Abstract
Chronic cerebral hypoperfusion (CCH) is postulated to underlie multiple pathophysiological processes in vascular dementia (VaD), including extracellular matrix dysfunction. While several extracellular matrix proteins, namely cyclophilin A (CypA), extracellular matrix metalloproteinase inducer (EMMPRIN) and gelatinases (matrix metalloproteinases, MMP-2 and -9) have been investigated in acute stroke, their involvement in CCH and VaD remains unclear. In this study, CypA-EMMPRIN-gelatinase proteins were analysed in a clinical cohort of 36 aged, cognitively unimpaired subjects and 48 VaD patients, as well as in a bilateral carotid artery stenosis mouse model of CCH. Lower CypA and higher EMMPRIN levels were found in both VaD serum and CCH mouse brain. Furthermore, gelatinases were differentially altered in CCH mice and VaD patients, with significant MMP-2 increase in CCH brain and serum, whilst serum MMP-9 was elevated in VaD but reduced in CCH, suggesting complex CypA-EMMPRIN-gelatinase regulatory mechanisms. Interestingly, subjects with cortical infarcts had higher serum MMP-2, while white matter hyperintensities, cortical infarcts and lacunes were associated with higher serum MMP-9. Taken together, our data indicate that perturbations of CypA-EMMPRIN signalling may be associated with gelatinase-mediated vascular sequelae, highlighting the potential utility of the CypA-EMMPRIN-gelatinase pathway as clinical biomarkers and therapeutic targets in VaD.
Keywords
Introduction
Vascular dementia (VaD) is the second most common form of dementia after Alzheimer’s disease, accounting for 20% to 30% of all dementia cases worldwide (World Alzheimer Report 2014). VaD has a predominantly vascular aetiology arising from multiple causes and risk factors. 1 However, one of the major pathophysiological features of VaD is a state of chronic cerebral hypoperfusion (CCH) throughout the different regions of the brain including the white matter regions.2,3 Recent evidence shows that CCH is associated with several other VaD-related, interlinked pathophysiological mechanisms, including inflammation, 4 oxidative stresss, 5 blood-brain barrier (BBB) dysfunction 6 and cerebrovascular diseases such as white matter lesions 7 in animal models.
In ongoing research efforts to investigate molecular mechanisms of VaD, animal models of CCH using a bilateral carotid artery stenosis (BCAS) procedure have shown BBB damage and white matter lesions.8,9 While the exact mechanisms are not fully understood, CCH has been postulated to trigger inflammation which in turn plays an important role in remodelling of the vascular extracellular matrix (ECM), a process also known to underlie BBB breakdown and demyelination. 10 These findings underscore the potential involvement of CCH in key pathophysiological processes of VaD.11,12 However, there is a dearth of research into the molecular mechanisms underlying CCH-associated signalling upstream of ECM modifications.
The cerebral ECM is a delicate environment that fills the parenchymal space, and a well-regulated ECM is critical in maintaining the neurovascular network. Recent studies have proposed the role of cyclophilin A (CypA) signalling to matrix metalloproteinases (MMPs) in cerebrovascular integrity, with subarachnoid haemorrhage associated with CypA signalling perturbations.13–15 CypA acts as the intracellular target for the immunosuppressant drug cyclosporine A, 16 but could also be secreted into the extracellular space, whereupon it binds to its receptor, the extracellular matrix metalloproteinase inducer (EMMPRIN, also termed CD147). 17 EMMPRIN is a highly glycosylated transmembrane receptor which has MMP-inducing activity. 18 Activated MMPs, including gelatinases which comprise MMP-2 and MMP-9, are known to trigger vascular remodelling by cleaving tight junctions and basement membrane of the ECM. 19 Gelatinase induction can also lead to demyelination and subsequent white matter disease.20–22 Although the physiological and pathophysiological aspects of gelatinase-mediated vascular remodelling, as well as its upstream signalling and regulation, remain unclear,23,24 the CypA-EMMRPIN-gelatinase pathway may be one potential candidate for this regulatory role.
While gelatinases have been consistently found to be dysregulated in post-mortem brains25–27 and blood of stroke patients,28–30 their involvement in VaD is unclear. Similarly, whilst several animal studies demonstrated CypA and EMMPRIN upregulation in post-stroke brains,31–34 these models focused on acute stroke conditions which may not represent the CCH process in VaD. The present study aimed to investigate the associations between CypA and key ECM proteins as well as their potential clinical utility by measuring serum levels of CypA, EMMPRIN, MMP-2 and MMP-9 in in a cohort of cognitively normal subjects and VaD patients. Putative associations between the serum biomarkers with neuroimaging markers of cerebrovascular disease, namely white matter hyperintensities (WMH, an established marker of white matter lesions), lacunes and cortical infarcts are also examined. Additionally, the effects of CCH on the CypA-EMMPRIN-gelatinase pathway were studied in BCAS-treated mice. This novel multi-modal approach in turn allows further evaluation of the overarching hypothesis that CCH-associated dysregulation of the CypA-EMMPRIN-gelatinase pathway is linked to cerebrovascular diseases in VaD, with concomitant insights into neurochemical mechanisms, identification of potential therapeutic targets as well as biomarkers.
Materials and methods
Clinical study cohort: demographic and disease factors
A case-control study design was adopted, with cross-sectional analyses performed on the data collected at baseline. All subjects were recruited from two study sites in Singapore (the memory clinics from National University Hospital and Saint Luke’s Hospital), while all standard physical, clinical, blood tests and neuropsychological assessments, as well as neuroimaging scans were performed at the National University of Singapore as previously described. 35 The study was conducted in accordance with the Declaration of Helsinki. Ethics approval was obtained from the National Healthcare Group Domain Specific Review Board (NHG-DSRB references: 2010/00017 for study protocol DEM4233 and 2018/01098 for study protocol DEM4333). Written informed consent was obtained from all participants in their preferred language prior to study recruitment.
Participants with no cognitive impairment (NCI, defined as cognitively normal on objective neuropsychological assessments) were recruited from both memory clinics and from the community, while VaD subjects were diagnosed based on the Diagnostic and Statistical Manual of Mental Disorders, Fourth edition (DSM-IV) and the National Institute of Neurological Disorders and Stroke-Association Internationale pour la Recherché et l' Enseignement en Neuroscience (NINDS-AIREN) criteria. 36 Subjects who did not fulfil diagnostic criteria for dementia and dementia subjects with non-vascular etiologies (e.g., Alzheimer’s disease) were excluded from this study. For all participants, clinical interviews and examination of medical records were performed to ascertain demographic and vascular risk factors, including education level, hypertension, diabetes mellitus, hyperlipidemia, and cardiovascular diseases (see Table 1).
Baseline characteristics of the clinical study cohort.
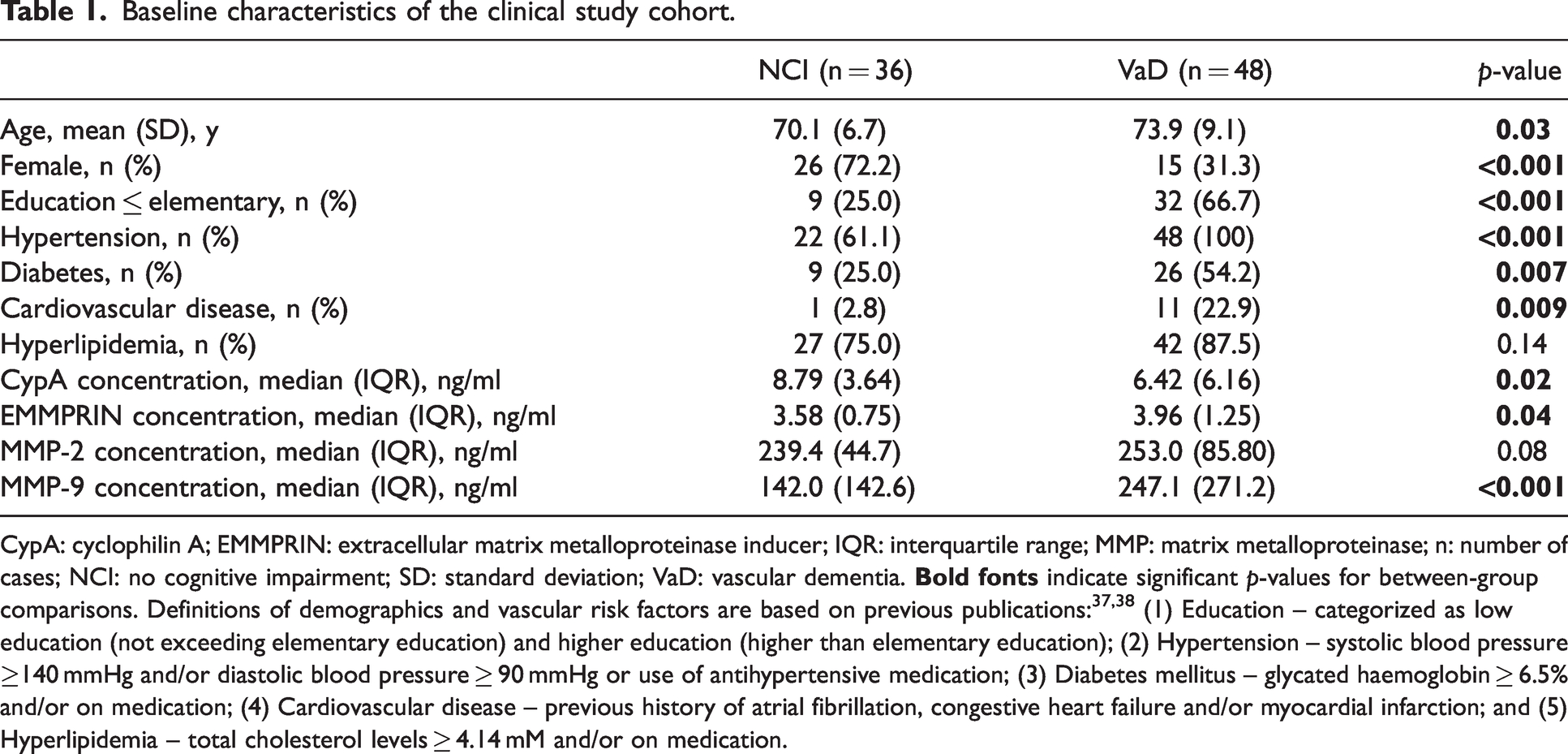
CypA: cyclophilin A; EMMPRIN: extracellular matrix metalloproteinase inducer; IQR: interquartile range; MMP: matrix metalloproteinase; n: number of cases; NCI: no cognitive impairment; SD: standard deviation; VaD: vascular dementia.
Clinical study cohort: Neuroimaging assessments
Magnetic resonance imaging (MRI) scans were performed at the Clinical Imaging Research Centre of the National University of Singapore, using a 3-Tesla (3T) Siemens Magnetom Trio Tim scanner with a 32-channel head coil. The sequences included T1-weighted, T2-weighted and fluid-attenuated inversion recovery (FLAIR), and were graded by two clinicians blinded to clinical data. Presence of lacunes, cortical infarcts and WMH were defined using STRIVE criteria, 39 while severity of WMH were graded using the modified Fazekas scale, 40 where scores ≥2 denote confluent lesions. 41
Clinical study cohort: Blood biomarker measurements
Non-fasting bloods were drawn from study participants into serum separating tubes and processed by centrifugation at 2000 g for 10 min at 4°C, followed by extraction of the upper serum layer and storage at −80°C until use. Serum CypA (Catalogue number RD191329200R, BioVendor-Laboratorni medicina a.s., Brno, Czech Republic), EMMPRIN, MMP-2 and MMP-9 (Quantikine®, Catalogue numbers DEMP00, MMP200 and DMP900, R&D Systems, Inc. Minneapolis, MN, USA) concentrations were measured using quantitative sandwich enzyme-linked immunosorbent assays (ELISAs) in accordance with manufacturer’s instructions. Briefly, serum samples were diluted 3-, 10-, 20- and 100-folds respectively for CypA, EMMPRIN, MMP-2 and MMP-9 in calibrator diluent buffer before adding to the antibody-coated plate. Detection was achieved by the addition of the stabilized chromogen (tetramethylbenzidine) and colour development was stopped after 10 to 30 minutes by the addition of an acidic stop solution. Absorbance was measured at 450 nm on a microplate reader (BioTek, Winooski, VT, USA) with background subtraction at 630 nm (for CypA) or 570 nm (for EMMPRIN, MMP-2 and MMP-9). A standard curve ranging was generated for each assay (CypA assay ranged from 0 to 20 ng/ml, EMMPRIN assay ranged from 0 to 4000 pg/ml, MMP-2 assay ranged from 0 to 32 ng/ml while MMP-9 assay ranged from 0 to 20 ng/ml) and fitted to a 4-parameter logistic model with weighted R-squared correlation coefficients consistently >0.99 in our hands. To obtain the actual biomarker concentrations in serum, sample concentrations read from the corresponding standard curves were multiplied by the respective dilution factors. All the blood samples were measured in duplicates and analysed blinded to subject characteristics and clinical status.
Mice for experimental study
All experimental procedures were approved by the National University of Singapore and the Institutional Animal Care and Use Committee (IACUC, protocol number: R17-1240). Animal care, handling and housing were carried out according to the National Advisory Committee for laboratory Animal Research (NACLAR) Guidelines on the Care and Use of Animals for Scientific Purposes (Singapore Animals and Birds Act, Chapter 7, Section 80). 42 All efforts to minimize suffering and animal count were made in accordance with the Responsible Care and Use of Laboratory Animals (RCULA) guidelines for the use of common laboratory mammals. Fourteen to sixteen weeks old C57BL/6J male mice were obtained from InVivos Pte. Ltd., Singapore and maintained in a murine pathogen-free animal facility at the National University of Singapore. All mice were given access to food and water ad libitum. 8-10 animals were used in total for each experimental group. All experiments in the manuscript were performed and reported according to ARRIVE (Animal Research: Reporting In Vivo Experiments) guidelines.
Bilateral common carotid artery stenosis (BCAS) surgery in mice
Mice were subjected to either sham or BCAS procedure using microcoils to induce CCH as previously described. 43 Briefly, animals were first anaesthetized with 1.5–2% isoflurane at a flow rate of 0.4–0.8 liter/minute. Through a midline cervical incision, both the left and right common carotid arteries (CCA) were exposed and freed from their sheaths. Microcoils specifically designed for mice (piano wire diameter 0.08 mm, internal microcoil diameter 0.18 mm, coiling pitch 0.5 mm, and total length 2.5 mm, from Sawane Spring Co Ltd, Japan) were twined on each artery via rotation around the artery. The site of incision was then closed, and the mice were removed from the isoflurane vaporiser system for observation until they regained consciousness and recovered completely. For the sham groups, the CCA were exposed after cervical incision, then closed prior to animal recovery from anaesthesia. Some of the animals from each experimental group were also subject to cerebral blood flow (CBF) measurements pre- and post-surgery (see below) before being euthanised 30 days after surgery for removal of brain tissues and subsequent studies.
CBF measurements in mice
Measurements of CBF (as visual validation for cerebral perfusion changes) before any surgery (baseline) and immediately after surgery were performed with a High-Resolution Laser Speckle Contrast Imaging (PSI system, Perimed AB, Jakobsberg, Sweden). Regions of Interest (ROIs) between bregma and lambda across the area of both hemispheres of the brain were recorded (see Figure 1(a) and (b)). Under isoflurane anaesthesia, an incision was made at the skull after the hair was removed. The exposed skull was cleaned gently with sterile phosphate-buffered saline (PBS) using a cotton applicator and kept moist using a non-toxic silicone oil. Perfusion images were acquired using the PeriCAM PSI system (Perimed AB, Jakobsberg, Sweden) with a 70-mW built-in laser diode for illumination and a 1388 × 1038 pixels charge-coupled device (CCD) camera installed 10 cm above the skull (speed 19 Hz, and exposure time 6 ms). Acquired images were analysed and the rate of CBF was calculated as an absolute value of cerebral perfusion in perfusion units using dedicated software (PIMSoft version 1.4, Perimed AB, Jakobsberg, Sweden). Mice were placed on a heat pad which was maintained at 37 ± 0.5°C throughout the experimental procedure.
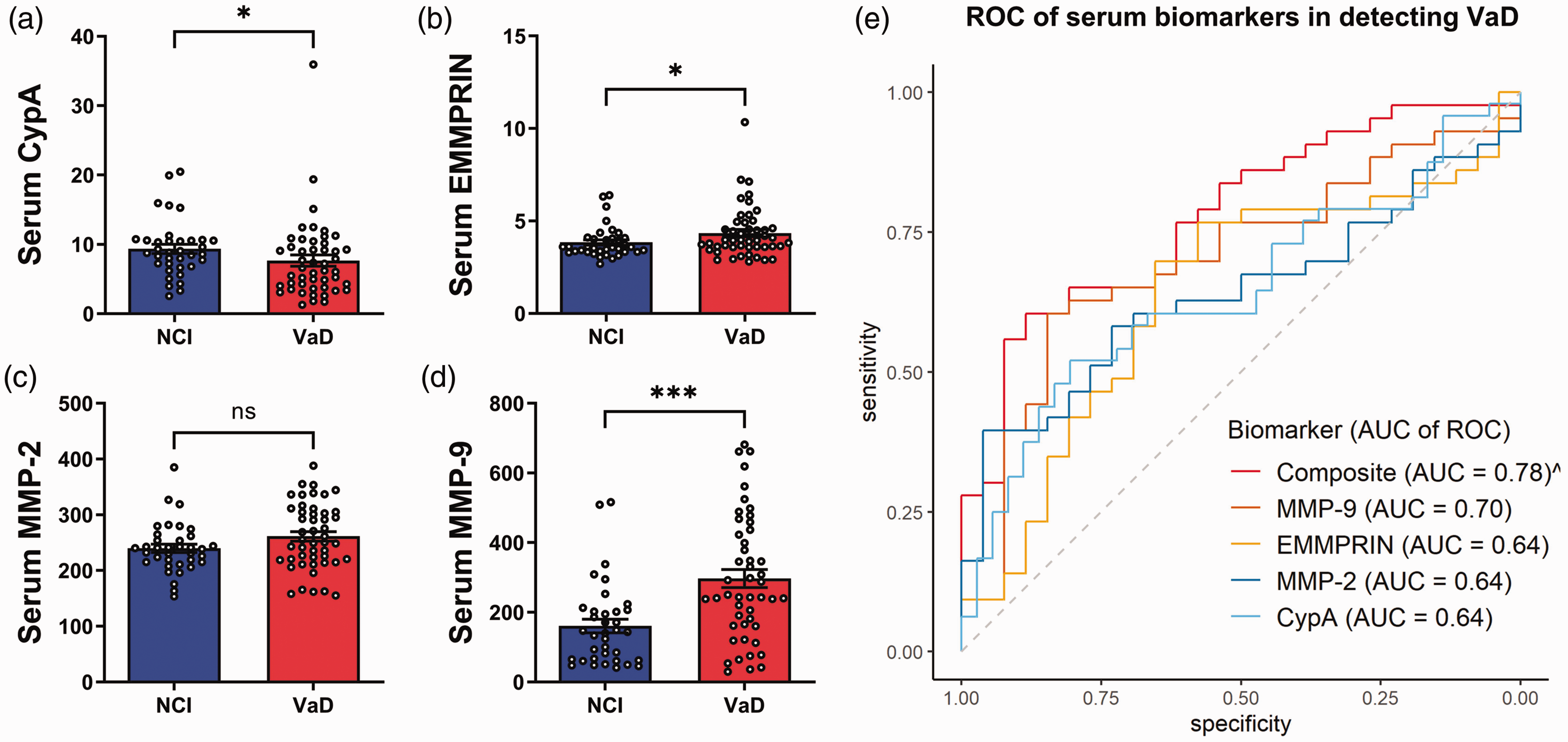
Serum concentrations of CypA and ECM proteins, and their respective ROC curves in detecting VaD in a clinical study cohort. Bar graphs of (a) CypA, (b) EMMPRIN, (c) MMP-2, (d) MMP-9 serum concentrations (mean ± SEM in ng/mL, with white dots indicating individual measurements) in a cohort of NCI (n = 36) and VaD (n = 48) subjects and (e) ROC analyses indicating the diagnostic performance of each serum biomarker as well as that of a composite panel of all four serum biomarkers in detecting VaD. AUC: area under curve; NCI: no cognitive impairment; ROC: receiver operating characteristic; VaD: vascular dementia.
Brain CypA, EMMPRIN and MMPs measurements in mice
Mouse brains were harvested after euthanasia by carbon dioxide. The cortical and hippocampal regions were immediately dissected on ice and frozen in dry ice for immunoblotting. Immunoblots were performed as previously described. 44 Briefly, brain tissues were homogenised in a lysis buffer and combined with 2× Laemmli Sample Buffer (Bio-Rad Laboratories, Inc., Hercules, CA, USA), followed by heating at 95°C for 10 minutes. Protein samples were then separated on 7.5–12.5% v/v sodium dodecyl sulfate polyacrylamide gel electrophoresis before being transferred onto nitrocellulose membranes. The membranes were then blocked with 1× tris-buffered saline containing 0.1% Tween 20 (TBST) and 1% fish skin gelatin (FSG) solution at room temperature for 1 hour before incubation with the following primary antibodies: CypA (Cell Signaling Technology, #2175S), EMMPRIN (Santa-Cruz, sc-9756), MMP-2 (Cell Signaling Technology, #87809S), MMP-9 (Millipore, #AB19016) and β-actin (Sigma-Aldrich, A5441), overnight at 4°C with shaking. Following primary antibody incubation, membranes were washed three times with TBST before incubating with horseradish peroxidase (HRP)-conjugated secondary antibodies (Goat Anti-Rabbit—Cell Signaling Technology, Danvers, MA, USA; Goat Anti-Mouse—Sigma-Aldrich, St. Louis, MO, USA) for 1 h at 24°C with shaking. All the above-mentioned primary and secondary antibodies were diluted at 1:1000 in TBST containing 1% FSG solution before incubation. Following secondary antibody incubation, membranes were washed three times with TBST. Enhanced chemiluminescence HRP substrate (Bio-Rad Laboratories, Inc., Hercules, CA, USA) was applied and the membranes were imaged using ChemiDocXRS + imaging system (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Immunoreactive bands were quantified using Image J software (Version 1.46; National Institute of Health, Bethesda, MD, USA), where protein densitometry of each brain biomarker was expressed relative to the densitometry of the corresponding β-actin.
Blood gelatinase concentrations in mice
Mouse blood were collected after euthanasia by carbon dioxide. Blood sample is withdrawn from the caudal vena cava and gentle aspiration of blood into a syringe. A large volume of blood (0.6-0.8 ml) free from haemolysis and contamination is collected from each mouse (n = 7 in each experimental group). Blood was left over two nights at 4°C without any agitation to allow for coagulation. The serum was collected after centrifugation of the blood sample and frozen in −80°C. Serum gelatinases (MMP-2 and MMP-9) were measured using followed the same protocol described previously under “Clinical study cohort: Blood biomarkers measurements”, with 40 times sample dilution (Quantikine®, Catalogue numbers MMP200 and MMPT90, R&D Systems, Inc. Minneapolis, MN, USA).
Luxol fast blue (LFB) myelin staining in mice
Mouse brains were harvested and placed in vials containing 4% paraformaldehyde solution for immersion-fixation overnight at 4°C. Thereafter, brain tissues were fixed in 10% neutral buffered formalin in a tissue processing protocol totalling 25 hours and embedded in paraffin wax blocks. Coronal sections of 4-µm thickness were obtained via microtome sectioning. White matter fibre density and presence of any white matter lesions were observed via microscope after LFB staining as previously described. 45 Briefly, the coronal sections were de-waxed and rehydrated before immersion into LFB solution (Abcam PLC, Cambridge, UK) at 37°C overnight. Excess staining was removed by rinsing with 95% ethanol followed by water. To differentiate grey and white matter, sections were treated with 0.05% lithium carbonate solution (Abcam PLC, Cambridge, UK) for 20 s. The sections were then passed through 70% ethanol twice briefly until nuclei were discoloured, before immersion in cresyl violet solution (Abcam PLC, Cambridge, UK) for 5 minutes with gentle shaking and rinsed in water to allow for standard histological staining for neurons. The sections were then dehydrated at an increasing ethanol gradient (70%–100%) and mounted on microscope slide using a mounting agent. Brightfield images were taken under 4× and 60× magnifications using an upright fluorescent microscope (model BX53, Olympus Corp, Tokyo, Japan). White matter density was evaluated at five brain regions: corpus callosum (paramedian), corpus callosum (medial), caudoputamen, internal capsule and the optic tract based on a previously described grading method. The severity of the WMLs were observed to be either normal (Grade 0) or abnormal disarrangement of nerve fibres (Grade 1), formation of marked vacuoles (Grade 2) and disappearance of myelinated fibers (Grade 3). 46
Statistical analyses
Statistical analyses were performed using SPSS Statistics (version 27, IBM Corp., Armork, NY, USA) and Prism (version 9, GraphPad Software, San Diego, CA, USA). Tests of normality were first carried out for all variables (Shapiro-Wilk test for animal data and Kolmogorov-Smirnov test for clinical data) for selection of subsequent parametric or non-parametric tests. For inter-group comparisons (Sham-operated vs. BCAS mice; or NCI vs. VaD subjects), Student’s T tests were used for the animal measurements and clinical study subjects’ age, Mann-Whitney U tests were performed for human serum markers, and Chi-square tests were used for categorical data like gender, education status and presence of vascular risk factors. Levels of each serum marker from the clinical cohort were also compared between absence or presence of confluent WMH (Fazekas scores ≥2), lacunes (≥1) or cortical infarcts (≥1) using Mann-Whitney U tests. Spearman’s correlation between each serum marker and MRI markers of cerebrovascular disease in continuous scales, namely Fazekas scores, lacune count and cortical infarct count, were also assessed. Binary logistic regression analyses were performed for the clinical cohort, with each blood biomarker (in tertiles) included as a determinant variable while VaD was included as an outcome variable. The regression analyses were repeated using Firth’s method, with each model adjusted for education, hypertension, diabetes and cardiovascular disease as covariates, as these variables were not matched between NCI and VaD (see Table 1). Associations between each blood biomarker and VaD were expressed in odds ratio (OR) and 95% confidence interval (CI). Diagnostic accuracies were further assessed by receiver operating characteristic (ROC) curve analyses, using R (version 4.0.2, R Foundation for Statistical Computing, Vienna, Austria) and RStudio (version 1.2, RStudio Inc., Boston, MA, USA) with package pROC. 47 Predicted probabilities of each continuous blood biomarker value, as well as a composite panel consisting of all four biomarkers in discriminating VaD from NCI were obtained from unadjusted binary logistic regression models built using the same approach described above. Predicted probabilities were then set as predictor, while VaD was set as the outcome in the ROC analyses. Areas under curves (AUC) and 95% CIs of each ROC analysis were computed using DeLong’s method, 48 whereas the optimal threshold cut-off, sensitivity and specificity values were calculated at Youden index thresholds using bootstrap procedures with 2,000 iterations. 49 The performance ability of the composite biomarker panel was compared with the individual blood biomarker by assessing the goodness of fit of the models using likelihood ratio tests. P-values less than 0.05 in all the above analyses were considered statistically significant.
Results
Baseline characteristics of clinical cohort
A total of 86 subjects with NCI without significant cerebrovascular disease and 59 with VaD were recruited from August 2010 until April 2020. Among these, 36 NCI and 48 VaD subjects had sufficient serum samples available for the biomarker assays. Compared to NCI, VaD subjects had lower education, higher prevalence of hypertension, diabetes and cardiovascular disease (Table 1). These variables were thus included as covariates in the subsequent regression models.
Altered serum ECM protein concentrations in VaD
With respect to serum measurements, there were significantly higher levels of CypA (p = 0.02), EMMPRIN (p = 0.04) and MMP-9 (p < 0.001) in VaD compared to NCI. MMP-2 also showed a nonsignificant trend towards increase in VaD (p = 0.08, see Figures 1(a) to (d) and Table 1). Similarly, univariate binary logistic regression models in Table 2 showed significant associations between lower CypA (OR = 4.2; 95% CI = 1.3 to 13.6), higher EMMPRIN (OR = 4.5; 95% CI = 1.5 to 13.9) and higher MMP-9 (OR = 8.3; 95% CI = 2.4 to 28.6) with VaD. After adjustments for all relevant vascular risk factors, significant associations with VaD were retained only for EMMPRIN (OR = 6.8; 95% CI = 1.1 to 40.5). The association between lower CypA and VaD remained significant after adjustment for age and gender only (OR = 3.8; 95% CI = 1.0 to 14.1) whereas the association between higher MMP-9 and VaD remained significant after adjustment for all risk factors except hypertension (OR = 7.2; 95% CI = 1.5 to 32.4).
Associations between serum biomarkers and VaD.
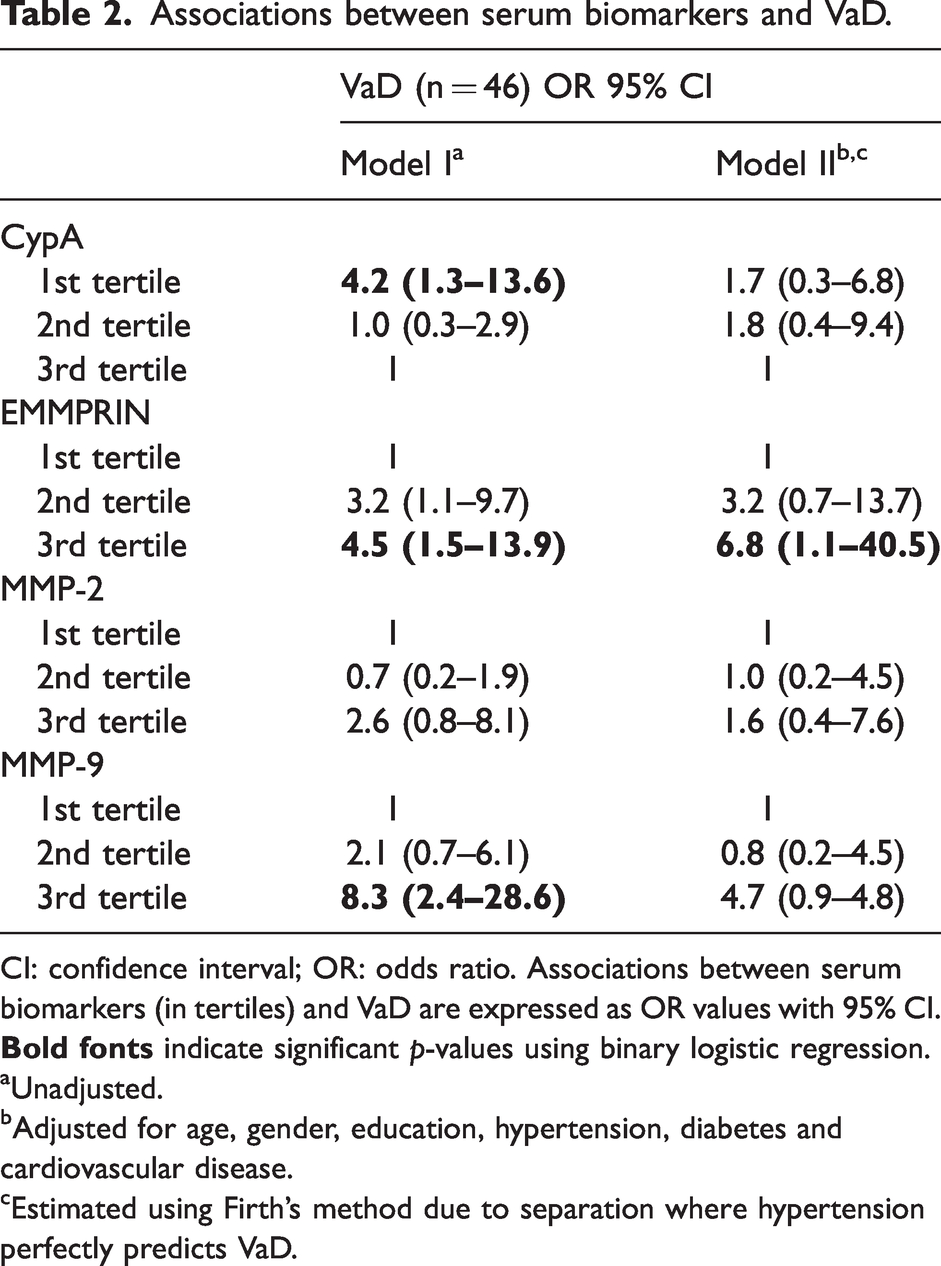
CI: confidence interval; OR: odds ratio. Associations between serum biomarkers (in tertiles) and VaD are expressed as OR values with 95% CI.
aUnadjusted.
bAdjusted for age, gender, education, hypertension, diabetes and cardiovascular disease.
cEstimated using Firth’s method due to separation where hypertension perfectly predicts VaD.
ECM proteins in serum as potential biomarkers for detecting VaD
Using ROC analyses, the potential diagnostic value of each serum biomarker in detecting VaD was assessed. The unadjusted ROC analyses summarised in Figure 1(e) and Supplementary Table S1 showed that the blood biomarkers alone were moderately useful in discriminating VaD cases from NCI controls, with AUCs ranging from 0.64 to 0.7. However, the discriminative ability of the biomarkers was improved after combining all four blood biomarkers into a composite panel (AUC = 0.78, sensitivity = 67%, specificity = 89%). The goodness of fit of the composite panel was also significantly better than those of the individual biomarkers (likelihood ratio tests, p < 0.05).
Associations of serum gelatinases with cerebrovascular disease
Given previous findings of the involvement of gelatinases in vascular remodelling and cerebrovascular disease,50–52 further analyses were performed to investigate putative links between MMP2/MMP9 and various MRI markers of cerebrovascular disease including WMH, lacunes and infarcts, which are prominent pathophysiological features in both CCH and VaD. Figures 2(d) to (f) show that serum MMP-9 concentrations were significantly higher in subjects with confluent WMH (p = 0.01) as well as with presence of lacunes (p = 0.04) and cortical infarcts (p = 0.02), whilst serum MMP-2 increases reached statistical significance only in subjects with cortical infarcts (Figure 2(c), p = 0.01). Furthermore, Fazekas scores for WMH and cortical infarct counts showed moderate (Rho values = 0.2–0.3) but significant correlations with both MMP-2 and MMP-9 (Supplementary Table S2).
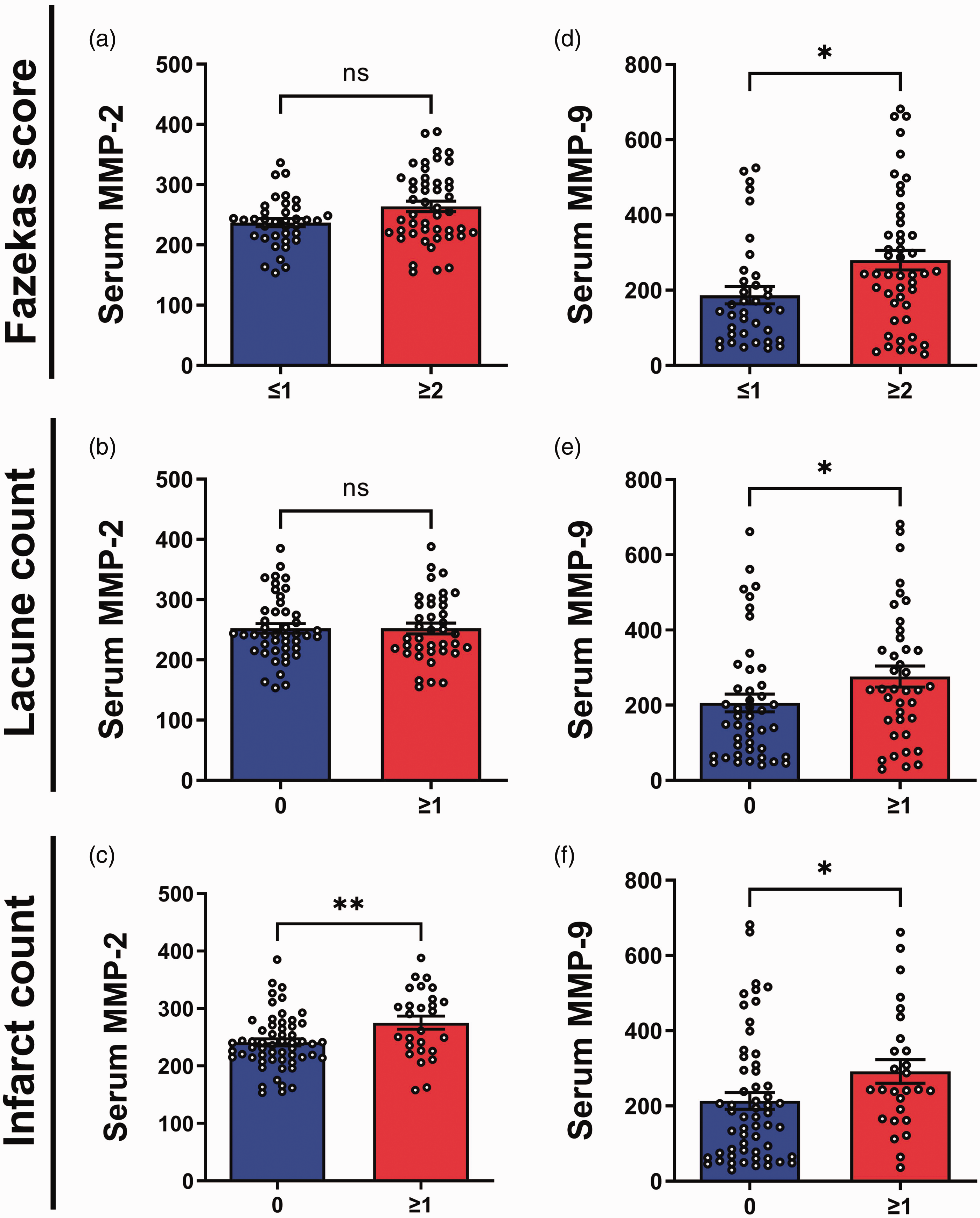
Associations of serum gelatinases with cerebrovascular diseases in a clinical study cohort. Bar graphs of (a–c) MMP-2 and (d–f) MMP-9 serum concentrations (mean ± SEM in ng/mL, with white dots indicating individual measurements) with absence or presence of (a,d) confluent white matter hyperintensities (as measured by Fazekas score ≥2), (b,e) lacunes or (c,f) cortical infarcts in a combined cohort (n = 84).
Characteristics of BCAS mice
Validation of CCH in the BCAS mice was performed via CBF measurements using a Laser Speckle Imaging technique (see Materials and Methods). Figures 3(a) and (b) showed the CBF images of sham-operated and BCAS mice as well as their respective quantification. In the sham-operated group, basal CBF remained unchanged throughout the duration of the procedure. However, in BCAS mice, CBF showed a drastic decrease post-surgery. Furthermore, Figure 3(c) demonstrated that disruptions of LFB staining indicating white matter rarefaction and lesions were apparent in the corpus callosum (paramedian and medial areas), caudoputamen, internal capsule and optic tract in the BCAS group 30 days post-surgery, in agreement with previous studies. 45 All five brain regions in BCAS animals showed grades 2 or 3 severity levels, as compared to grade 0 in Sham conditions. Taken together, BCAS induced CBF reductions and white matter lesions, providing validation for subsequent analyses as a model of CCH-associated cerebrovascular disease and VaD.11,12
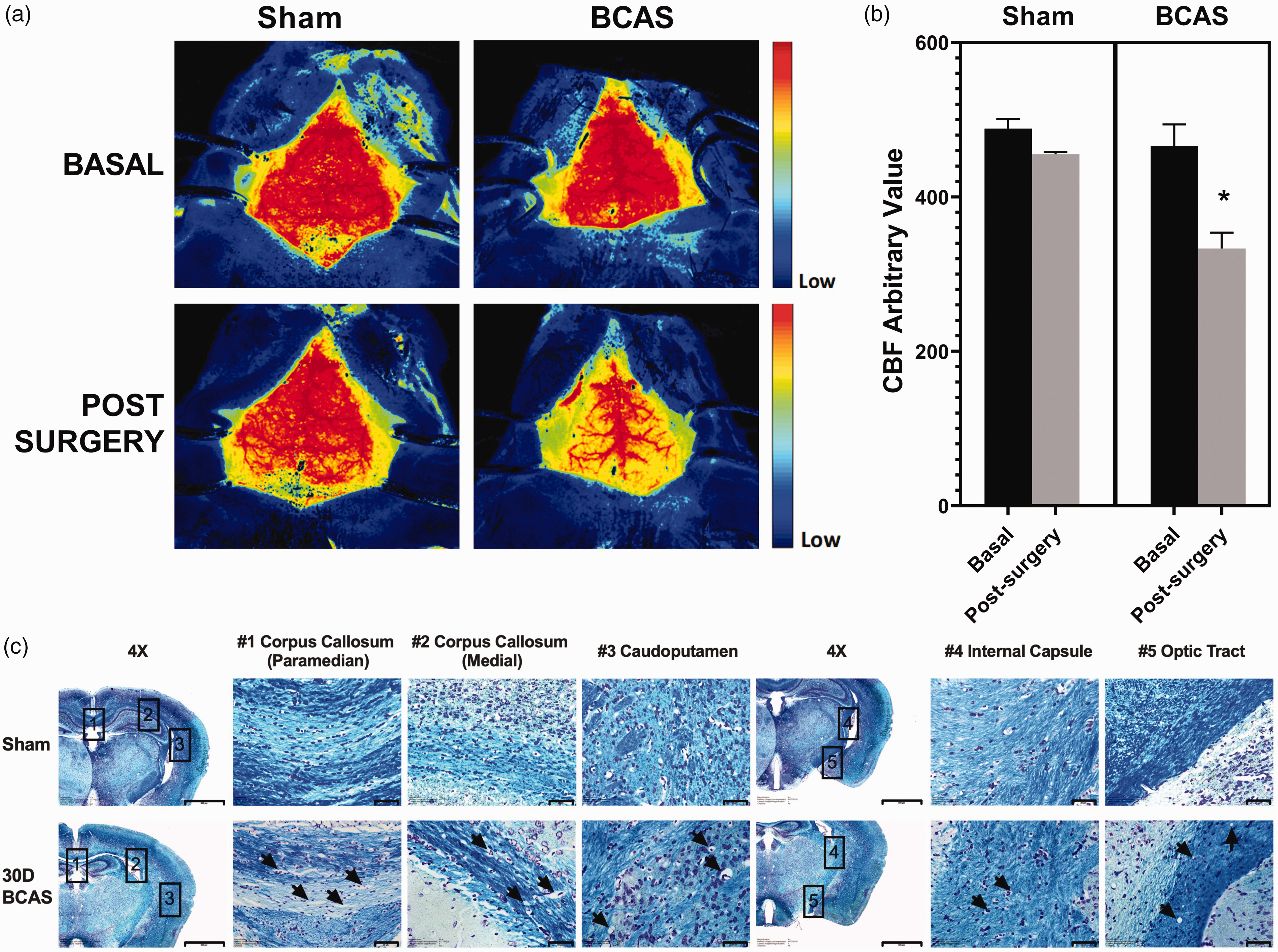
Effects of BCAS-induced chronic hypoperfusion on CBF and white matter integrity. (a) Representative CBF images with ROIs between bregma and lambda across the area of both hemispheres of the brain, (b) bar graphs showing mean CBF (±standard error of the mean, SEM) measured before (“Basal”) and immediately after the procedure (“Post-Surgery”) in both the sham-operated and BCAS C57BL/6J mice and (c) Representative images of LFB staining in sham-operated and BCAS mice 30 days after procedures demonstrated white matter rarefaction and formation of vacuoles (indicated by arrows) in the corpus callosum (paramedian and medial, regions “#1” and “#2”), caudoputamen (region “#3”), internal capsule (region “#4”) and optic tract (region “#5”) following BCAS. Coronal sections were imaged at 4x magnification, scale bar 500μm; zoomed in images were imaged at 60x magnification, scale bar 20μm. Images were taken under identical exposures and conditions. BCAS: bilateral carotid artery stenosis; CBF: cerebral blood flow; LFB: Luxol Fast Blue; ROI: region of interest.
BCAS-induced alterations of ECM protein expression
To assess potential ECM protein changes associated with BCAS, immunoreactivities of brain CypA, EMMPRIN and MMP-2 and MMP-9 were measured. Figures 4(a) and (b) showed that CypA was significantly decreased in the cortex and hippocampus of BCAS mice compared to sham-operated controls. In contrast, EMMPRIN was increased in the BCAS group, although statistical significance was reached only in the cortex (Figures 4(c) and (d)). MMP-2 was significantly elevated in BCAS cortex as well as hippocampus ( Figures 4(c) and (f)), while MMP-9 immunoreactivity was unchanged (Figures 4(g) and (h)). To further study alterations of gelatinases during BCAS-induced CCH, MMP-2 and MMP-9 concentrations in the serum of BCAS and sham animals were measured. Analysis of serum revealed a significant increase in MMP-2 following BCAS compared to sham controls (Supplementary Figure S1). In contrast, a significant reduction in the MMP-9 was observed in the BCAS group. Taken together, these observations suggest complex alterations of the CypA-EMMPRIN-gelatinase pathway in BCAS-associated CCH.
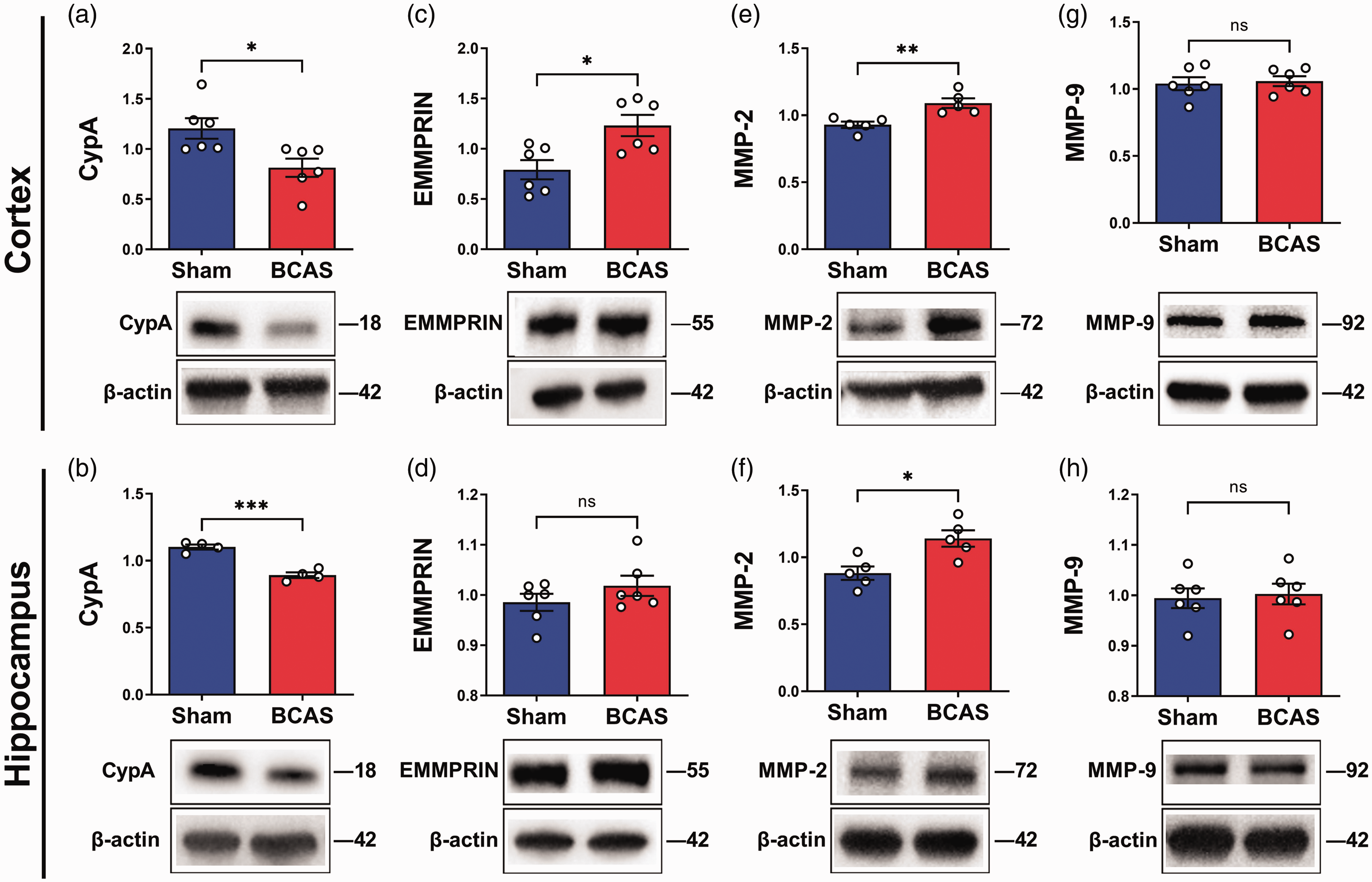
Effects of BCAS-induced chronic hypoperfusion on immunoreactivities of CypA and ECM proteins in mouse cortex and hippocampus. Representative immunoblots and bar graphs of immunoreactivities (in mean ± SEM arbitrary units normalized to β-actin, with white dots indicating individual measurements) of (a,b) CypA, (c,d) EMMPRIN, (e,f) MMP-2 and (g,h) MMP-9 in cortical and hippocampal tissues of sham-operated or BCAS mice 30 days after the procedures. Molecular weights (in kDa) are indicated on the right of each blot (all unedited blots can be found in Supplementary Figure S2). CypA: cyclophilin A; ECM: extracellular matrix; EMMPRIN: extracellular matrix metalloproteinase inducer; MMP: matrix metalloproteinase.
Discussions
EMMPRIN-mediated signalling to gelatinases is known to be involved in vascular remodelling and cerebrovascular disease,18,23,24 but its potential role in the pathophysiology of CCH and its known clinical sequela in VaD is unclear. Using both a rodent model of CCH as well as serum samples from a VaD patient cohort, we now report convergent data pointing to an altered CypA-EMMPRIN-gelatinase pathway which is implicated in both VaD and CCH. Specifically, we found decreased CypA concentrations contrasting with increased EMMPRIN in the serum of VaD patients, in parallel with lower CypA and higher EMMPRIN immunoreactivities in the cortex of BCAS mice.
Previous studies have reported the upregulation of CypA and EMMPRIN post-stroke, both in animal models31–34 as well as in clinical studies.53,54 It is intriguing that while CypA has repeatedly been reported to be upregulated in stroke, this study observed reductions of CypA both in BCAS-induced CCH mouse model as well as in VaD patients. This may be a result of the pathophysiological differences between acute stroke and chronic VaD. For example, CypA upregulation is well known to trigger apoptosis, oxidative stress and inflammation, processes which may aid in recovery after tissue damage in acute settings. 55 However, the aforementioned processes may become dysregulated and maladaptive in chronic conditions such as those associated with CCH and VaD.11,23 In this study, the upregulation of EMMPRIN in CCH and VaD may represent one such mechanism of dysregulation, leading to excessive apoptosis or inflammation, 56 as well as downstream perturbations of gelatinase levels and function (see below). In this context, the observed reductions of CypA may then be an attempt to counteract the signalling dysregulation arising from elevated EMMPRIN. This suggests that CypA may have opposing pathophysiological roles in the acute and chronic phases, and the observed CypA decreases in the BCAS model and in VaD may be an adaptive response to CCH-associated EMMPRIN dysregulation.
Notably, this study also demonstrated changes in gelatinases, namely MMP-2 and MMP-9, which are known downstream targets of EMMPRIN activation. While both gelatinases showed trends towards increases in the VaD cohort (with MMP-9 reaching statistical significance), higher MMP-2 were observed in the brain and serum of BCAS-induced mice. We observed a reduction of MMP-9 in the serum of BCAS-induced mice although there were no significant alteration in the brain. In this context, differential expressions between the two gelatinase subtypes, as well as differences in brain versus peripheral levels have previously been reported. For example, similar to the current BCAS data, a complete occlusion of bilateral carotid arteries resulted in increased brain MMP-2 but not MMP-9, 57 with similar trends also observed in post-mortem VaD brains. 21 Conversely, increased peripheral MMP-9 but not MMP-2 was observed in the cerebrospinal fluid of VaD patients. 58 Although both gelatinases are principally involved in vascular matrix remodelling, there are differences in activity temporal profiles throughout the disease process,25,26,30 in the specificities of substrate recognition, 59 as well as in subtype-specific associations with pathogenic mechanisms (e.g., MMP-2 correlated with inflammation and is also the primary modulator of ECM after vascular injury, while MMP-9 was associated with endothelial dysfunction and lipid metabolism),57,60 which may explain the observed differences between MMP-2 and MMP-9 in the BCAS model versus VaD. The different trends between the two gelatinases in human and mice may also be due to species difference. In humans, complex diseases are usually associated with perturbation of multiple pathways; and VaD pathophysiology may likewise involve processes besides cerebral hypoperfusion as modelled in the BCAS mice. Furthermore, MMP-9 inhibitors have been reported to reduce acute ischemic injury but were ineffective in chronic hypertensive animals. 61 Taken together, these results suggest that further enzymatic activity- and timepoint- based studies are needed to better delineate the specific involvement of MMP-2 and MMP-9 in CCH and VaD.
Interestingly, this study further showed that the increased MMP-9 in VaD also associated with confluent WMH as well as the presence of lacunes and cortical infarcts, while MMP-2 was associated only with cortical infarcts. The link between the CypA pathway and MRI markers of cerebrovascular disease is in concordance with a previous study which showed co-expression of MMP-9 with ischemic infarction and haemorrhage in post-stroke brains. 27 Additionally, previous studies also reported an association between blood MMP-9 levels and infarct volumes 28 as well as between MMP-2 levels and WMH volume 62 in acute ischemic stroke patients. WMH is known to be a contributor to the development of infarcts,63–66 sharing common pathophysiological mechanisms such as inflammation.67–69 The data therefore suggests involvement of EMMPRIN-induced gelatinase dysregulation in cerebrovascular disease markers of VaD. However, further validation using a larger, longitudinal study is required.
Strengths of the current study include the use of both animal model and clinical cohort with neuroimaging, which allows further insights into VaD pathophysiology from both a neurochemical and clinical perspective, as well as being able to assess the potential utility of ECM proteins as biomarkers of VaD and cerebrovascular disease. Moreover, possible confounding effects of demographic characteristics and vascular risk factors were taken into account using multiple regression analyses.
The present study also has several limitations. Firstly, despite having the advantage of having data from both animal model and clinical cohort, there are fundamental differences between an experimental animal model and human disease cases. Therefore, the pathophysiological validity of BCAS-induced CCH as the model for VaD and vascular cognitive impairment awaits further assessment. While our study suggested a role of the CypA-EMMPRIN-gelatinase pathway in BCAS-induced CCH and its related vascular features like white matter lesions, causal effects can only be ascertained with genetic manipulation (for e.g., EMMPRIN knockdown), which awaits future investigations. Similarly, the cross-sectional design of the clinical study was not designed to infer any causal or longitudinal relationships between biomarker alternations and vascular pathology. Furthermore, potential confounding effects by other ECM proteins linked to gelatinase pathways, such as tissue inhibitors of metalloproteinases (TIMPs), were not accounted for in the present study. Given that TIMPs were shown to be associated with VaD,70–72 it is therefore worthwhile assessing the potential roles of TIMPs and other ECM components in cerebrovascular disease pathogenesis. Moreover, as both the immunoblots and ELISAs employed in this study recognized pro- and cleaved forms of the gelatinases, we were thus unable to distinguish between active and inactive forms of the proteins. Further studies using technique such as in situ zymography could be used to determine the proteolytic activities of gelatinases and thus provide further insights into the nature of gelatinase involvement in cerebrovascular disease. Lastly, the current study included only VaD, which is part of a larger spectrum of vascular cognitive impairment. The potential implication of CypA-EMMPRIN-gelatinase dysregulation in other types of vascular cognitive impairment, as well as other dementia aetiologies such as Alzheimer’s disease thus awaits further investigation.
Conclusion
Using a well-validated animal model together with a well-characterized clinical cohort of VaD subjects, we demonstrated dysregulation of the CypA-EMMPRIN-gelatinase pathway is associated with VaD. Preliminary data from our BCAS-induced mouse model as well as from neuroimaging observation provided insights into the effect of cerebrovascular disease such as CCH and white matter lesions in this pathway dysregulation. Our findings provide impetus for further investigations of the biomarker utility of ECM proteins, and also point to components of the pathway, such as EMMPRIN, as potential targets for VaD pharmacotherapy.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221146401 - Supplemental material for Chronic cerebral hypoperfusion alters the CypA-EMMPRIN-gelatinase pathway: Implications for vascular dementia
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221146401 for Chronic cerebral hypoperfusion alters the CypA-EMMPRIN-gelatinase pathway: Implications for vascular dementia by Yuek Ling Chai, Vismitha Rajeev, Luting Poh, Sharmelee Selvaraji, Saima Hilal, Christopher P Chen, Dong-Gyu Jo, Edward H Koo, Thiruma V Arumugam and Mitchell KP Lai in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Medical Research Council (MOH-000500-01, MOH-000707-01 to CPC, MKPL; NMRC/CG/M006/2017 to CPC; NMRC-CBRG-0102/2016, NMRC/OFIRG/0036/2017 to TVA), the Yong Loo Lin School of Medicine (Post-Doctoral Fellowship Award NUSMED/2021/PDF/05 to YLC), the National University Health System (A-0006090-00-00 to SH) and a start-up fund from La Trobe University, Melbourne to TVA.
Acknowledgements
The authors would like to thank all patients and their families for their participation in this study. Dr Tan Boon Yeow from St Luke’s Hospital, Singapore, is acknowledged for his contributions to patient recruitment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
EHK, TVA and MKPL conceived and designed the study. DGJ, EHK, TVA and MKPL developed the methodology and provided domain expertise. YLC, VR, LP, SS and TVA performed the experiments. CPC and SH provided clinical data. YLC and VR analysed the data and wrote the manuscript. CPC, TVA and MKPL provided overall supervision. All authors have read, revised and approved the submitted version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
