Abstract
We hypothesized that cerebral small vessel disease (CSVD) burden might not relevantly affect leptomeningeal collateral supply in patients with acute ischemic stroke (AIS) due to large-vessel occlusion (LVO). In n = 154 patients with anterior circulation LVO, CSVD imaging markers (white matter hyperintensities [WMH], lacunes, cerebral microbleeds and enlarged perivascular spaces) were assessed with MRI, using established criteria. Besides the extent of WMH, assessed using total Fazekas sum score, overall CSVD burden was determined with a total CSVD summary score ranging from 0–4. A quantitative and rater-independent collateral vessel index was computed from automated processing of T2*-weighted time series in perfusion-weighted imaging (PWI) to assess the pial collateral status. The overall burden of WMH and CSVD were not significantly associated with poor collaterals (adjusted odds ratios 0.830 (0.328–2.104) and 0.995 (0.666–1.488), p = 0.695 and p = 0.982) and did not modify the significant relationship of leptomeningeal collaterals with clinical stroke severity, ischemic core volume and infarct growth rate. Quantitative and objective analysis of collaterals with a signal variance-based approach in PWI revealed no overt association between CSVD burden and collaterals in LVO patients. Factors favoring or impairing collateral supply in case of acute cerebral ischemia warrant further exploration in future studies.
Introduction
In patients with acute ischemic stroke (AIS) due to large-vessel occlusion (LVO), pial collateral blood vessels (syn. leptomeningeal anastomoses) are fundamentally important as they limit the rate of progression of the ischemic tissue damage and ensure tissue viability for a certain period of time, maintaining the possibility to intervene with recanalization therapies.1–3 Furthermore, sufficiently developed collaterals guide the process of reperfusion in terms of increasing the possibility of successful recanalization and tissue reperfusion as well as decreasing the risk of complications such as reperfusion injury and hemorrhagic transformation and are a major predictor of favorable functional outcome after endovascular thrombectomy (EVT).4–6 The prognostic implications of collateral flow with regard to functional outcome remain largely unchanged despite the recently established notion that also patients with large ischemic core volumes can benefit from EVT, 7 since collateral profiles independently predict functional outcome also in this subgroup of LVO patients. 8 There are considerable interindividual differences in the capacity of pial collateral blood vessels, resulting in a relevant proportion of patients having poor collateral circulation in the case of cerebral ischemia. 9 The individual abundance, including both number and diameter, of cortical anastomoses connecting major cerebral vascular territories is the result of collaterogenesis during development and mainly genetically determined.9,10 Besides the fact that the magnitude of collateral supply is naturally more or less developed from person to person, the autoregulatory capacity of leptomeningeal blood vessels is a major determinant of collateral blood flow.9,10 In case of acute cerebral artery occlusion with a drop of perfusion pressure for antegrade blood flow to the dependent vascular territory, collateral perfusion is driven by autoregulatory vasodilation with opening of leptomeningeal anastomoses along pressure gradients.9–12 An impact or detrimental effect of cardiovascular risk factors and comorbidities on the functional capacity of collaterals may be reasonably assumed.10–12 However, the overall relationship between the degree of collateral supply and demographic and clinical variables is debated and factors associated with certain collateral profiles have not been identified consistently so far.13–15
In recent years, several studies have investigated the relationship between pial collateral supply and imaging markers of cerebral small vessel disease (CSVD).16,17 Lipohyalinosis, arteriolosclerosis and fibrinoid necrosis are considered the main elements in the pathogenesis of CSVD, leading to structural alterations of the microvascular wall, luminal narrowing and vessel occlusion with subsequent chronic cerebral hypoperfusion and ischemia. 18 Furthermore, the pathology underlying CSVD, which may not be exclusively restricted to perforating arteries of the cerebral white matter, is associated with an overall reduction of the cerebral microvascular density and impairment of autoregulatory mechanisms. 18 Since smaller pial arteries and arterioles form part of the cerebral microvasculature, CSVD is a plausible factor, which may have a detrimental impact on collaterals in terms of an impaired functional capacity or rarefication of the collateral vasculature. 9 In this regard, poor collateral supply in the context of CSVD might be either mediated by arterial hypertension, which is the most common risk factor for CSVD18,19 and associated with impaired collateral blood flow,10,11,20 or due to direct involvement of leptomeningeal collateral vessels by CSVD-related vascular pathology. Although there seems to be some evidence pointing towards an association of increasing cerebral white matter hyperintensity (WMH) burden as the main feature of CSVD with worse collateral supply, studies in the literature have so far yielded somewhat inconsistent results.16,17 Whereas the respective studies are heterogeneous regarding the employed imaging methodologies and analysis techniques, they share the application of rather approximative and rater-dependent grading scales 21 which were used to assess pial collateralization.16,17 Therefore, authors have argued for the application of more uniform and comparable imaging methodologies for collateral assessment to produce more generalizable results and overcome the current inconsistencies.22,23 Recently developed collateral assessment in magnetic resonance imaging (MRI), which is based on the analysis of signal variance in T2*-weighted time series of perfusion-weighted imaging (PWI), yields a quantitative and objective, thus observer-independent measure of collateral supply with a continuous variable.24,25 In this study, we aimed to investigate the potential influence of CSVD on pial collateralization in patients with AIS due to LVO of the anterior circulation, using quantitative mapping of collaterals with MRI based on signal variance in PWI source data.
Materials and methods
Patients
Patients with acute ischemic stroke caused by LVO of the middle cerebral artery and/or the intracranial internal carotid artery who underwent a complete standardized stroke imaging protocol with diffusion‐weighted imaging (DWI) and PWI were included in this retrospective study. Persistent vessel occlusion at the time of MRI verified on MR angiography as well as complete bolus arrival in PWI as assessable on the scanner’s workstation were required. Furthermore, diffusion- and perfusion-weighted images had to be free of major motion-related artifacts, since automated image analysis was planned. Due to a former institutional paradigm for which MRI was the imaging modality of choice at admission in certain cases (e.g. unknown symptom onset, drip-and-ship patients transferred from primary care hospitals), the local database of the Institute of Neuroradiology at Goethe University Frankfurt from the period between January 1, 2012, and October 31, 2018 was screened for the above‐mentioned inclusion criteria. In addition, including imaging examinations from the year 2018, the internal database of the Department of Neuroradiology, University of Heidelberg was screened for patients meeting our inclusion criteria. The present cohort along with the criteria applied for patient selection have been described previously in detail. 26 The study was approved by the local institutional review board (Goethe University Frankfurt, Faculty of Medicine, approval number: 400/18) and was conducted in compliance with the Declaration of Helsinki and its further amendments. Informed consent from individual patients was waived because of the retrospective character of the study.
Magnetic resonance imaging protocol
MRI data were acquired as part of the clinical routine protocol in both institutions on Siemens 3 Tesla scanners (Magnetom Verio/Skyra and Magnetom Trio and Verio; Siemens Healthcare) equipped with a body transmit and an 8‐channel phased‐array head receive coil, respectively, a 12‐channel head matrix coil. The protocol comprised DWI for diagnosis of acute ischemic stroke and dynamic susceptibility contrast (DSC)-PWI to estimate the amount of tissue at‐risk. Further parenchymal imaging was performed with additional T2- and T2*-weighted sequences. For the detection of LVO, a 3‐dimensional time‐of‐flight magnetic resonance angiography was included in all cases. Specific acquisition parameters of the respective MRI protocols have been described in detail elsewhere.24,27
Image postprocessing and analysis
Ischemic core segmentation and quantitative assessment of collateral supply
Image postprocessing and analysis were performed automatically using in-house-built shell and MATLAB scripts, implementing tools provided in the FMRIB’s Software Library toolbox (FSL, version 5.0.7). The ischemic core at admission was defined using an established and validated upper threshold of 620 × 10−6 mm2/s on apparent diffusion coefficient (ADC) maps 28 (Figure 1(a)). Each automatically segmented ADC lesion was thoroughly inspected for accuracy and manually corrected if necessary. During this procedure, the corresponding diffusion-weighted image (b = 1,000 s/mm2) was taken into consideration. The segmented infarct core was used for volumetric assessment. The individual infarct growth rate (mL/h) was determined by dividing the ischemic volume at presentation by the time from symptom onset to imaging.29–31 For automated quantitative and observer-independent assessment of collateral supply, T2*-weighted PWI time series were processed as described in detail in previous publications.24,25 In brief, after motion correction of the T2*-weighted time series, maps representing voxels with a high coefficient of variance (CoV) in the signal-time curves were computed and further processed with a correction procedure to generate maps of the pial collateral vasculature (Figure 1(b)). In terms of their anatomical correspondence to leptomeningeal anastomoses connecting major arterial territories, the collateral vessel maps had been validated against an atlas of the cerebral vasculature in stereotactic standard space 32 during the development of the method for collateral mapping. 24 This approach allowed for verifying the anatomical overlap of regions (tubular structures) of high signal variance in PWI time series along the cranial and lateral convexity with cerebral blood vessels of the pial compartment. Based on the collateral vessel map, a collateral vessel index in perfusion-weighted imaging (CVIPWI) was calculated by dividing the volumetric abundance of the leptomeningeal collaterals along the lateral and cranial convexity in the vascular territory of the middle, the anterior and the posterior cerebral artery of the affected side by that of the unaffected side.24,25 A recently established CVIPWI threshold value was used to stratify patients according to fair to good (CVIPWI ≥ 0.96) and poor (CVIPWI < 0.96) collateral supply. 24 In terms of a clinical validation, the significant associations of the CVIPWI with clinical stroke severity, ischemic core volume and functional outcome found in previous studies confirm its plausibility and validity as a measure of collateral blood flow and support its use as a parameter for outcome prediction in LVO patients undergoing EVT.24,26
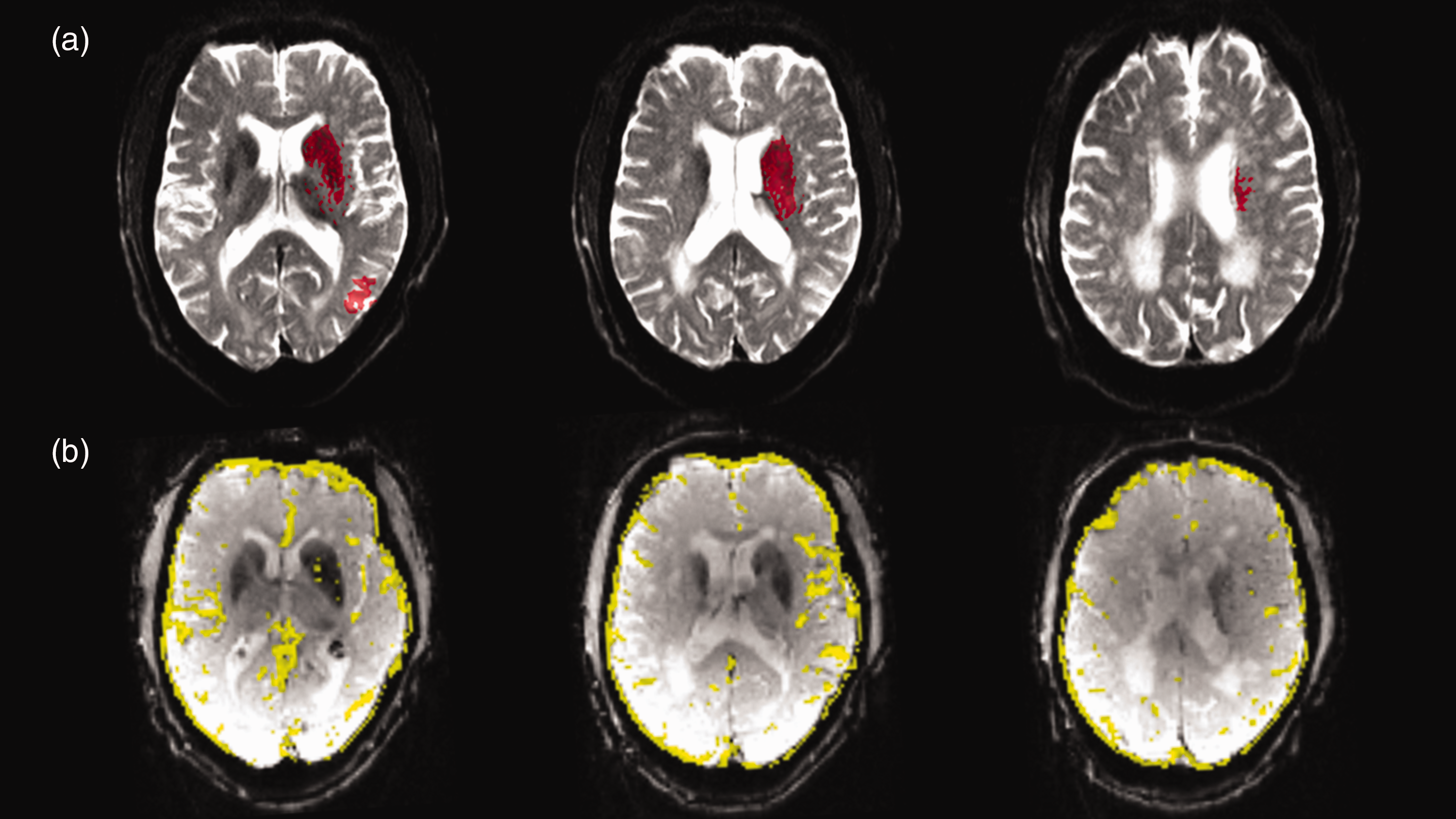
Illustration of ischemic core segmentation and collateral vessel maps a patient with left-sided MCA M1 occlusion. (a) Ischemic core obtained from automatic segmentation (red areas) overlaid on the first (b = 0) diffusion-weighted image and (b) collateral vessel map computed based on signal variance in the T2*-weighted PWI time series in yellow overlaid on the first volume of the PWI source data.
Assessment of CSVD features and severity
Markers of CSVD and their severity were assessed by two experienced investigators (one neuroradiologist and one stroke neurologist) blinded to the patients’ collateral status and clinical information, using established consensus criteria and proposed MR imaging sequences.19,33 CSVD markers consisted of WMH, chronic lacunes, enlarged perivascular perivascular spaces (EPVS) and cerebral microbleeds (CMB). Since among imaging markers of CSVD especially the extent of WMH was associated with unfavorable collateral profiles in previous studies,29,34,35 the individual WMH burden was evaluated separately from other CSVD markers in detail: grading of WMH severity was performed based on the Fazekas scale, 36 separately for the periventricular and the deep white matter. A total Fazekas sum score (ranging from 0–6) was calculated from the individual score for the periventricular and deep WMH.29,35 According to the total Fazekas score, patients were stratified into three groups of WMH severity: absent WMH (total Fazekas score 0), slight WMH (total Fazekas score 1–2) and moderate to severe WMH (total Fazekas score 3–6). 35 Furthermore, a four-item CSVD score ranging from 0–4 was determined, which represents the different CSVD types and the degree of CSVD severity.34,37 In each individual case, 1 point was given for the presence of ≥1 lacunae, 1 point for the presence of ≥1 CMB, 1 point for the presence of moderate-to-severe EPVS and 1 point for perivascular WMH with a Fazekas score of 3 or deep WMH with a Fazekas score of 2 to 3, as previously described. 34
Statistical analysis
Categorical variables were described using counts and percentages, while continuous and ordinal variables were described as mean ± SD or median and interquartile range (IQR). Proportions were calculated using χ2 statistics, whereas continuous and ordinal variables were compared using the Mann–Whitney U test or t-test, depending on the distribution of the data. Group comparisons of not normally distributed data between multiple groups were performed with the Kruskal-Wallis H test. The burden of WMH and the overall CSVD burden was compared between patients with fair – good and poor collateral supply. In order to examine the relationship between CSVD and collateral supply, multivariate binary logistic regressions were performed to calculate the crude and the adjusted odds ratios for poor collateral supply. As described previously in the literature, regressions were adjusted for age, sex and vascular risk factors including arterial hypertension, diabetes mellitus, atrial fibrillation and smoking. 26 Although arterial hypertension is a common causal factor of both CSVD and poor collateral status,18–20 regression analyses were adjusted for hypertension to ensure the comparability with previous studies in the literature.34,38 Furthermore, hypertension is associated with poor collateral supply due to structural remodeling and increased vascular tone of leptomeningeal collateral vessels through various pathophysiological mechanisms, resulting in reduced collateral perfusion.10–12 This association may be independent from the presence of lipohyalinosis as the pathological correlate of CSVD, which may also extend to smaller pial arteries and arterioles, and the presence of corresponding established imaging markers of CSVD. Statistical analyses were performed using JASP 0.18.3 (The University of Amsterdam). A p-value <0.05 was considered statistically significant.
Results
Demographic and clinical baseline characteristics
A total of 155 patients with persistent LVO at the time of MRI were included in the final analysis. Of this patient collective, n = 75 (48.4%) were female and the entire cohort had a mean age of 69 ± 15.7 years. MR imaging sequences for the assessment of CSVD were not available for one patient. The complete set of MR imaging sequences including T2*-weighted images was available for n = 105 patients, for which all items of the total CSVD summary score could be assessed. All other items of the CSVD score were assessable for n = 154 patients. In patients without available T2*-weighted images, no truncated CSVD score was used. The mean time from symptom to imaging was 7.4 hours and the major part of the patients (65.8%) presented with MCA M1 occlusion. Median (IQR) NIHSS at presentation was 13 (8–17) and the mean ischemic core volume was 28.4 ± 29.7 mL, with a mean IGR of 6 ± 9.1 mL/h. 37.4% of all patients received i.v. thrombolysis and the majority (68.4%) underwent EVT. Details on demographic and clinical baseline characteristics along with cardiovascular comorbidities are provided in Table 1.
Results of the univariate analyses after stratification of the patient collective according to collateral status.
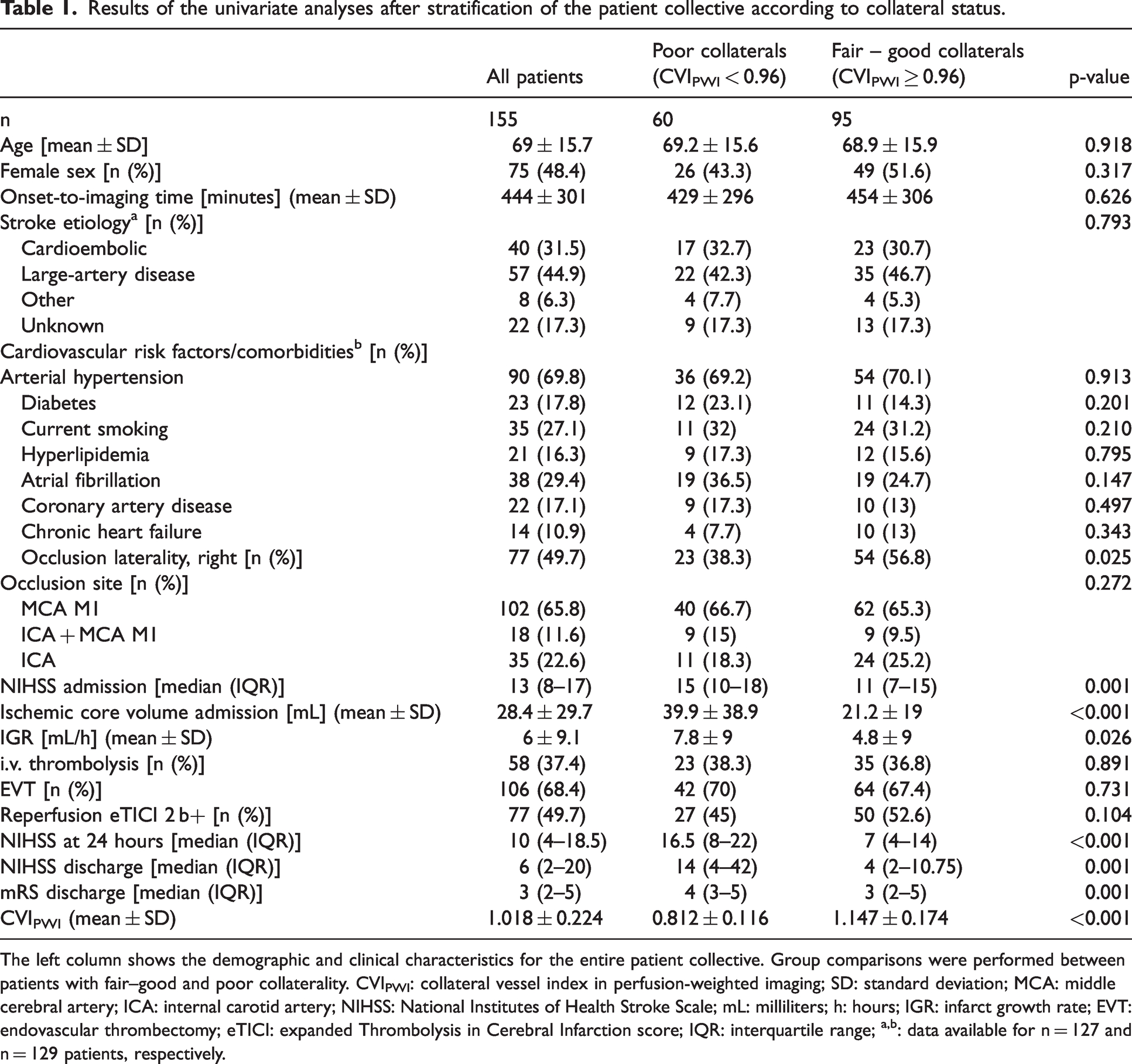
The left column shows the demographic and clinical characteristics for the entire patient collective. Group comparisons were performed between patients with fair–good and poor collaterality. CVIPWI: collateral vessel index in perfusion-weighted imaging; SD: standard deviation; MCA: middle cerebral artery; ICA: internal carotid artery; NIHSS: National Institutes of Health Stroke Scale; mL: milliliters; h: hours; IGR: infarct growth rate; EVT: endovascular thrombectomy; eTICI: expanded Thrombolysis in Cerebral Infarction score; IQR: interquartile range; a,b: data available for n = 127 and n = 129 patients, respectively.
Clinical stroke severity, infarct volumes and early functional outcome in patients with fair – good vs. patients with poor leptomeningeal collaterals
Patients with fair to good collaterals exhibited significantly lower scores on the NIHSS at presentation (p = 0.001), lower ischemic core volumes and IGR (p < 0.001, p = 0.026), lower NIHSS at 24 hours (p < 0.001) as well as lower scores on the NIHSS and mRS at discharge (p = 0.001) compared to patients with poor collateral supply. After adjustment for the covariate age, CVIPWI showed a significant negative correlation with the NIHSS on admission (r = −0.249, p = 0.002), the baseline ischemic core volume (r = −0.321, p < 0.001) and the IGR (r = −0.260, p = 0.002).
Extent of WMH and overall burden of CSVD in patients with fair – good vs. patients with poor leptomeningeal collaterals
There was no significant group difference with regard to the median Fazekas sum score (p = 0.632) between patients with fair – good and patients with poor collateral supply. Furthermore, the proportion of patients with absent WMH (Fazekas sum score 0), slight WMH (Fazekas sum score 1–2) and moderate to severe WMH (Fazekas sum score 3–6) was comparable between the groups without significant differences (Table 2). The total CSVD score as well as its four components were not differently distributed between the two groups stratified according to the degree of collateral supply (Table 2). Correlations of pial collateral supply (CVIPWI) with NIHSS at presentation, ischemic core volume and IGR did not change relevantly when additionally adjusted for the total Fazekas score (r = −0.256, p = 0.002; r = −0.323, p < 0.001 and r = −0.267, p = 0.001). These correlations could be reproduced in the subgroup of patients with available T2*-weighted images (r = 0.340, p < 0.001, r = −0.414, p < 0.001 and r = −0.380, p < 0.001), again without relevant changes after adjustment for the total CSVD score as a covariate (r = 0.340, p < 0.001, r = −0.435, p < 0.001 and r = −0.399, p < 0.001).
Comparison of CSVD imaging markers between patients with poor and patients with fair – good collateral supply.
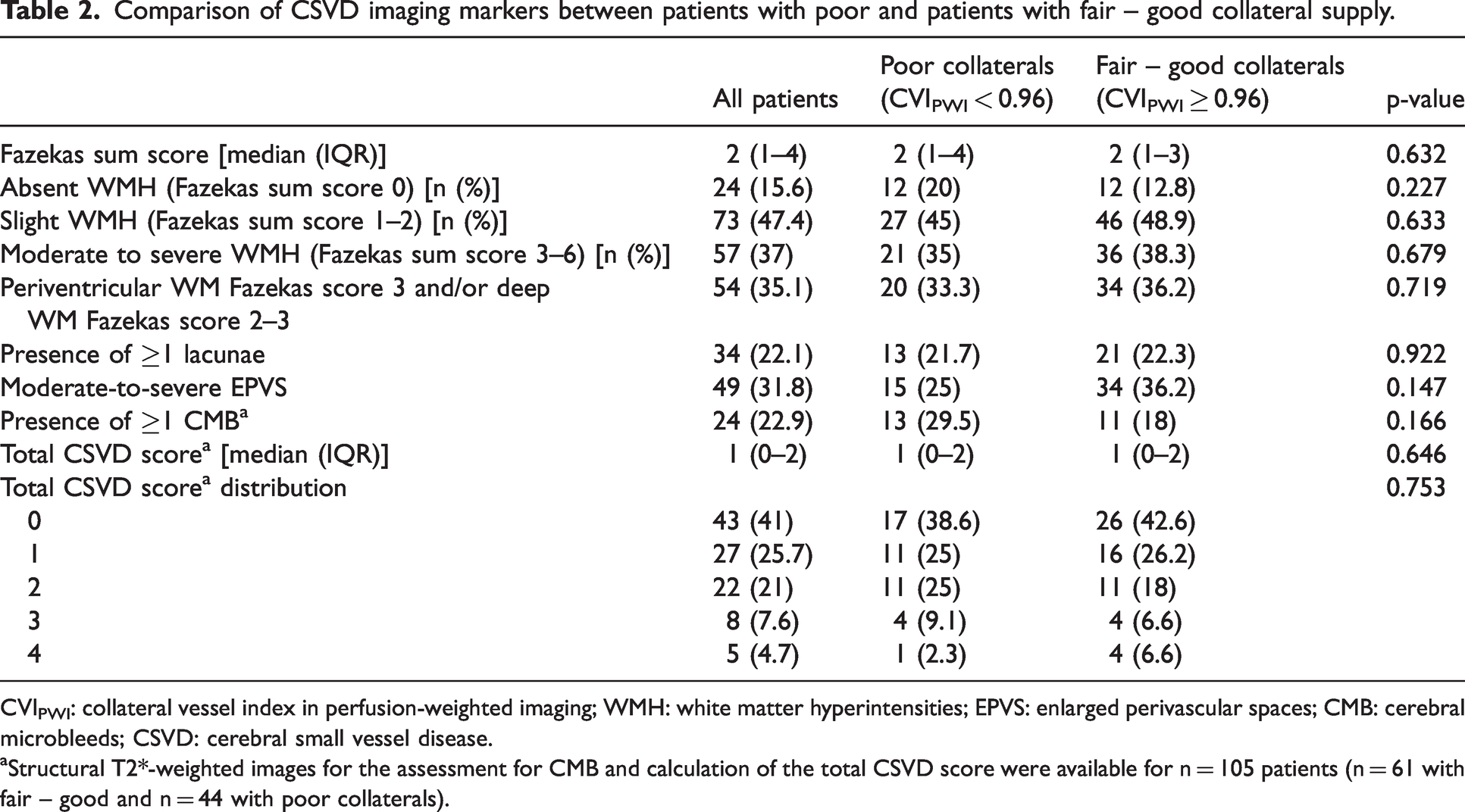
CVIPWI: collateral vessel index in perfusion-weighted imaging; WMH: white matter hyperintensities; EPVS: enlarged perivascular spaces; CMB: cerebral microbleeds; CSVD: cerebral small vessel disease.
Structural T2*-weighted images for the assessment for CMB and calculation of the total CSVD score were available for n = 105 patients (n = 61 with fair – good and n = 44 with poor collaterals).
Magnitude of collateral supply among different degrees of CSVD burden
After group stratification of the entire patient collective according to the overall burden of WMH (Fazekas sum score), there was no significant difference (p = 0.785) in the degree of leptomeningeal collateralization reflected by the CVIPWI across the three groups (Figure 2(a)). Using the Fazekas sum score (ranging from 0–6) for group stratification based on WMH severity, no significant difference in CVIPWI (p = 0.990) was found either (Figure 2(b)). Furthermore, there were no significant differences in CVIPWI across various total CSVD scores (p = 0.952) (Figure 2(c)).
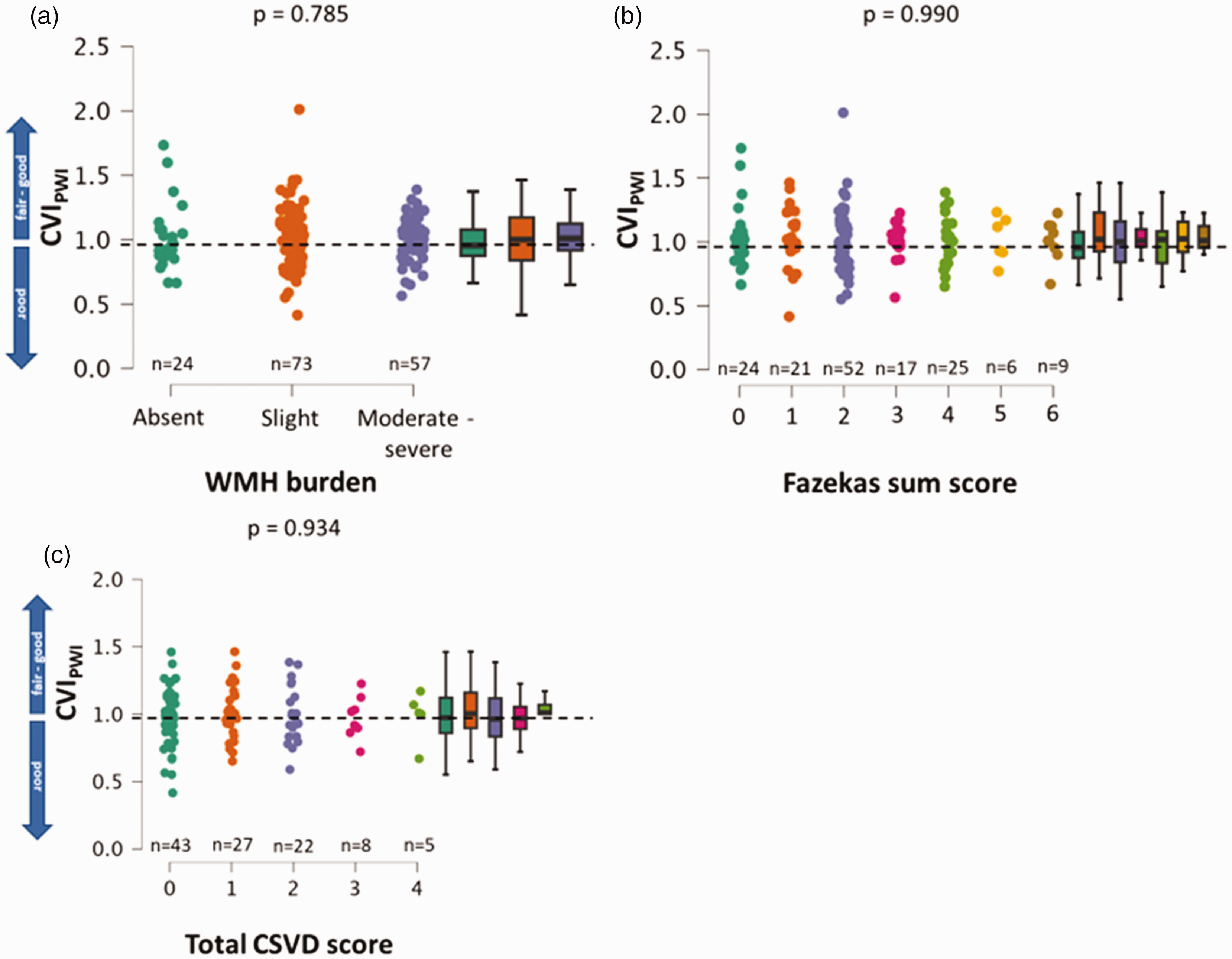
Illustration of the degree of pial collateral supply across different groups based on overall WMH burden. (a) Group stratification according to: absence of WMH (Fazekas sum score 0), slight WMH (Fazekas sum score 1–2) and moderate to severe WMH (Fazekas sum score 3–6). (b) Group stratification according to the full range of Fazekas sum scores and (c) group stratification based on the total CSVD score. The dashed horizontal lines indicate the CVIPWI threshold of 0.96 differentiating fair to good from poor collateral supply. CVIPWI: collateral vessel index in perfusion-weighted imaging; WMH: white matter hyperintensity.
Association of CSVD burden and poor collateral supply
Neither the extent of WMH (unadjusted OR for Fazekas sum score 0.958 (0.785–1.168), p = 0.668) nor the overall CSVD burden (unadjusted OR for total CSVD summary score: 1.024 (0.733–1.43), p = 0.890) were significantly associated with poor collateral supply). After adjustment for age, (female) sex, arterial hypertension, diabetes, smoking and atrial fibrillation, there was no relevant change in the odds ratios and significance levels for the association between WMH burden and poor collaterals (Table 3). Furthermore, also the different components of the total CSVD summary score did not show any significant association with poor collateral supply (Table 3).
Regression analyses yielding crude and adjusted odds ratios for poor leptomeningeal collateral supply (CVIPWI < 0.96).
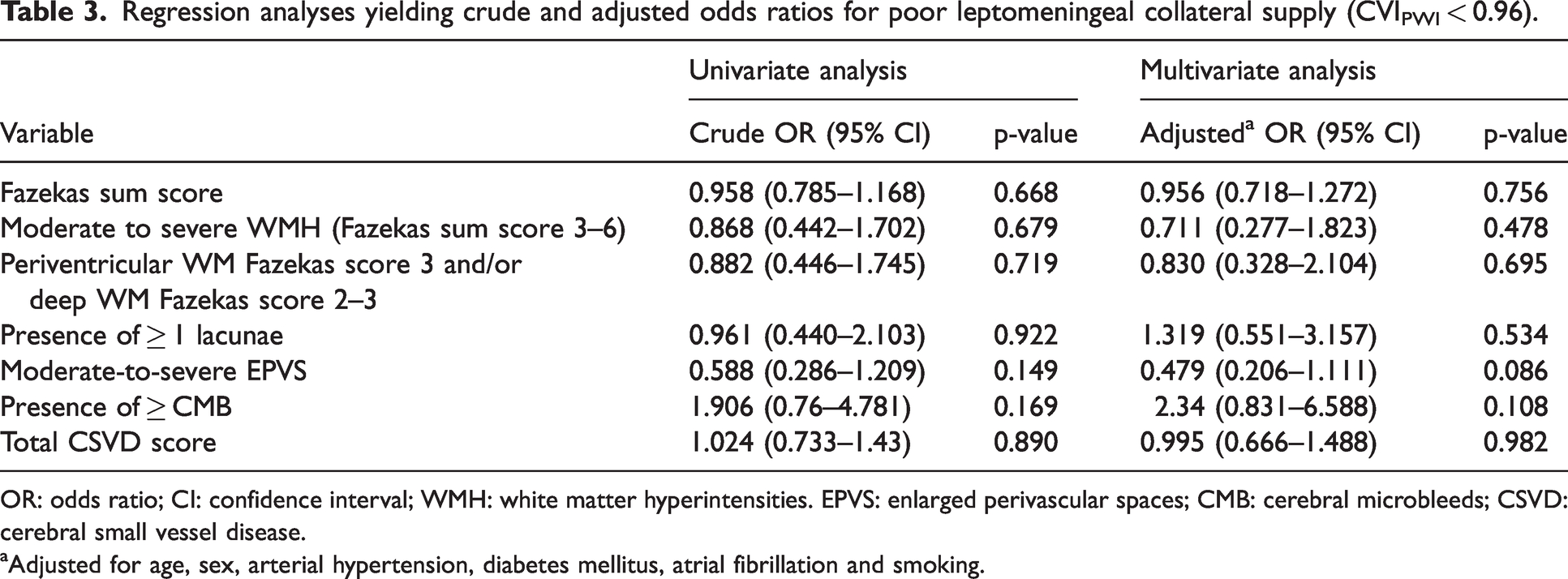
OR: odds ratio; CI: confidence interval; WMH: white matter hyperintensities. EPVS: enlarged perivascular spaces; CMB: cerebral microbleeds; CSVD: cerebral small vessel disease.
Adjusted for age, sex, arterial hypertension, diabetes mellitus, atrial fibrillation and smoking.
Discussion
In this study, we investigated the association of CSVD with pial collaterals in patients with AIS due to LVO of the anterior circulation. Based on T2*-weighted DSC-PWI source data, a collateral vessel index was used as a quantitative measure and continuous variable for the assessment of collateral supply. WMH were assessed by the means of the established Fazekas scale 36 and categorized based on the total Fazekas score, an approach comparable to the ones employed in previous research on the relationship between WMH burden and pial collaterals.29,35 The overall CSVD burden was determined by the means of a 4-item summary score ranging from 0–4 based on summing up the presence of common CSVD features on MR images34,37 We did not find any significant association between the extent of WMH or the overall CSVD burden and leptomeningeal collateral supply (Tables 2 and 3, Figure 2).
Although “recruitment” of collaterals is a widely used term in the context of collateral supply in AIS, rather than being a process of active recruitment, (retrograde) collateral perfusion in the case of acute vessel occlusion occurs passively along pressure gradients through the mechanism of autoregulatiory vasodilation.11,12,39 Although the findings are controversial, CSVD and accompanying vascular risk factors have been associated with the collateral circulation in clinical and experimental studies through an increased myogenic tone, reduced lumen diameter and an impaired vasodilatatory response of leptomeningeal anastomoses to abruptly lowered perfusion pressure in the vascular territory affected by acute vessel occusion.11,12,40 Several studies have pointed towards an association between more severe CSVD and a less favorable collateral profile in patients with AIS due to LVO of the anterior circulation.16,29,34,40 Among common CSVD features, especially cerebral leukoaraiosis as the predominant manifestation of CSVD was identified as a relevant factor associated with impaired leptomeningeal collateral supply.16,34,35 Cerebral WMH have been associated with higher ischemic core volume at presentation and greater infarct growth, both in the first hours after symptom onset and from initial imaging to follow-up after thrombectomy.29,30 Apart from associations with imaging markers related to the clinical outcome after LVO and recanalization therapies, a higher WMH burden is linked to worse early and long-term functional status.29,41,42 Apart from a reduced neuronal plasticity as the consequence of an impaired structural and functional reserve in CSVD,18,43 the described associations might be attributed to a potential impact of CSVD on the collateral circulation. A recent study, which demonstrated a significant association of more severe leukoaraiosis with poor collaterals, faster infarct growth as well as a higher likelihood of unfavorable outcome after thrombectomy, suggested that the association between more severe leukoaraiosis and a fast infarct core progression type is largely mediated by an unfavorable collateral profile. 29 In contrast, in our study the significant correlations of pial collateral supply with ischemic core volume, IGR and initial stroke severity in terms of the NIHSS score at presentation did not change relevantly after adjustment for the extent of WMH (total Fazekas score) or the overall load of CSVD-related imaging features as covariates, suggesting that none of these relationships was influenced to a notable extent by CSVD. Therefore, there seems to be no influence of CSVD on the abundance of leptomeningeal collaterals as reflected by the CVIPWI (Tables 2 and 3, Figure 2) and their functionality with regard to ensuring retrograde collateral perfusion to the region of ischemia. The significant associations of the CVIPWI with ischemic lesion volumes, clinical stroke severity and early functional outcome (Table 1) in this study support its use as a measure of leptomeningeal collateral supply with a clinical meaning, which is closely linked to the pathophysiology of cerebral ischemia. Mean ischemic core volumes of patients with poor collateral supply in this study (Table 1) were comparable to infarct core volumes found in patients identified as fast progressors in a recent analysis from the SELECT trial. 44 However, only about 15% (n = 23, data not shown in detail) of the patients in our cohort had an IGR exceeding the previously described threshold (≥10 mL/h) for differentiating fast from slow progressors.44,45 Consequently, patients with rapid early stroke progression were included in our study, but represented a minor part of the entire cohort. The formerly applied institutional paradigms regarding imaging-based patient triage for EVT in cases with unknown symptom onset, in advanced time windows and drip-and-ship patients may have led to a selection of slow progressors in our study, since the majority of patients exhibited a favorable collateral profile and relatively low IGR (Table 1). This selection of rather slow progressors may have affected our results since CSVD and cardiovascular risk factors may be less impactful in patients with an overall robust collateral profile and slower infarct progression. Thus, although we included patients with a broad spectrum of collateral profiles, it remains to be investigated in future studies whether CSVD is associated with poor collaterals and further accelerates infarct growth especially in fast or ultrafast progressors with large infarct volumes.
Taken together, the results of our study are in line with some studies in the literature,38,46,47 which is still overall heterogeneous concerning the association between CSVD and the collateral circulation, although recent meta-analyses suggest that overall recruitment of collaterals may be impaired in patients with leukoaraiosis as the main characteristic imaging finding of CSVD.16,17 The main difference of our study compared to other studies in the field is the use of an observer-independent objective and quantitative measure of collateral supply.24,26 Although several studies have used time-resolved MRI-based approaches or even digital subtraction angiography (DSA) as the gold standard for collateral imaging in AIS, their results are limited by the application of somewhat gross and rater-dependent grading systems, which in contrast to our methodological approach (Figure 2(a) to (c)) do not produce continuous variables and may not capture smaller interindividual variations in pial collateral supply. Therefore, they do not allow for appreciating the full spectrum of collateral supply in the respective patient cohorts.21,29,35,38,46 Furthermore, the PWI-based collateral imaging approach exploits signal signal variance as the criterion for segmentation of collaterals across the entire scanning time in the time series and therefore allows for detecting also delayed filling of smaller collateral vessels in the late venous phase, which may still represent sufficient collateral blood flow.24,25 Table 4 summarizes studies with the main objective to investigate the association between CSVD burden and pial collaterals in AIS patients and shows their main characteristics, including the number of investigated subjects, main results, applied imaging modalities and potential limitations. The study by Lin et al. investigated the impact of overall CSVD burden on collateral supply, using single-phase computed tomography angiography (CTA) as imaging modality for collateral assessment. 34 Among the different components of overall CSVD burden, WMH was significantly associated with poor collateral supply after adjustment for covariates. 34 However, single-phase CTA as employed in their study, largely captures retrograde collateral filling of larger arterial branches in the early phase after the contrast injection, while delayed filling of smaller pial collateral vessels in the late venous phase cannot be detected. Consequently, single-phase CTA may lead to a systematic underestimation of collateral supply.34,48 Collateral mapping with a collateral index as used in our study, which compresses the entire information on collateral filling across time by displaying the abundance of pial collateral vessels, is not time-resolved which means that collateral perfusion cannot be timely localized or related to a certain time point in the PWI source data. Therefore it is not fully comparable to tracing and grading collateral flow based on subtraction images across different time frames in PWI time series.49,50 The latter method described in the literature allows for assessing the extent and the time point of maximum collateral perfusion, as well as its relation to perfusion of the adjacent brain tissue.49,50 However, apart from completely relying on grading systems, this method is time-consuming and not feasible for the clinical setting. Furthermore, the abundance of collateral vessels segmented on the basis of signal variance may be more clinically significant as an outcome parameter compared to manual tracing of collateral filling across time series. Although it represents a newly developed imaging method, the use of the signal variance-based collateral vessel index (CVIPWI) as a measure of pial collateral supply in AIS is supported by a growing number of studies, which consistently demonstrated significant associations of CVIPWI with clinical stroke severity and baseline ischemic core volume and suggest its utility as a prognostic imaging marker for the prediction of functional outcome after EVT.24,26,51,52
Overview of cited studies investigating the association of small vessel disease burden and the degree of leptomeningeal supply in acute stroke patients.
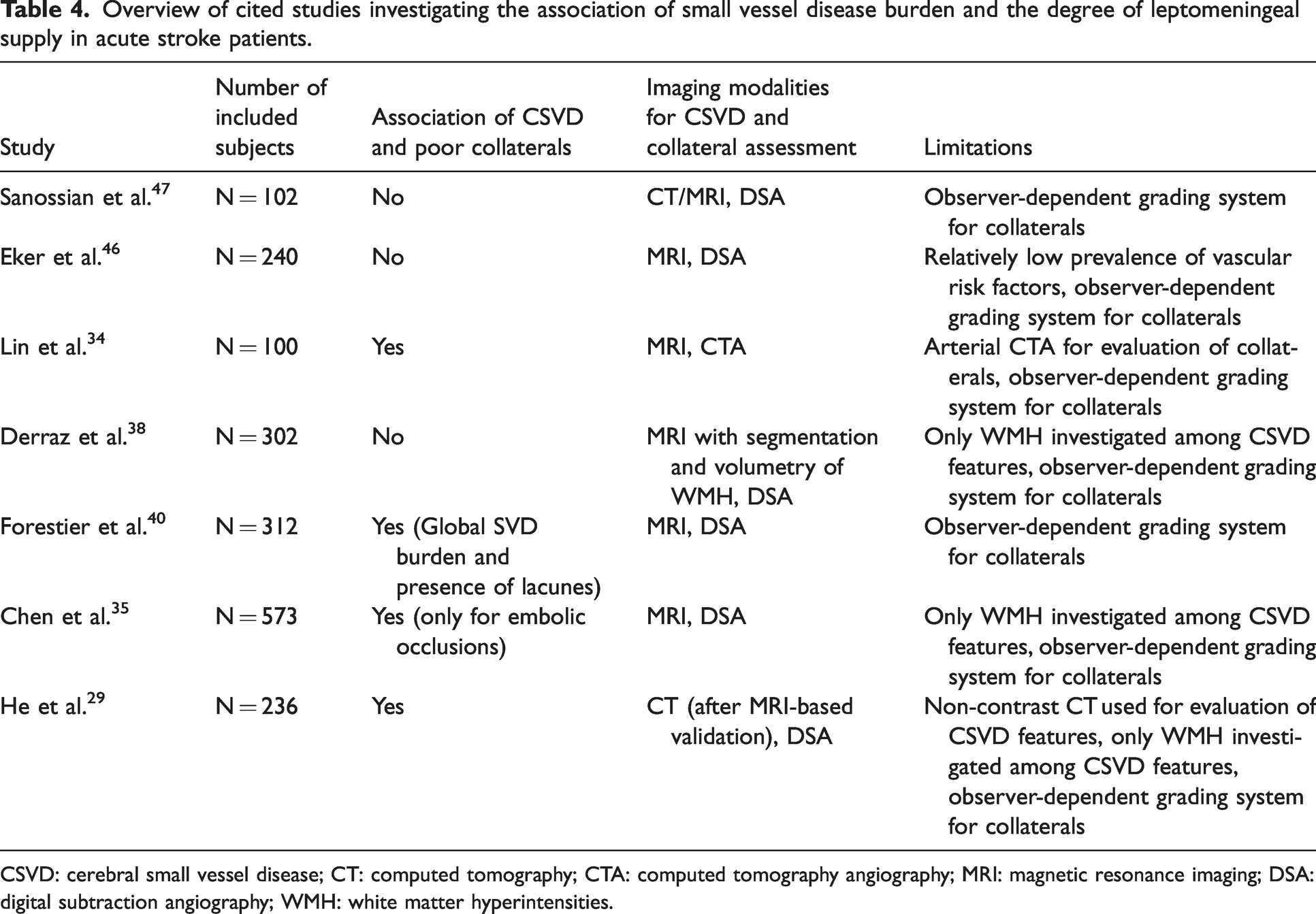
CSVD: cerebral small vessel disease; CT: computed tomography; CTA: computed tomography angiography; MRI: magnetic resonance imaging; DSA: digital subtraction angiography; WMH: white matter hyperintensities.
Notably, the major part of the studies reporting an impact of CSVD markers on leptomeningeal collateralization in AIS patients included mainly Asian cohorts and patient collectives with a relatively high prevalence of vascular risk factors such as arterial hypertension and diabetes mellitus.16,22,29,34,35,40 In our patient collective, the prevalence of arterial hypertension and diabetes mellitus was relatively high with approximately 70% (without any notable difference between patients with fair – good and poor collateral supply), respectively around 20% in the entire patient collective (the latter with a trend to a higher prevalence in the subgroup with poor collateral supply, Table 1) and comparable to the overall burden of cardiovascular risk factors and comorbidities found in similar studies which described a significant association between global CSVD measures or leukoaraiosis and leptomeningeal collateralization.34,35 Leukoaraiosis and overall CSVD severity in terms of the percentage of patients with moderate to severe WMH and high CSVD summary scores in our cohort was largely comparable to other studies.29,34,35 The majority of patients presented slight WMH, while only a small proportion exhibited no WMH. Interestingly, the latter proportion of patients was larger in the subgroup with a poor collateral profile, while the proportion of patients with a CSVD score of 4 was higher in the group with fair – good collateral supply (Table 2). Moderate to severe WMH were present in approximately 37% of all patients, with a relatively low number of patients showing severe WMH with a total Fazekas score ≥5 or high overall CSVD burden (Table 2, Figure 2(b) and (c)). However, since no trend at all was detected in terms of worse collateral supply at more severe WMH or high CSVD burden across the range of Fazekas and CSVD scores (Figure 2(a) to (c)), we do not suspect a skewing of our results towards the null as the main explanation for our negative finding. Unfortunately, data on ethnicity were not available for our patient cohort. However, given the differences in the relevance of stroke etiologies (with intracranial atherosclerosis as the most frequent etiology in Asian cohorts) as well as differences in the prevalence and phenotypes of CSVD in various patient populations, ethnicity might be a relevant factor with regard to the association between CSVD features and pial collaterals which should be systematically considered in future studies. 26
Limitations
This study has several limitations. First of all, it is a retrospective study including a relatively small selected population from two centers. Consequently, the findings of our study must be interpreted and generalized with caution, especially because our study was not powered to investigate the association between CSVD and leptomeningeal collaterals depending on different stroke etiologies. In addition, the lack of comprehensive and homogeneous follow-up imaging data with MRI prevented us from performing more detailed analysis with respect to the potential interaction between cerebral leukoaraiosis and collateral supply in the context of infarct growth after EVT. Finally, instead of enrolling a large number of subjects with rather slight WMH or low to moderate overall CSVD burden (Table 2), the inclusion of patients with more severe strokes and higher CSVD severity would have enabled a more accurate analysis of the association between the extent of CSVD and pial collaterals.
Conclusions
Quantitative analysis of collateral supply with a time-resolved imaging approach revealed no overt association between markers of CSVD and poor leptomeningeal collaterals in patients with AIS due to anterior circulation LVO. Given the heterogeneous results of prior studies in the growing body of literature on the potential influence of CSVD on pial collateralization in AIS, the actual factors favoring and impairing collateral supply in the case of acute cerebral ischemia warrant further exploration in future studies. Future research efforts in this field should be based on multicenter approaches with automated image analysis and the application of uniform objective imaging methodologies for collateral assessment. Besides the mere abundance of the pial collateral vasculature, functional properties of collateral vessels should be investigated for an association with leukoaraiosis and other manifestations of CSVD. The signal variance-based CVIPWI calculated from DSC-PWI time series as employed in this study might be a useful measure for objective rater-independent characterization of pial collateral profiles in clinical stroke studies.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Christoph Polkowski: Conceptualization of the study, literature research, collection of clinical and imaging data, data interpretation, writing.
Niklas Helwig: Conceptualization of the study, literature research, collection of clinical and imaging data, data interpretation, writing.
Fatih Seker: Collection of clinical and imaging data, interpretation, critical review of the manuscript.
Markus A. Möhlenbruch: Collection of clinical and imaging data, interpretation, critical review of the manuscript.
Marlies Wagner: Conceptualization of the study, literature research, MR image analysis, statistical analysis, writing.
Alexander Seiler: Conceptualization of the study, literature research, MR image analysis, statistical analysis, interpretation, writing.
