Abstract
The Alzheimer’s disease (AD) continuum is characterized by amyloid and tau protein deposition, which is partly attributable to the dysfunction of the brain clearance system. However, the specific phase in the AD continuum wherein aberrant clearance is present remains unclear. This study aimed to assess noninvasive magnetic resonance imaging (MRI) indices related to brain clearance functions, such as choroid plexus volume (CPV), lateral ventricular volume (LVV), and the index of diffusivity along the perivascular space (ALPS index), across the Alzheimer’s disease (AD) spectrum. The CPV, LVV, and ALPS index in amyloid beta (Aβ)-negative healthy controls (HCs) and Aβ-positive HCs as well as in patients with Aβ-negative subjective cognitive decline (SCD), with Aβ-positive SCD, with mild cognitive impairment, and with AD were evaluated. The CPV and LVV were higher, whereas the ALPS index was lower in the patients with more severe disease. The ALPS index was significantly lower in Aβ-positive HCs than in Aβ-negative HCs. In SCD patients and those in the AD continuum, the MRI-based clearance markers were correlated with P-tau and T-tau protein levels and cognitive scores. In summary, brain clearance markers on MRI are associated with tau deposition, neurodegeneration and cognitive dysfunction.
Keywords
Introduction
Alzheimer’s disease (AD), the predominant cause of dementia, typically starts asymptomatically and progresses gradually.1,2 AD is characterized by the extracellular aggregation of amyloid-beta 42 (Aβ42) peptides and neurodegeneration associated with intracellular neuroreticular wiring of the phosphorylated tau (P-tau). 3 The National Institute on Aging and Alzheimer’s Association defined AD based on the cardinal clinical manifestations of amyloidosis (A), tauopathy (T), and neurodegeneration/neuronal injury (N), determined by the presence or absence of their corresponding biomarkers.4,5 The combination of ATN biomarkers and presence of amyloidosis (A+) effectively captures pathological changes associated with the AD continuum. The AD continuum is characterized by amyloid and tau deposition, which is partly attributable to the dysfunction of the brain clearance system, including the glymphatic system (GS). 6 Nevertheless, the specific phase in the AD continuum wherein aberrant clearance occurs remains unclear. Recently, the subjective cognitive decline (SCD) was established as a prodromal phase of AD, preceding even mild cognitive impairment (MCI). SCD represents an earlier state of cognitive decline indicated by performance in cognitive tests and daily functions. 7 Since abnormal brain clearance function triggers tau and Aβ accumulation, elucidating alternations in the brain clearance system during the SCD and MCI phases may help predict the subsequent progression to dementia.
The GS hypothesis has garnered attention in recent years because its therapeutic potential of clearing brain aggregates. The cerebrospinal fluid (CSF) then interacts with intracerebral interstitial fluid and metabolic waste products. Consequently, the exchange of CSF/intracerebral interstitial fluid and the removal of waste substances such as amyloid beta (Aβ) are facilitated through a peri-venous drainage pathway. Traditionally, gadolinium-based contrast agents (GBCAs) have been used in tracer studies using magnetic resonance imaging (MRI) to evaluate glymphatic function. 8 However, GBCA administration is an invasive procedure. Noninvasive MRI techniques, devoid of tracers, have thus been developed to deduce perivascular network activity. Notably, a significant correlation was discovered between the index of diffusivity along the perivascular space (ALPS index) and glymphatic function measures, as determined using MRI following the intrathecal administration of GBCA. 9
The choroid plexus (CP) is involved in CSF production, formation of the blood-CSF barrier, and peripheral-central immune surveillance. 10 Several recent studies have shown that the choroid plexus volume (CPV) is correlated with AD and its severity. 11 CPV increases with age, whereas CSF production and permeability decrease. 12 In addition, the expansion of lateral ventricular volume (LVV) aligns with increased Aβ plaques and tau neurofibrillary tangles, 13 indicating impaired clearance against the backdrop of altered CSF-blood-brain barrier function. 14 Despite being a driving force of the GS, the relationship of decreased CSF production with GS remains unclear.
Although prior investigations on the correlation between CPV and the AD spectrum (e.g., SCD and MCI in the AD continuum), 13 yielded notable findings, comprehensive evaluations of multifaceted brain clearance functions, including the CPV, ALPS index, and LVV, have not been established (Figure 1). Furthermore, no studies have included Aβ-positive healthy individuals, and literature examining the relationship between these indicators and remanent tau protein, or clinical scores is scarce. Thus, this study aimed to investigate noninvasive MRI measures and their role in intricate intracerebral clearance functions, encompassing the GS, CP, and lateral ventricles, within the AD continuum. Importantly, we aimed to examine potential CSF biomarkers and their correlation with cognitive function.
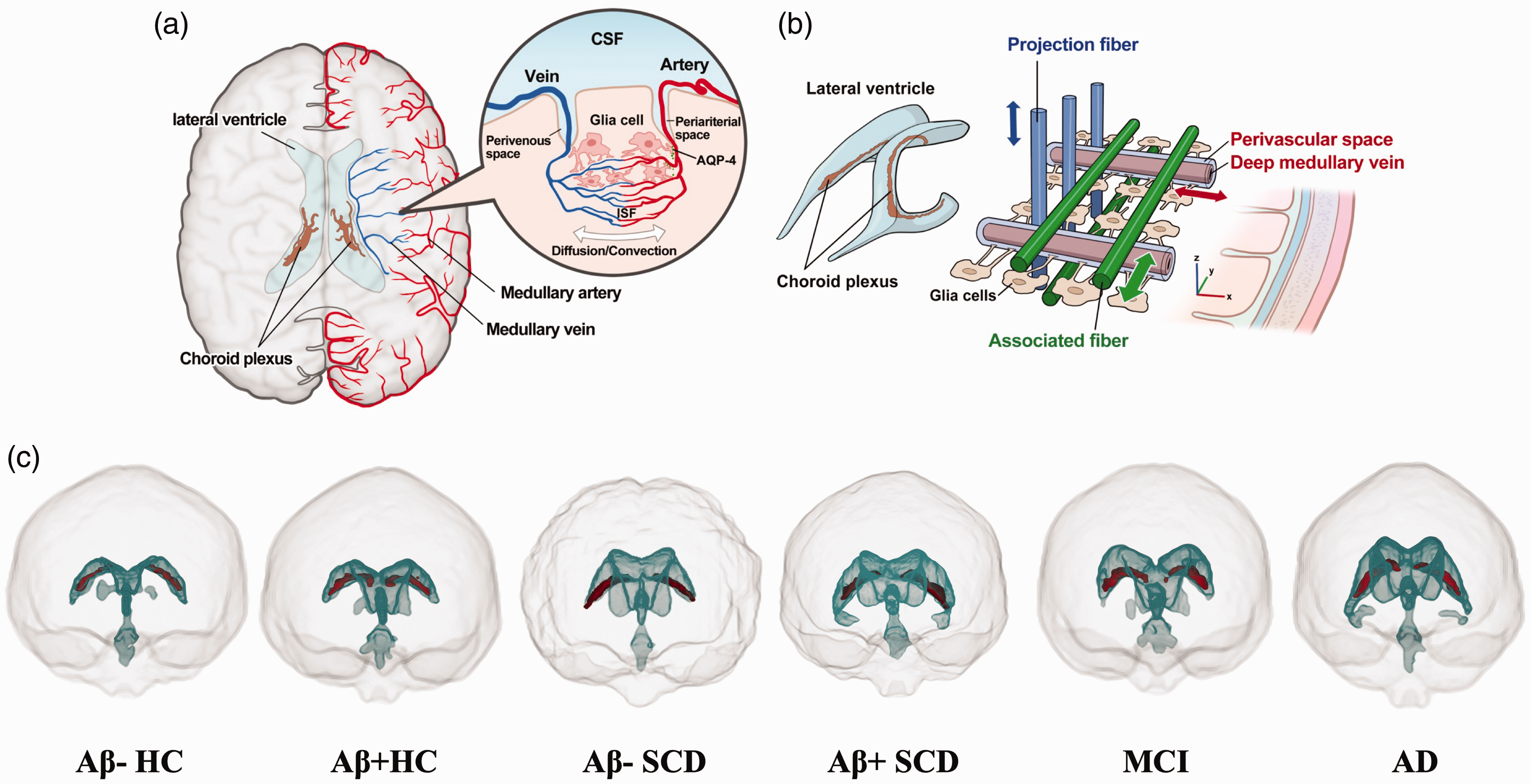
Glymphatic system and MRI indices. (a) Schematic drawing of the principle of the glymphatic system. (b) ALPS index, with the axes highlighted in appropriate color and (c) Volume of the CPV and LVV across the AD spectrum. Aβ: amyloid beta; AD: Alzheimer disease; ALPS index: index of diffusivity along the perivascular space; CPV: choroid plexus volume; CSF: cerebrospinal fluid; HC: healthy controls; LVV: lateral ventricular volume; MCI: mild cognitive impairment; SCD: subjective cognitive decline.
Materials and methods
Study design and participants
This study utilized data sourced from the ADNI-2 database (https://adni.loni.usc.edu). Aβ positivity was defined as Aβ-PET [AV45] standard uptake value ratio (SUVR) >1.11. This criterion was consistently applied across all participant groups, including HC, SCD, MCI, and AD patients. The CSF levels of Aβ42, P-tau, and total tau (T-tau) were obtained from the ADNI-2 database. The inclusion criteria, neuropsychological scores, and additional biomarkers evaluated are detailed in Supplemental Method 1.
MRI data acquisition
The MRI data acquisition is detailed in Supplemental Method 2.
MRI volumetric analysis
All T1W images were preprocessed utilizing FreeSurfer 7.1.1 (https://surfer.nmr.mgh.harvard.edu); this involved correction for subject motion, denoising, normalization of nonuniform intensity, and skull removal. The recon-all pipeline was used to derive the volumes of the CP, LV, and intracranial volume (ICV). The segmented CP and LV masks of all study participants were manually checked in a blinded manner by a neuroradiologists (M.N.) with 5 years of experience, and minimal manual correction was performed using ITK-SNAP version 3.8.0 (http://www.itksnap.org). Subsequently, the CPV fraction (CPVF = CPV/ICV) and LVV fraction (LVVF = LVV/ICV) were calculated to eliminate inter-individual variability in brain size.
ALPS index calculation
The ALPS index was computed from DW images using a semiautomated and reliable pipeline devised and validated by Taoka and Naganawa. 15 The details are provided in Supplemental Method 3.
Statistical analyses
Fisher’s exact test was used to compare the sex and APOE ε4 carriers of the groups. Significant differences in age, MRI measurements, neuropsychological scores, and CSF biomarker levels among the groups were identified using Tukey’s test following a one-way analysis of variance. Statistical analyses were conducted using two complementary approaches. A general linear model was employed to compare CPVF, LVVF, and ALPS index among groups, with age, sex, education, and white matter lesion volume fraction (WMLVF) serving as covariates. Additionally, partial Spearman’s rank correlation tests, adjusted for age, sex, education, and WMLVF, were performed to examine the relationships among MRI measurements, neuropsychological scores, and CSF biomarker levels. The volume of white matter lesions was quantified using the Lesion Segmentation Tool within SPM 12 (www.fil.ion.ucl.ac.uk), based on FLAIR imaging. This volume was then normalized by the ICV to derive the WMLVF. The effect size was subsequently calculated using Cohen’s d. Further, partial Spearman’s rank correlation tests adjusted for age, sex, education, and WMLVF were performed to examine the correlation among MRI measurements, neuropsychological scores, and CSF biomarker (Aβ, TAU, P-Tau) levels in each group, as well as in the combination of the Aβ-positive HC, SCD, MCI, and AD groups. All statistical analyses were performed using SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Statistical significance was determined using a p-value threshold <.005, and P < .05 indicated a trend. 16
Standard protocol approvals, registrations, and patient consents
Prior to commencement, this research received approval from the ethics committees at each participating institution. All ADNI studies were conducted according to the Good Clinical Practice guidelines, the Declaration of Helsinki, and U.S. 21 CFR Part 50 (Protection of Human Subjects), and Part 56 (Institutional Review Boards). Written informed consent was obtained from all participants before protocol-specific procedures were performed. The ADNI protocol was approved by the Institutional Review Boards of all participating institutions.
Results
Demographic and clinical characteristics of the study population
The study included 21 Aβ-negative HCs (10 men and 11 women; mean age, 72.28 ± 3.03 years), 10 Aβ-positive HCs (2 men and 8 women; mean age, 74.8 ± 4.03 years), 21 Aβ-negative SCD patients (9 men and 12 women; mean age, 71.51 ± 1.26 years), 9 Aβ-positive SCD patients (1 man and 8 women; mean age, 73.51 ± 1.33 years), 19 MCI patients (11 men and 8 women; mean age, 74.44 ± 0.98 years), and 26 AD patients (16 men and 20 women; mean age, 74.08 ± 1.83 years) (Table 1). There were no significant differences in sex, age, or years of education among the groups.
Clinicodemographic characteristics of the study participants.
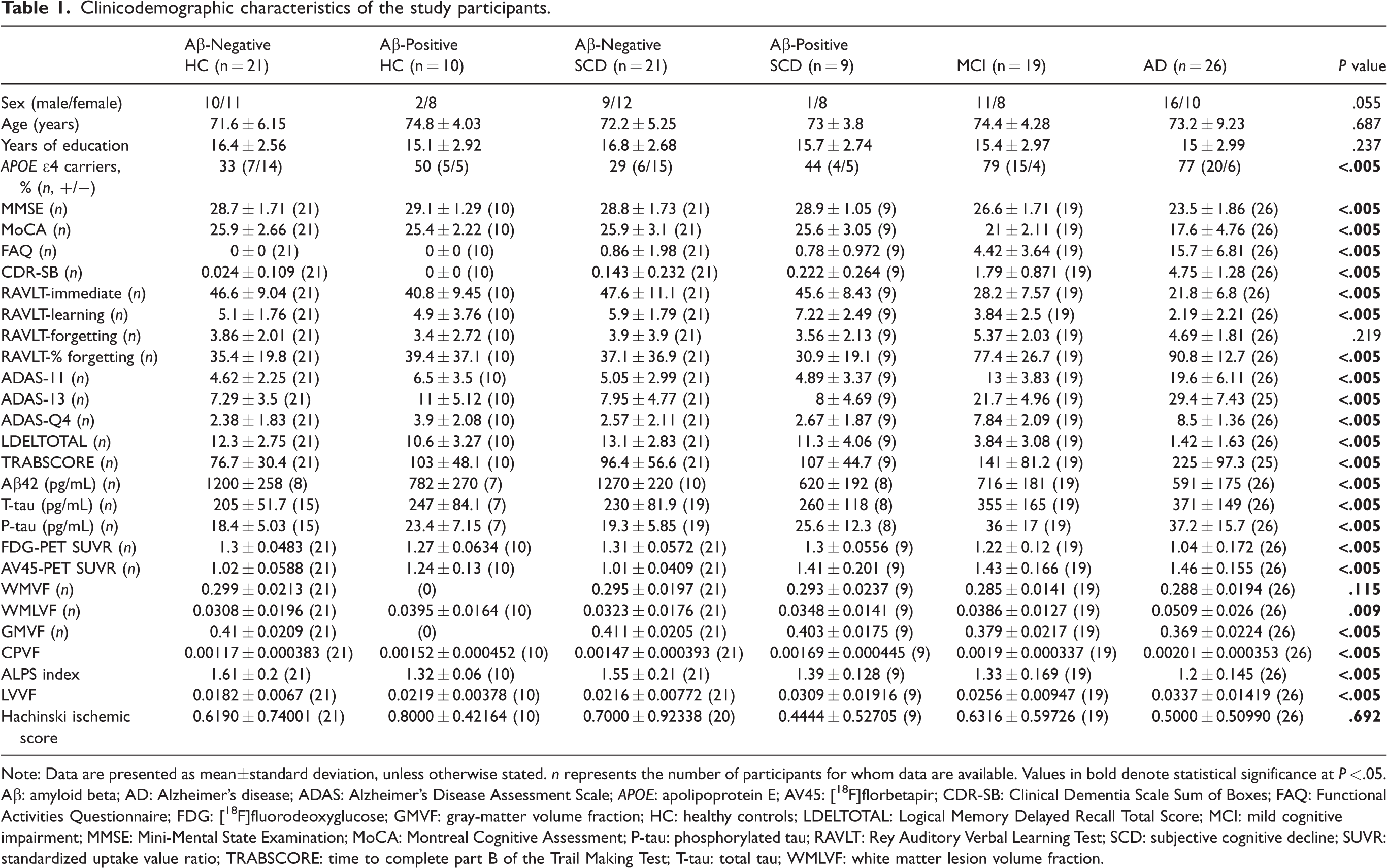
Note: Data are presented as mean±standard deviation, unless otherwise stated. n represents the number of participants for whom data are available. Values in bold denote statistical significance at P < .05.
Aβ: amyloid beta; AD: Alzheimer’s disease; ADAS: Alzheimer’s Disease Assessment Scale; APOE: apolipoprotein E; AV45: [18F]florbetapir; CDR-SB: Clinical Dementia Scale Sum of Boxes; FAQ: Functional Activities Questionnaire; FDG: [18F]fluorodeoxyglucose; GMVF: gray-matter volume fraction; HC: healthy controls; LDELTOTAL: Logical Memory Delayed Recall Total Score; MCI: mild cognitive impairment; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; P-tau: phosphorylated tau; RAVLT: Rey Auditory Verbal Learning Test; SCD: subjective cognitive decline; SUVR: standardized uptake value ratio; TRABSCORE: time to complete part B of the Trail Making Test; T-tau: total tau; WMLVF: white matter lesion volume fraction.
Among-group differences in MRI measurements
The comparison of MRI measurements between the Aβ-negative HC, Aβ-positive HC, Aβ-negative SCD, Aβ-positive SCD, MCI, and AD groups are shown in Figure 2. The CPVF was significantly higher in the Aβ-positive SCD (P = .016, d = 1.25), MCI (P < .005, d = 1.97), and AD (P < .005, d = 2.25) groups than in Aβ-negative HC group. The LVVF was also significantly higher in the AD group than in Aβ-negative HC group (P < .005, d = 1.32). No other significant differences were found, but there was a trend toward higher levels in the Aβ-negative HC, Aβ-positive SCD, and MCI groups.
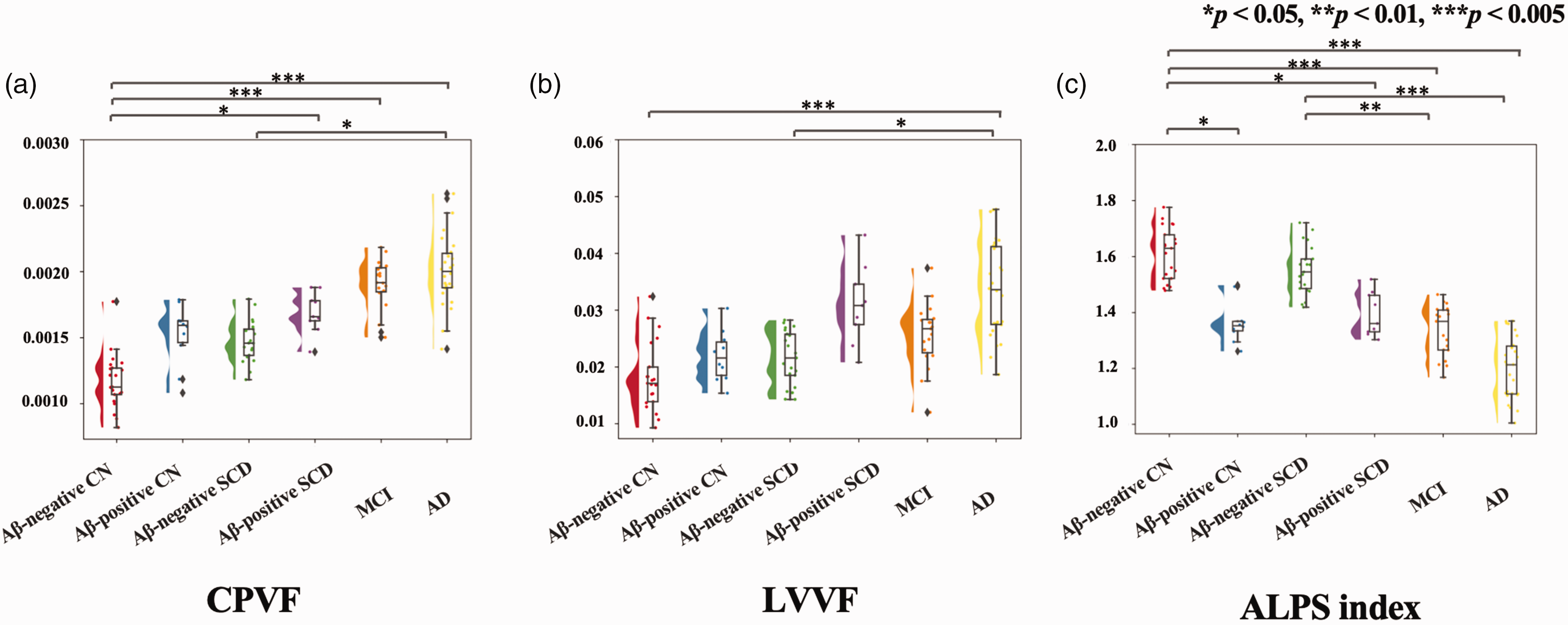
Between-group differences in magnetic resonance imaging measures. Violin-and-box plots of the CPVF (a), LVVF (b), and ALPS index (c) among Aβ- and Aβ-positive HCs, Aβ-positive and Aβ-negative participants with SCD, participants with MCI, and participants with AD. P values correspond to the general linear model analysis. Aβ: amyloid beta; AD: Alzheimer disease; ALPS index: index of diffusivity along the perivascular space; CPVF: choroid plexus volume fraction; LVVF: lateral ventricular volume fraction; MCI: mild cognitive impairment; SCD: subjective cognitive decline.
Regarding the ALPS index, it was significantly lower in the Aβ-positive SCD (P = .025, d = 1.17), MCI (P < .005, d = 1.46), and AD (P < .005, d = 2.35) groups than in the Aβ-negative HC group. It was also significantly lower in the MCI (P = .002, d = 1.13) and AD (P < .005, d = 1.97) groups than in the Aβ-negative SCD group. Furthermore, the ALPS index was significantly different between the Aβ-positive and Aβ-negative HC groups (P = .01, d = 1.42).
Correlation analyses
The results of the correlation analysis are summarized in Table 2, Figure 3, and Supplemental Figure 1. In the Aβ-positive SCD group, CPVF and LVVF showed the trend toward a correlation with T-tau (P = .046, rs = −0.954; P = .032, rs = −0.968) and P-tau (P = .046, rs = −0.954; P = .032, rs = −0.968). Furthermore, in the AD continuum, CPVF was correlated with the MoCA (P < .005, rs = −0.512) and CDR-SB (P < .005, rs = 0.438) results, LVVF was correlated with cognitive indicators such as the MMSE (P = .008, rs = −0.328) and MoCA (P < .005, rs = −0.348) results, and the ALPS index was correlated with the MMSE (P < .005, rs = 0.493) and CDR-SB (P < .005, rs = −0.526) results. Regarding the correlation among MRI measurements, there was a positive correlation between CPVF and LVVF (P < .005, rs = 0.507), whereas there was a negative correlation between the ALPS index and CPVF (P < .005, rs = −0.430) and between the ALPS index and LVVF (P < .005, rs = −0.507) in the AD continuum (Supplemental Figure 2).
Relationship between MRI measurements and clinical scores.
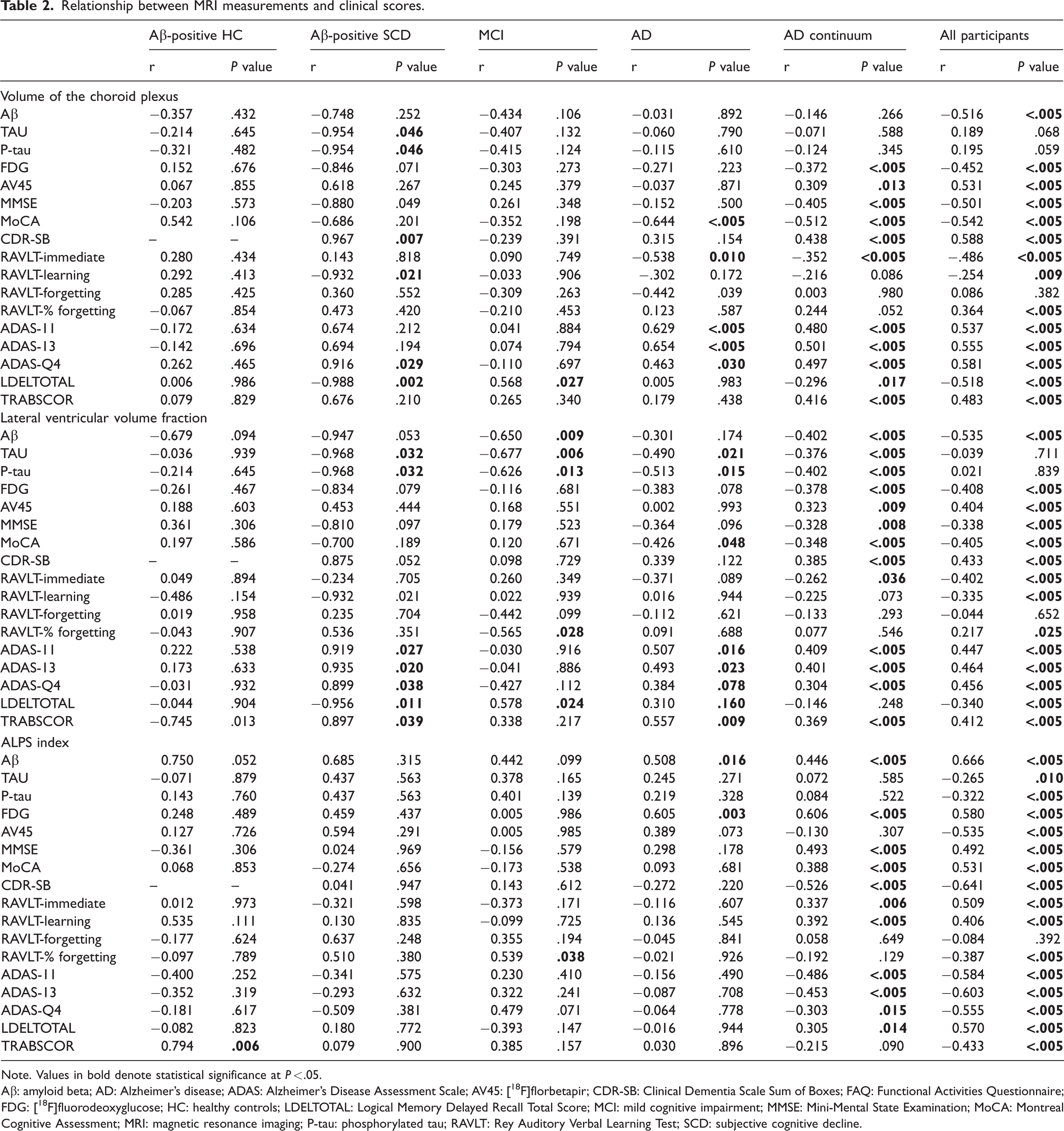
Note. Values in bold denote statistical significance at P < .05.
Aβ: amyloid beta; AD: Alzheimer’s disease; ADAS: Alzheimer’s Disease Assessment Scale; AV45: [18F]florbetapir; CDR-SB: Clinical Dementia Scale Sum of Boxes; FAQ: Functional Activities Questionnaire; FDG: [18F]fluorodeoxyglucose; HC: healthy controls; LDELTOTAL: Logical Memory Delayed Recall Total Score; MCI: mild cognitive impairment; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; MRI: magnetic resonance imaging; P-tau: phosphorylated tau; RAVLT: Rey Auditory Verbal Learning Test; SCD: subjective cognitive decline.
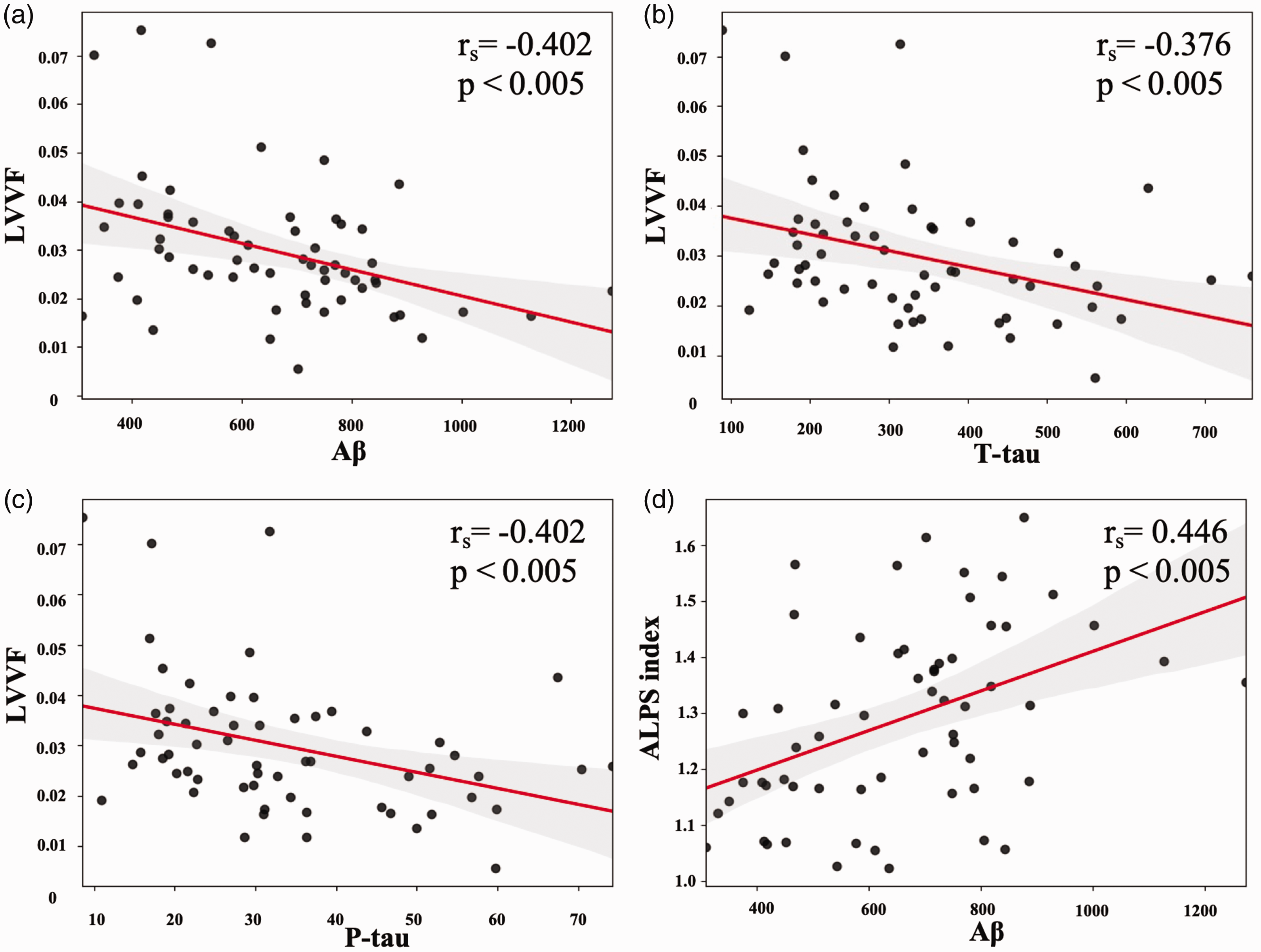
Relationship between magnetic resonance imaging and cerebrospinal fluid markers in the AD continuum demonstrated in dot plot diagrams. The LVVF is negatively correlated with Aβ (a), T-tau (b), and P-tau (c), whereas the ALPS index is positively correlated with Aβ (d). Aβ: amyloid beta; AD: Alzheimer disease; ALPS index: index of diffusivity along the perivascular space; LVVF: lateral ventricular volume fraction; P-tau: phosphorylated tau; T-tau: total tau.
Discussion
Despite prior investigations into the correlation between CPV and the AD spectrum, multifaceted brain clearance functions are not fully understood. This study found that the CPVF was significantly higher in Aβ-positive SCD, MCI, and AD patients than in Aβ-negative HCs, and the effect size was increased with the disease stage. For LVVF, it was significantly higher in AD patients than in Aβ-negative HCs, and the effect size was also increased with disease stage severity. The ALPS index was significantly lower in Aβ-positive HCs, Aβ-positive SCD patients, MCI patients, and AD patients than in Aβ-negative HCs, and the effect size was increased with the disease severity. Notably, the ALPS index was significantly lower in Aβ-positive HCs than in Aβ-negative HCs. There was also a trend of CPVF and LVVF correlating with P-tau and T-tau in Aβ-positive SCD patients. Furthermore, in the AD continuum, CPVF was correlated with the MoCA and CDR results, LVVF with cognitive indicators such as MMSE and MoCA results, and the ALPS index with the MMSE and CDR results. The correlation of MRI clearance markers with P-tau (tau deposition), T-tau (neurodegeneration), and cognitive score (clinical cognitive dysfunction) in SCD patients preliminarily suggests that impaired clearance is associated with tau deposition, neurodegeneration, and cognitive dysfunction. Collectively, these results indicate that in the absence of Aβ accumulation, clearance function is normal, but the clearance mechanism may already be impaired in SCD, a preclinical manifestation of AD.
The CPVF was significantly higher in Aβ-positive SCD patients than in Aβ-negative HCs in this study. These findings are consistent with previous reports showing significantly greater CPVF in AD patients than in both HCs and MCI patients.11,17 Meanwhile, in contrast to our study’s findings, Tadayon et al. 17 reported no significant variations in CPVF between SCD patients and HCs. This apparent discrepancy might be attributed to the fact that the SCD patients were further divided into the Aβ-positive or Aβ-negative subgroups in our study. This subcategorization may have increased the sensitivity to detect differences in CPVF. Group comparisons suggested that CPVF clearance had already started in SCD.
The alteration in CPVF, which is considered an indirect indicator of glymphatic clearance, 18 may be particularly pronounced in SCD, concurrent with AD pathology. The observed increase in CPVF in patients with AD-related cognitive impairment can be attributed to interstitial fibrosis, interstitial calcification, vascular thickening, or inflammation, ultimately compromising CSF production and clearance. 19 Notably, a trend toward a correlation between CPV and CSF clearance has been found in the healthy older individuals. 19 Inflammatory cells, particularly the microglia, play an important role in the clearance of pathological proteins in AD, 20 and P-tau has been linked to these processes. 21 The significant correlation between CPVF and P-tau in this study suggests a potential relationship between tau clearance and CPVF. Additionally, T-tau reflects the extent of neuronal injury and neurodegeneration. 22 The correlation between CPVF and T-tau in our study further implies a link between CPVF and neuronal cell injury. Moreover, previous studies have identified correlations between the CPV and cognitive functions assessed by the MMSE for patients in the AD spectrum. 11 The current study also found a correlation between CPVF and neuropsychological testing, suggesting that CPVF might be associated with clinical symptoms. CPVF was correlated not only with P-tau, but also with T-tau, a marker of neuronal cell injury, and with the cognitive function scale. These findings suggested that impaired CPVF clearance was also closely associated with neuronal cell injury and cognitive impairment in AD.
LVVF was significantly higher in patients with AD than in HCs in this study. This association of LVV with AD severity is consistent with previous findings. 23 LVV enlargement in AD is an indicator of disturbed CSF dynamics and signifies impaired CSF clearance mechanisms. 24 Consequently, the observed changes in LVVF, representing brain clearance indicators, suggest that alterations in CSF dynamics may already manifest in the presence of AD. LVVF was significantly correlated with T-tau and P-tau proteins in Aβ-positive SCD, MCI, and AD patients; furthermore, it was correlated with Aβ levels in the AD continuum. The correlations between LVV and cognitive evaluation scores, such as the MMSE and ADAS scores, further suggest that LVV clearance may be closely linked to neuronal and cognitive impairments in AD, similar to CPVF clearance observed in our study. However, additional comprehensive studies using larger sample sizes and advanced imaging techniques are necessary to further investigate the relationship between CSF production and clearance.
In this study, the ALPS index was significantly lower in patients with Aβ-positive SCD, MCI, and AD than in Aβ-negative HCs. This aligns with a previous finding of lower ALPS indices in patients with MCI and AD than in HCs.25,26 Another study showed that the ALPS index could accurately measure GS activity. 27 In addition, although the difference was not significant, the ALPS index was lower in Aβ-positive HCs than in Aβ-negative SCD patients, suggesting reduced clearance in individuals with Aβ accumulation. Furthermore, the ALPS index was significantly lower in Aβ-positive HCs than in Aβ-negative HCs. These outcomes suggest that in the absence of Aβ accumulation, clearance function remains normal. The ALPS index exhibited a positive and significant correlation with CSF Aβ within the AD continuum. This correlation is consistent with a previous report suggesting perivascular accumulation of Aβ as a cause of impaired perivascular clearance. 26 In our study, T-tau and P-tau were negatively correlated in all participants. A previous report, using the ALPS index and tau positron emission tomography, reported that the ALPS index may reflect cognitive dysfunction related to amyloid and tau deposition, 28 and consistent findings were observed in the current study.
In addition, the ALPS index showed significant correlations with various cognitive measures in the AD continuum, suggesting its potential as an early AD screening indicator.25,29 To the best of our knowledge, this study is the first to compare Aβ-positive HCs and SCD to Aβ-negative HCs. Our results support that in SCD, alterations in the ALPS index may already occur, with the presence of AD pathology.
In AD, various structural and functional alterations occur in the brain, including changes in CSF dynamics and barrier integrity.30,31 The presence of toxic proteins such as Aβ and tau around the ventricular regions suggests abnormal clearance mechanisms32,33 which may explain the positive correlations between CPVF and LVVF with T-tau, P-tau, and cognitive scores in our study. The ALPS index’s correlation with glucose metabolism 26 and its role in mediating Aβ and tau deposition28,34 indicate that impaired glymphatic function and neuronal degeneration contribute to cognitive decline in AD.
This study has some limitations. First, a longitudinal design is essential for evaluating the evolution of CPV and the angioarchitectural index of perivascular spaces in patients with subcortical dementia that progressed to MCI. The relatively small sample size and imbalance in participant numbers among the groups highlight the need for larger studies. Additionally, the angioarchitectural index of perivascular spaces only reflects the events in regional area (corona radiata) and does not account for the diffusion in perivascular spaces. Finally, histopathological validation was not conducted. Despite these limitations, this study provides insights into the relationship between brain waste removal and cognitive decline. Furthermore, our study may also provide new research directions for investigating brain structures and regions associated with AD. Future research with longitudinal designs and larger sample sizes is imperative to validate these findings.
In summary, brain clearance markers on MRI are correlated with P-tau, T-tau, and cognitive function scores, implying that impaired brain clearance is associated with tau deposition, neurodegeneration, and cognitive dysfunction. Changes in MRI measures related to brain clearance function can be detected in early SCD associated with AD pathology.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251321305 - Supplemental material for Magnetic resonance imaging indices for early Alzheimer’s disease detection: Brain clearance markers
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251321305 for Magnetic resonance imaging indices for early Alzheimer’s disease detection: Brain clearance markers by Moto Nakaya, Koji Kamagata, Kaito Takabayashi, Christina Andica, Wataru Uchida, Akifumi Hagiwara, Toshiaki Akashi, Akihiko Wada, Toshiaki Taoka, Shinji Naganawa, Osamu Abe and Shigeki Aoki in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X251321305 - Supplemental material for Magnetic resonance imaging indices for early Alzheimer’s disease detection: Brain clearance markers
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X251321305 for Magnetic resonance imaging indices for early Alzheimer’s disease detection: Brain clearance markers by Moto Nakaya, Koji Kamagata, Kaito Takabayashi, Christina Andica, Wataru Uchida, Akifumi Hagiwara, Toshiaki Akashi, Akihiko Wada, Toshiaki Taoka, Shinji Naganawa, Osamu Abe and Shigeki Aoki in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-3-jcb-10.1177_0271678X251321305 - Supplemental material for Magnetic resonance imaging indices for early Alzheimer’s disease detection: Brain clearance markers
Supplemental material, sj-pdf-3-jcb-10.1177_0271678X251321305 for Magnetic resonance imaging indices for early Alzheimer’s disease detection: Brain clearance markers by Moto Nakaya, Koji Kamagata, Kaito Takabayashi, Christina Andica, Wataru Uchida, Akifumi Hagiwara, Toshiaki Akashi, Akihiko Wada, Toshiaki Taoka, Shinji Naganawa, Osamu Abe and Shigeki Aoki in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Data availability
The data used in this study are publicly available in the ADNI-2 database (adni.loni.usc.edu). A list of the participants included in this study is available on request from the corresponding author. The data supporting the findings of this study are available on request from the corresponding author (K.K.) who has full access to the data used in the analyses in the manuscript. Raw data were generated at the Juntendo University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly supported by the Juntendo Research Branding Project, the Japan Society for the Promotion of Science Grants-in-Aid for Scientific Research (KAKENHI; grant numbers 23H02865, 23K14927 and 23K27556), a Grant-in-Aid for Special Research in Subsidies for Ordinary Expenses of Private Schools from the Promotion and Mutual Aid Corporation for Private Schools of Japan, and the Brain/MINDS Beyond Program of the Japan Agency for Medical Research and Development (AMED) under grant numbers JP18dm0307004 and JP19dm0307101 and AMED under grant number JP21wm0425006 and JP24wm0625310. This work was supported by JST FOREST Program, Grant Number JPMJFR231P.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
All authors made substantial contributions to the study concept, data analysis, and interpretation; M.N. and K.K. K.T. drafted the manuscript or revised it critically for important intellectual content; C.A., U.W., A.H., T.A., A.W., T.T., S.N., O.A. approved the final version of the manuscript to be published; and S.A. agreed to be accountable for all aspects of the work.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
