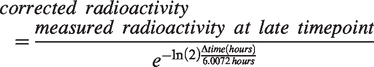Abstract
Many groups have reported lymphatic and glymphatic structures in animal and human brains, but tracer injection into the human brain to demonstrate real-time lymphatic drainage and mapping has not been described. We enrolled patients undergoing standard-of-care resection or stereotactic biopsy for suspected intracranial tumors. Patients received peritumoral injections of 99mTc-tilmanocept followed by planar or tomographic imaging. Fourteen patients with suspected brain tumors were enrolled. One was excluded from analysis because of tracer leakage during injection. There was no drainage of 99mTc-tilmanocept to regional lymph nodes in any of the patients. On average, after correcting for radioactive decay, 70.7% (95% CI: 59.9%, 81.6%) of the tracer in the injection site and 78.1% (95% CI: 71.1%, 85.1%) in the whole-head on the day of surgery remained the morning after, with variable radioactivity in the subarachnoid space. The retained fraction was much greater than expected based on the clearance rate from non-brain injection sites. In this pilot study, the lymphatic tracer 99mTc-tilmanocept was injected into the brain parenchyma, and there was no drainage outside the brain to the cervical lymph nodes. Our work demonstrates an inefficiency of drainage from peritumoral brain parenchyma and highlights a therapeutic opportunity to improve immunosurveillance of the brain.
Abbreviations
Food and Drug Administration
magnetic resonance imaging
single-photon emission computed tomography
Introduction
Significant work over the last several years has advanced our understanding of glymphatic and lymphatic drainage from the brain. 1 In particular, several key preclinical observations have established a mechanistic framework that helps explain the physiology of intracerebral and intraparenchymal fluid flow. Multiple groups have recently demonstrated the presence of the glymphatic system within a perivascular system that clears intraparenchymal waste and solutes towards the subarachnoid space. However, we are still trying to understand the mechanism behind potential antigen movement from the parenchymal tissue to secondary lymphoid structures such as cervical lymph nodes. Specifically, albumin injected into the brain parenchyma drains to cervical lymph nodes in rabbits and mice through a previously unclear mechanism.2,3 However, dural lymphatic vessels have recently been discovered in mice4,5 and humans. 6 In mice, dural lymphatics drain into deep cervical lymph nodes, 5 which may drain CSF in humans.6,7 However, it remains unclear whether antigens from brain tissue—as opposed to CSF—readily drain into lymphatic pathways and secondary lymphoid tissues in humans, or egress from the CNS via other routes to secondary lymphoid structures. To our knowledge, no group has studied drainage of a lymphatic tracer injected directly into human brain parenchyma to explore this question.
99mTc-tilmanocept is a radiopharmaceutical agent used for mapping tumor draining lymph nodes. 8 It is commonly used during sentinel lymph node biopsy in extracranial tumors, especially breast and skin tumors, to determine the presence of local nodal metastatic disease. Patients receive intratumoral or peritumoral injections of 99mTc-tilmanocept before surgical resection of the suspected primary mass. The total injection volume ranges from 0.5 to 5.0 ml. Within 10 minutes to 30 hours after injection, planar or tomographic scintigraphy can identify the sentinel lymph node(s), which appear as foci of increased tracer uptake. 99mTc-tilmanocept binds with high specificity to CD206, the mannose receptor, on macrophages and dendritic cells.8,9 The equilibrium binding constant of 99mTc-tilmanocept for CD206 is 0.12 nM. 10 The average molecular weight of 99mTc-tilmanocept is 17 kDa (range 15.2–23.4 kDa) and the diameter is 7.1 nm. 11 99mTc-tilmanocept is an ideal tracer for lymphatic mapping because it drains quickly from the injection site to lymph nodes with a short clearance half-life relative to its physical half-life. The injection site clearance half-life, a measure of how quickly a substance leaves the injection site, is 2.72 ± 1.57 hours after injection into the breast 10 and 2.17 ± 0.96 hours after injection into the skin, 12 with additional trials showing similar results.13,14 The physical half-life of technetium-99 m is 6 hours. The size of 99mTc-tilmanocept should ostensibly not prohibit drainage from brain tissue, as intraparenchymal injections of the larger ovalbumin molecule (40 kDa) in mice or albumin (66 kDa) in rabbits led to rapid clearance from the interstitial fluid within 2 hours.2,3
In this study, we tested the hypothesis that 99mTc-tilmanocept injected into the brain parenchyma of patients with brain tumors would transit to the cervical lymph nodes, thereby allowing us to map antigen egress into extracranial secondary lymphoid tissues. Here, we show a paucity of tracer movement after injection directly into the brain parenchyma due to a presumed inefficiency of glymphatic and lymphatic drainage.
Materials and methods
Ethics statement
The protocol was approved by the Washington University in St. Louis Human Research Protection Office and the Radioactive Drug Research Committee. The US Food and Drug Administration confirmed that an investigational new drug application was unnecessary for peritumoral injection of 99mTc-tilmanocept in the brain in accordance with 21 CFR 312.2(b). All patients gave written informed consent in accordance with the Declaration of Helsinki.
Patients
Eligible patients were at least 18 years old and candidates for neurosurgical resection or stereotactic biopsy of suspected benign or malignant tumors. Exclusion criteria included dextran hypersensitivity, pregnancy, and prior resection of head or neck lymph nodes. Fourteen patients were enrolled between January 2021 and April 2022. One patient was withdrawn before imaging due to leakage during tracer injection. The study was registered at clinicaltrials.gov (NCT04096716).
Patients 1–6 and 10 were injected with 0.5 mCi 99mTc-tilmanocept (50 µg mass dose). Patients 7–9 and 11–13 received a dosage of 1 mCi (same mass dose).
Twelve patients received injections of 99mTc-tilmanocept 1 cm into tumor-adjacent brain parenchyma at 4–5 locations assessed as safe by the operating surgeon after complete resection. Injections were completed over the course of 10 seconds, with the needle held in place for an additional 20 seconds, to prevent backflow out of the injection site, prior to proceeding to the subsequent injection sites. The volume of each injection was 100 µL, with a total injection volume of 400–500 µL per patient. A 25-gauge tuberculin needle was used for all injections.
One patient received stereotactic-guided injection into tumor-adjacent brain following biopsy. In this patient, 200 µL of 99mTc-tilmanocept was injected over 4 minutes through the same needle used to complete the stereotactic biopsy. The needle was held in place for an additional 5 minutes to prevent backflow out of the injection site prior to removing the needle.
Imaging
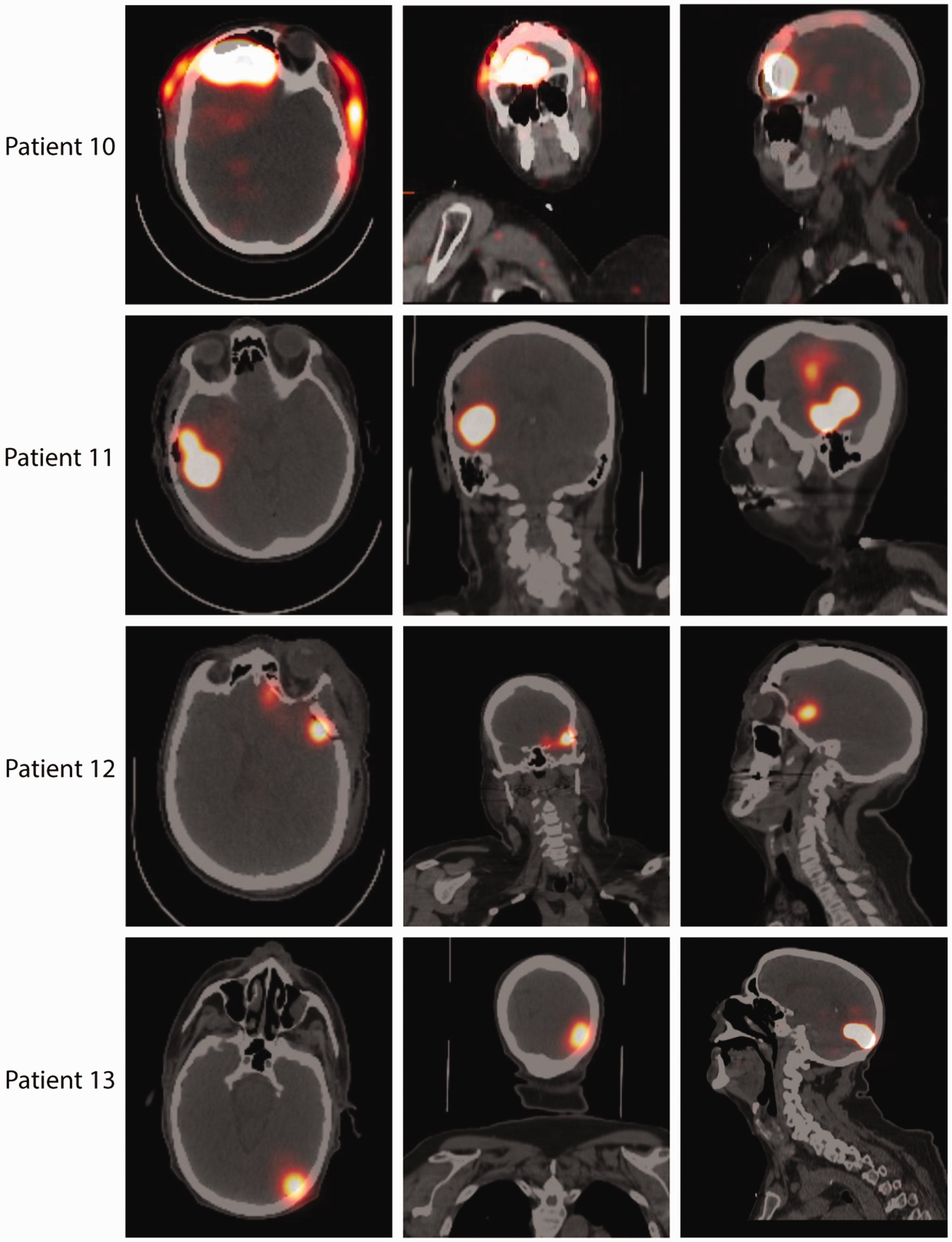
Planar lymphoscintigraphy was performed in 6 patients within 24 hours of surgery using a portable scintillation camera (Digirad Ergo Imaging). The first images were obtained as soon as the patient was clinically stable to undergo imaging on the day of surgery. The second timepoint was taken on the morning after surgery. We used a 5-minute acquisition per view (typically anterior and lateral projections) for the early timepoint, and a 10-minute acquisition for the late timepoint. We analyzed radioactivity counts per minute with MIM v6.9.3. Late timepoint counts were adjusted for physical decay:
We imaged the subsequent cohort of 7 patients with single-photon emission computed tomography/computed tomography (SPECT/CT) within 24 hours of injection using a GE Discovery NM/CT 670 Pro CZT scanner.
Immunohistochemistry
Peritumoral brain tissue sections were obtained at surgery and noted by the neurosurgeon to be from the enhancing section of tumor on preoperative MRI or from the non-enhancing adjacent white matter. The sections were stained with 4′,6-diamidino-2-phenylindole (DAPI, Invitrogen) and anti-CD206/MRC1 rabbit IgG mAb (clone E2L9N, Cell Signaling Technologies). Donkey anti-rabbit IgG pAb conjugated to Alexa Fluor 647 was used for secondary staining (clone ab 150075, abcam). Images were assessed using a Zeiss LSM 880 microscope.
Results
We enrolled 14 patients with suspected brain tumors but excluded 1 from analysis because of tracer leakage. Six patients were female (46%) and the median age was 64 (interquartile range 48-68 years). No patients experienced an adverse event.
Lymphoscintigraphy of the brain shows no drainage to head or neck lymph nodes
On planar lymphoscintigraphy of the first 6 patients, we saw no drainage of 99mTc-tilmanocept from the injection site to lymph nodes on the day of surgery or the morning after (Figure 1(a)). However, variable activity was apparent in the subarachnoid space, particularly on the late images presumably due to back-leakage through the needle tracks into the resection cavity or perivascular clearance towards the subarachnoid space though the glymphatic system. The raw tracer signal counts decreased between the first and second time points (Figure 1(b)), which, as expected, was mostly due to radioactive decay. Based on the reported injection site clearance half-life of 2.72 hours, 10 we expected a mean remaining fraction of 1.12% (95% CI: 1.07%, 1.17%) after accounting for radioactive decay. Surprisingly, after accounting for expected radioactive decay, a mean of 70.7% (95% CI: 59.9%, 81.6%) of radioactivity in the injection site and 78.1% (95% CI: 71.1%, 85.1%) in the whole head on the day of surgery remained the following morning. Thus, the results suggest that the tracer leaves the brain inefficiently.
Planar and SPECT/CT images after 99mTc-tilmanocept injection. (a) Planar images for each patient are shown from left to right: anterior day of surgery, anterior day after surgery, right lateral day of surgery, right lateral day after surgery. Patient 1: Female, 40 years old, right frontal lobe 99mTc-tilmanocept injection, radiation gliosis without neoplasm; Patient 2: Male, 72 years old, right temporal lobe injection, lung metastasis; Patient 3: Male, 67 years old, right frontal lobe injection, anaplastic oligodendroglioma; Patient 4: Male, 48 years old, right parietal injection, glioblastoma; Patient 5: Female, 68 years old, right frontal lobe injection, lung metastasis; and Patient 6: Male, 21 years old, right frontal lobe injection, low grade astrocytoma. (b) Graph of raw counts per minute (not decay corrected) for all images (anterior and lateral) of patients 1–6, each line represents change in radioactivity in the whole head and (c) SPECT/CT axial, coronal, and sagittal images are shown for each patient. Patient 7: Male, 51 years old, right parietal lobe melanoma metastasis, 3.5 hr after injection; Patient 8: Female, 86 years old, right frontal lobe breast carcinoma metastasis, 5 hr after injection; Patient 9: Male, 64 years old, right parietal lobe glioblastoma, 18.5 hr after injection.
Next, we considered that planar lymphoscintigraphy may not be sensitive enough to detect minimal drainage to lymph nodes, so we imaged 3 additional patients with suspected intraparenchymal malignancy using SPECT/CT, and again observed no detectable signal in lymph nodes regardless of timepoint or disease pathology (Figure 1(c)).
99mTc-tilmanocept is not sequestered by a dense population of CD206+ cells
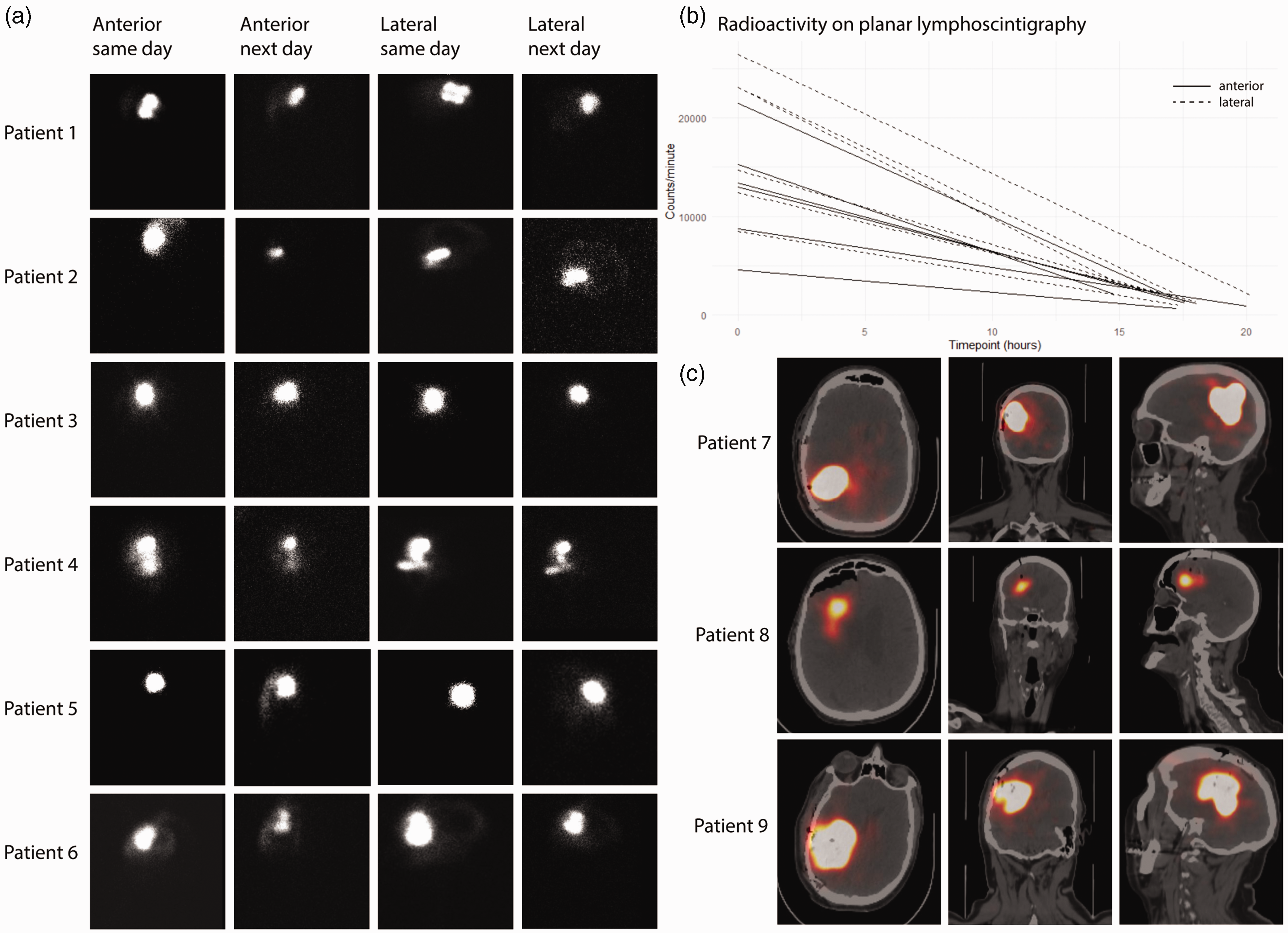
Given the high retention of the tracer signal, we investigated whether cells expressing CD206 were present at increased frequency in the peritumoral brain parenchyma and, therefore, able to sequester 99mTc-tilmanocept. With high specificity, tilmanocept co-localizes with antibody-stained CD206 on cell surfaces. 9 Therefore, we stained tissue sections from a resected glioma and associated adjacent white matter removed as part of the surgery with anti-CD206. Notably, the adjacent white matter was from the non-enhancing rim of tissue at the most shallow aspect of the resection cavity. Any injection of 99mTc-tilmanocept would be 1 to 1.5 cm deeper than the adjacent white matter we sampled, and, thus, even less likely to contain infiltrative tumor. We saw CD206+ cells within the tumor, but not within the adjacent white matter (Figure 2(a) to (d)).
Presence of CD206 in the brain. (a) Normal human tonsil (positive control, Novus Biologicals) stained with DAPI (blue) and anti-CD206 (magenta). (b) Glioblastoma tumor stained with DAPI (blue) and anti-CD206 (magenta). (c and d) Adjacent white matter stained with DAPI (blue) and anti-CD206 (magenta). (e) Tissue gene expression of MRC1 (normalized transcripts per million) from the Human Protein Atlas (mean nTPM for Cerebral Cortex: 0.669, White Matter 0.506, Breast: 16.3, Skin: 2.1, Lymph Node: 15.2, and Spleen: 50.6) and (f) Tissue sections stained for CD206 from the Human Protein Atlas.
To corroborate this finding, we interrogated the Human Protein Atlas to examine the expression patterns of the MRC1 gene (which encodes CD206) and the CD206 protein. 15 We confirmed that expression of the MRC1 gene and CD206 protein in the cerebral cortex and white matter was minimal (Figure 2(e) and (f)). Furthermore, MRC1 gene expression and CD206 protein expression in the brain was less than in breast and skin tissue, two sites that are commonly injected with 99mTc-tilmanocept. As expected, the sequencing data and tissue sections, respectively, showed robust expression of MRC1 or C206 in the lymph node and spleen samples. Together, these data suggest that 99mTc-tilmanocept is likely not sequestered at the injection site by CD206 expression.
99mTc-tilmanocept drainage to lymph nodes does not improve in the absence of parenchymal disease or after minimally invasive surgery
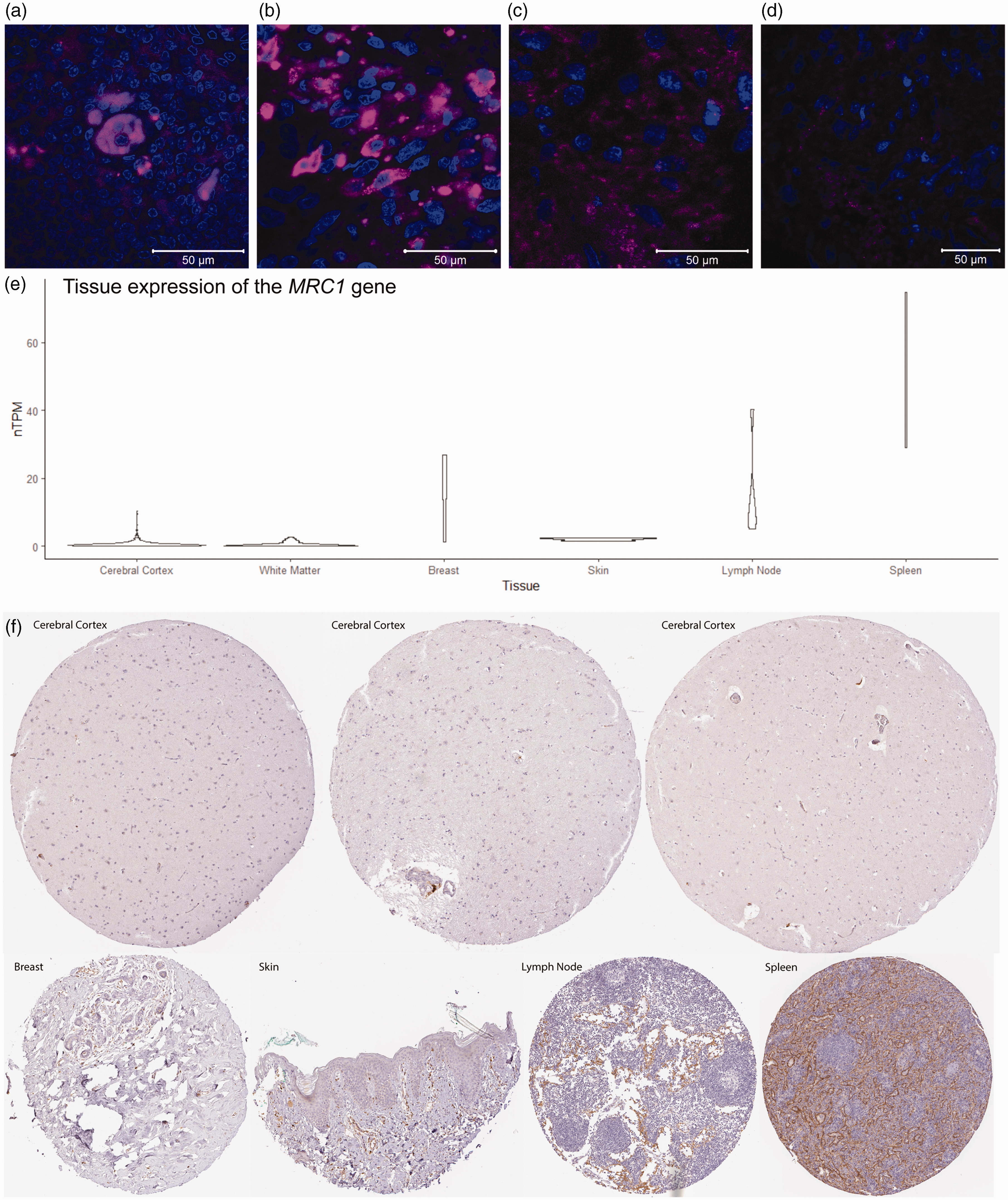
We considered that infiltrative, intrinsic brain tumors may perturb glymphatic flow. Therefore, 3 patients with meningioma, a benign tumor of the dura that does not invade the brain parenchyma, were enrolled. Again, we saw no drainage to lymph nodes following peritumoral injection of the unaffected adjacent brain parenchyma (Figure 3). Based on prior evidence demonstrating reduced glymphatic flow following craniectomy, we considered that the craniotomy procedure itself may impair lymphatic drainage. 16 We observed no drainage beyond the injection site, including no drainage to cervical lymph nodes, in patient 13 who underwent stereotactic needle biopsy with minimal anatomic perturbation (Figure 3).
SPECT/CT axial, coronal, and sagittal images after resection or biopsy and 99mTc-tilmanocept injection. Patient 10: Female, 40 years old, right frontal lobe meningioma, 20 hours after tumor resection and injection, a small amount of tracer is visible extracranially, likely in the subgaleal space; Patient 11: Female, 65 years old, right temporal meningioma, 4 hours after tumor resection and injection; Patient 12: Female, 54 years old, right frontotemporal meningioma, 16.25 hours after tumor resection and injection; Patient 13: Male, 65 years old, left occipital glioblastoma, 3.5 hours after stereotactic biopsy and injection.
Discussion
Here we report the first study in which a lymphatic tracer was injected directly into human brain parenchyma. We tested the hypothesis that 99mTc-tilmanocept would egress from brain tissue to extracranial secondary lymphoid tissues, thereby mapping draining lymph nodes from the brain. Surprisingly, 99mTc-tilmanocept migration from the injection site to regional lymph nodes was absent, or at least not detectable by planar scintigraphy or SPECT/CT. The basis for this observation is unclear. Our results suggest that the lack of drainage was not due to sequestration by CD206+ cells, perturbation of flow by malignancy, or altered flow from invasive surgical technique.
We observed some 99mTc-tilmanocept egress from the parenchyma presumably through CSF and venous drainage. 99mTc-tilmanocept could have cleared from the parenchyma through a combination of glymphatic drainage to CSF and blood-brain-barrier breakdown around the tumor with subsequent venous drainage. It is possible that a perioperative, peritumoral infiltration of macrophages or dendritic cells may clear a portion of the 99mTc-tilmanocept, but this is less likely because of the lack of CD206+ cells in the adjacent white matter; cells lacking this receptor would be unlikely to internalize the tracer. Nevertheless, although a modest amount of 99mTc-tilmanocept left the injection site, significantly more tracer was retained than we expected. This dearth of drainage is highly distinct from that seen at extracranial sites with intact lymphatics. The only reports of an absence of 99mTc-tilmanocept drainage to lymph nodes are in patients with damaged lymphatics and subsequent lymphedema.17–20 In comparison, following peritumoral injection at extracranial sites such as the skin, breast, or oral cavity, 99mTc-tilmanocept drains to lymph nodes within minutes.8,11,14,21
We could not detect how much, if any, 99mTc-tilmanocept leaves the brain immediately post-injection because of the necessary delay, for patient safety, between the intraoperative injection and the first scan. However, the rate of clearance from the brain is nonetheless clearly slower than expected when compared to the rate of clearance from extracranial soft-tissue injection sites. We recognize that the study was limited to imaging within 24 hours of surgery because of the 6-hour half-life of technetium-99 m. In this trial using 99mTc-tilmanocept lymphoscintigraphy, radioactivity rapidly decays to levels that would not permit detection of slow drainage to lymph nodes at a later timepoint. Certain properties of any tracer, including 99mTc-tilmanocept, may limit its usefulness in studying glymphatic and lymphatic drainage from the brain. Although 99mTc-tilmanocept is a relatively small molecule, even smaller molecules may traffic more quickly through the glymphatic system. However, a key limitation with an extremely small molecule in studying the connection from the intraparenchymal compartment to cervical lymph nodes is maintaining a tracer that is detectable noninvasively but is still a ligand for a receptor within lymph nodes. Smaller tracers also may not accurately model the movement, or lack thereof, of larger molecules, such as tumor antigens. Indeed, the potential need for a smaller tracer to study glymphatic flow as opposed to lymphatic drainage from extracranial sites may speak to the relative inefficiency of glymphatic and lymphatic drainage from the brain compared to extracranial lymphatics. This may help explain why extracranial tumors frequently metastasize, whereas metastasis of brain cancers is rare.
In comparison, intrathecal gadobutrol, a gadolinium-based magnetic resonance imaging (MRI) contrast, has a molecular weight of 604 Da and lacks a binding target within cervical lymph nodes. 7 Gadolinium-based contrast agents are commonly used for monitoring tumor progression in clinical practice because they enter lesions with blood-brain-barrier breakdown, such as tumors. When injected intrathecally, gadobutrol may drain to cervical lymph nodes. 7 To our knowledge, intraparenchymal administration of gadolinium-based contrast has not been reported. We believe that such a small molecule would eventually be cleared after intraparenchymal injection, but the safety of this would need to be established before MRI could be evaluated as an alternative method to study this phenomenon. We envision that a tracer with a longer physical half-life may show drainage at later timepoints, but assessing these kinetics is not possible with currently available FDA-approved technetium-99 m tracers. We believe that our work shows that intraoperative intraparenchymal injection is a potentially useful and safe route of administration for other tracers to study glymphatic clearance in humans.
Although we did not observe significant tracer transit outside of the brain, glymphatic and lymphatic structures within the brain have been described definitively within the last several years.4–6,22 The scarcity of drainage observed in our study is consistent with a previous report that CSF drainage to cervical lymph nodes in humans is much slower than in animals 7 and suggests that drainage through these systems may be inefficient—at least, in the clinical setting in which a brain tumor is present. Further, surgical intrastriatal tracer administration in rats impairs global glymphatic function for at least 2 hours. 23 It may be possible that intraparenchymal injection in humans impairs glymphatic function for at least 24 hours and, thus, our results must be applied to the natural state of glymphatic clearance of waste from the neuropil with caution. Other methods and/or technologies may be required to accurately characterize these systems in the human brain.
The introduction of tracer in the peritumoral region immediately following resection is a very close approximation of how tumor antigens, in this particular setting, may be expected to transit. The presumed inefficient lymphatic drainage therefore highlights a potential therapeutic opportunity to enhance brain immunosurveillance that may be relevant for brain tumors as well as other CNS pathologic states. Indeed, recent data suggest that augmentation of lymphatic vasculature in mouse brains improves antitumor immunity. 24 Thus, increasing interstitial fluid flow towards lymphatics may potentiate endogenous priming of tumor specific immune responses and enhance immunotherapy strategies in brain tumors and other diseases. Ultimately, the dynamics of how antigens and molecules may move from the brain parenchyma remains unclear in humans, and thus our study represents one approach to begin exploring these questions. Further work is needed to understand the basis of molecular transit in the brain and its implications for CNS immunity.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Cancer Research Institute Lloyd J. Old STAR Award. ATC was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number TL1TR002344. TMJ was supported by the National Cancer Institute of the National Institutes of Health under Award Number K12 CA167540. This study used the Siteman Cancer Center Imaging and Response Assessment Core supported in part by NCI Grant number P30 CA91842. 99mTc-tilmanocept was provided by Cardinal Health through the Investigator Initiated Study program.
Acknowledgements
The authors thank Jennifer Frye, CNMT and Austin Turner, PhD, CNMT for coordination of imaging procedures, and David Schwab, BA and Konstantina Stavroulaki, MS for subject recruitment and data collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
ATC wrote the study protocol. ATC, RD, BAS, TMJ, and GPD revised the protocol. ATC, AKV, JTW, JLD, ECL, AHK, RD, and GPD performed or provided intraoperative oversight of the 99mtechnetium tilmanocept injections (under the supervision of BAS as the authorized user of the radiopharmaceutical). PRP performed the IHC staining and imaging. ATC and RD acquired tissue and supervised injections. ATC, RD, and BAS analyzed lymphoscintigraphy images. All authors contributed to the discussion of results and writing of the manuscript. GPD had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.