Abstract
Tissue hypoxia and blood–brain barrier (BBB) dysfunction are key features of transient ischaemic attack (TIA) and ischaemic stroke. The neurovascular unit maintains brain homeostasis and coordinates stress responses. Extracellular vesicles (EVs) are emerging as important mediators of cell communication in hypoxia, impacting BBB integrity and enabling bidirectional movement. This study examined EV production by neurovascular cells in normoxia (21% O2) and hypoxia (1% O2) and compared these profiles with circulating EVs in TIA patients. Human brain endothelial cells and astrocytes were cultured under normoxic or hypoxic conditions for up to 24 h. EVs were isolated and analysed via nanoparticle tracking and flow cytometry. A co-culture transwell model assessed BBB permeability under controlled experimental conditions. Circulating EVs from TIA patients, TIA mimics and healthy controls were analysed for cell origin, phenotype and function. Hypoxia increased EV production from endothelial cells, disrupted BBB integrity and promoted EV movement across the barrier. TIA patients had distinct EV profiles, with elevated endothelial-derived (CD9+/CD144+) and astrocyte-derived (CD9+/GFAP+) EVs and proteins. Both cell- and patient-derived EVs enhanced clot formation and resistance to lysis. These findings suggest EVs contribute to post-TIA thrombotic risk. Astrocyte-derived EVs may serve as rapid, cost-effective biomarkers to distinguish TIA from mimics.
Introduction
Stroke remains a major public health burden, affecting over 100,000 individuals annually in the UK, often resulting in long-term disability and reduced quality of life.1,2 An additional 46,000 individuals experience a transient ischaemic attack (TIA) annually. TIA, a temporary episode of cerebral ischaemia, is frequently described as a warning sign for stroke. Although symptoms typically resolve within 4 h, up to 25% of TIA patients experience an ischaemic stroke (IS) within 12 months, with the highest risk occurring in the first 24 h.3–5
UK national guidelines, including NICE NG128 and the National Clinical Guidelines for Stroke 6 recommend urgent assessment and intervention – ideally within 24 h post-TIA, including aspirin initiation and specialist referral. However, significant delays persist, particularly in Wales, where stable patients are often referred to TIA clinics weeks after symptom onset. These guidelines also emphasise secondary prevention through medication and lifestyle modification, yet post-TIA follow-up remains inconsistent and limited. A key clinical challenge is identifying TIA patients at highest risk of subsequent stroke, enabling targeted interventions to mitigate healthcare and societal impacts.
IS and TIA share a common mechanism: vascular occlusion leading to cerebral hypoxia, resulting in impaired glucose and oxygen delivery, causing cellular activation and potential neurodegeneration. 7 Stroke patients exhibit a hypercoagulable state, and emerging evidence suggests TIA patients may exhibit similar profiles, with elevated fibrinogen levels and dysregulated clotting dynamics being observed post-TIA and IS.8–10
Extracellular vesicles (EVs) are membrane-bound particles released by most cell types and are increasingly recognised as a form of cell-to-cell communication and as key players in cardiovascular disease progression, 11 yet their role in TIA remains underexplored. EVs contribute to coagulation and inflammation, making them potential diagnostic, prognostic and therapeutic targets. Multiple studies have shown EVs activate both intrinsic and extrinsic coagulation pathways10,12–14 and may increase in number following stroke.10,12,14–17 Some studies correlate EV levels with infarct size and stroke severity, supporting their potential as biomarkers of recovery. 18
The pro-coagulant activity of EVs in TIA remains unknown and forms the focus of the PREDICT-EV clinical study. 19 Hypoxia, a hallmark of TIA and IS, triggers EV release from endothelial cells (ECs). 20 Hypoxia-induced endothelial EVs (CD9+/CD144+/CD62E+) exhibit a procoagulant phenotype, including surface expression of tissue factor (TF), which initiates the extrinsic coagulation cascade.4,12,13 These EVs enhance platelet activation, promote thrombin generation and exhibit resistance to fibrinolysis.21,22 They are found in thrombogenic settings such as atherosclerotic plaques and acute coronary syndrome plasma,23–26 supporting their role in clot risk enhancement.
Astrocytes also secrete EVs under physiological conditions that contribute to neuroprotection, neurogenesis and angiogenesis.27–29 Astrocyte-derived EVs facilitate communication with ECs and pericytes, helping maintain the blood–brain barrier (BBB). Although the role of astrocyte–EVs beyond the CNS is less clear, evidence suggests they can cross a compromised BBB and may adopt different functions in peripheral circulation. 29
In neuroinflammatory conditions, astrocyte–EVs have been shown to initiate systemic immune responses. In a mouse model, Dickens et al. demonstrated these cross the BBB and target PPARα in peripheral organs, such as the liver, altering NF-κB signalling to drive leukocyte recruitment back to the brain to initiate repair pathways. 30 This highlights their direct role in brain–immune system communication. Edwardson et al. recently reported elevated astrocyte–EVs at 5, 15 and 30 days post-stroke compared to controls, suggesting their potential as biomarkers of BBB disruption. 31 Another study linked increased brain-derived EV (expressing glial fibrillary acidic protein (GFAP) – a protein primarily produced by astrocytes/glial cells in the brain) to more severe stroke symptoms, proposing a role in assessing BBB damage and stroke severity. 17 Speculatively, appearance of brain-derived EV in the systemic circulation may reflect hypoxia-induced BBB compromise following TIA.
This study aims to investigate whether hypoxia-induced EVs contribute to the increased coagulation risk observed in TIA patients. To this end, we developed an in vitro BBB model to examine EV production and function under short- and long-term hypoxia, and compared findings to clinical data from TIA patients, TIA mimics and healthy controls.
Methods
Cell culture
The primary immortalised human microvasculature brain ECs (HBEC-5i) and primary immortalised human astrocytes (SVG-p12) were purchased from ATCC, UK. As immortalised primary cell lines no ethical approval was required. ECs were maintained in 90% Dulbecco’s Modified Eagle Medium–F12 (DMEM–F12; GIBCO, UK) supplemented with 10% (v/v) foetal bovine serum (FBS; PAA Laboratories, UK), 1% streptomycin/penicillin (Invitrogen, UK) and 40 µg/mL of endothelial growth supplement (Merck, UK). Mono and co-cultures were shown through histological staining to express junctional proteins integral to BBB integrity (shown in Supplementary Material S1).
Astrocytes were maintained in Essential Modified Eagle Medium (EMEM) supplemented with 10% FBS and 1% streptomycin/penicillin. Both cell types were incubated at 37 °C in 21% O2/5% CO2. At ~90% confluence cells underwent passage using trypsin–EDTA (Invitrogen, UK). Cells exposed to hypoxia using an InVivo2 hypoxic workstation 400 (Baker Ruskinn, UK) and maintained at 1% O2 for 1, 2, 4 or 24 h. Separate mono- or co-culture wells were set up in order to allow for TEER measurement and EV isolation from supernatant at each time point, and pairwise analysis of normoxia versus hypoxia at each time point and across the experiment. One percent O2 was chosen specifically as cells retain >95% viability over 24 h, and for its equivalence to hypoxic conditions in vivo (pO2 ~7.6 mmHg) which is characteristic of ischaemic conditions, having been extensively established by ourselves and others.20,22,32
In vitro model of the blood–brain barrier (BBB)
This model was adapted from that described in detail previously. 33 In brief, cell culture insert membranes (Millicell®, Germany; 3 µm pore size at a concentration of 2 × 106 pores/cm2, 12 mm diameter) were coated with poly-L-lysine (2 µg/cm2) on the basolateral membrane and incubated for 1 h before trans well inserts were flipped inside the plates and the bottom plate removed. Three × 105 astrocytes in <100 µL of media were added to the basolateral membrane and returned to the incubator for 2–3 h for cells to adhere. Post incubation, the excess media was removed from the inserts, the bottom of the plate returned and flipped back over. Astrocyte medium was added on both sides and returned to the incubator until cells reached ~90% confluence. Once confluent, astrocyte medium was removed from the apical compartment and 100 µL of 0.1% gelatin was added before 1 × 105 endothelial cells in 100 µL of media were added to the apical membrane of the insert. The plate was returned to the incubator for a minimum of 5 h before topping the apical chamber up with 700 µL of endothelial cell medium. Cell growth was monitored until the formation of a continuous endothelial monolayer was formed.
Trans-endothelial electrical resistance (TEER)
TEER was used to assess barrier integrity of astrocyte and endothelial cell co-cultures and was measured daily until the threshold value of 60–80/cm2 Ohms was attained, and taken to reflect physiological BBB conditions. 33 Once the TEER threshold was reached and remained stable over 24 h, co-culture wells were set up pairwise and exposed to experimental conditions (i.e. hypoxia or normoxia). TEER was subsequently measured in co-culture wells at 0, 1, 2 and 4 h (representing hypoxia in a TIA model) and 0 and 24 h (representing an IS model). TEER measurement was via an endothelial volt-ohm meter (Millicell-ERS, EMD Millipore, Billerica, MA) with a chopstick electrode (Millicell ERS probe; EMD Millipore, Billerica, MA). Prior to measurement, the electrode was sterilised by immersion in 70% industrial methylated spirits for 15 min followed by three washes in DMEM media. The electrode was lowered into the trans well at a 90° angle with the longer tip outside and the shorter tip on the inside of the trans well insert. Wells containing trans well insert and media only acted as the background and the subsequent value subtracted from TEER readings of the co-cultures. Unit area resistance was calculated by dividing the resistance value (Ohms) by the insert membrane area of 1.1 cm2, giving values expressed as Ω.cm2 (experimental set up shown in Supplementary Material S2). In order to mimic the effects of hypoxia on BBB integrity, the mimetic agent Desferrioxamine was added to the culture medium (DFO; 100 µM under normoxic conditions) for 1, 2, 4 and 24 h (Supplementary Material S3).
Permeability (dextran–FITC) assay
To assess the integrity and intercellular permeability of the BBB co-culture model, dextran–FITC (70 kDa, Sigma–Aldrich, UK) at a concentration of 1 mg/mL was added to the apical or basolateral chamber at t = 0. Then 10 µL aliquots were sampled from the apical and basolateral chambers of the same culture well at 1, 2, 4 and 24 h into a 96 well-plate and diluted 1:10 with dH2O. Co-culture experiments were set up pairwise to enable comparison of all time points within a given well, in either normoxia or hypoxia. A trans well with media only was used as a negative control and the reading subtracted from sample values. Sample fluorescence was measured using a CLARIOstar (Labtech BMG, UK) at Ex 485 nm/Em 535 nm to track the dextran–FITC.
Extracellular vesicle isolation
Cell culture
Cultured cells were washed and pre-incubated in serum free media for 24 h before EV isolation, to effectively remove contaminating particulates. The supernatant was then removed and subjected to differential ultracentrifugation as previously described.20,22,34 Firstly, the supernatant was spun at 1000g to remove any detached cells, followed by 15,000g for 20 min to eliminate cell debris or apoptotic vesicles. Lastly, the supernatant was ultracentrifuged at 100,000g for 1 h at 4 °C, using a Sorvall Discovery 100SE ultracentrifuge (Thermo Fisher Scientific, UK) Rotina 380, to pellet EVs. The resultant EV pellets were resuspended in 1× PBS, double filtered using a 0.22 µm filter unit, typically at a 40-fold concentration (~25 µL PBS/mL of supernatant centrifuged).
Participant plasma
Whole blood samples drawn in Vacutainers containing citrate as anti-coagulant were centrifuged at 800g for 15 min to render the sample acellular, without causing platelet activation, before being centrifuged twice further at 2500g for 15 min. One mL of a-cellular plasma was processed to EV using Size Exclusion Chromatography (SEC) added to the top of the SEC column followed by 5 mL of PBS. Five hundred µL fractions were collected and fractions 5 to 10 were harvested to yield a concentrated EV sample relatively free of plasma-based contaminants (as we have employed previously and in accordance with ISEV guidelines35,36).
EV characterisation – nanoparticle tracking analysis (NTA)
EVs were analysed using a NanoSight LM10 Microscope (Malvern, UK) equipped with a 488 nm (blue light) laser and NTA software (v3.4). For accurate determination of size and concentration, EVs were diluted in PBS in the range of 107–109 EVs/mL. Each count was replicated five times. An automatic syringe pump (Harvard Apparatus, UK) was used to administer the sample at 20 µL/min over 60 s.
EV characterisation – flow cytometry
To confirm the cellular origin and character of cell derived EV, a concentration of 1 × 106 EVs/mL were labelled with 20 µL of PE-anti-CD9 (BD Pharmingen, USA) and 5 µL secondary APC labelled antibody (APC-anti-CD31, APC-anti-CD301, APC-anti-CD144, APC-anti-GFAP, Biolegend, UK; APC-anti-S100B, NOVUS, Biotechne; or APC-anti-vimentin, Mitenyl Biotec) for 30 min in the dark before collecting 20,000 events within the established CD9+ violet side scatter size gate to confirm EV identity, using a CytoFlex Cytometer (Beckman Coulter, UK).
For patient plasma EV samples, the cellular origin of EVs was also assessed. Briefly, 80 µL of plasma EV sample was labelled with 20 µL of PE-anti-CD9 and 5 µL of either APC-anti-CD144 (endothelial) or APC-anti-GFAP (astrocytes; Biolegend, UK).
Fluorescent labelling of EV for BBB experiments
One µL of Carboxyfluorescein Diacetate Succinimidyl Ester (CFSE) Cell Division Tracker (Biolegend, UK) was added to 10 µL of EVs at a concentration of 1 × 1010/mL and incubated at room temperature in the dark. The fluorescence was quenched by adding 90 µL of PBS to give a final concentration of 1 × 108 EVs/mL. Depending on the direction of transfer tested, 1 × 107 EVs/mL (10 µL) was added to the apical or basolateral compartment of the trans well. Following incubation 50 µL of culture supernatant was sampled from each side of the trans well and analysed using flow cytometry periodically (0, 1, 2, 4 and 24 h). Samples were labelled with anti-CD9-PE (20 µL) and incubated in the dark for 30 min. A flow cytometry gate was set up for CD9+ (PE) and CFSE+ (FITC) EV and 20,000 events captured. The total fluorescence was calculated for each well and results were expressed as ‘% of total fluorescence transfer’ across the cell barrier.
Turbidimetric clotting and lysis assay
Turbidometry was employed to analyse the effect of cell- or patient-derived EV on clot formation and lysis as previously described. 37 Briefly, EVs isolated from cells or patient plasma were added to healthy pooled human plasma at a normalised concentration of 1 × 109 EVs/mL. Thrombin, calcium and tissue plasminogen activator (tPA) were added at a final concentration of 0.03 U/mL thrombin, 7.5 mM CaCl2 and 83.2 ng/mL tPA, in a total assay volume of 150 µL. Samples were run in duplicate and pooled plasma alone (without EV added) was run as a negative control. Absorbance was measured every 12 s using a CLARIOstar (Labtech BMG, UK) at 340 nm over 1 h.
Patient cohort
Ethical considerations
This study was conducted according to the principles and guidelines of Declaration of Helsinki. On meeting the inclusion criteria patients were invited to participate in the study and provided written study information (ethical approval by IRAS, ref #281530, NHS REC approval number 21/wm/0044). A member of the R&D team was at hand to address any questions or concerns.
Healthy control subjects (N = 30) were recruited from a multi-phased screening programme at Cardiff Metropolitan University (ethics ID; Sta-2860).
Consent to participate
All participants gave informed and written consent prior to completing a health questionnaire to evaluate underlying co-morbidities, cardiovascular risk profiles, family history and other general health information.
TIA patients (N = 35) were recruited as part of the PREDICT-EV study 19 from TIA outpatient clinic, accident and emergency or direct from acute stroke wards in Cwm Taf Morgannwg Health Board, Wales, UK. TIA was first confirmed by the leading stroke clinician, according to clinical guidelines. A further group of patients (N = 30) who attended TIA clinic having been referred but who were subsequently diagnosed as not having suffered a TIA were also recruited. This group is referred to as ‘TIA mimics’.
Soluble GFAP analysis
Plasma concentration of soluble GFAP (sGFAP) was assessed using a commercially available enzyme linked immunosorbent assay (ELISA) kit (Thermo Fisher, UK) according to manufacturer’s instructions.
Statistics
All data presented in this paper was analysed using GraphPad Prism software and results are expressed as mean ± SD, for N number of experiments (each N representing n = 3 within experimental replicates). Following confirmation of normal distribution, the mean of two independent groups was analysed using an unpaired Student’s t-test. Where there were three or more groups, either a one-way or two-way ANOVA was used, as appropriate followed by a Tukey’s post hoc test to determine statistical significance. Categorical variables were compared between TIA and TIA-mimic groups using a Pearson’s chi-squared test. Patient data is presented as (healthy control vs TIA mimic vs TIA) unless otherwise stated. A p ⩽ 0.05 was regarded as statistically significant. *p ⩽ 0.05, **p ⩽ 0.01, ***p ⩽ 0.001, ****p ⩽ 0.0001.
Results
Effect of short- and long-term hypoxia on BBB EV production and character
Astrocytes exposed to hypoxia (1, 2, 4 and 24 h) showed no significant change in EV production compared to normoxia (t = 1: 540.33 ± 42.77 vs 535.33 ± 348.22 EVs/cell, t = 2: 631.67 ± 243.66 vs 390.67 ± 231.85 EVs/cell, t = 4: 766.00 ± 445.06 vs 973.33 ± 341.06 EVs/cell, t = 24: 2786.33 ± 229.07 vs 2444.00 ± 280.71 EVs/cell, p > 0.05 for all comparisons; Figure 1(a)). However, there was a significant increase in the production of astrocyte EV overtime (p < 0.001), independent of oxygen condition. There was also no significant change in the production of EV from endothelial cells at 1, 2 and 4 h in hypoxia, however, at 24 h significantly (p < 0.001) more endothelial EV were produced in hypoxia when compared with normoxia (t = 1: 514.67 ± 120.67 vs 448.67 ± 47.01 EVs/cell, t = 2: 510.67 ± 262.92 vs 431.33 ± 4.16 EVs/cell, t = 4: 850.00 ± 135.35 vs 496.00 ± 5.57 EVs/cell, p > 0.05 for all, t = 24: 5121.00 ± 279.38 vs 3598.00 ± 371.74 EVs/cell, p < 0.001; Figure 1(b)). There was also a significant (p < 0.001) increase in endothelial EV production over time, independent of oxygen condition. There was no significant difference in the mean astrocyte EV size in hypoxia compared with normoxia, at any time point or overtime (Figure 1(c)). Endothelial EV size was not significantly different comparing hypoxia to normoxia at any time point; however, EV became significantly (p < 0.05) larger over time in both conditions (Figure 1(d)).
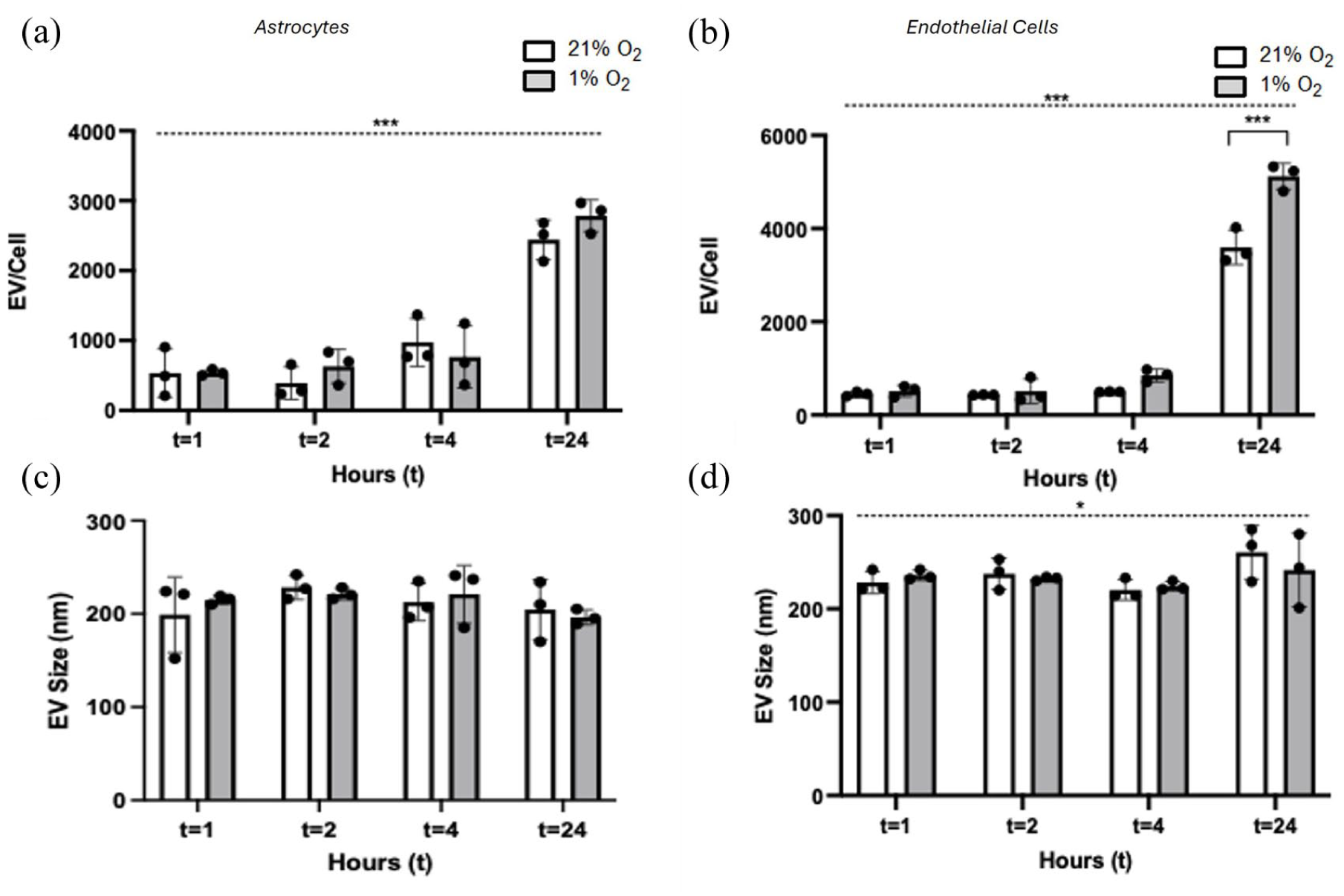
The effect of short-term and long-term hypoxia on EV production and size in primary immortalised astrocytes (left column) and endothelial cells (right column): (a) astrocyte EV production per cell subjected to normoxia (21% O2) and hypoxia (1% O2) for 1, 2, 4 and 24 h, (b) endothelial cell EV production per cell subjected to normoxia and hypoxia for 1, 2, 4 and 24 h, (c) the mean size of astrocyte EVs produced under normoxia and hypoxia conditions at 1, 2, 4 and 24 h and (d) the mean size of endothelial cell EVs produced under normoxia and hypoxia conditions at 1, 2, 4 and 24 h. Two-way ANOVA was performed and a post hoc Bonferroni test was applied. Results represent (N = 3) for all. Bar charts represent mean ± SD. Dotted line suggests relationship over time, independent of oxygen condition.
Effect of short- and long-term hypoxia on BBB integrity and permeability
Endothelial cells and astrocytes were co-cultured to confluence in the BBB model and exposed to hypoxia for 1, 2, 4 and 24 h. TEER was maintained at both 1 and 2 h in hypoxia, however, a significant decrease in TEER was observed in hypoxia at both 4 and 24 h (p < 0.001) when compared to normoxia (75.57 ± 7.78 vs 84.03 ± 8.44 Ω.cm2, 71.87 ± 0.64 vs 77.37 ± 3.54 Ω.cm2, 46.93 ± 2.29 vs 78.83 ± 3.54 Ω.cm2, 25.67 ± 5.54 vs 81.03 ± 2.78 Ω.cm2; Figure 2(a)). There was also a significant (p < 0.001) interaction between oxygen condition and TEER over time.
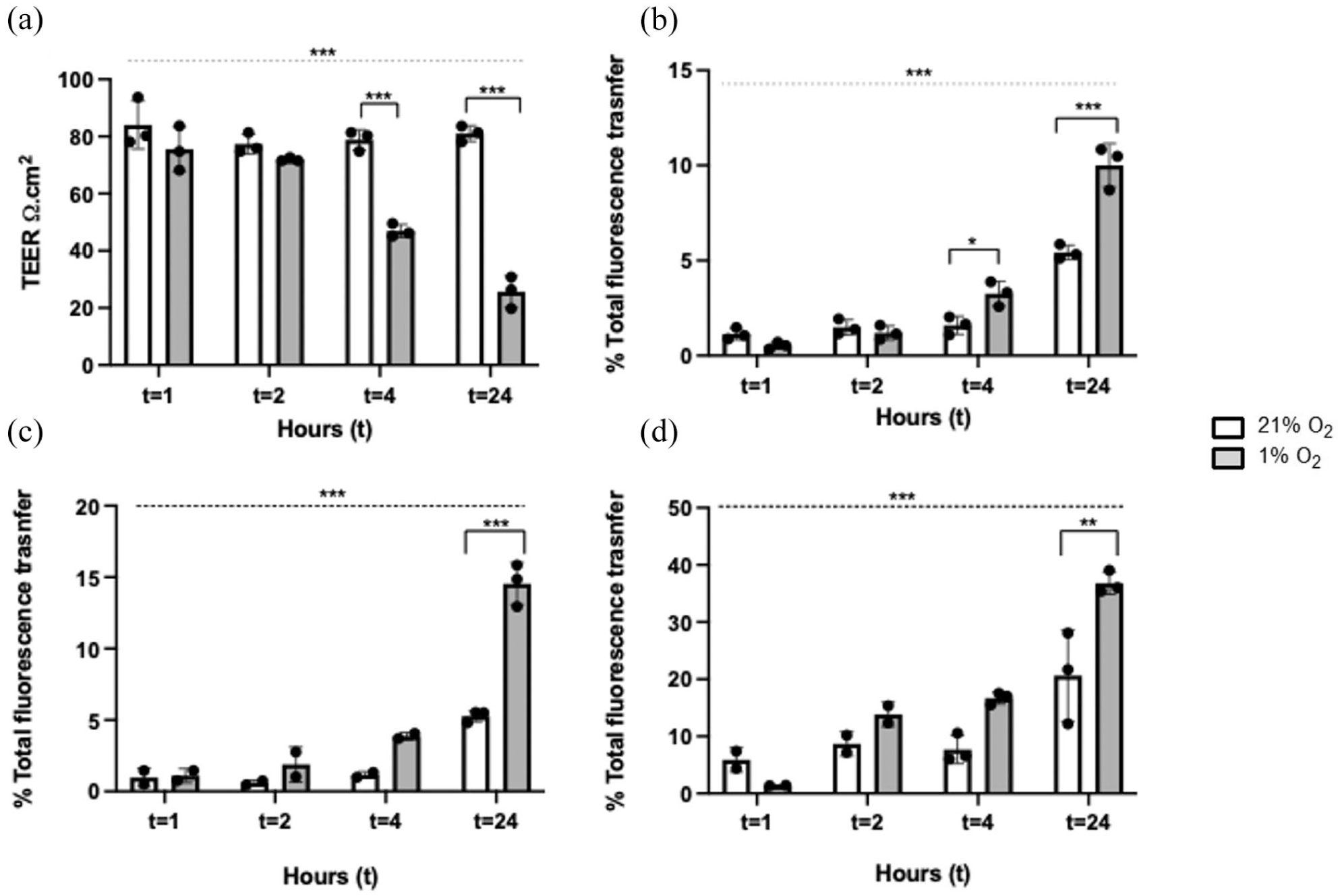
The effect of short-term and long-term hypoxia exposure on BBB integrity and permeability: (a) TEER measured in astrocyte and endothelial co-cultures exposed to hypoxia (1% O2) or normoxia (21% O2), over 1, 2, 4 or 24 h (t), (b) co-culture permeability to fluorescent dextran–FITC added to the apical chamber and the BBB exposed to either hypoxic or normoxic conditions, over time. Fluorescence determined in the basolateral compartment, (c) transfer of fluorescently labelled astrocyte EV from apical to basolateral compartment and (d) transfer of fluorescently labelled endothelial cell EV from basolateral to apical compartment. Two-way ANOVA was performed and a post hoc Bonferroni test was applied. Results represent (N = 3) for all. Bar charts represent mean ± SD. Dotted line suggests relationship over time, independent of oxygen condition.
Paracellular permeability across the BBB on exposure to hypoxia was assessed using dextran–FITC fluorescence tracing. No change in dextran–FITC permeability was observed at 1 or 2 h hypoxia compared to normoxia (t = 1: 0.53% ± 0.17% vs 1.14% ± 0.31%. t = 2: 1.2% ± 0.37% vs 1.49% ± 0.40%, p > 0.05) however, at 4 h a statistically significant increase in % of total fluorescence transferred to the basolateral chamber was observed (3.26% ± 0.66% vs 1.60% ± 0.48%, p < 0.001). A statistically significant increase in % of total fluorescence transfer to the basolateral chamber was also observed at 24 h hypoxia compared to normoxia (10.34% ± 1.15% vs 5.34% ± 0.38%, p < 0.001; Figure 2(b)). There was also a significant (p < 0.001) interaction between oxygen and dextran–FITC transfer over time.
Transfer of fluorescently labelled astrocyte EV from apical to basolateral compartment was measured in hypoxia over 1, 2, 4 and 24 h and compared with normoxia. No difference in the transfer of astrocyte EV, in hypoxia was observed in 1, 2 or 4 h. However, a significant (p < 0.001) increase in astrocyte EV transfer was observed following 24 h of hypoxia (14.54% ± 1.48% vs 5.28% ± 0.40%; Figure 2(c)). Similarly, fluorescently labelled endothelial EV transfer from the basolateral to apical compartment was measured. Results mirror those seen in astrocyte EV, with only a significant increase in EV movement observed after 24 h in hypoxia, when compared with normoxia (36.82% ± 1.97% vs 20.66% ± 7.99%, p < 0.01; Figure 2(d)). There was a significant (p < 0.001) interaction between oxygen and astrocyte EV (Figure 2(c)) or endothelial cell EV (Figure 2(d)) transfer, over time.
Impact of cell-derived EVs on clot formation and lysis
EVs derived from astrocytes or endothelial cells both in normoxia and hypoxia significantly reduced time to clot initiation (lag time) when compared to the no EV control (astrocyte EV; 177.38 ± 26.01 vs 197.38 ± 66.49 vs 528.00 ± 42.54 s, p < 0.001; endothelial EV; 324.00 ± 71.50 vs 294.80 ± 105.7 vs 528.38 ± 42.54, p < 0.001; Figure 3(a)). The maximal clot size, represented by maximum optical density (max OD) remained the same as the no EV control when either EV subtype was added under normoxic or hypoxic conditions (Figure 3(b)). The time taken for optical density to reach it’s max OD (taken to reflect the rate of clot formation) was significantly reduced when astrocyte EV derived under normoxia or hypoxia were compared to the no EV control (9.68 ± 2.59 vs 10.27 ± 2.17 vs 16.22 ± 1.75 min, p < 0.05 for both; Figure 3(c)). However, astrocyte EV derived from normoxia and hypoxia were comparable with each other. This was significantly faster compared to endothelial derived EV under similar conditions, respectively (14.42 ± 0.69 vs 9.6 ± 2.59 min, p < 0.01 for normoxic; 13.91 ± 1.59 vs 10.27 ± 2.17 min for hypoxic, p < 0.05). Further, EV derived from endothelial cells had no impact on the time to maximal clot formation. The time to 50% lysis from max OD (taken to reflect rate of clot breakdown) was significantly longer for endothelial EV derived from hypoxia compared to the no EV control (25.4 ± 4.56 vs 8.64 ± 3.83 min, p < 0.05; Figure 3(d)) whereas endothelial EV derived from normoxia, and astrocyte EV derived in either normoxia or hypoxia, had no impact on clot lysis.
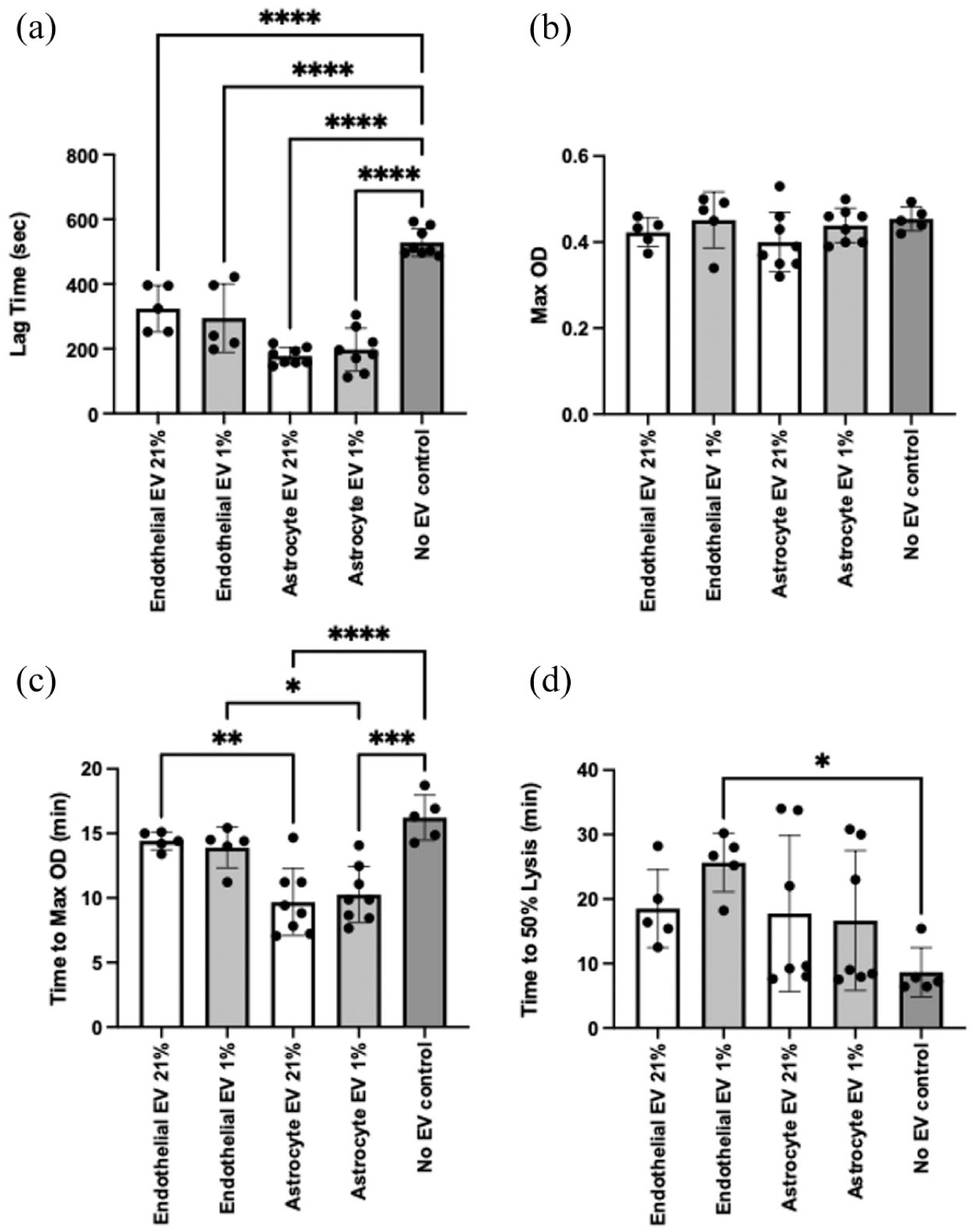
The effect of normoxic and hypoxic endothelial and astrocyte derived EVs on clot formation and lysis. Endothelial (N = 5) and astrocyte EVs (N = 7) produced under normoxic (21% O2) or hypoxic (1% O2) conditions were incubated with platelet poor plasma before clot formation was initiated with a combination of thrombin and calcium, and lysis induced by tPA. Results were compared to a no EV plasma control (N = 5). (a) Lag time to clot initiation, (b) max OD, representing maximal clot formation, (c) time to max OD and (d) time to 50% lysis. One-way ANOVA was performed, and a post hoc Bonferroni test was applied. Bar charts represent mean ± SD.
Patient studies
Participant characteristics
General cohort characteristics for each group (TIA, TIA mimics and healthy controls) are detailed in Table 1. The mean ages of participants in all three groups were significantly different (p < 0.001) from one another. The healthy control group exhibited the lowest prevalence of cardiovascular risk factors, and the TIA group exhibited the highest prevalence, of which only 5.3% reported no co-morbidities. Medications relevant to cardiovascular risk factors confirms significantly more TIA were being prescribed anti-hypertensives (p < 0.001), anti-platelets (p = 0.001) and statins (p = 0.09) compared to TIA mimics at presentation with TIA symptoms.
Cohort characteristics.
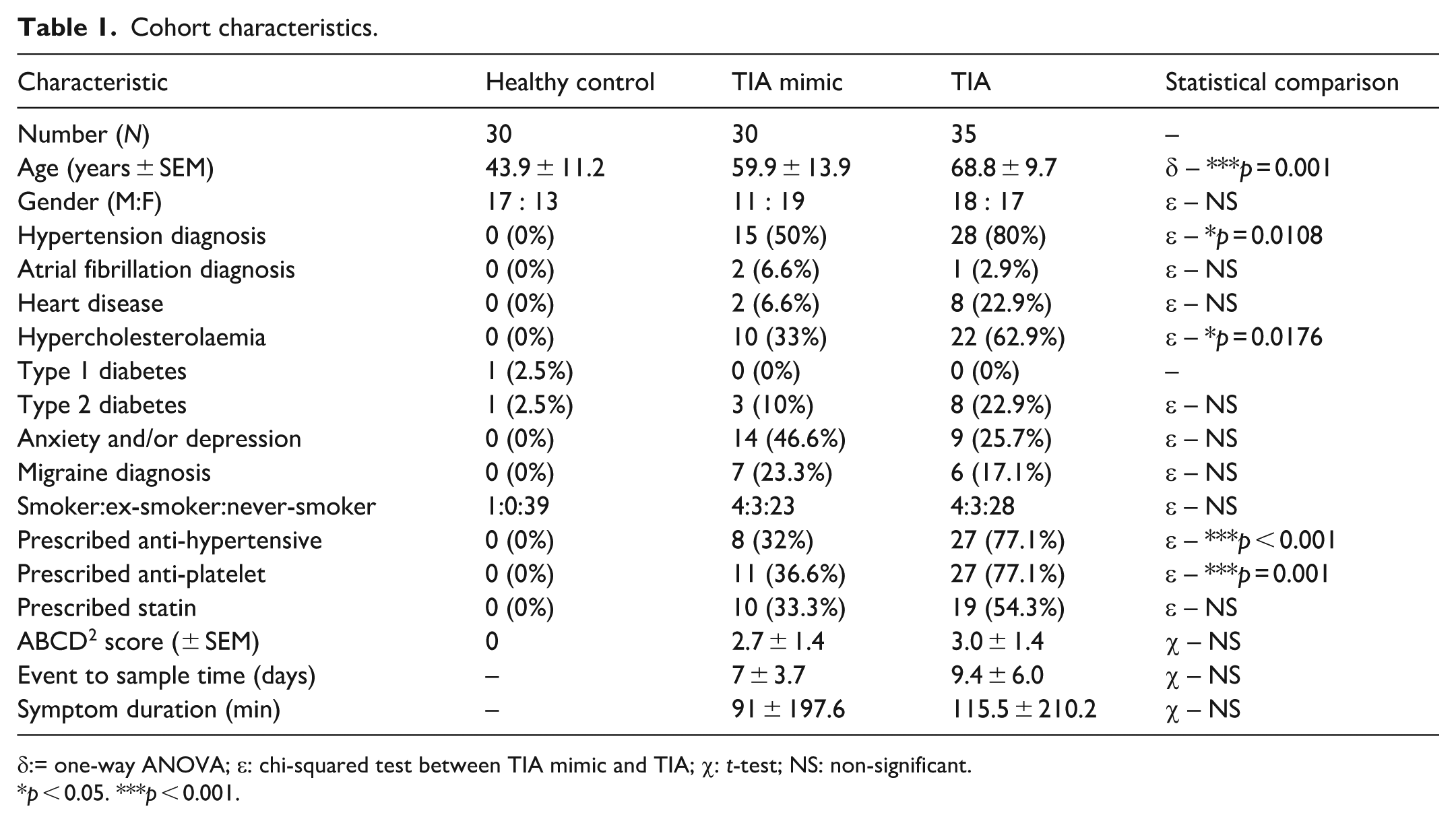
δ:= one-way ANOVA; ε: chi-squared test between TIA mimic and TIA; χ: t-test; NS: non-significant.
p < 0.05. ***p < 0.001.
Event characteristics for TIA patients and TIA mimics are also detailed in Table 1. The time between event and assessment at TIA clinic (hence recruitment to the study) was similar for TIA patients (9 days) and TIA mimics (7 days). No significant difference in symptom duration or ABCD2, a cumulative risk of stroke score, did not significantly differ comparing TIA patients with TIA mimics.
Haematological screening
TIA patients and TIA mimics had significantly longer pro-thrombin (PT) time when compared to healthy control subjects (13.79 ± 0.88 vs 13.71 ± 0.80 vs 11.55 ± 0.66 s, p < 0.001 and p < 0.05, respectively; Figure 4(a)). APPT was significantly higher in healthy controls compared to TIA mimics, whereas TIA patients did not show a different APPT (33.09 ± 3.34 vs 30.47 ± 3.38 vs 30.81 ± 4.80 s, p < 0.05; Figure 4(b)). Fibrinogen concentration was significantly increased in both TIA mimic and TIA patients in comparison to healthy controls (3.48 ± 1.13 vs 3.85 ± 0.86 vs 2.42 ± 0.68 g/L, p < 0.001 for both; Figure 4(c)). D-dimer concentration was significantly increased in TIA patients in comparison with healthy controls, whereas TIA mimics did not significantly differ (0.29 ± 0.22 vs 0.44 ± 0.24 vs 0.57 ± 0.63 µg/L, p < 0.05; Figure 4(d)).
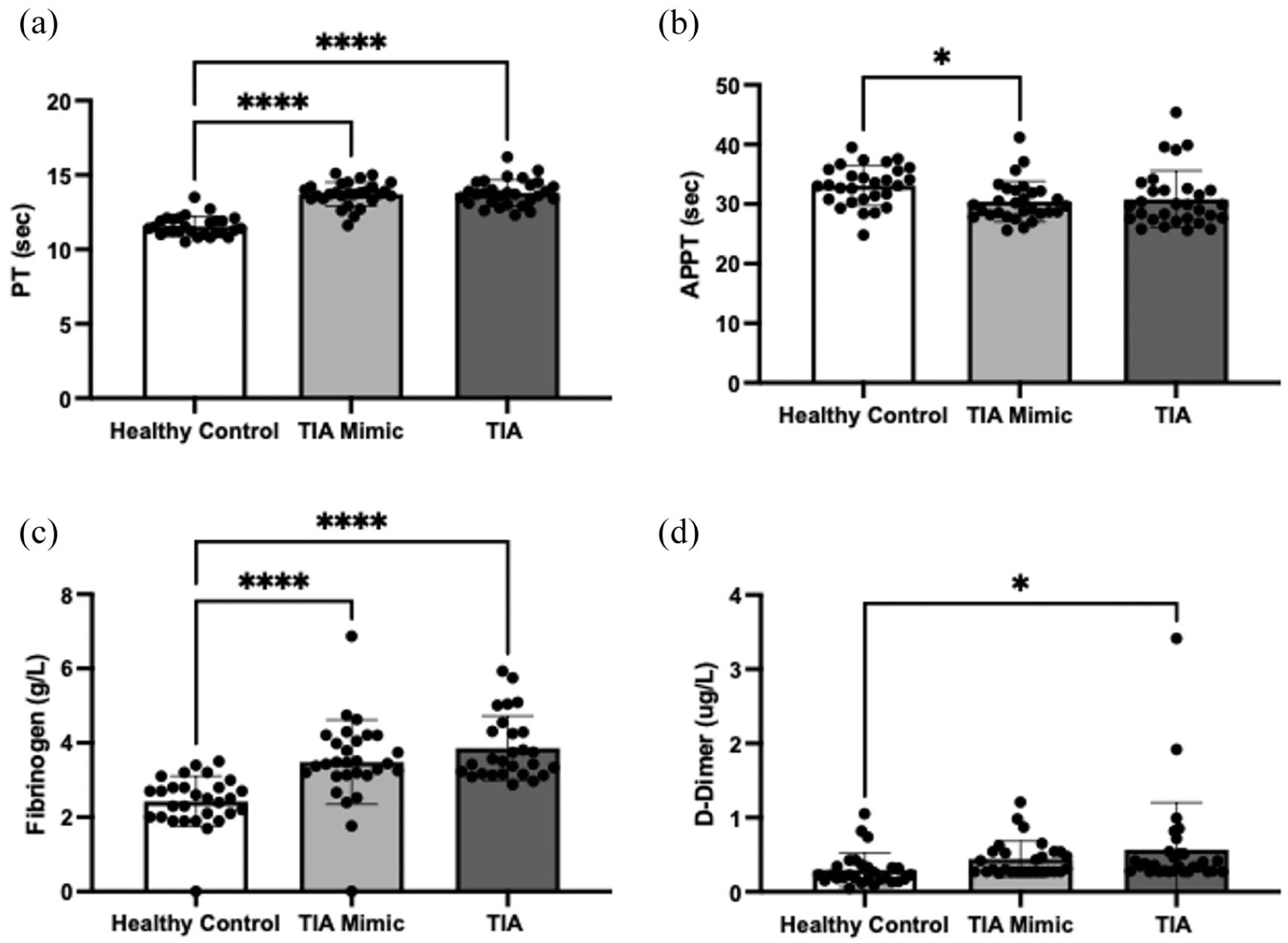
Haematological screening. Coagulation screening was used to determine changes in coagulative capacity in healthy controls (N = 30), TIA mimics (N = 30) and TIA patients (N = 35): (a) PT (s), (b) aPPT (s), (c) fibrinogen level (g/L) and (d) D-dimer concentration (µg/L) is shown. Bar charts represent mean ± SD.
Circulating levels of astrocyte- and endothelial-derived EVs
Total plasma EV concentration, represented by CD9+ events, was significantly greater in both the TIA and TIA mimics compared to healthy controls (2,230,900 ± 1,450,429 vs 1,717,000 ± 1,370,218 vs 615,228 ± 943,059 CD9+ EVs/mL, p < 0.001 and p < 0.01, respectively; Figure 5(a)).
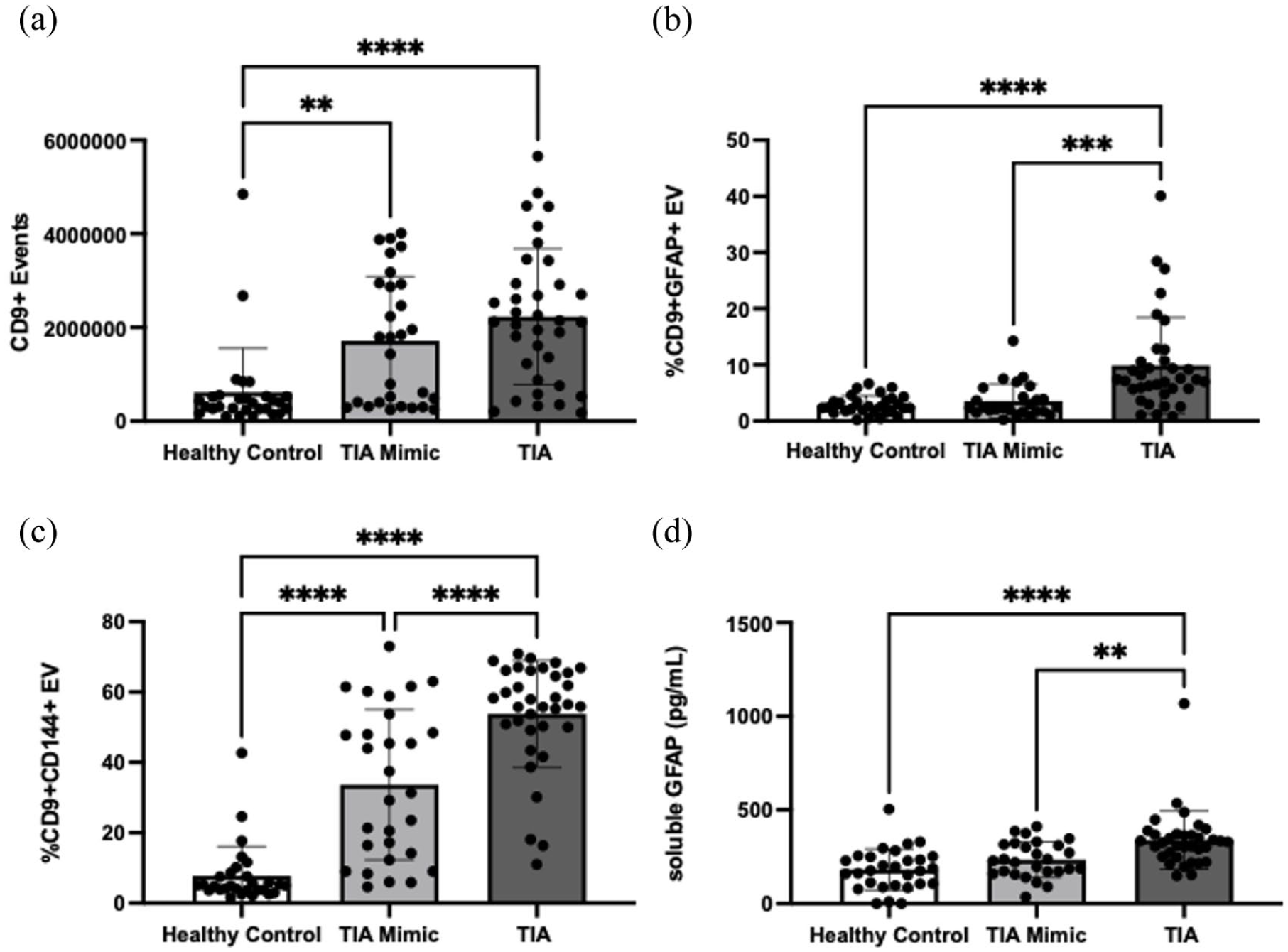
Circulating levels of Endothelial- and astrocyte-derived EV: (a) total circulating CD9+ EVs/mL, (b) circulating levels of CD9+/GFAP+ expressing EV, (c) plasma level of CD9+/CD144+ expressing EV and (d) Plasma level of soluble GFAP. All measures were assessed in healthy controls (N = 30), TIA mimics (N = 30) and TIA patients (N = 35). Bar charts represent mean ± SD.
Using CD9 to positively identify EV, the percentage of both CD9+/GFAP+ (astrocyte) and CD9+/CD144+ (endothelial) EV were determined in patient samples. TIA patients exhibited significantly greater CD9+/GFAP+ EV than both TIA mimics or healthy controls (9.90 ± 8.53 vs 3.58 ± 3.04 vs 2.82 ± 1.69, p < 0.001 for both; Figure 5(b)). Both TIA patient and TIA mimics had significantly more CD9+/CD144+ EV than healthy controls. TIA patients also exhibited significantly greater CD9+/CD144+ compared to TIA mimics (53.82 ± 15.25 vs 33.72 ± 21.40 vs 7.74 ± 8.37, p < 0.001 for all; Figure 5(c)).
The concentration of soluble GFAP in plasma was significantly higher in TIA patients compared to both TIA mimics (p < 0.01) and healthy controls (p < 0.001; 339.2 ± 155.7 vs 234.9 ± 95.29 vs 182.1 ± 110.2 pg/mL; Figure 5(d)).
There was no significant difference in circulating EV (CD9+), CD9+/CD144+, CD9+/GFAP+ or soluble GFAP levels in TIA patients on anti-platelet, anti-hypertensive or statin, compared with those not prescribed these medications.
Effect of patient-derived EV on clot formation and lysis
To assess the effect of patient EV on clot formation and lysis, EVs were isolated from participants and EV were added to pooled healthy platelet poor plasma. Clot initiation time (lag time) was significantly prolonged when TIA patient EV were added to pooled plasma, when compared with addition of either TIA mimic or healthy control derived EVs (450.8 ± 229.6 vs 323.3 ± 116.3 vs 297.3 ± 69.43 s, p < 0.01 and p < 0.001, respectively; Figure 6(a)). Maximal clot size (max OD) was significantly increased with both TIA patient and TIA mimic when compared with healthy control derived EVs (0.46 ± 0.04 vs 0.46 ± 0.06 vs 0.39 ± 0.08, p < 0.001; Figure 6(b)). The time from clot initiation to max OD was significantly longer for TIA derived EVs in comparison to both TIA mimic and healthy control EV (20.72 ± 8.29 vs 13.93 ± 3.10 vs 10.93 ± 2.24 min, p < 0.001; Figure 6(c)). Time to 50% lysis was quickest with addition of EV from healthy controls, with both TIA mimic (p < 0.01) and TIA patients (p < 0.001) EV exhibiting significantly longer lysis times (9.30 ± 1.57 vs 11.50 ± 3.02 vs 13.53 ± 3.28; Figure 6(d)). Addition of TIA patient EV led to a significantly longer lysis time in comparison to TIA mimics (p < 0.05).
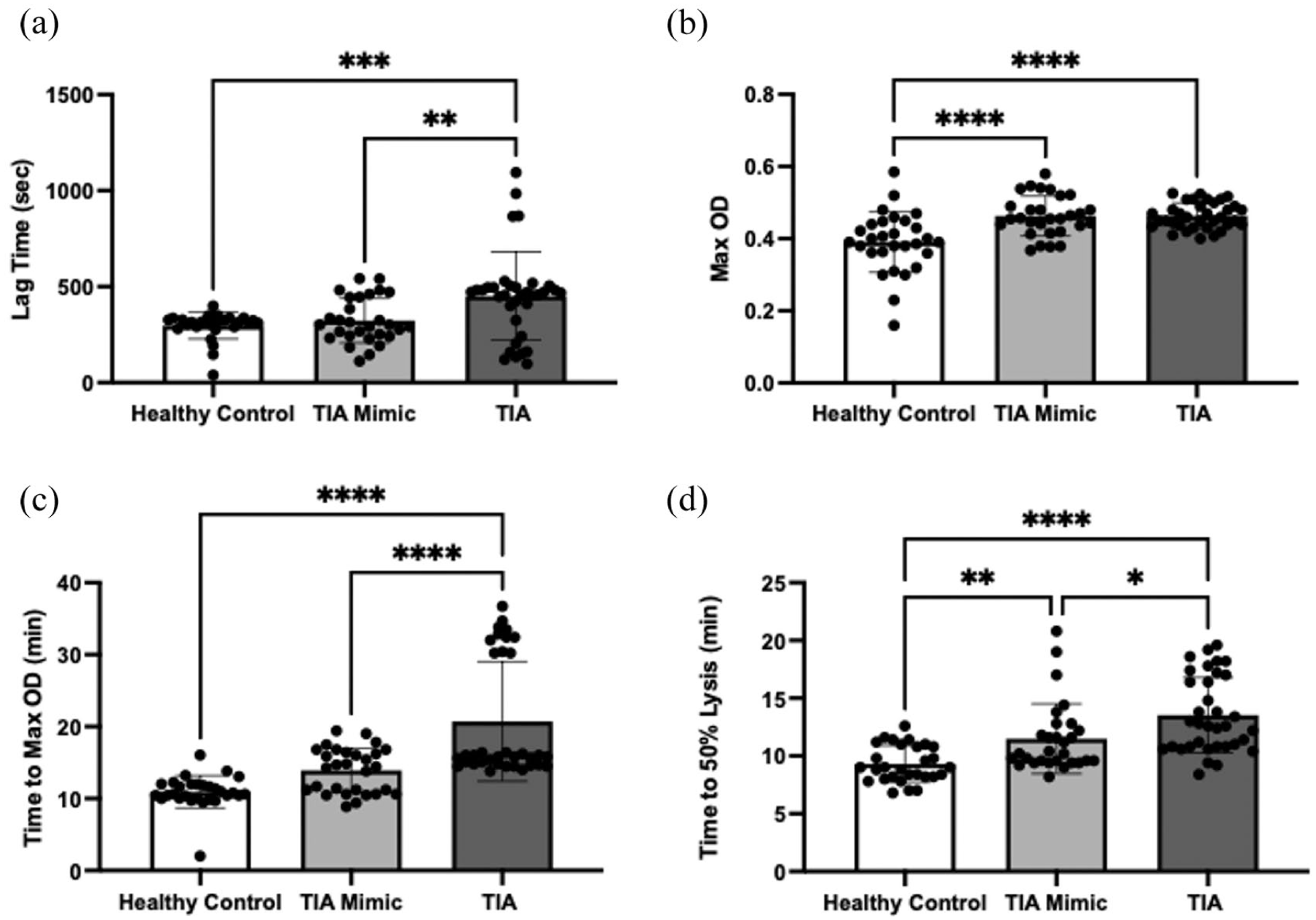
Effect of participant EV on coagulation of healthy pooled plasma. Thrombin and calcium induced turbidometry and tissue plasminogen activator-induced lysis. EVs from healthy controls (N = 30), TIA mimics (N = 30) and TIA patients (N = 35), were incubated with pooled healthy platelet poor plasma before clot initiation. A standardised EV concentration of 1 × 109 EVs/mL was used: (a) time for the clot formation to begin – lag time, measured in s, (b) maximum size of clot reached – max OD, (c) time to form maximum clot size – time to max OD measured in min and (d) time to 50% lysis of the clot formed – measured in min. Bar charts represent mean ± SD.
Discussion
This study demonstrates that hypoxic stress triggers distinct cellular and molecular responses in brain endothelial cells and astrocytes, resulting in altered EV production, BBB disruption and pro-thrombotic effects. Critically, we confirm hypoxia-induced astrocyte EV transfer across the BBB. These in vitro findings are supported by clinical data from TIA patients, which reveal EV phenotypes in blood consistent with those observed under controlled hypoxic conditions. Together, the data highlight EVs as both biomarkers and potential effectors of pathological processes and thrombotic risk in TIA.
Hypoxia-induced EV production and BBB disruption
Consistent with prior studies in non-brain endothelial cells20,22 our data confirm that hypoxia induces a time- and cell-specific increase in EV release from primary EC, but not from astrocytes. The production increase observed after 4 h of hypoxia suggests a transcriptionally regulated mechanism, likely driven by hypoxia-inducible factor 1-alpha (HIF-1α), which requires several hours to accumulate and drive target gene expression. 38 Use of the HIF stabiliser desferrioxamine (DFO) replicated hypoxia-induced barrier disruption and further supports HIF-1α involvement.20,39
The associated breakdown of barrier integrity under hypoxic conditions was demonstrated by a reduction in TEER and increased FITC–dextran permeability in co-culture models. These findings reflect in vivo data demonstrating BBB disruption following ischaemic injury, as observed in both human cerebrospinal fluid (CSF)/serum albumin ratios and in animal models.40–42 The observed bidirectional EV transfer across the barrier after 24-h hypoxia further implicates BBB dysfunction in facilitating peripheral dissemination of CNS-derived EVs.
Astrocyte EV function and pathogenic potential
Although ADEV production did not increase under hypoxic conditions, these vesicles are highly sensitive to microenvironmental cues and modulate their bio-cargo accordingly.43,44 Previous studies have shown that exposure to IL-10 promotes the release of EVs that enhance neurogenesis and survival, while pro-inflammatory cytokines such as, IL-1β and TNF-α lead to formation of EVs that suppress neuronal function and promote apoptosis.45–48 Astrocyte-EVs from ischaemia models have been found to carry miRNAs such as miR-190b, miR-92b-3p and miR-34c, which regulate autophagy, toll like receptor signalling and neuronal survival pathways.49–52 Additionally, astrocyte-EVs have been shown to transport neuroprotective proteins like apolipoproteins in response to oxidative stress, which is often intensified by hypoxia. 53
Thus, even without stimulating increased EV release, the cargo of astrocyte-derived EV produced under hypoxic or inflammatory stress can significantly impact downstream cellular responses, including those related to coagulation and inflammation.
EV transfer across the BBB and implications for circulating biomarkers
The appearance of astrocyte-EVs in the vascular compartment following hypoxia is notable, as it suggests BBB breakdown permits their release into the systemic circulation. This is supported by our clinical data, where both CD9+/GFAP+ EV and CD9+/CD144+ EV were significantly elevated in TIA patients but not in TIA mimics or healthy controls. Soluble GFAP, a protein produced exclusively by astrocytes/glial cells of the brain, a marker of astrocyte damage and BBB compromise, showed a similar increase uniquely in TIA patients. Previous studies have reported GFAP elevation in acute stroke that correlated with lesion size, NIHSS score and long-term outcomes.31,32,54–57 Importantly, our findings suggest that GFAP+ EV and soluble GFAP, may be particularly useful in distinguishing true TIAs from mimics.
Pro-thrombotic function of EVs and clotting risk
Hypoxia-induced EVs from both endothelial cells and astrocytes accelerated clot formation and increased clot strength in vitro, with endothelial EVs also enhancing resistance to tPA-mediated fibrinolysis. These findings align with previous reports demonstrating EV-driven alterations in fibrin architecture and reduced plasmin accessibility. 22 Collectively, these effects suggest that EVs may play a substantial role in stabilising thrombi, with potential clinical implications for stroke risk and responsiveness to thrombolytic therapy.
Notably, EVs isolated from TIA patients prolonged clot initiation time, despite containing elevated levels of procoagulant protein markers. This unexpected effect may be attributable to specific antithrombin III (AT-III) glycoforms present on the EV surface, which have been shown to bind thrombin and attenuate fibrin formation. 58 If validated, this would support a dual regulatory role for circulating EVs in fine-tuning thrombin activity and coagulation dynamics following transient ischaemic events.
At the same time, TIA-derived EVs significantly increased overall clot size and resistance to fibrinolysis, reinforcing the hypothesis that these vesicles contribute to a pro-thrombotic state after hypoxic insult and may predispose certain TIA patients to subsequent ischaemic events. These observations underscore the importance of EV profiling in risk stratification. Completion of the PREDICT study – which will characterise EV signatures in TIA patients and track those who subsequently develop stroke – will be critical for determining whether specific EV phenotypes can predict progression to stroke versus recovery without further events.
Clinical relevance and diagnostic potential of EV biomarkers
This PREDICT-EV cohort provided critical validation of in vitro findings and insight into the translational potential of EV markers. TIA patients exhibited elevated CD9+ and CD9+/CD144+ EV – markers previously associated with poor stroke outcomes.3,59,60 TIA patients uniquely showed elevated CD9+/GFAP+ EVs and soluble GFAP, suggesting these markers may help differentiate between true and mimicking events. Differentiating TIA from TIA mimics is a major problem facing healthcare professionals with only an estimated 50% of TIA patients referred to clinic later confirmed clinically as having suffered a TIA. Typically, this diagnosis relies heavily on patient reporting and on risk scoring systems (such as ABCD2) to determine the likelihood of stroke, as opposed to quantitative measures or confirmatory MRI (which are often inconclusive in the context of TIA). 61
Plasma or CSF levels of brain-derived metabolites have been shown to increase in several neurological disorders, including neurodegenerative disorders and traumatic brain injury (TBI).62,63 Examples of blood biomarkers that are elevated include GFAP, S-100β, neuron-specific enolase (NSE) and myelin basic protein (MBP).64–67 It is interesting therefore that Abbott developed the mTBI Alinity test which measures GFAP (and UCH-L1) for evaluating mild TBI. LVOne, which measures GFAP (and D-dimer) is the only lateral flow test currently available for point-of-care detection of sub-nanogram concentrations of GFAP, recently approved in the UK for detection of stroke patients with large vessel occlusions. 68 More research is needed to establish GFAP’s diagnostic value in TIAs and other neurological conditions, particularly in terms of prognosis and risk stratification.
Limitations
It is acknowledged that although patient-derived EV sub-types were specifically labelled they represent a proportion of the overall EV sample population (expressed as a % of CD9+ EV). Specific EV sub-types were not isolated prior to applying to in vitro tests. In our hands, methods to isolate specific EV sub-types are as yet unreliable and prone to non-specific binding (examples include immune capture or magnetic capture beads). In order to confirm cell of origin in vitro, EV from both astrocyte cells and endothelial cells were assessed for protein markers characteristic of the parent cell in normoxia and hypoxia (see Supplementary Material S4). Together with established markers of other cell types, these markers were also applied to patient EV samples. However, full proteinomic characterisation to obtain a patient EV signature was beyond the scope of this study. GFAP was used in the case of astrocyte EV; despite exhibiting relatively low expression this marker is highly specific for cells of glial origin (whereas S100B and vimentin were highly expressed but is produced by other cells). CD144 was used to identify endothelial EV as this has been used extensively by our group and others to characterise endothelial cells and EV. Importantly, hypoxia was shown not to have an effect on characteristic protein markers.
We applied well-established protocols to isolate and characterise EV in these studies, aligning with the guidelines set out by ISEV. 35 Although we acknowledge transmission electron microscopy (TEM) was not applied to analyse EV samples in these studies, we have previously undertaken detailed characterisation (including scanning EM/TEM) of endothelial-derived EV in normoxia and hypoxia and find little difference in overall structural integrity or appearance of endothelial EV as a direct result of hypoxia. 20
Patients in the TIA and TIA mimic cohorts were typically screened at ~9 and ~7 days, respectively. We recognise this is not aligned with National clinical guidelines but represents a real-life scenario within the majority of Health Boards across Wales. A small cohort of our patients (n = 5) were screened in A&E, and therefore more closely represent levels closer to the TIA event. It must be acknowledged therefore that the results reported, and our conclusions, represent a window into the condition only at this time and we are unable to extrapolate to levels at the point of TIA, or indeed, the levels prior to TIA. Nevertheless, it is interesting that levels of EV sub-types, and other markers show persistent elevation at up to 2 weeks post-TIA event, consistent with evidence the BBB remain permeable for several weeks following focal cerebral ischaemia. 69 In this context, specific EV subtypes and neurological markers offer a realistic opportunity given the limits of NHS triage and assessment of TIA.
No significant interactions were detected between medication at presentation and EV profiles within the TIA cohort. We would however acknowledge that the limited cohort numbers limits our ability to detect significant within-group effects. Long-term statin therapy, particularly with atorvastatin, lowers cholesterol, reduces endothelial activation and may decrease levels of endothelial-specific (CD146+) EVs. Similarly, third-generation anti-hypertensives can improve endothelial function and potentially reduce inflammatory EVs. Chronic antiplatelet therapy, especially with clopidogrel, can suppress platelet activation and thereby reduce circulating platelet-derived EVs, though total EV concentrations appear unaffected. Notably, despite 77% of TIA patients being on anti-hypertensives, they exhibited elevated mean arterial blood pressure (MABP), underscoring the need for rigorous risk assessment and treatment-to-target monitoring in this population. Further studies are needed to clarify the effects of these medications on EV populations.
Caution is warranted when drawing direct comparisons between a cell-culture model and patient presentations, as it is inherently challenging to reproduce the biological variability and complex in vivo conditions observed in humans. In our cohort, for example, patients reported TIA symptoms lasting 115.5 ± 70 min (Table 1), the precise degree of ischaemia and/or hypoxia could not be determined, and samples were collected at a single time point upon presentation. Each of these factors limits the validity of direct comparison with the in vitro model. The sensitivity of the in vitro assays should also be considered. Nevertheless, the in vitro results capture the immediate effects of a relatively brief hypoxic insult on BBB function and EV production, and these effects align with the patterns observed in our TIA cohort.
Future work should investigate the mechanistic role of EV surface-bound protein, assess the temporal evolution of EV subtypes post-TIA, and explore their interactions with thrombolytic therapies. Larger patient cohorts and earlier sampling post-event may improve the sensitivity and specificity of EV-based diagnostics.
Conclusions
The identification of GFAP+ and CD144+ EV as effectors of pathological processes and thrombotic risk, and potential as TIA-specific biomarkers is novel and clinically relevant. Given their ability to reflect BBB permeability and astrocyte activation, these vesicles may provide a minimally invasive means to diagnose TIA, assess severity and stratify future stroke risk. Longitudinal data from PREDICT-EV will be essential to validate these observations and determine their predictive value.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X261417191 – Supplemental material for Hypoxia stimulates blood–brain barrier disruption and systemic appearance of pro-coagulant, brain-derived extracellular vesicles: Implications in transient ischaemic attack patients
Supplemental material, sj-docx-1-jcb-10.1177_0271678X261417191 for Hypoxia stimulates blood–brain barrier disruption and systemic appearance of pro-coagulant, brain-derived extracellular vesicles: Implications in transient ischaemic attack patients by Rebecca Marie Raven, Jessica Olivia Williams, Cass Whelan, James White, Keith Morris, John Geen and Philip Eurig James in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The authors are indebted to the support and involvement of patients and public with lived experience as part of the Stroke Hub Wales PPI Group. We are also extremely grateful to the R&D team at CTMUHB, our volunteers and patients without whom this research would not be possible.
Author contributions
RMR had primary responsibility for in vitro model systems and initial drafting of BBB sections and interpretation of data. JOW made a substantial contribution to the concept and design and was responsible for healthy control recruitment, sample prep, data assessment and figure presentation. CW was responsible for acquisition of data and analysis of patient EV samples. JW was clinical lead and patient recruitment, assessment, characteristics and manuscript drafting. KM led on statistical comparison and data analysis/presentation. JG had primary responsibility for ethics and project governance, clinical diagnostics and manuscript review. PEJ was project lead, and had oversight on results/data interpretation, manuscript drafting and final review. All authors approved the version to be published.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by a Stroke Association Project Grant (PG22/23_S1100078). RR was supported as part of a PhD scholarship jointly funded by Cwm Taf Morgannwg University Health Board (CTMUHB) and Cardiff Metropolitan University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Consent for publication
Not applicable.
Data availability statement
The authors confirm that the data supporting the findings of this study will be made available on a free repository and are presented in PhD thesis entitled ‘The “hypoxic signature response” of cells and their derived extracellular vesicles in the neurovascular unit and their role in ischaemic disease’ by Dr. Rebecca Marie Raven, Cardiff Metropolitan University, and available on the University open access database.
Supplemental material
Supplemental material for this article is available online.
