Abstract
Leukocyte common antigen-related phosphatase (LAR) is widely expressed in the central nervous system and is known to regulate a variety of processes including cell growth, differentiation, and inflammation. However, little is currently known about LAR signaling mediated neuroinflammation after intracerebral hemorrhage (ICH). The objective of this study was to investigate the role of LAR in ICH using autologous blood injection-induced ICH mouse model. Expression of endogenous proteins, brain edema and neurological function after ICH were evaluated. Extracellular LAR peptide (ELP), an inhibitor of LAR, was administered to ICH mice and outcomes were evaluated. LAR activating-CRISPR or IRS inhibitor NT-157 was administered to elucidate the mechanism. The results showed that expressions of LAR, its endogenous agonist chondroitin sulfate proteoglycans (CSPGs) including neurocan and brevican, and downstream factor RhoA increased after ICH. Administration of ELP reduced brain edema, improved neurological function, and decreased microglia activation after ICH. ELP decreased RhoA and phosphorylated serine-IRS1, increased phosphorylated tyrosine-IRS1 and p-Akt, and attenuated neuroinflammation after ICH, which was reversed by LAR activating-CRISPR or NT-157. In conclusion, this study demonstrated that LAR contributed to neuroinflammation after ICH via RhoA/IRS-1 pathway, and ELP may be a potential therapeutic strategy to attenuate LAR mediated neuroinflammation after ICH.
Introduction
Intracerebral hemorrhage (ICH) has an annual incidence of 25 per 100000 1 and it is associated with high mortality and morbidity. 2 However, there is a lack of specific treatments to reduce the impact of secondary brain injury after ICH. Recent research efforts are being directed toward reducing secondary injury after ICH as effective treatment options. Neuroinflammation is one of the important factors that contributes to the progression of secondary brain injury. 3
Leukocyte common antigen-related phosphatase (LAR), also named Receptor-type tyrosine-protein phosphatase F (PTPRF), is a member of protein tyrosine phosphatase (PTP) family. This receptor is a signaling molecule comprised of an extracellular region, a single transmembrane region, and two tandem intracytoplasmic catalytic domains. 4 LAR has been reported to be expressed on neurons and microglia.5–7 Previous studies have shown that overactivation of LAR aggravated inflammation in spinal cord injury (SCI) mice model.6,8 However, whether inhibition of LAR has potential anti-neuroinflammatory function after ICH remains unknown.
Extracellular LAR peptide (ELP) is known to block LAR by binding to the fifth LAR fibronectin type III (FN-III) domain. 9 It was reported that deletion of LAR eliminated chondroitin sulfate proteoglycans (CSPGs)-induced Akt inactivation and RhoA activation in cerebellar granule neuron (CGN) cultures. Whereas systemic treatments with ELP and intracellular LAR peptide (ILP) improved locomotor recovery in SCI mice. 4 LAR signaling was found to regulate microglia by activating the RhoA/ROCK pathway. 6 Activation of RhoA/ROCK pathway was found to block muscle differentiation via increased serine phosphorylation of IRS-1/2, followed by decreases in tyrosine phosphorylation of IRS-1/2. 10 In addition, LAR reduced tyrosine phosphorylation of IRS-1 and its downstream molecule Akt. 11
Thus, the aim of this study was to investigate whether administration of ELP could attenuate neuroinflammation and thereby improve neurological outcomes after ICH in a mouse model and to determine the potential underlying anti-inflammatory mechanisms of ELP.
Materials and methods
Animals and experimental design
All the experimental protocols and procedures of this study were approved by the Institutional Animal Care and Use Committee at Loma Linda University and complied with the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals. All experiments were reported with the ARRIVE guidelines (Animal Research: Reporting in Vivo Experiments). A total of 220 adult male CD1 mice (8-week-old, weight 30–40 g; Charles River, Wilmington, MA, USA) were used in the study. The mice were housed in a temperature and humidity-controlled room for a minimum of 3 days before surgery with a 12-hour light/dark cycle and ad libitum access to food and water.
All mice were randomly assigned to different groups and the following experiments were conducted.
Experiment 1
The time course expressions of LAR, its endogenous ligand CSPGS including Neurocan and Brevican and downstream RhoA was characterized at different time points after ICH using western blot. The ipsilateral hemispheres of mice were collected for analysis from sham and ICH mice at 3 h, 6 h, 12 h, 24 h, 72 h after injury. Twenty-four mice were randomized into these 6 groups: sham (n = 4), ICH–3 h (n = 4), ICH-6 h (n = 4), ICH-12 h (n = 4), ICH-24 h (n = 4) and ICH-72 h (n = 4). Immunofluorescence was performed to evaluate the cellular localization of LAR in ICH-24 h mice (n = 3).
Experiment 2
This experiment was conducted to evaluate the beneficial effects of ELP in ICH model. ICH mice were randomly assigned to receive either vehicle (DMSO) or ELP which was given intranasally 1 h after injury. Neurobehavioral tests, brain water content and hematoma volume were evaluated at 24 h and 72 h after ICH. For 24 h outcome study, 30 mice were randomized into the following 5 groups: sham (n = 6), ICH + vehicle (n = 6), ICH + ELP 0.04 μg/g (n = 6), ICH + ELP 0.12 μg/g (n = 6) and ICH + ELP 0.36 μg/g (n = 6) to evaluate neurobehavior and brain water content at 24 h after ICH. The different dosages of ELP were used in accordance with a study in SCI model. 4 Additional 30 mice were assigned into the 5 groups described above to evaluate hematoma volume at 24 h after ICH. To determine the effects of ELP at 72 h after ICH, 18 mice were randomized into 3 groups: sham (n = 6), ICH + vehicle (n = 6), ICH + ELP 0.12 μg/g (n = 6), and neurobehavior and brain water content was evaluated at 72 h after ICH. Additional 18 mice were assigned into these 3 groups to evaluate hematoma volume at 72 h after ICH. Based on the results of 24 h outcome studies, the dosage of ELP 0.12 μg/g was chosen for 72 h outcomes and further studies.
Experiment 3
To assess the effects of ELP administration on microglia activation, 18 mice were randomly divided into three groups for immunofluorescence staining: sham (n = 6), ICH + vehicle (n = 6), ICH + ELP 0.12 μg/g (n = 6).
Experiment 4
To determine the effects of ELP on long-term neurobehavioral functions after ICH, 24 mice were randomly assigned to 3 groups: sham (n = 8), ICH + vehicle (n = 8) and ICH + ELP 0.12 μg/g (n = 8). Foot fault test and rotarod test were carried out on days 7, 14 and 21 after ICH. Morris water maze test was conducted on days 21–25 after ICH.
Experiment 5
To investigate the underlying mechanism of ELP/LAR mediated protective effects after ICH, 42 mice were randomized into 7 groups: sham (n =6), ICH + vehicle (n = 6), ICH + ELP 0.12 µg/g (n = 6), ICH + ELP 0.12 ug/g + Scramble CRISPR (n = 6), ICH + ELP 0.12 ug/g + LAR-activating CRISPR (n = 6), ICH + ELP 0.12 ug/g + DMSO (n = 6) and ICH + ELP 0.12 ug/g + NT157 (n = 6). The scramble CRISPR or LAR-activating CRISPR (2 μg in 2 μl, Santa Cruz Biotech, Santa Cruz, CA) were injected via intracerebroventricular (i.c.v) route 48 h before ICH. The IRS inhibitor NT157 (50 mg/kg, Selleck Chemicals, Houston, TX) or DMSO was injected via intraperitoneal (i.p.) route 1 h before ICH.
ICH model
The autologous blood injection ICH model in mice was adapted from previously described studies.12,13 Briefly, the animals were anesthetized with a mixture of ketamine (100 mg/kg) and xylazine (10 mg/kg) (2:1, intraperitoneal injection) and then positioned on a stereotaxic frame (Kopf Instruments, Tujunga, CA). Artificial tear ointment (Rugby, Livonia, MI) was applied to keep the eyes moist during surgery. A cranial burr hole (1 mm) was drilled 2.2 mm right lateral and 0.2 mm anterior to the bregma. Autologous blood was collected from the right femoral artery and a total of 30 μl blood without anticoagulant was injected into the brain using a 1 ml syringe. The needle of the syringe was inserted to the right basal ganglia through the burr hole. The needle was first inserted to 3 mm ventral depth and 5 μl blood was injected at a rate 2 μl/min after which the needle was held in place for 5 min. Next, the needle was further inserted to a depth of 3.5 mm and the remaining 25 μl blood was then delivered at a rate of 2 μl/min. The needle was left in place for 10 min to prevent reflux and then withdrawn slowly. Bone wax was applied to seal the cranial burr hole and the scalp was sutured. Normal saline 0.4 ml was delivered subcutaneously to avoid postsurgical dehydration. Animals in sham groups were subjected to needle insertion only without blood injection following the same surgical procedure.
Western blotting
Mice were transcardially perfused with 50 ml of ice-cold phosphate-buffered saline (PBS, pH 7.4). The brains were collected. Ipsilateral hemispheres were homogenized in Ripa lysis buffer and centrifuged to collect the supernatant samples. Protein concentration was determined by detergent compatible assay (DC protein assay, Bio-Rad laboratories, CA). Equal amount of protein was then loaded and separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to the nitrocellulose membrane. After blocking with 5% nonfat milk or bovine serine albumin (BSA, Sigma, Burlington, MA), the membranes were incubated overnight at 4°C with primary antibodies: sheep anti-Neurocan (1 μg/ml, R&D, System, Minneapolis, MN), sheep anti-Brevican (0.1 μg/ml, R&D, System, Minneapolis, MN), Biotin-conjugated anti-LAR (1:1000, Seattle, WA), rabbit anti-RhoA (1:1000, CST, Danvers, MA), rabbit anti-IRS1 (1:1000, CST, Danvers, MA), rabbit anti-p-IRS1 (Ser307) (1:1000, CST, Danvers, MA), rabbit anti-p-IRS1 (Tyr896) (1:1000, Abcam, Cambridge, MA), rabbit anti-Akt (1:2000, CST, Danvers, MA), rabbit anti-p-Akt (1:2000, CST, Danvers, MA), rabbit anti-IL-1β (1:1000, Abcam, Cambridge, MA), rabbit anti-IL-10 (1:1000, Abcam, Cambridge, MA) and mouse anti-actin (1:5000, Santa Cruz, Santa Cruz, CA). The membranes were then processed with corresponding secondary antibodies (1:3000, Santa Cruz Biotech, Santa Cruz, CA) or HRP-conjugated Streptavidin (1:8000, SouthernBiotech, Birmingham, AL) for 2 h at room temperature. The bands were probed with an ECL Plus chemiluminescence regent Kit (Amersham Biosciences, Arlington Heights, PA) and visualized with the image system (Bio-Rad, Versa Doc, model 4000). Relative density of the protein immunoblot images were analyzed by ImageJ software (Image J 1.4, NIH, USA).
Immunofluorescence staining
Mice were perfused with ice-cold PBS followed by 10% formalin at 24 h after ICH. The whole brains were fixed in formalin for 24 h and then dehydrated in 30% sucrose until saturation. The brains were cut into 10 μm-thick coronal sections using a cryostat (CM3050S; Leica Microsystems). The brain sections were blocked with 5% donkey serum albumin at room temperature for 2 h followed by incubation with primary antibodies including rabbit anti-Iba-1 (1:400, Wako, Chuo-ku, Osaka, Japan), rabbit anti-NeuN (1:200, Abcam), rabbit anti-GFAP (1:100, Abcam), mouse anti-LAR (1:100, BD Biosciences, San Jose, CA) and rat anti-CD86 (1:200, BD Biosciences) at 4°C overnight. The sections were rinsed three times with PBS, and then incubated with appropriate secondary antibodies (1:200, Jackson Immunoresearch, West Grove, PA) at room temperature for 2 h. The perihematomal areas of stained sections were examined under a fluorescence microscope (Leica Microsystems, Germany).
Intranasal administration
Intranasal (i.n.) administration was performed as previously described. 13 ELP (GenScript, Piscataway, NJ) at three different dosages dissolved in DMSO was administered intranasally. A total volume of 20 μl was delivered into the bilateral nares, alternating one naris at a time, with 5 μl per naris every 5 min for a period of 20 min.
Intracerebroventricular administration
Intracerebroventricular administration was performed as previously described. 14 Briefly, a 1 mm burr cranial hole was drilled into the skull at the following co-ordinates relative to bregma: 0.3 mm posterior, 1.0 mm left lateral, and 2.3 mm deep. Then a 26-gauge needle of a 10-μl Hamilton syringe was inserted into the left lateral ventricle through the cranial hole and infusion was conducted at a rate of 0.667 μl/min. The needle was left in place for an additional 8 min after the end of infusion and then removed slowly. The burr hole was sealed with bone wax.
Brain water content measurement
Brain edema was assessed by measuring brain water content using wet/dry method as previously described. 15 Briefly, mice were sacrificed under anesthesia without cardiac perfusion at 24 h or 72 h after surgery. The brains were removed quickly and divided into five parts, namely, ipsilateral and contralateral cortices (Ipsi-CX and Cont-CX), ipsilateral and contralateral basal ganglia (Ipsi-BG and Cont-BG) and cerebellum (Cerebel). The wet weight of each part was measured immediately, and the dry weight was obtained after the brain tissue had been dried at 100°C for 24 h. Brain water content was calculated as [(wet weight – dry weight)/wet weight] × 100%.
Neurobehavioral function assessment
Neurobehavioral tests were performed as previously reported.13,16 Short-term neurobehavior was evaluated at 24 h and 72 h after ICH by conducting Garcia test, corner turn test and limb placement test. Long-term neurobehavioral tests consisted of foot fault test, rotarod test and Morris water maze. The foot fault and rotarod tests were performed at the first, second and third week after ICH to evaluate sensorimotor coordination and balance. Morris water maze test was conducted on days 21 to 25 after ICH to assess memory and spatial learning abilities. The escape latency, swim distance, probe quadrant duration and swim path were video recorded and analyzed.
Hematoma volume
Hematoma volume was measured as previously described. 17 Briefly, mice were transcardially perfused with ice-cold PBS at 24 h after injury. The brains were removed and divided into left and right hemispheres and preserved at −80°C until further analysis. The right hemispheres were homogenized and then centrifuged at 15000 r, 4°C for 30 minutes and the supernatant was collected. Drabkins reagent (Sigma Aldrich, St. Louis, MO) 0.4 ml was added to 0.1 ml supernatant mixed with and allowed to react for 15 minutes at room temperature in dark. The volume of hematoma was determined by a spectrophotometer (GENESYS 10S UV-Vis; Thermo Fisher Scientific) and quantified according to a standard curve.
Statistical analysis
All data were expressed as Mean ± SD. Statistical analysis was performed using GraphPad Prism v. 6.02. The data was analyzed with One-way ANOVA followed by Tukey or Two-way ANOVA followed by Bonferroni post-test was used to determine the differences among groups. Differences were considered significant at p < 0.05.
Results
Animal mortality and exclusion
All sham-operated mice survived in this study. The overall animal mortality was 4.5% (10/220). The mortality was not significantly different among the experimental groups. There were 3 mice excluded from this study due to no hemorrhage.
Time course expressions of CSPGs, LAR and RhoA after ICH
Western blot results showed that the expression of endogenous CSPGs (Neurocan and Brevican) increased from 3 h after ICH. The expression of Neurocan peaked at 24 h while Brevican level kept increasing over 72 h (Figure 1(a) to (c)). The results also showed significant increases in expressions of LAR and RhoA levels after ICH, which peaked at 24 h (Figure 1(a), (d) and (e)).
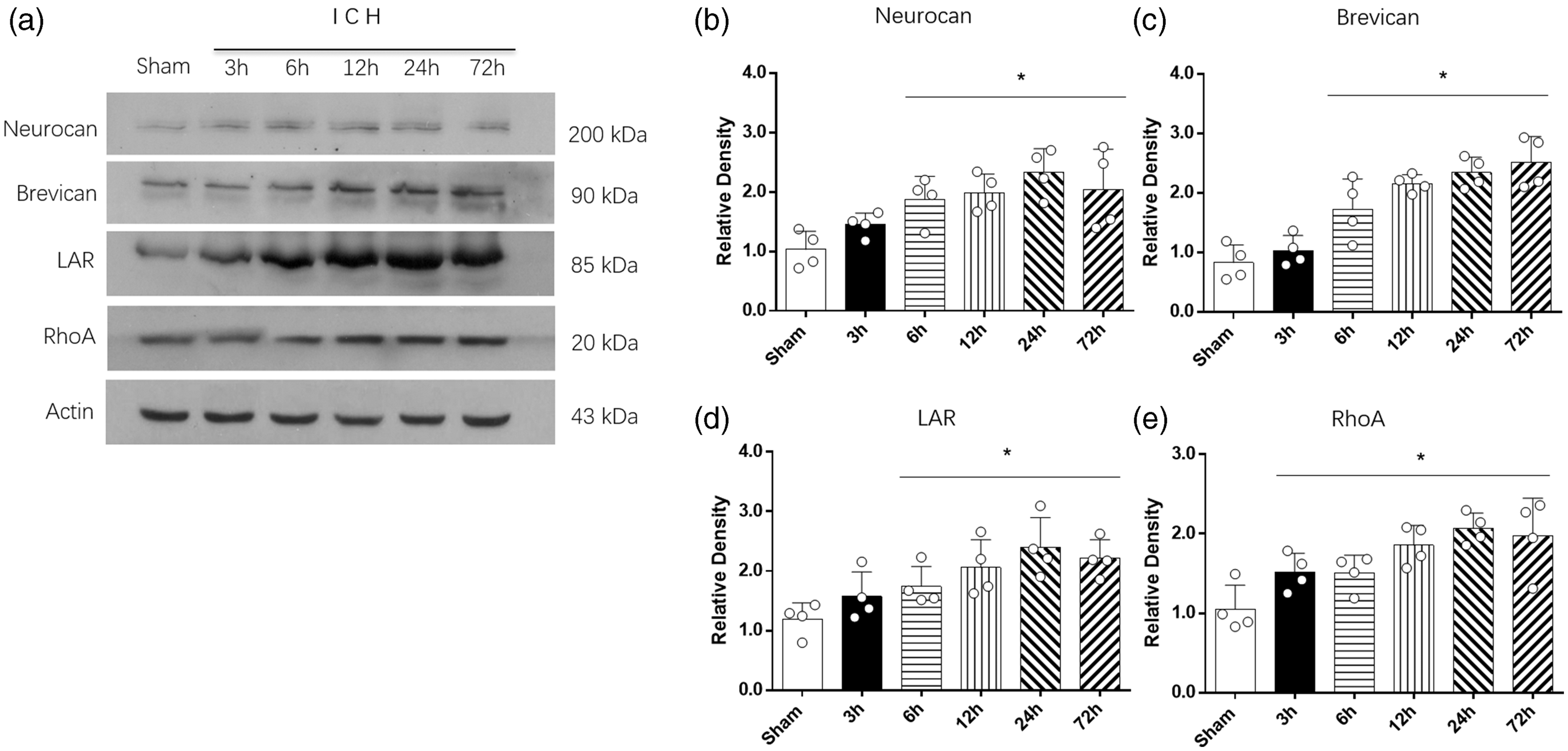
Time course expressions of CSPGs, LAR and RhoA after ICH. (a) Representative western blot bands and quantitative analyses of the time course expressions of (b) neurocan, (c) brevican, (d) LAR and (e) RhoA expression at 3 h to 72 h after ICH. *p < 0.05 vs. sham. Error bars are represented as mean ± SD, n = 4 mice per group.
Cellular localization of LAR after ICH
Double immunofluorescence staining was performed to detect the localization of LAR on microglia/macrophage (Iba-1), neurons (NeuN) and astrocytes (GFAP) at 24 h after ICH. LAR was found to co-localize with both microglia (Iba-1) and neurons (NeuN) at 24 h after ICH but there was no expression of LAR found on astrocytes (Figure 2).
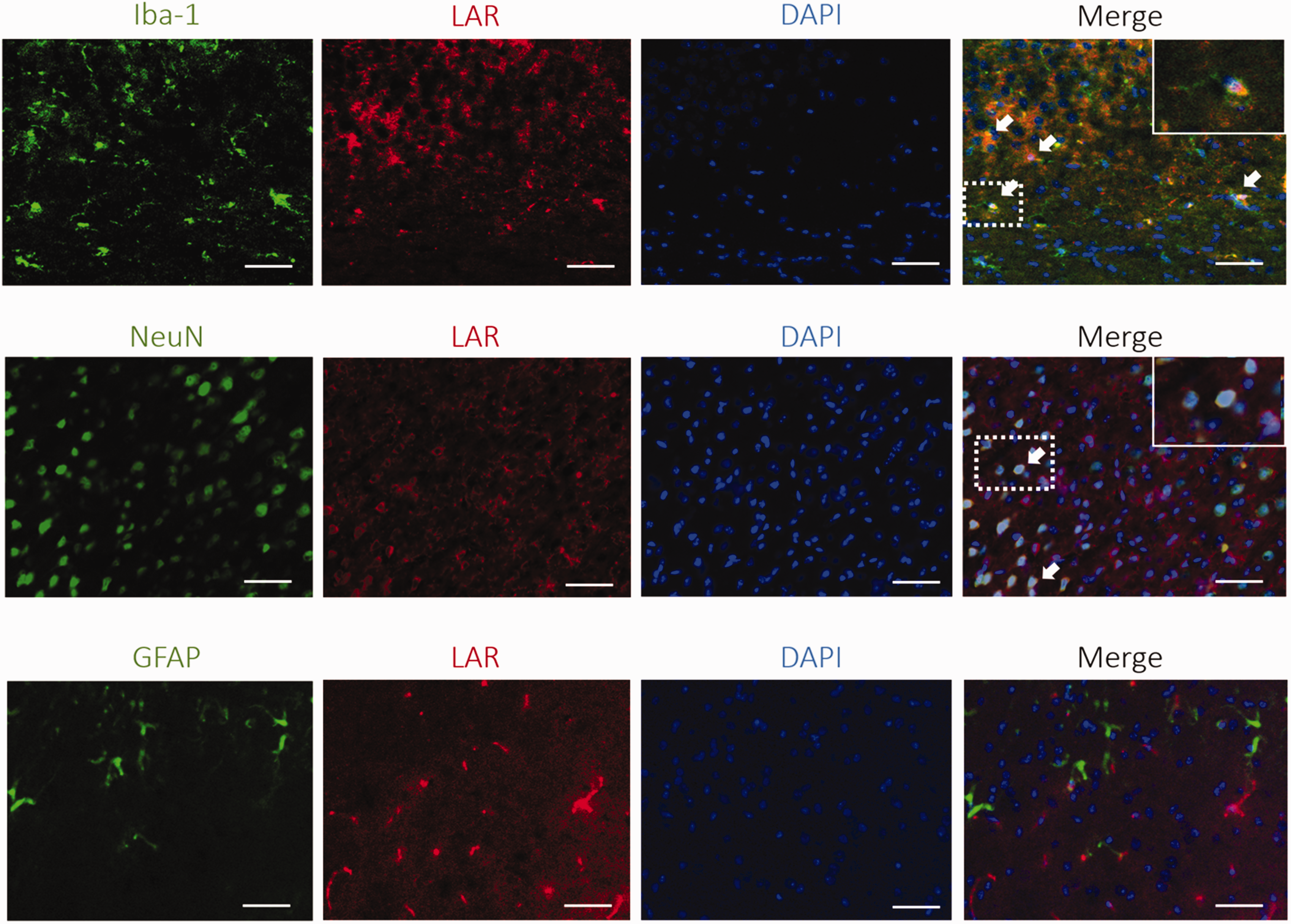
Cellular colocalization of LAR at 24 h after ICH. Co-localization of LAR (red) with microglia (Iba-1, green), neurons (NeuN, green) and astrocytes (GFAP, green) at 24 h after ICH. Scale bar = 50 μm, n = 3.
Administration of ELP alleviated short-term neurobehavioral deficits and brain edema at 24 h after ICH
ICH animals performed significantly worse than shams in short-term neurobehavioral tests including Garcia test (p < 0.01), corner turn test (p < 0.01) and forelimb placement test (p < 0.01) (Figure 3(a) to (c)). Likewise, ICH animals had higher brain water content in the ipsilateral basal ganglia and ipsilateral cortex compared to sham (p < 0.01, Figure 3(d)). Three doses of ELP (0.04 μg/g, 0.12 μg/g, 0.36 μg/g) were administered by i.n. route at 1 hour after ICH. Both middle and high dosage of ELP improved performance in Garcia test (p < 0.05) and corner turn test (p < 0.05) and reduced brain water content in ipsilateral basal ganglia compared to vehicle group (p < 0.05) (Figure 3(a), (b) and (d)). Neither vehicle nor ELP treatment had influence on hematoma volume at 24 h after ICH (Figure 3(e)).
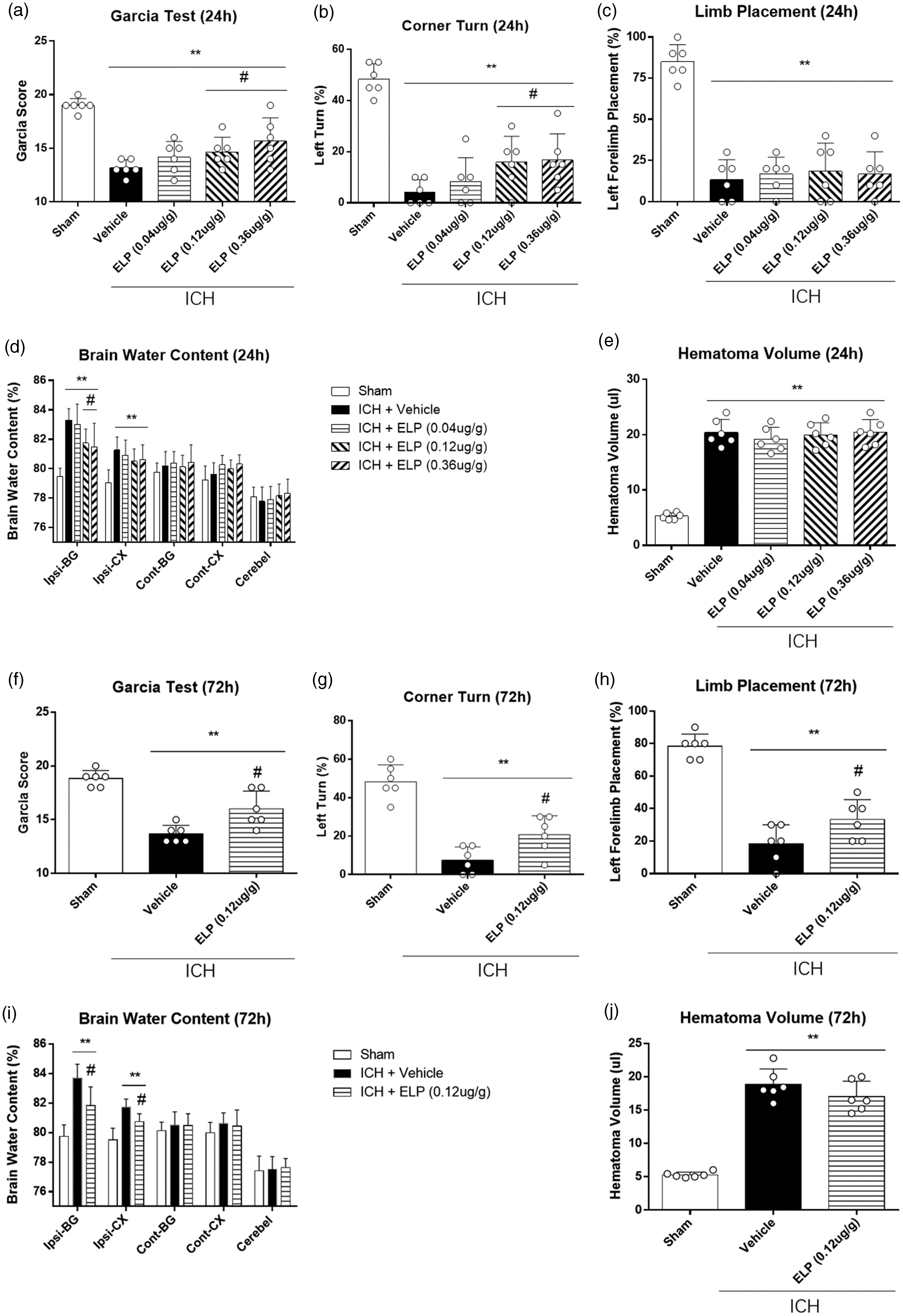
Effect of ELP on neurobehavior tests, brain water content, and hematoma volume after ICH. (a) Garcia test at 24 h, (b) corner turn test at 24 h, (c) forelimb placement test at 24 h, (d) brain water content at 24 h, (e) hematoma volume at 24 h. (f) Garcia test at 72 h, (g) corner turn test at 72 h, (h) forelimb placement test at 72 h, (i) brain water content at 72 h, and (j) hematoma volume at 72 h. **p < 0.01 vs. sham, #p < 0.05 vs. ICH + vehicle. Error bars are represented as mean ± SD, n = 6 mice per group. Ipsi-BG, ipsilateral basal ganglia; Ipsi-CX, ipsilateral cortex; Cont-BD, contralateral basal ganglia; Cont-CX, contralateral cortex; Cerebel, cerebellum.
ELP alleviated short-term neurobehavioral deficits and brain edema at 72 h after ICH
Seventy-two hours after ICH, mice had significantly worse neurobehavioral performance (p < 0.01, Figure 3(f) to (h)) and higher brain water content in ipsilateral basal ganglia and cortex than shams (p < 0.01, Figure 3(i)). Treatment with middle dosage ELP (0.12 μg/g) significantly improved performance in Garcia test, corner turn test and forelimb placement test (p < 0.05, Figure 3(f) to (h)) at 72 h as well as reduced brain water content in ipsilateral basal ganglia and cortex compared to vehicle (p < 0.05, Figure 3(i)) at 72 h after ICH. Neither vehicle nor ELP treatment had influence on hematoma volume at 72 h after ICH (Figure 3(j)).
ELP inhibited microglia activation at 24 h after ICH
The activated microglia were evaluated by immunostaining of M1 microglia marker CD86 in the perihematomal area at 24 h after ICH. The results showed that the number of CD86 positive cells were significantly increased in ICH + vehicle group compared to sham at 24 h after ICH (p < 0.01, Figure 4). Treatment with ELP (0.12 μg/g) significantly decreased the number of CD86 positive cells compared to vehicle (p < 0.05, Figure 4) at 24 h after ICH.
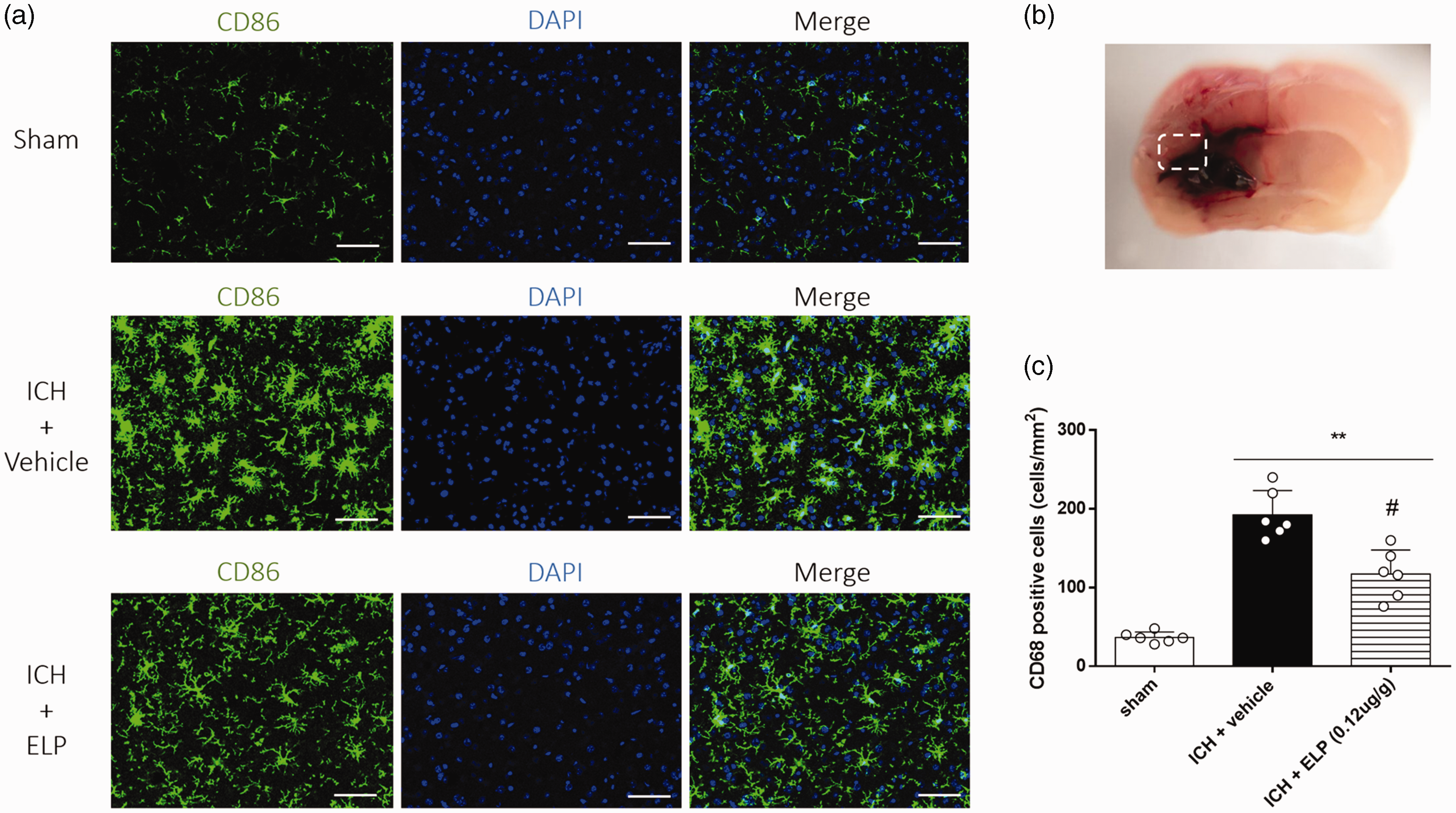
Effect of ELP on microglia activation at 24 h after ICH. (a) Representative images of immunofluorescence staining of CD86 (green) in the perihematomal area at 24 h after ICH, (b) brain sample with illustration showing the area for CD86 + cell counting in the perihematomal region, (c) quantitative analyses of CD86 + cells in the perihematomal area at 24 h after ICH. **p < 0.01 vs. sham, #p < 0.05 vs. ICH + vehicle. Error bars are represented as mean ± SD, n = 6 mice per group.
ELP improved long-term neurobehavioral outcomes after ICH
Mice in ICH + vehicle group had significantly more foot faults in the foot fault test and shorter falling latency in rotarod test than sham mice in the first, second and third weeks after ICH (p < 0.05, Figure 5(a) and (b)). However, administration of ELP but not vehicle significantly improved performance in foot fault test in the first, second and third weeks as well as in rotarod test in the second and third weeks after ICH (p < 0.05, Figure 5(a) and (b)).
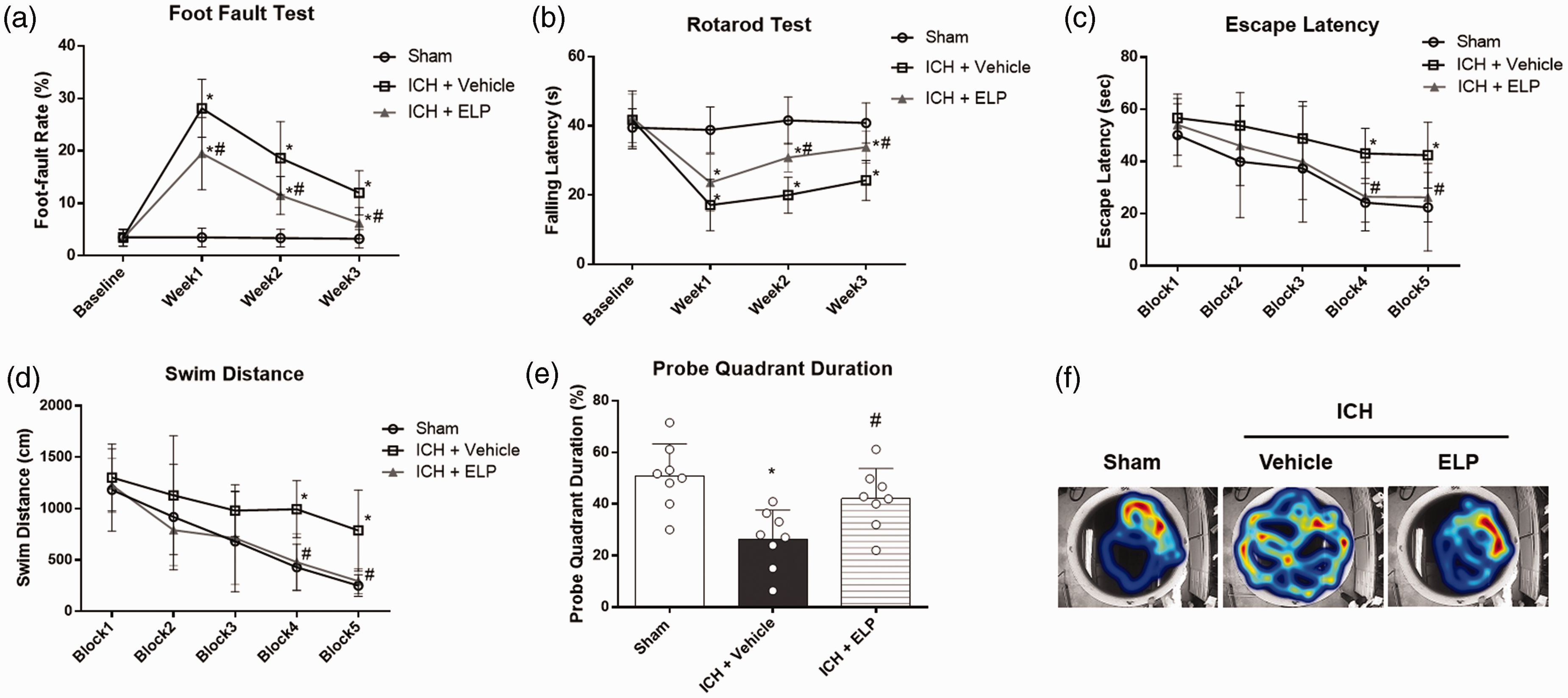
Effect of ELP on long-term neurobehavioral outcomes after ICH. (a) Foot fault test and (b) Rotarod test was conducted at the first, second and third weeks after ICH. (c) Escape latency and (d) swim distance evaluated in Morris water maze test on days 21 to 25 after ICH. (e) Probe quadrant duration of Morris water maze on day 25 after ICH. (f) Typical traces of Morris water maze testing on day 25 after ICH. *p < 0.05 vs. sham, #p < 0.05 vs. ICH + vehicle. Error bars are represented as mean ± SD, n = 8 mice per group.
In Morris water maze test, the escape latency and swim distance for mice to find the platform were significantly increased in the ICH + vehicle and ICH + ELP groups compared with the sham group on blocks 4 to 5 (p < 0.05, Figure 5(c) and (d)). ICH mice that received ELP (0.12 μg/g) treatment had decreased escape latency and shorter swim distance on blocks 4 to 5 (p < 0.05, Figure 5(c) and (d)). In the probe quadrant duration trial, mice in ICH + vehicle group spent less time in the target quadrant than sham (p < 0.05), while those in ELP treatment group showed improvement compared with ICH + vehicle group (p < 0.05, Figure 5(e) and (f)).
LAR activating CRISPR and NT157 reversed the protective effects of ELP after ICH
Administration of LAR Act-CRISPR reversed the protective effect of ELP on neurobehavioral improvement including Garcia test and corner turn test at 24 h after ICH (p < 0.05, Figure 6(a) and (b)) and the effect on brain water content (p < 0.05, Figure 6(c)). Similar results were found with the administration of NT157 (p < 0.05, Figure 6).
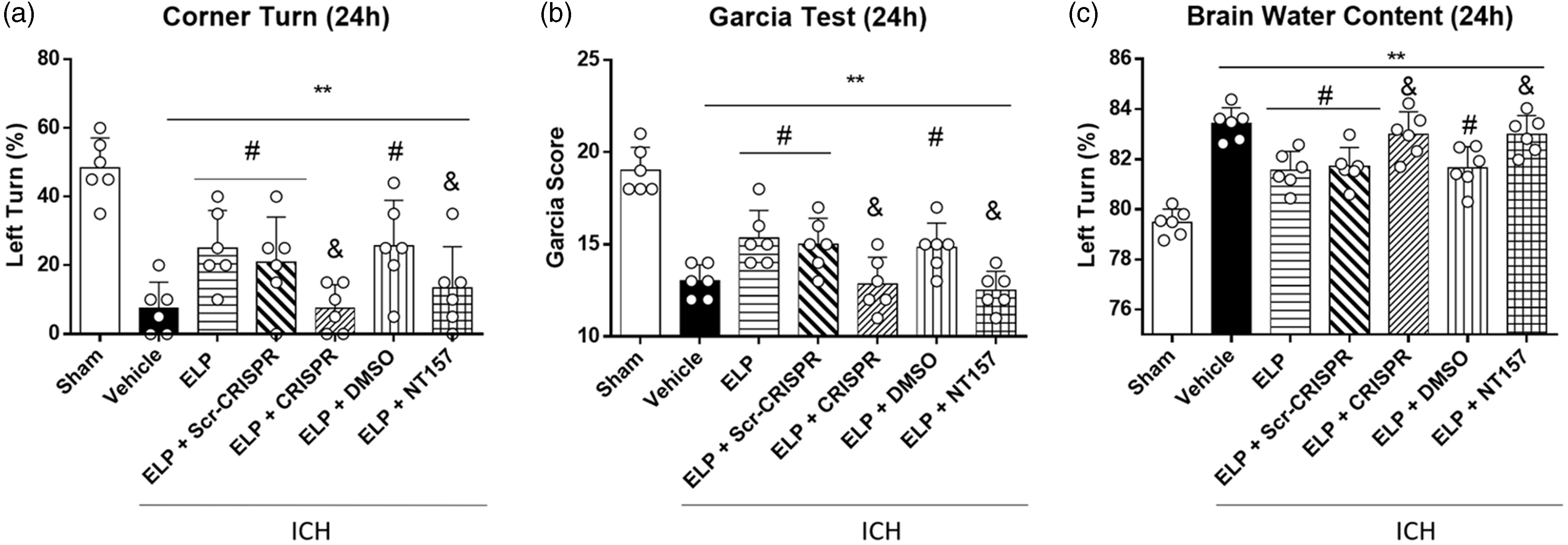
LAR Act-CRISPR and IRS-1 inhibitor NT157 reversed the protective effects of ELP on neurobehavior outcomes and brain water content after ICH. (a) Corner turn test, (b) Garcia test and (c) brain water content of ipsilateral basal ganglia evaluated at 24 h after ICH. **p < 0.01 vs. sham, #p < 0.05 vs. ICH + vehicle, &p < 0.05 vs. ICH + ELP + scramble CRISPR/DMSO. Error bars are represented as mean ± SD, n = 6 mice per group.
The expression of RhoA, serine phosphorylated IRS1 and IL-1b increased significantly at 24 h after ICH compared with sham (p < 0.05, Figure 7(a) to (b) and (f)). On the other hand, tyrosine phosphorylated IRS1, p-Akt and IL-10 were significantly downregulated at 24 h after ICH (p < 0.05, Figure 7(a), (d), (e) and (g)). Treatment with ELP (0.12 μg/g) decreased the expression of RhoA, serine phosphorylated IRS1 and IL-1b while it increased tyrosine phosphorylated IRS1, p-Akt and IL-10 compared with ICH + vehicle group (p < 0.05, Figure 7(a) to (g)), and these effects observed with ELP treatment were reversed by the administration of ELP-activating CRISPR but not the control scramble CRISPR (p < 0.05, Figure 7(a) to (g)). Additionally, the effects of ELP treatment on serine phosphorylated IRS1, tyrosine phosphorylated IRS1, p-Akt, IL-1b and IL-10 were reversed by the administration of NT157 (p < 0.05, Figure 7(h), (j) to (n)).
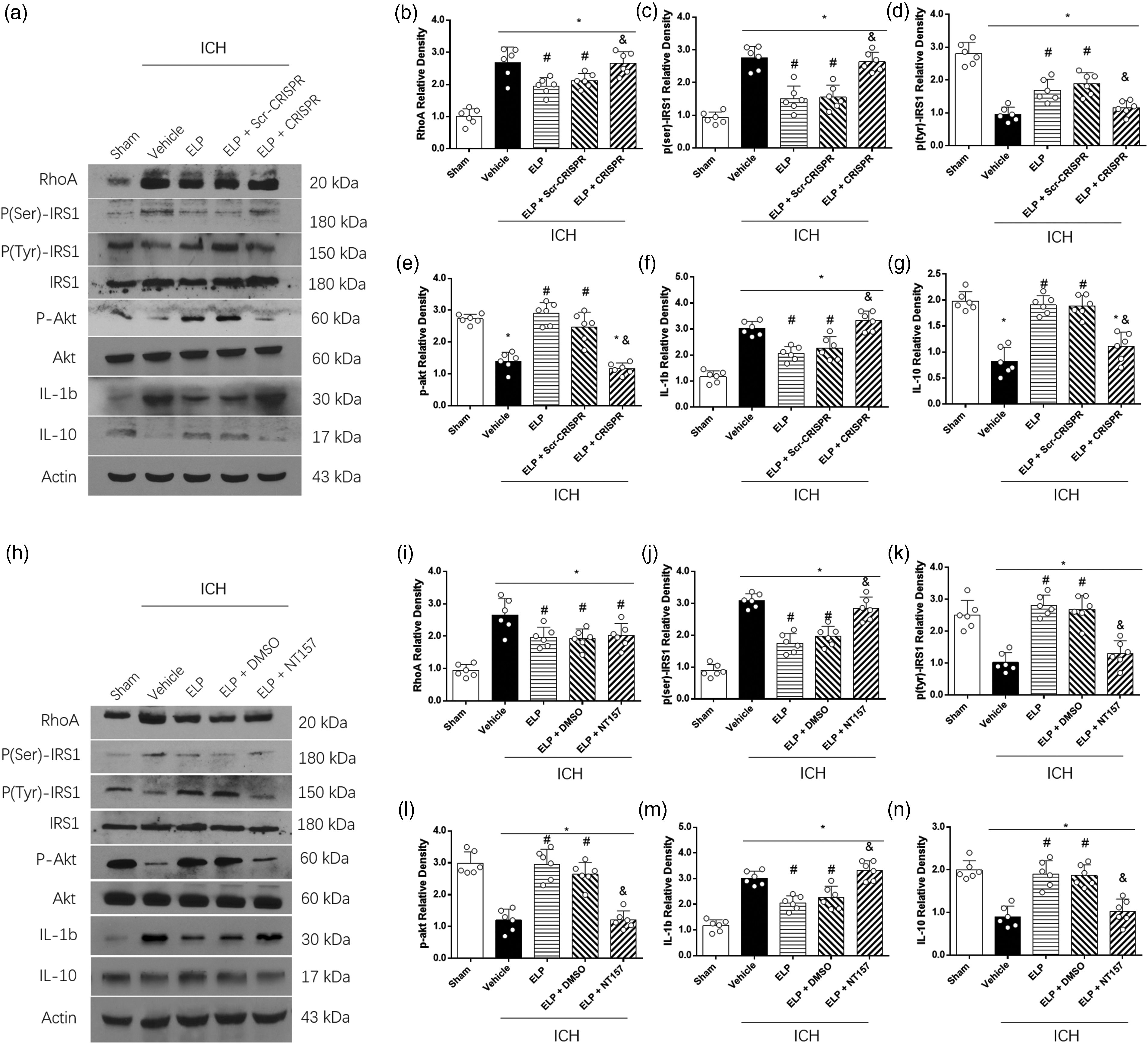
The effects of ELP, LAR Act-CRISPR and IRS1 inhibitor NT157 on the expression of the LAR downstream signaling proteins. (a, h) Representative western blot band and (b-g, i-n) quantitative analysis of RhoA, serine phosphorylated IRS1, tyrosine phosphorylated IRS1, phosphorylated Akt, IL-1b and IL-10 expressions at 24 h after ICH. *p < 0.05 vs. sham, #p < 0.05 vs. ICH + vehicle, &p < 0.05 vs. ICH + ELP + scramble CRISPR or ICH + ELP + DMSO. Error bars are represented as mean ± SD, n = 6 mice per group.
Discussion
In this study, we demonstrated that LAR played a role in the inflammatory process after ICH, and we determined the protective effects and underlying anti-inflammatory mechanisms of inhibiting LAR in ICH mice. We found that the expressions of LAR and endogenous CSPGs increased in the brain after ICH. Our results showed that inhibition of LAR with ELP alleviated brain edema and improved neurobehavioral outcomes both in the short-term and long-term neurological tests. In addition, treatment with ELP downregulated the expression of RhoA, serine phosphorylated IRS1 and IL-1b while upregulated tyrosine phosphorylated IRS1, phosphorylated Akt and IL-10. Furthermore, overactivation of LAR with Act-CRISPR and the IRS1 inhibitor NT157 reversed the neurobehavioral protective function and anti-inflammatory effects of ELP. Our findings suggested that LAR contributed to inflammation after ICH, and inhibition of LAR with ELP improved neurobehavioral outcomes and attenuated neuroinflammation at least partly via RhoA/IRS1/Akt signaling pathway.
LAR plays a critical role in cell growth, differentiation, mitotic cycle, and oncogenic transformation. Previous studies found that chondroitin sulfate proteoglycans (CSPGs), the endogenous agonist of LAR, as well as LAR are upregulated after neural injury in spinal cord injury and neuron culture,18,19 which is consistent with our findings. Studies have shown that treatment with Chondroitinase ABC, an inhibitor of CSPGs, improved outcomes in rodent models of ischemic stroke20,21 and spinal cord injury. 22 Furthermore, CSPGs were found to induce the activation of tumor-associated microglia. 5 Inhibiting LAR with ILP showed anti-neuroinflammatory effect via regulation of microglia. 6 However, to our knowledge there are no previous studies about the role of LAR in ICH. Therefore, we evaluated the potential anti-inflammatory effects of LAR inhibition by administering ELP after ICH in a rodent model.
We used a LAR inhibitor, extracellular LAR peptide (ELP), to determine the role of LAR in ICH. Systemic treatments with ELP/ILP were found to improve locomotor recovery in spinal cord injury (SCI) mice. 4 Previous studies have shown that macromolecules can be delivered intranasally to bypass the blood brain barrier. 23 Intranasal administration of peptides such as insulin with surfactants has been reported to be a sufficient and safe method for delivery to the brain. 24 Additionally, several studies in stroke models have utilized the intranasal method for delivery of neuroprotective agents to the brain.25,26 In our study, ELP was dissolved in DMSO and delivered intranasally to mice following the procedure for intranasal administration as previously published methods in stroke models.13,14 Brain edema is an important factor in the progression of secondary injury after ICH 27 and is related to hemolysis, which is also important in ICH. Hemolysis in the hematoma occurs early in 24 hours after ICH and contributes to the development of perihematomal edema.28,29 In this study, ICH mice received ELP as treatment at 1 h after ICH, and we evaluated brain edema by measuring brain water content and neurological function at 24 h and 72 h after ICH to determine the treatment effects of ELP. We found that ELP administration to ICH mice, reduced brain edema and improved neurological function. In addition, we measured hematoma volume in different groups at 24 h and 72 h after ICH. The results showed that treatment with ELP did not affect the hematoma volume after ICH, which indicated that the brain edema alleviation and neurobehavioral protective effects of ELP were not mediated by the resolution of hematoma, at least in the acute phase. We also conducted studies to determine whether ELP treatment had protective effects on long-term neurobehavioral outcomes after ICH, and we performed foot fault test and Rotarod test to evaluate sensorimotor function and Morris water maze test for cognitive function. The results showed that treatment with ELP improved long-term neurological function after ICH as shown by improved performance in movement coordination, spatial learning, and memory abilities in the neurological tests.
Microglia activation starts early after ICH 30 and promotes neuroinflammation, contributing to secondary injury after ICH.31,32 CD86 is known as a classical M1 microglia marker, which overexpresses after ICH. CSPGs were found to induce the activation of tumor-associated microglia. 5 Inhibition of LAR with ILP was found to have anti-neuroinflammatory effect via regulation of microglia in spinal cord injury. 6 To determine the role of activated microglia, we evaluated the levels of CD86 M1 microglia in the perihematomal area at 24 h after ICH. Our data showed that inhibition of LAR reduced M1 microglia activation after ICH and downregulated pro-inflammatory factor IL-1b and upregulated anti-inflammatory factor IL-10, which was consistent with previous studies.
We investigated the underlying mechanism through which LAR potentially mediated neuroinflammation. Emerging studies have shown anti-inflammatory effects of Akt. For instance, phosphorylation of Akt increased the phosphorylation of GSK-3β, which displayed anti-inflammatory effect.33,34 Activation of Akt also alleviated inflammation through Akt/CREB signaling axis in vitro. 35 Deletion of LAR eliminated CSPG-induced Akt inactivation and RhoA activation. Furthermore, previous study showed that LAR signaling regulated microglia activation via RhoA/ROCK pathway and the inhibition of LAR decreased IL-1β while increasing IL-10 in spinal cord injury. 6 The activation of RhoA/ROCK pathway was found to increase serine phosphorylation of IRS-1/2, followed by decreases in tyrosine phosphorylation of IRS-1/2. 10 Additionally, LAR reduced tyrosine phosphorylation of IRS-1 and its downstream molecule p-Akt. 11 We therefore hypothesized that RhoA and IRS-1 serine phosphorylation were involved in the inflammatory effects LAR. To elucidate the potential anti-inflammatory effects of ELP, we administered either LAR activating CRISPR or IRS inhibitor NT-157 prior to treatment with ELP. CRISPR was administered by intracerebroventricular injection, which was supported by previous studies that have reported the efficiency of intraparenchymal and intraventricular CRISPR injection.36,37 Intracerebroventricular injection of CRISPR provides a brain-wide genome editing and is now widely used in central nervous system studies.38,39 Likewise, the IRS inhibitor NT-157 was injected intraperitoneally in accordance to previous in vivo studies.40,41 Our results showed that ELP treatment downregulated RhoA and serine phosphorylation of IRS-1 while upregulating p-Akt following ICH. This was associated with the downregulation of pro-inflammatory IL-1β and upregulation of anti-inflammatory IL-10. Furthermore, administration of LAR activating CRISPR or IRS inhibitor NT-157 reversed these effects as well as abolished the protective effects of ELP on neurological outcomes after ICH. These results indicated that the neurobehavioral improvement after ICH and anti-inflammatory effects of ELP were mediated through LAR/RhoA/IRS-1/Akt signaling pathway. Interestingly, we found that the administration of LAR activating CRISPR or IRS inhibitor NT-157 also reversed the effect of ELP on brain water content after ICH. It was reported that LAR is expressed on vascular endothelium, 42 which potentially contributes to the edema reducing effects of ELP. In this study, we did not focus on the mechanisms of brain edema and the potential mechanisms of ELP in reducing brain edema will be explored in our future studies.
There are some limitations in our study. CSPGs include neurocan, brevian, versican, aggrecan and phosphacan. 43 This research was focused on the receptor LAR, hence only two kinds of CSPGs were measured in our study. Although, we found that ELP treatment reduced the expressions of RhoA and serine phosphorylated IRS1, we did not specifically utilize pharmacological intervention to study the role of RhoA/ROCK in our study. Previous studies have revealed the LAR/RhoA/ROCK pathway and RhoA/ROCK/IRS-1 pathway using Y27632, an inhibitor of ROCK.6,10 Additionally, NT-157 is an antagonist for both IRS-1 and IRS-2. 44 Further studies are needed to examine the LAR/RhoA/IRS-1 pathway. IRS-1/2 are usually combined in expression and phosphorylation,10,45 and IRS-2 may also be involved in the anti-inflammatory effects of ELP. In this study, only IRS-1 was measured. We evaluated p-Akt Ser473, this phosphorylation type of Akt was chosen since it is the downstream molecule according to previous study. 46 Whether IRS-2/Akt Thr308 may have contributed to the anti-inflammatory effect of ELP was not evaluated and therefore cannot be ruled out. Further studies are needed to find out whether IRS-2 plays a role in this pathway. In this study, we examined the anti-inflammatory effects of LAR inhibition, but the potential anti-apoptotic and blood-brain barrier preservation effects of ELP could not be excluded. Therefore, further research is needed to study the potential effects and the underlying mechanisms of LAR that may contribute to ICH pathology.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH grant R01NS091042 to JHZ.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
CSL and LST conceived and designed the study. PS was aware of the group allocation. CSL, XH, JYZ and JY acquired the data. CSL, XH and XHY analyzed and interpreted the data. CSL and LST wrote and prepared the manuscript. PS, JZ, Feng Gao and JPT reviewed the manuscript. JPT coordinated the study. All authors have read and approved the final manuscript.
