusion variability and modest perfusion reductions. Bilateral absence of the PcoAs was linked to significantly higher whole brain sCoV (most notably in the temporal lobes), lower occipital lobe CBF alongside higher occipital sCoV, and moderately lower parietal lobe CBF. Results reveal a directionally consistent pattern in which the loss of posterior collateral pathways appears to reduce both perfusion stability and magnitude in anatomically vulnerable regions. It is noteworthy that these effects were statistically independent of vascular risk factors (e.g. hypertension, diabetes, smoking), indicating that even among relatively healthy aging adults with minimal cardiovascular disease, prevalent PcoA absence may still relate to compromised cerebral hemodynamics or emerging risk.
The association of PcoA absence with elevated sCoV may reflect posterior perfusion instability and the potential compensatory limits of collaterals. Missing PcoAs limit posterior-to-anterior compensation capacity, and occipital and temporal lobes may be particularly vulnerable compared to anterior regions.49,50 Loss of PcoA support may shift perfusion toward longer, indirect, or unstable collateral routes, which may promote preserved but unstable blood flow (as observed in temporal lobes), compromised and unstable flow (as observed in occipital lobes), or overall prolonged ATT. In temporal lobes, mixed arterial inputs may enable compensatory adjustments when posterior collaterals are compromised, increasing blood flow heterogeneity despite its overall preservation. In contrast, sCoV may be more tightly coupled to CBF in occipital lobe regions with distal arterial positioning and limited collateral support, which increase compensation failure risk. Elevated sCoV patterns may thus serve as sensitive markers of posterior circulation stress, reflecting subtle compensation adjustments or emerging vascular insufficiency. 19 Importantly, the link between PcoA absence and higher sCoV may also reflect ATT-related measurement bias, particularly with our single-PLD ASL methodology. Definitive separation of ATT effects from true perfusion deficits would require multi-PLD ASL or PET. Findings suggest that the preservation of regional CBF may depend on the presence and redundancy of collateral pathways, with temporal lobes demonstrating compensatory capacity and occipital lobes susceptible to compromised flow.
Observed CBF reductions were modest (e.g. −9% in parietal lobe) compared with larger sCoV increases (e.g. +20% in temporal lobes), consistent with mild hypoperfusion in vulnerable territories. Parietal regions, largely supplied by the middle cerebral artery, may offer higher resilience to posterior circulation variants and the capacity to redistribute flow uniformly. Although missing PcoAs may affect perfusion to parietal regions with minor posterior inputs (e.g. watershed areas), most flow likely still arrives via well-established anterior arteries, maintaining uniform distribution. While ATT-related CBF underestimation remains possible even with stable sCoV, parietal lobe patterns of modest CBF reductions with preserved sCoV represent strong candidates for true hypoperfusion and stable flow redistribution rather than perfusion instability or collateral failure. Taken together, results suggest region-dependent hemodynamic consequences of PcoA absence, wherein parietal lobes maintain effective compensation with modestly reduced but uniform flow, temporal lobes demonstrate greater flow variability and likely reliance on indirect collateral routes, and occipital lobes appear the most hemodynamically vulnerable.
As expected, most CoW variants were observed among communicating arteries (91.3%), including missing PcoAs (47.9%), hypoplastic AcoA (40.3%), and hypoplastic PcoAs (38.2%). The high prevalence of PcoA variants, especially absence, likely reflects this segment’s vulnerability to regression, collapse, or stenosis. Most existing evidence suggests CoW configurations are established early, 51 but age-related changes (e.g. arterial stiffening, hypertension, atherosclerosis, microvascular rarefaction, reduced autoregulatory capacity) may also promote variant emergence, particularly since small caliber vessels are inherently fragile.52–54 Given our cohort’s healthy cardiovascular profile, variants likely represent baseline collateral anatomy, although age-related modification is still possible. If congenital, CoW variants may reflect inherent vascular configurations that have shaped lifelong resilience to hemodynamic stress, whereas variants acquired from cumulative degenerative processes may relate to emerging signatures of flow reorganization. Notably, while our TOF MRA imaging parameters offer relatively high-resolution for small artery detection, this method may still overestimate AcoA absence compared to previous studies. 55 PcoA or AcoA absence may thus arise from any combination of congenital differences, cumulative pathology, and methodological limitations.
Several subthreshold findings demonstrated notable effect sizes with unexpected regional or directional trends, likely reflecting small subgroup sizes, regional measurement variability, or physiological heterogeneity. These associations nonetheless provide insight into regional vulnerability and physiologically plausible compensation patterns (e.g. reduced anterior or watershed perfusion capacity when anterior-to-posterior pathways are eliminated 9 ; a shift in perfusion reliance to distal pathways when central CoW redundancy is lost2,56), which could offer hypotheses for future studies with larger samples or higher-sensitivity imaging. Given limited statistical power and unstable estimates, these findings should be interpreted cautiously and do not provide definitive evidence of physiological effects.
Our study has several limitations. Although TOF MRA likely captured most hemodynamically relevant segments,57,58 very small segments still capable of hemodynamic function 59 may still be missed or misclassified, creating heterogeneous subgroups that mask true effects. Combined ACoA/PCoA modeling may also reduce anatomical specificity and conflate distinct effects. Single-PLD ASL with a one-compartment model may underestimate CBF in regions with delayed ATT due to late blood arrival or the label decaying in transit. We are thus limited in our ability to distinguish true hypoperfusion from ATT artifact. While our rigorous quality-control procedures (e.g. motion exclusion, ENABLE filtering, vascular artifact screening), region-specific comparative analyses, and usage of a heart-healthy cohort reduce the risk of ATT-related bias, residual confounding cannot be ruled out. Although statistically significant, most observed associations were small relative to natural variability, limiting predictive or diagnostic value at the individual level. Nevertheless, findings may still be meaningful in the context of cardiovascular or demographic risk factors and offer new insights into early regional vulnerability and collateral support limits. Small effects are more susceptible to noise, measurement error, or unmeasured confounders, and residual confounding remains possible; subclinical cerebrovascular disease or physiological fluctuations (e.g. end-tidal CO2) may contribute variability. Finally, the cross-sectional design and small subgroup sizes for rarer variants limit causal inference. These factors may attenuate or obscure subtle effects, though observed patterns remain consistent and biologically plausible.
Despite these limitations, this study has several strengths. Our extensive MRA protocol, including double review by a board-certified neuroradiologist (LTD) and reconciliation by a secondary reviewer (CWB), reduces CoW measurement error. Classifications using quantitative thresholds (0.8 mm) increase sensitivity to subtle vessel variations and facilitate reproducibility. Comprehensive variant coding, capturing unilateral/bilateral, single-segment, and co-occurring variations (e.g. FTP) provides novel, fine-grained arterial specificity over global CoW patency approaches. Studying healthy older adults with low cardiovascular risk offers insight into early hemodynamic changes before overt pathology, minimizing confounding by major stenosis, stroke, or collateral failure, and enables isolation of anatomical variant effects. Concurrent CBF and sCoV analyses provide complementary information on perfusion magnitude and efficiency, revealing early-stage hemodynamic dysregulation patterns. While findings are broadly relevant (e.g. community-dwelling adults, high prevalence variants 60 ), our cohort may not fully reflect the general population, as participants were predominantly White (non-Hispanic), formally educated, and relatively healthy; associations may be stronger in higher-risk groups. Lastly, our adjustments for multiple comparisons and potential confounds (e.g. statistical outliers, cardiovascular risk factors, partial volume effects, vascular artifact) increase the robustness of findings. Future research is warranted to replicate findings, expand CoW characterizations, clarify mechanisms of early stage hemodynamic insufficiency (e.g. VEASL MRI), and assess the clinical significance of our findings.
In conclusion, this study demonstrates that structural variants of the CoW, particularly bilateral PcoA absence, exert regionally coherent effects on cerebral perfusion dynamics in community-dwelling, heart-healthy aging adults. Bilateral variants produced larger effects than unilateral, suggesting a dose-like relationship. Our findings expand frameworks for interpreting ASL perfusion heterogeneity in healthy aging. Combined sCoV and CBF profiles revealed region-specific patterns of effective flow redistribution, emerging hemodynamic stress, and potential measurement artifact risk. Over time, the subtle perfusion changes observed may increase vulnerability to age-related cerebrovascular dysfunction and cognitive decline. 61 However, future studies are needed to disentangle true perfusion deficits from transit time related confounds. These results provide novel insight into the link between common large artery variants and hemodynamic dysregulation, highlighting early hemodynamic vulnerabilities that may precede overt cerebrovascular disease and provide a framework for assessing subclinical vascular risk in healthy aging.
Footnotes
Acknowledgements
The authors would like to thank the dedicated Vanderbilt Memory and Aging Project participants, their loved ones, and our devoted staff and trainees who contributed to the recruitment, screening, and enrollment of the baseline cohort.
Non-standard abbreviations and acronyms
AcoA = anterior communicating artery, APOE = apolipoprotein E, ASL = arterial spin labeling, ATT = arterial transit time, CBF = cerebral blood flow, CMR = cardiac magnetic resonance imaging, CVD = cerebrovascular disease, CoW = circle of Willis, CI = 95% confidence interval, CU = cognitively unimpaired, DLMUSE = NiChart_DLMUSE pipeline; ENABLE = ENhancement of Automated Blood fLow Estimates, FSRP = Framingham Stroke Risk Profile, FTP = fetal-type posterior cerebral artery variant, PLD = post-labeling delay, PVC = partial volume corrected, VMAP = Vanderbilt Memory and Aging Project, MCI = mild cognitive impairment, MRA = magnetic resonance angiography, MRI = magnetic resonance imaging, PcoA = posterior communicating artery, ROI = region of interest.
Author contributions
FEC and ALJ conceived and designed the study. CWB, LTD, FEC, WHR, KRP, NS, TBJ, and TJH, curated and managed the imaging and clinical data. PZ, DL, and FEC performed the statistical analyses. FEC interpreted the results and drafted the manuscript. PZ created visual representations of results. ALJ supervised research activities and acquired funding and resources. All authors reviewed, provided critical revisions, and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by T32-AG058524 (FEC), Howard Hughes Medical Institute James H Gilliam Fellowship for Advanced Study (FEC), F31-AG059345 (FEC), Alzheimer’s Association IIRG-08-88733 (ALJ), R01-AG034962 (ALJ), R01-NS100980 (ALJ), K24-AG046373 (ALJ), F31-AG066358 (CWB), T32-AG058524 (CWB), UL1-TR000445 and UL1-TR002243 (Vanderbilt Clinical Translational Science Award), S10-OD023680 (Vanderbilt’s High-Performance Computer Cluster for Biomedical Research), S10-OD021771 (VUIIS Center for Human Imaging), P20-AG068082 (Vanderbilt Alzheimer’s Disease Research Center), Richard Eugene Hickman Alzheimer’s Disease Research Endowment, and the Vanderbilt Memory and Alzheimer’s Center.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TJH serves on the Scientific Advisory Board for Vivid Genomics, Deputy Editor for Alzheimer’s and Dementia: TRCI, and Senior Associate Editor for Alzheimer’s and Dementia. LTD serves as a consultant for Nashville Biosciences, LLC. ALJ has served as an advisor for Lantheus — Diagnostic and Therapeutic Innovations. DL is a member of the Data Monitoring Committee for a drug study sponsored by Regeneron Pharmaceuticals. All remaining authors have nothing to disclose.
