Abstract
Ischemic stroke results in blood-brain barrier (BBB) disruption, during which the reciprocal interaction between ischemic neurons and components of the BBB appears to play a critical role. However, the underlying mechanisms for BBB protection remain largely unknown. In this study, we found that Serpina3n, a serine protease inhibitor, was significantly upregulated in the ischemic brain, predominantly in ischemic neurons from 6 hours to 3 days after stroke. Using neuron-specific adeno-associated virus (AAV), intranasal delivery of recombinant protein, and immune-deficient Rag1−/− mice, we demonstrated that Serpina3n attenuated BBB disruption and immune cell infiltration following stroke by inhibiting the activity of granzyme B (GZMB) and neutrophil elastase (NE) secreted by T cells and neutrophils. Furthermore, we found that intranasal delivery of rSerpina3n significantly attenuated the neurologic deficits after stroke. In conclusion, Serpina3n is a novel ischemic neuron-derived proteinase inhibitor that counterbalances BBB disruption induced by peripheral T cell and neutrophil infiltration after ischemic stroke. These findings reveal a novel endogenous protective mechanism against BBB damage with Serpina3n being a potential therapeutic target in ischemic stroke.
Introduction
Stroke is a common cerebrovascular disease that affects millions of people annually across the world, of whom about 87% suffer from ischemic stroke.1,2 Ischemic stroke leads to severe neurological deficits. Ischemic stroke leads to severe neurological deficits. Established approaches to improve blood supply to the ischemic brain are currently limited to thrombolytic treatment and mechanical thrombectomy in the acute stage. 3 Due to the extremely narrow time window for recanalization procedures, only a small fraction of patients can benefit from such therapies. Thus, there is an urgent unmet need for treatments to prevent secondary brain injury, for instance resulting from oxidative stress and neuroinflammation.
Blood-brain barrier (BBB) damage is a critical pathological process that profoundly aggravates ischemic brain injury and long-term neurological outcomes. 4 There are complex interactions between the ischemic neurons and the compromised BBB after stroke. For example, immune cells detect danger-associated molecular patterns (DAMPs) released by injured neurons, including adenosine, heat shock proteins (HSPs), high mobility group box 1 (HMGB1), and interleukin (IL)-33, and initiate immune responses both in the brain and in the periphery.5–7 Peripheral immune cells infiltrate the brain across the damaged BBB and exacerbate neuroinflammation in the parenchyma. This further compromises BBB integrity, thereby forming a vicious cycle that augments long-term brain damage and neurological deficits after stroke. 8 In particular, neutrophils and T cells secreting serine proteases that degrade the basal membrane and junctional proteins of the BBB are among the most important infiltrating immune cell populations.9,10 Several studies have reported that depleting T cells or neutrophils is protective in BBB-disruption models.9,11–13 However, the exact mechanisms behind these protective effects are not fully understood.
Serpins are a family of structurally conserved protease inhibitors. 14 Serpina3n, the mouse homolog of the human SerpinA3, 15 is in astrocytes 16 after experimental stroke. This study revealed that Serpina3n is also expressed in neurons following stroke and revealed the spatial distribution and temporal profiles of this Serpina3n expression. Furthermore, the potential pathophysiological role of Serpina3n in BBB damage and neuroinflammation after ischemic stroke was investigated by investigating the expression pattern of Serpina3n and its functional roles in BBB damage, immune cell infiltration, and neurological impairment using a mouse model of focal cerebral ischemia and reperfusion. Our results identify Serpina3n as an endogenous neuroprotectant against BBB damage and neuroinflammation after stroke.
Materials and methods
Animals
Male C57BL/6 mice were purchased from Shanghai JSJ Laboratory Animals, Rag1−/− mice were purchased from Suzhou Cyagen Biosciences. Serpina3n−/− mice were purchased from Suzhou Cyagen Biosciences. All mice were housed under standard laboratory conditions (22°C and a 12‐hour light‐dark cycle with free access to food and water). All experiments were approved by the Renji Hospital Institutional Animal Care and Use Committee and performed by the Institutional Guide for the Care and Use of Laboratory Animals. Animal data reporting followed the ARRIVE 2.0 guidelines 17 and STAIR X Stroke recommendations. 18
Murine focal ischemia model
Male 8 to 10-week‐old C57BL/6 mice were subjected to transient tMCAO (tMCAO) as previously described. 19 Briefly, mice were anesthetized with 2% isoflurane in a 30% O2/70%N2 mixture. A skin incision was made at the neck of mice and the left CCA was exposed and ligated. We induced tMCAO by intraluminal occlusion of the left MCA with a silicone plug for 1 hour. Sham‐operated animals underwent anesthesia and surgical exposure of arteries but without artery occlusion. Body temperature was maintained at 37 ± 0.5°C with a heating pad during surgical procedures.
Randomization, blinding, and study timeline
All behavioral procedures, histological and flow cytometry assessments were performed by investigators blinded to experimental group assignments. Randomization was performed using the envelope method. The overall study timeline is as follows:
(1) Day 0: tMCAO; Hour 6, Day 1, Day 3, and Day 5: Collect tissue for Western blot, immunostaining. (2) Day 0: tMCAO; Day 3: Collect tissue for Evans blue staining, Western blot, immunostaining, flow cytometry, RNA sequencing. (3) Day 0: tMCAO; Day – 1–28: Day – 1, 1, 3, 5, 7, 14, 28 for neurological deficit score, adhesive removal, Y-maze, and grid walk; Day 30: Collect tissue.
Animals were excluded if the suture was not successfully placed to occlude the tMCAO which was monitored by two-dimensional laser speckle. The success of the tMCAO model was monitored by the two-dimensional laser speckle technique. Briefly, cerebral perfusion images were acquired using the PeriCam PSI System (Perimed) positioned above the mouse heads before, during ischemia, and after reperfusion. Speckle contrast was defined as the ratio of SD of pixel intensity to the mean pixel intensity. The speckle visibility relative to the velocity of the light-scattering particles was converted to correlation time values, which are inversely and linearly proportional to the mean blood velocity. CBF changes were expressed as a percentage of pre-tMCAO baselines.
Transcriptome experimental method and data analysis
Total RNA extraction
Total RNA was extracted from tissues using Trizol (Invitrogen, Carlsbad, CA, USA) according to manufacturer’s instruction. In brief, about 60 mg of tissues were frozen by liquid nitrogen in a 2 ml tube and subsequently homogenized for 2 minutes, followed by horizontal resting for 5 minutes. The homogenate was centrifuged for 5 minutes at 12,000 × g at 4°C. The supernatant was then transferred into a new EP tube containing 0.3 mL chloroform/isoamyl alcohol (24:1). The mix was shacked vigorously for 15 s, and then centrifuged at 12,000×g for 10 minutes at 4°C. After centrifugation, the upper aqueous phase containing RNA was transferred into a new tube and an equal volume of isopropyl alcohol was added before centrifuging at 13,600 rpm for 20 minutes at 4°C. After discarding the supernatant, the RNA pellet was washed twice with 1 mL 75% ethanol. Thereafter, the mix was centrifuged at 13,600 rpm for 3 minutes at 4°C to collect residual ethanol, followed by pellet air dry for 5–10 minutes in the biosafety cabinet. Finally, 25 μL–100μL of DEPC-treated water was added to dissolve the RNA. Subsequently, total RNA was qualified and quantified using a Nano Drop and Agilent 2100 bioanalyzer (Thermo Fisher Scientific, MA, USA).
mRNA library construction
Oligo(dT)-attached magnetic beads were used to purify mRNA. Purified mRNA was fragmented into small pieces with fragment buffer at an appropriate temperature. First-strand cDNA was generated using random hexamer-primed reverse transcription, followed by a second-strand cDNA synthesis. Afterwards, A-Tailing Mix and RNA Index Adapters were added to the incubation mix. cDNA fragments obtained from the previous step were amplified by PCR, and products were purified by Ampure XP Beads, then dissolved in EB solution. The product was validated on the Agilent Technologies 2100 bioanalyzer for quality control. The double stranded PCR products from the previous step were heat-denatured and circularized by a splint oligo sequence to get the final library. The single strand circle DNA (ssCir DNA) was formatted as the final library. The final library was amplified with phi29 to make DNA nanoball (DNB) which had more than 300 copies of one molecular, DNBs were loaded into the patterned nanoarray and pair end 100 bases reads were generated on BGIseq500 platform (BGI-Shenzhen, China).
Data analysis
The sequencing data was filtered with SOAPnuke (v1.5.2) 20 to remove reads (i) containing sequencing adapter; (ii) whose low-quality base ratio (base quality less than or equal to 5) is more than 20%; (iii) whose unknown base (‘N' base) ratio is more than 5%. Thereafter, clean reads were obtained and stored in FASTQ format before being mapped to the reference genome using HISAT2 (v2.0.4). 21 After that, Ericscript (v0.5.5) 22 and rMATS (V3.2.5) 23 were used to create fusion genes and differential splicing genes (DSGs), respectively. Bowtie2 (v2.2.5) 24 was applied to align the clean reads to the gene set, a database for this brain tissue built by BGI (Beijing Genomic Institute in ShenZhen), which was known and novel, coding transcripts were included, then expression level of gene was calculated by RSEM (v1.2.12). 25 The heatmap was drawn by pheatmap (v1.0.8) 26 according to the gene expression in different samples. Essentially, differential expression analysis was performed using the DESeq2(v1.4.5) 27 with Q value ≤0.05. To gain insight into the change of phenotype, GO (http://www.geneontology.org/) and KEGG (https://www.kegg.jp/) enrichment analysis of annotated different expression genes were performed by Phyper (https://en.wikipedia.org/wiki/Hypergeometric_distribution) based on a hypergeometric test. The significant levels of terms and pathways were corrected by Q value with a rigorous threshold (Q value ≤0.05) by Bonferroni. The STRING database (https://string-db.org/) was used for protein interaction analysis to reveal the relationship between target genes.
Serpina3n knock-down with adeno-associated virus
In vivo gene knock-down was achieved by Adeno-associated virus (AAV) vectors. Adeno-associated shSerpina3n virus (pAAV-U6-MCS-CAG-shRNA (Serpina3n)-EGFP, AAV-PHP.eB) and neuron-specific adeno-associated shSerpina3n virus (pAAV-hSyn-EGFP-5′miR-30a-shRNA (Serpina3n)-3′-miR-30a-WPREs, AAV-PHP.eB) were chemically synthesized by Genechem (Shanghai, China). The virus serotype was PHP.eB. AAV-shCtrl (pAAV-U6-MCS-CAG-shRNA (Scramble)-EGFP; pAAV-hSyn-EGFP-5′miR-30a-shRNA (Scramble)-3′-miR-30a-WPREs) was generated after cloning short-hairpin RNA (shRNA) fragments into the AAV vector GV478 (Shanghai Genechem Company Limited). AAV packaging was performed by cotransfecting AAV-293 cells with the recombinant AAV vector, pAAV-RC vector, and pHelper vector. AAVs were collected from the AAV-293 cell supernatant, condensed, and purified for further animal experiments. AAV was diluted in phosphate-buffered saline (PBS) to 2 × 10 11 genome copies/100 μl. Three weeks prior to tMCAO, mice were injected intravenously with 100 μl of AAV suspension (approximately 2 × 1011 genome copies/mouse).
Recombinant Serpina3n intranasal delivery
Mice were fully anesthetized by intraperitoneal(i.p.) injection of pentobarbital sodium (50 mg/kg) and then infected intranasally by dropping 4 μl of PBS containing 0 nmol/kg, 0.6 nmol/kg, 2 nmol/kg, or 6 nmol/kg recombinant Serpina3n(rSerpina3n) (4709-PI, R&D Systems) at 30 minutes, 24 hours, and 48 hours after tMCAO. PBS intranasal delivery served as control.
Neutrophil depletion
Neutrophils were depleted using anti-Gr1 mAb (BE0075, BioXCell).28–30 Specifically, starting at 1 day before tMCAO and 1 day after tMCAO, mice were injected i.p. with anti-Gr-1 mAb (0.1 mg/day) to deplete neutrophils or with rat IgG (BP0089, BioXCell) as a control. The efficiency of neutrophil depletion in tMCAO mice was confirmed by flow cytometry.
Western blot
Brain lysates of tMCAO and sham mice were prepared by homogenizing tissue in radioimmuno precipitation assay buffer (RIPA) solution (P0013B, Beyotime Biotechnology, China) containing a protease inhibitor cocktail (P1005, Beyotime Biotechnology, China). Proteins were separated on 12.5% polyacrylamide gels (PG113, EpiZyme Biotechnology, China) and transferred onto a nitrocellulose membrane (IPFL00010; Millipore) for 45 min at 0.4 A (constant). The membrane was blocked by incubation in 5% milk in 0.1 M PBS, 0.1% Tween-20 for 1 hour at room temperature (RT). The membrane was stained with anti-Serpina3n (1:2000, AF4709, R&D Systems), anti-NE (1:1000, 63610, CST), anti-GZMB (1:1000, 17215, CST), anti-GAPDH (1:5000, 5174, CST), and anti-β-tubulin (1:5000, 2128, CST). Membranes were then incubated with appropriate secondary HRP-conjugated antibody (1:10000, ABclonal, China, 1 h RT). Visualized using Immobilon Western HRP Substrate (Millipore) on ChemiDoc Imaging System (Bio-Rad). The optical density of the signals on the membrane was quantified using grayscale measurements in NIH Image J (1.52a) and converted to fold change.
Immunofluorescence
Coronal sections were incubated with 10% normal donkey serum in PBS containing 1% Triton X‐100 at RT for 30 minutes, followed by incubation with appropriate primary antibodies overnight at 4°C in the same buffer. The anti-Serpina3n(1:200, AF4709, R&D System), anti-NeuN(1:500, MAB377, Millipore), anti‐Iba‐1(1:500, ab178846, Abcam), anti‐GFAP (1:500, ab7290, Abcam), anti-Olig2(1:500, ab9610, Millipore), anti-PDGFRa(1:500, ab203491, Abcam), anti-MAP2(1:500, ab183830, Abcam), anti-CD31(1:500, AF3628, R&D System), anti-ZO1(1:150, 40-2200, Invitrogen), anti-Fibrinogen(1:150, ab193932, Abcam), anti-CD3ε(1:100, 78588, CST), and anti-Ly6G(1:100, ab25377, Abcam) antibodies were used. After primary antibody incubation, sections were washed with PBS containing 0.3% Triton X‐100 three times at RT before incubation with appropriate fluorescent‐labeled secondary antibodies (1:1000, Abcam) for 1 hour at RT. DAPI (1:2000, D9542, Sigma-Aldrich) counterstaining was performed to visualize nuclei. Sections were then washed with PBS containing 0.3% Triton X‐100 and mounted with water‐based mounting medium containing antifading agents (0100-01, Southernbiotech). All the confocal images were captured on a laser scanning confocal microscope (Olympus Fluoview FV3000, Olympus). The numbers of target immunopositive cells were quantified by a blinded investigator using NIH Image J (1.52a). Three randomly selected microscopic fields in the cortical infract core and border zone area of each section were analyzed for each brain by a blinded investigator.
Flow cytometry
Peripheral blood single cell suspensions were prepared using red blood cell lysis buffer (555899, BD) and filtered through a 70 µm nylon membrane. The lesioned hemisphere in tMCAO mice or the corresponding one in sham animals was homogenized using the gentle MACS Dissociator with Neural Tissue Dissociation Kit (130-093-231, Miltenyi Biotec) according to manufacturer’s instructions. For immune cell characterization, antibodies being specific for mouse cell surface markers anti-CD3-PE-Cy7 (552774, BD), anti-Ly6G-BV421 (562737, BD), anti-CD11b-APC (17-0118-42, eBioscience) were used. BD FACS Verse (BD Bioscience) was used to collect stained cells and FlowJo software (TreeStar) was used to analyze the data.
Measurements of infarct volume
Infarct volume was evaluated by microtubule-associated protein 2 (MAP-2) immunofluorescence staining as described above and determined with NIH Image J (1.52a) analysis by an investigator who was blinded to the experimental group assignment.
Assessment of the BBB leakage
Evans blue staining
BBB leakage was evaluated by Evans blue (sc-203736, Santa Cruz) staining. Briefly, 2% Evans blue was injected intravenously 48 hours after tMCAO or following the sham operation. 24 hours after the Evans blue injection, the mice were sacrificed and perfused with 20 ml saline. Both brain hemispheres were isolated and separated. Next, each hemisphere was weighed, homogenized in 1 ml 50% trichloroacetic acid (TCA), and centrifuged (12000 rpm) for 20 minutes at RT. The supernatant was diluted 1:4 with ethanol and Evans blue was measured with a SpectraMax M2e plate reader (Molecular Devices) (A620/A650) and presented as ng/mg in tMCAO or sham brain tissue.
Plasma IgG and fibrinogen extravasation
Extravasation of plasma IgG or fibrinogen in tMCAO or sham brain tissue was determined with immunofluorescence staining, as described above.
Neurological function evaluation
The modified Garcia Score, 31 grid walk, 32 adhesive test, 33 and Y-maze 34 were performed as previously described to assess sensorimotor functions before and after surgery by investigators who were blinded to experimental group assignments. The modified Garcia Score is a well‐established sensorimotor assessment system consisting of seven individual tests, of which one measures sensory function while four-measure motor function. We scored each test from 0 to 3 (maximal score = 15): (a) body proprioception, (b) forelimb walking, (c) limb symmetry, (d) lateral turning, and (e) climbing as described. In the grid walk test, mice were placed on an elevated steel grid and the foot fault errors (when the animal’s forelimb was misplaced and fell through the grid) were recorded as the mice moved. Data are presented as the footfault index (left forelimb foot fault errors/total amount of left forelimb steps – right impaired forelimb foot fault errors/total amount of right forelimb steps). In the adhesive removal test, two pieces of adhesive tape (0.3 * 0.4 cm2) were applied with equal pressure on each animal paw. The order of placement of the adhesive (right or left) was alternated between each animal and each session. Then, the mouse was gently placed in a Perspex box, and the seconds to remove each adhesive tape were recorded. Mice were trained daily before surgery (for 1 week) and regularly tested after stroke. The Y-maze equipment consisted of three arms (40 cm in length, 10 cm in width, and 12 cm in height). At the beginning of the test, the mice of each group were placed in one fixed arm, and the number of arm alternations was observed using a video camera. An alternation was defined as the entrance into each of the three arms during rotation. The alternations were calculated according to the equation: Alternation (%) = ([Number of alternations]/[Total arm invasion –2]) * 100.
Granzyme B and neutrophil elastase activity assay
The activity of GZMB and NE in the brain tissue of mice was measured by using the chromogenic substrate Granzyme B Activity Assay Kit (MAK246, Sigma-Aldrich) and Neutrophil Elastase Activity Assay Kit (MAK176, Sigma-Aldrich). Tissue (50 mg) from the ischemic brain of the tMCAO mouse was homogenized in 200 µL of ice-cold Granzyme B Assay Buffer or NE Assay Buffer. Centrifuged the samples at 13,000 g for 10 minutes and added 50 µL of the supernatant to 50 µL of the Reaction Mix (For Granzyme B: 45 µL of Granzyme B Assay Buffer and 5 µL of Granzyme B Substrate; For NE: 48 µL of NE Assay Buffer and 2 µL of NE Substrate) in a final volume of 100 µl. The fluorescence intensity was measured using λEx = 380 nm/λEm = 500 nm every 5 minutes during 35 minutes in 96-well plates with the SpectraMax M2e plate reader (Molecular Devices).
Statistical analysis
All statistics were performed using GraphPad Prism v.6 or the implemented statistical tests of the respective R packages. The Shapiro-Wilk normality test was initially performed on all datasets. A two-tailed Student’s t-test was used for the pairwise comparison between two groups. The rest of the data were analyzed using a one-way or two-way ANOVA as appropriate. Multiple comparison procedures were carried out to identify specific between-group differences using post hoc Tukey's tests. Results were presented as mean ± SD. The correlation analyses were performed using Pearson correlation analysis. P ≤ 0.05 was considered statistically significant.
Results
Serpina3n is up-regulated in the cerebral infarction area after tMCAO
To investigate the transcriptional profiles of the serine protease inhibitor (Serpin) family after stroke, tissues from the mouse cerebral infarct area 3 days after tMCAO (n = 3) and sham group (n = 3) were processed for RNA sequencing (RNA-seq) (Figure 1(a), Supplementary Fig. 1). We found a significant change in 4999 unique transcripts (P < 0.01, 2-fold threshold) between the tMCAO and sham groups (Supplementary Fig. 1(A)). Gene Set Enrichment Analysis (GSEA) and GO enrichment analysis of DEGs (different expression genes) revealed that the Gene Ontology terms related to immune inflammation, such as innate immune response (GO:0045087), inflammatory response (GO:0006954), and immune response (GO:0006955) were strongly enriched in tMCAO mice (Supplementary Fig. 1(D), (E)). Using differential gene expression analysis, we found that the expressions of only two Serpin family members, Serpina3n and Serpine1, were highly upregulated (Figure 1(a)). Serpine1 is the inhibitor of tissue plasminogen activator (tPA) and urokinase plasminogen activator (uPA). Interestingly, Serpina3n is a secretory serine protease inhibitor 15 that can inactivate serine proteases, thus leading to suppressed inflammation and cell apoptosis. 35 We subjected C57/BL6 mice to 1 hour of tMCAO followed by 6 hours, 24 hours, 72 hours, and 120 hours of reperfusion and found the expression of Serpina3n was upregulated (peaking at 3 days) after tMCAO as revealed by western blot and immunofluorescence (Figure 1(b), Supplementary Fig. 2(A)). We used STRING DB network analysis (https://string-db.org/) 36 to plot the proteins that interacted with Serpina3n and found a strong interaction between neutrophil elastase (Elane) and Serpina3n (Supplementary Fig. 1(F)). These results suggest that Serpina3n is an endogenous serine protease inhibitor induced by cerebral ischemic brain injury that may impact neuroinflammation after stroke.
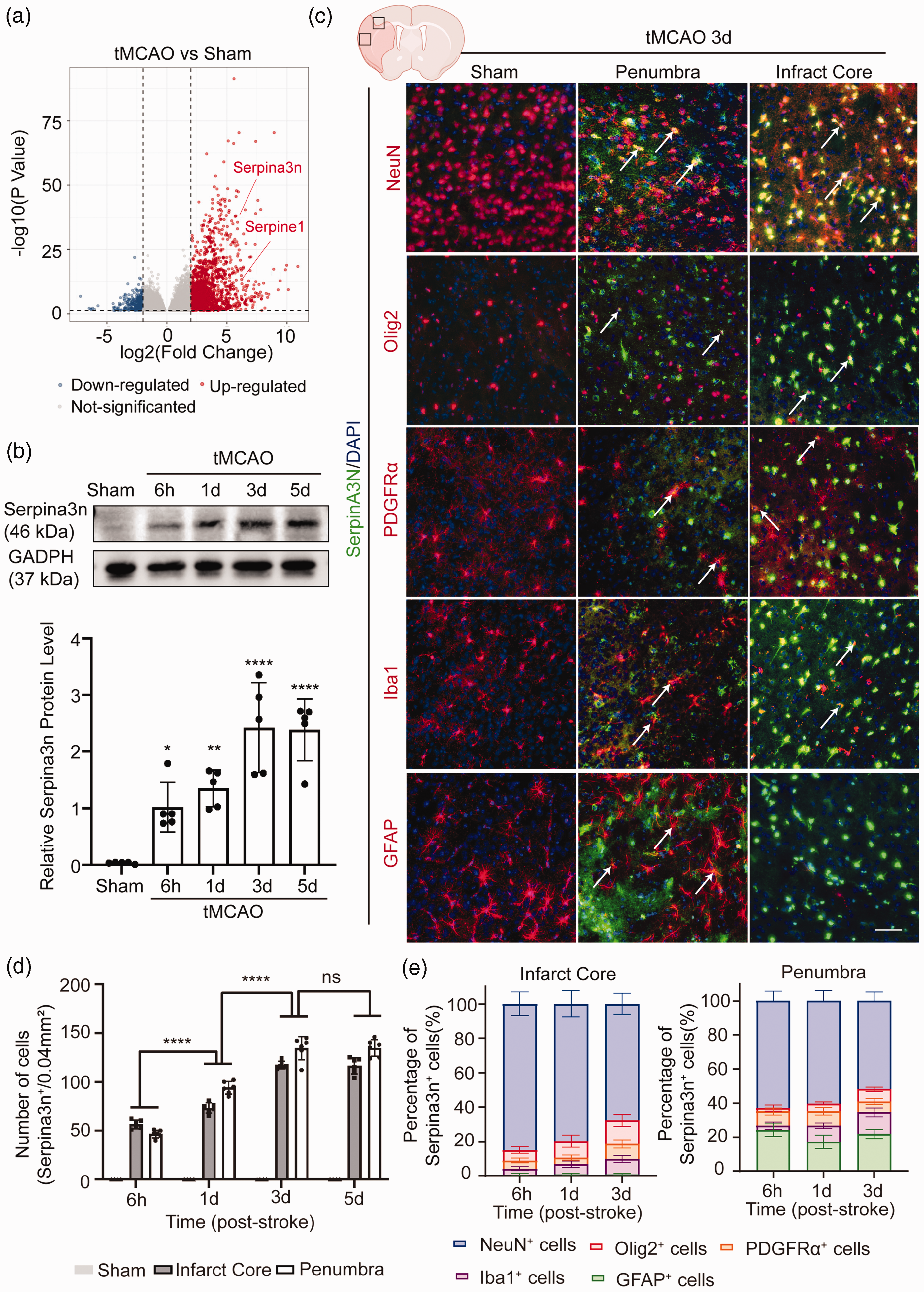
Serpina3n expression is upregulated after tMCAO. (a) Differentially expressed genes (DEGs) are displayed on a volcano plot. Red and blue spots represent genes whose expression was significantly increased and decreased, respectively, after tMCAO compared to sham controls (P < 0.01, | Log2 Fold Change ≥2) Gray spots represent genes without significant changes. (b) Representative immunoblots for Serpina3n of the infarcted cerebral cortex at 6h, 1d, 3d, and 5d after tMCAO or sham operation. The band intensities are relative to GAPDH. All data are presented as means ± SD, n = 5 per group. *P ≤ 0.05, **P ≤ 0.01, ****P ≤ 0.0001, one‐way ANOVA, and post hoc Tukey's tests. (c) Representative images of double-label immunofluorescence for Serpina3n and the cell-type markers NeuN, Olig2, PDGFRa, Iba1, and GFAP in the infarct core and peri-infarct regions of the brain at 3d after tMCAO or sham operation. Scale bar, 50 µm. (d) Quantification of Serpina3n+ cells in the brain's infarct core and peri-infarct regions at 6h, 1d, and 3d after tMCAO or sham operation. All data are presented as means ± SD, n = 6 per group. ****P ≤ 0.0001, ns P > 0.05, two‐way ANOVA, and post hoc Tukey's tests and (e) Quantification of the proportion of Serpina3n+ cells in the infarct core and peri-infarct regions of the brain at 6h, 1d, and 3d after tMCAO or sham operation. All data are presented as means ± SD, n = 6 per group.
To investigate the temporal and spatial distribution as well as the cell type of Serpina3n protein expression after stroke, mouse brain sections were co-stained with anti-NeuN (neurons), anti-olig2 (oligodendrocytes), anti-PDGFRa (oligodendrocyte progenitor cells, OPCs), anti-Iba1 (microglia/macrophages), anti-GFAP (astrocytes) and anti-Serpina3n antibodies (Figure 1(c), Supplementary Fig. 3). We found that the NeuN+Serpina3n+ cells were the most abundant cell types in Serpina3n+ cells (accounting for 80.05% at 6 hours, 79.73% at 24 hours, 67.76% at 72 hours and 62.82% at 6 hours, 60.25% at 24 hours, 51.80% at 72 hours in infarction core and ischemic penumbra areas respectively) (Figure 1(e), Supplementary Fig. 2(B)). Most PDGFRa+Serpina3n+, Iba1+SerpinA3+ and GFAP+Serpina3n+ cells were observed in penumbra areas (Supplementary Fig. 2(B)). These results suggest that neurons are the main source of Serpina3n expression in cortical areas after stroke.
Serpina3n knock-out increases infarct volume and exacerbates neurologic deficits after stroke
Serpina3n knock-out (Serpina3n−/−) mice were used to eliminate the expression of Serpina3n and MAP2 immunohistochemistry was applied to compare infarct volumes. Serpina3n knock-out significantly increased the infarct volume after tMCAO (Figure 2(a) and (b)). We also examined BBB disruption by quantifying the extravasation of plasma-derived IgG and found that Serpina3n knock-out significantly aggravated the BBB extravasation in stroke mice (Figure 2(a) and (b)). These results suggest that Serpina3n knock-out increases infarct volume and BBB disruption in the ischemic brain after stroke.
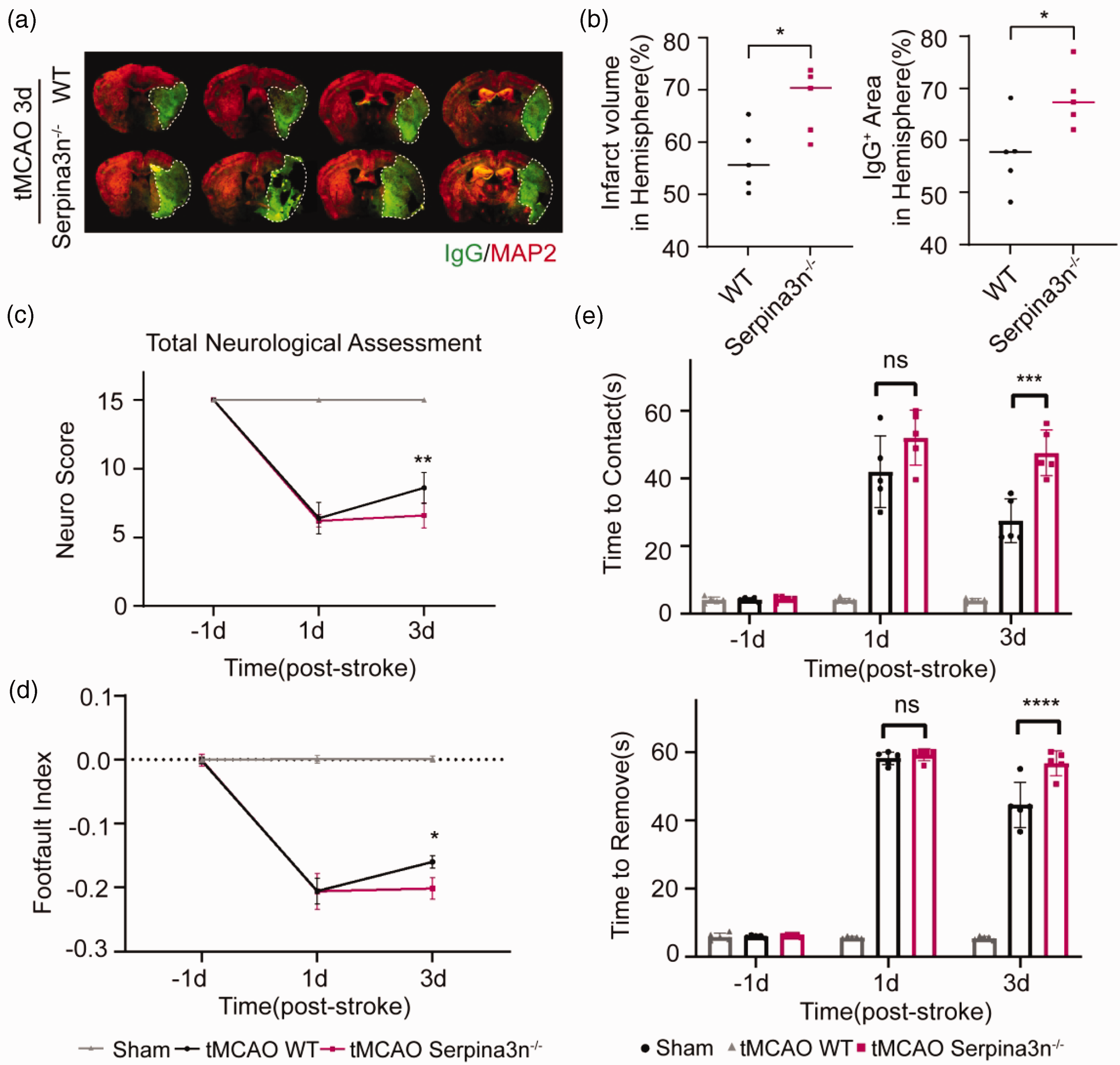
Serpina3n knock-out increases infarct volume and exacerbates neurologic deficits after stroke. (a, b) Double-label immunofluorescence for MAP2 and IgG in the brain of Serpina3n knock-out (Serpina3n−/−) or wild type (WT) mice at 3d after tMCAO. Quantification of infarct volume (MAP2 negative) and IgG extravasation area in ischemic brains. All data are presented as means ± SD, n = 5 per group. *P ≤ 0.05, one‐way ANOVA, and post hoc Tukey's tests. (c–e) Sensorimotor functions were significantly aggravated in Serpina3n knock-out mice after tMCAO compared to WT mice, as assessed by the Garcia Score (c), the grid-walk test (d), the adhesive removal test (e). (c) Serpina3n knock-out mice demonstrated reduced total assessment scores (tMCAO Serpina3n−/− vs tMCAO WT). (d–e) Serpina3n knock-out mice exhibited more profound sensorimotor deficits, including more extended sticker contact and removal time and more forelimb foot faults (tMCAO Serpina3n−/− vs tMCAO WT). All data are presented as means ± SD, n = 5 per group. *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001, ****P ≤ 0.0001, ns P > 0.05, two‐way ANOVA, and post hoc Tukey's tests.
We next examined the effect of Serpina3n knock-out on functional recovery after tMCAO using three different behavioral tests, including modified Garcia score, grid-walking test, and adhesive removal test. We found that the Garcia scores of Serpina3n−/− mice were significantly decreased (Figure 2(c)). In the adhesive removal test, the time to first touch the tape (time-to-contact) and the time removed the tape (time-to-remove) were significantly longer Serpina3n−/− mice (Figure 2(d)). In the grid walking test, the foot fault indexes (left forelimb foot fault errors/total amount of left forelimb steps – right impaired forelimb foot fault errors/total amount of right forelimb steps) were significantly lower in Serpina3n−/− mice (Figure 2(e)), suggesting that Serpina3n knock-out impairs post-stroke functional recovery.
Neuron-specific Serpina3n knock-down aggravates ischemia-induced BBB disruption and infiltration of T-cells and neutrophils following stroke
Ischemia-induced BBB disruption leads to extravasation of various peripheral immune cells that release proteinases that could compromise the integrity of BBB. 37 Since Serpina3n is a proteinase inhibitor that may counteract the effect of proteinases,35,38 we hypothesized that the endogenously secreted Serpina3n, especially Serpina3n secreted by neurons, could be beneficial to maintain BBB integrity after stroke. We carried out total or neuron-specific Serpina3n knock-down by infecting mice intravenously with Serpina3n shRNA AAV (shSerpina3n) or neuron-specific Serpina3n shRNA AAV (shSerpina3n (Neu)) three weeks before tMCAO. Efficient Serpina3n knock-down was confirmed by western blot and immunofluorescence at 3 days after tMCAO (Supplementary Fig. 4(A)-(D)). Both total and neuron-specific Serpina3n knock-down increased the infarct volume of the ischemic brain after stroke (Supplementary fig. 4(E)). We also found that ischemia-induced BBB leakage was significantly increased both in total and neuron-specific Serpina3n knock-down mice as shown by the increased permeability to Evans blue dye, IgG, and exogenous fibrinogen extravasation when compared with those in scramble shRNA-treated mice 3 days after tMCAO (Figure 3(a) and (b), Supplementary Fig. 5 (A), (B)). Ischemia stroke also led to a significantly downregulated expression of tight junction protein Zona occludens-1 (ZO-1), indicating a structurally disrupted BBB integrity. 39 Remarkably, a further decrease in ZO1 expression was observed in the infarct area of total and neuron-specific Serpina3n knock-down mice, as compared to that of scramble shRNA-treated mice (Figure 3(c), Supplementary Fig. 5(C)). Consistent with the notion that compromised BBB integrity can lead to exacerbated infiltration of peripheral immune cells after stroke, 40 we found the numbers of infiltrating T-cells and neutrophils were significantly increased in the total and neuron-specific Serpina3n shRNA-treated mice after stroke without changes of T-cells and neutrophils in periphery blood after stroke (Figure 3(d) to (g), Supplementary Fig. 5(D)-(G), Supplementary Fig. (6)). Thus, total and neuron-specific Serpina3n deficiency leads to exacerbated BBB disruption and increased the infiltration of T-cells and neutrophils into brain parenchymal after stroke.
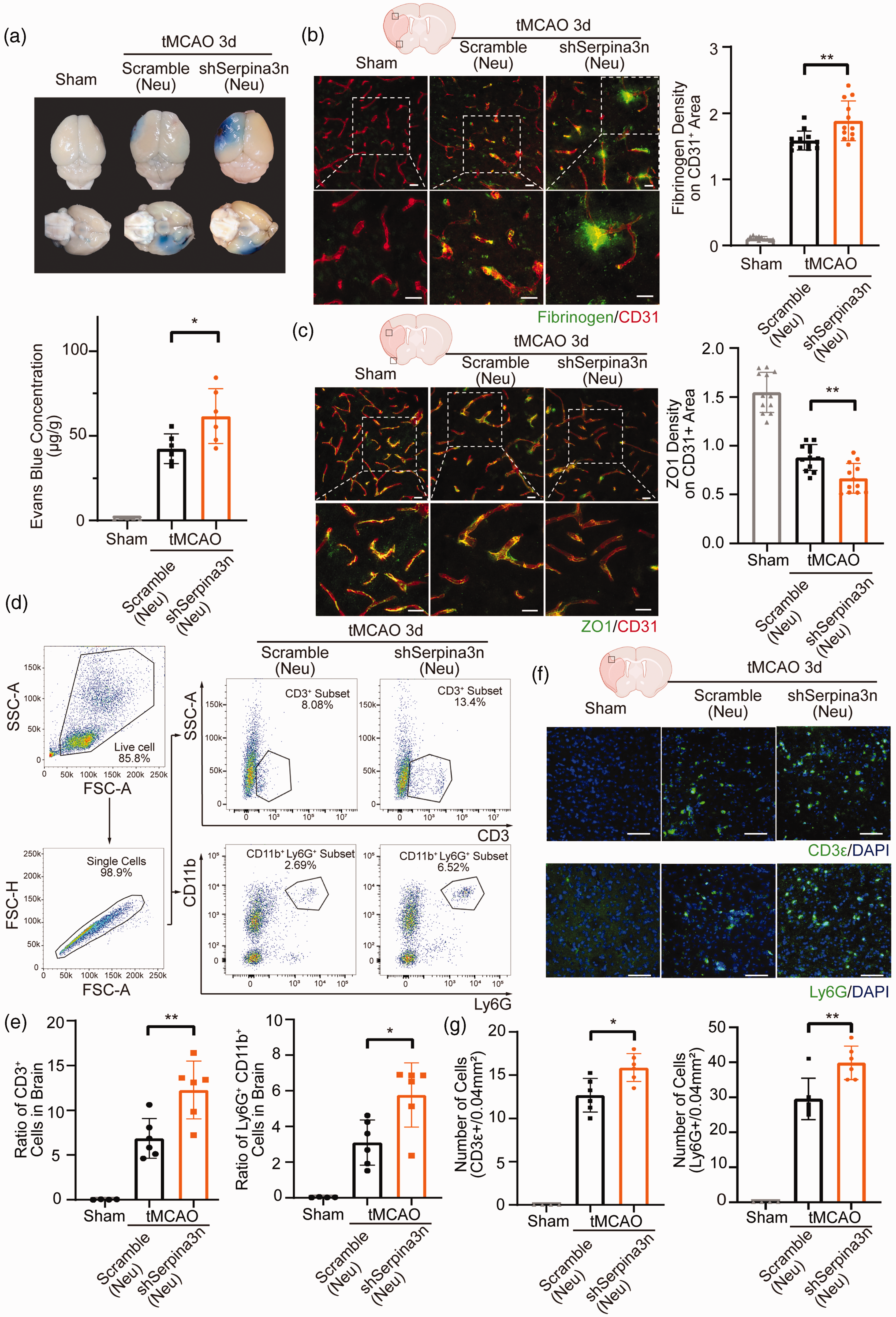
Neuron-specific Serpina3n knock-down aggravates ischemia-induced BBB disruption and infiltration of T-cells and neutrophils following stroke. (a) Representative photographs of Evans Blue staining and quantifications of Evans Blue concentration in the brain at 3d after sham operation or tMCAO with neuron-specific Serpina3n shRNA-AAV treatment. (b) Double-label immunofluorescence for CD31 and Fibrinogen in the infarcted regions at 3d after sham operation or tMCAO with neuron-specific Serpina3nshRNA-AAV treatment. Quantification of Fibrinogen extravasation in CD31+ areas. Scale bar, 50 µm. (c) Double-label immunofluorescence for CD31 and ZO1 in the infarcted regions at 3d after sham operation or tMCAO with neuron-specific Serpina3n shRNA-AAV treatment. Scale bar, 50 µm. (d) Representative dot plots and gating strategy for CD3+ cells and CD11b+ Ly6G+ Cells sorting followed by flow cytometry in mouse brain 3d after tMCAO with neuron-specific Serpina3n shRNA-AAV treatment. (e) Quantification of flow cytometry of CD3+ and CD11b+ Ly6G+ cells in the brain at 3d after tMCAO with neuron-specific Serpina3n shRNA-AAV treatment. (f) Representative photographs of CD3e and Ly6G immunostaining at 3d after tMCAO with neuron-specific Serpina3n shRNA-AAV treatment. Scale bar, 50 µm. (g) Quantification of CD3+ cells and Ly6G+ cells in the brain at 3d after sham operation or tMCAO with neuron-specific Serpina3n shRNA-AAV treatment. All data are presented as means ± SD, n = 6 per group ((b, c) 2 technical replicates from every biologically independent experiment per group). *P ≤ 0.05, **P ≤ 0.01, one‐way ANOVA, and post hoc Tukey's tests.
Serpina3n knock-down exacerbates neurologic deficits after stroke
To investigate the role of Serpina3n on the long-term stroke outcomes, we delivered total Serpina3n knock-down AAV (shSerpina3n) or control AAV(Scramble) intravenously 3 weeks before tMCAO and performed neurobehavioral tests in a randomized and blinded fashion, such as Garcia score, adhesive removal test and grid walking test to access the sensory-motor function and Y-maze spontaneous alternation test to access the cognitive function. Adhesive removal and grid walking tests were performed before tMCAO and 1 day after tMCAO to examine the neurological deficits (Supplementary Fig. 7).
We found that the Garcia scores of Serpina3n shRNA-treated mice were significantly decreased compared to those of the scramble shRNA-treated mice (Supplementary Fig. 7(A)). In the adhesive removal test, the time-to-contact and the time-to-remove of the Serpina3n shRNA-treated mice were significantly longer than those of the scramble shRNA-treated mice (Supplementary Fig. 7(B)). In the grid walking test, the foot fault indexes (left forelimb foot fault errors/total amount of left forelimb steps – right impaired forelimb foot fault errors/total amount of right forelimb steps) of the Serpina3n shRNA-treated mice were significantly lower than those of the scramble shRNA-treated mice (Supplementary Fig. 7(C)). As for Y-maze spontaneous alternation test, the alternation of the Serpina3n shRNA-treated mice was significantly reduced compared to those of the scramble shRNA-treated mice (Supplementary Fig. 7(D)), suggesting a better cognitive function of the Serpina3n shRNA-treated mice. These results suggest that Serpina3n knock-down resulted in worsened sensory-motor and cognitive function of post-stroke mice.
Intranasal Serpina3n delivery attenuates BBB disruption and immune cell infiltration following stroke
Intranasal delivery of proteins and peptides is a promising minimally invasive route for treating central nervous system disorders, such as stroke. 41 Since our data showed that Serpina3n deficiency resulted in augmentation of BBB disruption and infiltration of T cells and neutrophils into brain parenchymal, we explored the potential therapeutic effect of intranasal delivery of Serpina3n recombinant protein (rSerpina3n) in the tMCAO model. A preliminary dose-response experiment determined that 2.0 nmol/kg of rSerpina3n administrated on 3 successive days after tMCAO was the optimal regimen protecting against BBB leakage (Supplementary Fig. 8(A)); thus, this treatment regimen was used in all subsequent experiments. The protein level of Serpina3n in the treated mice brain was significantly increased compared to those control mice both in the sham and tMCAO groups (Supplementary Fig. 8(B)). We found that rSerpina3n treatment reduced infarct volume in the ischemic brain after stroke (Supplementary Fig. 8(G)). As compared to the control PBS-treated group, rSerpina3n-treated group exhibited significantly decreased BBB permeability to Evans blue dye, IgG, and fibrinogen extravasation at 3 days after tMCAO (Figure 4(a) and (b), Supplementary Fig. 8(G)). Significantly increased levels of ZO1 expression were also detected in the brain of rSerpina3n-treated mice compared to that of PBS-treated mice (Figure 4(c)). In addition, rSerpina3n treatment significantly reduced the number of infiltrating T-cells and neutrophils but did not affect the number of T-cells in cervical lymph nodes and periphery blood after tMCAO (Figure 4(d) to (g), Supplementary Fig. 8(C)-(F)). Collectively, these data suggested that intranasal treatment with rSerpina3n could attenuate BBB leakage after stroke.
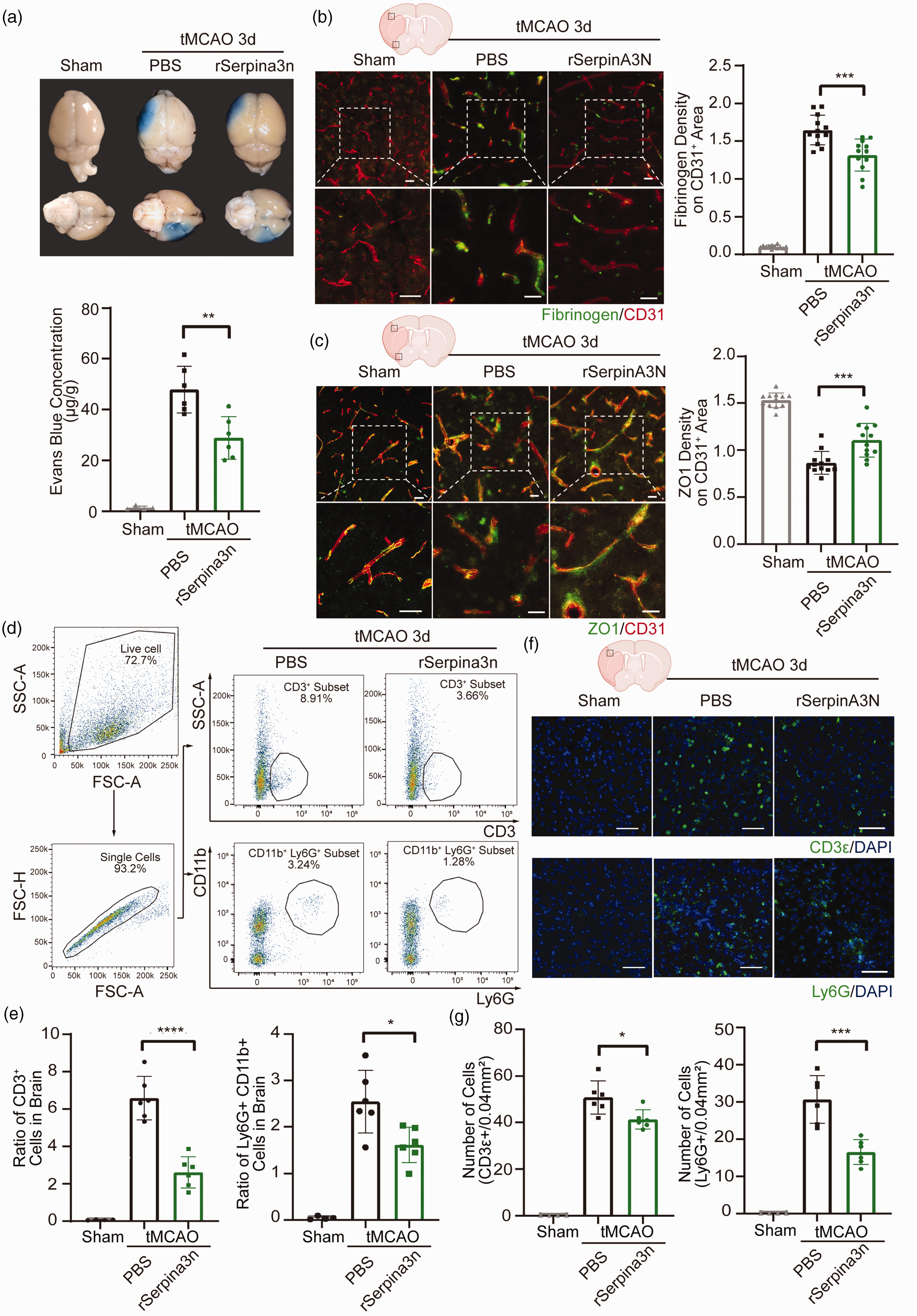
Intranasal Serpina3n delivery attenuates BBB disruption and immune cell infiltration following stroke. (a) Representative photographs and quantification of Evans blue staining in the brain at 3d after tMCAO or sham operation with rSerpina3n or PBStreatment. (b) Double-label immunofluorescent staining for CD31 and Fibrinogen in the infarcted regions and quantification of Fibrinogen extravasation in CD31+ areas at 3d after sham operation or tMCAO with the treatment of rSerpina3n or PBS. Scale bar: 50 mm. (c) Double-label immunofluorescence for CD31 and ZO1 in the infarcted regions and quantification of ZO1 expression on CD31+ area at 3d after sham operation or tMCAO with the treatment of rSerpina3n or PBS. Scale bar, 50 µm. (d) Representative dot plots and gating strategy for CD3+ cells and CD11b+ Ly6G+ Cells sorting followed by flow cytometry in mouse brain 3d after tMCAO with the treatment of rSerpina3n or PBS. (e) Quantification of flow cytometry of CD3+ and CD11b+ Ly6G+ cells in the brain at 3d after tMCAO with rSerpina3n or PBS treatment. (f) Representative photographs of CD3e and Ly6G immunostaining at 3d after tMCAO with rSerpina3n or PBS treatment. Scale bar, 50 µm. (g) Quantification of CD3+ cells and Ly6G+ cells in the brain at 3d after sham operation or tMCAO with rSerpina3n or PBS treatment. All data are presented as means ± SD, n = 6 per group ((b, c) 2 technical replicates from every biologically independent experiment per group). *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001, ****P ≤ 0.0001, one‐way ANOVA, and post hoc Tukey's tests.
Serpina3n attenuates ischemia-induced BBB disruption by inhibiting granzyme B and neutrophil elastase
Peripheral immune cells are critical regulators of the BBB integrity in many diseases, in which the infiltrating neutrophils, T-cells, and macrophages compromise BBB through secreting serine proteinases.42–44 We next explored the role of secretory serine proteinases in Serpina3n-afforded protection against BBB damage after ischemic stroke. We found that shRNA-AAV-mediated Serpina3n neuron-specific knock-down significantly increased the activities of GZMB and NE in ischemic brain compared to the scrambled shRNA-treated mice. In contrast, rSerpina3n-treated mice had decreased GZMB and NE activities compared to those PBS-treated control mice (Figure 5(a) and (b)). In addition, SerpinA3N knock-down lead to a high GZMB and NE protein level and rSerpina3n treatment decreased the GZMB and NE protein level (Supplementary Fig. 9(A), (B)).
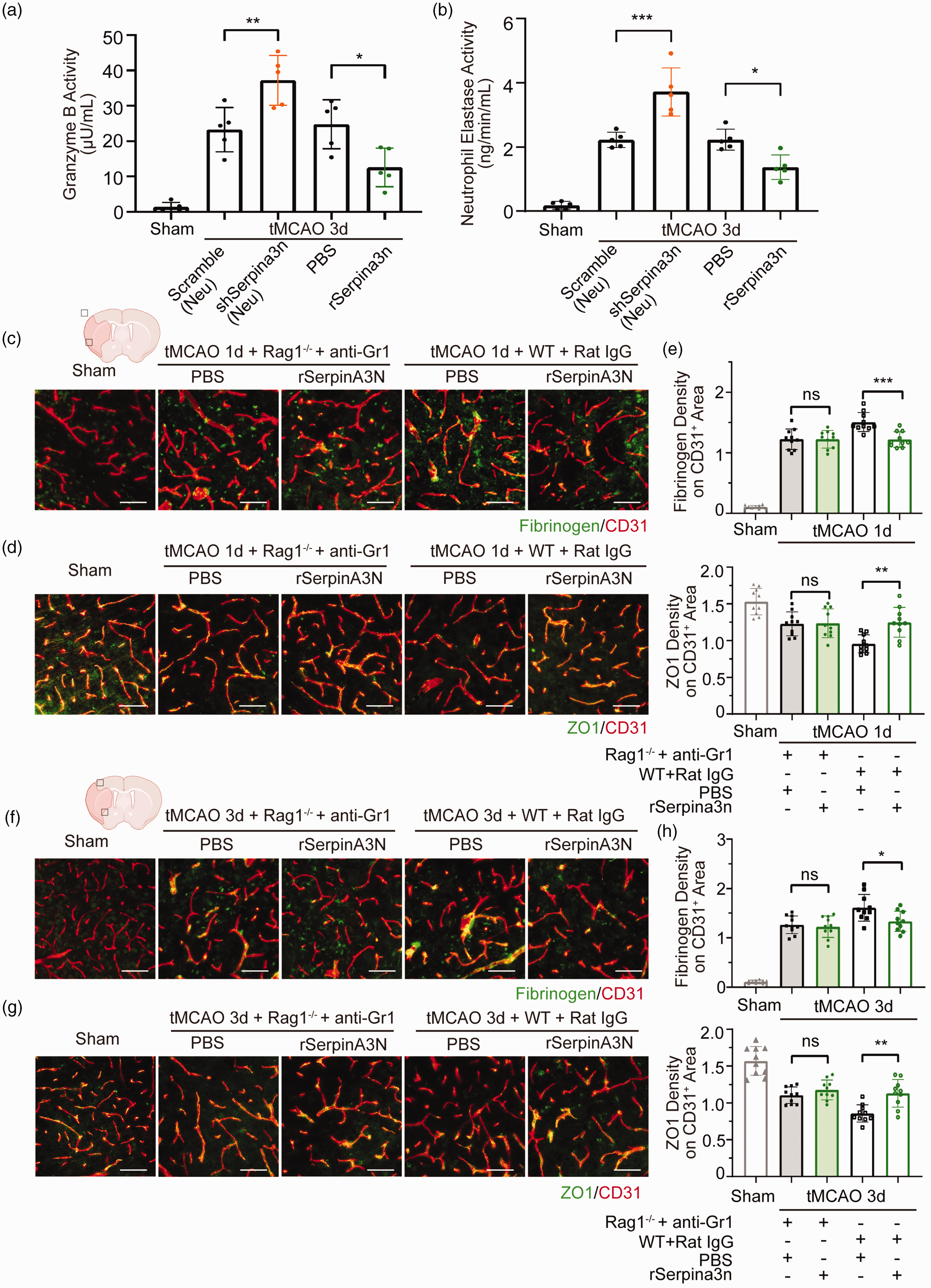
Serpina3n protects against BBB disruption by inhibiting granzyme B and Neutrophil Elastase after cerebral ischemia. (a, b) GZMB and NE activity in the brain parenchyma of neuron-specific Serpina3n shRNA-AAV and rSerpina3n treatment mice after tMCAO or sham operation. Data are presented as means ± SD, n = 5 per group. *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001, one‐way ANOVA, and post hoc Tukey's tests. (c–h) Double-label immunofluorescent staining for CD31 and Fibrinogen in the infarcted regions and quantification of Fibrinogen extravasation in CD31+ areas (top of e, h) of Gr1 antibody treatment Rag1−/− and rat IgG treatment WT mice with rSerpina3n or PBS treatment at 1 day (c) and 3day (f) after tMCAO or sham operation. Double-label immunofluorescence for CD31 and ZO1 in the infarcted regions and quantification of ZO1 expression on CD31+ area (bottom of e, h) of Gr1 antibody treatment Rag1−/− and rat IgG treatment WT mice with rSerpina3n or PBS treatment at 1 day (d) and 3day (g) after tMCAO or sham operation. Scale bar, 50 µm. Data are presented as means ± SD, n = 5 per group ((e, h) 2 technical replicates from every biologically independent experiment per group). *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001, ns P > 0.05, one‐way ANOVA, and post hoc Tukey's tests.
To test the hypothesis that rSerpina3n-afforded protection was via the suppressed infiltration of peripheral immune cells, both T-cells (Rag1−/− mice) and neutrophils (Gr1 antibody treatment, Supplementary Fig. 9(C)) were depleted in rSerpina3n intranasally administered mouse 1 day or 3 days after tMCAO. We found that the effect of rSerpina3n treatment on BBB damage was abolished (Figure 5(c) to (h)). However, when rSerpina3n was administered intranasally into neutrophil-depleted mice or T-cell-deficient Rag1−/− mice 1 day or 3 days after tMCAO, the protective effect of rSerpina3n treatment on BBB damage could still be observed (Supplementary Fig. 10, 11), suggesting that the protection of Serpina3n on BBB integrity was dependent both on neutrophils and T-cells. In conclusion, these results thus support our hypothesis that Serpina3n protects against BBB damage after cerebral ischemia by suppressing the infiltration of T cells and neutrophils into the brain.
Serpina3n intranasal treatment releases ischemic-stroke neurologic deficits
To investigate the effect of rSerpina3n treatment on long-term stroke outcomes, mice were treated with rSerpina3n intranasally for 3 days after tMCAO, and Garcia score, adhesive removal test, grid walking test, and Y-maze spontaneous alternation test were examined for 4 weeks after tMCAO (Figure 6). We found that Garcia scores of the rSerpina3n treatment group were higher than the PBS control group (Figure 6(a)). Time-to-contact and time-to-remove of the rSerpina3n treatment group were shorter compared with the PBS control group (Figure 6(b)). In the grid walking test, foot fault indexes of rSerpina3n treatment group mice were higher than PBS control group (Figure 6(c)). In the Y-maze spontaneous alternation test, the alternation of the rSerpina3n treatment group increased compared with the PBS control group (Figure 6(d)). In conclusion, rSerpina3n intranasal treatment improved functional recovery after stroke.
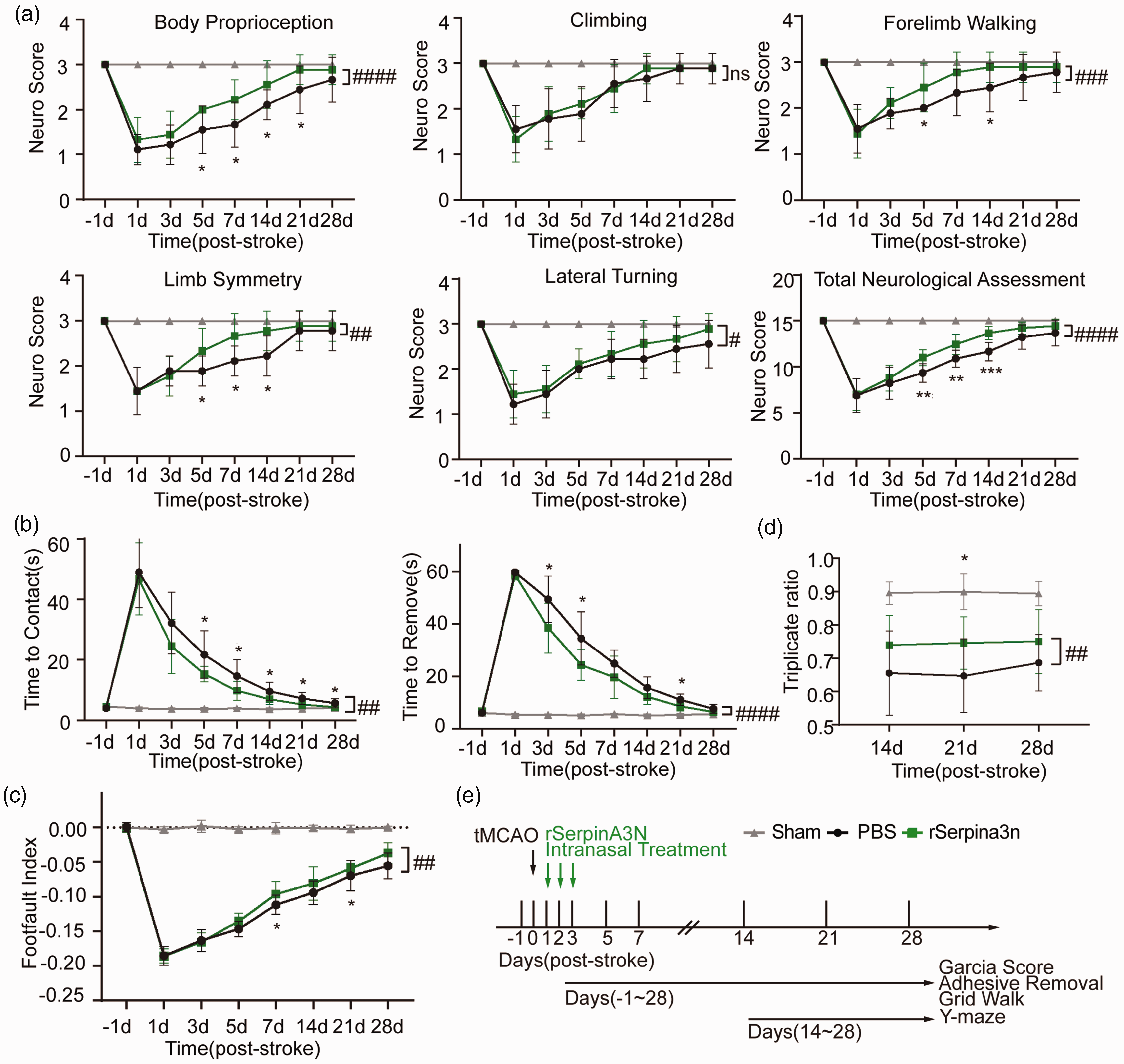
Serpina3n intranasal treatment improves long-term functional outcomes after cerebral ischemia. (a–d) Sensorimotor and cognitive functions were significantly improved in rSerpina3n‐treated mice after tMCAO compared to PBS-treated mice, as assessed by the Garcia Score (a), the adhesive removal test (b), the grid-walk test (c), and the Y-maze test (d). (a) rSerpina3n‐treated mice demonstrated improved body proprioception, lateral turning, and total assessment scores. (b–c) rSerpina3n‐treated mice exhibited less profound sensorimotor deficits, including reduced sticker contact and removal time and fewer forelimb foot faults. (d) rSerpina3n-treated stroke mice exhibited milder cognitive deficits than PBS-treated mice, with higher triplicate ratios. All data are presented as means ± SD, n = 9 per group. *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001 (PBS vs rSerpina3n at 5d, 7d, 14d, 21d, 28d), #P ≤ 0.05, ## P ≤ 0.01, ###P ≤ 0.001, #### P ≤ 0.0001, ns P > 0.05 (PBS vs rSerpina3n), two‐way ANOVA, and post hoc Tukey's tests. (e) Schematic illustration of the experimental design.
Discussion
This study demonstrated that Serpina3n, a serine protease inhibitor mainly expressed in neurons, protects against BBB damage and promotes long-term neurological recovery after stroke. Mechanistically, Serpina3n prevents BBB disruption by suppressing the infiltration of T cells and neutrophils and the detrimental effects of serine proteases. Thus, this study reveals a novel endogenous protective mechanism against stroke-induced BBB damage. This was confirmed by findings showing that shRNA AAV-mediated Serpina3n knockdown worsened BBB damage after cerebral ischemia while intranasal delivery of rSerpina3n attenuated it.
As one of the early pathological processes after stroke, the loss of BBB integrity can facilitate the infiltration of blood-borne cells, macromolecules, and fluids into the brain parenchyma,45,46 augmenting neuroinflammation and apoptotic cell death. 47 The infiltrating neutrophils and T cells further exacerbate BBB damage and leakage after stroke,9,11–13 perhaps by releasing the BBB-damaging proteases such as MMP9 and MMP2.48,49 Previous studies suggest that Serpina3n confers beneficial effects on abdominal aortic aneurysms, diabetic wound healing, and other diseases by inhibiting serine proteases secreted by T cells and neutrophils.35,50–52 The data presented here demonstrated that Serpina3n-afforded protective effects against ischemic BBB damage are associated with decreases in intracerebral infiltration of T cells and neutrophils and production of GZMB and NE. We also found that BBB disruption was reduced in Rag1−/− mice and Gr1 antibody-treated mice after tMCAO, which is consistent with the previously reported neuroprotective effects of depleting T cells or neutrophils.12,53 The reduction or lack of beneficial effects on BBB damage by Serpina3n in the absence of neutrophils or/and T cells further supports a causative role for the decreased infiltration of peripheral immune cells in Serpina3n-afforded BBB protection. However, our data do not prove that the decreased production of GZMB and NE is directly involved in Serpina3n-afforded protection against BBB damage, as the infiltrating neutrophils or T cells may release other BBB-damaging proteases as well.
Previous studies showed that Serpina3n expression is upregulated in astrocytes after stroke and in hippocampal neurons after hippocampal stab injury, respectively.16,54 In the current study, we found that Serpina3n expression is induced in a wide range of cell types, including neurons, astrocytes, microglia, and OPCs in the ischemic brain regions, indicating that the protective effects of Serpina3n against ischemic brain injury may be multifaceted. The neuron expressed Serpina3n may be directly responsible for its anti-neuronal apoptosis effects, as suggested by studies using animal models of multiple sclerosis 51 or the hippocampal stab injury. 54 The astrocyte- and microglia-enriched expression of Serpina3n may exert anti-inflammatory effects, which, subsequently, might have contributed to BBB protection after cerebral ischemia, as both astrocytes and microglia actively regulate BBB permeability after ischemic brain injury through the release of pro-inflammatory cytokines and chemokines.55–57 In the current study, we also found increased expression of Serpina3n in OPC and oligodendrocytes (OL) in the post-ischemic brain. The functional impact of Serpina3n in OPCs/OLs is unknown; however, since OPCs and OLs are essential for the myelination and remyelination processes in the brain, 58 it’s possible that Serpina3n could contribute to post-stroke white matter repair. Thus, future studies are warranted to determine whether Serpina3n is also critically involved in brain repair and functional recovery after stroke.
There are several limitations to this study. First, we achieved neuron-specific Serpina3n knock-down using shRNA-AAV with the hSyn promotor. It will be important in future studies to confirm the neuron cell-type specific mechanisms underlying the beneficial impact of Serpina3n on ischemic brain injury with conditional knock-out mice. Secondly, we speculated that Serpina3n reduces BBB damage by inhibiting the serine proteases GZMB and NE that are presumably produced by neutrophils and T cells. However, we have not directly tested this hypothesis. GZMB can also be produced and released by NK cells. 59 Moreover, neutrophils and T cells are known to release other proteases, such as MMP9 and MMP2, that contribute to BBB damage and dysfunction.48,49 Employing specific knock-out mice for GZMB or NE will help address this scientific question. Third, neurological deficits after stroke are evaluated in different sensorimotor and cognitive tests, but some differences are relatively small. Nevertheless, we paid attention to designing our test battery to contain moderately sensitive, easy-to-perform standard tests (e.g., Garcia score, adhesive removal test) with very specific and sensitive readouts such as the foot fault analysis and the Y-maze for cognitive function. 60 Finally, the current study only tested male mice. However, remarkable sex differences exist for brain physiology and stroke pathophysiology, and response to therapeutic treatment.61–63 Downstream research will need to investigate Serpina3n effects in mixed-sex study cohorts. Finally, to increase the translational potential of Serpina3n as a therapeutic target for stroke, comorbidities, such as aging, should be studied. We have, however, investigated the effects of Serpina3n in an ischemia-reperfusion paradigm and started to evaluate the effects of different cerebral cell populations what is in good alignment with current recommendations in the field.18,64
In summary, our data demonstrate that Serpina3n protects against BBB damage after stroke by inhibiting the intracerebral infiltration of neutrophils and T cells and their release of GZMB and NE while post-stroke intranasal delivery of rSerpina3n improves long-term neurological outcome.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221113897 - Supplemental material for Neuronal Serpina3n is an endogenous protector against blood brain barrier damage following cerebral ischemic stroke
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221113897 for Neuronal Serpina3n is an endogenous protector against blood brain barrier damage following cerebral ischemic stroke by Fengshi Li, Yueman Zhang, Ruqi Li, Yan Li, Shenghao Ding, Jianpo Zhou, Tianchen Huang, Chen Chen, Bingwei Lu, Weifeng Yu, Johannes Boltze, Peiying Li, Jieqing Wan in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: P.L. is supported by the National Natural Science Foundation of China (NSFC, 81722017, 91957111, 81971096, 82061130224), New Frontier Technology Joint Research sponsored by Shanghai Shenkang Hospital Development Center (SHDC12019102), Shanghai Municipal Education Commission-Gaofeng Clinical Medical Grant Support (20181805), “Shuguang Program” supported by Shanghai Education Development Foundation and Shanghai Municipal Education Commission (20SG17), and Shanghai Outstanding Academic Leaders’ Program from Science and Technology Commission of Shanghai Municipality (20XD1422400), Newton Advanced Fellowship grant provided by the UK Academy of Medical Sciences (NAF\R11\1010; together with J.B.), Innovation Program of Shanghai Municipal Education Commission (2019-01-07-00-01-E00074 to Weifeng Yu); J.W. is supported by the Shanghai Jiao Tong University (DLY201821, ZH2018ZDA07), and Science and Technology Commission of Shanghai Municipality (17411954000).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
FL, YZ, RL, TH, and CC performed the experiments. FL, YZ, SD, JZ, and YL collected the data and performed the analysis. FL and PL wrote the manuscript. FL, BL, WY, JB, and PL designed the experiments. PL and JW supervised the project. PL, JW, and JB edited the final manuscript. All authors agreed on the final version.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
