Abstract
Brain oscillations recorded in the extracellular space are among the most important aspects of neurophysiology data reflecting the activity and function of neurons in a population or a network. The signal strength and patterns of brain oscillations can be powerful biomarkers used for disease detection and prediction of the recovery of function. Electrophysiological signals can also serve as an index for many cutting-edge technologies aiming to interface between the nervous system and neuroprosthetic devices and to monitor the efficacy of boosting neural activity. In this review, we provided an overview of the basic knowledge regarding local field potential, electro- or magneto- encephalography signals, and their biological relevance, followed by a summary of the findings reported in various clinical and experimental stroke studies. We reviewed evidence of stroke-induced changes in hippocampal oscillations and disruption of communication between brain networks as potential mechanisms underlying post-stroke cognitive dysfunction. We also discussed the promise of brain stimulation in promoting post stroke functional recovery via restoring neural activity and enhancing brain plasticity.
Introduction
Local field potential (LFP), commonly referred to as electroencephalography (EEG) in clinical neuroscience, records synaptic potential generated by groups of excitatory and inhibitory neurons as oscillating waves within the range of detection by the electrodes. Powered by evolving data science in decoding and pattern recognition, electrophysiology has provided crucial information in improving our understanding of normal brain function as well as altered function due to injury or diseases. The frequencies of these brain oscillations include the physiological range of <1 Hz to 250 Hz and can extend to a higher frequency range under pathological conditions such as seizure activity. Although the origin of brain oscillations can be widespread, it is generally believed that each wave serves a specific function among certain brain structures and networks. Brain oscillations exhibit complex patterns of interaction and regulation within brain structures, as well as between them, binding networks together to achieve optimal functionality. Brain injuries and diseases often affect this tightly regulated coordination disproportionately, resulting in impaired brain function.
The generation and maintenance of a membrane potential involves the consumption of ATP; thus, cerebral blood flow (CBF) has a considerable effect on brain oscillations, with those in the higher frequency range being particularly prone to the immediate ramifications of energy deprivation. Assessing brain oscillation changes as biomarkers of disease progression, prognosis, and neurodegeneration in brain ischemia is especially valuable in brain health science. Increased signal power in low-frequency oscillations coincided with the reduction of high-frequency oscillations that were often observed among patients and laboratory animals following ischemic stroke (Examples of EEG traces and topographical maps are shown in Figure 1). Deafferentation of subcortical structures such as the thalamus following cortical lesion can also lead to propagation of low-frequency oscillations. Despite ample data from earlier studies in clinical strokes, EEGs no longer play a significant role in stroke detection or diagnosis due to the development of better-suited neuroimaging modalities. Magnetoencephalography (MEG),4,5 which measures a brain’s magnetic fields generated by electric currents, has a higher spatial resolution and deeper brain region accessibility has emerged as a more fitting electrophysiological tool in predicting stroke prognosis and functional recovery. In addition, an Electrocorticography (ECoG) with flexible microelectrode arrays records within the subdural space, can reach very high spatial resolution, and has a reduced noise level compared to EEG.6,7 Unlike ECoG, MEG is non-invasive and often used to assess novel stroke rehabilitation strategies nowadays.
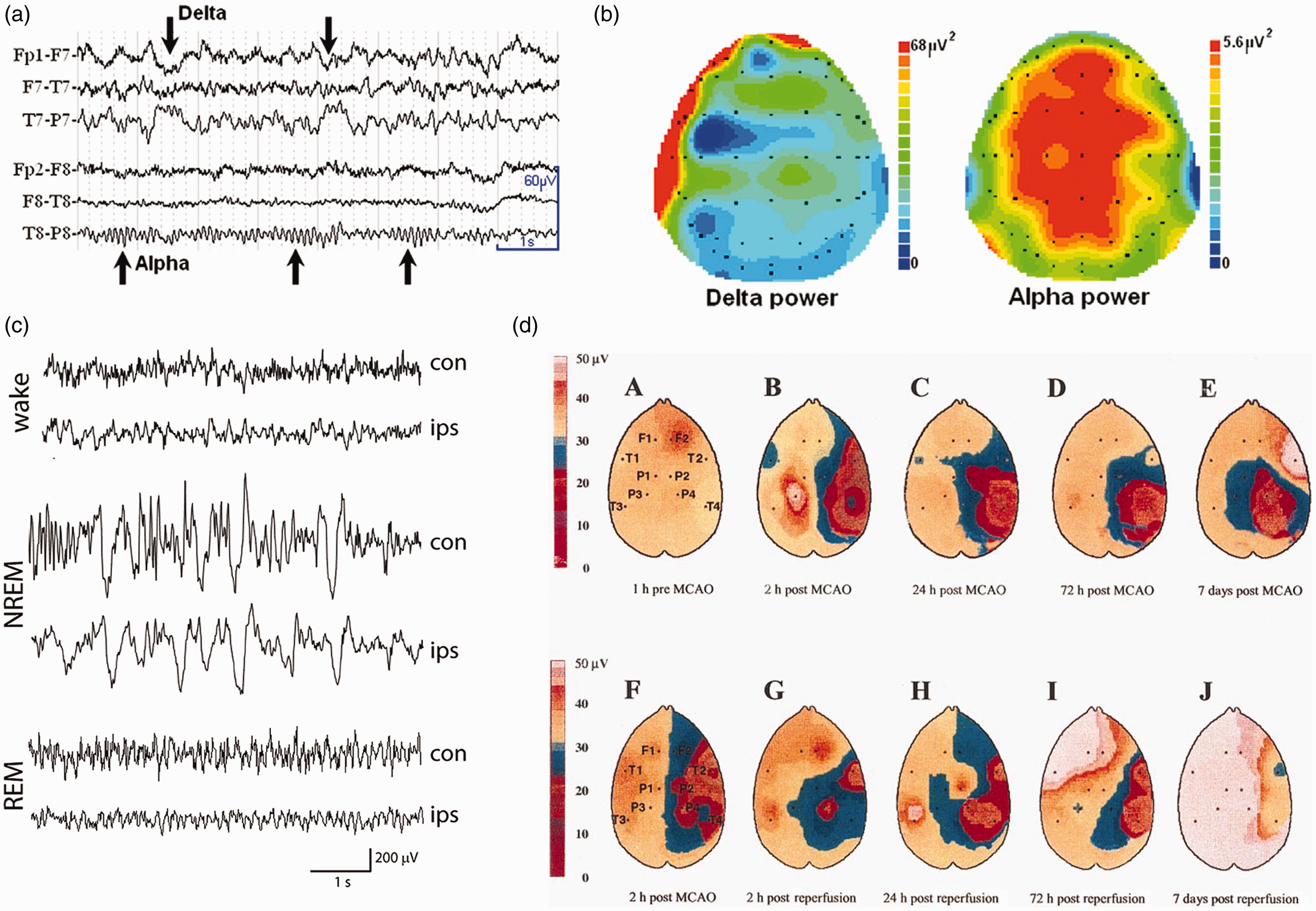
Examples of EEG traces and topographical maps from human and rat stroke. (a) The presence of delta wave in raw EEG traces recorded from a patient with left middle cerebral artery stroke (MCA) 7 hours after the onset of symptoms (NIHSS = 7). (b) Topographical maps showing the distribution of mean delta and alpha power across the scalp, with increased delta power in the frontal and temporal channels (adopted from figure 1, Finnigan and van Putten 1 ) (c) Representative EEG traces of wake, rapid eye movement (REM) and nonrapid eye movement (NREM) sleep in ipsilesional (ips) and contralesional (con) hemisphere recorded 4 days after MCAO in a rat with pre-implanted epidural electrodes (adopted from figure 3, Leemburg S et al., 2018 2 ) (d) Representative topographic EEG brain mappings of the amplitude of the global band selected at 5 time points from one rat with permanent MCA occlusion (a–e) and another with 2-hour temporary MCA occlusion (tMCAO) (f–j). (adopted from figure 9, Lu X et al., 2001 3 ).
Contrary to the non-invasive nature of EEG, penetrating electrodes used in experimental studies enabled the investigation of deeper brain structures remote from the ischemic infarct. Among those structures is the hippocampus, which is crucial for memory function but is often spared from ischemic injury caused by Middle Cerebral Artery Occlusion (MCAO). Sharp wave associated ripples (SPW-R), a hippocampus-specific oscillation, is crucial in memory consolidation, relying on an intricate balance between excitatory and inhibitory networks and are vulnerable to the disruption caused by ischemia. In this review, we will start by providing an overview on the basics of LFP/EEG signals and their biological relevance, followed by a summary of findings reported in clinical and experimental strokes. We will discuss recent findings from experimental stroke studies highlighting how ischemic injury alters hippocampal brain oscillation and communication between brain networks. Lastly, we will discuss emerging technologies such as Brain Machine Interface (BMI) and brain stimulation and their potential in promoting functional recovery post stroke.
Brain oscillations detected by extracellular recordings
Major brain oscillation and their biological function in humans and animals
Most human extracellular electrophysiological data were initially derived from conventional EEG recordings of electrical activity in the superficial layers of the brain using non-invasive scalp macro-electrodes, 8 as opposed to the local field potential recordings from high impedance micro-electrodes placed in the deeper brain locations that have been used for experimental studies. Over time, non-invasive recording technology has evolved to high-density scalp EEGs, 9 high spatiotemporal resolution Magnetoencephalography (MEG), which is less influenced by the conductivity of the extracellular space, and high spatial resolution low noise Electrocorticography’s (ECoG) that records activity in the subdural space bypassing the skull and in-between tissue that distort the signal. Commonly described oscillations in mammalian brains range from 0.05 to 250 Hz in frequency, including the best-known waves δ (1–4 Hz), θ (4–8 Hz), α (8–12 Hz), β (12–30 Hz) and γ (30–80 Hz). Examples of lesser-known oscillations include slow waves (0.3–1 Hz) that are slower than the δ band, and high frequency oscillations (HFO) (80–250 Hz) that are faster than the γ band. 10 Among the fast oscillations, SPW-Rs (150–250 Hz) are a type of hippocampal oscillation crucial for memory consolidation, 11 while pathological High Frequency Oscillations (pHFOs) (200–600 Hz) are often recorded in the dentate gyrus during seizure generation and are distinct from physiological SPW-Rs. 11 Compared to the penetrating electrodes used in animal studies, the non-invasive human scalp EEG detection is limited in frequency range and prone to artifacts and noise.
A consensus regarding the anatomical regions where brain oscillations originate in mammalian brains has not been established due to inconsistent findings. Specific brain regions or networks may serve as the generator for some oscillatory frequencies, while others act as the resonators that respond to certain firing frequencies. 12 The two main oscillation generators of the non-REM sleep-associated slow-wave were reportedly located in the pyramidal neurons of the layers II/III, V, and VI of the neocortices, and in the Nucleus Reticularis Thalami (NRT) neurons of the thalamus. Corticothalamic, thalamocortical, and intracortical connections are all involved in synchronizing activities between the 2 generators. 13 Similarly, the δ wave is generated by the thalamus and pyramidal cells located in layers II–VI of the cortices, whereas higher frequency oscillations like α or β are believed to be generated by the cells in layers IV and V of the cortex.14,15 The hippocampus is known to resonate θ oscillation that propagates through the septo-temporal axis via the volume conduction, since inactivation or lesion of the septum perturbs the hippocampal θ oscillations. 16 However, discrepant studies have reported the hippocampal and thalamic origins of the θ and α waves, respectively. In contrast, the γ rhythm is widely spread among many brain structures including the cortex, midbrain and the hippocampus.17–19 Low-frequency waves such as θ and δ propagate through the entire brain as directional waves, whereas high-frequency ones like α, β, and γ tend to stay localized to small structures and are driven by θ and δ.20,21
The functional role of each brain oscillation is complex, spanning from sensory motor to cognitive domains. Contributing to a dominant brain state, the θ rhythm is involved in movement and cognitive activity during the awake state, as well as memory consolidation during non-REM sleep.22,23 Conversely, the hippocampal non-θ state, also known as the slow-wave state (SWS), is associated with immobility or SWS sleep.24,25 Manipulating the hippocampal θ rhythm alters cognition, further suggesting a causal role of the hippocampal θ in cognition. 26 Cortical γ oscillations, including low (30-60Hz) and high γ oscillations (60-150Hz), are known to play a role in sensory stimulation, declarative memory, attention selection, cognition, and assist in encoding and retrieval of memory traces in the hippocampal formation.27–29 SPW-R, another novel and highly significant brain oscillation involved in cognition and memory, is discussed below. However, there are limitations in translating findings from animal studies to human physiology with respect to age effect, the origin of brain oscillations and how they function across networks due to the species difference in brain structure and organization.
Sharp wave associated ripples (SPW-R)
Experimental studies in recent decades have revealed a type of hippocampal oscillation with a frequency of 150-250 Hz that has not been described in human scalp EEG studies, known as the SPW-R. Detected in the CA1 pyramidal layer of the hippocampus, SPW-R s are concurrent with the firing of sharp waves in the stratum radiatum, as the name implies. By now, ample experimental and human data have established that SPW-Rs play a crucial role in the formation and subsequent stabilization of enduring memories at the cortical level during system-level memory consolidation. 11 It has been reported the rate of SPW-Rs increase in the hippocampus of rats after learning a new task such as an odor-learning association or radial maze, 30 and their sequential activity is a replay of firing sequences of the same neurons activated during learning and exploration.31–36 Conversely, the selective elimination of SPW-Rs via timed electrode stimulation during post-learning sleep resulted in the impairment of long-term memory.34–37 Using intracranial recordings in human subjects engaging in episodic free recall of previously viewed pictures, a recent study found that content-selective increase in hippocampal SPW-R firing emerged 1–2 seconds before a recall event. SPW-R rate during encoding could also predict subsequent free recall accuracy, indicating that human memory reactivation during the free recall was coupled to hippocampal SPW-R events. 38
SPW-Rs belong to a complex oscillatory network, the coupling of which coordinates specific information transfer among brain regions during memory consolidation. Combining neurophysiology recordings and fMRI techniques, a seminal study revealed the global effect of SPW-R in the brain. It was found that most of the cortical regions are activated during SPW-R, in contrast to the diencephalic, midbrain and brainstem regions are selectively inhibited. 39 This suggests that silencing the output of subcortical areas involved in sensory processing may minimize interference from regions like the thalamus and provide a privileged interaction state between hippocampus and cortex during memory consolidation. A later study confirmed the activity in the mediodorsal thalamic nucleus was suppressed during SPW-R firing in the awake state and around spindle-uncoupled SPW-R but was increased around spindle-coupled SPW-R during non-REM sleep. 40 This study confirmed that blocking thalamocortical relay of sensory information flow facilitates memory consolidation in “off-line” learning during spindle-uncoupled SPW-R. This in turn promotes hippocampo-cortical communication because mPFC is more responsive to hippocampal input during spindle-uncoupled SPW-R. The interaction between hippocampal SPW-R s and cortical slow waves appears to be bidirectional during NREM sleep. 41 In addition to the mediodorsal thalamic nucleus, other midline thalamic neurons such as the calretinin-positive neurons in the reuniens nucleus can also mediate hippocampus and mPFC interaction via SPW-R. 42
Locally, both excitatory and inhibitory neurons are needed for proper SPW-R firing. Although the excitability of the pyramidal cells plays a key role in the generation of high frequency oscillatory events in the hippocampus, GABAergic activity is also crucially involved in maintaining SPW-R including GABAAR mediated signaling. 43 Direct evidence in support of the involvement of GABA in SPW-R regulation came from pharmacological studies in which focal pharmacological blockades of GABAA receptors abolished SPW-R, 44 while an antagonist of a GABAAR5 enhanced the power and duration of the SPW-R.45,46 Specific targeting of GABA signaling via transgenic technology also affects ripples. By genetically reducing AMPA receptors in parvalbumin GABAergic interneurons in mutant mice, GABA-mediated tonic inhibition was altered and ripple oscillation was augmented. 47 Disturbance of physiological ripple oscillation has been reported after experimental stroke during acute and chronic phases.48,49 Apart from ischemic stroke, changes in SPW-Rs are also observed in murine neurodegenerative disease models. For example, diminished ripple occurrence rates were found in aged mice with human Apolipoprotein E4 knocked in, 50 which led to the loss of hilar GABAergic interneurons, 51 while rTg4510 transgenic mice, a model of tauopathy and dementia, had reduced inhibitory control of the hippocampal network and smaller amplitude-SPW-R. 52 Therefore, a well-orchestrated action between excitation and inhibition is required for the normal physiological function of SPW-R since synchronous activation of excitatory pyramidal cells and certain classes of inhibitory GABAergic interneurons are necessary for SPW-R generation.43,53
It has recently been shown that prolonging SPW-R via optogenetic stimulation of the CA1 neurons improves memory performance. 54 SPW-R can also be enhanced pharmacologically, GABA inhibitor L655,708 reportedly enhances the power and duration of the SPW-R in rats, 46 and increases the SPW-R cluster in CA1, 45 although the efficacy of L655,708 in restoring SPW-R in disease or neurodegeneration has not been established.
Complex interactions among brain waves
Most of the oscillations also show complex patterns of interaction with other waveforms or spike activities from other brain regions. Interaction between oscillations of different frequencies can occur via phase-amplitude coupling, resulting in synchronization of the amplitude envelope of faster rhythms within the phase of slower rhythms. The best-known example of cross-frequency phase coupling is the θ modulation of γ amplitude that is known to play a critical role in cognition. During coupling, θ oscillations provide a temporal reference for the exchange of information among different brain networks, whereas faster γ-frequency oscillations nested within θ cycles underlie local information processing in the cortex.55,56 Furthermore, the extent of cortical neurons modulated by θ rhythm depends on neuronal location and subtype. A greater proportion of interneurons, for example, 32% in the parietal cortex and 46% in the prefrontal cortex, are modulated by θ waves compared to that in pyramidal neurons (11% in the parietal cortex and 28% in the prefrontal cortex). 57 Breakdown of the phase-amplitude coupling disrupts communication between neural networks and causes disturbance of brain physiology or diseases. For example, the hippocampal θ-γ coupling is stronger during memory replay, while poor coupling predicts poor memory performance.58,59 Similarly, reduced modulation of cortical γ amplitude during hippocampal θ phase was reported in a mouse model for Alzheimer’s Disease with cognitive deficit, 60 and in schizophrenic patients associated with working memory dysfunction. 61 A review of methods for EEG/LFP signal processing and data analysis can be found in supplemental material.
EEG changes after cerebral ischemia
Effect of cerebral blood flow on EEG
The extracellular signal recorded in an EEG is the net summation of the electric current flow of charges in and out of the neurons, known as the current sink and source, respectively. Once activated, neurons release neurotransmitters into the synaptic cleft that either excite/depolarize or inhibit/hyperpolarize the adjacent postsynaptic neurons. The Excitatory Postsynaptic Potential (EPSP) depolarizes the postsynaptic neurons due to the release of excitatory neurotransmitters such as glutamate and acetylcholine, while Inhibitory Postsynaptic Potential (IPSP) hyperpolarizes neurons due to the release of inhibitory neurotransmitters such as γ-amino butyric acid (GABA) and glycine. Consequently, the summation of the IPSP and EPSP induces a graded potential in the neuron so that when the membrane potential reaches the threshold, it induces an action potential that can be propagated between neurons. EEG detects field potential as IPSP or EPSP generated by neurons because those events are longer in duration than the action potential (up to 10 milliseconds vs. a few milliseconds). An EEG trace reflects the interaction of signals of a group of excitatory and inhibitory neurons within the detection range of the electrodes, in which the EEG waves oscillate with alternating rises and falls in amplitude. 10
The maintenance of ionic gradients and membrane potential consumes energy, thus the reduction of cerebral blood flow (CBF) as a result of brain ischemia affects brain oscillations with differential sensitivity, in which the signal power of high frequency waves are first to be decreased at the early stage of hemodynamic failure. 10 Besides CBF reduction following ischemia, the concurrent increase of local glutamate release is also known to exacerbate the decrease of EEG power. 62 At the network level, attenuation of the excitatory drive onto the thalamus or enhanced subcortical inhibition from cortical lesions are known to cause thalamo-cortical dysrhythmia, often resulting in the propagation of low frequency oscillations (in the δ and θ range).63,64 These stroke-induced thalamo-cortical pathological rhythms may underlie stroke-induced diaschisis such as neuropathic pain or hemispatial neglect.
Clinical Applications of EEG in acute ischemic stroke
Apart from being a non-invasive and a widely available device, the ability of an EEG to detect changes in brain electrical activity in real-time makes it a good candidate for monitoring the evolution and treatment of stroke. We discuss how the EEG signals are modified in stroke and how they can be used as biomarkers to predict prognosis after stroke.
Several reports indicate the utility of an EEG in acute ischemic stroke (Table 1). An EEG is highly sensitive to detect electrophysiological activities induced by ischemic stroke and could even detect changes in patients with no abnormal signs in the initial CT scan.67,76 Due to advances in technology, it is possible to use an EEG anytime and anywhere through a more downsized and manageable portable version. 77 These options can be helpful in situations where time is of the essence, such as transportation of a patient via ambulance, or an initial assessment by paramedics. The prospect of being able to identify ischemic strokes earlier with the use of portable EEGs can expedite the necessary emergency treatments for ischemic stroke, such as intravenous thrombolysis and mechanical thrombectomy with the goal of improving the outcomes for said patients.
Summary of EEG characteristics in acute ischemic stroke.
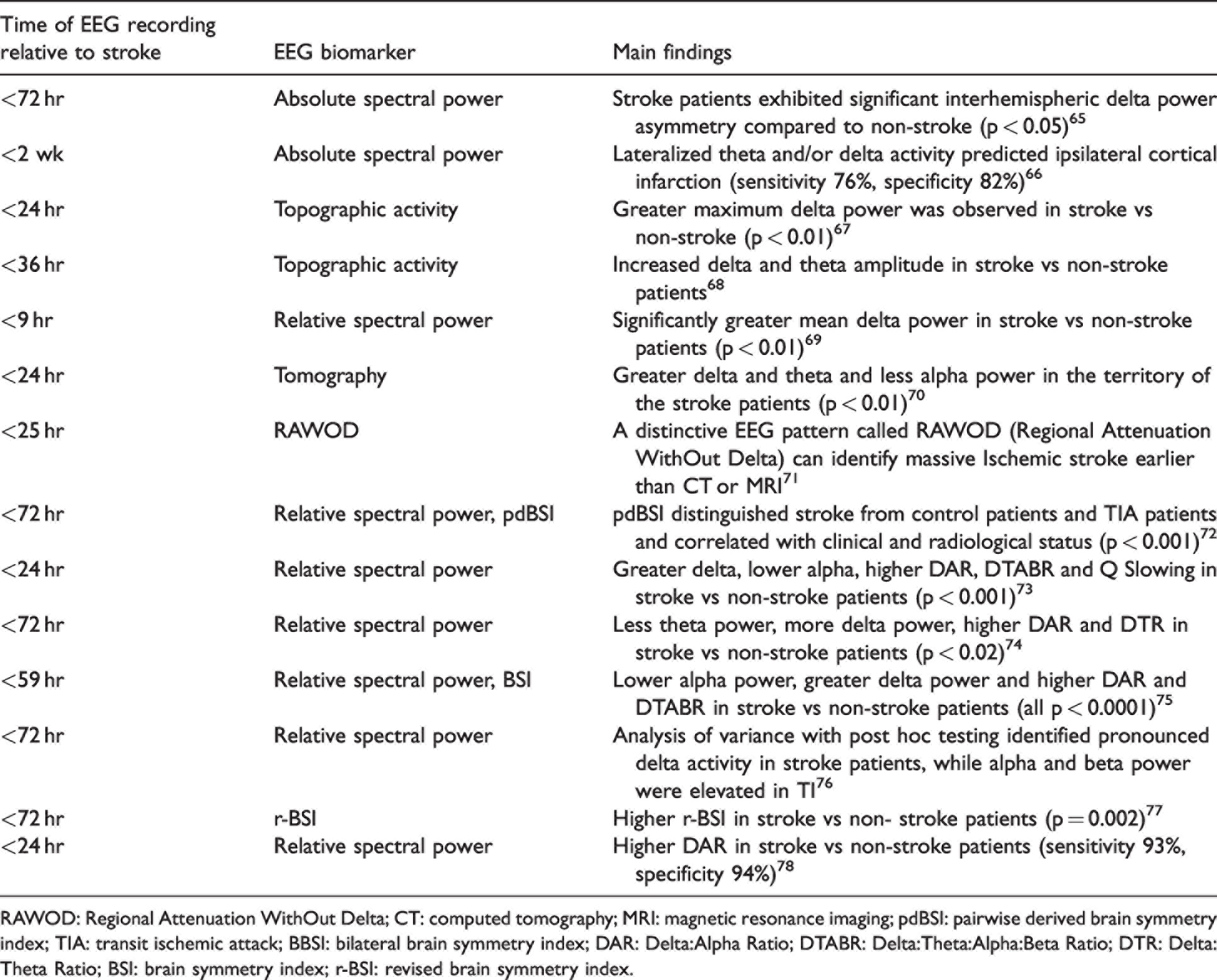
RAWOD: Regional Attenuation WithOut Delta; CT: computed tomography; MRI: magnetic resonance imaging; pdBSI: pairwise derived brain symmetry index; TIA: transit ischemic attack; BBSI: bilateral brain symmetry index; DAR: Delta:Alpha Ratio; DTABR: Delta:Theta:Alpha:Beta Ratio; DTR: Delta:Theta Ratio; BSI: brain symmetry index; r-BSI: revised brain symmetry index.
Similar to other experimental findings, greater δ power and lesser α or β activity was observed among stroke patients compared to healthy controls,71–73,92 while increased or decreased θ power have both been reported. 74 An asymmetric index such as the Brain Symmetry Index (BSI) or the global pair wise derived Brain Symmetry Index (pdBSI) is particularly instrumental in reflecting changes between both hemispheres over time. A significant asymmetry shown in BSI and pdBSI in brain waves was found in stroke patients compared to control.1,72,77 Some studies indicated the size or location of the ischemic region was correlated to EEG changes. 92 Larger infarct volume was often associated with increased δ and θ power but with decreased α and β power.69,93,94 Another study showed a trend towards a reduction in faster waves in the contralesional hemisphere when infarct size was larger. 93 As opposed to large infarct strokes, subtle focal θ activity, asymmetries in the α oscillation and mu rhythm were often detected in lacunar infarcts. 95 Abnormal EEG patterns were also reported and could result from the deafferentation of subcortical structures. 92 Interestingly, evidence suggests that EEG detection tends to show better sensitivity in the anterior circulation than the posterior circulation.66,67 In addition, some recent studies suggest that an EEG can distinguish a transient ischemic attack (TIA) from an ischemic stroke. Evoked potentials and spectral power across all bands, with greater δ power, less α power and less β power were identified in TIA compared to ischemic stroke. 76
Continuous EEG monitoring during thrombolysis
Reduction of δ power before symptomatic recovery within 20 min after r-tPA administration was reported in human stroke patients,96,97 consistent with finding in rodent EEGs in which a significant increase in the amplitude and recovery of the BSI after early recombinant tissue Plasminogen Activator (r-tPA) administration (1–3 hr after photothrombotic stroke). 98 Another study showed a clear improvement of the BSI during the 6 hours after r-tPA administration. 99 These studies suggest that continuous EEG monitoring with real-time brain electrical activity information may help to identify successful recanalization by r-tPA. This identification could enable physicians to consider additional treatments such as intra-arterial thrombolysis or mechanical thrombectomy accordingly.
Moreover, continuous EEG monitoring may also detect secondary severe adverse events, such as massive hemorrhagic transformation, severe cerebral edema, stenosis, or re-occlusion in an early stage. However, a big limitation of the thrombolysis studies is the lack of real-time intra-arterial monitoring of the clot dissolving. Therefore, a study combining continuous EEG with intra-arterial therapy will clarify more detailed changes before and after recanalization.
EEG as an electrophysiological biomarkers of stroke prognosis
Continuous EEG monitoring of ischemic stroke has become not only a tool to diagnose, but also a tool to predict the prognosis as an electrophysiological biomarker in the evaluation of functional outcome are frequently reported with the Modified Rankin Scale (mRS), Barthel Index/modified Barthel Index (BI/mBI), National Institutes of Health Stroke Scale (NIHSS) or mortality rate. Focal brain lesions may functionally impair remote regions, a phenomenon known as “diaschisis” in which the excitability and metabolism of the remote regions including the hemisphere contralateral to the stroke side are reduced.100,101
Greater δ and θ activity along with greater interhemispheric asymmetry, and lesser α and β activity, appear to predict a bad prognosis in both acute and chronic phase ischemic stroke. One study reported the greater δ and θ power and lesser β and γ power within 24 hr from the onset was associated with poor mRS at discharge. 102 Another study found that lower BI at 21 days was associated with higher BSI at admission. 103 Furthermore, the NIHSS value was found to correlate with sBSI and pdBSI values,104,105 while a greater contralateral θ power was associated with increased mortality at discharge or six months post-stroke. Conversely, a lack of slow activity with minimal decrease in other background frequencies predicts a good outcome. 106
EEG parameters evaluated during the pre-thrombolytic phase are indicative when predicting the prognosis of thrombolysis. One study reported the correlation between outcomes after treatment and the pre-thrombolysis hyper-acute EEG parameters. Relative δ power and α power, δ/α ratio, and (δ + θ)/(α + β) ratio significantly correlated with the seven-day and 12-month NIHSS, respectively. In an EEG study using the wireless and portable techniques, a good outcome as mRS ≤ 2 at 12 months was found strongly correlated to δ/α ratio, 73 suggesting these parameters in predicting short/long-term outcomes were not only feasible but might also contribute to the development of better treatment strategies. In conclusion, during ischemic stroke, the increase in lower frequencies and a decrease in faster frequencies are the main features. Persisting changes are associated with poor long-term functional outcome. The δ wave change appears to be the most reliable index for reduced CBF and brain metabolism during focal ischemia.
EEG/MEG to monitor post stroke functional recovery and efficacy of rehabilitation
Magnetoencephalography (MEG) is a non-invasive measurement of the brain's magnetic fields generated by electrical currents. The initial design of MEG by David Cohen in 1968 employed a copper induction coil for detection, which was plagued with noise. 107 Upon subsequent improvement, a superconducting quantum interference detector was able to successfully detect and measure the magnetic field. Direct measurement of brain function became possible with this technology that provides a very high temporal and spatial resolution image of neuronal activity. Since its early days being deployed as a device to evaluate the epileptic foci pre-surgically, 108 the clinical utility of MEG has expanded into many functional domains such as functional mapping of eloquent cortex. Mapping of the language cortex was found to be more accurate with MEG than with EEG due to the reduced noise level and lesser artifacts in the former.4,5,109 As a result, MEG is now widely used for pre-operative brain mapping before brain tumor surgery because it is capable of detecting functional brain regions within the tumor. 110 The latest MEG has more than 250 sensors and provides a high spatial resolution and accessibility to deeper brain structures compared to the EEG. Recently, MEG has been introduced to solve the complex problems of recovery and brain reorganization after stroke. It is particularly instrumental in predicting the prognosis and in evaluating the efficacy of rehabilitation methods (Supplemental Table 1), which have become the new focus in the clinical use of MEG.
Unlike scalp EEG that detects electrical fields generated by extracellular currents, MEG primarily detects the magnetic fields induced by intracellular currents. As such, MEG possesses some major advantages over EEG. First, the decay of magnetic fields as a function of distance is more pronounced than electrical fields, making MEG especially sensitive to superficial cortical activity. Consequently, MEG requires a smaller area of synchronized cortical activity to detect an epileptic spike relative to EEG. Second, MEG provides better spatial resolution of source localization than EEG, since magnetic fields are not distorted by tissue conductivity of the skull and CSF as in electrical fields. MEG is often combined with MRI to obtain an optimal structural perspective critical for brain mapping purposes (Figure 2). Although MEG is promising for diagnosing functional recovery and reorganization following a stroke, there are some limitations when applying it in actual clinical practice. MEG is much more expensive and time consuming compared to EEG. Additionally, it requires highly trained staff for data acquisition and analysis, limiting its accessibility. Despite its higher spatial resolution, the distance MEG is able to reach is inversely proportional to the magnetic flux density, making it difficult to assess deep brain structures. Under these conditions, the use of fMRI or PET would be good alternatives to assess these deeper structures.
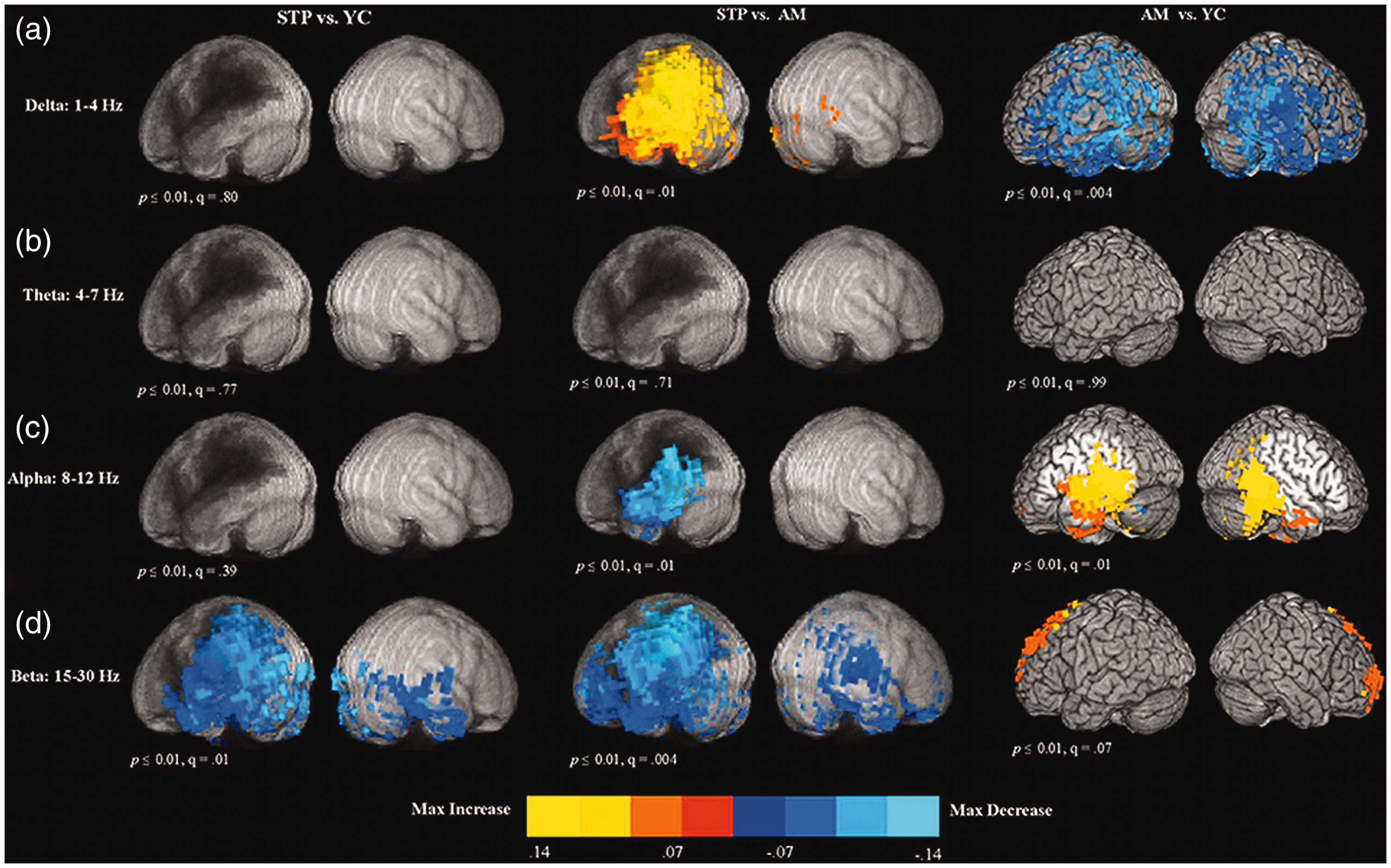
Age and stroke effect on brain oscillations detected by MEG. The maps represent the voxel-wise contrast resulting from a t-test comparing stroke and age effect (STP, stroke; YC, young controls; AM, age-matched). Resting-state data was collected for 5 min during MEG acquisition, followed by structural MRI scans. T-test comparison maps of relative delta power (a), theta power (b), alpha power (c) and beta power (d). Stroke increased the power of delta but decreased that of alpha and beta near lesion regions. To the contrary, age decreased delta, but increased alpha and beta power in specific brain regions (adopted from figure 4, Kielar A et al., 2016 111 ).
Like an EEG, MEG detected a bilateral increase of the slow-wave activity and increase of interhemispheric asymmetry in the acute phase following a unilateral ischemic stroke as a prediction of a worse functional outcome. 112 One study performed follow-up recordings of MEG in 16 patients with acute ischemic stroke of the MCA territory and found ∼10-Hz oscillations enhancement in the temporoparietal lobes as a sign of recovery. 113 Another report evaluating 12 patients with ischemic or hemorrhagic stroke confirmed poor functional recovery among patients who showed no response to stimulation in the somatosensory evoked field. 114 This observation led to the hypothesis that intensive sensory peripheral stimulation may maximize the use of residual sensory function and thus contribute to the improvement of the sensory deficit. By calculating the size of hand representation in the primary somatosensory cortex, another study determined how upper extremity motor function was affected in 15 stroke patients and found that the enlargement of the hand representation predicted good recovery. 115 MEG can provide a novel way of indirectly assessing primary motor cortex projection and the extent of recovery of sensorimotor function by localizing cortical sources of response to median nerve stimulation in stroke patients, 116 which has proven to be of particular value among those who cannot perform motor tasks due to paralysis.
The application of various rehabilitation strategies in stroke subjects, such as Mirror Therapy (MT), Rhythmic Auditory Stimulation (RAS), Music Supported Therapy (MST) and speech therapy have been effectively evaluated by MEG in recent years. By creating a reflective illusion of a disabled limb with positive visual feedback of limb movement using a mirror, MT has shown to reduce the initial asymmetry in movement-related β desynchronization between hemispheres, according to a spinal cord injury study. 117 Another series of studies validated the benefit of using pulsed rhythmic or musical stimulation-supported motor rehabilitation to improve limb function in stroke patients with RAS or MST accompanied by showing enhanced connectivity between the auditory and motor network nodes for β and γ oscillations by MEG. 118 MEG was also introduced to evaluate the therapeutic effect of speech therapy in patients with aphasia to optimize the rehabilitation process, in which increased activation in the right hemisphere after speech therapy appeared to be related to language function recovery. 119
Using EEG/MEG in combination with brain machine interfaces (BMIs) for stroke rehabilitation
The latest challenging application of MEG in stroke rehabilitation incorporates Brain–Machine Interface (BMI) 120 which circumvents the lack of feedback information commonly seen in stroke patients with low-mobility. 121 A recently developed BMI system allows stroke patients to control and manipulate a mechanical orthosis fixed to their paralyzed hand using mu-rhythm oscillations data detected by MEG in real time. 122 Compared to physiotherapy where patients can only move passively while simultaneously being told to imagine the active movement, BMI training is more proactive in improving a patient’s motor abilities. 123 With BMI, MEG is transformed from a passive recording device into an active treatment and rehabilitation instrument in which the rapidly evolving technology is certainly anticipated. Similarly, studies have used EEG-based recordings to perform a virtual reach while simultaneously exciting a specific part of the peripheral nervous system according to Hebbian learning.124,125 There is a large body of work regarding plasticity and the impact of BMI124–126 and selective attention127,128 that can lead to functional recovery from stroke. However, the functional recovery achieved with BMI technology among those patients remains modest due to several limitations: 1) Our insufficient knowledge of the neurophysiology correlates of recovery; 2) A substantial lack of understanding on strategies for targeted plasticity such as the timing between detected neural activity and peripheral stimulation; 3) Although BMI techniques focus on strengthening the connections between the brain and periphery through the residual connections, emerging evidence suggests that targeted rewiring of the brain may lead to better a functional outcome124,129 (see section ‘Targeted stimulation-based therapies for stroke’ for future potential directions).
Local field potential changes after experimental stroke
Local field potential changes near ischemic infarct after experimental MCA occlusion
Comparable to the changes observed in human stroke patients, the most consistent LFP/EEG characteristic during the acute and chronic phases of rodent focal ischemia was the increase of low-frequency and decrease of high-frequency oscillations (Table 2). Within a few minutes after MCAO, severe voltage suppression of LFP/EEG amplitude was observed in the ischemic hemisphere. Specifically following the temporary middle cerebral artery occlusion (tMCAO), the distribution of LFP/EEG changed from 25%–45% to 85% of δ oscillations, from 40% to 7% of θ oscillations, from 12%–15% to 5% of α oscillations, and from 3-20% down to only 3% of β oscillations.3,130 Following permanent MCAO (pMCAO), a reduction in LFP/EEG power across all frequency ranges was found in the ischemic ipsilateral cortex of rats acutely at 1–3 h and was associated with a decrease of 30% of CBF compared to baseline. Increased pdBSI was observed only in the acute phase of ischemia (from 15 min to one hour after occlusion) in young rats but up to 14 days in 12-month-old rats. 91
Representative EEG/LFP studies in rat models of MCA stroke.
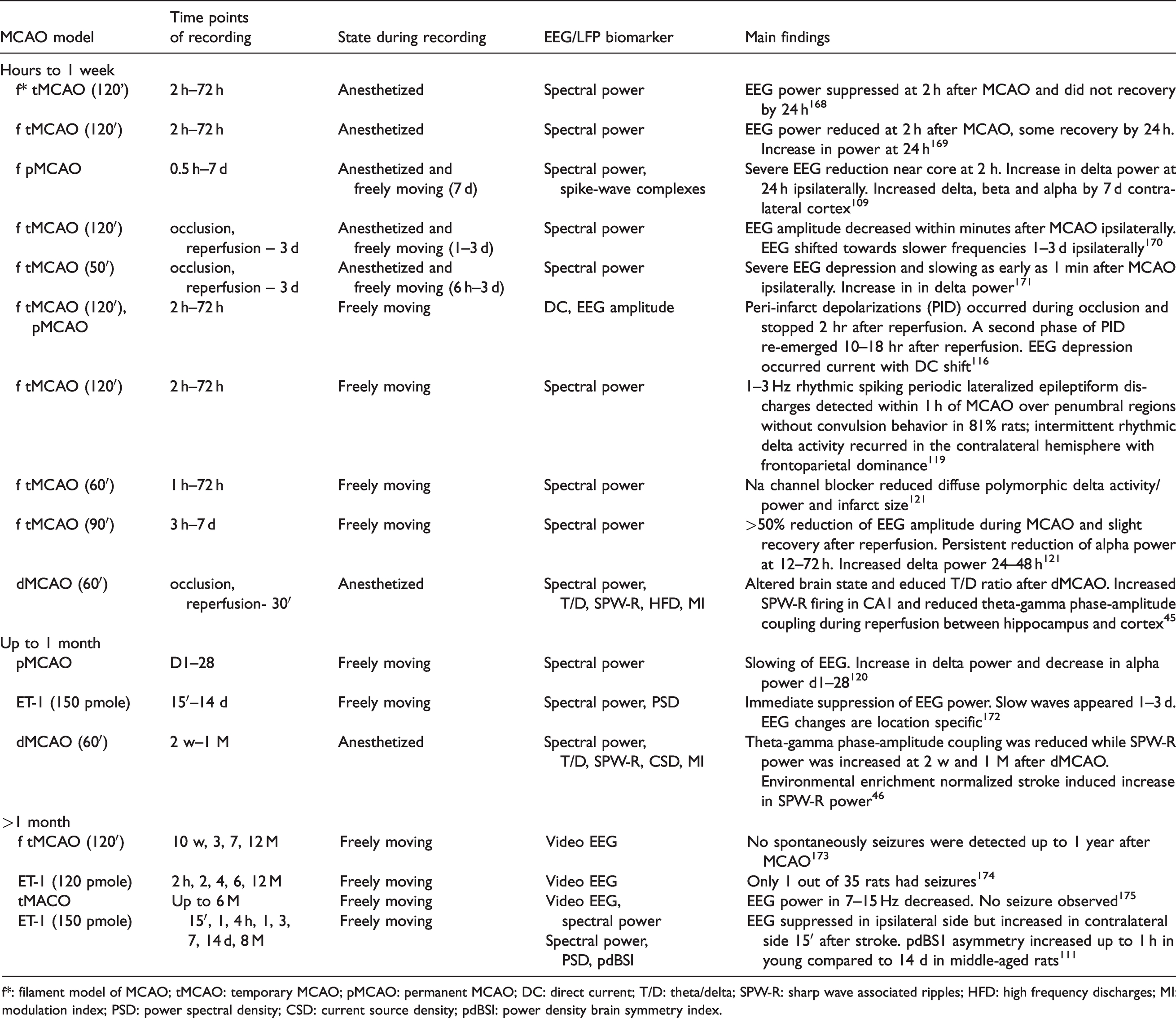
f*: filament model of MCAO; tMCAO: temporary MCAO; pMCAO: permanent MCAO; DC: direct current; T/D: theta/delta; SPW-R: sharp wave associated ripples; HFD: high frequency discharges; MI: modulation index; PSD: power spectral density; CSD: current source density; pdBSI: power density brain symmetry index.
Unlike neurons in the ischemic core where they can only depolarize in the presence of CBF at 20% below normal level, neurons in the penumbra can still repolarize following depolarization, but at the cost of perpetual depletion of valuable energy and the risk of core expansion. 131 During this critical period, sensory stimulation of the susceptible hot zone by tactile stimulation of the forelimb could trigger Peri-Infarct Depolarizations (PIDs) or spreading depression (SD), manifested as direct coupled potential shifts and suppressed electrocorticogram activity.132,133 Therefore, post-stroke LFP/EEG monitoring can provide information into the evolution of the ischemic core and penumbra.104,134 Detection of spreading depolarization requires direct current (DC) coupled recordings of local field potentials (LFPs),83,133 although sometimes the spectral profile of the electrical events assessed by EEG and ECoG have been suggested to provide relevant information about SD, including its prediction.135,136
Because the LFP/EEG amplitude depends on CBF, the recovery of LFP/EEG anomaly is often reported after ischemic reperfusion, but to various extents and speeds. For example, an increase of δ amplitude was observed in the ipsilateral hemisphere as early as one minute following intraluminal suture occlusion of the proximal portion of the MCA and lasted until the chronic phase, seven days or beyond;91,130 or even in the contralateral cortex one to seven days after tMCAO.3,84,91 However, the recovery of α or β amplitudes may take longer. One report showed that α amplitude decreased from day one to day 28 after pMCAO, another found a 35% reduction in the amplitude of α and β oscillations in the ipsilateral hemisphere three to seven days after tMCAO. 86 However, some studies reported an increase of β oscillation activity by seven days in the contralateral cortex after stroke in tMCAO rat models. 3
Similar to human stroke findings, some evidence implicates a correlation between the infarct volume and LFP/EEG changes. Acute δ oscillations increase, δ change index, pdBSI, relative α percentage, relative α-β percentage, relative δ-θ percentage, δ/α ratio, or δ-θ/α-β ratio were also reported to correlate with a larger infarct volume. 85 Additionally, commonly reported abnormal LFP/EEG patterns in experimental strokes included polymorphic slow-wave δ activity; intermittent rhythmic δ activity consisting of readily identifiable brief (<10 s) rhythmic large-amplitude bursts in the 4–7 Hz range; non-convulsive seizures, electro graphically defined as rhythmic discharges with spike components of 1–4 Hz frequency; occasional rhythmic spike-and-waves and polyspike discharges, not accompanied by motor convulsions; periodic lateralized epileptic discharge, consisting of interictal spikes; and sharp or slow-wave recurring with a variable period of 1–8 s.84,85 These changes are separate from the generalized or regional slow-wave activity and show a focal attenuation of a specific rhythm, usually the faster activity frequencies, and general attenuation or suppression of one or more brain oscillations.
Local field potential changes in brain regions remote from MCA infarct
Recent advances in functional connectivity suggest shared neuronal activation patterns define brain networks linking anatomically separate brain regions.137,138 The hippocampus is involved in memory function, yet direct ischemic lesions in the hippocampus are rarely observed in MCA stroke, 48 raising the question of how function is affected in regions remote from the stroke epicenter via stroke-induced change of brain connectivity and reorganization.139–141 By means of a meso-scale connectome analysis using the neuroVIISAS platform, 142 we previously found that all 6 damaged cortical regions caused by distal occlusion of the MCA (dMCAO) including the parietal cortex and somatosensory cortex, had a relatively high connectivity with brain regions involved in processing spatial information, 143 including reciprocal connections with the perirhinal cortex, lateral entorhinal cortex, postrhinal cortex, and non-reciprocal connections with the CA1 (Figure 3). The adjacency matrix clearly shows complete output connectivity of the lesioned regions with the ipsilateral entorhinal cortex. Thus, each of the lesioned regions is connected to the main input interface of the spatial learning system, suggesting that dMCAO leads to deafferentation of the hippocampal circuitry.
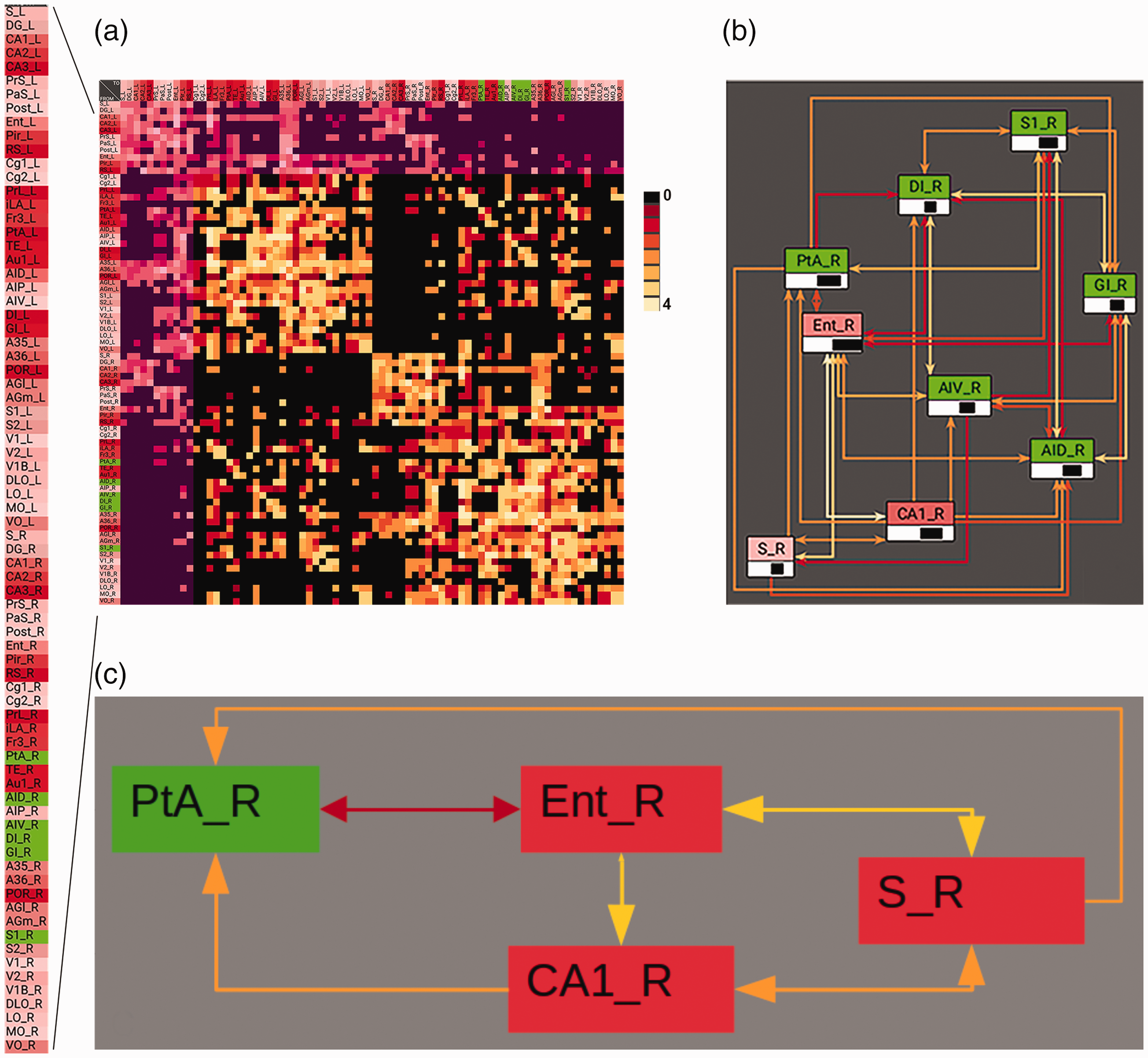
The lesioned cortical regions in the dMCAO stroke model are highly connected to ipsilateral limbic regions. Distal MCAO results in unilateral destruction of 6 cortical regions S1, GI, DI, PtA, AIV, AID (light green) and their neuronal connectivity. (a) Overview of weighted and directional neuronal connections of neocortical and allocortical (magenta) regions. _L: left hemispheric regions, _R: right hemispheric regions. Significantly denser intrahemispheric connections between the lesioned regions and other brain regions compared to interhemispheric connections; Connection weights (0–4) were shown in color (black to yellow). The dense reciprocal and overlapping connectivity is evident between the lesioned regions and the limbic regions. The connectivity of lesioned right hemispheric regions was highlighted with the main input region (entorhinal cortex), output region (subiculum), and CA1 region of the hippocampal formation for ipsilesional hemisphere (b) and hippocampal-parietal cortex interconnectivity (c). Abbreviations: S: subiculum; DG: dentate gyrus; CA1/CA2/CA3: fields of hippocampus; Prs: presubiculum; PaS: parasubiculum; PostS: Postsubiculum; Ent: entorhinal cortex; Pir: piriform cortex; RS: retrosplenial regions; Cg1: cingulate cortex area 1; Cg2: cingulate cortex area 2; PrL: prelimbic cortex; iLA: Infralimbic area; Fr3: Frontal cortex area 3; PtA: parietal association cortex; TE: temporal cortex; Au1: primary auditory cortex; AID: agranular insular cortex dorsal part; AIP: agranular insular cortex posterior part; AIV: agranular insular cortex ventral part; DI: dysgranular insular cortex; GI: granular insular cortex; A35: perirhinal cortex A36: ectorhinal cortex; POR: postrhinal cortex; AGl: lateral agranular prefrontal cortex AGm: medial agranular prefrontal cortex; S1: primary somatosensory cortex; S2: secondary somatosensory cortex; V1: primary visual cortex; V2: secondary visual cortex; V1B: primary visual cortex binocular area; DLO:, dorsolateral orbital cortex; LO: lateral orbital cortex; MO: medial orbital cortex; VO: ventral orbital cortex.
During acute stroke
By recording local field potentials under urethane anesthesia via a linear electrode array spanning the dorsohippocampus to deep cortical layers above, we found that dMCAO acutely altered the brain state in the hippocampus with reduced high-θ state and increased low-θ state, rendering the LFP with a decreased T/D ratio as compared to the pre stroke recording. 48 This change is similar to the finding in human EEGs near ischemic infarct soon after stroke onset, suggesting that dMCAO affects neural activity in remote brain regions. During MCA occlusion, there was a temporary reduction of θ and γ power bilaterally in the hippocampus, followed by a gradual recovery towards baseline level. The disturbance of signal power in δ, θ and γ persisted after ischemic reperfusion. 48 There was also a significant increase in SPW-R s firing during ischemic reperfusion following MCA occlusion in the CA1 as accompanied by the sharp waves in the stratum radiatum compared to a few ripple events during baseline recording. In addition to SPW-R s, the occurrence of High-Frequency Discharges (HFD) in the range of 250–450 Hz was also increased during ischemic reperfusion following dMCAO (Figure 4), resulting in the increased proportion of HFDs in the pathological range compared to the physiological range of ripples. 48 The perturbation of either SPW-Rs or HFDs after stroke could be attributed to altered excitatory and inhibitory drives. For example, GABAaR and GABAbR are downregulated after hippocampal ischemia in guinea pigs 144 and focal ischemia in mice. 145
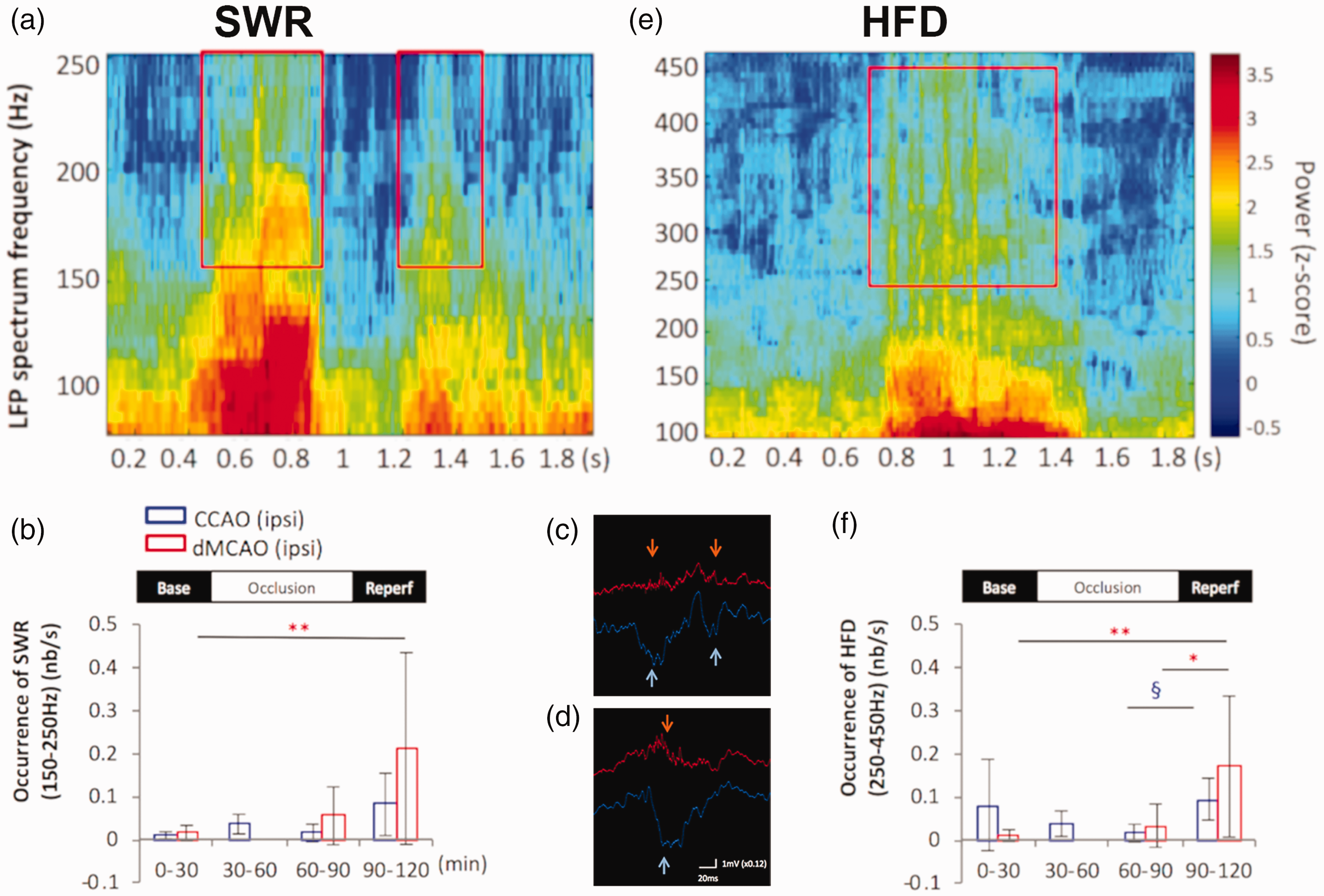
Cortical ischemic stroke increased sharp wave associated ripples (SPW-R) and high-frequency discharges (HFD) in the remote hippocampus. a and e, Frequency spectrograms of the LFP recording from CA1 pyramidal layer during dMCAO display the presence of SPW-R in the frequency range of 150–250 Hz (a) and the high frequency discharges in the range of 250–450 Hz embedded in the ripple discharges (e). An increase in the occurrence of SPW-R (b) and HFDs (f) was detected in the ipsilesional CA1 pyramidal layers during MCA reperfusion compared to baseline in the bar graphs. Representative traces show each sharp wave event in the radiatum (light blue arrows) is accompanied by SPW-R (orange arrows) in the CA1 pyramidal layer before stroke (c) and after ischemic-reperfusion with a longer duration of SPW-R (d). Modified from He J et al., figure 2. 48
Unlike the dMCAO model that produced an ischemic lesion mainly in the somatosensory cortex, unilateral hippocampal photothrombotic stroke caused an extensive hippocampal infarction and reduced ipsilateral γ activity and elicited a direct current voltage shift. However, some HFDs in the range of 250–450 Hz recorded in the hippocampal photothrombotic model reminiscent of epileptiform discharges were also detected during the acute stage of our dMCAO model. This suggests that brain regions remote to the ischemic core could also suffer from similar electrophysiological perturbations.
Both cortical low (30–60 Hz) and high γ (60–120 Hz) bands engaged in dynamic and differential phase-amplitude modulation during acute stroke as demonstrated by the changes of the Modulation Index (MI). Increased MI between cortical high γ bands and hippocampal θ (4–7 Hz) (MIHiG-θ) or low frequency oscillations (0–4 Hz) (MIHiG-Lowfreq) was observed during MCA occlusion and the reperfusion period in the ipsilateral CA1 layer during high theta duration (HTD) but not low theta duration (LTD) phase. On the other hand, increased coupling of cortical low γ and hippocampal low frequency oscillations (MILoG-Lowfreq) were found during MCA occlusion in bilateral CA1. Furthermore, the phase of the δ wave along with cortical high γ coupled during MCA occlusion continued to shift towards lower frequency oscillation (<1.5 Hz) during reperfusion. Increased MIHiG in θ and low frequencies phases were also detected in the ipsilateral stratum lacunosum-moleculare layer during the dMCAO occlusion period.
The unexpected increase of coupling between cortical γ power and hippocampal θ as well as low frequency oscillations during dMCAO occlusion and after reperfusion may reflect an attempt to compensate for the loss of connectivity between the cortex and hippocampus during acute ischemic stroke. The loss of communication between brain regions and the associated increase in phase-amplitude modulation of cortical low γ was previously reported in the near death state of Wistar rats, though with α- (10–15 Hz) and θ oscillations (5–10 Hz). 146 Whether the increased θ-γ coupling during acute stroke is an effort of the brain to compensate for the temporary loss of cortical input to the hippocampus in the background of large-scale reduction of CBF remains to be verified. In summary, cortical stroke caused an imbalance of excitatory and inhibitory circuits in the remote hippocampus region and disrupted the cortico-hippocampal networks.
During chronic stroke
Cognitive impairment is not uncommon among patients after a stroke, even following first-ever stroke. 147 However, how neural activity is chronically affected by cortical stroke in the hippocampal region remote to ischemic infarct is not well characterized. Similar to acute stroke, brain state was disrupted 2-4 weeks after dMCAO as shown by a decreased duration of high θ state detected in the bilateral hippocampus, pointing to the persistent nature of altered brain state after ischemic insult. δ power appeared to be significantly reduced two weeks after dMCAO compared to non-stroke controls in both the cortex and hippocampus, while γ power peaked at two weeks and gradually reversed back to prestroke level one month after the stroke. Unlike the temporary reduction of hippocampal θ and γ power seen during acute stroke, γ power was elevated during chronic stroke possibly due to a compensatory response to the acutely reduced γ oscillatory activity.
Characteristics of SPW-Rs are chronically altered following dMCAO. The signal power of SPW-Rs was increased in the bilateral CA1 pyramidal layer both two weeks and one month after dMCAO. Interestingly, the duration of SPW-R is prolonged two weeks after stroke but is shortened one month after stroke compared to that of the non-stroke control rats. During SPW-R firing, current through the CA1 field of the hippocampus as measured by current source density analysis, revealed pairs of dipoles with the apparent source centered in the pyramidal layer and the sink in the radiatum. Following dMCAO, the strength of the current dipole increases above baseline at two weeks and then decreases below at one month 49 (Figure 5). Thus, it appears there is a compensatory hyperactivity as a result of stroke that peaks at two weeks after stroke, followed by frequency-specific normalization or restoration to pre stroke level around one month post stroke.
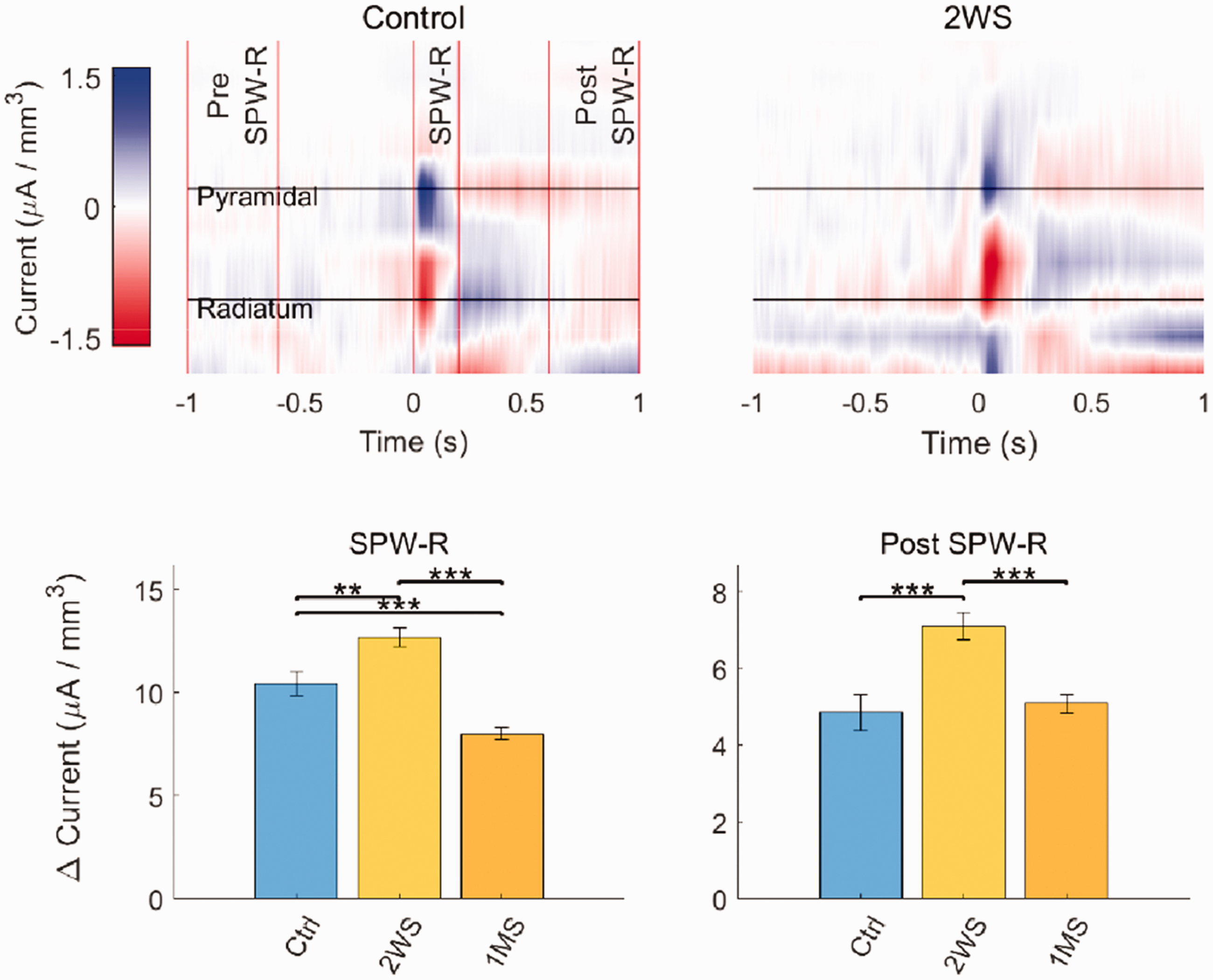
Cortical stroke changed the current flow in the hippocampal CA1 layer. Current source density map of ipsilesional CA1 in relation to the timing of ripple firing: before (Pre SPW-R), during, (SPW-R) and after SPW-R (Post SPW-R). Change in local current flow was determined using the difference between the minimum and maximum dipole amplitudes during each time window. An increased current difference was detected at 2 weeks after dMCAO (2 WS) and dissipated at 1 month after stroke (1MS). Modified from Ip Z et al., figure 4. 49
Phase Amplitude Coupling (PAC) between cortex and hippocampus and within the hippocampus is present during high θ state where θ-γ coupling, and δ-high γ coupling is present bi-directionally. θ-γ coupling between the hippocampus and cortex, as well as within the pyramidal layer was significantly lower at 1MS compared to non-stroke control (Figure 6). During the low θ state, only δ-high γ coupling was present which did not change during chronic stroke. The diminished PAC between hippocampal θ and cortical γ one month after stroke suggests a persistent breakdown of network communication resulting from ischemic injury, likely caused by the deafferentation of the hippocampal regions from cortical stroke.
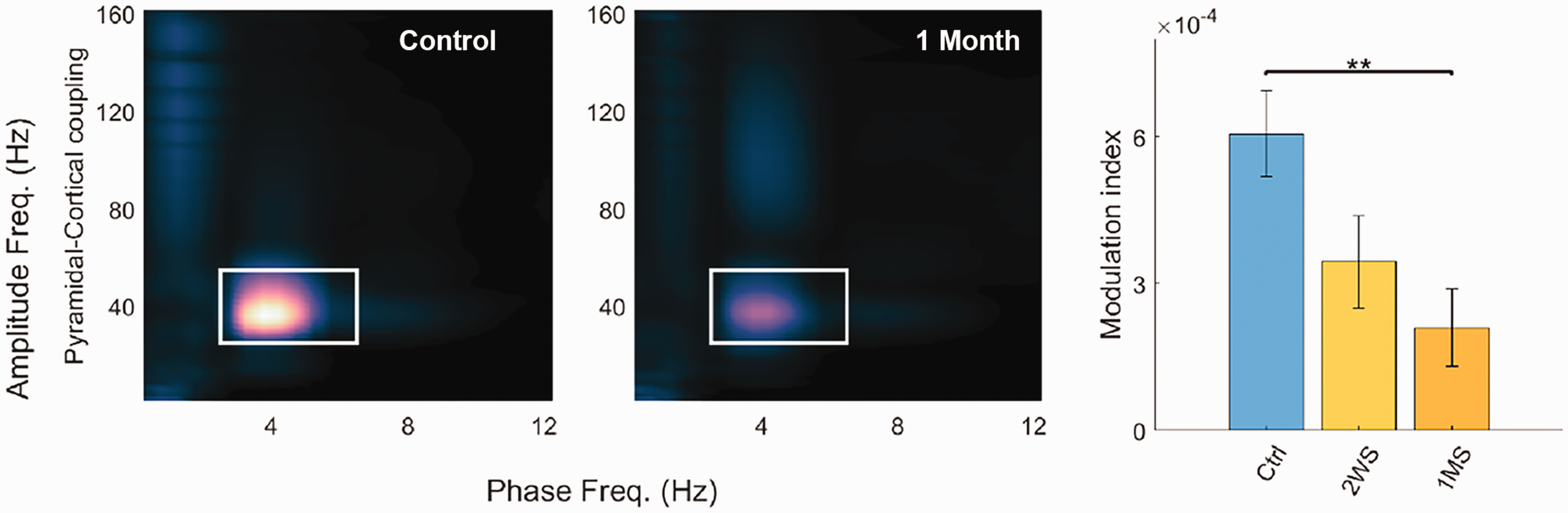
Cortical stroke reduced coupling between CA1 θ and cortical γ oscillations. Phase amplitude comodulograms showing modulation between cortex and hippocampus for control (Ctrl) and 1 month after dMCAO (1MS), in which θ-γ coupling was demarcated by white rectangle. Significant reduction of average θ-γ modulation index was detected in rats 1 month after stroke compared to the control rats. **: p < 0.01. Modified from Ip Z et al., figure 5. 49
Effect of environmental enrichment on brain oscillations in chronic stroke and implication in the recovery of function
Environmental Enrichment (EE) has been widely studied in laboratory animals as an effective method to enhance neuroplasticity, neurogenesis, and to improve behavioral recovery from various brain injury models. 148 With respect to stroke, EE has exerted synergistic effects in enhancing post stroke motor function recovery in rats when combined with task-specific reach training, 149 aside from the many challenges and limitations in translating EE from the bench to clinical practice in rehabilitation medicine. Despite the early skepticism on the efficacy of EE, arguing that the human environment is already enriched compared to the impoverished laboratory animal’s environment, increasing evidence has revealed that most stroke patients were in isolation and physically inactive in the hospital setting; 150 this suggests a need to further stimulate and enrich the experience of post stroke patients. Recent evidence suggests that embedding an enriched environment in an acute stroke unit could increase activity in stroke patients. 151 Nevertheless, some foreseeable difficulties have impeded the implementation of EE in clinical practice such as the lack of standard EE paradigms that are essential while conducting clinical trials, or the lack of knowledge in the extent and duration of enrichment to achieve maximal functional recovery, that may vary on an individual level.
EE has consistently been shown to improve the recovery of motor and spatial memory function following dMCAO stroke (Figure 7). Stroke-induced hypoactivation of limbic regions following maze learning can be visualized via Fos imaging, a proxy of neuronal activation. Housing in the EE for one month after stroke reversed the hypoactivation (Figure 8). As a physiological correlate of neural function, we wanted to determine whether EE also improved the perturbation of brain oscillations caused by ischemic stroke. EE by itself reduced the signal power of γ in the hippocampus and restored chronic stroke-induced increases of γ power, although it had no effect on δ nor θ among stroke and non-stroke animals. Exposure to EE restored HTD in stroke rats, while EE and stroke alone reduced the HTD state. EE mitigated the stroke induced increase to SPW-Rs signal power and decreased its duration, suggesting that EE counterbalanced the excitatory and inhibitory networks disrupted by stroke. 49 Although prolonging SPW-R via optogenetic stimulation of the CA1 neurons reportedly also improved the memory performance, 54 further studies are needed to understand the optimal SPW-R duration for memory function to differentiate the effects of stroke and EE on the increased duration of SPW-R.
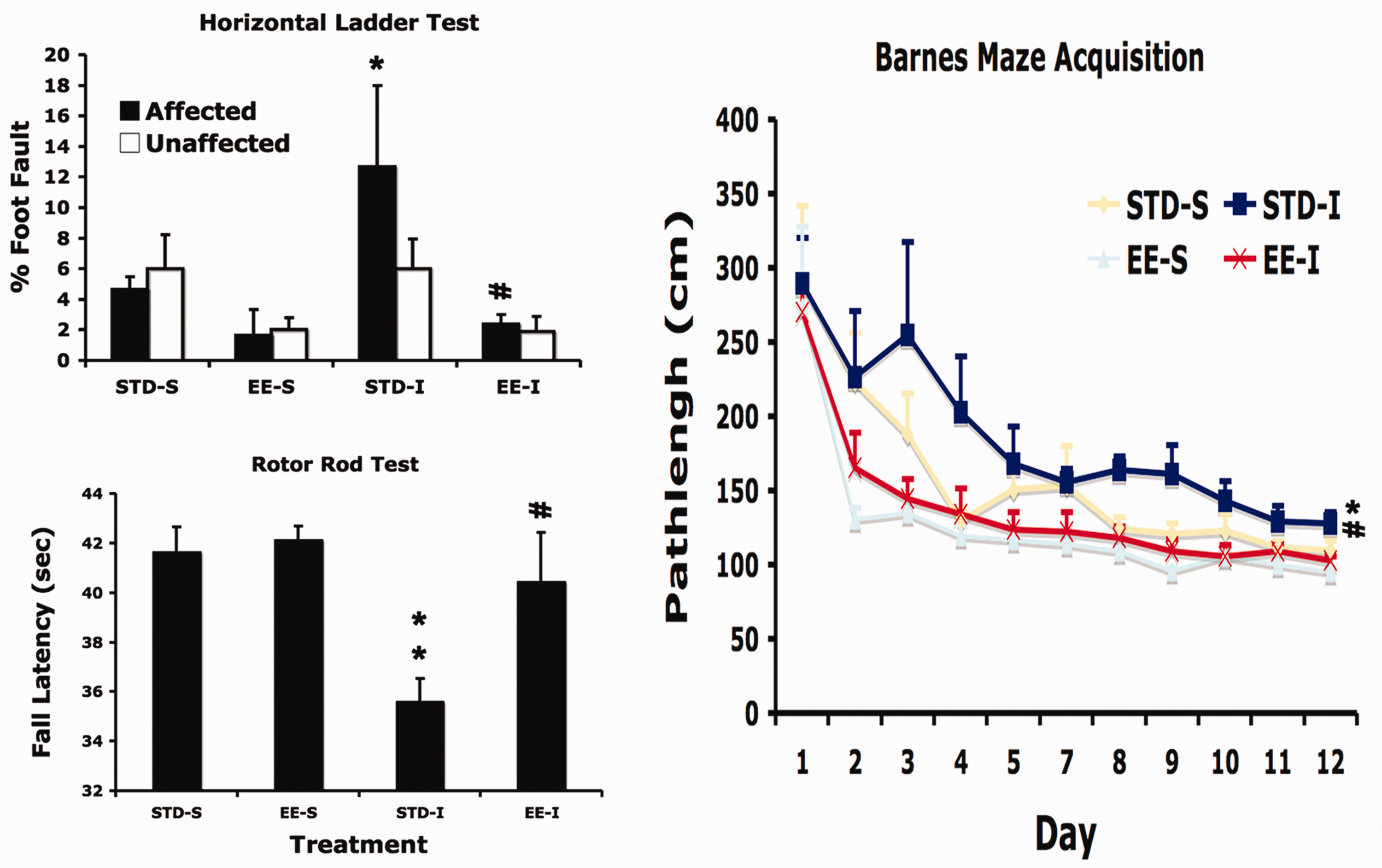
Environmental enrichment reduced cortical stroke induced functional impairment. Rats were subjected to either ischemic (I) or sham (S) surgery and housed in either standard- (STD) or enriched environment (EE) one week after surgery for one month. Two-way ANOVA detected a global effect of stroke on motor function impairment 5 weeks after stroke (Ladder test: p < 0.05; Rotor rod: p < 0.01), while EE had a beneficial effect on both motor and memory function (Ladder test: p < 0.05; Rotor rod test: p < 0.005; Barnes maze test: p < 0.01). Post hoc test detected significant difference between STD-S and STD-I groups (*: p < 0.05; **: p < 0.01), and between STD-I and EE-I groups (#: p < 0.05; ##: p < 0.01).
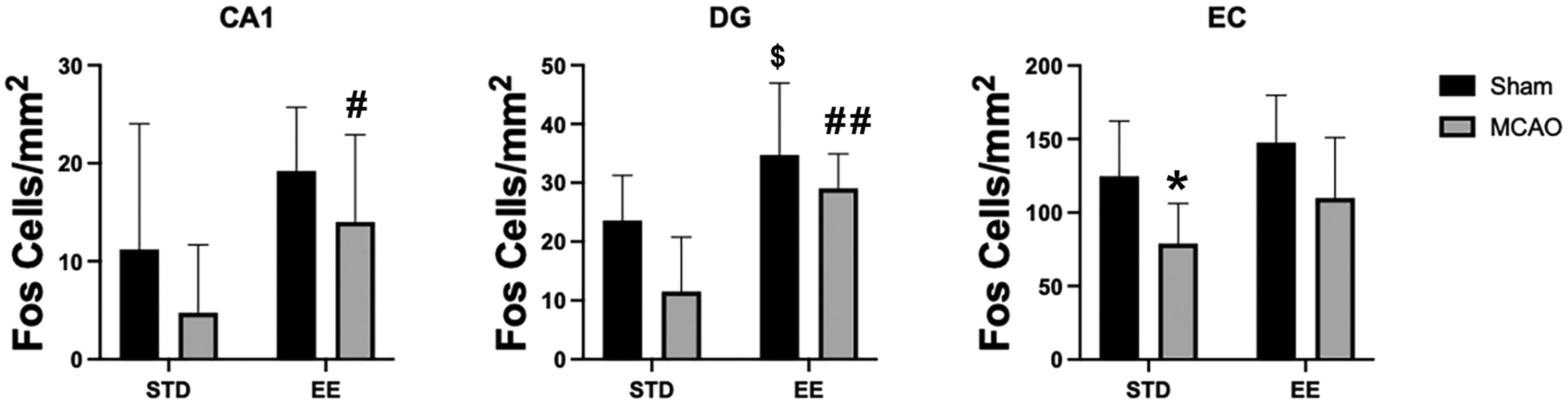
Environmental enrichment reversed cortical stroke induced hypoactivation in limbic regions including hippocampal CA1, dentate gyrus (DG) and entorhinal cortex (EC). Ninety minutes after 5-days of training in the Barnes maze, rats were processed for Fos immunohistochemistry. Two-way ANOVA detected a global negative effect of stroke (CA1: p < 0.05; DG: p < 0.005; EC: p < 0.001) and positive effect of enriched environment (EE) (CA1: p < 0.005; DG: p < 0.001; EC: p < 0.01) on Fos immunoreactivity. Specifically, stroke caused hypoactivation in the EC in the STD groups (*p < 0.05). EE restored neuronal activation in the CA1 and DG following learning in the Barnes maze relative to standard environment (STD) (Sham EE vs Sham STD: $p < 0.05; MCAO EE vs MCAO STD: #p < 0.05, ##p < 0.01).
Contrary to its known benefit on synaptic plasticity and cognition, EE unexpectedly lowers the levels of θ-γ coupling in the hippocampus. 49 Via mechanisms that are currently unknown, these results suggest there is a reduction of information flow between the cortico-hippocampal networks for both stroke and EE groups. Our results begin to uncover the complex interaction between stroke and EE, albeit further studies in awake animals are warranted to validate these findings and determine the underlying mechanisms. Nonetheless, these neural features may be areas of interest to further understand the network imbalances caused by stroke and to investigate future therapeutic interventions.
Targeted stimulation-based therapies for stroke
Brain stimulation is believed to modulate the excitability of neural circuits and increase the likelihood of neuroplastic change. Because neurons that fire together, wire together, 152 various approaches combining brain stimulation and rehabilitative training have been proposed to take advantage of stimulation-induced neuroplasticity for neurorehabilitation. 153 In particular it has been shown that repetitive Transcranial Magnetic Stimulation (rTMS), transcranial Direct Current Stimulation (tDCS), and Epidural Cortical Stimulation (ECS) can enhance neural plasticity in the motor cortex post-stroke. The modulatory effects of rTMS have been used in stroke rehabilitation to both induce cortical excitation 154 using high-frequency (≥3Hz) rTMS applied to the lesioned motor cortex and induce cortical inhibition 155 using low frequency (near 1 Hz) rTMS to contralesional motor cortex. 156 Similar to rTMS, tDCS has been used for stroke rehabilitation in the motor cortex to both excite lesioned areas using anodal tDCS and inhibit contralesional areas using cathodal tDCS. The rationale behind using these techniques being, that exciting ipsilesional areas promotes rewiring of perilesional neurons to take over lost function and speed the recovery process, while inhibiting the contralesional areas may restore the balance between the two hemispheres. 156 Furthermore, a large body of animal and human studies have shown brain stimulation promotes motor recovery following stroke. 157 This led to the use of ECS in humans which initially looked promising but failed to show significant efficacy in large clinical trials. Although rTMS, tDCS, and ECS all modulate cortical excitability, the induced changes in motor recovery are most effective when combined with motor training. 156 The effects of these stimulation techniques alone on brain physiology and functional recovery remain poorly understood.
More recently, other studies have reported reorganization of the brain using targeted electrical or optogenetic stimulation,158–163 that show promise for using these paradigms for neurorehabilitation. In particular, closed-loop activity-dependent stimulation has received more attention due to its dependence of stimulation protocols on the state of the brain.158–160 In these techniques the stimulation parameters are informed by the activity of the network or a nearby area. Notably, the stimulation protocols are informed by the Hebbian Plasticity model. At the cellular level, connections between two neurons are strengthened when the firing of one neuron repeatedly contributes to the firing of the second, a mechanism termed Hebbian plasticity. 152 Spike Timimg Dependent Plasticity (STDP), according to Hebbian rules, 152 states that the probability of connectivity change between two neurons depends on the relative firing time between them. Based on STDP, the probability of any change between two neurons is close to zero if the delay between their firing is more than 100 ms. This probability is highest for small delays such as 5 ms. Activity-dependent stimulation according to STDP rules have been shown to induce more robust neural plasticity,156,159,160 and restore normal patterns of functional connectivity between cortical regions. 156 It has indicated that activity-dependent brain stimulation in accordance with Hebbian principles 152 may lead to greater recovery from the motor deficits caused by stroke.156,164 Targeted stimulation in these studies have shown the feasibility of fine-scale changes in functional connectivity both locally 160 and at the network level.162,163
Despite the therapeutic promise these techniques have shown, there are major discrepancies that need to be addressed before these techniques can be used for stroke therapy. Previous stimulation techniques haven’t been specific enough, and risk unintentionally affecting large areas of the brain and their connections. Technologies with high spatial resolution capabilities are needed to control the off target 163 and widespread network effects. 162 Furthermore, the brain’s natural connectivity may play an important role in the plasticity mechanisms and has yet to be incorporated in generating the stimulation protocols. Recent findings show that the underlying network structure is an important mediator of the response to brain stimulation. 165 Moreover, since each injury is unique in various aspects, an optimal stimulation protocol needs to attend to the patient’s individualized needs. As stated earlier, strokes may impact both local and distant circuits, which can be reflected in the neurophysiological measures such as coherence and cross-frequency coupling. 166 Recent evidence suggests that brain stimulation has the potential to induce plastic changes and compensate for these functional connectivity alterations following neural damage.162,166 Our understanding of the effects of stimulation on brain oscillations and the underlying mechanisms that lead to plasticity change are still in their infancy. Emerging technologies enabling large-scale manipulations of brain circuits such as optogenetics in non-human primates167–169 can provide significant insight into these underlying mechanisms and galvanize the creation of more sophisticated and personalized stimulation-based therapies for stroke patients.
Apart from direct brain stimulation of the motor cortex, sensory stimulation has been attempted as a treatment of stroke. Although spreading depression170,171 was reported to be triggered by sensory stimulation following stroke, 132 recent work has contradicted this finding by showing the protective effect of peripheral stimulation within 5 hours of stroke.172–174 Simultaneous neural recording during sensory stimulation has shown a decrease in the degree of synchrony in LFP that was elevated due to stroke, taking it back to pre-stroke levels. It is possible that the extent of residual blood flow surrounding the stimulation determines the outcome of stimulation. Neural recordings across multiple brain areas during a stroke and following ischemic lesion formation could be critical in understanding the current contradictory results reported on sensory stimulation following stroke.
Conclusions
The integration of electrophysiological biomarkers obtained from extracellular recordings during acute or chronic stroke will be of great significance to clinical and pre-clinical stroke studies. In the clinical realm, spatial resolution and signal quality continues to improve due to emerging technologies. In both clinical and preclinical stroke research, advances in data mining techniques and sophisticated computer modeling have substantially improved our understanding of the underlying neural mechanisms in response and adaptation to ischemic injury and therapies.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221105677 - Supplemental material for Pathological changes of brain oscillations following ischemic stroke
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221105677 for Pathological changes of brain oscillations following ischemic stroke by Yoshimichi Sato, Oliver Schmitt, Zachary Ip, Gratianne Rabiller, Shunsuke Omodaka, Teiji Tominaga, Azadeh Yazdan-Shahmorad and Jialing Liu in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grant R01NS102886 (JL), R21NS120193 (JL), Research Career Scientist award IK6BX004600 (JL), the Eunice Kennedy Shiver National Institute of Child Health & Human Development of the National Institutes of Health under Award Number K12HD073945 (AY), and the Center for Neurotechnology (CNT, a National Science Foundation Engineering Research Center under Grant EEC-1028725) (AY).
Acknowledgements
The authors would like to thank Mr. Jaime Falcone-Juengert for manuscript editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
YS contributes to the section of clinical stroke and recovery
OS contributes to the stroke and connectome interpretation
ZI contributes to the chronic experimental stroke interpretation
GR contributes to the basic LPF/EEG concepts and acute experimental stroke interpretation
SO contributes to the clinical stroke knowledge
TT contributes to the clinical stroke knowledge and overall support
AYS contributes to the concept of brain stimulation and BMI
JL contributes to the overall concept, writing and interpretation of clinical and preclinical stroke studies including those from her own lab.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
