Abstract
Interest in cerebrospinal fluid (CSF) physiology and its relevance to neurological disease has increased markedly in recent years. Classical descriptions portray CSF as a unidirectional flow from the choroid plexus to the dural venous sinuses and rarely distinguish between its solutes and the solvent (water) component, which constitutes ~99% of CSF. We conducted a systematic literature review to evaluate current evidence for water exchange between CSF and blood across the blood–CSF barriers (BCSFB). Eighteen studies met the inclusion criteria: 15 in experimental animals and six with humans, spanning more than 70 years and employing diverse methodologies. This literature review shows that CSF water moves freely and bidirectionally between CSF and blood across multiple BCSFB sites along the craniospinal axis, including the choroid plexus, ependymal surfaces, pial vessels, and perivascular spaces. The net direction of movement varies locally with hydrostatic, osmotic, and molecular gradients that transiently favor either inflow or outflow. Blood–CSF water exchange occurs predominantly by diffusion and is modulated by aquaporins and local vascular forces. These findings challenge the classical concept of unidirectional CSF production and absorption, supporting instead a dynamic equilibrium where distributed, gradient-driven water flux maintains brain water homeostasis.
Introduction
The year 2025 marks several milestones in cerebrospinal fluid (CSF) research. In 1825, François Magendie published Mémoire sur un liquide qui se trouve dans le crâne, introducing the concept of CSF, 1 and a century later, in 1925, Harvey Cushing coined the term “third circulation.” 2 Over time, CSF has been regarded as a single fluid with well-defined production sites (primarily the choroid plexus), fixed circulatory pathways (from the ventricles to extracerebral exit routes), and dedicated absorption sites (mainly the dural venous sinuses).
This classical textbook model has been increasingly questioned.3,4 Recent work highlights discrepancies between the traditional “third circulation” and emerging views of CSF as a fluid undergoing exchange at multiple sites. 4 In parallel, the discoveries of the glymphatic and meningeal lymphatic systems have emphasized the importance of CSF in solute transport, metabolic waste clearance, and immune surveillance. 5 Although these discoveries have shifted our understanding of CSF physiology, the prevailing framework still largely treats CSF as a uniform solution produced primarily by the choroid plexus.6,7
CSF consists of ~99% water (H2O) and ~1% solutes, including electrolytes, trophic factors, and metabolic by-products. Yet the water component is seldom considered separately in physiological models, and many techniques for measuring CSF “production” implicitly treat CSF as a homogenous entity. A recent methodological review underscored this limitation. 8
According to the Starling principle, water and solute exchange across systemic capillaries are closely coupled.9,10 Whether this paradigm fully applies to the vasculature of the central nervous system (CNS) remains uncertain; it is unclear to what extent water may cross CNS barriers independently of solutes. A clearer distinction between CSF solvent and solute components is therefore essential, especially regarding water exchange between CSF and blood across the blood–CSF barriers (BCSFB). To address this gap, we conducted a systematic literature review to evaluate current evidence for water exchange between blood and CSF in terms of (i) direction (production vs absorption), (ii) magnitude, (iii) anatomical distribution along the craniospinal axis, and (iv) regulatory mechanisms (driving forces, water-solute dissociation, and molecular pathways).
In this review, we distinguish between human and animal data and examined the influence of neurological disease on these processes. Furthermore, given the current interest in the glymphatic system and the central role of aquaporine-4 (AQP4) in glymphatic flux, we also consider how the identified evidence aligns with or challenges the glymphatic framework.
Materials and methods
Search strategy and selection criteria
Firstly, we conducted a comprehensive literature search across EMBASE (Ovid), MEDLINE (Ovid), and Scopus (Elsevier), with no restrictions on language, publication date, document type, or publication status. This search was guided by the following research question: What is the current evidence regarding water exchange between CSF and blood in terms of (i) direction (production vs absorption), (ii) magnitude, (iii) anatomical distribution along the craniospinal axis, and (iv) regulatory mechanisms (driving forces, water-solute dissociation, and molecular pathways)? Secondly, a citation search based on the initial literature search result was conducted using Citation Chaser, Research Rabbit, Scopus, and Web of Science.
The final search was completed in April 2025, and the complete search strategy is summarized in Supplementary Table 1.
Both authors independently screened titles, abstracts, and full texts using Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia), including only original research articles published in English. The full-text exclusion criteria were as follows: (1) full text not available after reasonable effort; (2) not a primary research study, including book chapters; (3) outcomes not relevant to the review question; and (4) not relevant to the review question. Any discrepancies were resolved between the two reviewers.
Data extraction
Extracted data included: (1) species studied, (2) disease model or diagnosis, (3) methods and outcome measures used to assess blood–CSF water exchange, (4) direction of water movement (CSF → blood or blood → CSF), (5) magnitude measures of blood–CSF water exchange, (6) anatomical site of exchange (e.g. brain ventricles, spinal cord), (7) mechanisms regulating the water exchange, and (8) author’s interpretation of findings. Results were analyzed separately for animal and human studies.
Results
Study selection
A total of 2,299 articles were identified through the systematic literature and citation search, and imported into Covidence for screening. After automatic and manual removal of 103 duplicates, 2,196 titles and abstracts were screened for relevance. Sixty-eight full-text manuscripts were assessed for eligibility, of which 18 met all inclusion criteria and form the evidence base for this review. Among these, 15 studies reported animal data, six included human data, and three contained both. The full list of included studies is provided in Supplementary Table 2. Figure 1 displays the PRISMA 2020 flow diagram for the review (http://www.prisma-statement.org/PRISMAStatement/PRISMAStatement.aspx). 11
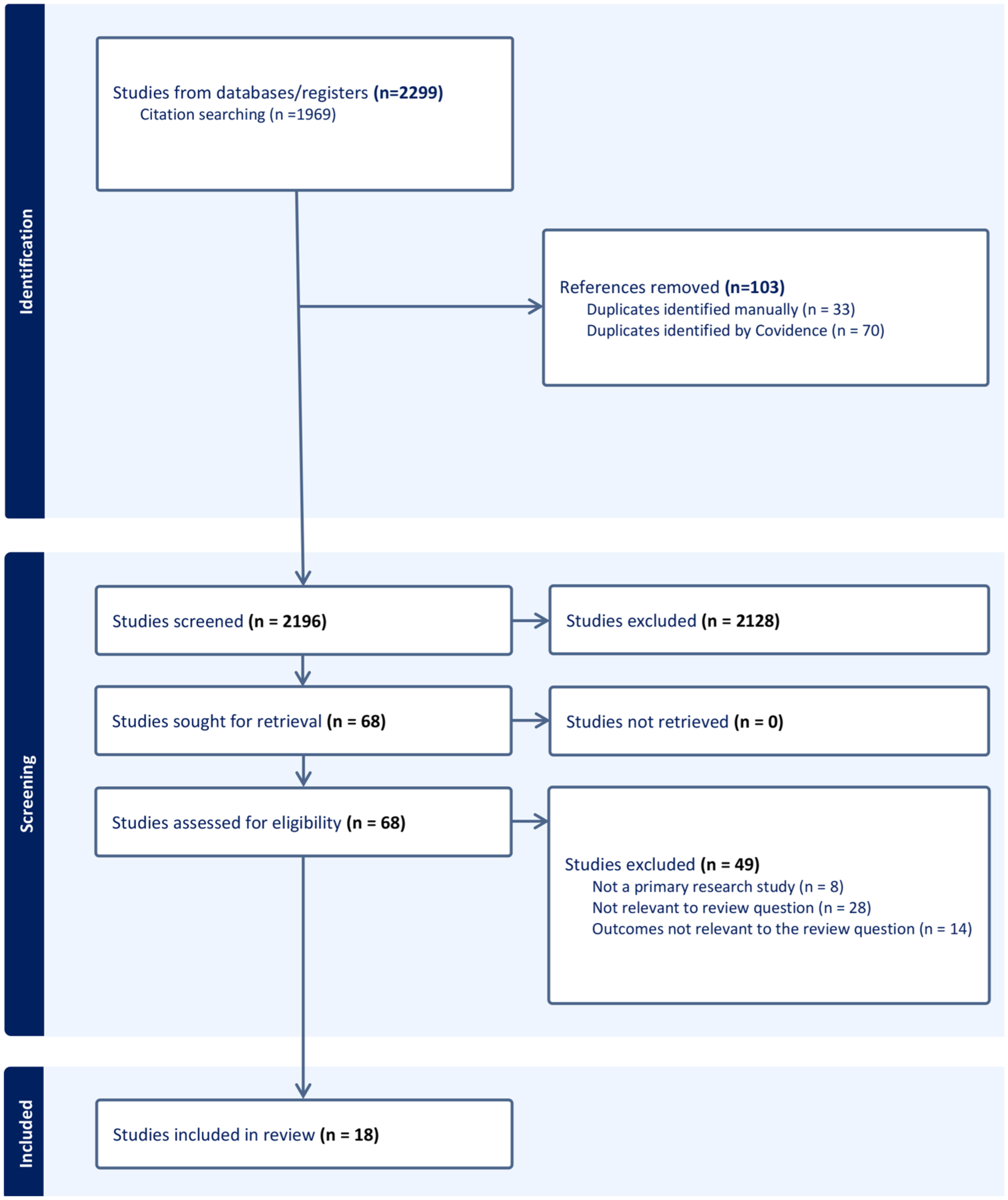
PRISMA 2020 flow diagram for the systematic review that include searches of databases. The PRISMA plot 11 with included and excluded studies.
The included studies span more than 70 years of research. Table 1 summarizes the 15 animal studies and Table 2 summarizes the six human studies investigating blood–CSF water exchange. Over this period, the tracers and imaging methods evolved substantially: heavy water (D2O) in the 1950s,12–14 tritiated water (T2O) from the 1960s to the early 2000s,15–21 and H217O magnetic resonance imaging (MRI) techniques during the past decade.22–25 More recently, arterial spin labeling (ASL) MRI has been applied to investigate water transfer from blood to CSF.26–29
Animal studies on water exchange between blood and CSF.

AQP: aquaporin; ASL: arterial spin labeling; BCSFB: blood–CSF-barrier; CASL: continuous arterial spin labeling; CM: cisterna magna; CP: choroid plexus; CSF: cerebrospinal fluid; CV: cerebral ventricles; DEX-I, dexmedetomidine; HC: hydrocephalus; ICV: intracerebroventricular; ISO, isoflurane; IV: intravenous; JJVCPE, JJ vicinal coupling proton exchange; MRI: magnetic resonance imaging; NR: not reported; SAS: subarachnoid space; SHR: spontaneous hypertension rat; SP, senile plaque; SSS: superior sagittal sinus; TBW: total body water.
Human studies on water exchange between blood and CSF.

AQP: aquaporin; CP: choroid plexus; CSF: cerebrospinal fluid; CM: cisterna magna; CV: cerebral ventricles; HC: hydrocephalus; ICV: intracerebroventricular; IV: intravenous; MRI: magnetic resonance imaging; NR: not reported; SAS: subarachnoid space; TBW: total body water.
Direction and magnitude of CSF–blood water exchange
Bering first demonstrated that intravenous D2O in dogs resulted in rapid exchange with brain tissue (12–20 s) and CSF (3–8 min), with cisternal CSF equilibrating faster (~3 min) than ventricular CSF (~8 min). 12 In monkeys, cisternal D2O likewise equilibrated rapidly with blood. 13 Several studies across nonhuman species confirmed rapid water passage from ventricular CSF to blood, 17 and from the subarachnoid space (SAS) to venous blood via pial arteries, with mean transit times of ~14 s (range 5–28 s). 18
In humans (“healthy” individuals aged 6 months–87 years), Bering measured half-times for CSF compartments to reach blood D2O concentrations after intravenous administration, reporting: cisterna magna, 1.5–6 min, cerebral ventricles 2–37 min, and lumbar SAS 7–38 min. 12 Migliore et al. 21 later showed that T2O administered either intravenously or intraventricularly equilibrated between blood and CSF within ~1.5 h in individuals aged 8–50 years without CSF pathology.
Modern H217O- and ASL–MRI studies corroborate this rapid bidirectional exchange. In rodents and humans, blood-to-CSF delivery at the choroid plexus and cortical SAS occurs with characteristic time constants of ~1 min.26,28 Using ultra-long echo time ASL–MRI, Petitclerc et al. 28 quantified blood-to-CSF transfer times of ~60 s for both choroid plexus and SAS, and ~80 s in white matter. Notably, CSF–ASL signal was broadly distributed around the cortex, indicating that water exchange is not confined to the choroid plexus.
Collectively, these findings show that water exchange between blood and CSF is rapid, continuous, and bidirectional, rather than a unidirectional process of CSF “production” and “absorption.”
The reported (semi-)quantitative estimates suggest that the total volume of water exchanged between blood and CSF is large relative to the CSF pool (Supplementary Table 3 and Table 3). Based on isotopic and MRI-derived half-times, we estimate the effective flux to about 0.5–2.0 mL/min, which would be sufficient to renew the entire CSF water content (~150 mL) several times per day.
Anatomical distribution of exchange along the craniospinal axis
Evidence for blood–CSF water exchange is found at multiple interfaces extending from the cerebral ventricles to the lumbar SAS. At the choroid plexus, ASL and H217O MRI in rodents and humans demonstrate rapid blood-to-CSF water transfer, closely linked to perfusion.26,28 Across ventricular ependyma and periventricular capillaries, classical isotope studies indicate diffusion-dominated exchange with permeability coefficients in the range of 10−4–10−3 cm/s.12,16 At pial and cortical vessels, water moves from CSF to venous blood within seconds, independently of ion transport, 18 indicating a highly permeable interface. Evidence of spinal water exchange comes from early D2O studies in which tracer appeared in lumbar CSF even when cranial CSF pathways were obstructed,12,13 demonstrating that exchange also occurs locally along the spinal SAS.
Integrating data across compartments suggests that total CSF–blood water exchange is distributed approximately as follows: ~25% via the choroid plexus, ~20% via the ventricular wall, ~25% via cortical and pial surfaces, and ~30% via the spinal SAS (Table 3). Exchange is faster in cisternal and spinal compartments than within the ventricles, reflecting greater surface area and vascular density. These findings suggest that blood–CSF water turnover is a system-wide process rather than a localized secretion, and that the craniospinal compartment operates as a continuous diffusion interface between vascular and CSF water pools.
Estimated water exchange between blood and CSF across craniospinal sites.
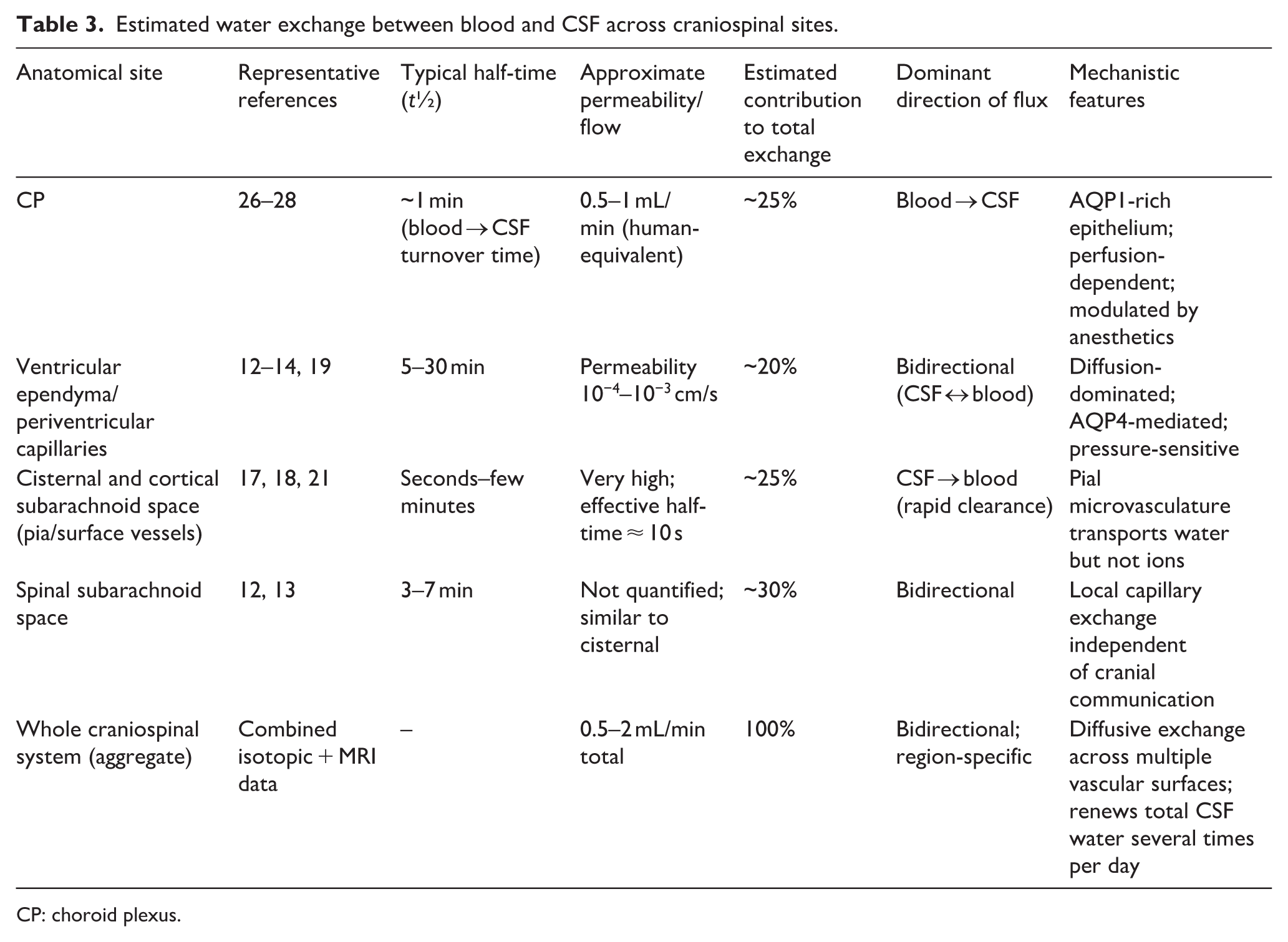
CP: choroid plexus.
Tracer studies also indicate that the choroid plexus removal does not substantially alter the appearance of D2O, 24Na, or 131I-albumin in CSF after intravenous administration,12,14 further supporting a distributed exchange system.
Driving forces governing water exchange
Experimental manipulation of pressure, osmolarity, and hormonal state suggests that the direction of blood–CSF water flux is dynamic and locally regulated. Early isotope studies identified diffusion as the principal mechanism of water transfer across blood–CSF interfaces,12,15 and Migliore et al. 21 likewise attributed blood-to-CSF water movement in humans to simple diffusion.
Hydrostatic forces also contribute: elevations in intraventricular pressure increased T2O efflux from ventricles to blood, 15 whereas increased choroid plexus perfusion or arterial pressure enhanced blood-to-CSF inflow. 26 Osmotic gradients can reverse the direction of movement, as demonstrated by the introduction of hyperosmolar mannitol into CSF, which shifted net flux toward blood. 16 Hormonal modulation was evident when vasopressin increased ependymal permeability and accelerated CSF clearance. 19
Evidence was given that permeability is adjustable: deletion or pharmacologic modulation of AQP4 altered the rate of water exchange without changing its direction, indicating that aquaporins regulate conductivity rather than net flux polarity.22–24
Table 4 summarizes studies demonstrating that water exchange across blood–CSF interfaces occur largely independently of ionic or macromolecular transport. Bering observed distinct exchange patterns for D2O, 24Na, 42K, and 131I-albumin in dogs 14 : water moved rapidly, whereas ions and proteins crossed more slowly. Similar findings were reported in monkeys: after cisternal injection, T2O equilibrated with blood within ~10 min, 22Na required ~45 min, and 131I-albumin moved even more slowly, likely influenced by sagittal sinus absorption. 17 In goats, ventricular permeability coefficients differed markedly (T2O: 1.7 ± 0.5 cm2/min; 24Na: 0.18 ± 0.01 cm2/min; 42K: 0.80 ± 0.13 cm2/min for). 15 In dogs and cats, T2O rapidly appeared in venous blood after subarachnoid or ventricular administration, while 22Na and 3H-inulin exhibited markedly slower movement.16,18 In humans, Migliore et al. 21 found that T2O exchanged rapidly, whereas other tracers (24Na, 36Cl, 42K, 32P, 82Br, 131I-albumin) showed delayed and heterogeneous kinetics.
Studies comparing water and ionic tracers.
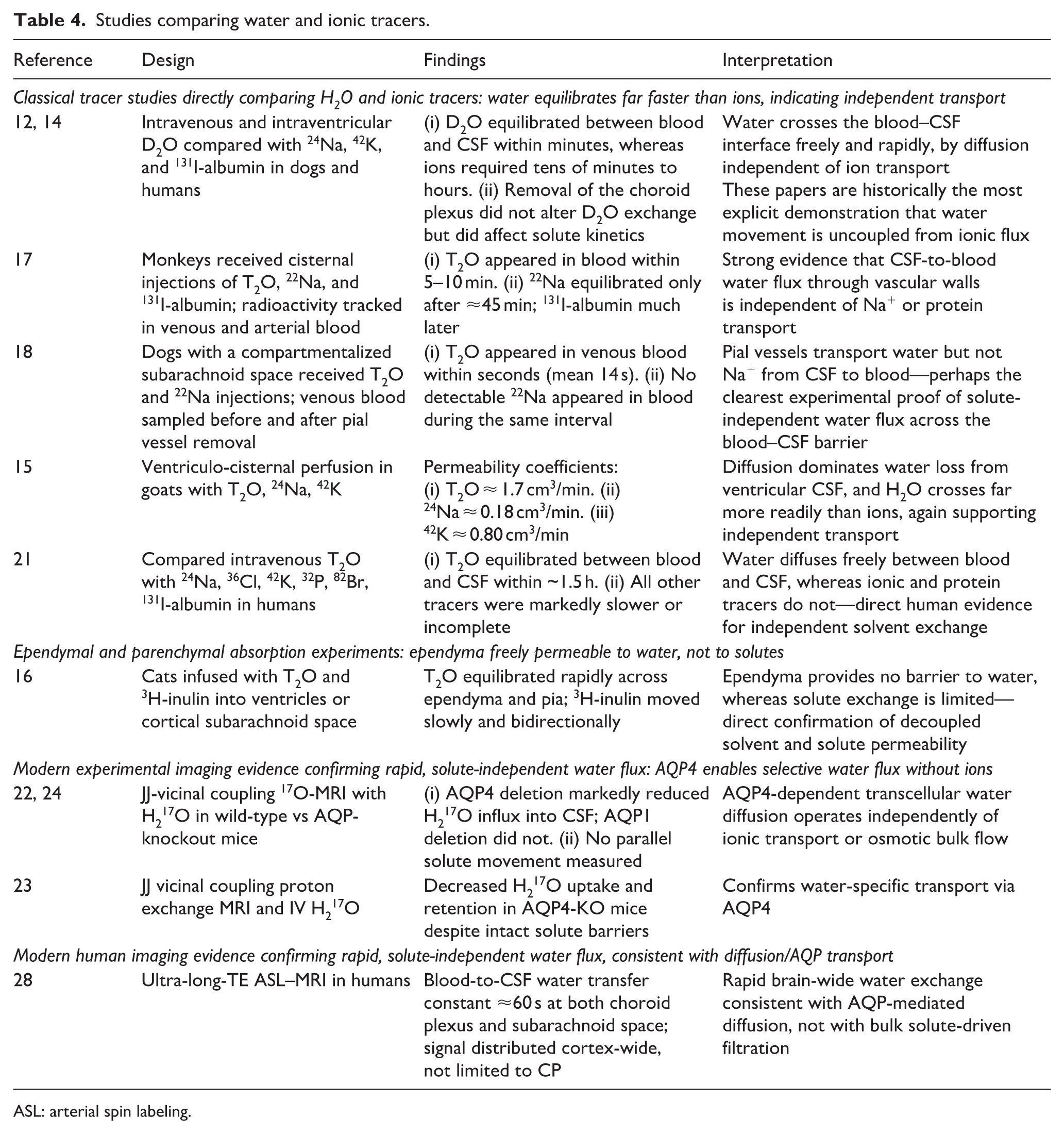
ASL: arterial spin labeling.
Together, the reported results indicate that hydrostatic, osmotic, and molecular gradients jointly determine the local balance between inflow and outflow, making net water transport highly context dependent. The pronounced dissociation between water and solute flux may be most parsimoniously explained by AQP-mediated diffusion through selective, high-permeability membranes, allowing water to cross rapidly while restricting ionic and macromolecular passage.
Molecular and cellular mechanisms of water transfer
Aquaporin water channels provide the molecular basis for rapid water exchange between blood and CSF. AQP1, expressed on choroid plexus epithelial cells, facilitates trans-epithelial water movement from blood to ventricular CSF. AQP4, densely localized to astrocytic endfeet at perivascular, ependymal, and pial surfaces, 30 governs permeability on the brain side of the vascular wall. Knockout or mis-localization of AQP4 reduces water exchange by ~20%–40% in optical and MRI studies, 24 confirming its rate-limiting role.
Three pharmacologic modulators demonstrated effects on water transport:
i. Arginine vasopressin (AVP) enhances CSF-to-blood water efflux. In cats, AVP increased the T2O distribution volume, raised ependymal permeability, and shortened capillary transfer half-time during ventriculo-cisternal infusion experiments. 19 The net effect was accelerated removal of water from ventricular CSF into brain tissue and blood (enhanced CSF-to-blood absorption), 19 consistent with increased transcellular permeability at the CSF–brain interface.
ii. Aquaporin-4 (AQP4) modulation alters parenchymal water flux and affects water transport across the BBB and BCSFB. In AQP4-knockout mice, H217O penetration into third ventricle CSF after intravenous administration was significantly reduced, whereas AQP1-knockout mice showed no difference, implicating AQP4 in blood-to-CSF water entry. 24 Intravenous H217O combined with 17O-MRI showed reduced brain water signal in AQP4-knockout mice, reflecting impaired BBB water transport. 23 Conversely, the AQP4 facilitator TGN-073 enhanced cortical water flux and increased interstitial fluid (ISF) turnover, interpreted as faster ISF-to-perivascular/blood exchange. 25 Notably, CSF water transport itself was not measurably increased, suggesting a predominantly parenchymal/perivascular mode of action rather than enhanced blood → CSF entry.
iii. Anesthetic state modulates blood-to-CSF delivery at the choroid plexus. In rats, continuous ASL–MRI demonstrated that BCSFB-mediated water delivery from arterial blood to ventricular CSF was higher under isoflurane than dexmedetomidine anesthesia, with corresponding differences in arterial transit times. 26 Although not a therapeutic modulation, this provides a clear proof-of-principle that pharmacology can tune blood-to-CSF water transport at the choroid plexus.
Together, these findings indicate that water movement across blood-to-CSF interfaces is a dynamic and regulatable process, not a fixed passive property. Because water crossing the capillary wall must pass through sequential barriers, the endothelium and the AQP4-rich astrocytic endfeet, changes in AQP4 expression or polarization alter the overall hydraulic conductivity between blood and CSF, even though AQP4 is not located within the endothelial membrane itself.
Impact of disease
Several studies identified alterations in blood–CSF water exchange associated with neurological and psychiatric conditions.
In APP–PS1 transgenic mice (a model of Alzheimer’s disease), intravenous H217O combined with JJ vicinal coupling proton exchange MRI (JJVCPE–MRI) demonstrated reduced water flux into third-ventricle CSF, suggesting impaired blood-to-CSF water entry early in the disease course. 22 In spontaneously hypertensive rats, ASL–MRI revealed a 35.8% reduction in BCSFB-mediated ventricular water delivery compared with normotensive controls (14.4 ± 1.92 vs 9.22 ± 1.20 µL/min). 27 Conversely, in another Alzheimer’s mouse model (sXTg), the same group reported increased choroid plexus-mediated ventricular water delivery, 29 highlighting model-specific differences in CP perfusion and water flux.
Early D2O studies in hydrocephalus also demonstrated altered exchange. Bering reported markedly prolonged half-times for D2O entry into the ventricles (60–245 min) and lumbar SAS (9–36 min) in hydrocephalic infants compared with non-hydrocephalic children and adults (ventricles, 2–37 min; lumbar SAS, 7–38 min). 12 He attributed the slowed exchange to a reduced ventricular surface-area-to-volume ratio. Importantly, ventricular wall exchange persisted despite Sylvian aqueduct obstruction and occurred at similar rates regardless of whether D2O was administered intravenously or intraventricularly. 13 In one case of non-communicating hydrocephalus, D2O appeared in lumbar CSF only after its appearance in blood, indicating a blood-borne route. 13 Diverting CSF did not change ventricular-to-blood D2O exchange rates, leading Bering to conclude that physiological CSF pressure has little influence on water exchange, which is primarily diffusion-driven. 13
Altered water exchange has also been observed in psychiatric disease. Coppen et al. 20 administered T2O intravenously and intrathecally in patients with major depression. Individuals with current depression exhibited slower blood-to-CSF transfer (half-time 29.4 ± 6.0 min; transfer constant 0.021 ± 0.008 min) than recovered patients (24.3 ± 4.7 min; 0.025 ± 0.008 min), suggesting that impaired CSF–blood water exchange may accompany mood disorders.
Discussion
This systematic review identified a small but informative body of literature on blood–CSF water exchange spanning more than 70 years and comprising 18 studies—15 in animals, six in humans, and three that included both. Collectively, these studies show that water moves rapidly and bidirectionally across the blood–CSF barriers, including the vasculature of the choroid plexus, ventricular walls, subarachnoid space, and perivascular compartments within the parenchyma (Figure 2). Exchange occurs predominantly by diffusion and is modulated by hydrostatic gradients and surface area. Contrary to the traditional view, 7 the choroid plexus appears to play a more limited role in water entry into the CSF than previously assumed. Importantly, water transfer can occur independently of solute movement, including ions (Na+, K+) and larger molecules such as inulin and albumin. Evidence for molecular regulation is more limited but highlights the roles for AQP4 and arginine vasopressin in modulating permeability.
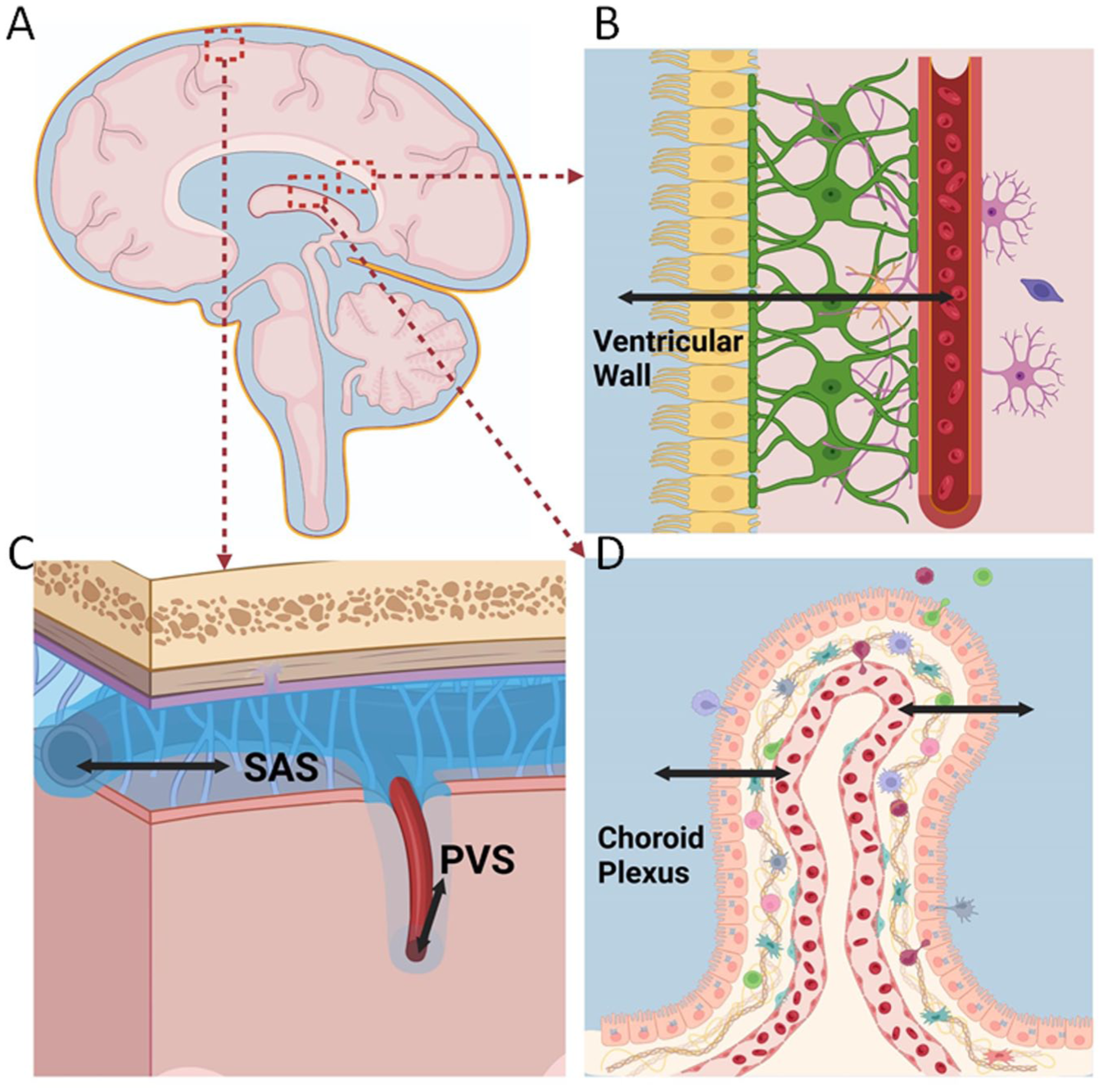
Locations for water passage between CSF and blood at blood–CSF-barriers. (a) The blood–CSF-barriers examined in the included reviews include, (b) ventricular wall, that is, lined by the ependymal cells, (c) the pial arteries in SAS and the brain parenchyma (that corresponds to blood vessels within the parenchymal perivascular spaces), and (d) the choroid plexus. Illustration: Dr. Cesar Luis Vera Quesada, Oslo University Hospital, University of Oslo.
As summarized in Tables 1 and 2, the methods for assessing blood–CSF water exchange have evolved considerably over time. Early invasive techniques using D2O and T2O provided quantitative estimates of exchange but offered limited spatial and temporal resolution. More recent non-invasive approaches—including ASL–MRI and 17O-sensitive MRI—allow anatomical mapping and mechanistic assessment of blood–CSF water transfer, although they currently lack the direct quantitative precision of classical tracer studies.
Reinterpreting “production” and “absorption” as bidirectional equilibrium
Across animal and human studies using D2O, T2O, and H217O tracers, water was found to move rapidly and bidirectionally between blood and CSF. Importantly, this reciprocal exchange persisted even after choroid plexus isolation, directly challenging the classical view that CSF is formed and absorbed at discrete, anatomically fixed sites. Instead, the apparent “production” or “absorption” observed under physiological or pathological conditions reflects transient dominance of inflow or outflow driven by local hydrostatic, osmotic, or permeability gradients. CSF homeostasis is therefore best conceptualized as a steady-state equilibrium maintained by distributed, bidirectional water flux.
The traditional “third circulation” model, in which CSF is produced primarily by the choroid plexus and absorbed into the dural sinus veins to establish a unidirectional bulk flow, 2 is inconsistent with evidence showing water entry and exit at multiple CNS interfaces. While some authors have argued that widespread vascular exchange precludes the existence of net CSF flow, claiming, for example, that no net movement occurs through the Sylvian aqueduct, 4 this position contradicts phase-contrast MRI studies demonstrating net CSF (water) flow through both the aqueduct and the cranio-cervical junction.31,32 Thus, even though water may enter the CSF from the entire CNS vasculature, local pressure gradients can still generate regions of net directional flow within the ventricular and subarachnoid spaces.
The long-standing perspective that CSF is a single, uniform entity produced almost exclusively by the choroid plexus can be partly traced to methodological conventions. A key example is the ventriculo-cisternal perfusion technique, 15 which estimates “CSF production” by measuring the clearance of inulin, a tracer molecule of 3–5 kDa (with a broader range of 1–10 kDa). Because this method quantifies the removal of a solute, it implicitly treats CSF as a homogeneous fluid and overlooks the fact that its major constituent—water—has distinct exchange pathways and kinetics. As evidenced by the studies in this review, CSF water derives from multiple sources throughout the CNS, not solely the choroid plexus, and diseases can selectively alter these local water-exchange mechanisms.
Thus, referring to “CSF production” without acknowledging the diverse origins and behaviors of its solvent and solute components oversimplifies the physiology. A more nuanced framework that recognizes the heterogeneous composition of CSF, its multiple entry routes, and the regulatable nature of water exchange provides a more accurate foundation for understanding both normal CSF dynamics and their alterations in disease.
Anatomical distribution of exchange along the craniospinal axis
Water exchange occurs throughout the craniospinal system, involving the choroid plexus,26,28,29 ventricular ependyma,14–16 cortical pial vessels,17,18 perivascular and parenchymal interfaces,22–24 and the spinal subarachnoid space.12,13 The consistency of tracer kinetics and MRI findings across these interfaces indicates that the CSF system functions as a distributed exchange network rather than a linear production-absorption circuit. Both spinal and cortical vessels contribute substantially to total water flux, suggesting that the entire perivascular continuum participates in maintaining CSF–blood water equilibrium.
Several findings highlight the importance of the spinal compartment. Intravenous D2O appeared in the lumbar SAS at similar rates in individuals with intact cranial communication and in those with isolated lumbar CSF spaces, 12 implying that water entry into lumbar CSF occurs locally and independently of cranial pathways. Consistent with this interpretation, modern MRI studies have repeatedly demonstrated net upward CSF water flow at the cranio-cervical junction in supine individuals.31,32 Moreover, the average time for a lumbar administered CSF tracer to reach the cisterna magna was 20 ± 23 min, 33 indicating rapid CSF flow in the cranial direction. Given the increased hydrostatic pressure gradients in the upright posture, spinal contributions to CSF water entry are expected to be even more prominent when standing.
Across these regions, the apparent direction of CSF–blood water movement is not fixed but reflects the balance of local hydrostatic, osmotic, and permeability conditions. Elevated arterial or choroid plexus perfusion pressure enhances blood-to-CSF inflow, whereas increased ventricular pressure, CSF hyperosmolarity, or vasopressin stimulation favors CSF-to-blood efflux. Because these gradients vary spatially and dynamically, the craniospinal system operates as a bidirectional equilibrium, in which traditional notions of “production” and “absorption” reflect transient, region-specific biases rather than anatomically segregated processes.
Mechanistic basis of water exchange
Quantitative data from early tracer work and modern MRI methods converge on the conclusion that water exchange between blood and CSF is rapid, continuous, and large relative to total CSF volume, occurring on the order of seconds to minutes rather than hours. Bering’s early D2O studies showed that CSF tracer disappearance closely paralleled declines in blood concentrations, consistent with ongoing equilibration rather than unidirectional “production” or “absorption.”
Diffusion appears to be the principal mechanism driving this exchange. Partial-pressure and concentration gradients of H2O across vascular, ependymal, and pial interfaces provide sufficient chemical potential to support substantial flux, even in the absence of solute movement. Small hydrostatic differences further augment transcapillary flow due to the finite hydraulic conductivity of brain microvessels. For example, experimentally increasing intraventricular pressure enhanced T2O efflux, 15 demonstrating a pressure-dependent component superimposed on diffusion. Thus, what is traditionally interpreted as “production” or “absorption” likely reflects regional differences in diffusion rates, surface area, and local driving forces rather than directional flow in a single circuit.
At any given site, the net direction of water movement is set by the interplay of hydrostatic, osmotic, and permeability gradients. Elevated ventricular pressure favors CSF-to-blood movement, whereas increased arterial or choroidal perfusion promotes blood-to-CSF entry. Hyperosmolar CSF enhances absorption; hypo-osmolar plasma enhances entry. Vasopressin and AQP4 regulation alter effective permeability and thus shift the balance accordingly. These regionally variable and dynamic gradients explain how the system maintains stable CSF volume despite fluctuating pressures and osmolarities.
The short half-times for water equilibration (seconds to minutes) contrast sharply with those for ions (Na+, K+) or macromolecules (inulin, albumin), which require tens of minutes to hours (Table 4). This dissociation demonstrates that water transport is not obligatorily coupled to solute flux. CSF and interstitial water are in near-continuous exchange with capillary and perivascular water, forming a dynamic fluid continuum rather than a slowly circulating, isolated compartment. For instance, rapid water transfer at pial arteries occurred without accompanying Na+ exchange. 18 This implies that CSF composition is not uniform and likely varies according to different sites of exchange—a view supported by documented differences between lumbar and ventricular CSF. 34
The classical Starling principle that couples solvent and solute transport, 9 does not fully apply to brain microvessels. Instead, selective hydraulic permeability and aquaporin-mediated water diffusion allow rapid water passage across endothelial, ependymal, and glial barriers even when ion gradients are minimal. AQP enables single-file water movement along osmotic or pseudo-osmotic gradients without solute transfer, resolving the apparent paradox posed of solvent-solute uncoupling. Consistently, AQP4-knockout animals show reduced H217O penetration into CSF and brain interstitial tissue, confirming its key role (see Tables 1 and 222–24). AQP1 in the choroid plexus facilitates trans-epithelial water flow but appears less essential for whole-system CSF–blood exchange. The intracellular and extracellular water phases of the CNS form a nearly continuous medium, allowing diffusive exchange across ependyma, pia, and perivascular spaces even when solute diffusion is restricted.
Hormonal and physiological state further modulate exchange. Vasopressin increased ependymal permeability and enlarged the T2O distribution space in cats, 19 likely via AVP-AQP4 signaling, while anesthetic state (isoflurane vs dexmedetomidine) altered choroid plexus blood-to-CSF transfer on ASL–MRI. These observations reinforce that water flux across the BCSFB is a regulated physiological process, not a fixed passive property.
Molecular and cellular mechanisms of water transfer
The reviewed studies provide consistent evidence that AQP4 is the principal molecular regulators of water exchange between blood, interstitial fluid, and CSF. AQP4 is densely expressed in astrocytic endfeet along perivascular, subpial, and ependymal surfaces, that is, the membranes that interface with CSF and the microvasculature. 30 In AQP4-knockout mice, H217O influx into ventricular CSF was significantly reduced, whereas AQP1 deletion in choroid plexus had no measurable effect, 24 indicating that AQP4, not AQP1, mediates the dominant water pathway across the CNS fluid barriers.
Structurally, water transfer from blood to the interstitial space of the brain parenchyma requires passage through three layers: (i) The endothelial membrane. (ii) Basement membrane. (iii) AQP4-rich astrocytic endfeet. These layers act as serial hydraulic resistors that together determine the effective permeability of the brain-CSF surface:

Ultrastructural studies35,36 show AQP4 clustered on both the perivascular and neuropil-facing surfaces of astrocytic endfeet, though less polarized to the perivascular membrane in humans than mice. 36 In vivo imaging and genetic models37,38 demonstrate that deletion or loss of AQP4 polarization markedly reduces fluid and solute flux between perivascular and interstitial compartments. These data support a transcellular “hydraulic bridge” model, in which water entering the perivascular basal lamina crosses the endfoot cytoplasm via paired AQP4 domains, allowing rapid exchange while maintaining ionic segregation. This explains how water—but not ions or macromolecules—can move freely between blood and CSF.
Regulatory mechanisms further tune this equilibrium. Vasopressin increases ependymal permeability and accelerates CSF clearance, likely via AQP4-dependent pathways, while anesthetic agents modulate blood-to-CSF inflow at the choroid plexus in ASL–MRI studies. Together, these findings show that the molecular machinery governing water transfer is not static but dynamically regulated by physiological and pharmacological states.
Glymphatic transport versus blood–CSF water exchange
How do the present observations relate to the glymphatic framework? At first glance, the glymphatic model, characterized by net CSF influx along periarterial spaces and efflux along perivenous pathways, 38 appears inconsistent with the bidirectional transvascular water exchange demonstrated in the reviewed literature. However, these processes operate at different spatial and temporal scales and are best understood as complementary rather than contradictory.
Glymphatic flow reflects bulk convective transport of fluid and solutes within the perivascular continuum. 38 In contrast, the tracer and MRI data reviewed here quantify microscopic, bidirectional exchange of water molecules across endothelial and astrocytic membranes. Continuous molecular-level exchange does not preclude a net convective bias; rather, both processes can coexist. In this framework, AQP4 provides the low-resistance transcellular pathway that links microvascular water permeability to macroscopic glymphatic transport.
Converging evidence supports this integrated model: AQP4 deletion slows glymphatic tracer clearance,38,39 and H217O studies show that AQP4 also mediates rapid bidirectional water exchange across the neurovascular unit.23,25 Human evidence includes patients with the dementia subtype idiopathic normal pressure hydrocephalus (iNPH), who present with loss of perivascular AQP440,41 and impaired glymphatic function shown by intrathecal contrast-enhanced MRI.42,43
Thus, the same perivascular channel underlies both microscopic exchange and macroscale convective flow, with the prevailing driving forces (pressure, osmolarity, or permeability) determining whether net movement occurs. Glymphatic flow describes the direction of bulk movement, whereas CSF–blood exchange explains how water is continuously renewed and redistributed.
Together, these findings suggest that the CSF–blood exchange network provides the hydraulic substrate on which the glymphatic circulation operates. Factors that alter local pressure gradients, osmolarity, endothelial permeability, or AQP4 function are therefore expected to modulate both microscale water exchange and macroscale solute clearance in the brain.
Blood–CSF water exchange in disease
Pathological conditions modify the regulation and efficiency of blood–CSF water exchange. Experimental studies show reduced water flux in models of hypertension 27 and Alzheimer’s disease, 29 whereas enhanced exchange has been observed in certain early Alzheimer’s stages, 29 possibly reflecting compensatory choroid plexus hyperperfusion. In humans, altered T2O entry into CSF in patients with depression suggests that psychiatric disease may also involve subtle disturbances in water homeostasis.
The available literature on disease-related changes remains limited but informative. Bering’s investigations of infantile hydrocephalus demonstrated markedly slowed blood-to-CSF water exchange, implicating the surface-area-to-volume ratio of the ventricles as a key determinant of water movement. 13 Notably, water absorption across the ventricular wall persisted even when the Sylvian aqueduct was occluded, indicating that exchange can occur independently of intact CSF pathways. Furthermore, choroid plexus removal did not alter water transfer between blood and CSF, 14 reinforcing the concept of a distributed exchange system.
Other pathological models provide converging evidence. Experimental hypertension reduces blood-to-CSF delivery at the choroid plexus, 27 and both preclinical and transgenic Alzheimer’s models show altered BCSFB water transport.22,29 These disturbances may reflect impaired perfusion, vascular stiffening, or inflammatory changes affecting barrier permeability.
Loss of AQP4 polarization or genetic deletion further decreases water turnover, contributing to impaired interstitial and CSF clearance. In hydrocephalus, reduced ependymal permeability slows CSF-to-blood efflux, promoting ventricular expansion. Although initial dilatation temporarily increases the surface area available for exchange, progressive enlargement ultimately reduces the surface-area-to-volume ratio, limiting compensatory capacity. Hypertension and neurodegeneration disease may similarly alter local pressure, osmotic gradients, or membrane permeability, shifting the equilibrium toward net retention or depletion of CSF water.
Limitations
This review is subject to several notable limitations. First, the literature on blood–CSF water exchange is sparse and relies heavily on older studies that used invasive tracer methods with limited spatial and temporal resolution. Although foundational, these techniques may not adequately capture the complexity of rapid, regionally variable water exchange that modern imaging approaches can reveal.
Second, the majority of available data derive from animal models, with relatively few studies in humans. This limits the direct translational relevance of findings and underscores the need for more human-focused research.
Third, significant gaps remain in our understanding about the molecular and pharmacological mechanisms that regulate blood–CSF water flux, particularly in neurological disease. Continued development of advanced, non-invasive imaging and molecular approaches will be essential to clarify these mechanisms and to characterize how they are altered in pathology.
Conclusions
Together, the findings of this systematic review indicate that CSF water exchange is a distributed, diffusion-dominated process occurring across multiple interfaces rather than a unidirectional secretory–absorptive system centered on the choroid plexus. Water exchanges continuously between blood, interstitial fluid, and CSF along the entire craniospinal axis, driven by local gradients in hydrostatic pressure, surface area, permeability, and osmolality. This exchange can occur independently of solute transport, underscoring the need to distinguish CSF water dynamics from solute circulation. Recognizing CSF as a heterogeneous fluid, whose water and solute components follow distinct anatomical and molecular pathways, fundamentally reframes classical notions of “production” and “absorption” as manifestations of transient, region-specific biases in a broader bidirectional equilibrium. AQP4 provides the principal low-resistance pathway linking vascular, glial, and ependymal interfaces, while disease states such as hydrocephalus, hypertension, and neurodegeneration likely disrupt this finely regulated hydraulic network. Despite these insights, contemporary evidence remains limited, and the molecular and pharmacological determinants of blood–CSF water exchange are poorly understood. Future work should integrate quantitative radiotracer imaging, high-resolution multimodal MRI, and molecular perturbations to map regional water fluxes, clarify spinal contributions, and determine how these processes are altered in neurological disease-ultimately informing therapeutic strategies that target aquaporin function or local fluid gradients to restore CSF homeostasis.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251413926 – Supplemental material for Water exchange across the blood–CSF barrier: A systematic review
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251413926 for Water exchange across the blood–CSF barrier: A systematic review by Trine Hjørnevik and Per Kristian Eide in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The authors would like to thank Academic Librarian Hilde Strømme at the University of Oslo Library of Medicine and Science for her contributions to the literature search.
Author contributions
TH and PKE contributed to the conception and design of the study. TH and PKE contributed to the acquisition and analysis of data. TH and PKE contributed to the writing. Both authors approved the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the South-Eastern Norway Regional Health Authority, Norway (grants 2025024).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data sharing is not applicable to this article as no datasets were generated during the current study.
Supplementary material
Supplemental material for this article is available online.
