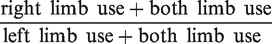Abstract
Ischemic stroke is a leading cause of disability and death globally. Stem cell therapies are emerging as a frontier for enhancing post-stroke recovery, with Muse cells—a subclass of pluripotent stem cells—demonstrating considerable promise. Muse cells are notable not only for their potential in cell replacement but also for their role in modulating immune responses following cerebral infarction. In the present study, we administered Muse cells intravenously to mice after inducing a stroke via distal middle cerebral artery occlusion. We evaluated motor outcomes, splenocyte populations, cytokine profiles, and gene expression 2 weeks after inducing stroke. Additionally, comparisons were drawn between outcomes in splenectomized mice and those receiving adoptive splenocyte transfer to discern the specific influence of the spleen on treatment efficacy. Our findings revealed that Muse cell therapy facilitates motor recovery, an effect that is compromised in the absence of the spleen. Spleens in treated mice exhibited a shift in neutrophil counts, increased cytokine activity, and a notable uptick in the expression of genes related to protein folding. These insights affirm the potential therapeutic effect of Muse cells in post-stroke treatment strategies, with their efficacy attributed, at least in part, to immunomodulatory pathways involving the spleen.
Introduction
Ischemic stroke is a major cause of death and disability worldwide.1,2 Although recombinant tissue plasminogen activator and mechanical thrombectomy have significantly improved the outcome of occlusive ischemic stroke,3,4 most stroke survivors suffer irreversible brain damage leading to severe morbidity, creating an urgent need to develop treatment strategies that promote neurologic recovery after stroke.
Stem cell therapy is a promising treatment strategy for ischemic stroke.5,6 The protective effects of mesenchymal stromal cells (MSCs) against ischemic stroke pathology have been evaluated in several in vitro and in vivo studies. The mechanisms by which MSCs contribute to neurologic recovery following ischemic stroke, however, are not fully understood, but may include mitochondrial transfer7,8 and trophic effects through exosome secretion.9,10 Many clinical trials have failed to replicate the promising results of MSC treatment following ischemic stroke,11 –13 however, which may be due to the insufficient regenerative capacity of MSCs. Homing of transplanted stem cells to the injured tissues and differentiation of the transplanted stem cells into target cells may also be important factors for sufficient neurologic recovery.
Multilineage-differentiating stress-enduring (Muse) cells are nontumorigenic endogenous pluripotent stem cells easily isolated as SSEA-3+ cells that mobilize from the bone marrow to the peripheral blood and distribute to organ connective tissues.14 –16 We previously demonstrated that transplanted Muse cells autonomously migrate into peri-infarct areas and differentiate into neurons and oligodendrocytes to support neurologic recovery after ischemic stroke in an immunodeficient rodent model.17 –19 Indirect mechanisms underlying the therapeutic effects of Muse cells to support neurologic recovery, however, remain largely unknown. A recent human clinical trial using CL2020, an allogeneic Muse cell-based product, demonstrated promising results, suggesting effective treatment possibilities for subacute ischemic stroke with a notable response rate and manageable safety profile. 20
Here, we investigated the immunomodulatory effects of human Muse cells to enhance our understanding of their physiologic roles in neurologic recovery after ischemic stroke following systemic xenotransplantation without immunosuppression.
Methods
Distal Middle cerebral artery occlusion surgery
All animal experiments comply with the Animal Research: Reporting of In Vivo Experiments guidelines and are carried out in accordance with the Code of Ethics of the World Medical Association as well as the Tohoku University Guidelines based on the International Guiding Principles for Biomedical Research Involving Animals. The animal protocols were approved by Tohoku University’s Administrative Panel on Laboratory Animal Care (Access number: 2018MdA-204). Male mice (CB-17/Icr-+/+-Jc1, 8–10 weeks of age) were purchased from Clea-Japan Inc. Mice were housed under a 12-h light/dark cycle with ad libitum access to regular food and water. The animals underwent left distal middle cerebral artery occlusion (dMCAO) as previously described.21,22 Briefly, mice were anesthetized with 2% isoflurane in a mixture of 30% oxygen and 70% nitrous oxide. Rectal temperature was maintained at 37°C during surgery. The temporalis muscle between the left eye and ear was retracted along a 4-mm skin incision. A small bone window (∼1 mm diameter) was created to expose the distal middle cerebral artery (MCA). The MCA was cauterized just distal to the lenticulostriate branches. The incision was closed with 6-0 nylon sutures. Animals were monitored in a warmed cage during recovery from anesthesia. Just prior to MCA occlusion, the neck was dissected and the left common carotid artery was temporarily occluded with 6-0 nylon before the bifurcation. The carotid artery ligation was removed after 1 h.
Intravenous administration of muse cells
Green fluorescent protein (GFP)-labeled MSCs were prepared by labeling human bone marrow-derived MSCs (BM-MSCs, Lonza, Basel, Switzerland) with lentivirus GFP as previously described, 23 For lentivirus production, pMD2G, pCMV deltaR8.74, and pWPXL-GFP were transfected into LentiX-293T packaging cells (Takara Bio Inc, Shiga, Japan) using Lipofectamine 2000 (Thermo Fisher Scientific, Waltham, MA, USA). After 3 days, the viral supernatant was collected, centrifuged, and filtered through a 0.45-µm filter. To collect human Muse cells, MSCs or GFP-labeled MSCs were incubated with anti-stage specific embryonic antigen (SSEA)-3 antibody (1:200; Cat # 330302, Biolegend, San Diego, CA, USA) and stained with secondary antibody allophycocyanin (APC)-conjugated anti-rat IgM (1:100; Cat # 112-136-075, Jackson ImmunoResearch Laboratories, Inc., West Grove, PA, USA) as previously described.14,15 SSEA-3-negative cells were used as “non-Muse cells”. The gating strategy for isolation is shown in Supplementary Figure 1.
We also used the clinical-grade Muse cell-based product CL2020 provided by Life Science Institute Inc. These cells were produced from human MSCs by exposing them to a combination of stressors and then confirmed to be positive for both pluripotency marker SSEA-3 and mesenchymal marker CD105, but negative for leukocyte marker CD45.19,24
Prior to injection, Muse cells were thawed, washed, and suspended in Hank’s Balanced Salt Solution (HBSS, Cat # 14025076, Gibco). Cells were counted and checked for viability via Trypan blue exclusion. In the present study, only Muse cells with viability >95% were used. Muse cells (5 × 104 cells) in 200 µl HBSS were intravenously injected at a rate of 0.2 ml/min via the jugular vein at 48 h after dMCAO. Stroke-injured control animals were administered vehicle (HBSS) in the same manner as the Muse cell injections.
For histologic assessment, we evaluated the location of GFP-labeled Muse cells. For flow cytometry, RNA-sequencing, and reverse transcription-polymerase chain reaction (RT-PCR), we used Muse cells without labeling. For behavioral assessment and adoptive cell transfer studies, we used CL2020.
Behavioral analysis
Long-term dMCAO-induced paralysis was assessed using the cylinder test. The cylinder test was administered before dMCAO surgery, 1 day after dMCAO surgery (1 day before Muse cell administration), and at 1, 2, 4, 6, and 8 weeks after Muse cell administration by an investigator (A.K.) blinded to the experimental groups. The cylinder test was adapted for use with mice to assess forelimb use and rotation asymmetry. Mice were placed in a transparent glass cylinder (diameter 9 cm, height 15 cm) and videotaped from the side during the test. The cylinder was placed next to a mirror to enable visualization of limb use movements from all angles. Forelimb use at the first contact against the wall after rearing and during lateral exploration was recorded according to previously described criteria.25,26 The test continued until a total of 20 vertical movements were recorded. The asymmetry score was obtained as follows:
In the adoptive cell transfer experiment, the cylinder and adhesive tape removal tests were used to assess hemiparalysis. The adhesive tape removal test is used to assess post-stroke sensorimotor loss. Mice were placed in a transparent box (15 cm × 25 cm) and small adhesive tape strips (3 mm × 4 mm) were applied on each forepaw with equal pressure. The time required for the mouse to remove the tape strips (maximum 120 s) was recorded. 27 Behavioral assessment was performed before dMCAO surgery, 1 day after dMCAO surgery, and 1 day after adoptive splenocyte transfer by an investigator blinded to the experimental groups.
Immunohistochemistry
At 2 days, and 2 and 8 weeks after Muse cell administration, animals were anesthetized with isoflurane and perfused intracardially with cold saline followed by 2% paraformaldehyde. The organs were immersion-fixed in the same solution at 4°C overnight. Fixed tissues were cryoprotected by immersion in 10% sucrose for ≤2 h, 20% sucrose for ≤6 h, and 30% sucrose for ≤18 h before embedding in OCT compound (Cat # 25608-930, Sakura Finetek, USA), and then cut into 7-µm thick sections using a cryostat. Brain samples were stained with cresyl violet solution and infarct volume was calculated as the sum of lesion areas in 5 slices. Other samples, including the brain, were examined with fluorescence immunohistochemistry. The presence of engrafted cells and evaluation of their differentiation into hematopoietic cells was assessed using primary antibodies against GFP (1:500, ab6673, Abcam) and CD45 (1:100, ab10558, Abcam). The samples were incubated with either anti-goat IgG conjugated with Alexa-488 (1:500, A11055, Invitrogen) or anti-rabbit IgG conjugated with Alexa Fluor-568 (1:500, A10042, Invitrogen). Nuclei were counterstained with 4’6-diamidino-2-phenylindole (1:500, D9542, MilliporeSigma). To suppress autofluorescence, we used Trueblack lipofuscin autofluorescence (Cat # 23007, Biotium) according to the manufacturer’s protocol. Images were captured by laser confocal microscopy (FV3000, Olympus).
Splenectomy and adoptive transfer of spleen cells
At 2 weeks after administration of Muse cells or vehicle, mice were anesthetized with 2% isoflurane in a mixture of 30% oxygen and 70% nitrous oxide and placed in the supine position. A small 1-cm incision was made in the left upper quadrant of the abdomen followed by retraction of the spleen and ligation of the splenic hilum. After removing the spleen, the incision was closed with a running suture. The spleens were mechanically dissociated into a single-cell suspension, filtered through a 40-µm filter, and centrifuged at 400 g for 5 min. The pellets were incubated in 3 mL of red blood cell lysis buffer (Cat # 00-4333-57, Invitrogen) on ice for 5 min. The samples were then centrifuged at 400 g for 5 min and resuspended in 2% fetal bovine serum. Isolated splenocytes were used for the adoptive cell transfer experiment. 28 Isolated splenocytes from mice intravenously injected with Muse cells or vehicle (controls) were counted with a hemocytometer using the trypan blue exclusion method. Ten million splenocytes from the donor mice were suspended in 200 µL HBSS and transferred intravenously through the jugular vein 48 h after dMCAO.
Flow cytometry
Flow cytometry was conducted according to previously described methods29,30 with minor modifications. At 2 weeks after administration of Muse cells or vehicle, mice were anesthetized and splenectomy was performed as described above. The spleens were mechanically dissociated into a single-cell suspension, filtered through a 40-µm filter, and centrifuged at 400 g for 5 min. The pellets were incubated in 3 mL of red blood lysis buffer (Cat # 00-4333-57, Invitrogen) on ice for 5 min. The samples were then centrifuged at 400 g for 5 min and resuspended in 2% fetal bovine serum. The number of viable splenocytes in the cell suspension was counted with trypan blue staining in a 96-well plate with 1 × 105 cells/well. Isolated splenocytes were incubated with anti-mouse CD16/32 (Cat # 101320, Biolegend) on ice for 5 min to block nonspecific binding of immunoglobulin to macrophage Fc receptors. Next, to evaluate the lymphocytes, splenocytes were incubated with APC-conjugated anti-mouse CD45 (Cat # 103112, Biolegend), phycoerythrin (PE)-conjugated anti-mouse CD19 (Cat #1 52408, Biolegend), PE-Cy7–conjugated anti-mouse CD3 (Cat # 100220, Biolegend), fluorescein isothiocyanate-conjugated anti-mouse CD4 (Cat # 100406, Biolegend), and Pacific blue-conjugated anti-mouse CD8 (Cat # 100725, Biolegend). To evaluate the myeloid cells, splenocytes were incubated with APC-conjugated anti-mouse CD45, fluorescein isothiocyanate-conjugated anti-mouse Ly-6G (Cat # 127606, Biolegend), PE-conjugated anti-mouse CD11b (Cat # 101208, Biolegend), and APC-Cy7–conjugated anti-mouse CD11c (Cat # 117324, Biolegend). The labeled cell suspension was analyzed using fluorescence-activated cell sorting (FACS Canto II; BD Biosciences, San Jose, CA, USA) with the gating strategy shown in Supplementary Figure 2. All the data were analyzed using Flowjo software 10.7.1 (Three Star, Ashland, OR, USA).
Enzyme-linked immunosorbent assay
Peripheral blood was collected in collection tubes on day 14 after Muse cell administration. Peripheral blood was immediately centrifuged at 15,000 rpm for 15 min at 4°C. The serum was decanted, immediately frozen, and maintained at −80°C until analysis. Serum concentrations of interleukin-10 (IL-10) and transforming growth factor-beta (TGF-β1) were analyzed by enzyme-linked immunosorbent assay (ELISA) using commercially available kits (M1000B and MB100B, R & D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions.
Real-time quantitative polymerase chain reaction
Total RNA was extracted from spleens using an RNeasy Mini kit (Cat # 74104, The Netherlands) according to the manufacturer’s instructions. We confirmed all the samples had an RNA integrity value >7.0 using the Agilent Bioanalyzer 2100. Complementary DNA was synthesized using Superscript IV VILO (Cat # 11756050, Invitrogen) according to the manufacturer’s protocol as previously described with minor modifications.29,30 Real-time quantitative PCR (RT-qPCR) was performed with TaqMan primer probes. The cycling parameters were as follows: hold at 95°C for 20 s, followed by 4 cycles of PCR amplification comprising denaturation at 95°C for 1 s, and annealing and extension at 60°C for 20 s. Specific probes for mouse beta-actin (Actb; Mm00607939_s1), Il-10 (Mm01288386_m1), Tgfb1 (Mm01178820_m1), Il-4 (Mm00445259_m1), tumor necrosis factor-alpha (Tnfa; Mm00443258_m1), interferon-gamma (Ifng; Mm01168134_m1), and Il-1b (Mm00434228_m1) were used. RT-qPCR was conducted with triplicates in each sample. Expression levels of target genes were calculated using the ΔΔCt method. Actb was used as an internal control.
RNA-sequencing
mRNA sequencing was performed using a NEBNext Ultra II Directional RNA Library Prep kit (Cat # E7765S, Biolabs) according to the manufacturer’s instructions. 31 Quality control of libraries was performed using an Agilent DNA 1000 kit. The NEBNext Library Quant Kit was used to quantify the libraries. Libraries were pooled and sequenced on a Hiseq-X Ten System (150 bp, paired-end) by Macrogen Japan. All sequencing experiments were performed with 3 replicates per group. Quality control for Raw fastq files was performed using FastQC. Reads were trimmed using Trimmomatic 32 and the adaptor sequences were removed. Reads were aligned to the mus musculus reference genome (mm10 downloaded from UCSC) using Hisat2. 33 Read counting and transcript assembly were performed using featurecounts 34 and differential gene expression analysis was performed using the Limma-voom package. 35 The criterion for statistical significance of differentially expressed genes (DEGs) was a fold change >1.5 plus a false discovery rate (FDR) <0.05. Volcano plots were created using R. Pre-ranked gene set enrichment analysis was conducted using eVitta. 31 T-distributed Stochastic Neighbor Embedding), heatmap, and Uniform Manifold Approximation and Projection (UMAP) clustering were conducted using Omics Playground V2.8.9 in a local Docker instance.
Statistical analysis
Data are expressed as mean ± standard deviation (SD). Statistical analysis was performed using GraphPad Prism 9.2.0 (GraphPad Software, San Diego, CA, USA). The behavioral assessment was analyzed using repeated-measures analysis of variance with Bonferroni post-hoc tests. FACS analysis and spleen volume were assessed by 1-way analysis of variance followed by Dunnett multiple comparison tests among the Pre-stroke, Vehicle, and Muse groups. An unpaired t-test was used to compare the infarct volume and mRNA expression in the spleen between the Muse and Vehicle groups. All analyses were 2-tailed, and a p-value of less than 0.05 was considered significant.
Results
Intravenous injection of muse cells improves motor function after stroke
First, to test the therapeutic effects of Muse cells in a model of dMCAO, we used a model in which GFP-labeled Muse cells, GFP-labeled non-Muse cells, or vehicle were administered intravenously 2 days after inducing ischemic stroke by dMCAO. The result of the cylinder test demonstrated that the asymmetry score significantly decreased after dMCAO, indicating greater reliance on the unaffected (ipsilateral) limb, in all groups except the sham-operated group. As reported in a previous study, the hemiplegia induced by dMCAO persisted for 8 weeks in the Vehicle-treated group. 36 The asymmetry score gradually increased in the GFP-labeled Muse cell group, and was significantly higher than that in the Vehicle-treated group from 28 days through 56 days post-administration, indicating improvement. No significant difference in the asymmetry scores was detected between the GFP-labeled non-Muse cell group and the Vehicle-treated group (Figure 1(a)). The same experiment was performed using CL2020, a human Muse cell preparation, to confirm the efficacy of a clinical-grade Muse cell product. The asymmetry score was significantly increased in the CL2020 group at 42 and 56 days post-administration compared with the HBSS group (Figure 1(b)). Nissl staining revealed no clear difference in the infarct volume between the GFP-labeled Muse cell-treated and Vehicle-treated groups at 2 days after administration (Figure 1(c)). To determine whether Muse cells contribute to reducing infarct lesions over time, we evaluated infarct volume at day 14 post-stroke and there was no difference between two groups (Supplementary Figure 3).
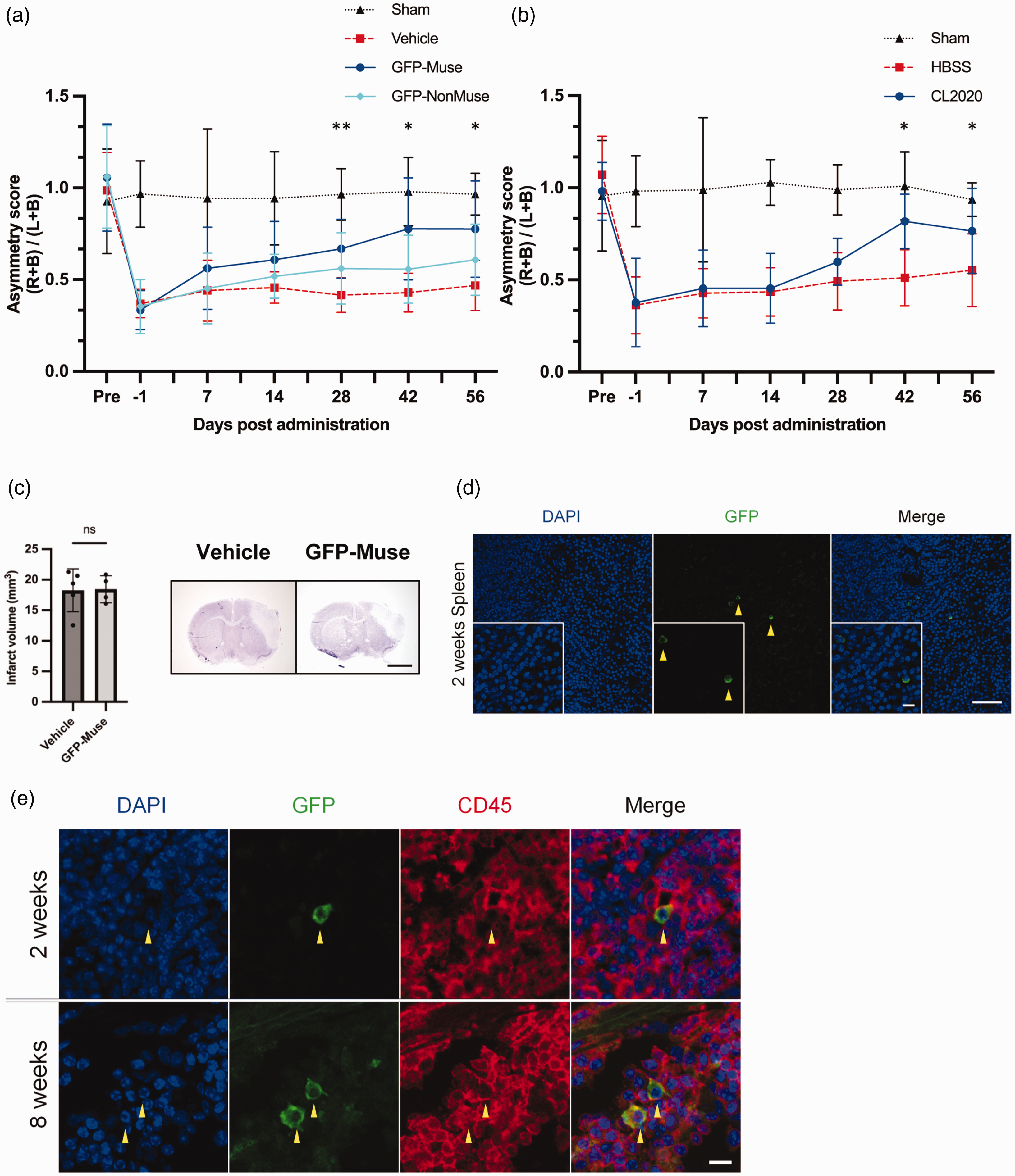
Behavioral assessment, infarct volume, and immunohistology assessment after Muse cell administration. (a) Functional recovery assessed by the cylinder test after intravenous administration of GFP-labeled Muse cells, GFP-labeled non-Muse cells, or vehicle. The asymmetry score of the GFP-labeled Muse cell group is significantly higher than that of the Vehicle group from 28 to 56 days after administration (n = 6 for Sham, n = 12 each for GFP-labeled Muse, GFP-labeled Non-Muse, and Vehicle). Data represent mean ± SD. *p < 0.05, **p < 0.01. (b) Functional recovery assessed by the cylinder test after intravenous administration of CL2020 or HBSS. The asymmetry score of CL2020 is significantly higher than that of the HBSS group at 42 and 56 days after administration (n = 11/group). Data represent mean ± SD. *p < 0.05. B, both left and right; L, left; R, right. (c) Nissl staining of the representative coronal section 1 mm anterior to bregma after dMCAO and Muse cell administration. No difference in the infarct volume was detected between the Vehicle and GFP-labeled Muse cell treatment (n = 4–5). ns: not significant. Scale bar: 2 mm. (d) GFP-labeled Muse cells were detected only in the spleen 2 weeks after intravenous administration. Scale bar 50 µm in the big box and 10 µm in the small box and (e) immunofluorescent study showing that GFP-labeled Muse cells co-stained with leukocyte marker CD45 were not observed 2 weeks after intravenous administration, but were observed 8 weeks after administration. Scale bar 10 µm (n = 3).
The brain and spleen were excised and immunostained at 2- or 8-weeks post-administration to investigate the distribution of the administered Muse cells. In contrast to our previous findings in immunodeficient mice, 19 intravenously administered Muse cells were not detected in the brain in the immunosuppression-free model used in the present study (data not shown), whereas they were identified in the spleen (Figure 1(d)). Intravenously administered Muse cells, which were CD45-negative prior to administration and at 2 weeks post-administration, became CD45 positive in the spleen at 8 weeks post-administration (Figure 1(e)). GFP-positive Muse cells were predominantly located in the red pulp of the spleen rather than in the lymphoid follicles at both 2- or 8-weeks post-administration (Supplementary Figure 4).
The spleen is essential for the protective effects of muse cells against stroke
To evaluate the involvement of the spleen in the functional improvement observed in the Muse cell group, a model was developed in which the spleen was excised 2 days before dMCAO. The outcomes indicated no significant difference in the asymmetry scores of either the GFP-labeled Muse cell- or GFP-labeled non-Muse cell-treated groups compared with the Vehicle-treated group up to day 56 post-administration in this model (Figure 2(a)). A similar trend was observed between the CL2020-treated and HBSS-treated groups (Figure 2(b)). These outcomes suggested that the intravenously administered Muse cells improved neurologic function through a spleen-dependent mechanism. Therefore, an adoptive cell transfer experiment was conducted to determine whether splenocytes stimulated after Muse cell administration could improve neurologic function (Figure 2(c)). Stroke-injured mice intravenously administered splenocytes obtained from the CL2020-treated group exhibited significantly higher asymmetry scores in the cylinder test and a significantly shorter tape removal time in the adhesive tape removal test on the first day following cell transfer compared with stroke-injured mice injected with splenocytes obtained from the HBSS-treated group (Figure 2(d)), suggesting that splenocytes played a crucial role in the neural recovery observed following Muse cell administration after dMCAO-induced stroke.
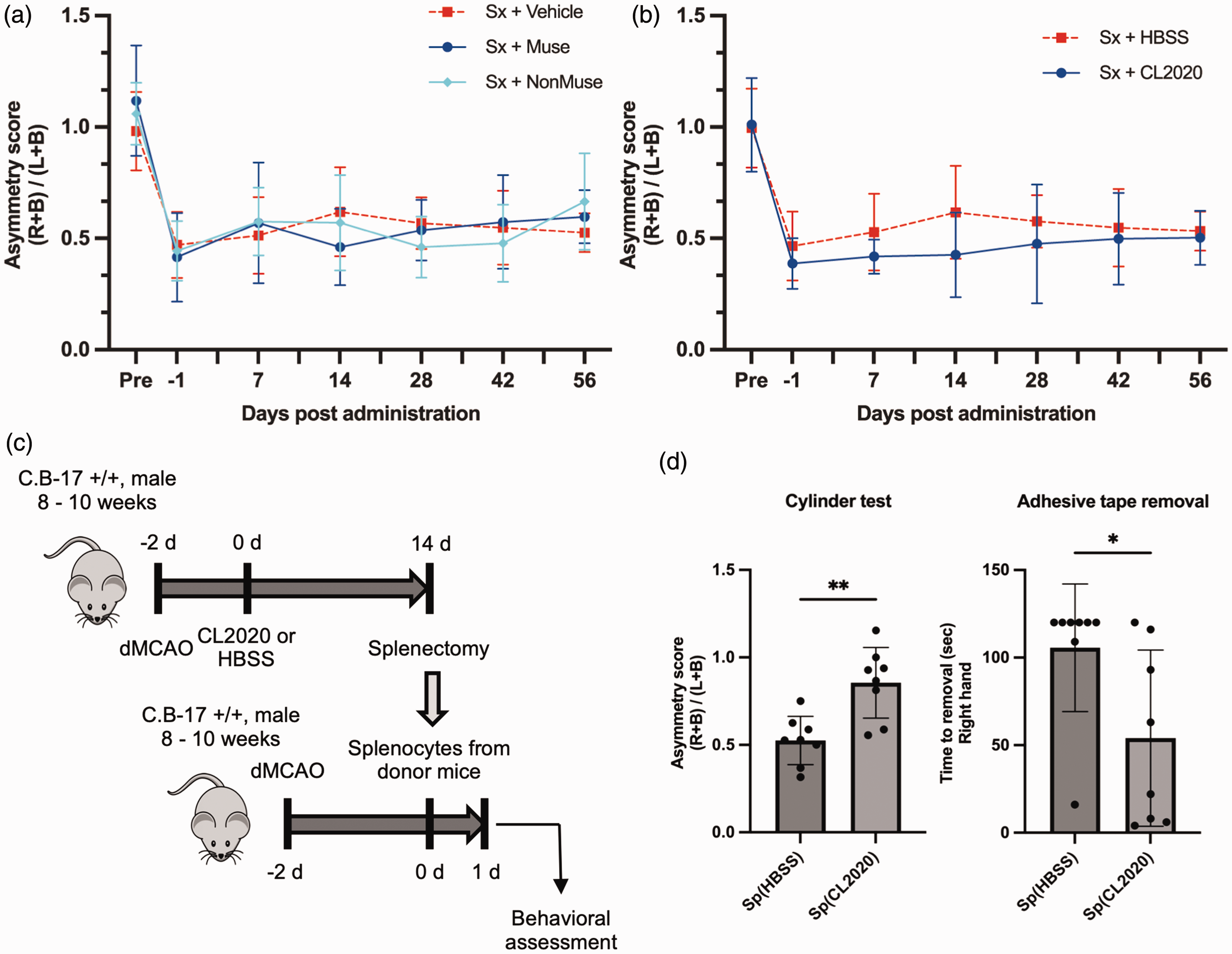
Behavior assessment in splenectomized mice without and with splenocyte transfer. (a) Results of the cylinder test to assess motor function. The mice were splenectomized 2 days before stroke. No long-term improvement in motor function was observed in the Vehicle, Muse cell, and Non-Muse cell groups (n = 12/group). (b) Results of the cylinder test to assess motor function. Similar results were observed following CL2020 and HBSS administration. (n = 11/group). B, both left and right; L, left; R, right; Sx, splenectomy. (c) Scheme of the adoptive cell transfer experiment. d, days and (d) motor function in the recipient mice was evaluated by the cylinder and adhesive tape removal tests. The asymmetry score was significantly higher in the Sp(CL2020) group, and the time to adhesive tape removal was significantly lower in the Sp(CL2020) group (n = 8/group). Sp(HBSS), splenocytes from HBSS-administered mice; Sp(CL2020), splenocytes from CL2020-administered mice. Data represent mean ± SD. *p < 0.05, **p < 0.01.
Muse cell injection induced significant changes in the spleen after ischemic stroke
Spleen weight was predominantly decreased in both the Vehicle and Muse-cell groups at 2 days after injection (4 days after dMCAO) and recovered to normal at 2 weeks after treatment. Spleen weight did not differ significantly between the 2 groups at any of the time points examined (Figure 3(a)). We then examined the resident immune cell types in the spleen using flow cytometry. Spleens extracted from naïve mice were also collected for reference as the “Pre-stroke” value. The mean percentages of CD3+ T cells, CD3+CD4+ T cells, CD3+CD8+ T cells, and CD19+ B cells did not differ significantly among the 3 groups (Figure 3(b)). The percentage of CD11b+Ly6G+ polymorphonuclear neutrophils was significantly higher in the Vehicle group, but not in the Muse group. The percentages of CD11b+CD11c+ classical dendritic cells and CD11b+Ly6G- monocytes/macrophages did not differ significantly among the 3 groups (Figure 3(b)). The results of RT-PCR using RNA extracted from splenocytes showed that Il-10, Tgfb1, Tnfa, and Ifng levels were significantly higher in the Muse group than in the Vehicle group (Figure 3(c)). ELISA analysis of the serum revealed no significant difference in the IL-10 and TGF-β1 levels between the 2 groups (Figure 3(d)).
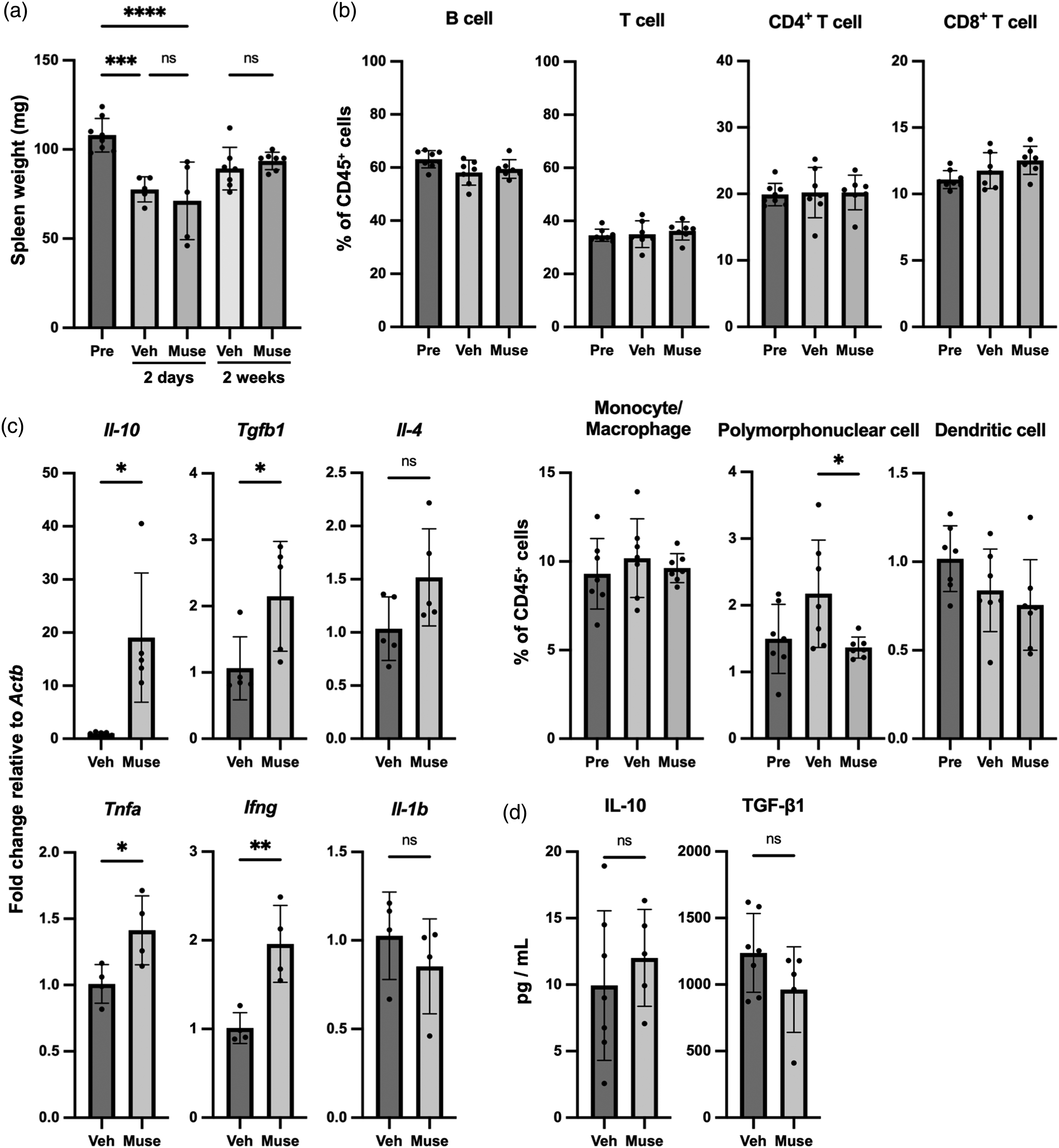
Muse cell effects on the spleen 2 weeks after administration. (a) Spleen weight among 5 groups: Pre-stroke (n = 8), stroke with Vehicle or Muse cell treatment after 2 days (n = 5), stroke with Vehicle or Muse cell treatment after 2 weeks (n = 7/group). Spleen weight decreased in both the Muse cell and Vehicle-treated groups 2 days after stroke and recovered 2 weeks later. No differences were observed between the groups during the entire period. Data represent mean+SD. ***p < 0.001, ****p < 0.0001, ns, not significant. (b) Cell fractionation in the spleen before and 2 weeks after cell administration. The ratio of CD19+ B cells, CD3+ T cells, CD3+CD4+ T cells, CD3+CD8+ T cells, CD11b+Ly6G− monocytes/macrophages, and CD11b+CD11c+ dendritic cells to CD45+ cells did not differ among the 3 groups: Pre-stroke, Vehicle, and Muse cell groups. The percentage of polymorphonuclear cells was significantly lower in the Muse cell group than in the Vehicle group (n = 5/group). *p < 0.05. (c) RT-PCR revealed the upregulation of both pro-inflammatory and anti-inflammatory cytokines in the spleen after Muse cell treatment (n = 4–5). Data represent mean+SD. *p < 0.05, **p < 0.01 and (d) ELISA of the anti-inflammatory cytokines in the serum. No difference was detected between the Vehicle- and Muse cell-treated groups (Vehicle n = 7, Muse n = 5). Pre: Pre-stroke, Veh: Vehicle.
We next conducted bulk RNA sequencing to analyze transcriptomic changes occurring in the splenocytes extracted from Vehicle- or Muse-treated animals post-stroke. We compared Muse-treated animals with Pre-stroke animals (Figure 4(a)), Vehicle-treated animals with Pre-stroke animals (Figure 4(b)), and Muse-treated animals with Vehicle-treated animals (Figure 4(c)). Gene UMAP clustering of the 3 groups revealed unique clustering patterns occurring after Muse or Vehicle treatment when compared with the Pre-stroke group or with each other (Figure 4(d)). We further conducted correlation analysis between the DEGs in the three comparisons (Figure 4(e)). Pearson’s correlation revealed low correlation between the three groups. Such low correlation (r < 0.5) indicates unique gene expression patterns occurring in the Muse or vehicle group, and further indicates that the transcriptional response after Muse treatment is unique. Clustering analysis coupled with gene ontology biologic processes (GOBP) analysis revealed unique clustering patterns in each group (Figure 5(a)). Interestingly, Muse cells had a distinct cluster showing enrichment of terms related to unfolded protein response (UPR) and chaperone function. To further clarify the gene set enrichment divergence in each group, we analyzed UMAP clustering patterns for GOBP enrichment (Figure 5(b)). Again, the Muse group exhibited unique clustering patterns showing enrichment for protein folding and chaperone function, a pattern that was not observed in the Vehicle-treated group. The GOBP activation matrix further clarified these interesting clustering patterns in GOBP enrichment (Figure 5(c)). Pathways related to protein folding and heat-shock response were significantly enriched in the Muse group, whereas pathways linked to immune responses were downregulated in the Muse group. Furthermore, we validated such findings by utilizing another gene set enrichment analysis method. Pre-ranked gene set enrichment for GOBP in Muse vs Pre-stroke (Figure 6(a)), Vehicle vs Pre-stroke (Figure 6(b)), and Muse vs Vehicle (Figure 6(c)) revealed the strong upregulation of pathways related to protein unfolding and chaperone function in the Muse group. While the Vehicle group showed enrichment for chaperone function and protein folding, enrichment of these processes was much stronger in the Muse group compared with the Vehicle group. In addition, we observed a strong downregulation of pathways related to translation, mitochondrial respiration, and bioenergetics in the Muse group compared with the Vehicle group (Figure 6(c)).
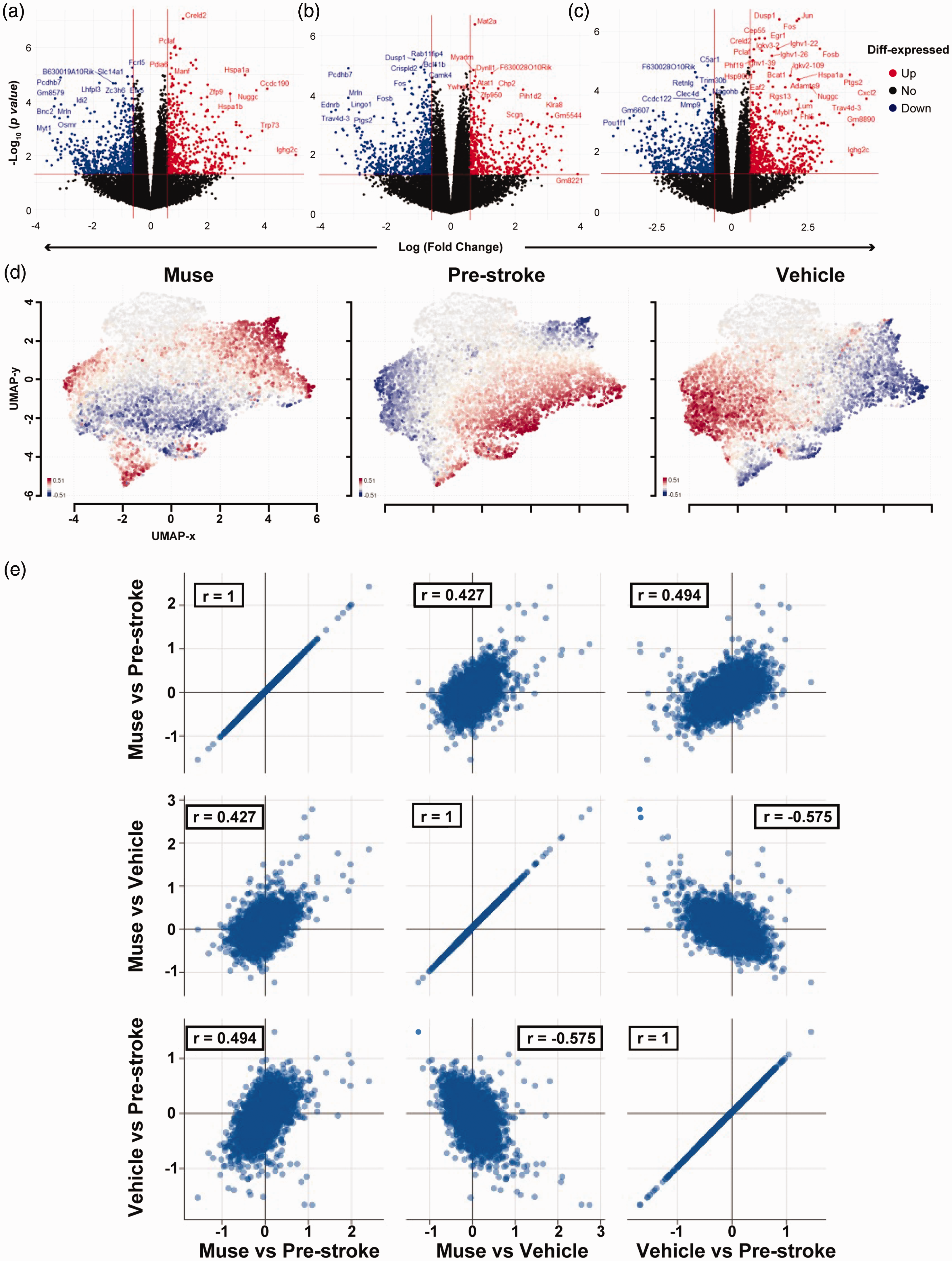
RNA-seq after Muse cell therapy. (a) Volcano plot of differentially expressed genes in Muse cell treatment vs Pre-stroke, (b) Vehicle vs Pre-stroke, and (c) Muse cells vs Vehicle. (d) Gene UMAP clustering of the 3 groups showing unique gene expression patterns in the Muse cell-treated group and (e) Pearson’s correlation analysis among the 3 groups showing modest correlations.
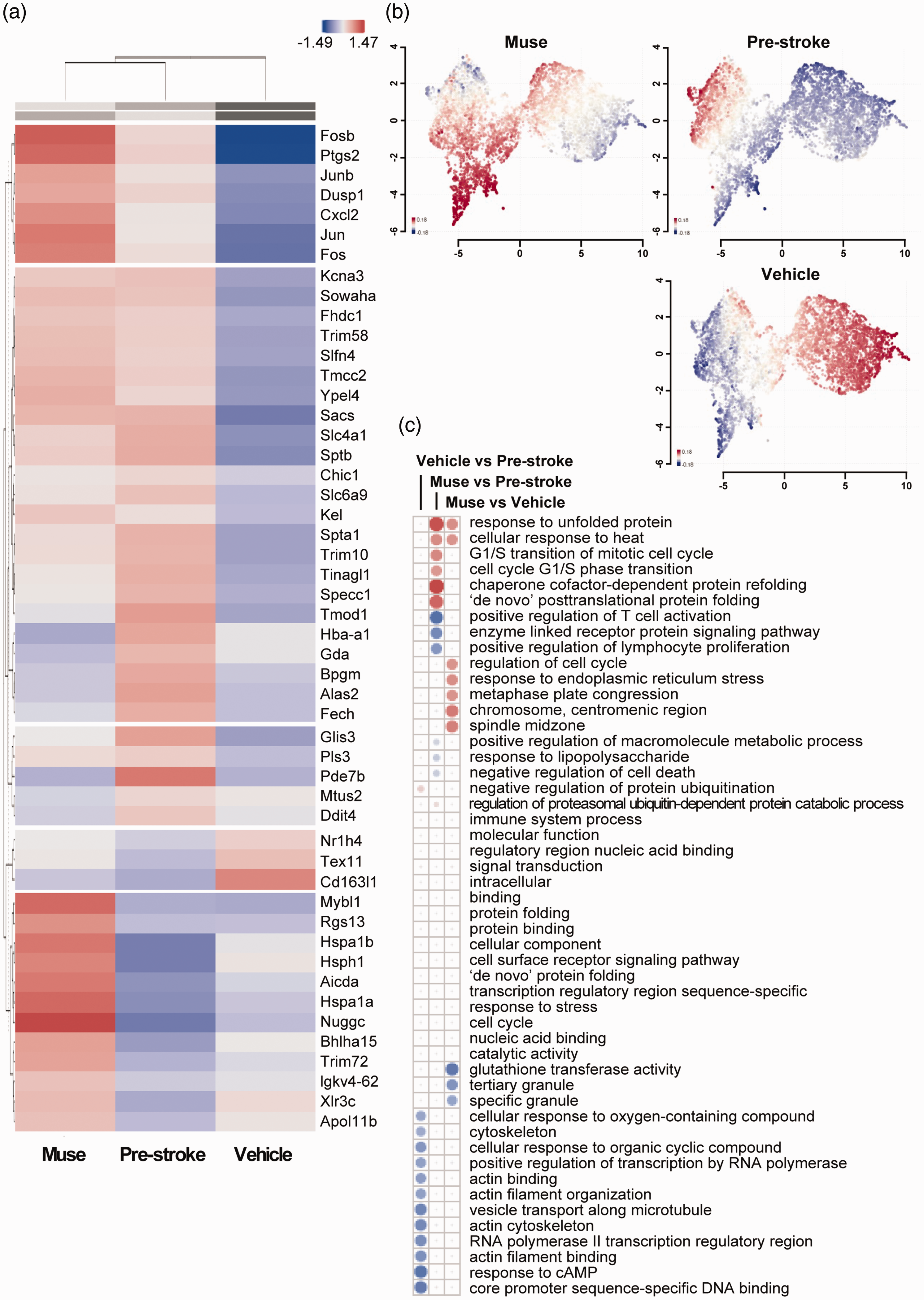
Enrichment of gene ontology biologic processes (GOBP) terms shown in each comparison. (a) Heatmap clustering of the 3 groups. (b) Gene set UMAP clustering showing unique GOBP enrichment in each group and (c) activation matrix for GOBP enrichment showing enrichment of GOBP terms related to unfolded protein response and cell cycle in the Muse cell group vs the other groups but not in the Vehicle vs Pre-stroke comparison.
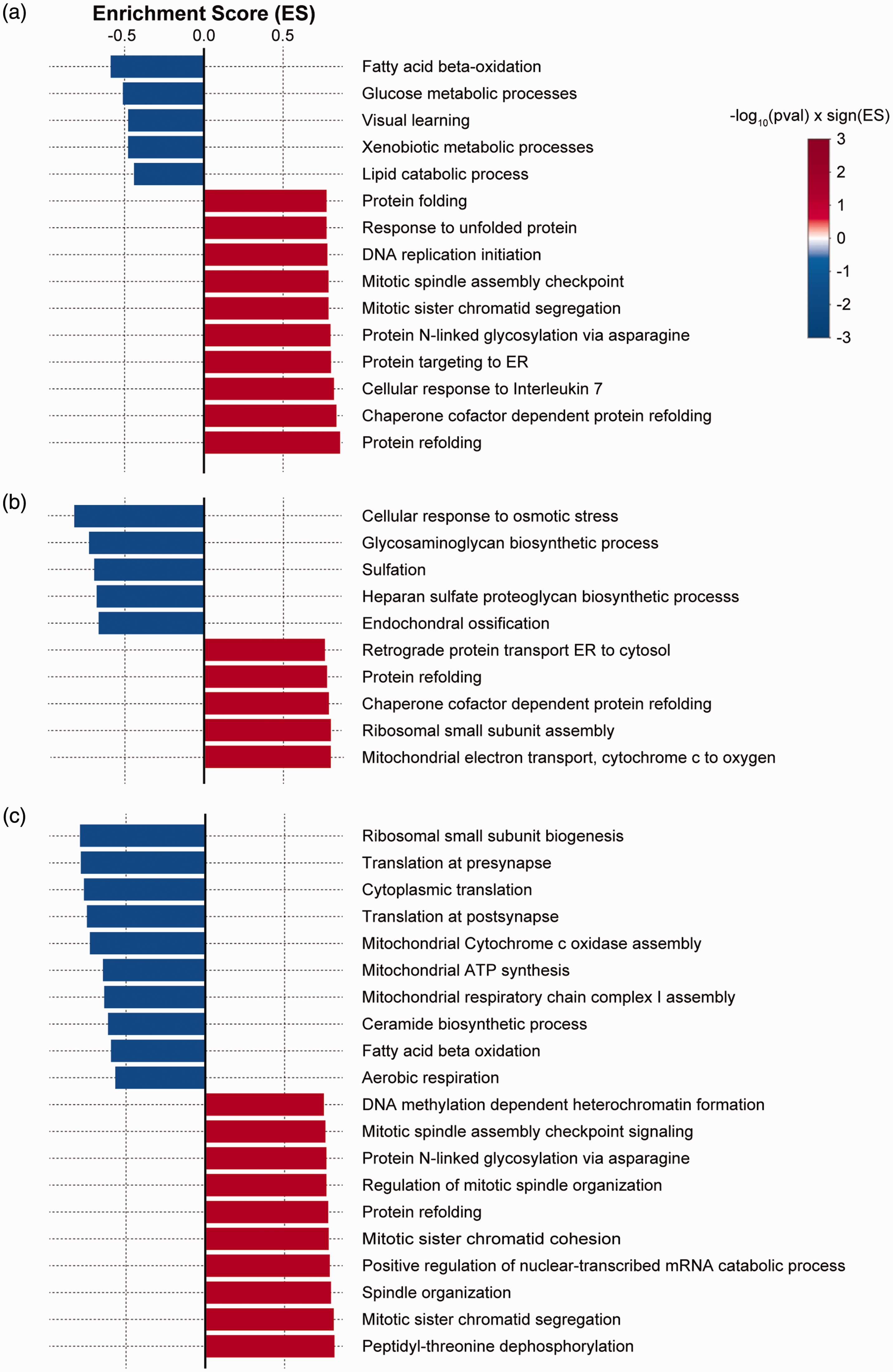
Pre-ranked gene set enrichment analysis. (a) Muse cells vs Pre-stroke, (b) Vehicle vs Pre-stroke and (c) Muse cells vs Vehicle. Upregulation of pathways related to protein unfolding and chaperone function observed in the Muse cell-treated group.
Discussion
The findings of the present study clearly revealed a beneficial role of Muse cells in post-stroke recovery without the need for immunosuppression. Mice administered GFP-labeled Muse cells displayed significantly improved motor function following dMCAO, an effect that was notably absent in mice treated with non-Muse cells or vehicle alone. We corroborated these results using a clinical grade Muse cell preparation, CL2020. In this work, we didn’t observe a reduction of infarct size. This finding supports the notion that the observed functional recovery is not due to reduction in infarct volume but potentially due to other mechanisms such as immunomodulation. In line with our findings, Uchida et al. reported that even with localized brain administration of human-derived Muse cells in an immunosuppressive environment following tMCAO in rats, there was no significant reduction in infarct volume, although motor function improvements were observed. 17 Interestingly, Muse cells were primarily found in the spleen rather than the brain post-administration in this immunosuppression-free model. This organ-specific targeting was further validated by the finding that splenectomy blocked the therapeutic benefits of Muse cells. Conversely, splenocytes transferred from Muse cell-treated mice into stroke-affected mice restored motor function, emphasizing the spleen's integral role in recovery. The rapid functional recovery observed following the injection of Muse cell-treated splenocytes could be explained by immediate immunomodulatory effects. Our data demonstrate that Muse cell-treated splenocytes lead to a reduction in the proportion of neutrophils in the spleen at 2 weeks post administration. Since neutrophils are known to promote inflammation during the acute phase of stroke (especially in 1–3 days post-stroke), 37 their reduction likely mitigates acute inflammation, contributing to the early functional improvements observed. This immediate suppression of acute inflammation by Muse cell-treated splenocytes highlights the potential of Muse cells in modulating early immune responses to promote neural recovery. Additionally, the PCR results showed an increase in both pro-inflammatory and anti-inflammatory cytokine gene expression in the Muse cell-treated group, with the ratio skewed toward anti-inflammatory cytokines. Together, these findings indicate that Muse cells play a vital role in immunomodulation via targeting the spleen, which contributes to improved motor function in post-stroke scenarios. Beyond immunomodulation, the therapeutic effects of Muse cells may also involve other mechanisms, such as mitochondrial transfer or the release of extracellular vesicles, as reported in other stem cell therapies.38,39 These mechanisms represent promising areas for future research to fully understand the multifaceted roles of Muse cells in stroke recovery.
Previous studies highlighted a significant role of the spleen in treatments for cerebral infarction involving intravenous stem cell infusion. For example, Acosta et al. 40 reported that when 400,000 human BM-MSCs were intravenously administered to rats following the induction of cerebral infarction, more cells were observed to accumulate in the spleen than in the brain. Similarly, Yang et al. 41 reported that intravenous administration of ∼3 million human-derived multipotent adult progenitor cells in rats at 24 h following the induction of cerebral infarction not only mitigated spleen shrinkage but also ameliorated the stroke-induced motor dysfunction. Importantly, 3 additional studies produced similar findings using distinct cell types: human umbilical cord blood cells, 42 hematopoietic stem cells, 43 and human amnion epithelial cells. 44 Our study builds on this growing body of evidence by demonstrating that Muse cells also contribute to spleen-mediated immunomodulation in ischemic stroke. This contribution is particularly noteworthy, as it represents the first documentation of such an effect with Muse cells, adding a new layer of complexity to our understanding of their versatile therapeutic potential.
Intravenously administered Muse cells target injured areas in diverse conditions, including myocardial infarction, 45 cerebral infarction, 19 liver fibrosis, 46 and spinal cord injury.24,47 Interestingly, in our study conducted in an immunosuppression-free environment, Muse cells did not gravitate toward the brain, the primary injury site, but instead notably accumulated in the spleen. This pattern is consistent with observations in other models involving aortic aneurysm and focal segmental glomerulosclerosis, where Muse cells also congregated in the spleen;48,49 the role of Muse cells in the spleen, however, was not addressed in these studies. The observation that Muse cells in the spleen express CD45 at 56 days but not at 14 days suggests a time-dependent differentiation process influenced by the local tissue environment. Muse cells are known for their ability to differentiate into all three germ layers (triploblastic lineages) through a unique mechanism involving the phagocytosis of apoptotic cells. 50 It was shown that Muse cells can engulf apoptotic cell fragments, which then release their contents, such as transcription factors, into the cytoplasm of the Muse cells. These factors are subsequently translocated into the nucleus and bind to the stem cell genome, triggering differentiation into specific cell lineages. This differentiation likely includes the acquisition of CD45 expression as Muse cells adapt to the splenic environment and differentiate into specific cell types relevant to immune function.
Moreover, the anti-inflammatory effects of Muse cells are documented in a lung disease model and a perinatal hypoxic-ischemic encephalopathy model.51,52 The different therapeutic effects of Muse cells on damaged tissues, which appear to be influenced by the use of immunosuppression and the type of cell transplantation—xenogeneic or allogeneic—also suggest that the spleen is involved as a possible site for Muse cell accumulation and their subsequent anti-inflammatory activities.
The UMAP clustering and correlation analyses revealed distinct gene expression patterns in Muse-treated animals compared with the Vehicle group and Pre-stroke controls. Notably, muse cell-treated animals exhibited an upregulation of pathways related to protein folding and chaperone function in the spleen. This finding dovetails with existing literature highlighting the critical role of the UPR in stem cell differentiation and survival, particularly in stress conditions. 53 In MSCs, from which Muse cells are derived, the UPR plays a pivotal role in dictating cell fate. For example, the UPR modulates processes such as apoptosis and differentiation into various specialized cell types, including osteoblasts, chondrocytes, and adipocytes. 54 Moreover, the UPR is sensitive to external stressors like palmitate and arsenic trioxide, which induce apoptosis in MSCs in a concentration- and time-dependent manner through endoplasmic reticulum stress pathways. 55 Thus, the UPR serves as a dynamic regulatory mechanism, allowing MSCs to either mitigate stress and survive or, under persistent stress conditions, undergo apoptosis. This adaptability ensures that only the fittest cells contribute to tissue regeneration and homeostasis. The enrichment for UPR and chaperone function pathways in the Muse-treated group is particularly noteworthy. Given the role of the UPR in cellular homeostasis and adaptive stress responses, our observations could explain how Muse cells, despite not migrating to the brain, exert beneficial effects via spleen-mediated immunomodulation. The UPR's adaptability allows it to serve as a dynamic regulatory mechanism, particularly relevant to the plasticity and differentiation potential of Muse cells suggesting that Muse cells are actively engaging in tissue-specific differentiation processes in the spleen. This underscores the capacity of Muse cells to reprogram the molecular landscape of the spleen, potentially leading to the observed therapeutic effects post-stroke. Additionally, RNA sequencing data revealed that genes involved in translation and mitochondrial function were significantly downregulated in the Muse cell-treated group compared to the Vehicle group. This includes genes associated with mitochondrial ATP synthesis and mitochondrial cytochrome c oxidase assembly. By reducing the demands on mitochondrial activity, Muse cells might alleviate cellular stress and contribute to their therapeutic benefits. This mechanism is supported by studies on MSCs, which have demonstrated similar effects in various models of injury.7,8 MSCs harbor protective roles on injured cells from mitochondria-related apoptosis and oxidative damage by reducing the release of cytochrome c into the cytoplasm. 56 Additionally, MSCs secrete a series of cytokines and growth factors that upregulate anti-apoptotic proteins (BCL-XL, BCL-2) and downregulate pro-apoptotic proteins (BAX, BAK, BAD), thereby minimizing mitochondrial injury. 57 These findings suggest that by modulating mitochondrial function and reducing cellular stress, Muse cells can provide significant therapeutic benefits. Surprisingly, these pathways were not among the top downregulated pathways in the Muse vs Pre-stroke comparison, indicating that the downregulated pathways in the Muse group might contribute to the observed therapeutic effects. Importantly, upregulation of heat-shock response proteins and chaperones, and downregulation of translation and ribosome biogenesis indicate significant changes occurring at the level of mRNA translation and protein synthesis that affect spleen function in the Muse group and may account for the observed effects. These translational changes can also explain the disagreement between our qPCR and ELISA results. These effects, coupled with the changes in mitochondrial bioenergetics, indicate significant changes in the transcriptional, translational, and metabolic landscape of the spleen following Muse cell therapy post-stroke that require further analysis.
Despite our study's compelling findings, there are some limitations. First, a thorough quantitative assessment of Muse cell distribution across different organs other than the brain and spleen was not performed. Second, the role of the Muse cell interactions with other spleen-based immune cells, especially regarding the increased presence of neutrophils, remains unclear. While the current study confirmed the presence of intravenously administered Muse cells in the spleen, their detailed behavior, interactions with existing immune cells, and proliferation post-administration require further investigation. Third, the stroke model we employed was a dMCAO, which results in a relatively limited infarct area compared to the commonly used transient MCAO model. This model primarily affects the somatosensory area, but also extends to part of the motor area. Previous report has shown that the cylinder test can evaluate both motor and sensory function deficits. 36 Larger infarctions or ischemia/reperfusion phenomenon may trigger more dynamic immune responses. Therefore, it is necessary to further investigate the behavior and mechanisms of Muse cells in other infarct models. Last, while our RNA sequencing data provide initial insights, corroborative molecular validation is still needed. Given the broad therapeutic applications of Muse cells, further research is crucial to gain a more complete understanding of their roles and mechanisms in the context of cerebral infarction.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241290363 - Supplemental material for Intravenous administration of muse cells improves cerebral ischemia outcome via immunomodulation in the spleen
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241290363 for Intravenous administration of muse cells improves cerebral ischemia outcome via immunomodulation in the spleen by Yuya Kato, Daiki Aburakawa, Ryosuke Tashiro, Yuan Zhou, Sherif Rashad, Hidenori Endo, Teiji Tominaga and Kuniyasu Niizuma in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Data availability statement
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Japan Society for the Promotion of Science Grant-in-Aid for Scientific Research B (20H03560) and a joint research grant from Life Science Institute Inc.
Acknowledgements
We thank Natsumi Konno, Marisa Ota, Azusa Kuroishi, and Lu Lu for invaluable technical support.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Niizuma is affiliated with the Graduate School of Biomedical Engineering, Tohoku University, and Dr. Tominaga is affiliated with the Tohoku University Graduate School of Medicine, which were parties to a joint research agreement with Life Science Institute Inc. Dr. Niizuma had a contract for a clinical trial agreement with Life Science Institute Inc. Dr. Tominaga received consulting fees from Life Science Institute Inc. Dr. Tominaga holds a patent for the application of Muse cells for the treatment of cerebral infarction. The other authors report no conflicts.
Authors’ contributions
Y.K., D.A., R.T., Y.Z., S.R., H.E., T.T., and K.N. participated in the interpretation of study results, and in the drafting, critical revision, and approval of the final version of the manuscript. Y.K., D.A., R.T., S.R., and K.N. were involved in the study design. Y.K., D.A., and Y.Z. contributed to data collection. Y.K. and S.R. conducted the statistical analyses. All authors had full access to all the data in this study and take final responsibility for the decision to submit this report for publication.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.