Abstract
Cerebral small vessel disease (CSVD) is associated with vascular risk factors (VRFs) and early executive function (EF) decline. Because periventricular white matter (PVWM) is the most weakly perfused brain region, it may be particularly sensitive to early manifestations of CSVD. Among 101 cognitively unimpaired adults, we assessed the cross-sectional correlations of arterial spin-labeled derived absolute and relative cerebral blood flow (CBF and rCBF, respectively) and diffusion tensor imaging (DTI) metrics in normal-appearing WM (NA-WM) and normal-appearing PVWM (NA-PVWM) with VRFs and EF, and evaluated the mediation and moderation relationships between these variables in amyloid β negative (Aβ−) subjects. CBF, rCBF, and DTI metrics were significantly different in NA-PVWM compared to NA-WM (p < 0.001). In multivariate analysis of the Aβ− group, NA-PVWM rCBF was associated with VRFs (p = 0.012), while NA-PVWM rCBF (p = 0.035) and mean diffusivity (MD) in NA-WM (p = 0.022) and NA-PVWM (p = 0.029) were associated with EF. The association between VRF and EF was not mediated by rCBF and/or MD. However, increased MD was significantly associated with poorer EF for NA-PVWM CBF ⩽16.3 mL/100 g/min and NA-PVWM rCBF ⩽0.35. NA-PVWM rCBF is associated with VRFs and EF and might be an early biomarker for CSVD, especially in the absence of amyloid pathology.
Keywords
Introduction
Cerebral small vessel disease (CSVD) is the primary disorder underlying vascular cognitive impairment (VCI) and refers to progressive pathologies damaging small end arteries, arterioles, capillaries, and venules, most notably arteriolosclerosis and cerebral amyloid angiopathy (CAA). 1 Aging and conventional vascular risk factors (VRFs) contribute to arteriosclerosis and result in reduced cerebral blood flow (CBF) and impaired cerebral autoregulation. These processes, along with the activation of inflammatory responses, lead to the loss of oligodendrocytes, demyelination, axonal damage, and decreased integrity of white matter (WM), which in turn affect cognition.1–4 Impairments in executive function (EF) and processing speed occur early in the course of the disease, while episodic memory, which is commonly affected in Alzheimer’s disease (AD), is relatively preserved.5–7
CSVD lacks specific predictive biomarkers. White matter hyperintensities (WMHs) on fluid-attenuated inversion recovery (FLAIR) T2-weighted magnetic resonance imaging (MRI) are commonly considered as an imaging hallmark of CSVD.8,9 However, WMHs are not specific for CSVD10,11 and CSVD-related brain damage is not limited to visible WM lesions. Diffusion tensor imaging (DTI) can detect microstructural changes in normal-appearing WM (NA-WM) before the emergence of WMHs, even in cognitively unimpaired (CU) adults.12–14 Longitudinal and cross-sectional studies suggest that common DTI metrics such as fractional anisotropy (FA), mean diffusivity (MD), axial diffusivity (AxD), and radial diffusivity (RD) in normal-appearing WM (NA-WM) are predictors of WMH progression and cognitive decline.15,16
CBF can be quantified noninvasively using arterial spin labeled (ASL) perfusion MRI. 17 Previous studies applying ASL to CSVD and VCI 18 have suggested that alterations in global or regional CBF occur before the clinical onset of VCI and can predict cognitive decline.18–21 Thus, CBF measurements may identify individuals at risk of VCI in preclinical stages. While most ASL MRI studies have focused on gray matter (GM) or whole brain CBF, recent improvements in ASL MRI technology now enable the quantification of WM CBF. 22 Periventricular WM (PVWM) is the most common site of WMHs in normal aging. 8 This region is supplied exclusively by small vessels (<100 μ) and is the most weakly perfused region in the brain.23–25 Accordingly, both CBF and WM microstructural integrity in the PVWM region might be affected by VRFs earlier than other regions and may potentially also contribute to early clinical manifestations of CSVD, such as EF impairment. In our previous study, reduced CBF in PVWM was observed across the Alzheimer’s disease (AD) spectrum and was associated with VRFs. 26
Although the current literature suggests an association between VRFs, global and regional CBF, and WM microstructural injury with cognitive decline,27–29 whether changes in CBF and WM microstructural integrity mediate the effect of VRFs on EF remains unclear. Additionally, whether hypoperfusion accelerates the impact of WM microstructural injury on cognition is also uncertain. In this study, we used ASL and DTI to examine changes in CBF and microstructural integrity in non-lesional WM as they relate to VRFs and EF in cognitively unimpaired (CU) older adults with a specific focus on comparing the periventricular region to the whole WM. Amyloid-beta (Aβ) pathology is also linked to WM changes 30 and interacts with VRFs in relation to CBF. 31 Since AD is the most common cause of dementia and often occurs in conjunction with VCI due to CSVD, in the current study, we also categorized participants based on Aβ status to detect the presence of preclinical AD and investigated the associations of imaging metrics with VRFs presence and EF in the total cohort and each group. We aimed to test the following hypotheses: (1) Perfusion and diffusion characteristics in normal-appearing PVWM (NA-PVWM) differ from normal-appearing whole-brain WM (NA-WM). (2) CBF in NA-PVWM is more sensitive to the presence of VRFs in normal aging than other imaging metrics such as CBF in whole WM, GM, and global CBF, DTI metrics, and WMH volume. (3) CBF and DTI metrics in NA-PVWM can predict early changes in EF in CU older adults. (4) VRF presence is associated with poorer EF, that is, mediated through changes in CBF and microstructural integrity. (5) CBF moderates the effect of microstructural WM injury, as measured by DTI metrics, on EF.
Methods
Study design and participants
Cross-sectional data was obtained from the Penn Alzheimer Disease Research Center (ADRC) cohort. We included a total of 101 CU adults older than 50 years, including 78 amyloid beta (Aβ) negative (Aβ−) and 23 Aβ positive (Aβ+) participants, based on the availability of DTI, ASL, and Aβ positron emission tomography (PET) scans. All participants completed multimodal neuroimaging, neurological examination, and extensive neuropsychological evaluation using the Uniform Data Set (UDS) v.3.0. Diagnostic categorization was determined by consensus using a comprehensive neuropsychological battery as well as neurological and supportive neuroimaging assessments by expert clinicians. The study design was in line with the Declaration of Helsinki. 32 The Penn ADRC study protocol was approved by the University of Pennsylvania Institutional Review Board (IRB; approval number: 068200), and informed written consent was obtained from all participants. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines. 33
VRFs and amyloid status assessment
VRFs included hypertension (defined as systolic blood pressure ⩾140 mmHg or diastolic blood pressure ⩾90 mmHg, measured by a trained research coordinator by averaging two blood pressure readings in both arms after >5 min of rest using a validated device, or being treated for hypertension), diabetes mellitus, obesity (BMI ⩾30), hypercholesterolemia (defined by physician judgment based on medical chart data, or use of lipid-lowering medications), and current smoking. We categorized participants into those with VRFs if they had at least one VRF and those without any VRFs.
To determine amyloid status, the temporally closest Aβ PET scan using either intravenous 18F-florbetaben or 18F-florbetapir as the tracer was used, 34 and amyloid status was classified based on visual read by an expert nuclear medicine physician (IMN) with 13 years of experience. 35
EF evaluation
Participants underwent the UDS v.3.0 neuropsychological battery. In UDS v.3.0, the trail-making test part B (TMT B) is used to measure EF. 36 For this study, we used TMT part B minus TMT part A (TMT B-A) to more specifically assess EF while accounting for the variations in processing speed, visual search, and motor function.37–39
MRI acquisition
A Siemens Prisma 3T MRI scanner was used for image acquisition. MRI sequences assessed in this study were as follows: T1-weighted structural MRI acquired using magnetization-prepared rapid gradient echo (MPRAGE) protocol with repetition time (TR) = 2400 ms, echo time (TE) = 2.24 ms, inversion time (TI) = 1060 ms, flip angle = 8°, voxel size = 0.8 × 0.8 × 0.8 mm3, field of view (FOV) = 240 × 256, 208 sagittal slices, and bandwidth = 210 Hz/px. T2-weighted fluid-attenuated inversion recovery sequence (FLAIR) acquired with TR = 6000 ms, TE = 289 ms, TI = 2200 ms, voxel size = 1 × 1 × 1 mm3, FOV = 221 × 258 mm², 160 sagittal slices, and bandwidth = 930 Hz/px, was acquired to identify WMH. DTI acquired using turbo-spin echo planar imaging with TR = 3027 ms, TE = 82.8 ms, flip angle = 78, voxel size = 1.5 × 1.5 × 1.5 mm3, acquisition matrix = 140 × 140°, 90 axial slices, 15 diffusion-encoding directions at b = 300, 30 directions at b = 800, 64 directions at b = 2000, and nine volumes at b = 0. ASL MRI was acquired using unbalanced pCASL with 1.8 s labeling duration and 1.8 s post-labeling delay (PLD) with 90% background suppression and a 1D-accelerated four-shot 3D stack-of-spirals acquisition (TR = 4.25 s, TEeff = 9.78 ms) at a nominal isotropic voxel resolution of 2.5 mm3 (matrix = 96 × 96, flip angle = 90°, FOV = 240 × 240 mm2, bandwidth = 400 Hz/px). Two M0 images, acquired without any magnetization preparation, were averaged and used to normalize the control-label difference for CBF quantification. Ten label/control pairs were acquired for signal averaging in ~5.5 min.
MRI data processing
WMH segmentation
WMH lesions on FLAIR images were segmented using the nnU-Net, a deep learning-based approach for medical image segmentation. 40 We previously described the detailed methodology for the model training. 41 The binary WMH masks were inspected for accuracy and manually corrected as needed. The WMH masks were mapped to the subject’s T1-weighted structural image. The total volume of WMH lesions for each subject was calculated, normalized to the intracranial volume (ICV), and log-transformed due to its skewed distribution.
ROIs
T1-weighted images were segmented into gray matter (GM), WM, and cerebrospinal fluid (CSF) tissue probability maps using SPM12. For extraction of mean CBF and DTI metrices, GM and WM masks were created by thresholding the probability maps to 0.9 and 0.99, respectively. A whole brain (WB) mask was obtained by thresholding the combined GM and WM probability maps to 85%.
We used a previously defined CBF-based functional PVWM ROI. 23 Briefly, a group-averaged CBF map was derived from 436 healthy middle-aged subjects enrolled in the Coronary Artery Risk Development in Young Adults (CARDIA) study. The lowest perfusion in the group-averaged map was found in the periventricular (PV) region. In the current study, we used a PVWM ROI in MNI152 space defined by group CBF ⩽15 ml/100 g/min (see Supplementary Figure 1). The PVWM ROI was transformed to the participant T1 space by applying the inverse warping of FSL FNIRT. For each participant, we discarded voxels overlapping the ventricles, as segmented by SPM, from the periventricular mask to account for possible non-linear registration inaccuracy. To generate NA-WM and NA-PVWM ROI masks, voxels within WMH lesion masks were excluded.
ASL processing and CBF quantification
ASL data were processed using in-house automated pipelines developed in MATLAB, FSL, and SPM12, 23 following the recommendations by Alsop et al. 17 The processing consists of motion correction, pairwise subtraction of the ASL control-label pairs, and conversion to absolute CBF quantification using a single-compartment model. We used an automated quality evaluation index (QEI) to objectively evaluate the quality of mean CBF maps. 42 QEI values vary between 0 and 1, and higher values suggest a better quality of CBF maps. CBF maps with QEI <0.5, a predetermined threshold shown to have good sensitivity and specificity for low-quality maps, 42 were discarded. Subsequently, CBF maps were co-registered to the T1-weighted imagea using the FSL boundary-based registration approach. Global CBF was defined as the mean CBF in the whole brain. Applying the ROI masks, we extracted the mean CBF in GM, NA-WM, and NA-PVWM. We additionally calculated NA-PVWM relative CBF (rCBF) by normalizing NA-PVWM CBF to the global CBF. 43
DTI processing
DTI data processing was performed using a previously described in-house pipeline 35 and included denoising, Gibbs-unringing, ANTs N4-bias correction using MRItrix3, and eddy correction and distortion correction using FSL eddy with outlier replacement.44–51 For quality control, we carried out both visual inspection and eddy QUAD and excluded scans with >3.5% of overall slice outliers, >20% outliers for a single shell, and CNR <3.5 SDs from the group mean. 52 FA, MD, RD, and AxD maps were generated using FSL dtifit through a voxel-wise tensor model. The FA maps were used for the co-registration of DTI data to T1-weighted images using ANTs linear affine registration. 53 We calculated the mean FA, MD, AxD, and RD in the NA-PVWM and NA-WM ROIs.
Structural MRI correlates of CSVD: Lacunar infarcts, Fazekas scores, and perivascular spaces (PVS)
FLAIR images in the study cohort were assessed for lacunar infarcts and Fazekas scores by JAD, a neurologist with expertise in neuroimaging. Only one participant had a lacunar infarct. Total Fazekas score was highly correlated with WMH volume (R = 0.836, p < 0.001). In the analyses, quantitative WMH volume was used to increase accuracy. Automated PVS segmentation was performed on T1-weighted images using an established method. 54 CSF spaces were excluded from the WM PVS segmentation. 55 The total PVS voxel count was calculated and normalized to the ICV and was log-transformed.
Statistical analysis
STATA version 15.1 56 and R were used for statistical analysis and Python and R were used to generate the plots.
We tested the normal distribution of all variables using the histograms and Shapiro–Wilk test. We used the two independent sample T-test and Wilcoxon-rank sum test to compare quantitative variables with and without a normal distribution, respectively, between 78 Aβ− and 23 Aβ+ participants. Either mean and standard deviation (SD) for variables with a normal distribution or median and interquartile range (IQR) for variables with a non-normal distribution were reported. Chi-squared and Fisher’s exact tests were used to compare qualitative variables between the two groups. We further compared CBF and DTI metrics in NA-PVWM and NA-WM using two sample T-tests. Associations between VRF presence and imaging metrics were assessed using linear regression analysis with each imaging metric as the dependent variable and VRF presence as the independent variable in simple linear regression models, and VRF presence, age, and sex as the independent variables in multiple linear regression models in the total cohort and each Aβ− and Aβ+ group. We used a similar regression model to assess the association of TMT B-A with VRF presence while controlling for age, sex, and years of education. Further, we evaluated the association of TMT B-A with each imaging metric through linear regression models adjusting for age, sex, and years of education in the total cohort and each Aβ− and Aβ+ group. Standardized β provides an approximation to the correlation coefficient and is comparable between variables of interest, and was reported for all the linear regression analyses. To better understand if there is any mediation or moderation relationship between VRFs, CBF, MD, and EF, we conducted mediation and interaction analyses. Mediation relationships between VRF presence, CBF, MD, and TMT B-A were assessed by Process package version 4.3.1 in R. 57 All interaction effects and Johnson-Neyman intervals were calculated using the Process and Interactions packages in R, respectively.57,58 To remove the possible confounding effects of amyloid pathology, we performed the mediation and interaction analyses only in the Aβ− older adults who represented a group unlikely to have the contribution of amyloid angiopathy. Two subjects with missing data were discarded from the mediation and moderation analyses (N = 76). Statistical power for moderation and mediation analyses was calculated through the Monte Carlo simulation method using the “Mass” Package in R and was estimated at 0.42 and 0.36 for moderation analyses with NA-PVWM CBF and NA-PVWM rCBF as moderators, respectively, and at 0.99, 0.98, and 0.02 for total, direct, and indirect effects, respectively, in the final mediation pathway. All statistical tests were two-sided, and statistical significance was set at 0.05 level. We did not correct for multiple comparisons due to the exploratory nature of this study. 59
Results
Baseline characteristics and imaging metrics in Aβ− and Aβ+ subjects
After the exclusion of participants with unknown amyloid status, 101 CU older adults were included in the analysis. The baseline characteristics and imaging metrics are compared between Aβ− and Aβ+ groups in Table 1. The mean age of the cohort was 71, ranging from 59 to 80 years and 62% were female. Ever smoking was significantly more frequent in Aβ− group compared to the Aβ+ group (p = 0.002).
Baseline characteristics and imaging metrics in Aβ− and Aβ+ subjects.
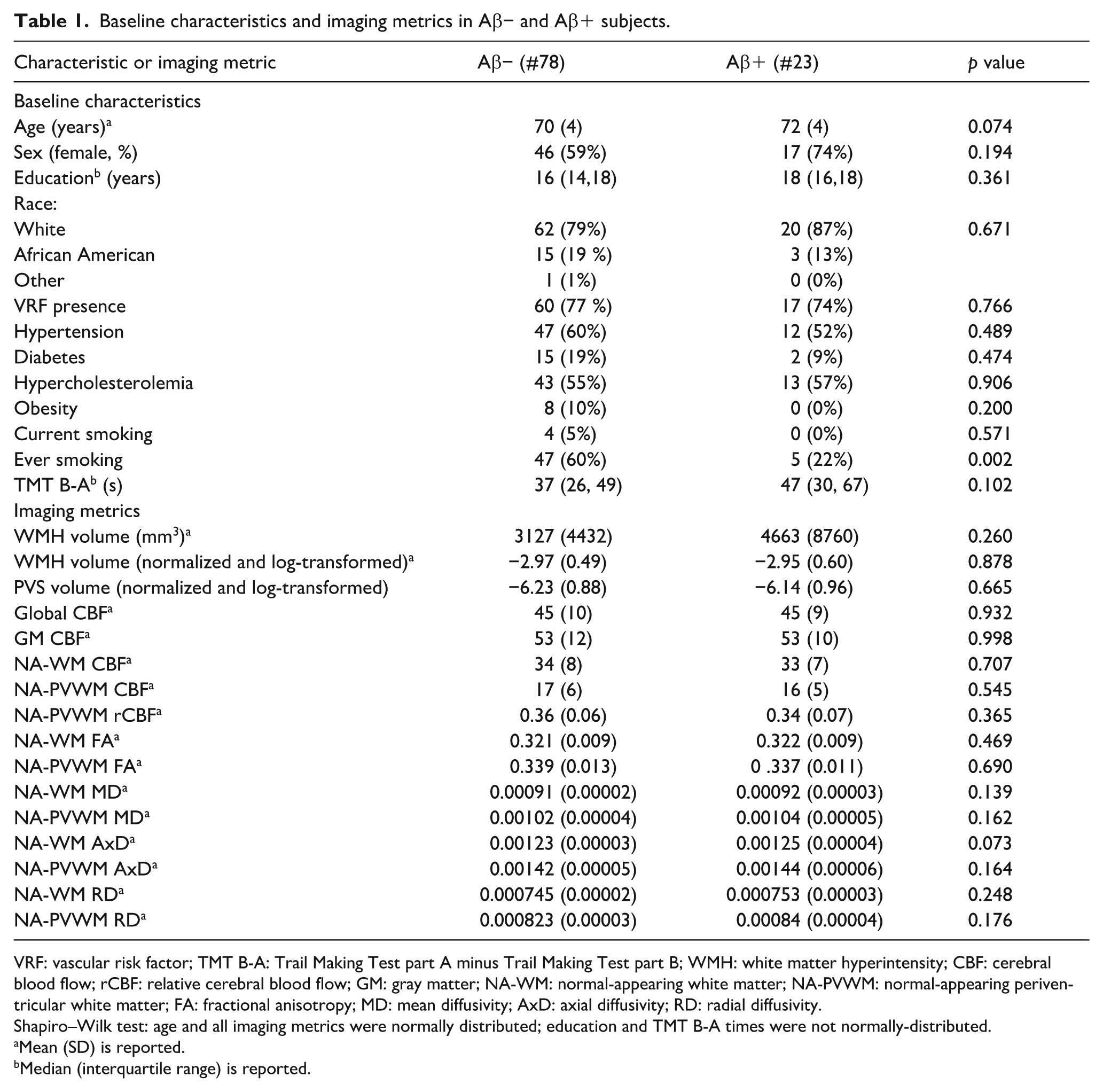
VRF: vascular risk factor; TMT B-A: Trail Making Test part A minus Trail Making Test part B; WMH: white matter hyperintensity; CBF: cerebral blood flow; rCBF: relative cerebral blood flow; GM: gray matter; NA-WM: normal-appearing white matter; NA-PVWM: normal-appearing periventricular white matter; FA: fractional anisotropy; MD: mean diffusivity; AxD: axial diffusivity; RD: radial diffusivity.
Shapiro–Wilk test: age and all imaging metrics were normally distributed; education and TMT B-A times were not normally-distributed.
Mean (SD) is reported.
Median (interquartile range) is reported.
Comparison of CBF and DTI metrics in NA-PVWM and NA-WM
In the total 101 participants, CBF and DTI metrics in NA-PVWM and NA-WM were compared as shown in Table 2. Both CBF and rCBF were significantly lower in NA-PVWM compared to the NA-WM. All DTI metrics were significantly higher in the NA-PVWM compared to the NA-WM (all p < 0.001).
Comparison of CBF and DTI metrics in NA-PVWM and NA-WM across the total cohort (N = 101).
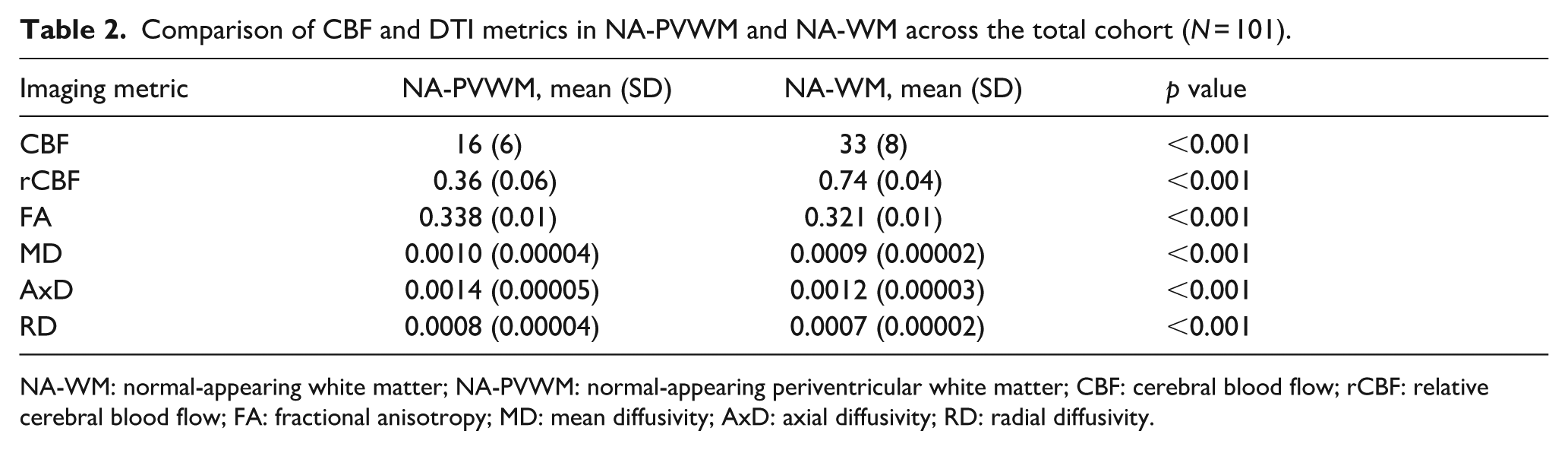
NA-WM: normal-appearing white matter; NA-PVWM: normal-appearing periventricular white matter; CBF: cerebral blood flow; rCBF: relative cerebral blood flow; FA: fractional anisotropy; MD: mean diffusivity; AxD: axial diffusivity; RD: radial diffusivity.
The association of VRF presence with imaging metrics and TMT B-A
The unadjusted and adjusted β for associations of VRF presence with imaging metrics (adjusted for age and sex) and with the TMT B-A (adjusted for age, sex, and education) are presented in Supplementary Table 1. Univariate analyses in the total cohort of 101 participants showed significant associations between VRF presence and CBF in all ROIs as well as rCBF in NA-PVWM. Among DTI metrics, FA in NA-PVWM and MD and RD in both NA-PVWM and NA-WM were significantly associated with VRF presence. Additionally, TMT B-A was significantly higher (indicating poorer performance) in those with VRF present (all p < 0.05). After adjusting for age and sex, none of the imaging measurements remained significantly associated with VRF presence. TMT B-A (adjusted β = 0.278, p = 0.009) remained significantly correlated with VRF presence after adjustment for covariates.
In the Aβ− group (N = 78), after adjustment for age and sex, NA-PVWM CBF and rCBF were significantly associated with VRF presence (adjusted β = −0.247, p = 0.043 and adjusted β = −0.324, p = 0.012, respectively). None of the DTI metrics were significantly associated with VRF presence in this group. After controlling for age, sex, and education, VRF presence significantly correlated with TMT B-A (adjusted β = 0.310, p = 0.012).
In the Aβ+ group (N = 23), only FA in NA-PVWM was significantly associated with VRF presence (adjusted β = −0.435, p = 0.043). Neither other imaging metrics nor TMT B-A were significantly correlated with VRF presence in this group.
WMH volume was not significantly associated with VRF presence either in the total cohort (unadjusted β = 0.109, p = 0.285) or in the Aβ− (unadjusted β = 0.166, p = 0.156) or Aβ+ (unadjusted β = −0.032, p = 0.884) groups. The results remained not significant after adjustment for age and sex.
There were no significant interactions between amyloid status and VRF presence in association with imaging metrics or TMT B-A performance (all p > 0.05; Supplementary Table 1).
The association of imaging metrics with EF
In the total cohort (N = 101), after adjustment for age, sex, and education, lower rCBF in NA-PVWM was associated with a poorer TMT B-A performance (adjusted β = −0.201, p = 0.048). Additionally, higher MD and RD in both WM ROIs and higher AxD in NA-WM were significantly associated with poorer TMT B-A performance (all p < 0.05).
Scatter plots of EF versus imaging metrics in the Aβ− group are shown in Figure 1. In the Aβ− group (N = 78), after controlling for age, sex, and education, NA-PVWM rCBF was significantly correlated with TMT B-A (adjusted β = −0.240, p = 0.035). Also, MD and RD in NA-PVWM and NA-WM and AxD in NA-WM were significantly associated with TMT B-A (all p < 0.05). In Aβ+ participants (N = 23), there was no significant association between any of the imaging metrics and TMT B-A performance (Supplementary Table 2).
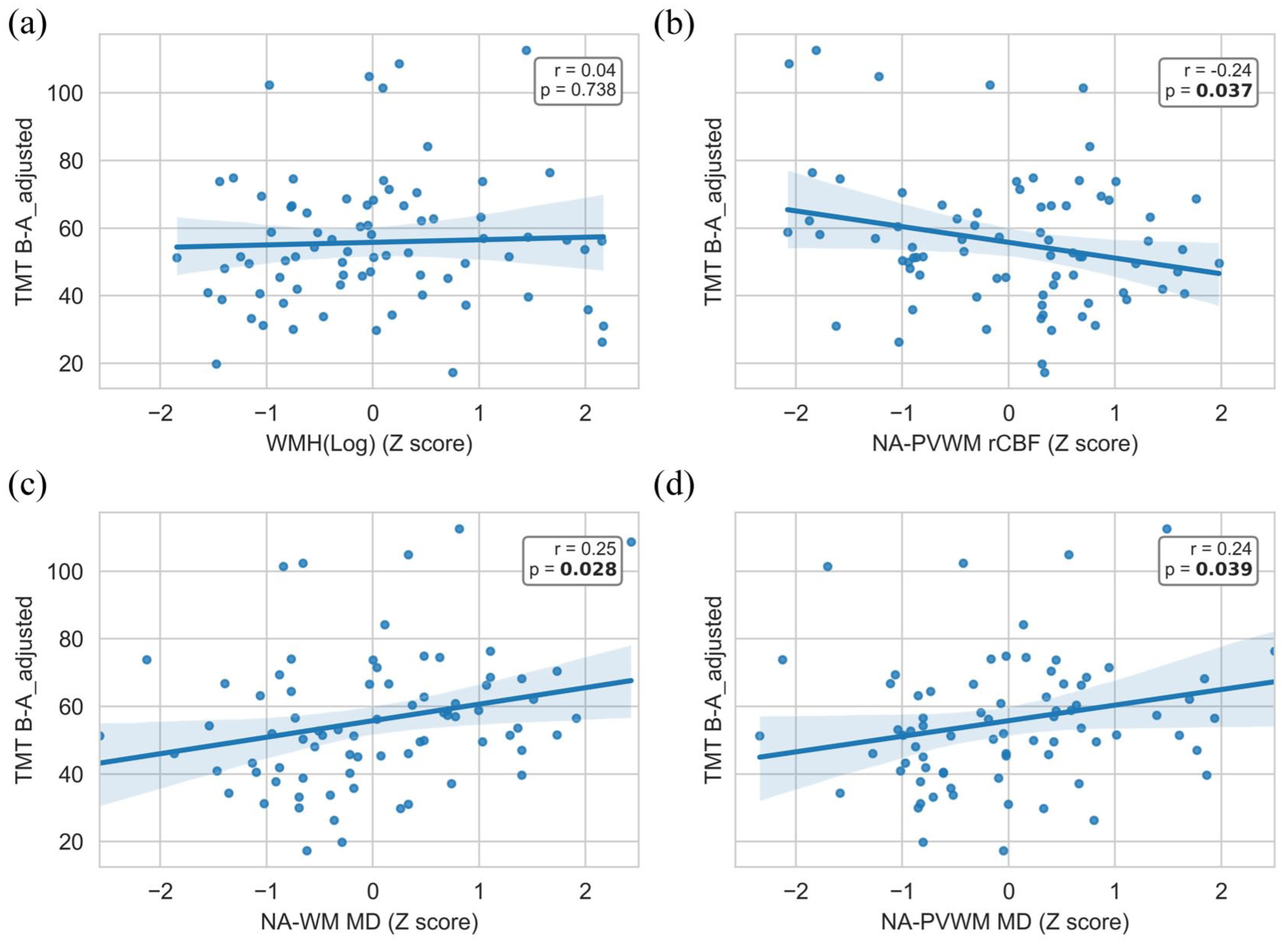
Scatter plots of TMT B-A adjusted for age, sex, and education versus WMH (a), NA-PVWM rCBF (b), NA-WM MD (c), NA-PVWM MD (d) Z scores in Aβ− participants.
WMH volume did not significantly correlate with TMT B-A in the total cohort and any group. In addition, there was no significant interaction between amyloid status and any imaging metrics in association with EF (Supplementary Table 2).
Mediation relationship between VRF presence, CBF, MD, and EF in the Aβ− group
Mediation analysis was performed among 76 participants with negative Aβ status. The association between NA-PVWM rCBF and TMT B-A performance was not significantly mediated by MD changes in the same region. Similarly, the association between VRF and TMT B-A performance was not mediated by changes in either rCBF or MD in the NA-PVWM, or through both rCBF and MD changes in the NA-PVWM (Table 3).
Mediation paths between VRF presence, CBF, MD, and TMT B-A in Aβ− group (N = 76) adjusted for age, sex, and education.
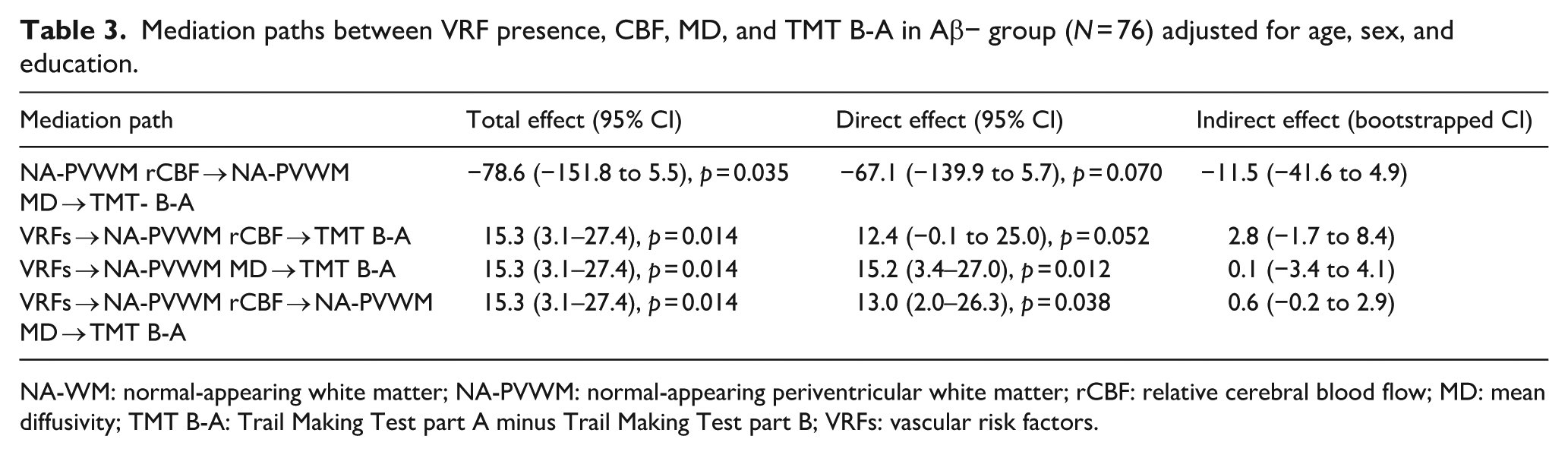
NA-WM: normal-appearing white matter; NA-PVWM: normal-appearing periventricular white matter; rCBF: relative cerebral blood flow; MD: mean diffusivity; TMT B-A: Trail Making Test part A minus Trail Making Test part B; VRFs: vascular risk factors.
Moderation effect of CBF on the relationship between MD and TMT B-A in the Aβ− group
Among 76 Aβ− participants, the moderating effects of NA-PVWM CBF and NA-PVWM rCBF on the relationship between NA-PVWM MD and TMT B-A were close to significant (NA-PVWM CBF* NA-PVWM MD effect size = −16982, p = 0.050, 95% CI = −33984, 19 and NA-PVWM rCBF* NA-PVWM MD effect size =−1402073, p = 0.088, 95% CI = −3018513, 214366, respectively). Figure 2 shows the NA-PVWM-related changes in EF in three different levels of CBF and rCBF (mean, low = mean − SD, and high = mean + SD). Johnson-Neyman interaction analysis showed that increased MD is significantly associated with poorer TMT B-A performance below the threshold of NA-PVWM CBF ⩽16.3 mL/100 g/min and NA-PVWM rCBF ⩽0.35, after controlling for age, sex, and education (Figure 3).
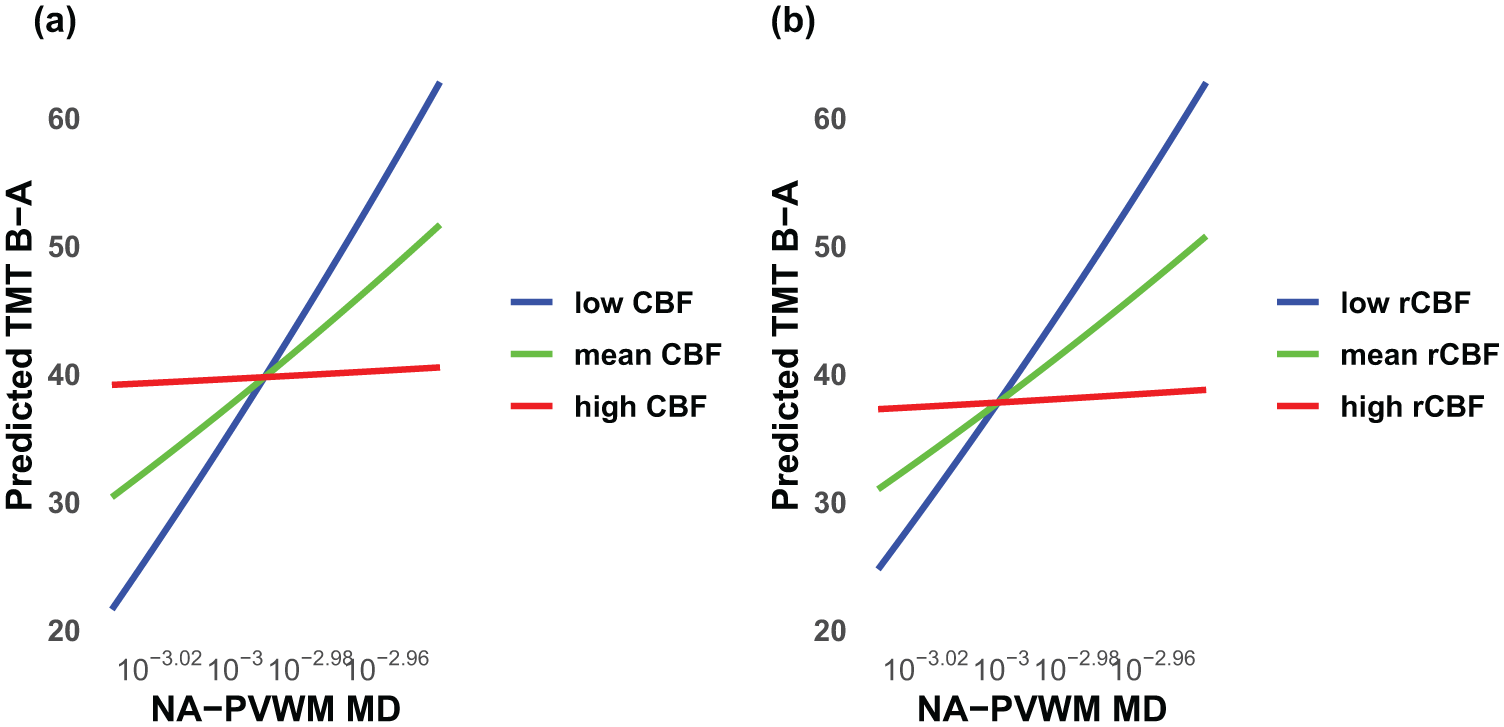
NA-PVWM MD-related TMT B-A changes in three levels (mean, low = mean − SD, high = mean + SD) of NA-PVWM CBF (a) and rCBF (b) in Aβ− participants controlled for age, sex, and education. Higher scores on TMT B-A represent poorer (slower) performance.
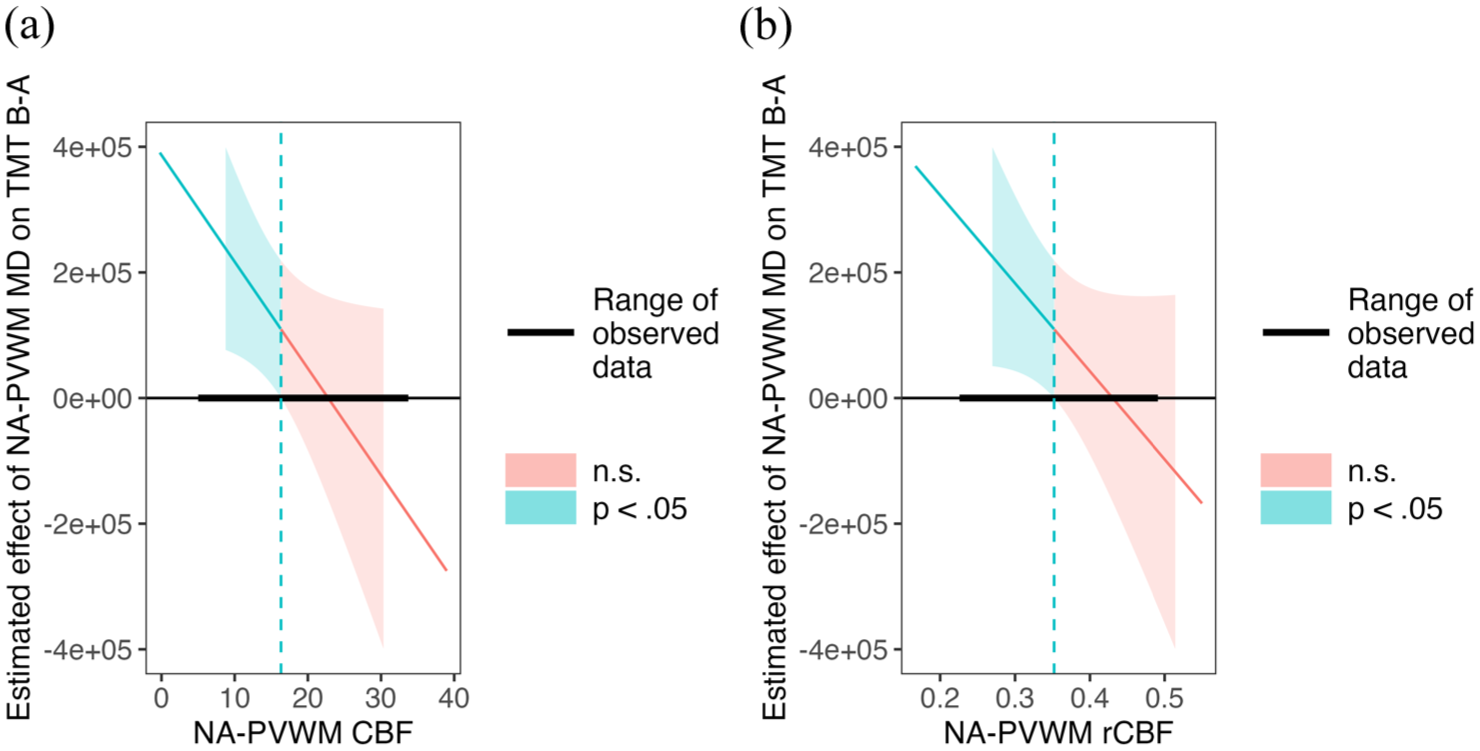
Johnson–Neyman plots showing regions of significance for the effect of NA-PVWM MD on TMT B-A across different values of NA-PVWM CBF (a) and NA-PVWM rCBF (b) in Aβ− participants controlled for age, sex, and education.
Discussion
In a cohort of CU older adults, we measured CBF and DTI metrics in NA-PVWM and NA-WM to evaluate whether these imaging metrics are associated with VRFs and EF changes, both of which might suggest the presence of early CSVD in a cognitively intact cohort. Our results demonstrated that the NA-PVWM has different perfusion and diffusion characteristics from the NA-WM in the whole brain. In addition, rCBF in the NA-PVWM was significantly associated with the presence of VRFs and EF impairment while the global, GM, and NA-WM CBF were not. Diffusion metrics in both NA-WM and NA-PVWM also correlated with EF. However, neither CBF nor MD significantly mediated the association of VRF presence with EF, though CBF and rCBF in NA-PVWM may moderate the relationship between microstructural integrity and EF, and lower NA-PVWM CBF and rCBF may contribute to accelerated EF decline with WM microstructural injury. Together, our findings suggest that rCBF in NA-PVWM might provide a biomarker of early CVSD.
NA-PVWM perfusion and diffusion characteristics
Significantly lower CBF and rCBF in NA-PVWM compared to the NA-WM is consistent with our previous studies and the known vasculature anatomy in this region.23,24,60,61 Significantly higher MD, AxD, and RD in NA-PVWM compared to the whole NA-WM might have several possible explanations. First, it might reflect increased microstructural injury, myelin loss, and axonal injury in this region. In addition, since PVWM has a high load of WMHs 8 and the PVWM is a small ROI, some of it may be penumbral. Studies have shown that MD increases in normal-appearing areas immediately adjacent to WMH lesions, while FA and CBF decreased in the WMH lesion penumbra.13,62 Furthermore, CSF has higher MD, AxD, and RD than NA-WM, 63 and partial volume effects due to CSF contamination could also have contributed to the observed effects despite our best efforts to ensure that the PVWM ROI does not overlap with ventricles. Our results also revealed an increased FA in NA-PVWM, which might reflect the presence of more linear fibers in this ROI than in WM more generally. 64
CBF and rCBF in NA-PVWM as an early biomarker of CVSD
Previous studies have demonstrated correlations between regional and global CBF and VRFs,65–67 though very few studies assessed the CBF in the PVWM region, 26 and did not focus on cognitively intact participants. Our results showed that only CBF and rCBF in NA-PVWM remained associated with VRFs in the Aβ− group after adjusting for covariates, while CBF in other regions, DTI metrics, and WMH volume did not. This observation supports our hypothesis that CBF and rCBF in NA-PVWM ROI may be more sensitive to early vascular pathology than the other imaging metrics measured in this study. In addition, a reduction in rCBF in NA-PVWM, among other ROIs, was associated with poorer EF, an early clinical correlate of CSVD. Because the NA-PVWM ROI is much smaller in volume than other ROIs and includes the most weakly perfused brain voxels, the presence of a significant VRF association lends further support to the sensitivity of this region to early CSVD changes. Continued refinements in ASL MRI acquisition methodology should allow CBF in PVWM to be measured more accurately in future work.
Although VRF prevalence was similar across the Aβ− and Aβ+ groups, the Aβ− group likely represented participants with purer vascular pathology in this study. The absence of the above associations in the Aβ+ group might be due to the smaller sample size in this group or reflect an effect of amyloid co-pathology. Indeed, the weaker associations between CBF metrics and EF in the total cohort compared to the Aβ− group suggest a complex effect of amyloid pathology on CBF. While our results failed to show any significant interactions between amyloid status and VRFs or imaging metrics, another recent study showed such interactions and suggested a compensatory increase of CBF in response to Aβ accumulation. 31
The lack of association between WMH volume with VRFs and EF might be due to low overall WMH volume in this cohort of cognitively intact older participants compared to previous studies.39,68 Hence, WMH changes might not be as sensitive as NA-PVWM rCBF in early CSVD.
The associations of DTI metrics and EF
The associations between diffusivity metrics and EF followed a more diffuse pattern and were not limited to the NA-PVWM. Although we accounted for visuomotor speed by subtracting TMT A from TMT B, TMT B-A is still time-dependent, and a global decrease in microstructural integrity across WM tracts affects processing speed. 69 This finding is consistent with our previous work showing that disconnection in multiple WM tracts, rather than a specific tract, is linked to TMT B-A performance decrements. 39
The mediation relationship between VRFs, CBF, MD, and EF
We assessed mediation paths to test the hypothesis that changes in CBF and, subsequently, in microstructural integrity mediate the effect of VRFs on EF (Table 3). We included only Aβ− participants in this analysis to avoid the confounding effect of Aβ co-pathology. Our analysis revealed that while both NAPVWM rCBF and MD are associated with EF, VRFs affect EF independently from changes in perfusion, microstructural integrity, or both. This aligns with a recent study by Shirzadi et al. suggesting that vascular risk affects cognitive decline in older adults beyond white matter injury and CBF. 27
WM microstructural injury in normal-appearing tissue precedes overt WMHs. 15 Although WMHs are typically assumed to be ischemic lesions related to VRFs, particularly hypertension,70–72 in a study of UK Biobank participants, Koohi et al. found that combined conventional VRFs only account for 15% of the variance in WMH volume, and over 60% of the variance remains unexplained. 68 This might explain the lack of causal relationship between VRF and WM microstructural injury as the precursor of WMHs, observed in our study.
The moderation effect of CBF on the association of microstructural integrity and EF
In the Aβ− group, mean NA-PVWM CBF and rCBF were 16.6 (ranging from 5.5 to 33) and 0.36 (ranging from 0.28 to 0.48), respectively. We found that for NA-PVWM CBF ⩽16.3 mL/100 g/min and NA-PVWM rCBF ⩽0.35, increased NA-PVWM MD significantly correlated with poorer EF performance. Moreover, with decreased perfusion, the magnitude of the effect of microstructural injury on EF increased. To the extent that MD provides an early marker of WM integrity changes, these findings suggest that higher perfusion may have a protective effect against WM injury-related cognitive decline. This might explain the better cognition with increased CBF observed in previous studies73,74 and underscores the potential benefits of interventions enhancing cerebral perfusion to prevent cognitive decline.
Limitations
Our study has several limitations. First, the ASL images were acquired with a single PLD. The optimal PLD is equal to the arterial transit time (ATT), which is the time required for the blood to reach the tissue of interest from the proximal labeling location. The current study used ASL data with a PLD of 1.8 s, which is close to the ATT of white matter reported to range between 1.5 and 1.7 s.22,75 One study reported that PVWM has an ATT of 1.8 s, 76 which could lead to a slight underestimation of CBF with our method. In addition, in the presence of VRFs, ATT might increase, which could lead to further CBF underestimation. Nevertheless, the underestimated CBF should still be proportionately related to true CBF, and hence, observed associations should remain valid. In addition, although the nominal resolution was 2.5 mm isotropic, the actual resolution in the slice direction was considerably lower due to T2 blurring. This could have caused some partial volume effects. In future work, further refinements in ASL MRI acquisition parameters could reduce these potential confounds.
Second, our sample size limited the statistical power and we did not correct for multiple comparisons due to the exploratory nature of this study. Due to our limited sample size, we also could not investigate the associations of each VRF with the imaging metric and EF separately. Instead, we simplified VRFs to their presence or absence. However, the effect of each VRF on the pathophysiology of CSVD and their interactions with each other might be more complicated. Future studies with larger sample sizes are needed to validate and generalize the observed findings.
Third, this study only included cross-sectional data. Although we used mediation analysis to infer the relationship between VRF, CBF, MD, and EF, it cannot be established whether the changes in CBF and WM integrity precede the cognitive decline. Longitudinal studies are best suited to investigate causal pathways and demonstrate the prediction of clinical outcomes. Hence, the utility of NA-PVWM rCBF as a biomarker to predict CSVD occurrence should be studied in a longitudinal cohort.
Fourth, our study lacked some radiological parameters related to CVSD. We did not have susceptibility weighted imaging needed to assess for cerebral microbleeds and we did not have 3D T2-weighted imaging needed to optimally quantify PVS volumes. Future studies may provide more insight into the associations of perfusion and diffusion metrics, particularly within PVWM, with those radiological biomarkers of CSVD.
Finally, we selected only TMT B-A as a measure of EF since other tests attributed to the EF in the UDS neuropsychological battery did not show strong correlations with imaging metrics due to a ceiling effect and small variance in this sample of CU adults. Therefore, future studies using additional measures of EF or a composite score will be needed for further validation of our findings.
Conclusions
Relative CBF in NA-PVWM is associated with vascular risk factors and reductions in executive function. Hence, NA-PVWM rCBF might provide a biomarker of early CSVD. Although the effect of VRFs on EF cannot be explained solely by CBF reduction and changes in WM microstructural integrity, increased perfusion in the PVWM region may reduce the effects of WM microstructural injury on EF, potentially slowing the progression to clinical CSVD. Additionally, concomitant AD pathology might obscure the effects of VRFs on MRI measures, making it harder to study VRFs-imaging relationships.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X251389043 – Supplemental material for Clinical correlates of perfusion and diffusion MRI metrics in cognitively unimpaired older adults
Supplemental material, sj-docx-1-jcb-10.1177_0271678X251389043 for Clinical correlates of perfusion and diffusion MRI metrics in cognitively unimpaired older adults by Banafsheh Shakibajahromi, Sudipto Dolui, Christopher A Brown, Mohammad Taghvaei, Shokufeh Sadaghiani, Pulkit Khandelwal, Sandhitsu Das, Paul A Yushkevich, Ilya M Nasrallah, Sharon X Xie, David A Wolk and John A Detre in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The authors thank LS Zhao for assistance in deriving PVS volumes from T1-weighted MRI data.
Author contributions
Banafsheh Shakibajahromi: formal analysis, methodology, investigation, data curation, writing—original draft, visualization. Sudipto Dolui: methodology, data curation, writing—review and editing. Christopher Brown: methodology, data curation, writing—review and editing. Mohammad Taghvaei: investigation, data curation. Shokufeh Sadaghiani: investigation, data curation. Pulkit Khandelwal: software, data curation. Sandhitsu Das: validation, resources, writing—review and editing. Paul Yushkevich: software, validation, resources, writing—review and editing. Ilya M Nasrallah: data curation, validation, resources, writing—review and editing, Sharon X Xie: methodology, validation, writing—review and editing. David Wolk: supervision, resources, writing—review and editing. John A Detre: conceptualization, resources, writing—review and editing, supervision, project administration, funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Health (NIH; grant numbers AG072979, NS111115, and EB031080) and the Pennsylvania Department of Health (grant number 2019NF4100087335).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DAW has served as a paid consultant to Qynapse, Beckman Coulter, and Eli Lilly. He serves on a DSMB for GSK. He also received grants from Biogen paid to his institution and received travel support from the Alzheimer’s Association.
Ethical considerations
The Penn ADRC study protocol was approved by the IRB (approval number: 068200).
Consent to participate
All study participants provided written informed consent.
Consent for publication
Not applicable.
Data availability statement
The data is available upon request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
