Abstract
Intracranial cardiac impulse propagation along penetrating arterioles is vital for both nutrient supply via blood circulation and waste clearance via CSF circulation. However, current neuroimaging methods are limited to simultaneously detecting impulse propagation at pial arteries, arterioles, and between them. We hypothesized that this propagation could be detected via paravascular CSF dynamics and that it may change with aging. Using dynamic diffusion-weighted imaging (dynDWI), we detected oscillatory CSF motion synchronized with the finger photoplethysmography in the subarachnoid space (SAS) and cerebral cortex, with a delay revealing an impulse propagation pathway from the SAS to the cortex, averaging 84 milliseconds. Data from 70 subjects aged 18 to 85 years showed a bimodal age-related change in the SAS-Cortex travel time: it initially increases with age, peaks around 45 years, then decreases. Computational biomechanical modeling of the cardiovascular system was performed and replicated this 84-millisecond delay. Sensitivity analysis suggests that age-related variations in travel time are primarily driven by changes in arteriolar compliance. These findings support the use of dynDWI for measuring intracranial impulse propagation and highlight its potential in assessing related vascular and waste clearance functions.
Keywords
Introduction
Each heartbeat generates an aortic pressure impulse that propagates through cerebral vascular trees, spanning from surface pial arteries to penetrating arterioles that dive into the gray matter, and further to microcirculation networks. Acting as resistance vessels, penetrating arterioles play a crucial role in dampening the incoming pulse pressure to protect delicate capillaries and maintain the integrity of the blood-brain barrier, ensuring a consistent flow within the microcirculation. Moreover, the paravascular space surrounding penetrating arterioles, known as the Virchow-Robin space, serves as a conduit for cerebrospinal fluid (CSF) entry into the brain tissue, facilitating crucial waste clearance mechanisms.1 –3 The pulsatile force exerted on these elastic arteries plays a role in driving paravascular CSF flow, aiding in the removal of brain waste products.4 –6 Despite its potential significance, research on impulse propagation from pial arteries to penetrating arterioles is scarce due to the lack of assessment tools. Current methods for studying intracranial impulses primarily rely on detecting blood flow, focusing on either pial arteries7 –10 or penetrating arterioles.11 –13 The challenge of simultaneously detecting both segments lies in the large differences in their flow velocities (20 cm/s in pial arteries vs. 1 cm/s in arterioles) and the small dimensions of penetrating arterioles (diameters below 200 μm), necessitating sub-millimeter imaging resolutions.
We hypothesize that the elastic expansion of pial arteries and penetrating arterioles can be detected via the movement of paravascular CSF. This hypothesis stems from emerging research on the coupling between vascular and paravascular fluid dynamics. Preclinical investigations employing two-photon microscopy and biomechanical modeling studies have consistently shown pulsation-driven paravascular CSF dynamics that oscillates in synchrony with pressure changes within the vessel.4,6,14,15 Recent human studies have corroborated this coupling in major and pial artery segments.16 –21
Thus, it is plausible that this coupling extends to smaller downstream arteriolar segments. We propose using dynamic diffusion-weighted imaging (dynDWI) to detect fluid dynamics along this pathway, manifesting as oscillatory incoherent motion driven by cardiac pulses. Diffusion-weighted imaging can measure incoherent paravascular CSF motion without resolving individual paravascular spaces, similar to how it measures axonal water motion without resolving individual axons. The CSF fluid motion within the Virchow-Robin space provides a unique opportunity to evaluate impulse propagation from pial arteries to arterioles in a way not possible in other organs.
Detecting impulse propagation along this short distance requires high temporal resolution to resolve time delays at the millisecond scale. To achieve this, we introduced a signal processing approach to enhance the resolvable time delay of dynDWI, achieving a temporal resolution of 2.5 milliseconds (msec). This enhancement was achieved by leveraging the high temporal resolution of the simultaneously acquired finger photoplethysmography signal and natural variations in heart rate. Through this approach, we demonstrated dynDWI’s ability to map impulse arrival time at both pial arteries and penetrating arterioles, as well as the propagation time between these segments. Subsequently, we applied this technique to a cohort aged 18 to 85 years to investigate age-related changes. Finally, we conducted biomechanical modeling to validate imaging findings and gain insights into the potential biomechanical factors contributing to alterations in impulse propagation speed in the aging brain.
Material and methods
Cross-correlation analysis for tracking impulse arrival times within the brain
To capture paravascular CSF dynamics, dynDWI with temporal repeats was collected for 5-minutes at a repetition time of 1.78 seconds. A b-value of 150 s/mm2 was applied to provide sensitivity to the incoherent motion of CSF while suppressing the signal from rapid blood flow.18,19 The diffusion gradient method has established efficacy in suppressing the fast-flowing blood signals within various segments, including major arteries,22,23 penetrating arterioles, and capillary networks.24 –27 Consequently, temporal variations in dynDWI signal primarily reflect the incoherent motion of paravascular fluid in response to pressure changes of the vessel wall.
Figure 1 provides an overview of the signal processing approach developed to extract the impulse arrival time. The dyn DWI signal was first converted to the apparent diffusion coefficient (ADC), with higher values indicating faster incoherent water motion. Cross-correlation between the dynamic ADC and the finger photoplethysmography (PPG) signal determined the impulse arrival time at brain voxels relative to the finger (Figure 1(a)). In cross-correlation, the finger PPG served as the reference, and dynamic ADC signals were shifted in 2.5 ms steps. At each shift, PPG was undersampled to match the dynDWI time points to calculate the cross-correlation coefficient, achieving a temporal resolution of 2.5 ms for impulse arrival time. The cross-correlation curve in Figure 1(b) shows that the brain impulse arrival precedes the finger impulse. The positive correlation coefficient was used to determine the arrival time because the CSF waveform and finger PPG share the same polarity at the systolic peak. This is based on our observations that the CSF waveform’s systolic peak closely resembles the pressure waveform 18 and that its peak timing aligns with the expansion of the arterial vessel wall.4,17
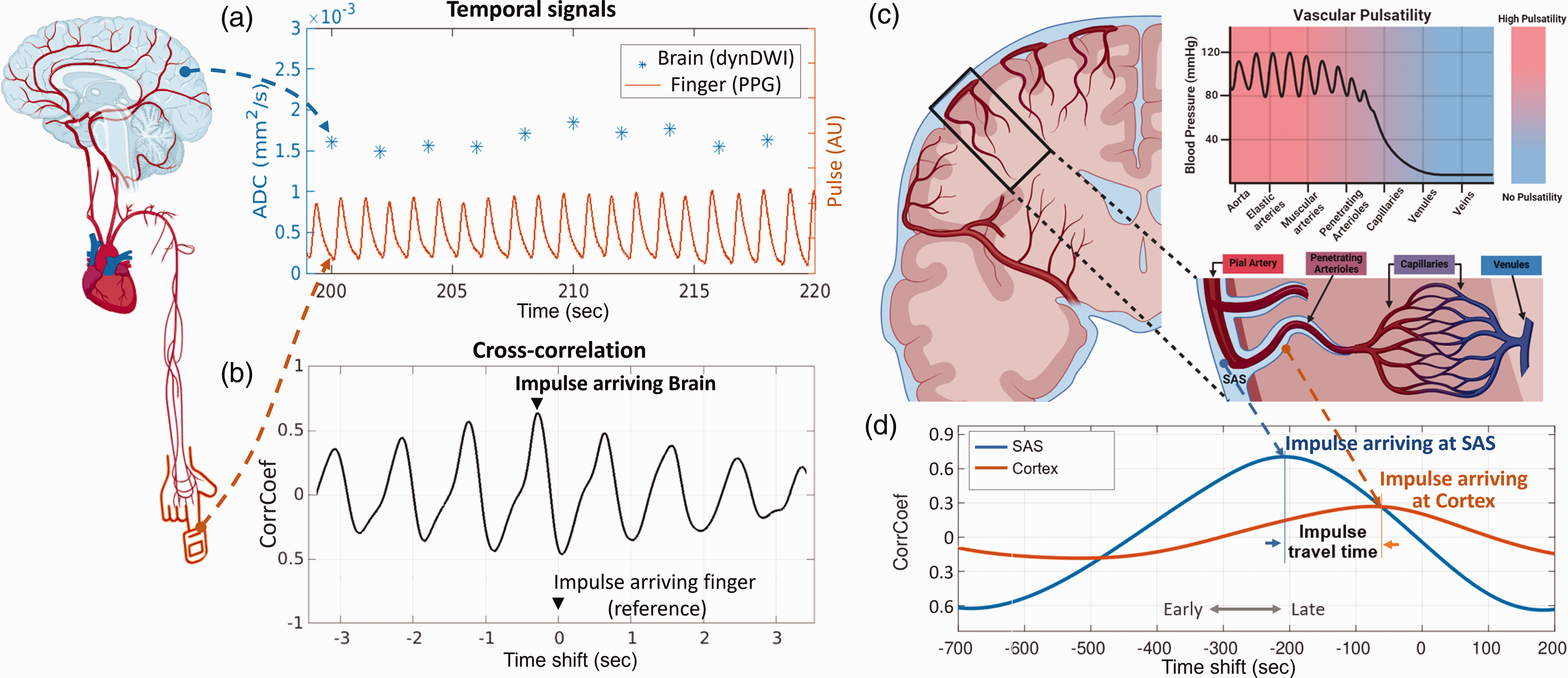
Schematic view of the cross-correlation analysis used to extract impulse arrival time within different brain regions. (a) Temporal signals of simultaneously acquired brain dynamic diffusion-weighted imaging (dynDWI) (1750 ms sampling rate) and finger Photoplethysmography (PPG) (2.5 ms sampling rate). (b) Peak correlation coefficient (CorrCoef) of the cross-correlation curve indicates the impulse arrival time at a brain voxel relative to finger (reference point, time shift = 0). This negative time shift illustrates impulse arriving at the brain earlier than the finger. (c) Illustration of the cerebral vascular tree, CSF-filled paravascular space, and vascular pulsatility changes along it and (d) The cross-correlation curves of one voxel from the subarachnoid space (SAS) and one from the cortex. The SAS voxel shows a higher CorrCoef and an earlier impulse arrival than the cortex.
This analysis was applied to voxels along vascular segments, from the pial arteries in the subarachnoid space (SAS) to arterioles within the cortex (Figure 1(c), (d)). Results indicate earlier impulse arrival and higher pulsatility in SAS, while the cortex shows later arrival and lower pulsatility (Figure 1(d)).
Human participants and data acquisition
Seventy healthy participants aged 18–85 years (28 males and 42 females) were enrolled in the study. All participants provided written informed consent according to procedures approved by the Institutional Committee for the Protection of Human Subjects at Indiana University. The study was conducted in adherence to the principles of the Belmont Report (Respect for Persons, Beneficence, and Justice) and federal regulations outlined in 45 CFR 46 and 21 CFR 50 and 56.
All MRI data were collected on a Siemens Prisma 3 T scanner with a 64-channel RF receiver head–neck coil. DynDWI was collected at b = 150 s/mm2 at three cardinal diffusion encoding directions (x/y/z), each repeated 45 times to capture signal changes at various phases of the cardiac cycle. Simultaneously, the scanner’s built-in finger photoplethysmography (PPG) recorded physiological data. Additional acquisition parameters included a voxel size of 1.8 × 1.8 × 4 mm3, a repetition time/echo time of 1750 msec/49.6 msec, a multi-band factor of 2, 36 slices for whole-brain coverage, ten b = 0 s/mm2 volumes, and a total acquisition time of 4 minutes and 58 seconds. T1-weighted anatomical imaging data (T1W) were collected using a three-dimensional (3 D) magnetization rapid gradient echo (MPRAGE) with 1.1 × 1.1 × 1.2 mm3 voxels.
ROI-wise and voxel-wise analyses
To observe the time window of intracranial impulse arrival time for all subjects, a global mean signal of three regions of interest (ROIs) was cross-correlated with PPG for each subject using a wide time shift window of [−3, 3] seconds. The three ROIs correspond to the pulsatile CSF surrounding surface arteries in the subarachnoid space (SAS), cortex, and white matter. Cortex and white matter ROIs were segmented on T1-weighted images (FMRIB Software Library [FSL]: fsl_anat) and then registered to the diffusion space using linear registration (FSL: epi_reg). The SAS mask was generated using a previously established method that detects pulsatile CSF voxels. This iterative algorithm identifies voxels within the subarachnoid space by discerning regions with significant ADC differences between systolic and diastolic phases (>200 × 10−6 mm2/s). The validity of this approach was confirmed in our previous publication by comparing it with artery regions identified in time-of-flight images of the same subjects. 19 ROI-wise analysis facilitated the observation of time delay patterns of all participants, with each participant providing one cross-correlation curve.
To visualize spatial pattern of impulse arrival time, voxel-wise analysis was performed for each subject, generating an ArrivalTime map and a CorrCoef map. The voxel-wise analysis applied a narrower time shift window of [−400, 400] msec in the cross-correlation, falling within a physiologically plausible range determined from the ROI-wise analysis (Figure 2(c) in the Results section) and consistent with prior studies. 18 This time window for impulse arrival time is significantly shorter than the blood transit time (∼1 sec from arteries to reach tissue) because the mechanical pressure wave travels much faster than the flow, though both are driven by cardiac pulsation. Narrowing this window also improves calculation speed and the robustness of arrival time extraction, resulting in ArrivalTime and CorrCoef maps for each subject. Mean ArrivalTime was subsequently quantified in each ROI. To mitigate partial volume effects with the SAS that could bias cortex and white matter quantification, voxels with ADC < 900 mm2/s were used to constrain cortex/white matter voxels for quantification.
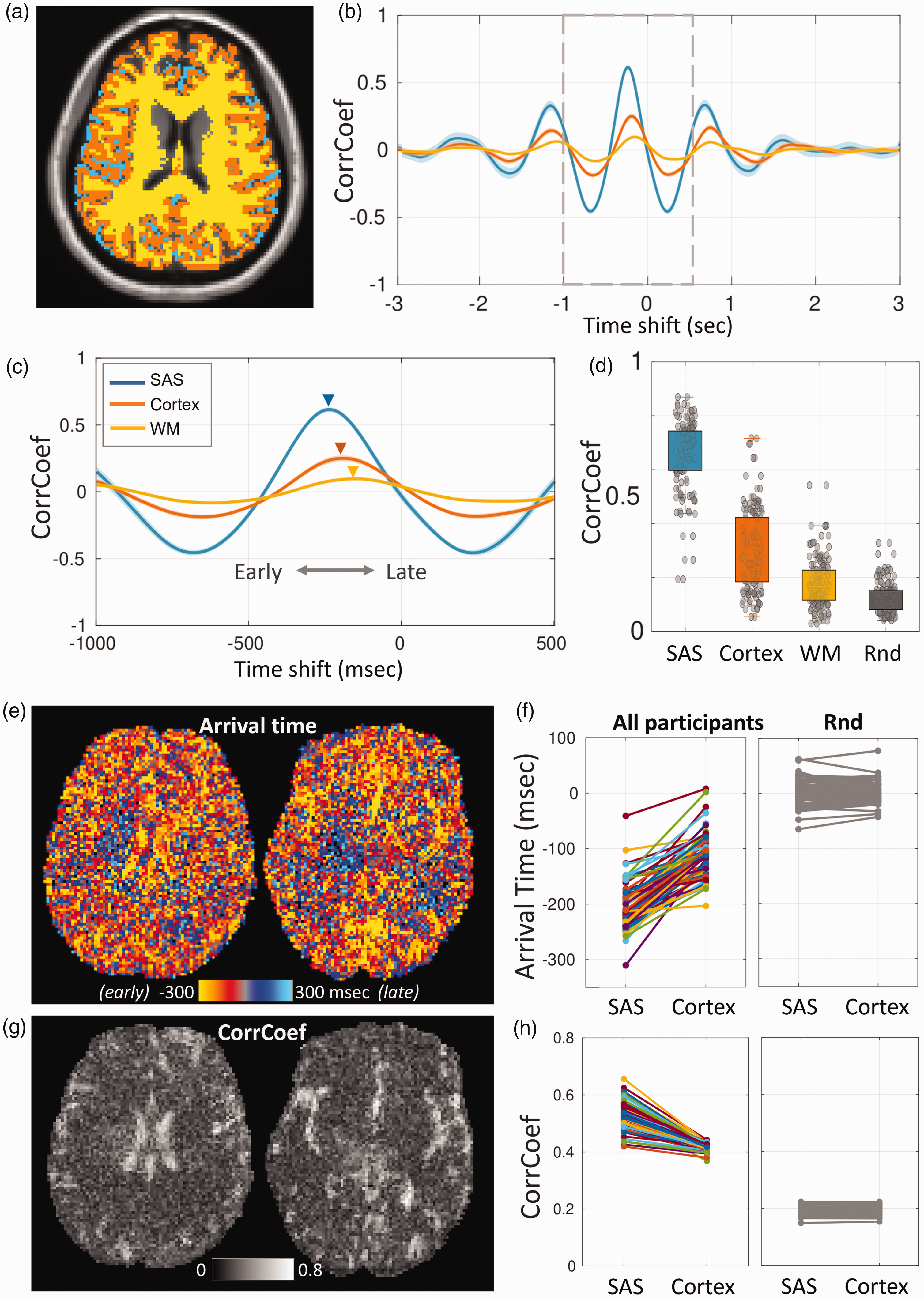
Slice-wise and voxel-wise analyses revealed consistent impulse arrival patterns, from SAS, to cortex, then to white matter. (a) An axial slice of middle brain with a mask overlay indicating SAS (blue), Cortex (orange), WM (yellow) regions. (b) Cross-correlation curves of 70 participants show a consistent arrival pattern, with the brain leading the finger. Within the brain, SAS leads Cortex and WM. The solid line and shaded area correspond to mean and standard error. (c) The 1500 ms zoom-in window of the cross-correlation. (d) The boxplot shows that the CorrCoef of SAS, Cortex, and WM are higher than that calculated using the simulated random noise, supporting that dynDWI signal is coupled to cardiac pulsation. (e, g) Map of impulse arrival time and CorrCoef for two axial slices of a presentative participant. SAS exhibits earlier impulse arrival and higher CorrCoef compared to Cortex. (f) Mean arrival time of SAS and Cortex from all participants (N = 70) showing SAS-early-Cortex-late trend. The random control analysis on the right shows no such trend and (h) The CorrCoef in SAS is higher than that in the cortex, which is not seen in the random control analysis on the right. Abbreviations: SAS – subarachnoid space; WM – white-matter; CorrCoef – correlation coefficient; Rnd: random control analysis.
To ascertain whether the observed ArrivalTime and CorrCoef were true signals or occurred by chance, negative control analyses were performed whenever possible. Specifically, for every cross-correlation performed, a negative control pair was generated by replacing dynDWI with Gaussian random noise and cross-correlating it with actual PPG data.
Assessment of reproducibility and the impact of b-value, blood signal, and waveform shape alterations on travel time
To assess reproducibility, four participants (2 females, 2 males, age <35 years old) underwent four repeats within the same scan session.
To verify that dynDWI signal originated from CSF independently of blood signal, we employed two approaches to suppress the blood signal and assess any alterations in TravelTime. Firstly, dynDWI was collected at varying b-values (b = 50, 100, 150, 200, 500 mm2/s), with higher b-values better suppressing the blood signal. This experiment was performed in three subjects (1 female, 2 males, age <35 years old). Secondly, blood-nulling dynDWI acquisitions were conducted on four participants (1 female, 3 males, age <35 years old). Blood-nulling dynDWI placed one or two saturation bands below the dynDWI imaging volume before every slice excitation pulse to suppress the incoming blood in the carotid and vertebral arteries.
Additionally, the cross-correlation between the CSF waveform and finger PPG may be influenced by changes in CSF waveform shape, such as the widened systolic peak observed in older brains.17,19 To assess whether waveform shape alterations confound travel time and its associations with age and other variables, waveform peak width (PeakWidth) was included as a covariate in the linear regression model.
Statistical analyses
A Student’s t-test and ANOVA were employed to evaluate differences between two groups and among multiple groups, respectively. The Shapiro-Wilk normality test was performed for each variable. For variables that did not follow a normal distribution (p < 0.05), the Mann-Whitney rank-sum test was used instead. Pearson’s correlation was utilized to assess associations between continuous variables across all participants. Variables that did not follow normal distributions were Box-Cox transformed before analyses. Linear regression models were employed to analyze the relationship between imaging metrics (dependent variable) and age, sex, heart rate, and cortical thickness (independent variables). In the presence of a bi-phasic trend between imaging metrics and age, second-order polynomial fitting was conducted to determine the turning point of age. TravelTime reproducibility was assessed using total-least-squares regression, Bland-Altman plots, and intraclass correlation coefficient (ICC) tests. To account for the four repeated measurements within each subject, we calculated the six unique pairs of measurements (24 pairs total across four subjects) and used these pairs as inputs for the Bland-Altman analysis.
Biomechanical model of cardiac impulse propagation
Computational biomechanical modeling was performed to validate image findings on the SSS-Cortex travel time. We employed a recently developed reduced-order model of the cardiovascular system capable of simulating pressure impulse travel along the arterial trees with cardiac pulsations. 28 The model consists of 83 systemic arteries within the trunk, head, and limbs, with morphologies derived from extensive review of modern high-resolution anatomical data sets in humans. The modeled arterial network consists of three main parts (Figure 5(a)–(c)): 1) a single ventricle pump at the heart, 2) transmission line RLC segments (resistance, inductance, and compliance) for major arteries (Figure 5(c)), and 3) three-element Windkessel boundaries for the terminal conditions to form a closed-loop system, representing the artery-to-arteriole branching along the SAS-cortex segment (Figure 5(b)).
We calculated pressure impulse travel time using the rate of change in the pulse waveform (dP/dt) measured at major cerebral arteries and terminal cerebrovascular beds, approximating SAS-cortex time delays. Simulations were performed separately for young and old, with age-specific parameters detailed in the Supplementary material.
Results
Consistent timing patterns demonstrate impulse propagation from SAS to cortex and white matter
Among the 70 individuals studied, the impulse propagation exhibits a consistent pattern, with the earliest arrival observed in SAS, followed by the cortex, and WM (Figure 2(c)). The correlation coefficient was highest in SAS with a median value of 0.69, indicating that 69% of the signal variation in this region was driven by cardiac pulsation. This reflected the strong influence of cardiac pulsation on CSF dynamics in the SAS. In contrast, this influence diminished in the cortex and white matter, where cardiac pulsation accounts for 27% and 17% of the signal variation, respectively (Figure 2(d)). Other physiological factors may underlie the lower cardiac pulsation component in the parenchymal tissue, such as respiration and low-frequency vasomotion.3,29 For each cross-correlation, a negative control result was generated by cross-correlating a simulated dynDWI signal of Gaussian random noise with the actual finger PPG. The correlation coefficients in all three regions were significantly higher than those of the negative control (Figure 2(d), t-test, p < 0.001, effect size d = 3.71, 1.10, 0.5, 95% CI [3.47, 3.95], [0.86, 1.34], [0.26, 0.74] for SAS, Cortex, and WM respectively). These results confirmed that the dynDWI signal fluctuations in all three ROIs were coupled with cardiac pulsations.
ArrivalTime and CorrCoef maps from a representative subject using voxel-wise analysis showed earlier arrival in SAS, then cortex, and lastly WM, with SAS having the highest CorrCoef (Figure 2(e)–(g)). ArrivalTime and CorrCoef data for all 70 individuals showed earlier arrival at the SAS than the cortex, with an arrival time of −196 ± 41 ms for the SAS and −112 ± 42 ms for the cortex (paired t-test: p < 0.001, effect size d = 1.83, 95% CI [1.60, 2.06]) (Figure 2(c) left). A negative value indicates an arrival time preceding the finger pulse. Negative control analysis showed no difference between regions, confirming the validity of the imaging results (Figure 2(c) right). Negative control analysis showed no arrival time difference between regions, with an arrival time centered around 0 and a CorrCoef around 0.2 (Figure 2(c) right).
SAS-Cortex TravelTime is age dependent
SAS-Cortex impulse TravelTime was quantified by subtracting the arrival time at the Cortex from that at the SAS. Plotting the TravelTime against age revealed a bi-model trend. The second-order polynomial fitting showed TravelTime was significantly associated with age (p = 0.012 for Age and p = 0.002 for Age2) with a peak TravelTime appearing around 44.6 years old. Linear regression was subsequently applied in each age group, with TravelTime as dependent variable and age, sex, heart rate (HR), and cortical thickness as independent variables. Below 44.6 years old, TravelTime increases with age (regression coefficient = 4.01, 95% CI [1.83, 6.19], p = 0.002). Above 44.6 y/o, TravelTime decreases with age (regression coefficient = −1.39, 95% CI [−2.64, −0.14], p = 0.036) (Figure 3(a)). The average TravelTime was 83.8 ± 38.0 msec across all participants, 99.9 ± 35.9 msec in the younger cohort, and 75.6 ± 37.6 msec in the older cohort. Additionally, a higher heart rate (HR) was significantly associated with a shorter TravelTime in the younger cohort (Figure 3(b)). This trend was not seen in the older cohort (Figure 3(c)).
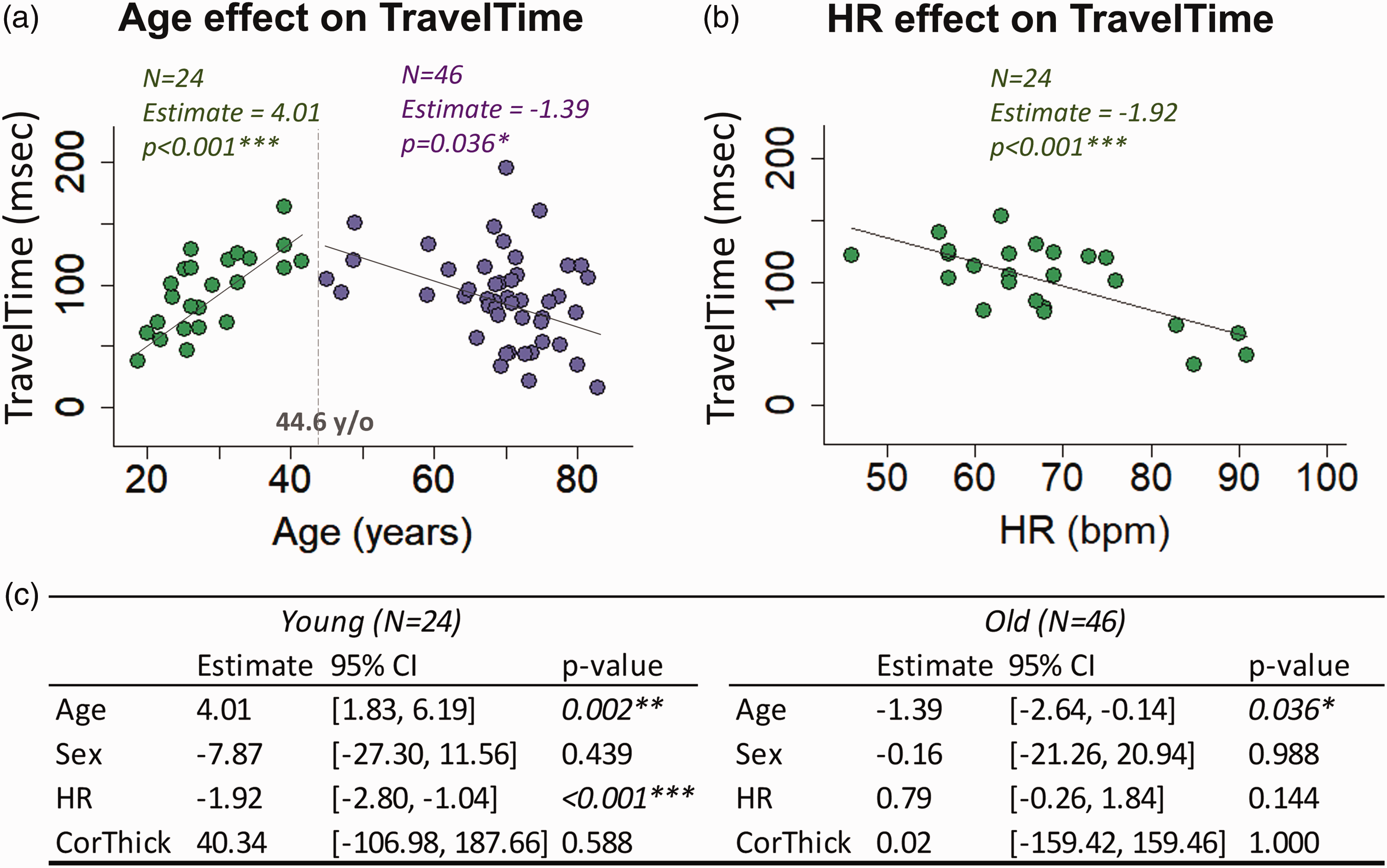
SAS-cortex impulse TravelTime exhibited a bi-modal trend with age (N = 70). (a) Under 44.6 years old, TravelTime increased with age (N = 24, p = 0.002, after controlling for Sex, HR, and cortical thickness). Above >44.6 years old, TravelTime decreased with age (N = 47, p = 0.036 after controlling for Sex, HR, and cortical thickness). (b) A higher heart rate was significantly associated with a decrease in TravelTime in the younger cohort (N = 24, p < 0.001) and (c) The table summarizes regression results within each age group. Abbreviations: SAS – subarachnoid space; HR – heart rate.
Impulse propagation measurements were reproducible and independent of the arterial blood flow
Figure 4(a) illustrates the reproducible impulse travel time measurements. The intraclass correlation coefficient (ICC) across four repeated scans of all participants was 0.87 for SAS arrival time (95% CI [0.60, 0.99], p < 0.001), 0.85 for Cortex arrival time (95% CI [0.53, 0.99], p < 0.001), 0.71 for TravelTime (95% CI [0.27, 0.98], p < 0.001), in the range of an excellent (ICC > 0.75) and good (0.6 < ICC < 0.75) level of agreement. Linear regression and Bland-Altman analysis demonstrated agreement between test-retest scans, with a mean difference of 0.69 ± 31.52 ms. A paired t-test (p = 0.88) indicated no significant difference between the test-retest measures.
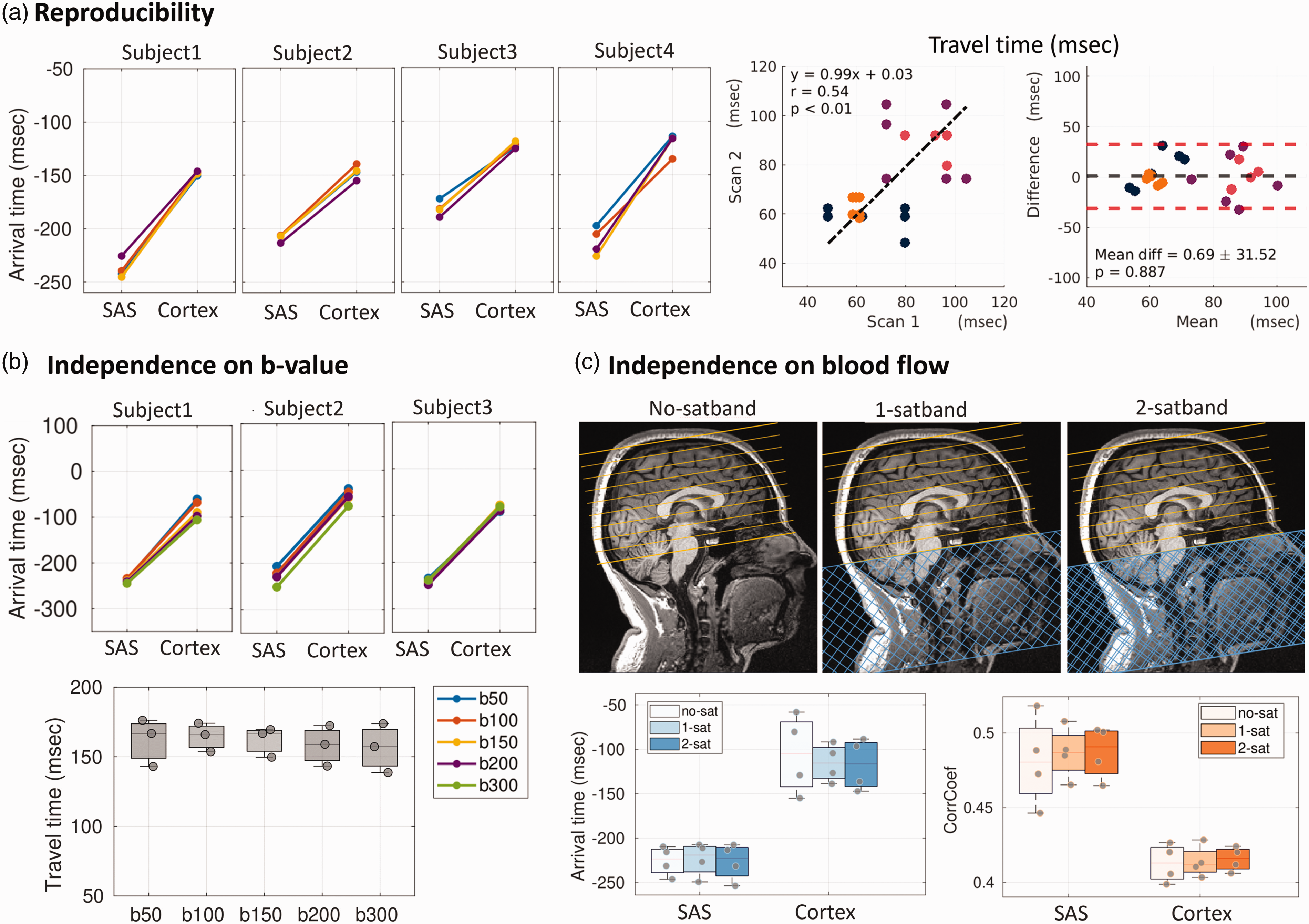
TravelTime showed good reproducibility and independence of b-value and blood flow. (a) dynDWI was repeated for four participants within the same imaging session. Impulse arrival time demonstrated reproducibility, with an intra-class correlation coefficient of 0.87, 0.85, and 0.71 for SAS arrival time, Cortex arrival time, and SAS-Cortex TravelTime, respectively. For TravelTime, linear regression showed strong linearity between test-retest results (slope = 0.99, Pearson’s r = 0.54, p < 0.01, each color represents the six pairs derived from four repeats for each subject). Bland-Altman analysis and a paired t-test (p = 0.88) confirmed no significant differences between test-retest measurements. (b) Varying b-values from 50 to 300 mm2/s did not impact TravelTime measurements (ANOVA, p > 0.1, effect size = 0.056) and (c) Saturation bands were positioned below the bottom slice to suppress the incoming blood signal. Different numbers of saturation bands were utilized to achieve varying levels of blood suppression, including no saturation band (no-sat), 1 saturation band (1-sat), and 2 saturation bands (2-sat). Boxplot results indicate that altering blood suppression does not affect the measurements of arrival time and CorrCoef (ANOVA, p > 0.1, effect size = 0.005).
Varying b-values results revealed that employing a higher b-value did not induce changes in impulse TravelTime (ANOVA, p > 0.1, effect size =0.056, Figure 4(b)). When saturation bands were positioned below the imaging volume to suppress incoming blood, the outcomes demonstrated no differences in the impulse arrival times between conditions without saturation band (No-satband), with one satband (1-satband), and with two overlapping satbands (2-satbands) (ANOVA, p > 0.1, effect size = 0.005, Figure 4(c)). These experiments collectively support the independence of the dynDWI signal from arterial blood contributions.
When comparing cardiac-aligned pulsation waveforms, the older brain showed a widened peak compared to the younger brain in both the SAS and Cortex (Supplementary Figure 1AB), consistent with prior observations. 19 Including PeakWidth as a covariate revealed no association with travel time and no impact on the observed travel time–age relationship (Supplementary Figure 1C). The consistent waveform shape changes between the SAS and Cortex likely have a similar effect on cross-correlation calculations, canceling out their influence on SAS-Cortex travel time.
Measured SAS-Cortex impulse travel time aligns with predicted values using biomechanical modeling
Model results showed the pressure impulse takes 71.3 msec from SAS to reach terminal vessels in a typical younger brain and 62.5 msec in a typical older brain. These values closely aligned with the TravelTime measurements using dynDWI, with 99.9 msec in the young and 75.6 in the old (Figure 3(a)).
When the heart rate varied from 50 to 90, the impulse travel time was shortened from 78 to 70 msec in young and from 65 to 61 msec in old, corresponding to a linear regression slope of −0.2 and −0.1 (Figure 5(e)). The direction of change and steeper slope in young people align with imaging findings. However, model simulations showed a smaller heart rate impact on impulse travel time compared to imaging data, showing a stronger HR effect on TravelTime (coefficient =−1.92, Figure 3(c)).
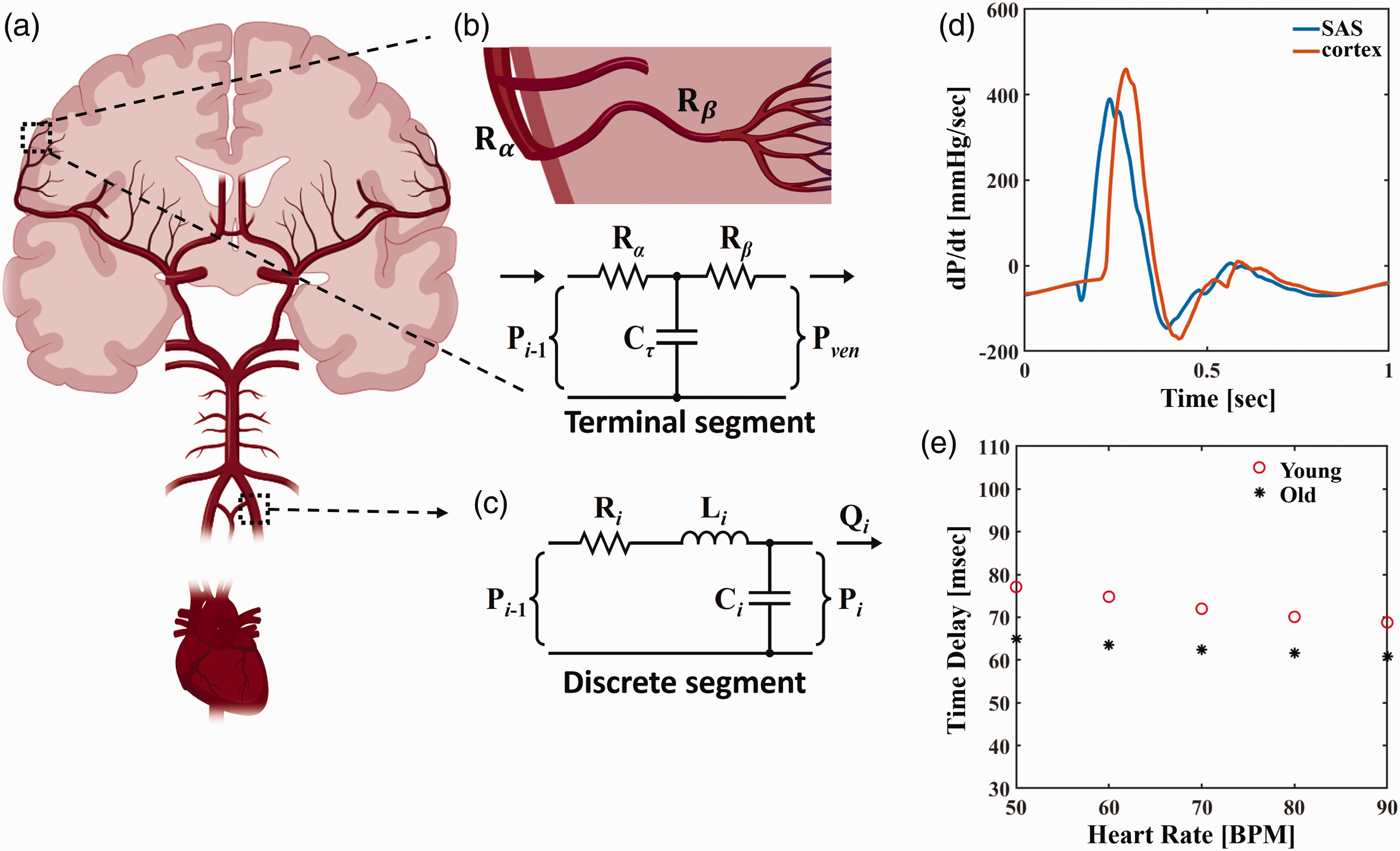
The components of the model and explanation of time delay measures via computational modeling. (a) The systemic model of the cardiovascular system is driven by cardiac pressure propagation, enabling probing for regional arterial pressure and flow waveforms. (b) Visual representation of Rα and Rβ terms associated with three-element Windkessel boundaries connecting terminal segments to the lumped venous compartment. (c) Discrete transmission line RLC segments with resistance “Ri,” inertance “Li,” and compliance “Ci” that were updated each time step. (d) Representative rate of pressure change (dP/dt) waveforms simulated from the model for calculating SAS-Cortex travel times. Cortex waveforms are representative of pressure changes in downstream terminal Windkessel boundaries, taken as proxy for small vessel (i.e., arteriole) pressurization and (e) Travel times are dependent on heart rate, with heart rate influencing young models of circulation greater than old circulations.
Biomechanical modeling suggests that SAS-Cortex impulse TravelTime is mainly influenced by terminal vessel compliance
To explore the vascular properties driving the age-related changes in TravelTime, we conducted a sensitivity analysis in biomedical modeling to assess how variations in model parameters affect travel time. Four model parameters were examined: the terminal resistance
Results show that terminal compliance had the most significant impact on travel time (Supplementary Figure 1). Varying terminal compliance (pial arteries, arterioles, and capillaries) decreased travel time by up to 46.9% in young and 68.1% in old subjects. In comparison, changes in discrete compliance resulted in an 18.7% shift in young and a 13.2% shift in old. Resistance effects, both discrete and terminal, were minimal in young models (less than 5%) but more pronounced in older cases (discrete, less than 1%; terminal, 18.6%).
Discussion
We conclude that impulse propagation from pial arteries to penetrating arterioles is detectable through the paravascular CSF using dynDWI. Results from 70 subjects consistently showed an early impulse arrival at pial arteries and a subsequent arrival at penetrating arterioles, with a mean travel time of 84 msec. Notably, this duration contrasts sharply with the 1–2 seconds propagation of blood flow, highlighting its distinct nature. Blood flow travels at a much lower velocity than the pressure impulse. Analogously, blood flow is like a moving train, while impulse propagation resembles its traveling sound – a mechanical wave that moves much faster than the train itself. The blood transit from major arteries to the capillary bed has traditionally been used to study tissue viability, utilizing perfusion imaging techniques such as arterial spin labeling,30 –32 or MR-encephalography (MREG) that detects oxy-/deoxy-hemoglobin fluctuations.33,34 Beyond its distinction from blood transit time, the spatial pattern of impulse propagation differs notably from the tissue pulsation patterns observed with other MRI techniques. Tissue motion in response to brain pulsation generates subtle sub-voxel coherent motion, which dynDWI is less sensitive to but is measurable through displacement encoding techniques (DENSE) and amplified MRI (aMRI).35 –39 Such tissue motion represents a macroscopic level compensation in response to cardiac impulse. This tissue motion exhibits a centrifugal, or inside-out, propagation pattern where the brainstem pulsates first due to the Circle of Willis pulsation. It then propagates upward to the ventricles, followed by the white matter and cortex. The pressure impulse propagation observed in our study diverges from the tissue pulsation pattern and follows a centripetal direction, from outward to inward: SAS → gray matter → white matter.
The SAS-Cortex travel time of 84 milliseconds appears slow compared to the pulse wave velocity (PWV) measured along the larger upstream arteries such as carotid and intracranial arteries. While both measurements assess impulse propagation, the pathways of propagation are significantly different. PWV is typically measured along major arteries, reflecting the stiffness of these artery segments. PWV values range from about 3 to 18 meters per second (m/s) at the carotid artery40 –42 and 8 to 30 m/s along the intracranial arteries,7,8 resulting in a travel time of only 2 to 13 msec to cover a distance of 4 millimeters, much shorter than the 84 msec observed in the SAS-Cortex pathway. In contrast, the SAS-parenchyma impulse travel focuses on the branching of vessels, transmitting from parent arteries on the brain surface to daughter arterioles within the parenchyma. These daughter arterioles are notably smaller in size and higher in density, designed to provide sufficient dampening of pressure pulses while sustaining constant, high blood flow to the vascular bed. Conceptually, large vessel PWV is like cars rushing on highways, whereas SAS-Cortex travel is akin to getting off highways to enter local streets. This timescale difference aligns well with biomechanical modeling results, which project a SAS-Cortex travel time of around 70 milliseconds, consistent with the 84 milliseconds in imaging findings.
The biomechanical modeling suggests that changes in the SAS-cortex impulse travel time mainly arise from alterations in the compliance of the daughter arterioles within the cortex, rather than those upstream arteries before the SAS segments. Compliance is determined by factors such as vessel radius, wall elasticity, and wall thickness, with higher compliance indicating better wall elasticity, larger vessel radius, and lower wall thickness.28,43,44
Incorporating insight from the biomechanical modeling, the observed age effect on SAS-cortex travel time can be partially explained by changes in arteriolar compliance. Our imaging results revealed a bimodal age effect on SAS-cortex impulse travel, with travel time increasing with age under 44.7 y/o and decreasing with age above 44.7 y/o. This may reflect bimodal changes in the compliance of arterioles determined by a combined effect of vessel diameters, density, elasticity, and wall thickness during mid-age and aging. The observed shortening in travel time above 44.7 aligns well with known changes in the vascular system during aging. The breakdown of elastin, accumulation of stiffer collagen fibers, and thickening of the vessel walls in age-related hypertension cause reduced compliance.45 –47 These vessel changes collectively cause a reduction in vessel compliance, explaining the observed shortened travel time with advanced age.
The more intriguing observation is the increase in travel time with age among the younger cohort under 45 years old. According to the Moens–Korteweg equation, PWV is proportional to the square root of stiffness (Young’s modulus) of the vessel wall and wall thickness, and inversely proportional to the square root of the wall diameter. Hence, increased travel time with decreased PWV could be linked to a decrease in arterial stiffness, a reduction in wall thickness, or an enlargement in vessel radius through middle age. Although all of these possibilities seem unlikely, further study is needed to evaluate both reproducibility and potential mechanisms. Nevertheless, these findings underscore the potential of dynDWI in measuring impulse propagation as indicators of arteriolar compliance. These indicators may shed light on both local intravascular blood flow, providing oxygen and nutrients, and paravascular cerebrospinal fluid (CSF) flow, facilitating the removal of waste products, in the underlying cerebral cortex.
Our study introduces new imaging and analytical methods, prompting discussion on key considerations. The imaging approach detects dynamic changes in incoherent CSF motion within the paravascular space, thus eliminating the need for super-resolution imaging to delineate singular paravascular spaces of arterioles. However, this raises uncertainties about whether these changes are specific to CSF or if they also include blood contributions. To assess this, we conducted experiments to progressively suppress blood signals using increased b-values and saturation bands at the neck regions. Our findings showed that these manipulations did not affect the measured pulsatile signal, indicating the effective elimination of blood contributions by the applied diffusion gradient, consistent with literature.22 –27 Nonetheless, the current resolution may result in partial voluming between the subarachnoid space and cortical regions. Future studies employing higher resolution for improved delineation of these regions would be beneficial. Additionally, cortical para-venous CSF may contribute to the measured dynDWI signal in the parenchyma. However, this contribution is likely small, given the half density of venules compared to arterioles 48 and their reduced pulsatility.13,49 In the subarachnoid space, our segmentation pipeline selects CSF regions with systole-diastole ADC differences exceeding 200 × 10–³ mm2/s. This approach primarily captures para-arterial CSF and excludes CSF near surface veins, characterized by lower pulsatility driven by Δ5 mmHg intracranial pressure fluctuations, in contrast to the higher pulsatility of para-arterial spaces influenced by Δ40 mmHg arterial pressure fluctuations.50,51 In signal processing, ensuring the quality of the PPG signal is crucial for the cross-correlation analysis. Factors such as cold hands, colored fingernails, or loose connections can compromise signal integrity. We excluded six datasets (8%) due to poor PPG signals. Verifying PPG signal quality during scans can mitigate the risk of erroneous data collection.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251320902 - Supplemental material for Dynamic diffusion-weighted imaging of intracranial cardiac impulse propagation along arteries to arterioles in the aging brain
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251320902 for Dynamic diffusion-weighted imaging of intracranial cardiac impulse propagation along arteries to arterioles in the aging brain by Qiuting Wen, Joseph Muskat, Charles F Babbs, Adam M Wright, Yi Zhao, Xiaopeng Zhou, Chengcheng Zhu, Yunjie Tong, Yu-Chien Wu, Shannon L Risacher and Andrew J Saykin in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the National Institute of Health, including RF1 AG083762, R01 AG053993 and P30 AG072976.
Acknowledgements
The authors thank Dr. Deva Chan and Dr. Vitaly Rayz from Purdue University for useful discussions. The illustrations in Figure 1(a,c) and Figure 5(a,b) were created with BioRender.com.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
