Abstract
In the resting, non-stimulated brain, metabolic demands are met exclusively by the delivery and extraction of glucose and oxygen at an ~6:1 ratio. Amongst healthy people at rest, there is marked variability in resting global cerebral blood flow (CBF) yet remarkably stable concentrations of circulating glucose and oxygen. Thus, we would expect interindividual variability in resting CBF to be inversely related to oxygen and glucose extraction, maintaining oxidative glucose metabolism. Herein, we investigated the fundamental relationship between CBF and substrate extraction in 75 healthy adults (27.3 ± 4.8 years) with resting measures of CBF and cross-brain concentrations of oxygen and glucose. We observed that the marked interindividual variability in CBF (<500 to >1200 mL/min) is inversely related to oxygen extraction (
Keywords
Introduction
The brain is a unique metabolic organ owing to its disproportionate metabolic demand relative to total body mass. 1 In the resting brain, the vast majority of the energetic demand is met by oxidative phosphorylation of glucose. 2 Interestingly, despite remarkably stable arterial concentrations of glucose and oxygen amongst healthy humans at rest, there is striking interindividual variability in delivery that is, cerebral blood flow (CBF). In healthy adults with no known cognitive or neurological disease, resting CBF can vary by 50% between individuals.3–5 Thus, the question arises: How can the brain’s metabolic demands, which are met almost exclusively by substrate delivery, be achieved with such extreme differences in CBF between individuals? Given consistent levels of circulating oxygen and glucose, the only conceivable way this would be possible is for cerebral oxygen and glucose extraction to adjust to different rates of delivery.
The brain’s primary energy source of oxidative glucose metabolism is evidenced by a resting oxygen to glucose index of 5.46, 6 nearly matching the 6:1 (oxygen:glucose) stoichiometric ratio required for oxidative phosphorylation. The ratio falling slightly below 6 indicates a small presence of aerobic glycolysis (AG; the metabolism of glucose without oxygen despite adequate oxygen availability 7 ). AG is an energetically-inefficient metabolic process (~95% lower energy yield) yet plays an important role in cellular processes such as NAD+ homeostasis, 8 neural development, 9 synaptic plasticity and memory formation.10,11 Conversely, although the mechanisms are unclear, a loss of AG in the resting brain is associated with aging and neurodegeneration.12,13 Upon brain activation, such as during physical exercise14,15 and sensory stimulation,2,16,17 there is an increase in CBF alongside a decrease in oxygen extraction 18 and no change in glucose extraction, 19 resulting in the disproportionate uptake of glucose relative to oxygen (i.e., AG).14,16,17 The link between AG and CBF upon brain activation indicates that glucose and oxygen metabolism may behave differently upon variation in delivery.
The relevance of understanding how brain glucose and oxygen extraction change with delivery are paramount in the myriad of conditions where CBF is known to be reduced 20 such as Alzheimer’s disease,21,22 hypertension, 23 and ischemic stroke. 24 Similarly, in response to probable reductions in CBF, many studies of healthy aging report stable CMRO225,26 alongside increasing OEF, while others report decreases in CMRglc,27–29 which is in accordance with an age-related decline in AG. 12 These outcomes support the notion that oxygen extraction is more responsive than glucose extraction to changes in CBF, which may also result in a decline of AG with reductions in CBF—a complementary yet inverse process with what is known to occur upon brain activation.
Here we have accumulated resting measures of CBF using duplex ultrasound alongside arterial and internal jugular bulb concentrations of oxygen and glucose in 75 healthy humans (21 females) aged 21–41 years. With this, we have examined and compared the interindividual relationships of cerebral oxygen extraction fraction (OEF) and glucose extraction fraction (GEF) with CBF and the resultant changes in glucose and oxygen metabolism relative to one another. We suggest that CBF may be a critical determinant for basal levels of CMRglc and AG due to a lack of adjustment in glucose extraction by the brain, reflecting what is well-established during brain activation.
Methods
Experimental model and subject details
We collected cerebral blood flow alongside arterial and internal jugular venous bulb blood sample data from the baseline measures of eight studies (three unpublished)30–34 in humans, all conducted at the University of British Columbia Okanagan. All studies recruited only healthy, physically-active, adults with no known cardiovascular or neurological diseases between the ages of 21–41 years old (mean 27.3 ± 4.8 years), with a total of 75 individuals (21 females). All studies were approved by the University of British Columbia Clinical Research Ethics Board and carried out in accordance with the Declaration of Helsinki and informed consent was obtained from all participants.
Method details
Cross-brain blood sampling
In each study, arterial and central venous catheters were placed with the participant supine, using a sterile technique under local anesthesia, and assisted via the use of ultrasound guidance. To accurately isolate cerebral metabolism it is critical that blood sampled from the internal jugular vein comprises no contamination from extra-cerebral blood. This was done via cranial advancement (~15 cm) of the catheter, placing the catheter tip in the jugular bulb. 35 Following this approach, it has been demonstrated to lead to >97% sampling of venous blood isolated from the brain. 36 Blood samples from the artery (brachial or radial) and internal jugular vein were drawn simultaneously and immediately analyzed using a commercial blood gas analyzer (ABL90 FLEX Radiometer).
Cerebral blood flow (CBF)
Simultaneous diameter and velocity of the right internal carotid artery (ICA) and left vertebral artery (VA) were measured using 10 MHz multi-frequency linear array duplex ultrasound (Terasmart uSMart 3300, Teratech, Burlington, MA, USA). Simultaneous B-mode imaging and pulse-wave mode were used to measure live arterial diameter and beat-by-beat peak velocity respectively.
Cerebral artery diameter and blood velocity were analyzed with custom wall tracking/edge detection software (BloodFlow Analysis, version 5.1). Volumetric blood flow (
Quantification and statistical analysis
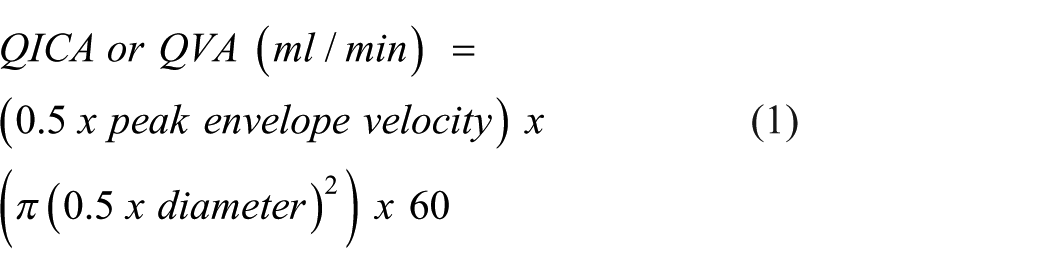
Global cerebral blood flow was calculated as twice the sum of unilateral assessments of ICA and VA blood flow:
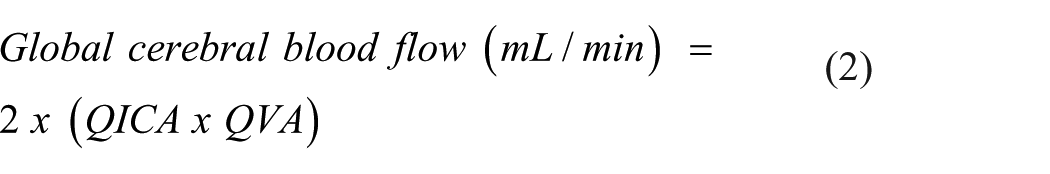
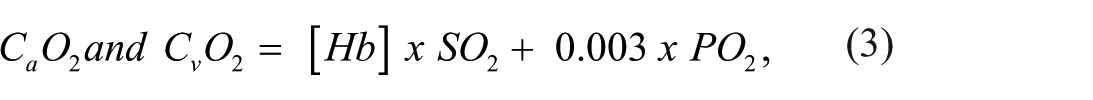
where arterial and venous oxygen content (CaO2 and CvO2) quantifies the total amount of oxygen in the blood such that [Hb] represents hemoglobin concentration, SO2 oxygen saturation and PO2 partial pressure of oxygen.
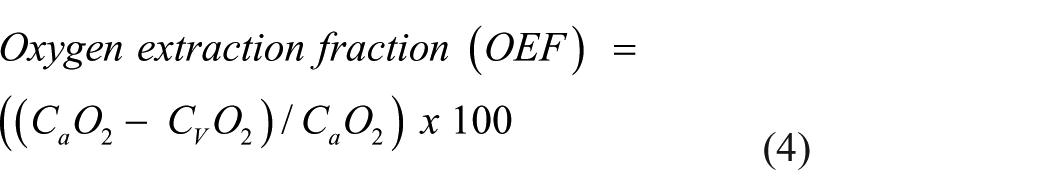
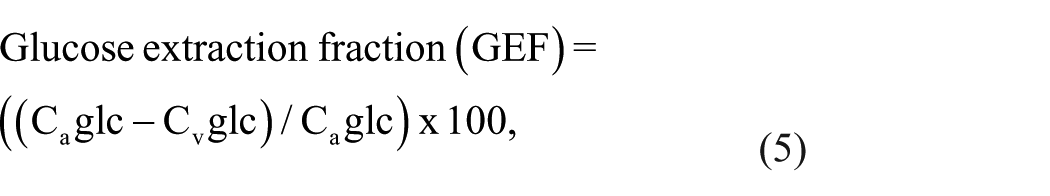
where Caglc and Cvglc represent the arterial and venous concentrations of glucose in mmol/L.
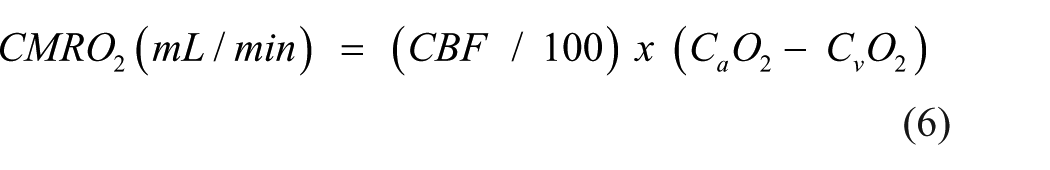
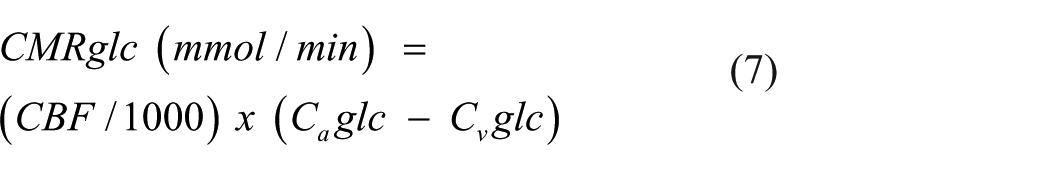
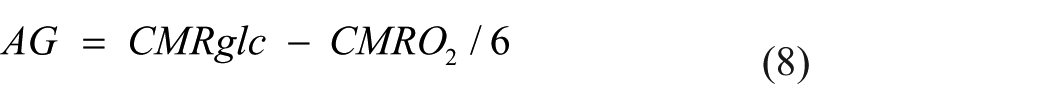
Here, oxygen content is converted to mmol/L for the calculation of CMRO2 and divided by 6 to account for the 1:6 stoichiometric ratio of glucose:oxygen. AG is defined as the metabolism of glucose without oxygen despite adequate oxygen availability.7,37 Thus, the calculated value for AG provides the rate of excess glucose extracted by the brain in mmol/min relative to what can be accounted for by oxidative phosphorylation. A greater positive value indicates greater rates of AG while a negative value indicates unaccounted for sources of carbohydrates (e.g., lactate, ketones, and brain glycogen) undergoing oxidation.
Statistical analysis
All relationships were determined by a linear mixed effects model in R (R Statistical Software v4.4.1; R Core Team 2024). PaCO2, study, and sex were included as covariates to account for the direct effects of PaCO2 on CBF, the effect of brain size between males and females, and any effect of differences in experimenter techniques or standardizations between studies. Fourteen participants were involved in two or more studies (at least 1 year apart), thus subject identifiers were used as random intercepts. The variance inflation factor (VIF) was used to assess the effect of collinearity between variables such that a VIF less than 2 was deemed acceptable and suggesting in minimal effects on model estimates. Significance was set at
Results
Oxygen extraction inversely changes with variation in CBF, glucose extraction does not
In 75 resting healthy adult subjects (age 21–41 years, mean 27.3 ± 4.8 years), we present striking interindividual variability in CBF with the lower 10th percentile having CBF of <500 mL/min, whereas the upper 10th percentile have a CBF of >1000 mL/min. Additionally, OEF is on average 3.4-fold larger than GEF, with average values of 33.1 ± 6.8% and 9.8 ± 2.3%, respectfully. We then examined the relationships between oxygen and glucose extraction fractions for a given CBF by plotting each subject’s OEF and GEF with their respective CBF (Figure 1(a) and (b)). We show that OEF decreases 1.4% for every dL/min increase in CBF (
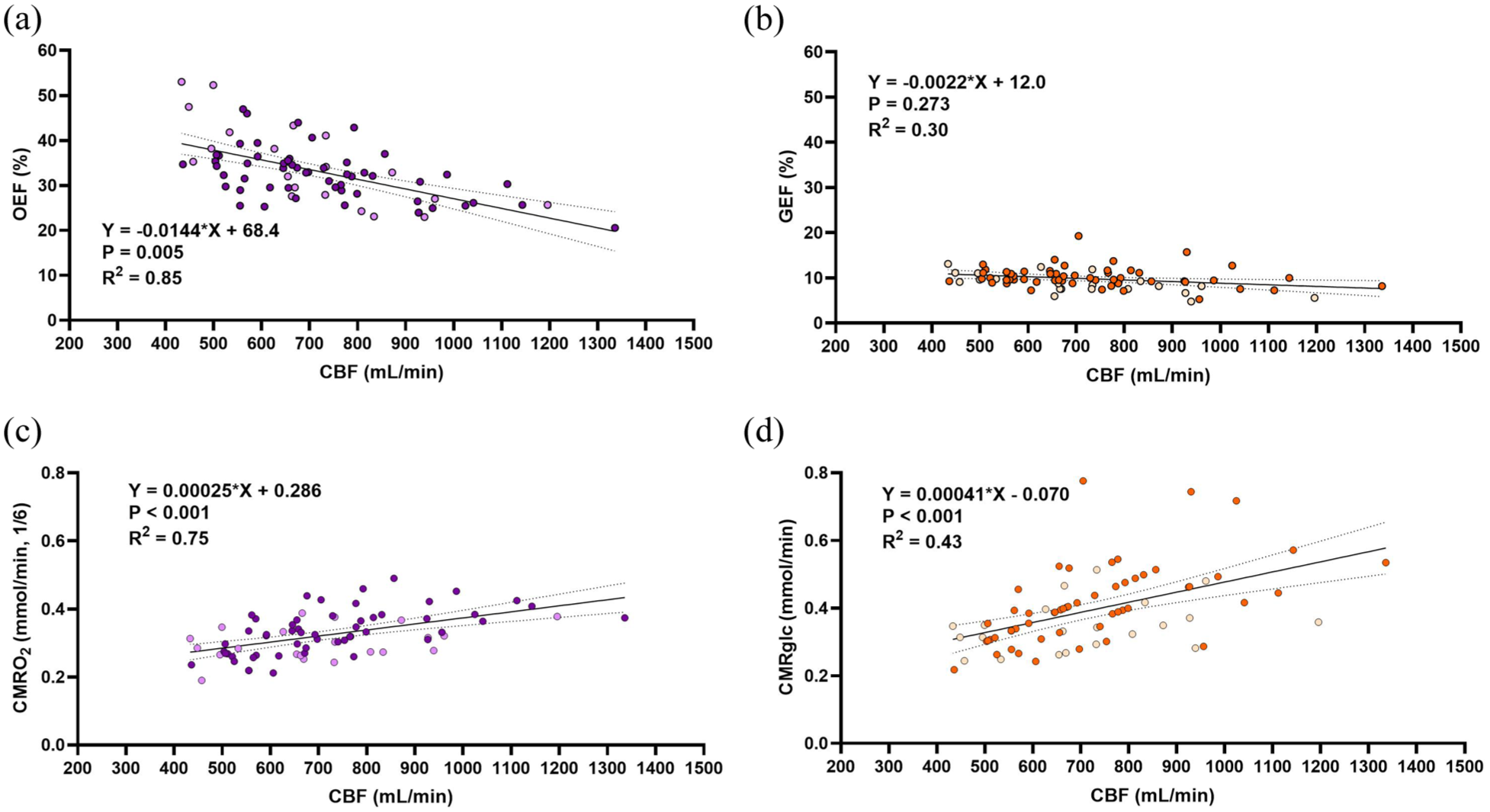
Oxygen extraction inversely changes with variation in CBF, glucose extraction does not. (a) Depicts the OEF-CBF relationship such that there is a significant inverse relationship between. (b) Depicts the GEF-CBF relationship such that there is a non-significant inverse relationship and a 6.5-fold lower slope than that of OEF-CBF (
We then determined the relationship between the metabolism of oxygen and glucose (CMRO2 and CMRglc) with the variability in CBF (Figure 1(c) and (d)). CMRO2 is scaled to it’s stoichiometric ratio for glucose (divided by 6) to account for the greater oxygen extraction required to facilitate oxidative glucose metabolism. Both relationships yield a significant, positive slope, suggesting that the significant changes in OEF do not fully accommodate differences in CBF, that is, a perfectly accommodating extraction fraction would yield stable CMRO2. However, upon comparing the slopes of the CMRO2-CBF and CMRglc-CBF relationships (Figure 2(a)), we revealed that CMRglc is 1.6 times more dependent on CBF compared to CMRO2 (slopes 0.00041 vs 0.00025,
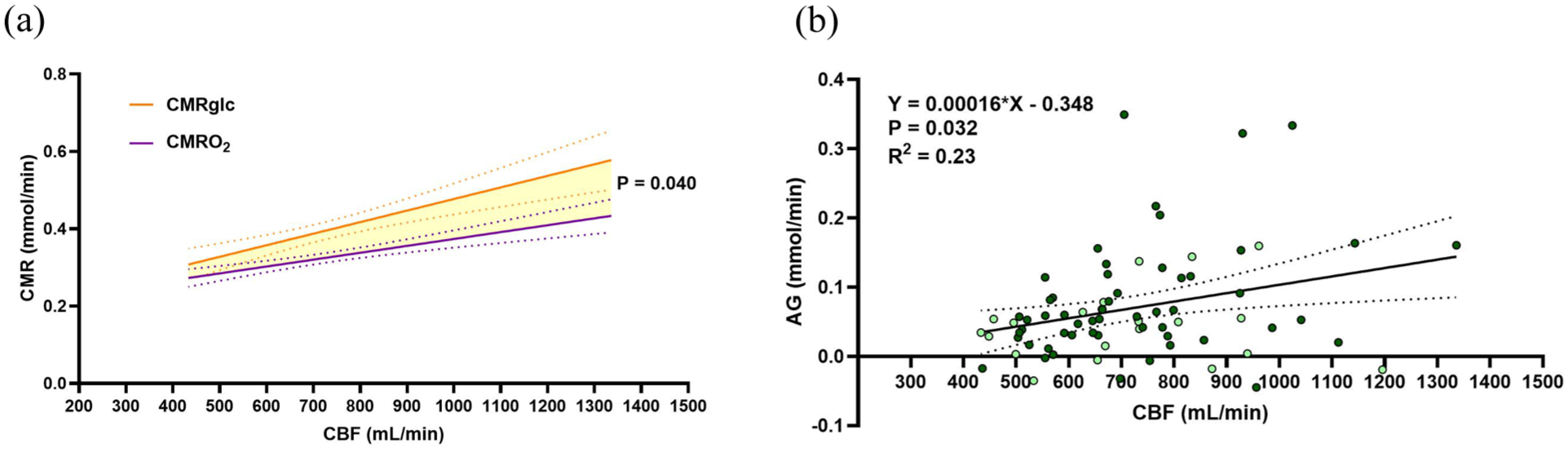
Inability of glucose extraction to accommodate low CBF underlies low rates of AG. (a) Depicts both the relationship of CMRglc and CMRO2 on the same axis, with a significant difference between slopes. (b) Depicts the AG-CBF relationship which indicates that with an increase in CBF there is an increase in excess glucose metabolism relative to oxygen. In both figures,
Variance inflation factors (VIF) were established to assess effects of multicollinearity on the previous regressions due to CBF being a component in the calculation of both CMRO2 and CMRglc. All values were below 2, indicating an acceptable level of collinearity that is unlikely to significantly impact model estimates.
Inability of glucose extraction to accommodate low CBF predicts low rates of AG
To extend on the finding that CMRglc varies more with interindividual differences in CBF (Figure 2(a)) we quantified individual resting levels of aerobic glycolysis according to equation (8) (see Methods for details on calculation and interpretation). A linear regression of AG and CBF (Figure 2(b)) resulted in a positive slope (
This analysis was repeated using normalized CBF (nCBF) expressed in terms of 100 g of brain mass. The average brain volume based on age and sex reported in a recent metanalysis 38 was used to estimate brain mass for each individual. 39 The overall outcomes were the same, demonstrating that brain mass does not explain the discrepancy between oxygen and glucose extraction across the wide range of CBF (see Supplemental Material).
Discussion
We have demonstrated that brain glucose extraction does not adapt to striking variability in resting CBF, while oxygen extraction does. Cerebral metabolism is derived from the product of the rate of substrate delivery and the arterial-venous difference (i.e., extraction).40,41 As such, the way substrate extraction responds (or does not respond) to changes in delivery will determine the metabolic rate. Thus, the greater dependence of glucose metabolism on delivery suggests that low CBF places greater strain on CMRglc compared to CMRO2. This inability of the brain to adjust glucose extraction underlies the observation that low CBF is directly related to low rates of AG. The inverse relationship of OEF with oxygen delivery is a well-established phenomena in the dynamic response to brain activation17,42 (providing the fundamental basis for BOLD-MRI), yet is completely unexplored in the resting brain across individuals where CBF varies substantially.
A possible explanation for the broader range of compensable oxygen metabolism relative to glucose is the difference in oxygen and glucose transport across the blood brain barrier and into cells. Oxygen follows passive diffusion which depends on oxygen tension gradients between blood and tissue, 43 while glucose influx requires facilitated diffusion through GLUT1/GLUT3 transporters,44,45 which can be influenced by transporter saturation and expression levels. Therefore, while CBF and extraction may covary differently for each substrate, the disproportionate rates of glucose and oxygen metabolism at either extreme of delivery could reflect the differing mechanisms of their transport. Additionally, an increase in substrate delivery can occur via increases in circulating concentrations. Recent work by Blazey et al., whereby circulating glucose was elevated to 17 mmol/L, showed an increase in CMRglc but not CMRO2, that is, elevated AG, in white matter with no change in CBF. 46 Thus, it appears that in the healthy brain CMRglc is somewhat dependent on glucose delivery, whether due to alterations in flow (CBF) or circulating glucose concentration. This is in stark contrast to CMRO2, which is stable during hyperglycemia, 46 hypoglycemia, 47 hypoxia and hyperoxia. 48 Although we cannot discern cause-effect with our data, the disparate relationships between glucose and oxygen delivery and extraction fraction help explain the relationship between AG and cerebral glucose and oxygen delivery.
The understanding that with age and in several pathological states (e.g., Alzheimer’s disease, ischemic stroke, and cognitive impairment) humans experience a decrease in CBF20,49,50 as well as glucose hypometabolism21,22 suggests that these two variables are inextricably linked. This link poses a ‘chicken or the egg’ dilemma: Does a decrease in CBF facilitate brain glucose hypometabolism or is reduced CBF a reflection of lower metabolic demand? Given the finding that the age-related reduction in cerebral glucose metabolism is driven almost entirely by a reduction in AG
12
—a non-oxidative and
When considering these data and the proposed link between substrate delivery and metabolism in healthy human adults at rest, it is important to consider what may be driving the marked variability in CBF. As discussed, cerebral activation results in an increase in CBF due to reasons such as the removal of protons and maintenance of partial pressures of carbon dioxide and oxygen. 18 An increase in CBF during activation may also be a response to an increased demand for glucose. 51 While all measures presented here were taken under quiet, resting conditions, it is possible that confounding brain activation is present in some of these individuals, driving the variability in CBF and AG. Moreover, arterial levels of carbon dioxide (PaCO2), are an important determinant of CBF during wakefulness.52,53 Likewise, studies have attributed variations in OEF to differences in PaCO2, both at rest 54 and under experimentally induced changes. 55 As such, PaCO2 was included as a covariate to account for subject differences in arterial CO2 levels.
Overall, interindividual variability in CBF is accompanied by variability in oxygen but not glucose extraction, and this underlies the tight coupling between CBF and non-oxidative glucose metabolism, i.e., aerobic glycolysis. This can be attributed to a smaller change in oxygen metabolism for a given CBF relative to glucose metabolism, an outcome which is well-established during intraindividual brain activation but undescribed in interindividual CBF variability. Future work should aim to examine this relationship in aging and pathological reductions of CBF to establish underlying reasons for reductions in glucose metabolism and AG.
Supplemental Material
sj-csv-2-jcb-10.1177_0271678X251400247 – Supplemental material for Brain glucose extraction is fixed at 10% despite twofold variability in resting cerebral blood flow in healthy humans
Supplemental material, sj-csv-2-jcb-10.1177_0271678X251400247 for Brain glucose extraction is fixed at 10% despite twofold variability in resting cerebral blood flow in healthy humans by Jennifer S Duffy, Hannah G Caldwell, Ryan L Hoiland, Connor A Howe, Kurt J Smith, Anthony R Bain, David B MacLeod, Philip N Ainslie and Travis D Gibbons in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X251400247 – Supplemental material for Brain glucose extraction is fixed at 10% despite twofold variability in resting cerebral blood flow in healthy humans
Supplemental material, sj-docx-1-jcb-10.1177_0271678X251400247 for Brain glucose extraction is fixed at 10% despite twofold variability in resting cerebral blood flow in healthy humans by Jennifer S Duffy, Hannah G Caldwell, Ryan L Hoiland, Connor A Howe, Kurt J Smith, Anthony R Bain, David B MacLeod, Philip N Ainslie and Travis D Gibbons in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Author contributions
Conceptualization and Methodology, JSD and TDG; Formal Analysis, JSD; Writing—Original Draft, JSD and TDG; Investigation, Data Curation, Writing—Review & Editing, JSD, HGC, RLH, CAH, KJS, ARB, DBM, PNA, TDG; Supervision, PNA and TDG.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The dataset supporting the findings of this study is available as a supplementary CSV file submitted alongside the manuscript. Further details are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
