Abstract
We performed a systematic review and meta-analysis on prospective studies that provided risk estimates for the impact of 3 different MRI markers of small vessel disease (SVD), namely white matter hyperintensities (WMH), cerebral microbleeds (CMB) and lacunes, on cognitive decline in relatively healthy older adults without cognitive deficits at baseline. A total of 23 prospective studies comprising 11,486 participants were included for analysis. Extracted data was pooled, reviewed and meta-analysed separately for global cognition, executive function, memory and attention. The pooled effect size for the association between cerebral SVD and cognitive decline was for global cognition −0.10 [−0.14; −0.05], for executive functioning −0.18 [−0.24; − 0.11], for memory −0.12 [−0.17; −0.07], and for attention −0.17 [−0.23; −0.11]. Results for the association of individual MRI markers of cerebral SVD were statistically significant for WMH and global cognition −0.15 [−0.24; −0.06], WMH and executive function −0.23 [−0.33; −0.13], WMH and memory −0.19 [−0.29; −0.09], WMH and attention −0.24 [−0.39; −0.08], CMB and executive function −0.07 [−0.13; −0.02], CMB and memory −0.11 [−0.21; −0.02] and CMB and attention −0.13 [−0.25; −0.02]. In conclusion, presence of MRI markers of cerebral SVD were found to predict an increased risk of cognitive decline in relatively healthy older adults. While WMH were found to significantly affect all cognitive domains, CMB influenced decline in executive functioning over time as well as (in some studies) decline in memory and attention.
Introduction
The prevalence of neurodegenerative diseases such as dementia is rapidly increasing due to the increase of the ageing population. Cerebral small vessel disease (SVD) is the most common pathology underlying vascular cognitive impairment. 1 SVD is associated with lesions in the brain commonly identified in neuroimaging studies of persons above the age of 60 years. 2 SVD can be either familial (rare) or more commonly non-familial (sporadic). Whilst the aetiology of sporadic SVD has been attributed to the aging process, these lesions have been suggested to represent pathological changes in the brain that are variable in nature and severity. 3 SVD has been associated with neurological impairments in motor function and cognition 4 and with an increased incidence of neurological disorders such as stroke and dementia. 5
Brain magnetic resonance imaging (MRI) markers of SVD comprise three main types, being white matter hyperintensities (WMH), cerebral microbleeds (CMB), and lacunes.6,7 WMH have a diffuse pattern and may reflect underlying loss of oligodendrocytes and demyelination or deterioration of white matter fibre tracts. 8 Cerebral microbleeds are small focal lesions that are caused by leakage of blood components from vessels resulting in a small area of focal hemosiderin deposition.9,10 A lacune is typically a lesion of less than 15 mm in diameter that can affect both the subcortical white matter as well as the deep grey matter of the brain and brainstem. 11
Increased sporadic SVD load is associated with diffuse loss of white matter and cortex; not only in connected, but also in unrelated brain areas.12,13 Due to the neurovascular changes caused by SVD, the cognitive function associated with the affected brain region may be affected.10,13 Whilst individuals with SVD may have no symptoms, increasing SVD load has been associated with varying levels of cognitive dysfunction such as cognitive impairment or dementia. 13 A systematic review of SVD and risk of Alzheimer’s disease found that SVD markers are associated with clinical dementia pathology, but the exact correlation between SVD markers and Alzheimer’s disease pathology is unclear. 14 Presence of SVD prior to stroke has been causally implicated in the risk of post-stroke cognitive impairment and dementia. 15 However, although several recent reviews have been performed regarding the effect of SVD and cognition in subjects with pathologies such as hypertension and stroke,16,17 to the best of our knowledge, to date no other review has specifically quantified the effect of SVD on cognitive decline in healthy persons without cognitive deficits at baseline. Furthermore, the longitudinal relationship between sporadic SVD and declines in specific cognitive domains (executive function, memory and attention) in cognitively intact older adults remains unclear. This knowledge is important as it can guide the choice for biomarkers in future lifestyle intervention studies or future treatment studies aimed at prevention of cognitive decline in healthy older adults.
In this study, we aimed at quantifying the effects of individual markers of sporadic SVD on cognitive decline in healthy older adults without cognitive deficit at baseline. We performed a systematic review and meta-analysis of available evidence from longitudinal studies on the association between MRI markers of cerebral SVD (WMH, CMB and lacunes) and cognitive decline in healthy older adults without cognitive impairment at baseline. Pooled analysis was conducted for studies investigating the role of global SVD (either WMH, CMB or lacunes) in cognitive decline in the cognitive domains of (1) global cognition, (2) attention, (3) executive function and (4) memory to assess the potential difference in outcomes relating to the different types of SVD and the separate cognitive domains. For each cognitive domain we performed additional meta-analyses for each MRI marker of SVD, being WMH, CMBs and lacunes, when sufficient studies were available per subgroup.
Material and methods
This systematic review and meta-analysis was done according to the 2009 PRISMA-guidelines.
Data sources and search strategy
We conducted a systematic literature search on 30-06-2023 on the association between sporadic cerebral small vessel disease and cognitive decline. PubMed, Embase, Web of Science, Cochrane Library, Emcare, PsycINFO and Academic Search Premier were searched. The design of the electronic search strategy was done in consultation with an expert reference librarian (J.W.S.). To avoid missing any relevant study in the search, broadly defined medical subject heading terms were used (see Supplemental data Appendix A1). Snowballing was applied by searching the reference list of identified key articles for relevant articles that could otherwise have been missed.
Inclusion and exclusion criteria
Extracted citations were screened for eligibility by two independent reviewers (A.J. and A.A.A.). To maximise the quality and comparability of the studies, general inclusion and exclusion criteria were defined beforehand (Table 1). To be included, studies had to be original studies of longitudinal study design with sporadic SVD as determinant and cognitive functioning as outcome variable. Furthermore, quantitative neuropsychological testing and brain imaging measures had to be available. Patients needed to have normal cognitive function at baseline and had to be free of co-morbidities related to cognitive decline other than cerebral SVD. A median patient age of 60 years or more was required. The screening process that was used to obtain the final study selection used in this review is provided in a flowchart (Figure 1).
Selection criteria for eligibility for inclusion or exclusion.
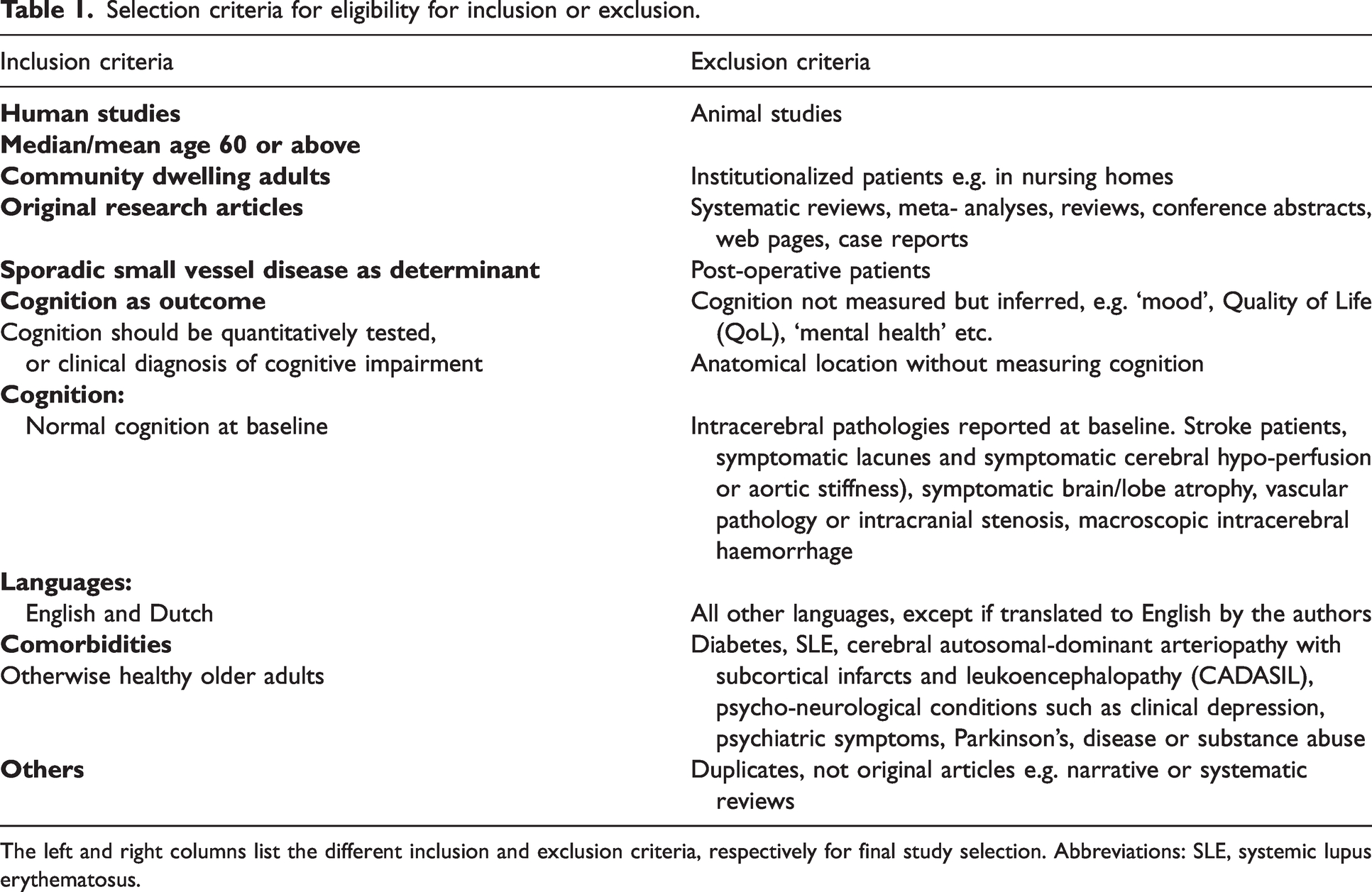
The left and right columns list the different inclusion and exclusion criteria, respectively for final study selection. Abbreviations: SLE, systemic lupus erythematosus.
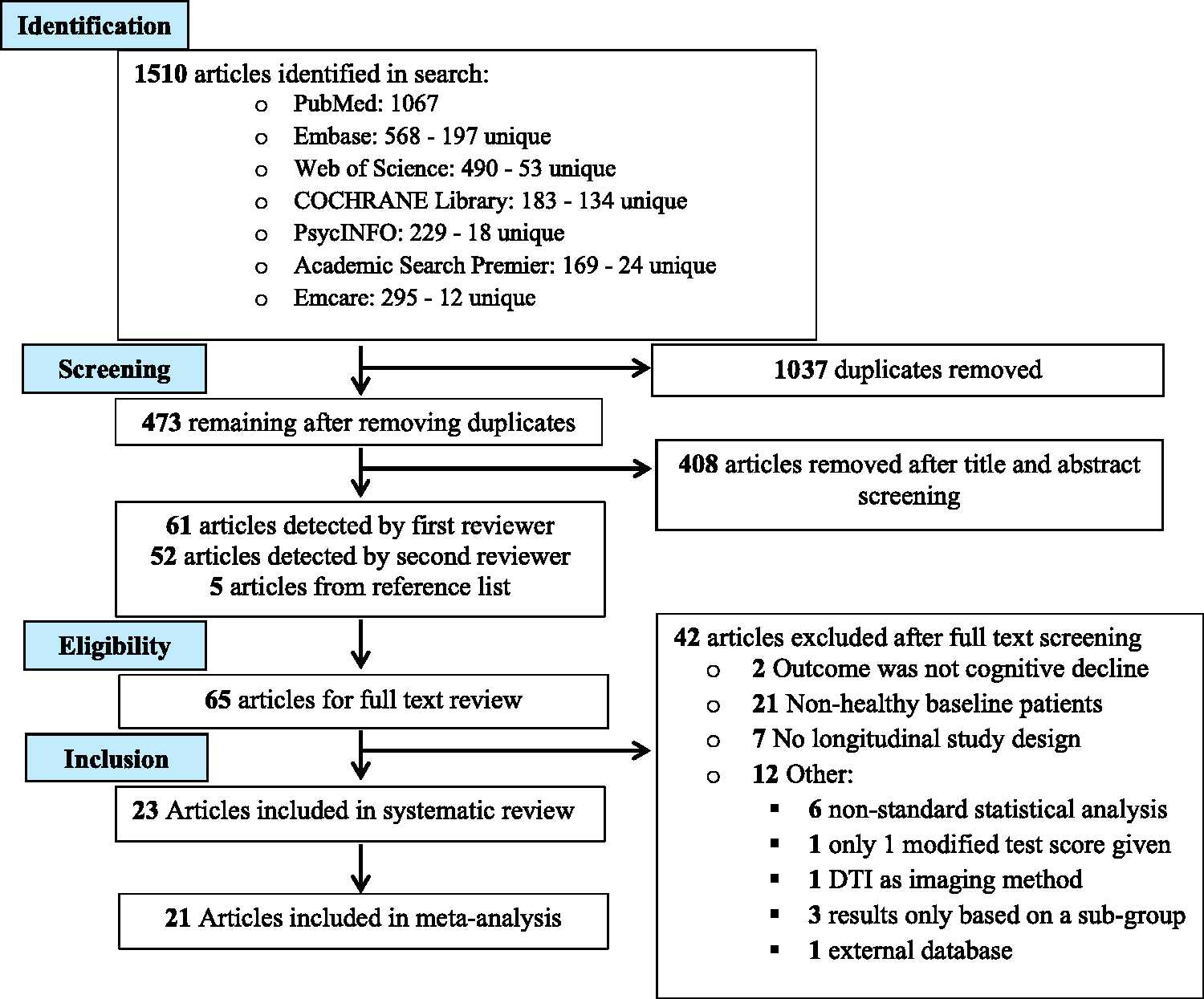
PRISMA flow diagram for the systematic review and meta-analysis on SVD and cognition describing the flow of study identification and selection. Database searches identified in total 1510 articles. After duplicates were removed, there were 473 unique articles eligible for title and abstract screening. After this first phase of screening, 65 articles were left to assess for eligibility by screening of the full-text articles. After this second phase of screening, 23 articles were included in the systematic review, and 21 articles were included in the meta-analysis.
Data collection process
For each included study, we extracted data concerning the total number of subjects, MRI methods used, patient (sub-)groups, follow-up duration and mean patient age, SVD types included, baseline cognition, analysed cognitive domains and neuropsychological test outcomes at study baseline and follow-up. A summary is presented in the results. Classifications and descriptions of the neuropsychological tests used within the scope of articles included in this review can be found in Supplementary tables A1 and A2, respectively. When such data was not available, we requested this from the study authors. As specified in the Acknowledgement section, requested information was received in the majority of cases.
Risk of bias in individual studies
To determine the risk of bias in individual studies we performed a quality scoring of each included study (Supplemental data Table A3) according to 17 study characteristics, which included a follow-up period of at least 2 years, a responder rate of follow-up of at least 75%, availability of multiple neuropsychological tests and statistical adjustment for confounders. Furthermore, a full patient-characteristics table, information on responders and non-responders and differences between subjects with complete and non-complete data was preferably available. We also determined whether the diagnosis of cognitive impairment of individual study subjects was made according to well-established criteria 18 and whether patients with baseline cognitive impairment were excluded.
Data synthesis and statistical analysis
For each of the cognitive domains, different tests were used encapsulating different cognitive abilities. We then extracted or calculated standardized effect estimates for each study test outcome categorized per cognitive domain according to the respective neuropsychological test performed. The most adjusted estimates were used for analysis.
When only direct test scores were provided, we calculated the standardized effect size based on the mean test score difference for baseline versus follow-up by using the “Practical Meta-Analysis Effect Size Calculator”. 19
If needed, we transformed hazard ratios (HR) or risk ratios (RR) into standardized effect sizes by using the formula:
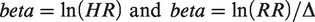
Once all outcomes were standardized, we conducted a random-effects meta-analysis using the metafor package in R (http://www.metafor-project.org/). 20
Pooled analysis was conducted for studies investigating the role of SVD in cognitive decline in the cognitive domains of (1) global cognition, (2) attention, (3) executive function and (4) memory. For each cognitive domain we performed a meta-analysis on the effects of global SVD (any WMH, CMB or lacune reported result) and additional meta-analysis on each separate MRI sub-marker of SVD, when sufficient studies were available per subgroup to perform a meta-analysis (minimum of 4).
We calculated the pooled effect sizes and corresponding confidence intervals using the ‘rma’ package, where the DerSimonian-Laird estimator was used for determining heterogeneity I2 scores. I2 values of <25% were considered reflective of low, between 25–50% of moderate and >50% of high heterogeneity between studies. We used the ‘forest’ function to create forest plots to visualize the meta-analysis results.
Assessment of quality of included studies
Quality scoring included 17 total criteria and a score of either 0 or 1 was given for each element having a qualitative effect on the meta-analysis and serving as a basis for the inclusion of the respective study into our systematic review. Elements include time of follow-up, a well-defined hypothesis, exclusion of baseline dementia and the breadth of cognitive testing. The full list of characteristics evaluated can be found in Supplementary data Table A3: Quality Scoring.
Risk of bias across studies
To determine risk of bias across studies we assessed studies for selective reporting of results and the risk of missing or inappropriate data by verifying whether all included cognitive domain neuropsychological testing scores per type of SVD, were provided at both baseline and follow-up. Furthermore, we assessed the risk of publication bias by making use of funnel plots made using the metafor package in R (Supplemental data Figures A1–4). Individual funnel plots were made for the log odds ratio in relation to the standard error using a fixed-effects model which included the total number of individual study participants and the (calculated) effect sizes.
Results
Study selection
We identified a total of 1510 studies from our search of scientific literature databases. After removing duplicates, we screened 473 titles and abstracts for relevance which resulted in the inclusion of 65 articles for full-text review. A total of 23 articles were included in this systematic review and 21 for the meta-analysis. The study flow chart is provided in Figure 1.
Study characteristics
Table 2 provides a detailed overview of the individual study characteristics and results of the 23 included longitudinal studies, which comprised a total of 11,486 participants with sporadic SVD. Mean age of included participants was 75 years. Five of the included studies measured all SVD markers, 12 studies measured WMH only and four studies measured CMB only. Four studies measured all cognitive domains (global, memory, attention, executive function). The average duration of follow- up was 5.7 years (range 2–30 years). Baseline cognition of included subjects ranged between studies from MMSE 25.7 ± 3.0 to 29 ± 1.4.
Characteristics of included studies.
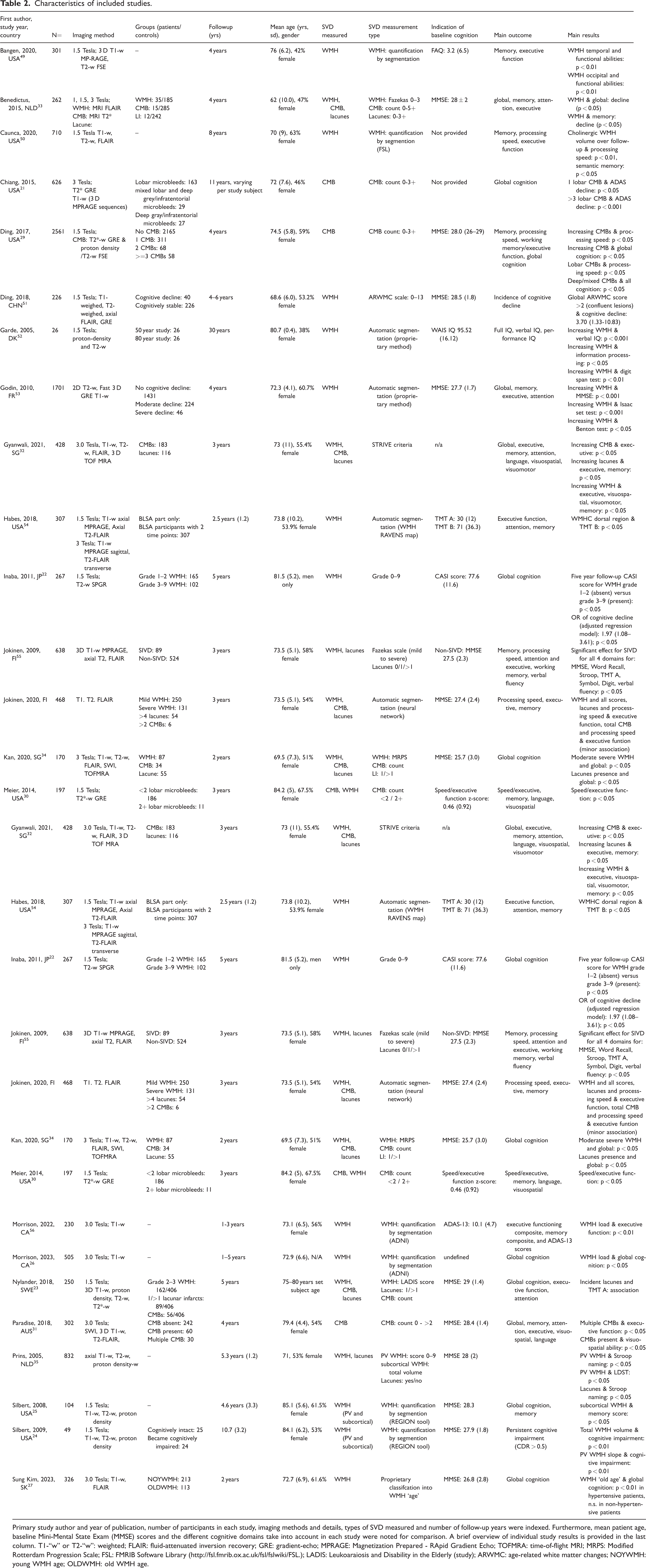
Primary study author and year of publication, number of participants in each study, imaging methods and details, types of SVD measured and number of follow-up years were indexed. Furthermore, mean patient age, baseline Mini-Mental State Exam (MMSE) scores and the different cognitive domains take into account in each study were noted for comparison. A brief overview of individual study results is provided in the last column. T1-“w” or T2-“w”: weighted; FLAIR: fluid-attenuated inversion recovery; GRE: gradient-echo; MPRAGE: Magnetization Prepared - RApid Gradient Echo; TOFMRA: time-of-flight MRI; MRPS: Modified Rotterdam Progression Scale; FSL: FMRIB Software Library (http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/FSL.); LADIS: Leukoaraiosis and Disability in the Elderly (study); ARWMC: age-related white matter changes; NOYWMH: young WMH age; OLDWMH: old WMH age.
Quality scoring and risk of bias
A quality scoring of each included study based on 17 predefined quality criteria (0/1 score) is available in Supplemental data Table A3. The quality scores of included studies ranged from 11–17 (maximum possible score of 17). Most studies reported extensive neuropsychological test scores or compound z- or beta-coefficients The remaining studies21 –28 provided alternative statistical outcome measures. We found a minor indication of the presence of publication bias in the sub-analysis for decline in attention (Supplemental data Figure A2).
Systematic review
A total of 15 of the 23 studies had a response rate higher than 75%. In total, 10 of the 23 studies had a lost-to-follow-up lower than 20%. All studies performed neuropsychological testing and multiple cognitive tests were done in 21 of the 23 studies. A brief summary of the main results of each study is presented in the last column of Table 2.
Data of 21 of the 23 studies were pooled and meta-analyzed. The studies by Morrison et al. 26 and Sung Kim et al. 27 provided calculations and results in a format that could unfortunately not be pooled with the rest of the data. To briefly summarize, the study by Morrison et al. 26 reported that a decrease in cognitive function was significantly higher in patients with subjective cognitive complaints at baseline, with concomitant higher WMH load, compared to those without (Cognitive Change Index, p < 0.05; Everyday Cognition Scale, p < 0.05). The study by Sung Kim et al. 27 categorized participants in two groups, namely an ‘old’ and ‘young’ WMH age calculated according to a proprietary algorithm, and found that ‘old’ WMH age in the sub-group with hypertension significantly correlated with decreased global cognitive performance at follow-up on the Mini Mental State Exam (p < 0.01), The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) neuropsychological battery (p < 0.01) and Frontal Assault Battery (p < 0.05) assessments, but not in non-hypertensive patients.
WMH
All 19 included studies on the relation between WMH and cognitive decline consisting of a total of n = 8,000 participants showed that greater WMH burden was consistently associated with cognitive decline.
CMBs
A total of 9 studies consisting of n = 5,264 participants studied the relation between CMBs and cognitive decline. Of these, 6 showed a significant association.21,28 –32 The study by Benedictus et al. 33 found no effect of presence of 1, 2–4 or ≥5 CMB’s on cognitive performance over time on any cognitive domain. The study by 34 found no association between CMB’s and increase in the neuropsychiatric inventory score (NPI), an independent predictor of cognitive decline. The study by Nylander et al. 23 found no relation between presence of CMB’s (n = 56 of total study population of N = 406) and three cognitive tests.
Lacunes
A total of 7 studies consisting of n = 3,048 participants of whom 684 subjects had confirmed lacunes studied the relation between lacunes and cognitive decline. Of these, five studies provided negative correlations between lacunes and cognition in the domains of executive and memory functioning, 32 processing speed and executive functioning, 28 global cognition, 34 executive function 23 and attention. 35
Meta-analysis
To quantitatively assess whether MRI markers of SVD were associated with cognitive decline in various domains we performed a meta-analysis of study effect estimates per cognitive domain.
Any SVD marker
The pooled effect of any SVD marker on global cognition, expressed as a weighted summary estimate from a linear mixed-effects model, was −0.10 [−0.14; −0.05] (p < 0.01) with a heterogeneity (I2) of 83% (Figure 2(a)). The pooled estimate of the effect of any SVD marker on decline in attention was −0.17 [−0.23; −0.11] (p < 0.05) with an I2 of 95% (Figure 2(b)). The pooled estimate of the effect of any SVD marker on decline in executive functioning was −0.18 [−0.24; −0.11] (p < 0.001) with an I2 of 97% (Figure 2(c)). The pooled estimate of the effect of any SVD on decline in memory was −0.12 [−0.17; −0.07] (p < 0.001) with an I2 of 93% (Figure 2(d)). These results indicate that all cognitive domains are negatively affected by increased general presence of MRI marker of cerebral SVD. However, we also found differences between SVD subgroups, outlined below.
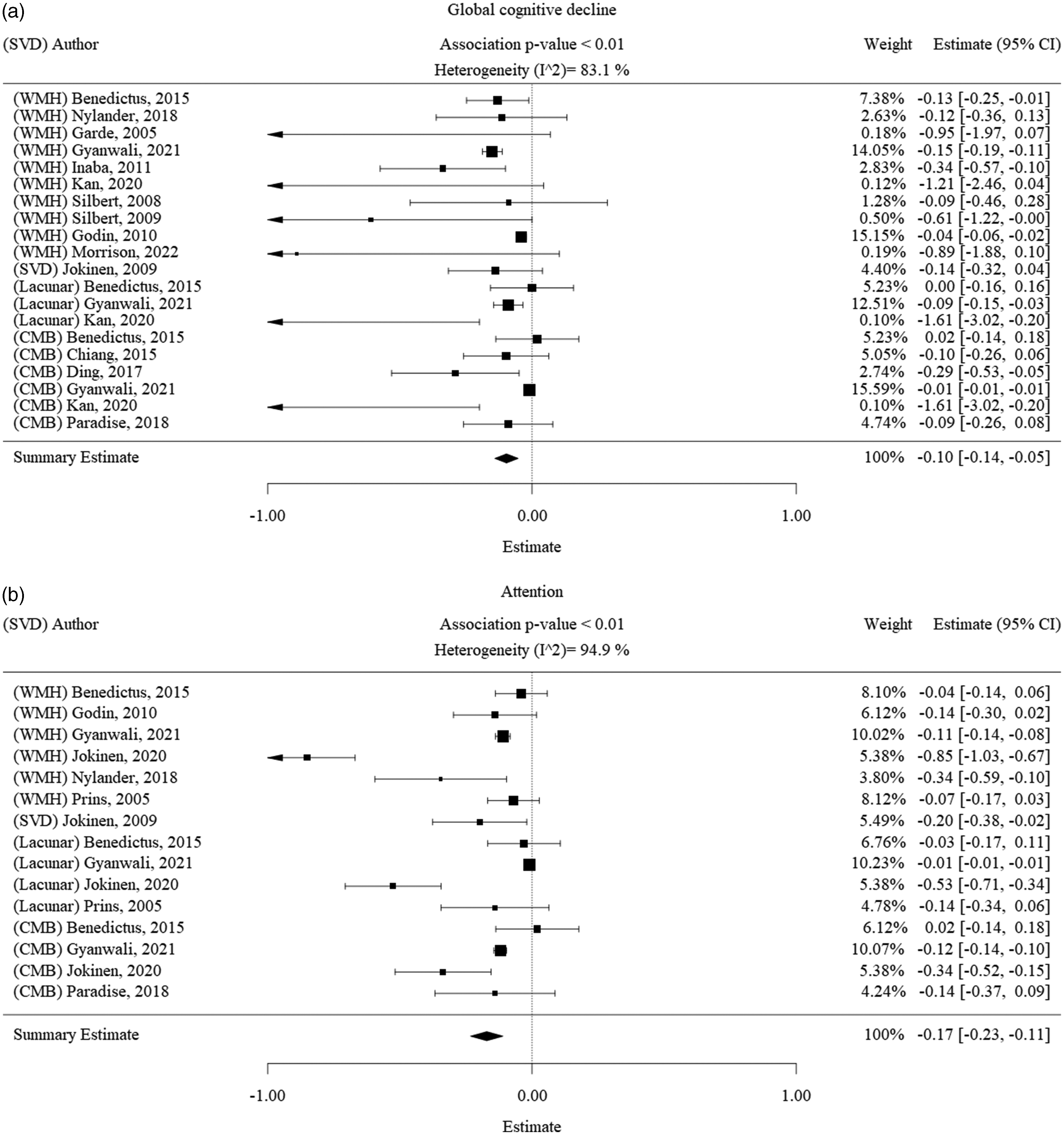
Forest plot of the association between SVD with cognitive decline. Results of the main meta-analysis visualized as forest plots, displaying the effect size per included study and the summary effect estimate for the presence of any SVD in relation to each cognitive domain, namely a.) SVD and global cognitive decline, b.) SVD and decline in attention, c.) SVD and decline in executive function and d.) SVD and decline in memory function. Squares indicate individual study effects. Size of the squares is proportional to the relative study weight. Bars indicate confidence interval of the study estimate. Diamond indicates the summary effect whereas the outer edges of the diamond indicate the confidence interval. The scale of the estimate ranges from −1 to +1 where −1 indicates high cognitive decline and +1 indicates high cognitive improvement.
WMH
The pooled estimate of the effect of WMH on global cognition was −0.15 [−0.24; −0.06] (p < 0.001) with a heterogeneity of 80% (Figure 3(a)). The pooled estimate of the effect of WMH on attention was −0.24 [−0.39; −0.08] (p < 0.005) with a heterogeneity of 93% (Figure 3(b)). The pooled estimate of the effect of WMH on executive function was −0.23 [−0.33; −0.13] (p < 0.001) with a heterogeneity of 97% (Figure 3(c)). The pooled estimate of the effect of WMH on memory was −0.19 [−0.29; −0.09] (p < 0.001) with a heterogeneity of 93% (Figure 3(d)).
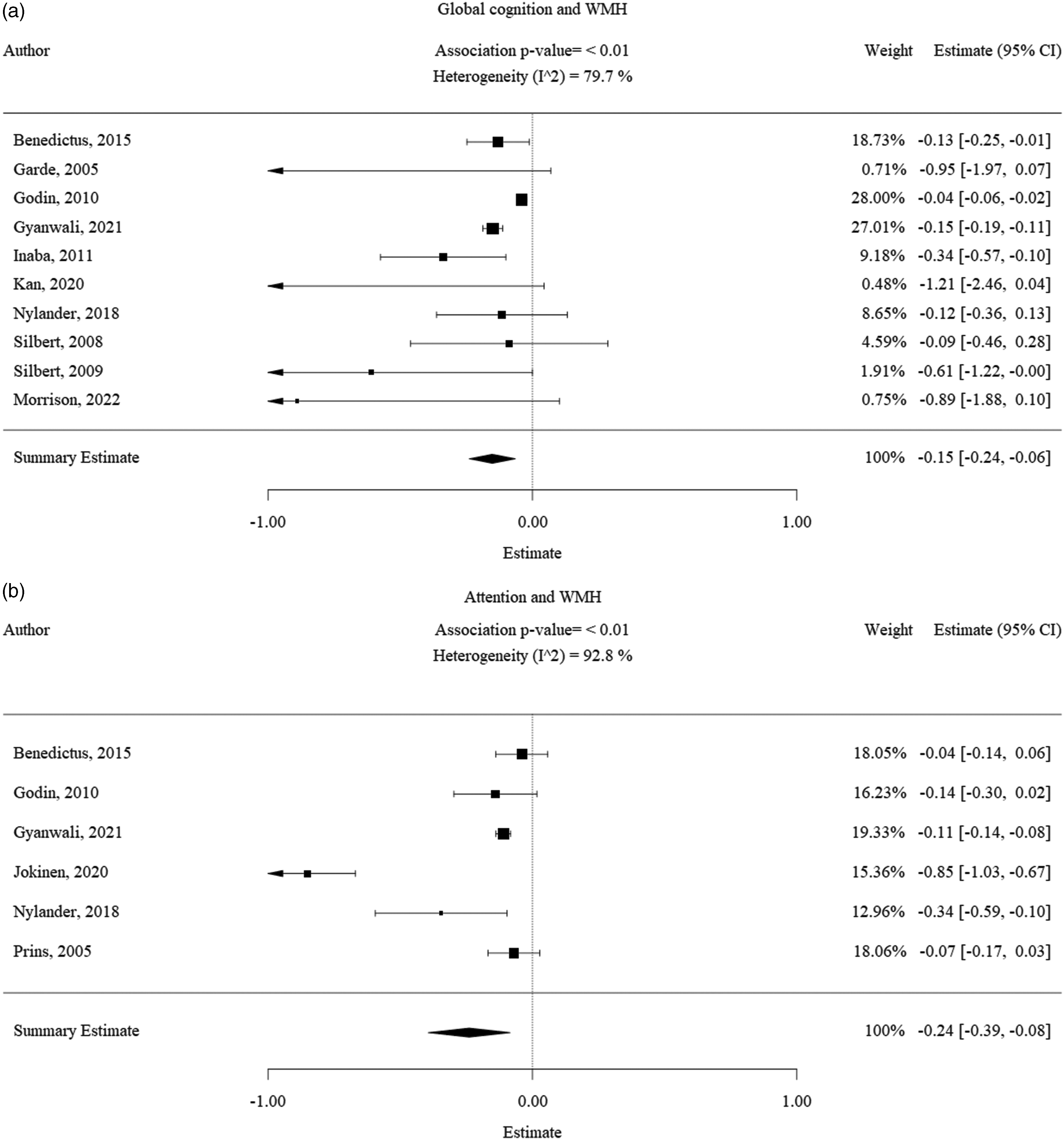
Forest plot of the association between WMH with cognitive decline. Results of the WMH subgroup meta-analysis visualized as forest plots, displaying the effect size per included study and the summary effect estimate for the presence of WMH in relation to each cognitive domain, namely a.) WMH and global cognitive decline, b.) WMH and decline in attention, c.) WMH and decline in executive function and d.) WMH and decline in memory function. Squares indicate individual study effects. Size of the squares is proportional to the relative study weight. Bars indicate confidence interval of the study estimate. Diamond indicates the summary effect whereas the outer edges of the diamond indicate the confidence interval. The scale of the estimate ranges from −1 to +1 where −1 indicates high cognitive decline and +1 indicates high cognitive improvement. All significance values that were less than 0.01 were rounded off to the P < 0.01. The exact significance values were mentioned in the text of the results.
CMBs
There was no significant pooled effect of CMB on global cognition (Figure 4(a)). The pooled effect estimate of CMBs on attention was −0.13 [−0.25; −0.02] (p < 0.05) with a heterogeneity of 65% (Figure 4(b)). The pooled estimate of the effect of CMBs on executive function was −0.04 [−0.07, −0.02] (p < 0.05) with a heterogeneity of 20% (Figure 4(c)). The pooled effect estimate of CMBs on memory was −0.11 [−0.21; −0.02] (p < 0.05) with a heterogeneity of 45% (Figure 4(d)).
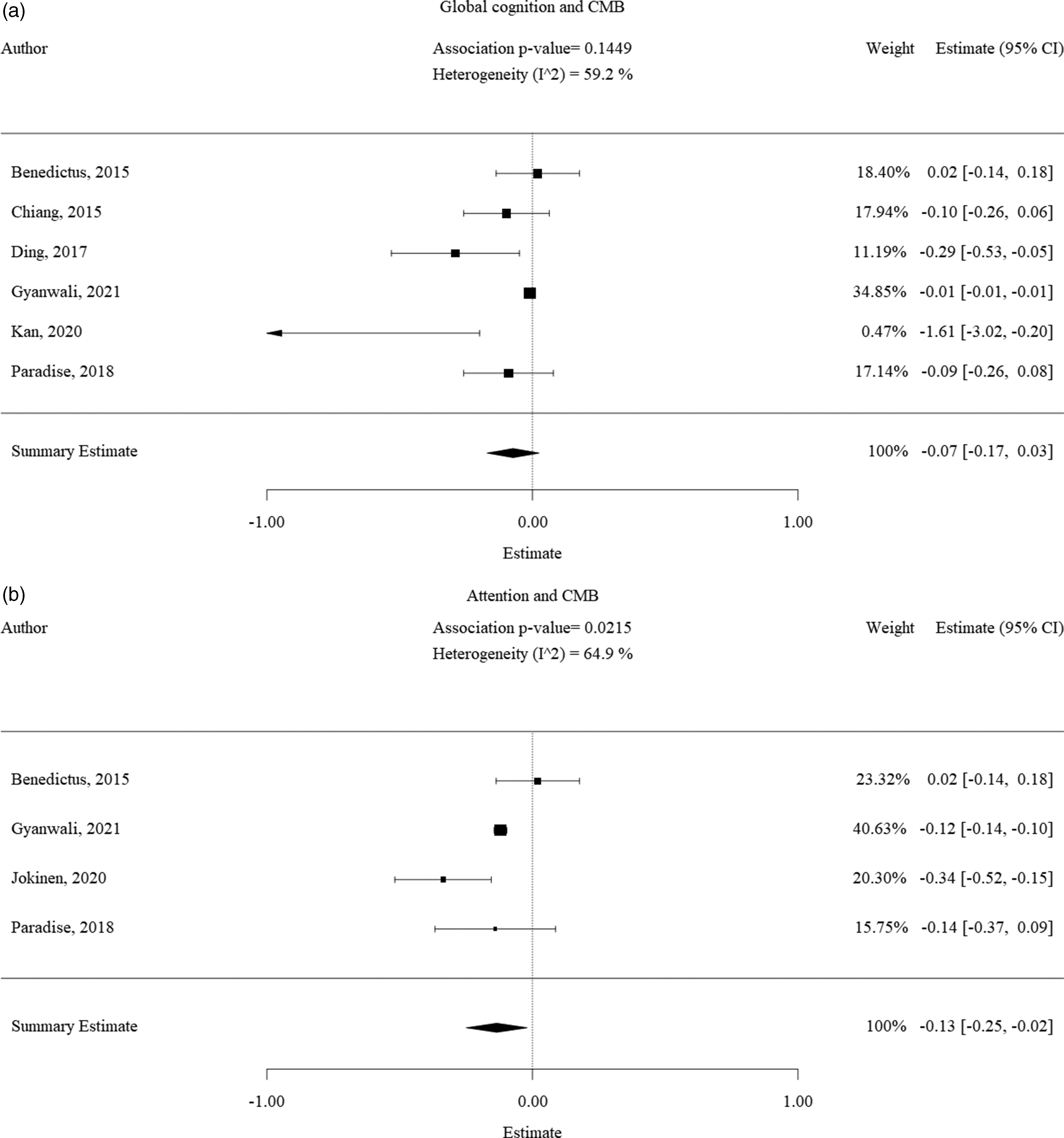
Forest plot of the association between CMB with cognitive decline. Results of the CMBs subgroup meta-analysis visualized as forest plots, displaying the effect size per included study and the summary effect estimate for the presence of CMBs in relation to each cognitive domain, namely a.) CMBs and global cognitive decline, CMBs and decline in attention c.) CMBs and decline in executive function and d.) CMBs and decline in memory function. Squares indicate individual study effect. Size of the squares is proportional to the relative study weight. Bars indicate confidence interval of the study estimate. Diamond indicates the summary effect whereas the outer edges of the diamond indicate the confidence interval. The scale of the estimate ranges from −1 to +1 where −1 indicates high cognitive decline and +1 indicates high cognitive improvement. All significance values that were less than 0.01 were rounded off to the P < 0.01. The exact significance values were mentioned in the text of the results.
Lacunes
There was sufficient data available to perform a meta-analysis for the association between lacunes and attention (Figure 5(a)) and memory (Figure 5(b)). A very minor effect was seen for memory functioning (effect size of −0,01 [−0,01; −0,01], mainly ascribed to the study by 32 which had a 99% study weight of the 4 included studies.
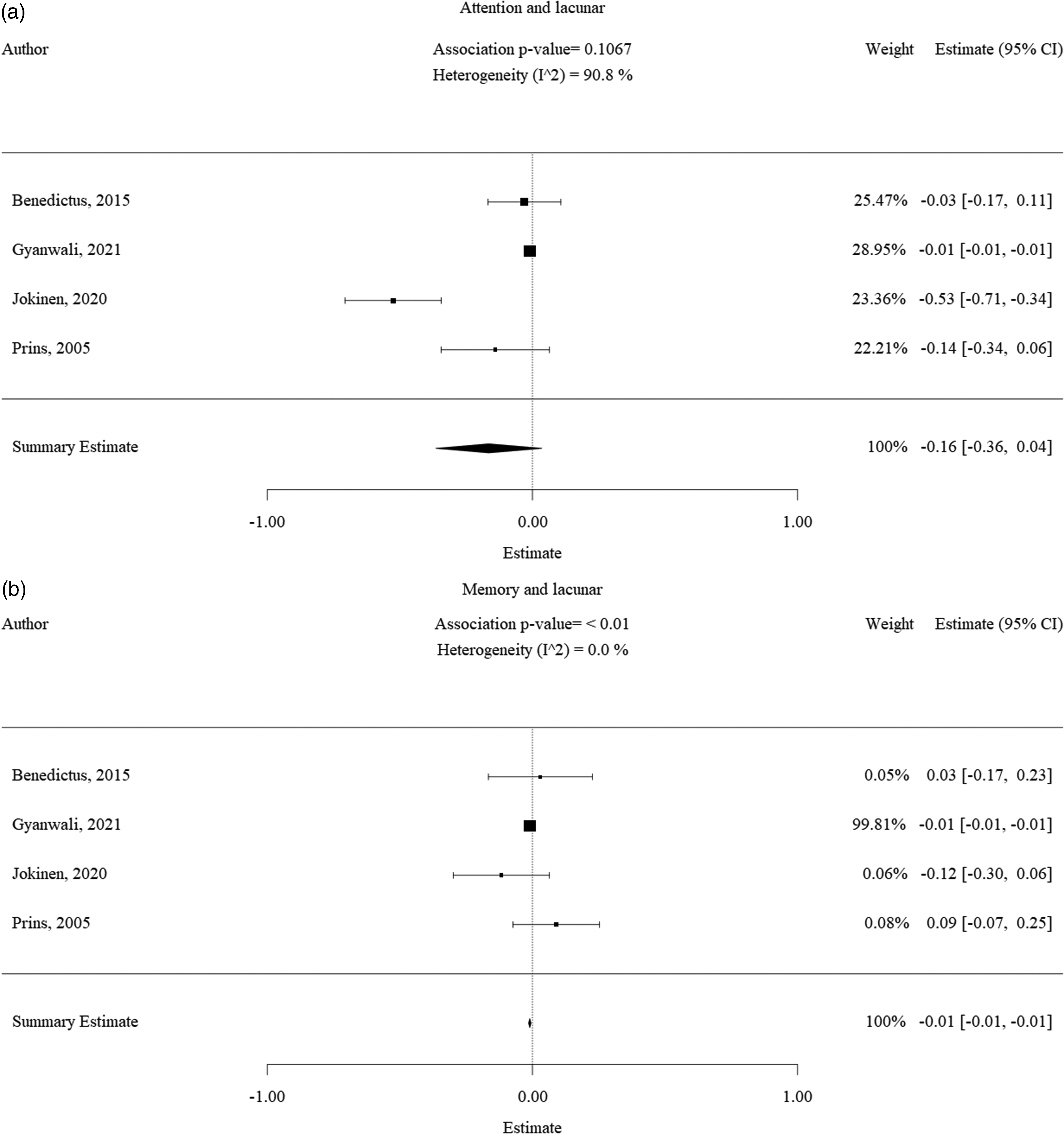
Forest plot of the association between lacunes with cognitive decline. Results of the lacune subgroup meta-analysis visualized as forest plots, displaying the effect size per included study and the summary effect estimate for the presence of lacunes in relation to each cognitive domain, namely a.) lacunes and decline in attention and, b.) lacunes and decline in memory function. Squares indicate individual study effect. Size of the squares is proportional to the relative study weight. Bars indicate confidence interval of the study estimate. Diamond indicates the summary effect whereas the outer edges of the diamond indicate the confidence interval. The scale of the estimate ranges from −1 to +1 where −1 indicates high cognitive decline and +1 indicates high cognitive improvement. All significance values that were less than 0.01 were rounded off to the P < 0.01. The exact significance values were mentioned in the text of the results.
Analyses for trends
SVD and clinical parameters
We aimed to visually inspect for potential trends in predictive performance of SVD in relation with clinical parameters of the included participants, such as clinical parameters gender and age. Lesion location was not take into account for the current review, although 10 of the 23 included studies reported on the difference of outcomes specifically per brain-region. However, the classification and categorization of regions differed per study making it difficult to summarize this data.
Gender
There is a mix of males and female in almost all the studies (F:M variation was between 38%–68%). Visual inspection indicated that the results of the associations between SVD types and decline of cognitive performance in different domains were consistent across studies, irrespective of the gender distribution within the studies (Figures 2 to 5).
Age
Ages included in this review ranged from 62 to 84 years with an average of around 75 years. Also here, visual inspection indicated that the results of the associations between SVD types and decline of cognitive performance in different domains were consistent across studies, irrespective of the age distribution within the studies (Figures 2 to 5).
Discussion
Although cerebral SVD has been implicated as having a causal role in vascular cognitive impairment (VCI) and dementia, the exact effects of specific SVD markers on decline in specific cognitive domains in healthy adults without cognitive deficits is yet to be quantified. The main finding of this systematic review and meta-analysis is that although MRI markers of cerebral SVD predict an increased risk of cognitive decline in all domains (global, cognitive, executive, attention) in relatively healthy older adults without cognitive deficits at baseline, not all MRI markers of SVD carried a similar weight in their effect on cognitive decline. Presence of WMH was the strongest predictor of cognitive decline over time, significantly affecting all cognitive domains. The contribution of lacunes and CMB was weaker. Notably, the effects of lacunes were mostly inconsistent across the included studies. To the best of our knowledge, this is the first systematic review and meta-analysis on the quantified effects of sporadic SVD on cognitive decline in healthy older adults over 60 years of age without cognitive deficits at baseline.
Our systematic review showed that the studies on CMB suffered from low numbers of individuals with CMB and a corresponding high variability in outcomes. Furthermore, data on the association between the presence of lacunes and cognitive decline were scarce, although significant findings in individual studies were reported.28,32 –35 The finding of a weaker yet significant effect of lacunes and CMB on cognitive decline has been documented in earlier studies done in (older) adults without large infarcts,28,36 but we have additionally provided evidence in healthy older individuals. As such, future studies on CMB should include larger samples, whereas studies investigating the role of lacunes in cognitive decline should still be established.
Based on the results of our meta-analysis, WMH volume in community-dwelling older individuals could be a prognostic biomarker for cognitive decline in global cognitive, executive, memory-related and attention-related functioning. This finding is in line with the observations following the LADIS and RUN DMC studies that found WMH to be a strong predictor of global cognitive decline in older individuals.37,38 Also, presence of CMB or an increased CMB count could be used as a biomarker for a potential decrease in executive functioning together with WMH volume.
A summary of the principal methodology used for quantifying SVD and cognition in the current studies indicated that presence and volume of markers of SVD were usually detected and quantified on 1.5–3.0 Tesla MRI scans, using T2-weighted or FLAIR sequences for WMH detection and T2* or susceptibility-weighted imaging (SWI) imaging to detect the presence of CMB’s. WMH volume was preferentially quantified using automatic segmentation tools, secondary to traditional scoring methods such as the Fazekas scale.10,39 CMB were preferentially counted numerically, similar to lacunes. Cognitive decline was preferentially established using multiple neuropsychological tests in tandem to obtain multiple performance measurements on each respective cognitive domain.
The finding that all studies had directionally consistent results towards a negative effect of SVD presence on cognitive functioning over time, is further supported by our risk-of-bias assessment across studies. Publication bias was minimal for all analysed cognitive domains, indicated by the observation that most study residuals fell within the pseudo-confidence region of the funnels. This implies that most studies investigating the association between SVD and cognitive decline derive similar findings, as reported here, and that the chance of any potentially missing or unpublished studies altering the significant findings of our meta-analysis is low.
Sporadic cerebral SVD is a multifactorial disease caused by diverse pathological processes, most common of which are arteriolosclerosis caused by hypertension, and other conventional vascular risk factors. 1 SVD has been associated with a wide range of clinical manifestations including motor impairment, (vascular) parkinsonism, impaired balance, falls, and behavioural symptoms, such as depression, apathy, and personality change. Furthermore, SVD has been implicated in the development of vascular cognitive impairment and dementia.13,40 Considering the health importance of SVD, it is important to attempt to project a clinical threshold level for determining when an individual will potentially experience cognitive decline due to the presence of cerebral SVD. This knowledge is important for future lifestyle intervention studies and treatment studies to prevent cognitive decline in older individuals based on cerebral SVD burden.
Based on the results of our review and the found summary estimates, a clinical threshold level for determining when an individual will potentially experience cognitive decline due to the presence of cerebral SVD and thus potentially requiring a change in patient management, can be set. This notion is also termed as the minimally clinical important difference (MCID) level. 41 The resulting MCID levels for the respective cognitive domains investigated in this study would as such be −0.10 [−0.14; −0.05] for global cognition, −0.12 [−0.17; −0.07] for memory, −0.17 [−0.23; −0.11] for attention and −0.18 [−0.24; −0.11] for executive function, respectively. These effect estimates can however only be compared to similar effect estimates, such as the z-score, for individuals that have undergone multiple neuropsychological tests. Importantly, our summary estimates only provide information on the transition from normal cognition up to mild cognitive impairment (MCI). The progression of MCI to dementia is a process that takes longer and has a different clinical onset. 18 With regards to the prevention and treatment of SVD related cognitive decline, Cannistraro et al. (2019) recommend to take into account the full risk profile of the patient, presence of multiple SVD markers and corresponding severity and the severity of the clinical sequelae. Lowering blood pressure is seen as the most prominent risk-modulator for SVD, next to the use of statins.10,42,43
Previously, Debette et al. (2010) found that WMH are an important risk factor for future development of cognitive dysfunction, especially in the areas of executive function and processing speed, based on the results of a subgroup meta-analysis that included 4 population based studies. 5 Our results add to the finding of this study that predominantly an increased WMH load influences cognitive decline albeit that we found WMH to have an effect on attention and memory functioning too. This effect was investigated earlier in a study by 44 who showed in an anatomical mapping of WMH in the brain that an increasing WMH burden corresponds directly to increased confluency of WMH from the periventricular regions to the deep white matter. This implies that as WMH burden increases, more brain regions are affected and correspondingly the effect on the specific functions in cognition is increased. This may explain why a higher WMH burden can potentially be linked to both cognitive decline and MCI as well as dementia. Indeed, an increasing WMH burden in the brain has previously shown to be correlated to an increased risk of developing all-cause dementia and Alzheimer’s disease in the general population (Bos et al. (2018; HR = 1.39 [1.00–1.94])). 45 CMB and lacunes, however, are known to be more diffusely dispersed throughout the brain and are smaller and more focal. As such, the effects of CMB may be less detrimental compared to WMH and can be more variable in nature depending on their location. As found in the current review, executive function was previously shown to be affected by cerebral microbleeds. 46 An earlier study by 9 found that cerebral microbleeds were located predominantly in the frontal lobes and basal ganglia, which are indeed the brain regions associated with executive function. In the current review we additionally found an association between CMBs and both memory and attention function.
An earlier systematic review of SVD and risk of Alzheimer’s disease found that SVD markers are associated with clinical dementia pathology, but the exact interplay between SVD markers and Alzheimer’s disease pathology is unclear. 14 Our current systematic review and meta-analysis shows that WMH and also CMB may cause decline in global cognitive function over time in healthy community-dwelling older individuals, next to the effects of WMH was shown to have on cognitive impairment in post-stroke individuals 17 and the effects of WMH as well as CMB were shown to have on the development of biomarkers CSF Aβ42 and Aβ pathology, respectively, these being related to Alzheimer’s disease. Importantly, in a recent meta-analysis of longitudinal studies, 47 although WMH volume was found to generally increase over time, large inter-individual differences were observed in the magnitude of WMH volume increase over time, with also evidence for WMH regression over time. In time, WMH volume regression could be an important intervention target for future studies.
Our study has several strengths. One strength is the extensive literature search that was performed based on a comprehensive list of study in- and exclusion criteria, providing us with the most recent study list available on this topic. Secondly, we performed an in-depth data synchronisation and meta-analysis based on the individual study effect sizes, hereby obtaining standardized and quantifiable outcomes. Thirdly, we performed subgroup calculations for both the different cognitive domains and types of SVD that allowed us to determine any potential directions of effect within our primary analysis.
Our study has a number of limitations. First is the heterogeneity of results between the included studies. Variety in the choice of cognitive tests that was used to assess the different cognitive domains could have been a contributory factor to this high observed heterogeneity after data were pooled and meta-analysed. Secondly, studies differed in the way MRI markers of cerebral SVD were determined and in what way the progression of SVD severity was accounted for in statistical analysis where a common method would have generated more comparable results. However, as all studies included were found to be directionally consistent, this effect was not deemed detrimental. Another limitation of our study is that although we did not include study participants that had vascular pathology at baseline of their respective study, we cannot totally exclude the possibility that some of the participants could have developed large vessel disease during the studies as these were not fully reported. If this indeed occurred, we cannot completely exclude that cognitive decline could be a consequence of such an event. Another limitation of the current study is that we were not able (due to lack of homogenous individual participant imaging data) to combine different variables into a multivariable prediction model to prospectively assess individual risk. To move the field forwards, future studies could use artificial intelligence based image analysis methods, such as deep learning, to allow for a more comprehensive analysis of imaging markers in larger data sets. 48
In conclusion, our systematic review and meta-analysis shows that individual MRI markers of cerebral SVD are associated with a different pattern of cognitive decline in healthy, community-dwelling, older adults without cognitive deficits at baseline. WMH were shown to be significantly correlated to longitudinal decline in all cognitive domains: global cognition, executive function, memory and attention, whilst CMB were shown to affect decline predominantly in executive function, while some studies also observed effects of CMB on attention-span and memory.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-3-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-3-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-4-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-4-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-5-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-5-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-6-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-6-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-7-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-7-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-8-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-8-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-9-jcb-10.1177_0271678X241235494 - Supplemental material for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis
Supplemental material, sj-pdf-9-jcb-10.1177_0271678X241235494 for Sporadic cerebral small vessel disease and cognitive decline in healthy older adults: A systematic review and meta-analysis by Alexander Jansma, Jeroen de Bresser, Jan W Schoones, Diana van Heemst and Abimbola A Akintola in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors’ contributions
Alexander Jansma made substantial contributions to concept and design, acquisition of data and analysis and interpretation of data, and drafted the article.
Abimbola Akintola made substantial contributions to concept and design, acquisition of data and analysis and interpretation of data, and revised the article critically for important intellectual content.
Jeroen de Bresser made substantial contributions to analysis and interpretation of data, and revised the article critically for important intellectual content.
Diana van Heemst made substantial contributions to analysis and interpretation of data, and revised the article critically for important intellectual content.
Jan W. Schoones made substantial contributions to acquisition of data, and revised the article critically for important intellectual content.
All authors approved the version to be published.
Acknowledgements
The authors would like to thank Dr. M. Habes (University of Pennsylvania), Dr. L. Silbert (University of Oregon), Dr. A. Brickman (Columbia University), Dr. E. Smith (University of Calgary), Dr. H. Jokinen-Salmela (University of Helsinki), Dr. K. Bangen (University of California San Diego) and C. Ni Kan (National University of Singapore) for providing additional (unpublished) data on request.
Code availability
Code for meta-analysis, forest plots and funnel plots is available upon request.
Data availability statement
Data on reviewed articles and meta-analysis is available upon request.
Declaration of conflicting interests
Alexander Jansma: reports no disclosures
Jeroen de Bresser:
The research of Jeroen de Bresser is supported by Alzheimer Nederland under grant WE.03-2019-08.
Diana van Heemst: reports no disclosures
Abimbola A. Akintola: reports no disclosures
Jan W. Schoones: reports no disclosures
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
