Abstract
Among opioids, buprenorphine presents a favorable safety profile with a limited risk of respiratory depression. However, fatalities have been reported when buprenorphine is combined to a benzodiazepine. Potentiation of buprenorphine interaction with opioid receptors (ORs) with benzodiazepines, and/or vice versa, is hypothesized to explain this drug-drug interaction (DDI). The mutual DDI between buprenorphine and benzodiazepines was investigated at the neuroreceptor level in nonhuman primates (n = 4 individuals) using brain PET imaging and kinetic modelling. The binding potential (BPND) of benzodiazepine receptor (BzR) was assessed using 11C-flumazenil PET imaging before and after administration of buprenorphine (0.2 mg, i.v.). Moreover, the brain kinetics and receptor binding of buprenorphine were investigated in the same individuals using 11C-buprenorphine PET imaging before and after administration of diazepam (10 mg, i.v.). Outcome parameters were compared using a two-way ANOVA. Buprenorphine did not impact the plasma nor brain kinetics of 11C-flumazenil. 11C-flumazenil BPND was unchanged following buprenorphine exposure, in any brain region (p > 0.05). Similarly, diazepam did not impact the plasma or brain kinetics of 11C-buprenorphine. 11C-buprenorphine volume of distribution (VT) was unchanged following diazepam exposure, in any brain region (p > 0.05). To conclude, our PET imaging findings do not support a neuropharmacokinetic or neuroreceptor-related mechanism of the buprenorphine/benzodiazepine interaction.
Introduction
The opioid overdose crisis started in 1999 with the increase of fatalities attributed to the irrational and over-prescription of opioids. 1 In this context, buprenorphine has become the leading opioid maintenance treatment prescribed in the US, ranking before methadone. 2 First-line prescription of buprenorphine is related to its respiratory safety profile. Compared to methadone, which is a full mu-opioid receptor (MOR) agonist, buprenorphine is described as a partial MOR and a kappa-opioid receptor (KOR) antagonist, resulting in ceiling effects regarding respiratory depression but not analgesia, even at high doses.3,4 The slow association/dissociation of buprenorphine at its target receptors may also contribute to its particular safety profile. 5
However, as soon as marketed in Europe, where it was first approved in the mid-1990s, deaths involving buprenorphine were reported. 6 Forensic studies have clearly shown that most of deaths occurred when buprenorphine was co-administered with another psychotropic drug, primarily a benzodiazepine.6 –8 Consistently, animal studies have shown that buprenorphine or benzodiazepines alone do not induce respiratory depression while the combination of both drugs does, 9 strongly suggesting a drug-drug interaction (DDI), which mechanisms still remains unclear. Several preclinical studies, mainly performed in rodents, do not support the hypothesis of peripheral pharmacokinetic DDI. Buprenorphine is metabolized by cytochrome P450 (CYP) 3 A to norbuprenorphine, which is a ∼10-fold more potent MOR agonist compared to buprenorphine in vitro. 10 In the rat, CYP3A induction increased the proportion of circulating norbuprenorphine but did not worsen buprenorphine-induced respiratory depression. 11 This experimental finding could be explained by the low brain penetration of norbuprenorphine, which transport across the blood-brain barrier (BBB) is negligible compared to the transport of buprenorphine itself.12,13 Therefore, it can be hypothesized that the DDI between buprenorphine and benzodiazepines may occur at the CNS level. This may involve interaction, cross-regulation or allosteric modulation of the neuroreceptors mediating the effects of each respective drug.
In vivo quantification of benzodiazepine receptors (BzR) is possible with Positron Emission Tomography (PET) imaging using 11C-flumazenil, a reversible BzR antagonist. 14 11C-flumazenil has been used to quantify the engagement of BzR by different pharmacological doses of benzodiazepines.15,16 Moreover, 11C-flumazenil PET imaging has been widely used to investigate regulation of BzRs in various pathological conditions such as epilepsy, 17 Alzheimer disease, 18 multiple sclerosis, 19 or ischemic stroke. 20 11C-flumazenil benefits from extensive research to validate pharmacokinetic models and quantitatively describe the availability of BzR in vivo. 21 In this setting, the partial saturation method has been validated in baboons 22 and humans,23,24 aiming to estimate both the density and affinity to BzRs from a single-scan session.
Several PET radioligands have been developed for quantification of opioid receptors (ORs) in the living brain. 25 The MOR 11C-carfentanil has been useful to investigate the dose and time-dependent occupancy of MORs by buprenorphine in humans. 25 We recently validated 11C-buprenorphine in rodents and non-human primates as a mean to specifically address the neuropharmacokinetics and binding of buprenorphine to its CNS targets in vivo.26,27 Blocking experiments performed in rats suggested that the specific binding of 11C-buprenorphine in the brain predominantly corresponds to its interaction with MORs rather than with other OR subtypes. 26
In the present study, the hypothesis of a mutual DDI between buprenorphine and diazepam, a widely used benzodiazepine, was investigated at the neuroreceptor level using brain PET imaging, according to a cross-over design in non-human primates. The impact of a pharmacological dose of buprenorphine on BzR was assessed using 11C-flumazenil PET imaging. Then, the impact of a pharmacological dose of diazepam on the brain kinetics and receptor binding of buprenorphine was investigated in the same individuals using 11C-buprenorphine PET imaging.
Material and methods
Chemicals and radiochemicals
Buprenorphine hydrochloride for intravenous (i.v.) injection (0.3 mg/mL) was obtained from Axience (Pantin, France). Naloxone hydrochloride for i.v. injection (0.4 mg/mL) was obtained from Aguettant (Lyon, France). Diazepam for intravenous (i.v.) injection (5 mg/mL) was obtained from Roche (Boulogne-Billancourt, France). Ketamine was obtained from Virbac (Carros, France). Propofol was purchased from Fresenius laboratory (Sèvres, France). Isoflurane was obtained from Abbvie (Rungis, France). Flumazenil (0.1 mg/mL) was obtained from Mylan (Paris, France).
11C-Buprenorphine was synthesized in-house according to the method described by Lever et al. 28 with minor modifications previously described. 26 Ready-to-inject 11C-buprenorphine (1.8–3.5 GBq, decay-corrected radiochemical yield: 15%) was obtained with a radiochemical purity above 98% and a molar activity of 28 GBq/μmol at end of synthesis (EOS).
11C-Flumazenil was synthesized based on a previously published method. 29 Ready-to-inject 11C-flumazenil (1.1–2.8 GBq, decay-corrected radiochemical yield: 23%) was obtained with a radiochemical purity above 98% and a molar activity of 21 GBq/μmol EOS.
Animals
All animal of the procedures were used in accordance with the recommendations of the European Community for the care and use of laboratory animals (2010/63/UE) and the French National Committees (French Decret 2013–118). Experimental protocols were validated by a local ethics committee for animal use (CETEA/A15–002 and A18–065) and approved by the French government. Four adult male Rhesus macaques (Macaca Mulatta; weight = 7.2 ± 1.2 kg, age = 5.6 ± 0.5 years during the study), which were obtained from Silabe (Simian Laboratory Europe, France). Animals were housed and acclimatized for at least 7 days before experiments. Macaques were housed under standard experimental conditions: room temperature (22 ± 1°C); light/dark cycle (14 h light/10 h dark); free access to water and food (fresh fruit preparations, pellets). Animals were kept in social groups with enrichment (toys and structures for resting, hiding and climbing). Animal data are reported in compliance with the ARRIVE guidelines 2.0.
Study design
The present study is composed of two sequential sets of experiments with 11C-buprenorphine and 11C-flumazenil, respectively designated as study 1 and study 2. All four macaques participated to study 1 and study 2, Allowing for paired comparison of outcome data. The number of individuals was set based on previous studies using a similar design.26,30 A minimum interval of two weeks was respected between two PET imaging experiments in the same individual.
Baseline scans were performed before pretreatement scans, assuming a possibility for long-term regulation of investigated neuroreceptor by the pretreatment.
Arterial input function and metabolism
During PET acquisition, in both studies 1 and 2, arterial blood samples (500 µL) were obtained at selected times after the radiotracer injection. Samples were centrifuged (5 min; 2,054 g; 4 °C) and the supernatant (200 µL) was gamma-counted for total plasma radioactivity. Additional plasma samples were obtained at 0, 5, 10, 15, 30, 60, and 90 min to measure the percentage of parent (unmetabolized) radiotracer using radio-HPLC and state of-the-art methodology. 31 The fraction of parent radiotracer in each sample was used to generate the metabolite-corrected arterial input function.
PET acquisition procedure
First, each monkey underwent an anatomical T1-weighted brain MR scan using an Achieva 1.5 T scanner (Philips Healthcare, Suresnes, France) on ketamine anesthesia (intramuscular injection, i.m.). PET acquisitions were performed using a HR+ Tomograph (Siemens Healthcare, Knoxville, TN, USA) in anesthetized macaques, as previously described. 32 Briefly, the macaque received ketamine (10 mg/kg, i.m.) to induce anesthesia. After intubation in supine position, venous catheters were inserted for radiotracer injection (sural vein), propofol infusion (sural vein), and drug injection for pre-treatment conditions (brachial vein). Another catheter was inserted in the femoral artery for arterial blood sampling. Macaques were positioned under the camera before the administration of a 2 mL i.v. bolus of propofol followed by a 1 mL/kg/h i.v. infusion under oxygen ventilation. The heart rate, oxygen saturation, respiratory rate, and endtidal CO2 were monitored during the PET scan procedure.
In study 1, macaques were i.v. injected with a microdose of 11C-buprenorphine (280 ± 60 MBq, molar activity at the time of injection (MAinj) = 19.3 ± 8.6 GBq/µmol).
In study 2, macaques were i.v. injected with 11C-flumazenil (277 ± 44 MBq, MAinj = 10.8 ± 4.9 GBq/µmol), co-injected in the same syringe with 10 µg/kg of unlabeled flumazenil to enable quantification of receptologic parameters using the partial saturation method, as previously described in humans. 24
Imaging data reconstruction and segmentation
The person analysing the data was not aware of the baseline/pretreatment status of each animal. PET data were analyzed using PMOD software (PMOD Technologies, Zürich, Switzerland). PET images were co-registered to corresponding T1-weigted MR images for each macaque. A macaque T1-weighted MR template 33 was generated from individual MR images. Transformation matrices were thereafter applied to the segmentation obtained from the template to generate time-activity curves in 13 brain structures selected as volumes of interest (VOI): frontal, orbital, cingulate, temporal, parietal and occipital cortices, and caudate nucleus, putamen, amygdala, thalamus, hypothalamus, pons and cerebellum.
PET quantification and data modeling
For 11C-buprenorphine and 11C-flumazenil PET experiments, kinetics of radioactivity in the selected brain regions and in arterial plasma samples were corrected for radioactive decay, injected dose of radioactivity and animal weight, and expressed in standardized uptake values (SUV). Brain exposure was estimated by the area under the time-activity curve (AUC) of the regional brain kinetics of PET radioligands.
In study 1, the brain distribution of 11C-buprenorphine (VT; mL/cm3) was estimated using the Logan plot analysis taking the metabolite-corrected input function into account. 34
In study 2, the regional brain distribution of 11C-flumazenil was estimated by VT. The binding potential of 11C-flumazenil was also estimated using the simplified reference tissue model (SRTM) using the pons as the reference region (BPND). Moreover, a graphical analysis based on the partial saturation method was tested for estimation of i) BAvail, which represents the concentration of receptors in the VOI, estimated graphically with the Scatchard analysis as the X-axis intercept and ii) AppKd represents the in vivo affinity, estimated graphically with the Scatchard analysis as the Y axis-intercept, as previously described. 24
Statistical analysis
Comparisons between the different experimental conditions were performed using R 35 and GraphPad Prism (v9.5, USA) softwares. Plasma exposures were compared using a Wilcoxon matched pairs test. Outcome parameters of brain PET data were compared using a Two-Way Repeated Measure ANOVA, assuming that both factors “Brain regions” and “Treatment” were matched for each individual. The distribution of residues was normal (Levene’s test) and homogeneous (Bartlett’s test). A Tukey’s post-hoc was performed for multiple comparisons test. A result was deemed significant when a two-tailed p value was <0.05.
Results
Impact of diazepam pretreatment on the kinetics of 11C-buprenorphine
In the study 1, diazepam did not impact the metabolism of 11C-buprenorphine. The proportion of parent 11C-buprenorphine was not different in the absence and presence of diazepam pre-treatment, at any time-point (Figure 1(a)). Plasma concentrations of parent 11C-buprenorphine peaked rapidly, followed by a fast washout of radioactivity to reach equilibrium (∼0.12–0.20; SUV unit) at 15 min after 11C-buprenorphine injection (Figure 1(b)). Diazepam did not impact the plasma exposure of 11C-buprenorphine with AUCplasma = 26.9 ± 1.9 and 27.3 ± 1.7 SUV.min in the absence and presence of diazepam, respectively (p = 0.89, Figure 1(c)).
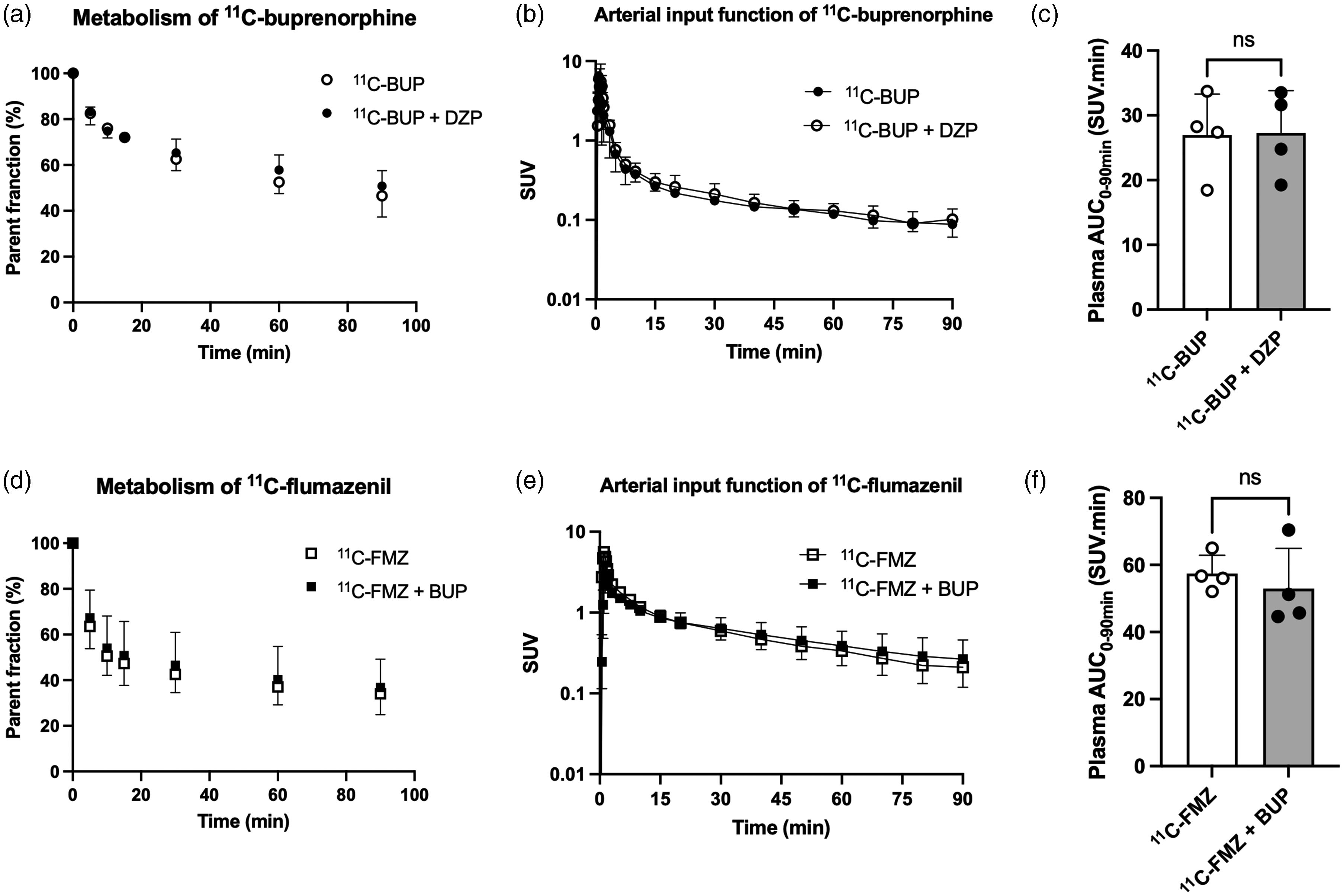
Metabolism and plasma pharmacokinetics of 11C-buprenorphine (11C-BUP) and 11C-flumazenil (11C-FMZ), with or without diazepam (DZP) or buprenorphine (BUP) pre-treatment, respectively. (a) Percentage of parent 11C-buprenorphine vs time in plasma. Arterial input function of unmetabolized 11C-buprenorphine (b) and corresponding areas under the curve (c). Percentage of parent 11C-flumazenil vs time (d). Arterial input function of unmetabolized 11C-flumazenil (e) and corresponding areas under the curve (f). 11C-Flumazenil vs time is shown in a and c. Data are shown as mean ± SD (n = 4). Statistical significance was set at p < 0.05 with ns: non-significant.
The brain kinetics and distribution of 11C-buprenorphine in macaques was consistent with previous findings 26 (Figure 2), with a significant difference between brain regions (p < 0.001) and maximal distribution in the OR-rich regions such as the putamen (VT = 11.2 ± 1.6 mL.cm−3) and caudate nucleus and (VT = 10.5 ± 1.4 mL.cm−3), and minimal distribution in the regions with poor expression of ORs such as the cerebellum (VT = 5.0 ± 0.7 mL.cm−3) and the occipital cortex and (VT = 5.0 ± 0.9 mL.cm−3, Figure 2). Pretreatment with diazepam slightly increased the VT of 11C-buprenorphine in some brain regions, although statistical significance was not reached (p > 0.05, Figure 2). Maximum change in the VT was observed in the cerebellum (+8.8 ± 14.8%), the orbital cortex (+7.4 ± 11.2%), the temporal cortex (+7.4 ± 13.5%) and the pons (+7.0 ± 8.1%).
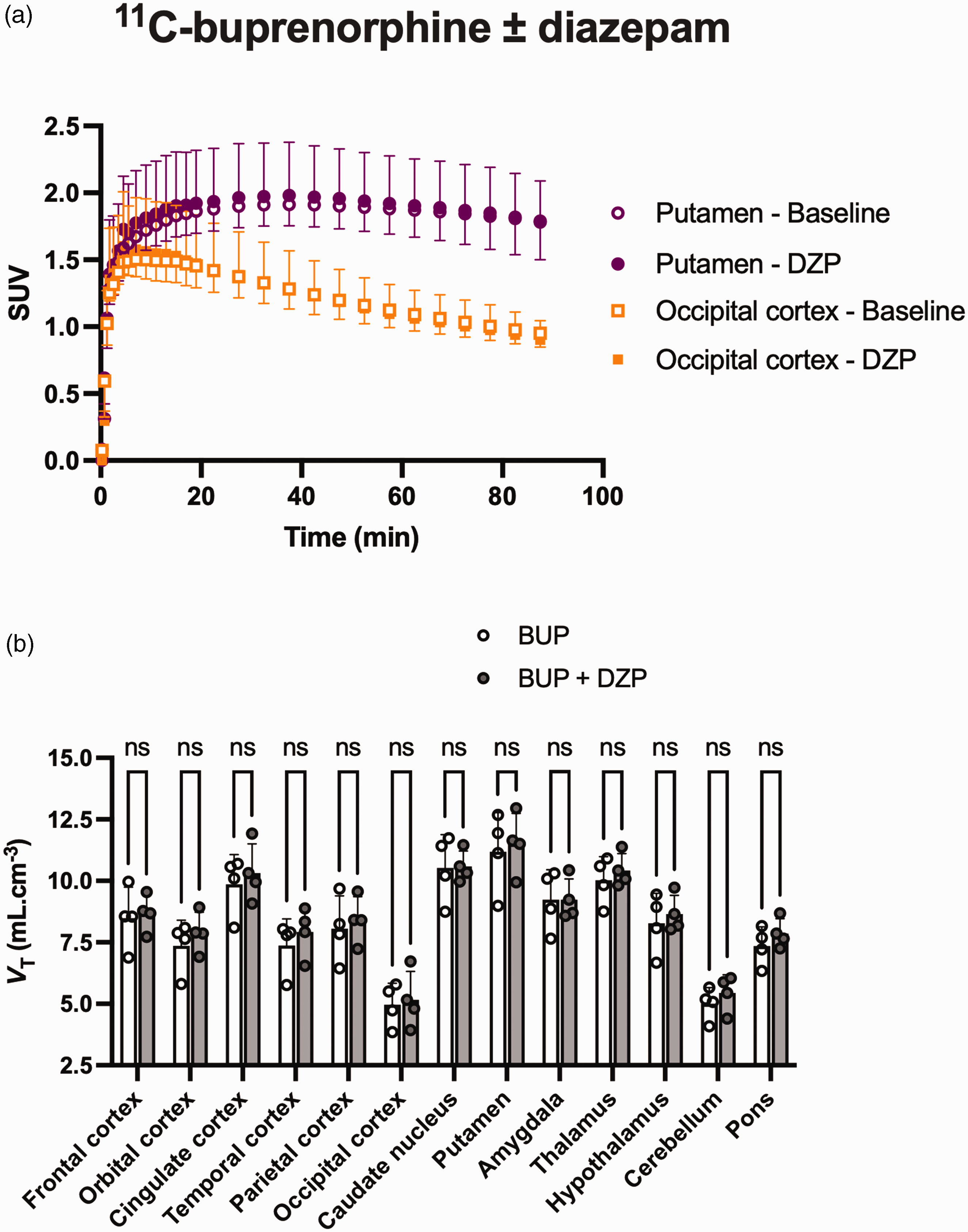
PET data of 11C-buprenorphine obtained with or without diazepam (DZP) pre-treatment in macaques. (a) 11C-Buprenorphine brain kinetics in the putamen and occipital cortex. (b) Regional VT estimated using the Logan plot analysis in each investigated brain region, with or without diazepam pre-treatment (b). Data are shown as mean ± SD (n = 4) with ns = non-significant.
Impact of buprenorphine pretreatment on the kinetics of 11C-flumazenil
Buprenorphine did not impact the plasma metabolism of 11C-flumazenil according to the results of the study 2. At each of the sampling times and up to 90 min, the proportion of the parent 11C-flumazenil fraction is not different with or without pre-treatment with buprenorphine (Figure 1(d)). Buprenorphine did not impact the plasma exposure of 11C-flumazenil (Figure 1(e)), with AUCplasma of 57.4 ± 5.5 and 55.0 ± 15.9 SUV.min with or without buprenorphine pre-treatment, respectively (p = 0.88, Figure 1(f)).
Regional VTs of 11C-flumazenil were significantly different across region (p < 0.001), with the lowest uptake in the pons. The VT of 11C-flumazenil in the pons was not different with or without buprenorphine pre-treatment (p = 0.99). Regional BPND using the pons as the reference region were significantly different among brain regions (p < 0.001) but were not different with or without buprenorphine pretreatment (p = 0.56) (Figure 3). In most experiments, linearity was not obtained for the Scatchard plot so that reliable estimation of BAvail and AppKd was not possible.
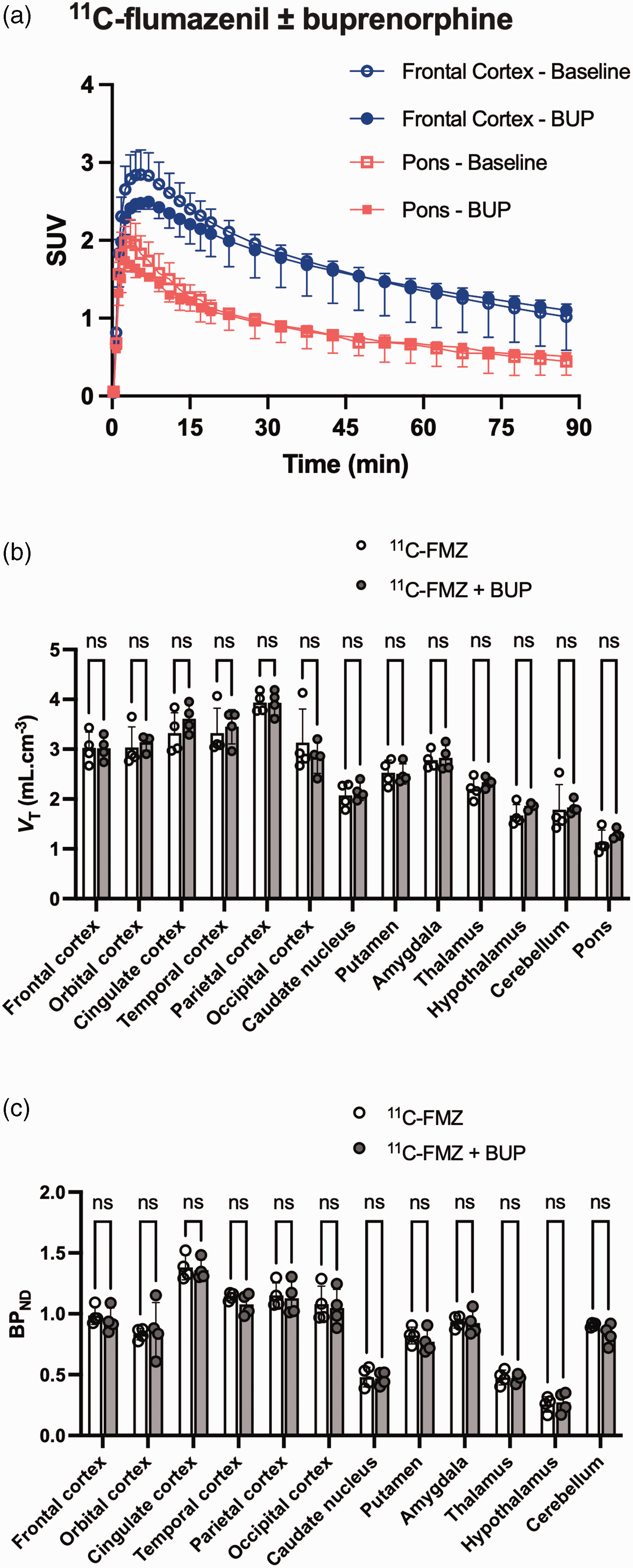
PET data of 11C-flumazenil obtained with or without buprenorphine (BUP) pre-treatment in macaques. (a) 11C-flumazenil brain kinetics for pons and frontal cortex regions. (b) Regional VT estimated using the Logan plot analysis in each investigated brain region, with or without diazepam pre-treatment and (c) Regional BPND measured using the SRTM with the pons as the reference region for each investigated brain region. Data are shown as mean ± SD (n = 4) with ns = non-significant.
Discussion
Previous studies have shown that combination of buprenorphine and benzodiazepines may lead to severe and often fatal respiratory depression, suggesting a DDI which mechanism remains poorly understood. 9 The functional interplay between the GABAergic and opioidergic systems, with the presence of MORs and BzRs on the same neurons, suggest an interactive role in controlling brain function. 36 Direct agonist effect of diazepam on MORs, and/or effect of buprenorphine on BzRs, is very unlikely, although this remained to be addressed in vivo.9,37 Moreover, the hypothesis of a positive allosteric modulation of BzRs and/or MORs by each respective drug remained to be investigated. The possibility for an allosteric modulation of MORs, which increased the affinity of orthosteric ligands has been reported.38,39 Translational studies have shown that some drugs like tiagabine or yohimbine may act as positive allosteric modulators of BzRs, which was confirmed in vivo using 11C-flumazenil PET imaging.40 –42 A cross-over PET study using 11C-buprenorphine and 11C-flumazenil was therefore conducted in non-human primates to address the hypothesis of a mutual DDI between diazepam and buprenorphine at the neuroreceptor level.
The impact of diazepam on the brain kinetics of buprenorphine was first investigated using 11C-buprenorphine PET imaging. Most imaging studies focusing on OR use dedicated PET radioligands such as 11C-carfentanil, which offer an insight into regulation of MORs in physiological conditions and disease settings. 25 In previously conducted rats and macaques studies, we showed that 11C-buprenorphine is suitable to investigate the brain exposure and receptor binding of buprenorphine, since predominantly driven by its affinity for MORs. 26 Therefore, this pharmacokinetic imaging approach was performed to more specifically investigate the impact of diazepam on both the brain exposure and binding to ORs in vivo. Only a microdose (∼10 µg) of 11C-buprenorphine could be administered to avoid respiratory depression in the presence of diazepam. The selected dose of diazepam used to precipitate the DDI (i.e. 10 mg i.v. bolus, 15 min before PET) corresponded to half the dose used in rats in this situation, 9 and five to ten-fold the human dose when i.v. injected. A previous PET study using 11C-flumazenil in baboons showed that a similar dose of diazepam (1.4 mg/kg), administered 20 min before PET, was associated with ∼70% occupancy of BzR in baboons. 30 This suggests that the buprenorphine-diazepam interaction, which is observed in the therapeutic range, 7 occurs in the situation of partial occupancy of BzRs in humans. Our results and kinetic modeling data suggested no or limited impact of diazepam on the brain exposure and binding of 11C-buprenorphine to brain regions, regardless of the local density of MORs, thus not supporting the hypothesis of an enhanced brain delivery of buprenorphine or regulation of the availability of MORs in the presence of diazepam. Our finding obtained in macaques is consistent with a previous study in rats showing that diazepam (20 mg/kg, s.c.) does not significantly change the brain kinetics of 11C-buprenorphine. 9 Of note, diazepam did not increase the binding of 11C-buprenorphine in the pons which contains important MOR-dependent loci for respiratory control such as the parabrachial nuclei or the Kölliker fuse. 43
The impact of a pharmacological dose of buprenorphine on the availability of BzR was thereafter investigated using 11C-flumazenil PET imaging. The selected dose of buprenorphine used to precipitate the DDI (0.2 mg i.v. bolus, which corresponds to 0.03 mg/kg in our animals) was administered in anesthetized macaques and shown to achieve ∼80% receptor occupancy. 26 11C-flumazenil is widely used in animals and humans and benefits from extensive research in pharmacokinetic modelling with the aim to better estimate the receptologic profile of BzR in vivo. 22 Moreover, flumazenil is a BzR antagonist and a pharmacological dose of unlabeled flumazenil could be safely administered after an unlabeled buprenorphine to explore DDIs using 11C-flumazenil PET, described as the partial saturation approach. 24 The selected dose of unlabeled flumazenil (10 µg/kg) used for partial saturation was previously shown to occupy ∼60% of available BzRs in baboons. 30 This corresponds to the recommended level for using the partial saturation method. 24 Consistently, regional BPND are lower than those reported in macaques using microdose 11C-flumazenil, in the absence of partial saturation. 44 In the partial saturation condition, level of available receptor is still substantial (∼40%). 45 However, it may be hypothesized that partial saturation, which reduces the dynamic range of available receptors, may reduce the sensitivity of 11C-flumazenil PET to detect changes in BzR availability. Nevertheless, buprenorphine did not change the regional VT of 11C-flumazenil. Neither was BPND, which was estimated using the pons as a reference region with low expression of BzRs, increased by buprenorphine pretreatment. Although microdose 11C-flumazenil PET has not been performed to confirm this result, this suggests a limited impact of buprenorphine pre-treatement on the brain distribution and binding of 11C-flumazenil to BzRs.
The partial saturation approach proposed by Delforge et al. has been described in baboons 22 and humans 24 aiming to estimate the density and the affinity of 11C-flumazenil from a single scanning section. 46 However, linearity of the Scatchard plot was not obtained in many macaques, in both baseline and buprenorphine conditions, so that BAvail and AppKd could not be reliably estimated (data not shown). The selected dose of unlabeled flumazenil used for partial saturation may be to low to achieve equilibrium during the scan in macaques. Further work is probably needed to optimize the dose of unlabeled flumazenil for partial saturation in the different animal species.
This study suggests a negligible impact of diazepam on the brain kinetics of buprenorphine, and the absence of regulation of MORs. Buprenorphine seems also not to impact the availability of BzRs. It is highly however likely that the interaction between buprenorphine and diazepam occurs at the level of their intraneuronal signalling pathways. 8 Vodovar et al. have investigated the diazepam/buprenorphine DDI in rats using plethysmography and suggested a possible pharmacodynamic mechanism. 9
This study has limitations, which may question the accuracy of our model to mimic the DDI between buprenorphine and diazepam observed in Human. First, only microdoses of the MOR agonist 11C-buprenorphine could be administered for safety reasons. However, non-linearity was not observed regarding the non-specific brain uptake of 11C-buprenorphine in the presence of co-injected doses of buprenorphine up to 0.03 mg/kg, whereas dose-dependent occupancy of ORs by buprenorphine was observed. 26 This suggests that microdose 11C-buprenorphine PET imaging reliably describes the neuropharmacokinetics of buprenorphine. Moreover, a single dose of buprenorphine and diazepam was tested. It may be hypothesized that lower or higher doses of each drug may have differently impacted the outcome of the study. Nevertheless, the selected doses of buprenorphine and diazepam were higher than those observed in reported cases of death associated with the combination of buprenorphine and diazepam. 7 The resolution of the PET scanner used in this study (∼7 mm) is too low to specifically investigate the binding of 11C-buprenorphine to the small intrapontine loci involved in respiratory control. 43 However, it can be assumed that subregion-specific regulation of MOR, that would not occur in any other MOR-containing brain regions, is very unlikely. Flumazenil is a BzR antagonist, which could be safely co-injected with 11C-flumazenil to investigate the DDI at pharmacological doses. However, this method only provides an insight on the regulation of BzRs and does not inform on the neuropharmacokinetic brain uptake of benzodiazepines such as diazepam used by patients. Finally, general anesthesia using ketamine followed by propofol was required to induce and maintain anesthesia during PET acquisition in non-human primates. PET acquisition in awake macaques is possible, although very limited by technical and ethical issues.44,47 The impact of propofol anesthesia on 11C-buprenorphine PET data is not known. Propofol anesthesia, but not ketamine, was shown to increase the binding of 11C-flumazenil.44,48 The same standardized protocol was used for all experiments, which enables comparison of PET data obtained in control and pretreated animals. However, the presence of anesthesia should be considered for clinical translation of the present PET data. The diazepam/buprenorphine interaction could be further investigated using 11C-buprenorphine and 11C-flumazenil PET imaging in humans, without anesthesia, to confirm the negative results obtained in anaesthetized macaques.
Conclusion
Using a cross-over study PET imaging study in nonhuman primates, our results suggest a limited impact of the regulation of BzR and MOR and do not support a neuropharmacokinetic or receptologic mechanism to explain the DDI between buprenorphine and benzodiazepines. This negative result supports that respiratory depression induced by diazepam/buprenorphine combination may result from a pharmacodynamic interaction induced by the combined effect of each respective drug on ventilation.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
We gratefully thank Jérôme Cayla, Maud Goislard and Vincent Brulon for technical assistance.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SA, DV, SC, BM and NT designed the research; SA, DV, SG, AS and FC performed the research; SA, DV, SC, LC, MB, BM and NT analyzed and interpreted the the data; SA, DV and NT wrote the initial draft of the manuscript. All authors revised the manuscript critically for important intellectual content and approved the submitted version.
