Abstract
Several studies suggested the association of migraine with deep white matter hyperintensities (WMHs). We aimed to explore the cerebrovascular reactivity (CVR), deep WMH burden, and their association in patients with migraine using a state-of-the-art methodology. A total of 31 patients with migraine without aura and 31 age/sex-matched controls underwent 3T MRI with prospective end-tidal carbon dioxide (CO2) targeting. We quantified deep WMH clusters using an automated segmentation tool and measured voxel-wise CVR by changes in blood oxygen level-dependent signal fitted to subjects’ end-tidal CO2. The association of migraine and CVR with the presence of WMH in each voxel and interaction of migraine and CVR on WMH were analysed. Patients had a higher number of deep WMHs than controls (p = 0.015). Migraine and reduced CVR were associated with increased probability of having WMHs in each voxel (adjusted OR 30.78 [95% CI 1.89–500.53], p = 0.016 and adjusted OR 0.30 [0.29–0.32], p < 0.001, respectively). Migraine had an effect modification on CVR on deep WMHs (p for interaction <0.001): i.e. the association between CVR and WMH was greater in patients than in controls. We suggest that the migraine-WMH association can be explained by the effect modification on the CVR.
Keywords
Introduction
Several studies suggested that migraine is associated with a greater burden of deep white matter hyperintensities (WMHs) compared with migraine-free controls, especially in non-elderly age groups. 1 –4 As WMHs are associated with an increased risk of stroke, 5 uncovering the pathophysiology of WMHs would be helpful for understanding the underlying mechanisms of stroke in patients with migraine. However, the mechanisms of WMH development, in association with migraine, have not been fully elucidated.
Recently, our group documented that a decreased vasodilatory response, as measured by cerebrovascular reactivity (CVR) to carbon dioxide (CO2), is associated with higher WMH loads, using transcranial Doppler in patients with migraine. 6 That provided the first evidence of a CVR-WMH association in migraine. This study, however, had the limitations that CVR was measured using transcranial Doppler, which has limited spatial resolution (i.e. it only reflects major arteries, while CO2-induced vasodilation occurs in the distal arteriolar beds), 7 and breath holding was used as the vasodilatory stimulus, by which the degree of arterial CO2 increment may differ between individuals. 8 Therefore, a further study was required to confirm the findings and to overcome the methodological limitations of the vasodilatory stimuli and recording method.
In the present study, we aimed to investigate the association between CVR and WMH in migraine using the most advanced techniques. To investigate CVR with the greatest temporal and spatial resolution, blood oxygen level-dependent (BOLD) MRI and a state-of-the-art prospective end-tidal CO2 targeting method were used. 9,10 Deep WMHs were segmented by a machine learning-based automated segmentation tool developed for this purpose, 11 and analyzed with regards to the corresponding voxels’ CVR in patients and matched controls.
Materials and methods
Patients
We recruited patients with episodic migraine at the Samsung Medical Center headache clinic between April, 2018 and December, 2018. Inclusion criteria were as follows: 1) adults aged 18–50 years; 2) diagnosis of migraine without aura based on the International Classification of Headache Disorders 3rd edition 12 ; 3) absence of vascular risk factors such as hypertension, diabetes, dyslipidemia, cardiac disease, stroke, and current or past history of smoking; and 4) no current use of any regular medications including migraine preventive agents. Patients with chronic migraine and/or medication-overuse headache were excluded.
Controls
Age-sex-matched headache-free controls were also recruited during the same study period. Inclusion criteria were as follows: 1) adults aged 18–50 years; 2) no headaches other than very low frequency tension-type headache as remembered by the subjects; 3) absence of vascular risk factors such as hypertension, diabetes, dyslipidemia, cardiac disease, stroke, and current or past history of smoking; and 4) no current use of any regular medications including migraine preventive agents. Controls were free from any other chronic pain disorders.
Patient consent and exclusion criteria
As this study involved experimental manipulation of CO2 levels, we thoroughly explained the study protocol to patients and controls, and screened for contraindications to hypercapnia, hyperventilation, or MRI. Subjects were excluded if they 1) could not breath 20 L for 1 minute because of cardiopulmonary disease (e.g. chronic obstructive pulmonary disease or heart failure), 2) had a disease that could be exacerbated by elevated PaCO2 in blood (e.g. interstitial lung disease or amyotrophic lateral sclerosis), 3) were unable to write the headache diary due to cognitive decline, 4) had contraindications to MRI, 5) were pregnant or anticipated a pregnancy during the study period, or 6) refused to participate. Only participants who provided written informed consent were included in this study.
Standard protocol approvals and registrations
The Samsung Medical Center Institutional Review Board approved this study. We conducted the study following the ethical standards of the Helsinki Declaration of 1975 (and as revised in 1983). This study is a cross-sectional analysis of an ongoing prospective follow-up study registered at ClinicalTrials.gov (NCT03494673).
Clinical evaluation
All patients and controls were evaluated using a structured headache questionnaire and a clinical interview. Migraine diagnosis was made by two headache neurologists (M.J.L. and S.C.) based on the International Classification of Headache Disorders 3rd edition. 12
MRI acquisition
All participants underwent 3T MRI using an identical scanner (Ingenia; Philips, Best, The Netherlands). Patients were interictal at the day of MRI acquisition. However, three patients took acute headache medication the day before the MRI exam. All participants were instructed to remain awake and relaxed with their eyes open and fixed on a specific point. 3D T1-weighted, fluid-attenuated inversion recovery (FLAIR), and BOLD MRI with and without CO2 manipulation were obtained from all participants. The imaging parameters of T1-weighted data were as follows: repetition time = 9.9 ms, echo time = 4.6 ms, field of view = 240 × 240 mm2, voxel size = 1 × 1 × 1 mm (acquired) and 0.5 × 0.5 × 0.5 mm (reconstructed), total number of slices = 360, and total scan time = 381 seconds. 3D FLAIR data was acquired with the same parameters as T1-weighted data except for repetition time (4,800 ms), echo time (300 ms), and inversion time (1,650 ms) with a total scan time of 341 seconds. Parameters for BOLD MRI were as follows: SENSE acceleration factor = 1, repetition time = 3,000 ms, echo time = 30 ms, flip angle = 90°, field of view = 220 ×220 mm2, pixel resolution = 1.25 × 1.25 mm2, slice thickness = 4 mm, number of slices = 33, and number of volumes = 280.
Prospective CO2 targeting
End-tidal pressures of carbon dioxide (EtCO2) and oxygen were monitored and manipulated using an automated gas blender that adjusts the gas composition and flow to a sequential gas delivery breathing circuit (RespirAct™; Thornhill Research Inc., Toronto, Canada). 13 The step-ramp sequence was used during BOLD measurements: baseline EtCO2 for 120 s (normocapnia), followed by an abrupt hypercapnic step change to +10 mmHg for 120 s, a return to baseline for 120 s, an abrupt decrease to 30 mmHg for 60 s and a gradual increase to 50 mmHg for 300 s, and a return to baseline for 120 s (Figure 1(a)). Throughout the sequence, end-tidal pressure of O2 was maintained at around 110 mmHg.
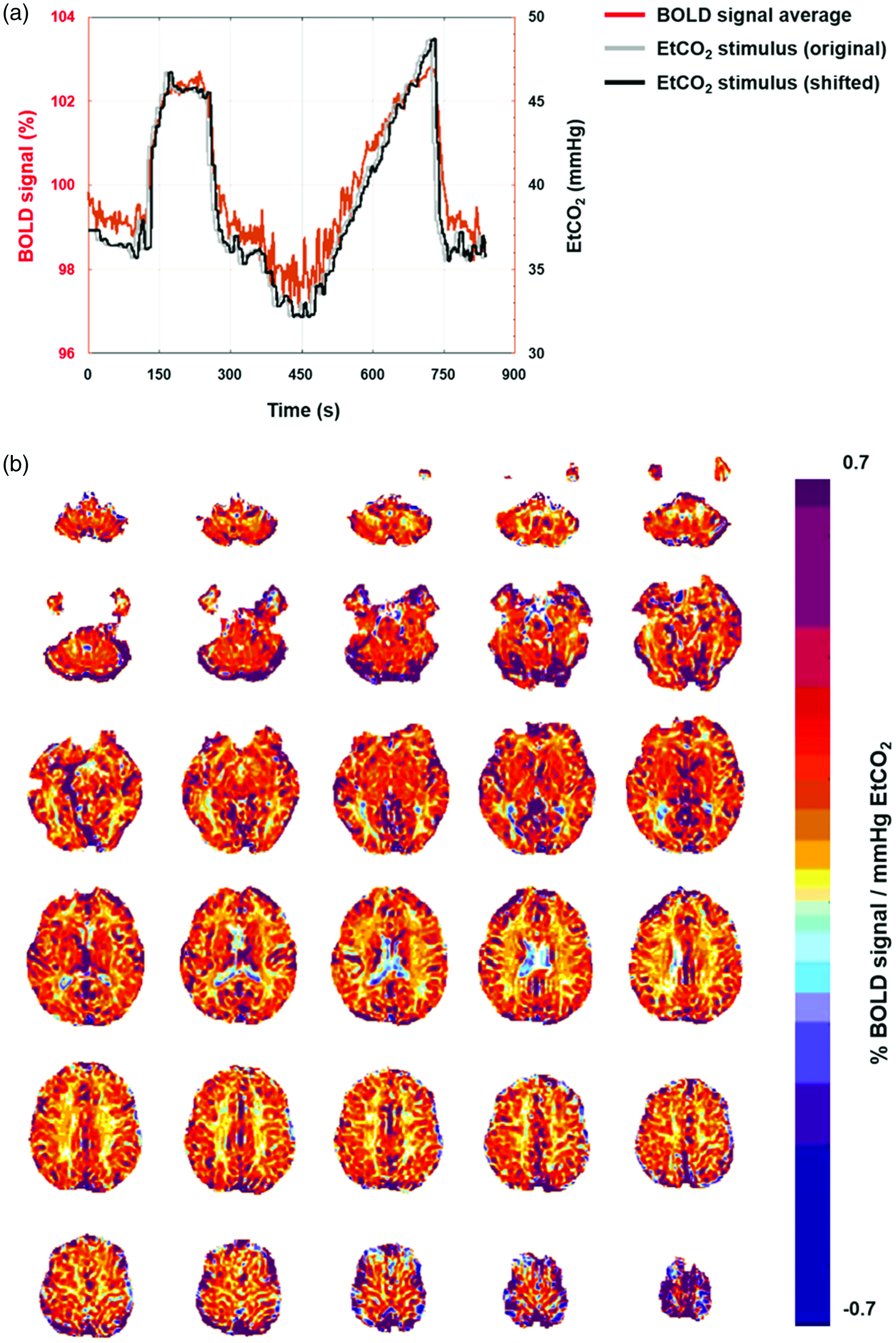
Cerebrovascular reactivity (CVR) map construction of representative patient and control subjects. (a) A plot of average BOLD signals (red), and original (grey) and time shifted EtCO2 (black) stimuli and (b) Estimated CVR maps of the whole brain.
Construction of BOLD-CVR map
The minimal preprocessing of BOLD MRI was performed using the FuNP (fusion of neuroimaging preprocessing) software that integrated AFNI and FSL. 14 –16 The steps include re-orientation, slice timing and head motion correction, skull stripping, and intensity normalization. We did not perform spatial smoothing not to distort CVR values in small deep WMHs as well as in the border between gray matter and white. CVR maps were constructed from the preprocessed BOLD MRI data using AFNI 15 by following established procedures previously published. 10,17 EtCO2 stimulus was shifted in time to have the maximum correlation with averaged BOLD signal across the whole brain. Then, a voxel-wise linear, least-squares regression was performed to fit the averaged BOLD signal to the adjusted EtCO2 stimulus. We followed a previously published formula: 10 Averaged brain signal(t) = CVR × EtCO2 + e(t), where CVR is the slope, e(t) residual time series (i.e., intercept term). The fitted regression coefficient for each voxel was considered as CVR, which is interpreted as the change of mean BOLD signal per EtCO2 (%/mmHg) (Figure 1(b)). We additionally constructed CVR maps by aligning EtCO2 stimulus with averaged BOLD signal within the gray matter to assess the consistency of our results.
WMH quantification
We quantified deep WMHs using the DEWS (DEep White matter hyperintensity Segmentation) tool, a fully automated machine learning-based pipeline developed for the detection of deep WMHs in non-elderly patients with migraine. 11 Using DEWS, contiguous WMH voxels of oval or round shape are considered a distinct WMH, thus the number and volume of separate deep WMH regions can be assessed as well as those of whole deep WMHs. Its rationale and details have been published previously. 11 All the WMH segmentation results were reviewed and manually corrected by a single investigator (MJL) to ensure accuracy. To compare CVR with WMHs and normal-appearing white matter (NAWM), we co-registered T1-weighted data onto the BOLD data with affine transformation and the inverse transform was applied to the CVR map to transform it to the high-resolution FLAIR space (Figure 2) not to discard small deep WMHs (Supplementary Fig 1). As the NAWM regions are relatively large compared to WMH regions, we additionally calculated CVR values within NAWM regions by randomly selecting sub-NAWM regions whose sizes were similar to deep WMHs to assess robustness. We repeated this process 1,000 times for each subject and assessed median and IQR of the CVR values in the selected NAWM regions.
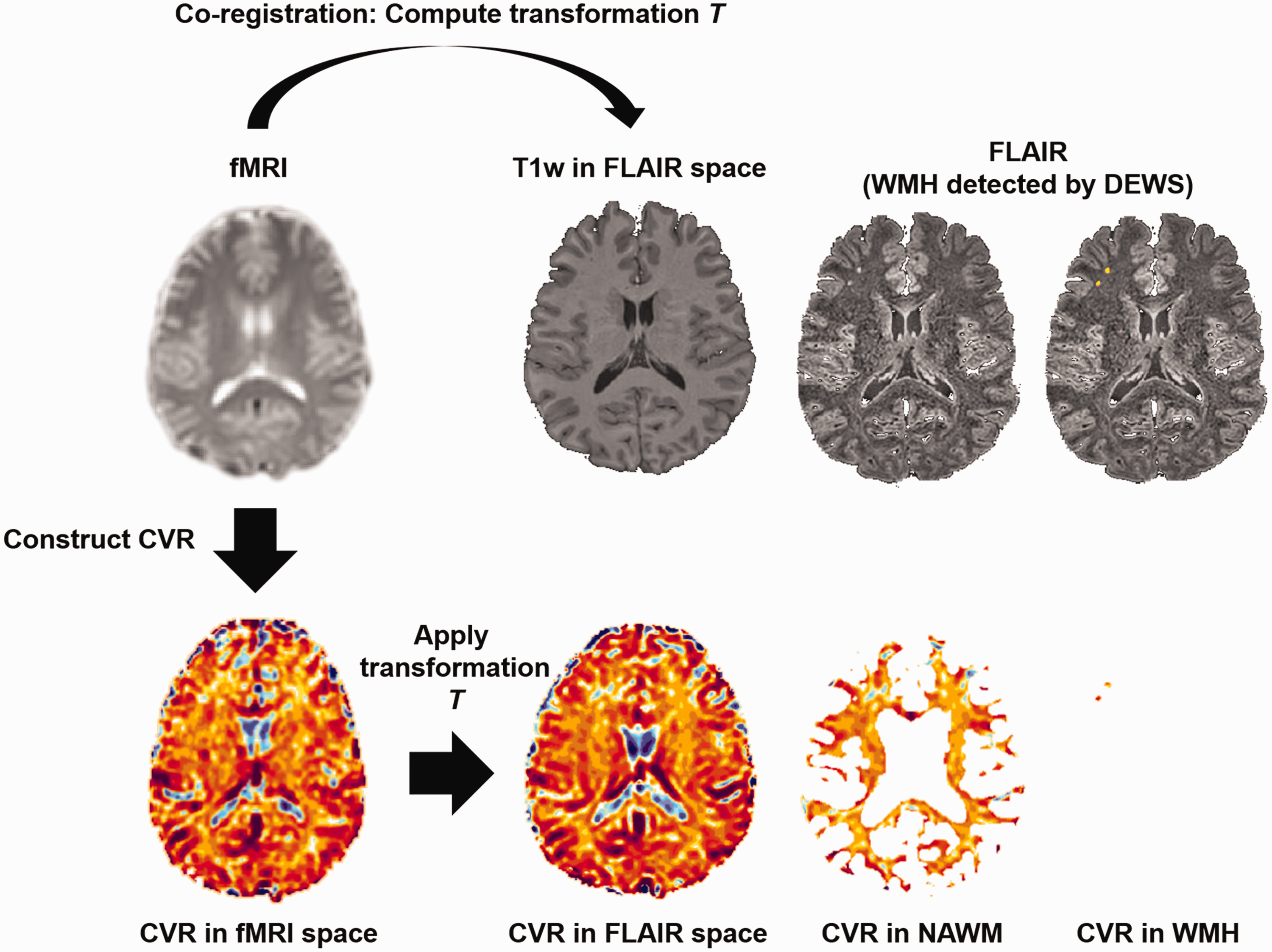
Cerebrovascular reactivity (CVR) for white matter hyperintensities (WMHs) and normal-appearing white matter (NAWM). T1-weighted data in the FLAIR space was registered to the fMRI data and the inverse transform was applied to transform CVR to the FLAIR space. In the FLAIR space, CVR was quantified with respect to the WMHs and NAWM.
Statistical analysis
Sample size was estimated based on the previous study results of the CVR difference of 0.03 between WMH and NAWM and the assumption of moderate correlation (i.e. correlation coefficient of at least 0.4) between CVRs of WMH and NAWM. 18 Using this estimate, a sample size of 24 subjects having WMH and NAWM would have provided a statistical power of 80% at the 5% (two-sided) level of significance.
We performed the Mann-Whitney test to compare the number of deep WMHs and mean CVR of the whole white matter between patients and controls. Then, CVR was compared between WMHs and NAWM, respectively, in patients and controls using the Wilcoxon signed-rank test. Using a full dataset of voxel-wise CVR and WMHs (yes or no), we performed the mixed-effects logistic regression model with a random subject effect to test if the CVR was associated with greater odds of having WMH in the corresponding voxel. Age, sex, and disease condition (migraine vs. control) were also tested and adjusted for. The synergistic effect between CVR and migraine was tested by entering the interaction term (Group [migraine vs. control] ×CVR) into the multivariable mixed-effects logistic regression model with a random subject effect.
Data are presented as the mean (SD) or median (interquartile range [IQR]), odds ratio (OR) (95% CI), or number (%). All analyses were performed using Stata 15.0 (StataCorp. 2017. Stata Statistical Software: Release 15; StataCorp LLC, College Station, TX, USA) and Matlab 2017 b (Mathworks Inc., Natick, MA, USA). Results with a two-tailed p-value <0.05 were considered statistically significant.
Results
Study participants
A total of 31 patients and 31 age-sex-matched controls were recruited. The demographics and characteristics of the participants are summarized in Table 1.
Demographic information of the study participants.
White matter hyperintensity
Deep WMHs profile of patients and controls are presented in Table 2. Deep WMH prevalence was higher in patients than in controls (64.5% vs. 38.7%, p = 0.042). Patients with migraine had a greater number of deep WMHs than controls (p = 0.015). The volume of deep WMHs was not significantly different between patients and controls (p = 0.302). Distribution of number and volume of deep WMHs in patients and controls are shown in Supplementary Figure 2.
Deep WMH and CVR comparison between patients and controls.
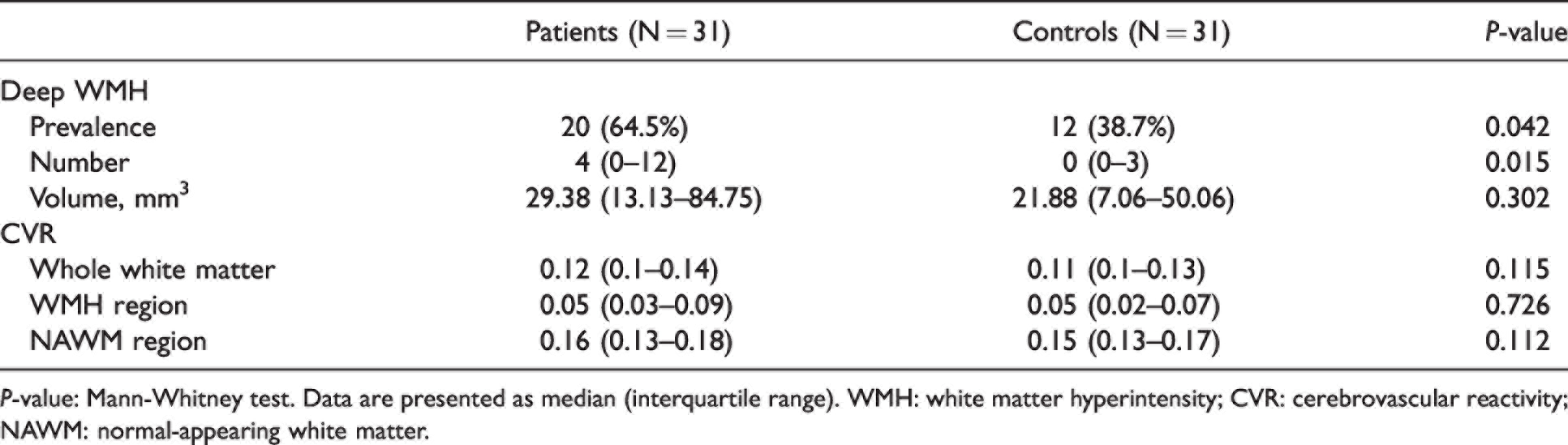
P-value: Mann-Whitney test. Data are presented as median (interquartile range). WMH: white matter hyperintensity; CVR: cerebrovascular reactivity; NAWM: normal-appearing white matter.
CVR comparison
CVR of the white matter, WMH, and NAWM are summarized in Table 2. A representative FLAIR and CVR images are shown in Figure 3. There were no differences in median CVR of the whole white matter (p = 0.115), WMH regions (p = 0.726), and NAWM regions (p = 0.112) between patients and controls. The CVR of WMH was lower than that of NAWM in both patients (median 0.05 [IQR 0.03–0.09] vs. 0.16 [0.13–0.18], p < 0.001) and controls (0.05 [IQR 0.02–0.07] vs. 0.15 [0.13–0.17], p = 0.012). Comparing CVR maps constructed based on averaged brain signals across the whole brain and those within gray matter showed highly consistent results (mean ± SD linear correlation r = 0.99 ± 0.006). In addition, the shifted TRs during alignment did not differ between the whole brain and gray matter-based signals (3.88 ± 1.57 for the whole-brain, 3.64 ± 1.57 for gray matter; p = 0.347), indicating robustness. Randomly selecting NAWM subregions whose sizes were similar to deep WMHs, we could find consistent results of CVR values (median 0.15 (IQR 0.12–0.16) for patients, 0.15 (0.12–0.17) for controls).
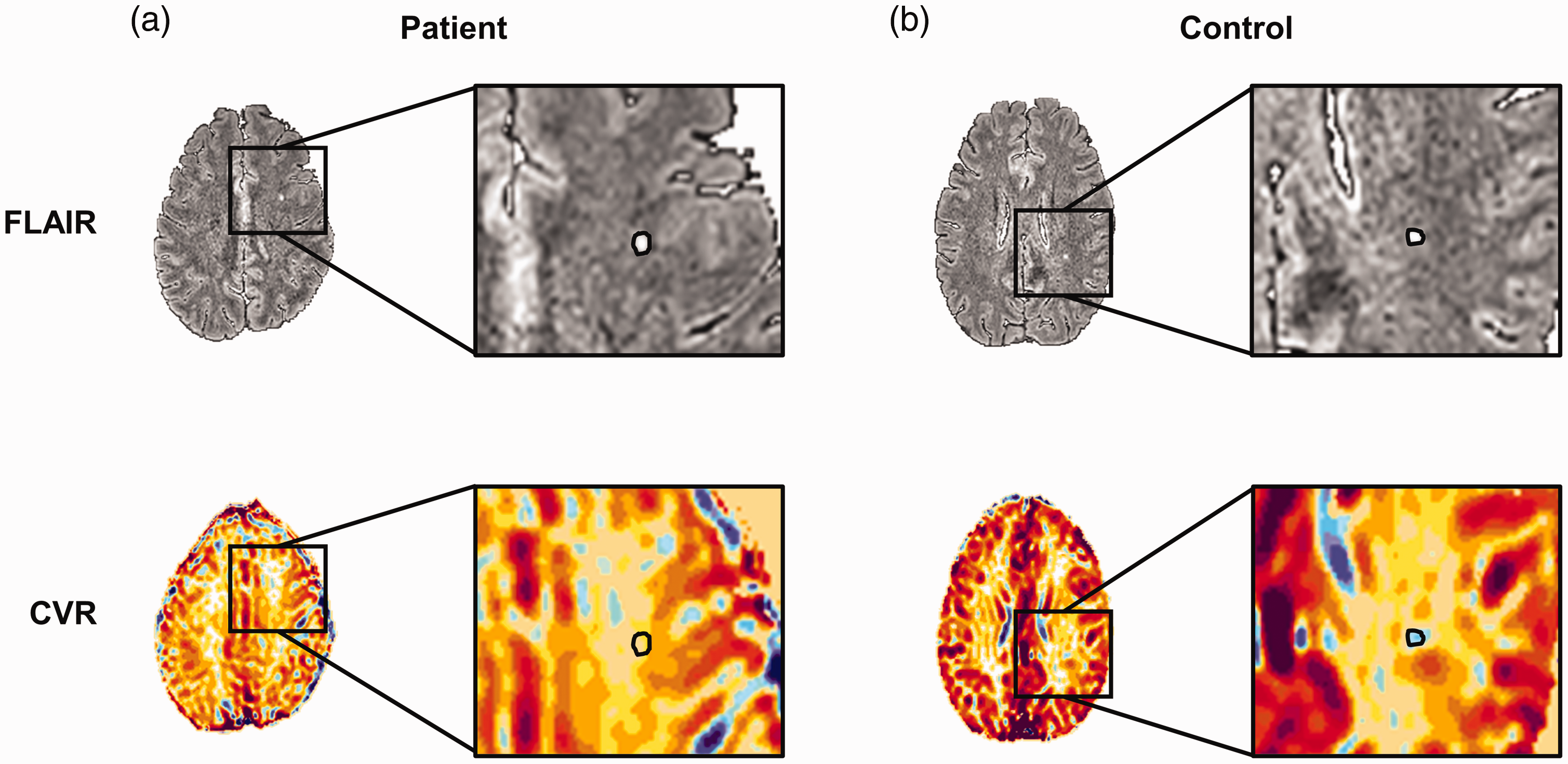
Representative maps of (top) FLAIR and (bottom) CVR of (a) a single patient and (b) control with deep WMHs marked with black circles.
Factors associated with WMH
Results from the analysis using the generalized linear mixed model are summarized in Table 3. In both univariable and multivariable models, migraine was significantly associated with an increased probability of deep WMH in a given voxel (adjusted OR 30.78, 95% CI 1.89–500.53, p = 0.016). Reduced CVR was associated with higher probability of deep WMH (adjusted OR 0.30, 95% CI 0.29–0.32, p < 0.001). Age was associated with presence of deep WMH (adjusted OR 1.26, 95% CI = 1.05–1.50, p = 0.012).
Univariable and multivariable analyses on the presence of deep white matter hyperintensity in a given voxel.
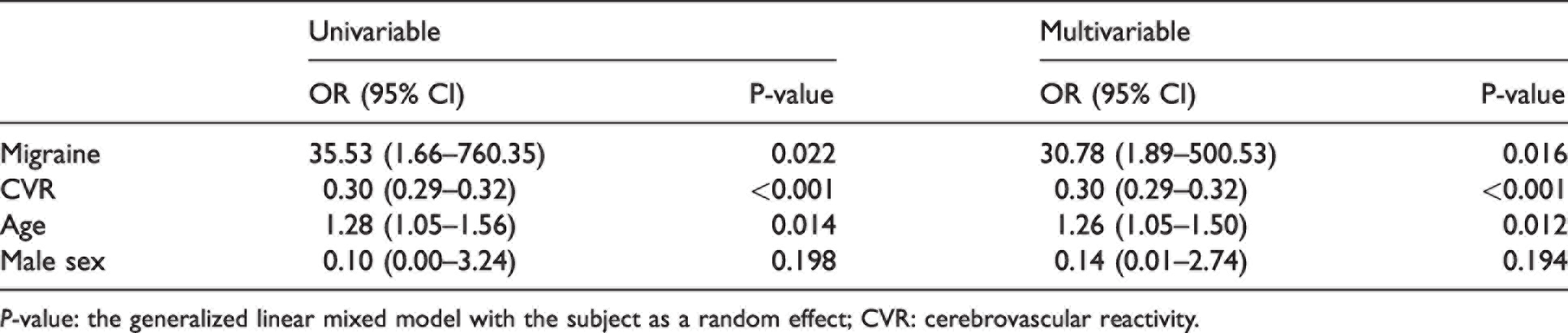
P-value: the generalized linear mixed model with the subject as a random effect; CVR: cerebrovascular reactivity.
Synergistic effect between migraine and CVR
As the patients and controls showed no difference in the mean CVR, but the numbers of WMHs were higher in patients, the synergistic effect of CVR and migraine was tested. The interaction between migraine and CVR was significant (p for interaction <0.001). When stratified by the presence of migraine, the association between CVR reduction and risk of WMH was greater in patients with migraine (OR 0.31, 95% CI = 0.30–0.33) than in migraine-free controls (OR 0.22, 95% CI 0.19–0.27) (Figure 4).
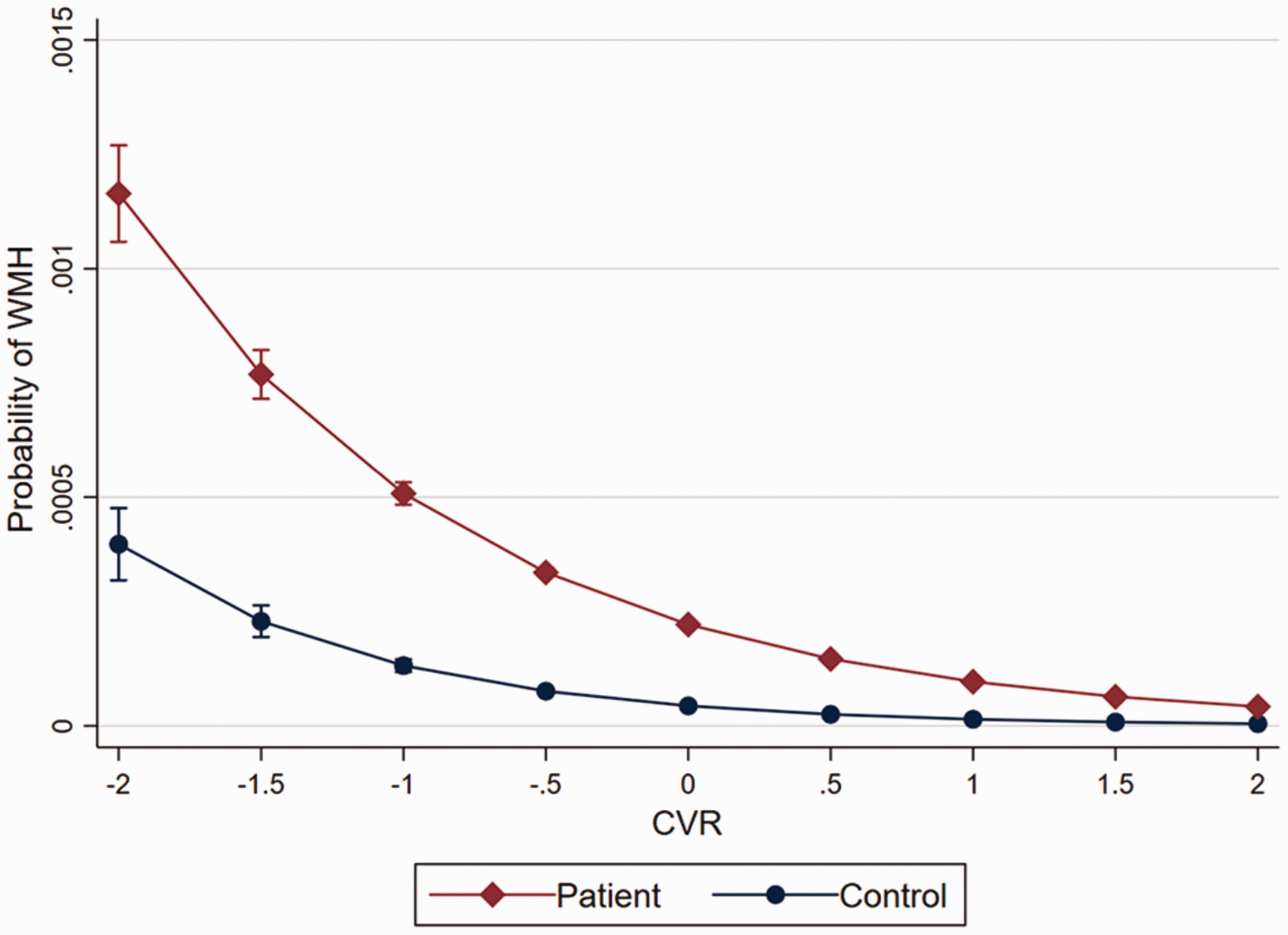
The probability of white matter hyperintensity (WMH) according to cerebrovascular reactivity (CVR) in patients and controls.
Discussion
In this study, we investigated voxel-wise CVR in the whole white matter using the prospective CO2 targeting method in patients with episodic migraine without aura. Our results showed that 1) patients had a greater prevalence and number of deep WMHs than in controls; 2) CVR of the white matter, WMH, and NAWM was not different between patients with migraine and matched controls; 3) migraine, reduced CVR, and advanced age were significantly associated with a possibility of having WMH in a given voxel; and 4) the effect of reduced CVR on the development of WMH was greater in patients with migraine than in controls.
Deep WMHs in migraine
To date, several studies have been conducted regarding the relationship between migraine and WMH. It has been debated whether patients with migraine have a higher prevalence and burden of WMHs compared to normal controls. However, as the prevalence of WMHs increases with age and comorbidities, the migraine-attributed increase in WMHs, if present, can be obscured by age-related changes. 19,20 Therefore, age and comorbidities should be considered in interpreting study results on WMHs in migraine. In addition, methodological parameters for the detection of WMHs, such as MRI resolution, definition of WMH, and manual counting vs. automated segmentation, can affect the results particularly in younger patients with very early stage of WMH development. 21 The type of WMH (deep vs. periventricular) should be also considered because they have different pathophysiologies and risk factors. 22 –25
Several clinic-based studies have reported a high prevalence of WMHs in patients with migraine. Most studies reported that deep WMH is the predominant type of WMH in patients with migraine, 20 and recent studies using 3T with a slice thickness of <3 mm reported a high prevalence of WMHs of about 70% in young (aged <50 years) migraineurs without vascular risk factors. 6,26 In particular, deep WMH was the predominant type of WMHs in patients aged <50 years and without vascular risk factors. 26 –29 Population-based studies showed conflicting results on the association between WMH and migraine. 1 –4,30,31 In these studies generally, participants were older and scan resolution was lower than in clinic-based studies and some did not separate deep WMHs from periventricular WMHs. In some population studies where results were positive, participants were relatively younger and a significant association was found specifically in deep WMH. 1 –4
At present, the mechanism of deep WMH development in patients with migraine is unclear as there is only a paucity of pathophysiology studies looking at differences between deep and periventricular WMHs. One autopsy study has shown that deep WMHs are more associated with markers of hypoxia and ischemia than periventricular WMHs. 32 However, conventional vascular risk factors are less associated with deep WMHs and more associated with periventricular WMHs in normal population or other disease groups. 22 –25 In migraine perspective, previously reported risk factors for WMH in patients with migraine were female sex, 1,3 migraine with aura, 2 migraine without aura, 3 migraine frequency, 1,33 and right-to-left shunt. 34 However, these results have not been replicated in other studies, 3,35 and some studies have shown conflicting results. 2,3 Our recent study showed reduced CVR was associated with higher deep WMH burden in patients with migraine, while such association was not found in normal controls, suggesting a role of CVR in deep WMH development in migraine. 6
Effect of CVR on WMH: current study findings
Our study showed no difference in mean CVR between patients with migraine and normal controls. Previous studies using transcranial Doppler showed controversial results regarding CVR impairment in patients with migraine. 36 –38 We hypothesized that impaired CVR is a feasible explanation for migraine-associated WMHs, as was evidenced in healthy elderlies 18 as well as our previous study in non-elderly patients with migraine. 6 However, the overall CVR of the whole white matter and NAWM were similar between patients with migraine and normal controls in the present study. Migraine itself, reduced CVR, and advanced age were all independent factors of WMH.
Nevertheless, our study showed that the effect of CVR on WMH was different between patients with migraine and normal controls. This effect modification can be explained by features of the migraine brain: hyperexcitability and spreading depolarization. Migraine brain has unique features of hyperexcitability and exaggerated sensory processing, 39 –42 which may result in an increase in cerebral blood flow due to increased metabolic demand. 43,44 Consequently, arterioles dilate to some degree at baseline, which prevents further dilatation in response to chemical stimuli (reduced vascular reservoir). This may explain vulnerability of migraine brain to ischemic insult and serve as a hypothetical background for the greater effect of reduced CVR in the development of WMH in patients with migraine. 45 In addition, spreading depolarization can cause more structural damage during ischemia, as evidenced in animal and human studies. 46,47
Patent foramen ovale (PFO) can also contribute the development of WMH. PFO has been reported as more prevalent in patients with migraine at least in several clinic-based studies and theoretically can be a source of deep WMH. 48,49 However, we did not test PFO for this study as the association between PFO and migraine without aura is less robust than in migraine with aura, and a previous study failed to document the association between PFO and WMH in patients with migraine. 50 Another potential confounding factor is the use of triptans. A small proportion of our patients took acute headache medications including triptans the day before the MRI scan. This may influence CVR with a negative direction. Nevertheless, CVR of patients were not reduced compared to controls in our study.
Effect of CVR on WMH: comparison to previous study findings
Our previous study showed an association between deep WMH burden and reduced CVR in patients with migraine. 6 The previous study had the limitations as stated in the Introduction section. The present study is the first to adopt the state-of-the-art gold standard of CVR measurement in the field of migraine research: BOLD MRI with the prospective CO2 targeting method. The rationale of BOLD MRI with the prospective CO2 targeting method to measure CVR has been published in several papers. 10,13,51 Although CVR is smaller in white matter, an adequate and sustained increase in CO2 using a prospective CO2 targeting method leads to detectable increase in white matter BOLD signal, and a linear correlation between PETCO2 and BOLD signal was well documented in white matter. 9 Using this method, the existence of CVR in white matter has been well validated not only in elderly or diseased population 18,52,53 but also in young healthy individuals in several other studies. 54,55 BOLD MRI has an excellent spatial and temporal resolution among other recording modalities such as transcranial Doppler, near-infrared spectroscopy, and single photon emission computed tomography, 7,9 and has superior signal-to-noise ratio compared with cerebral blood flow MRI. 56 This method allows the measurement of CVR in any region of the brain and the direct voxel-wise analysis between structural changes and CVR in the corresponding area. The prospective CO2 targeting method can guarantee the accurate delivery of vasodilatory stimuli by monitoring and controlling the EtCO2 levels. The prospective CO2 (actually O2 as well) targeting method eliminates the need for a feedback correction mechanism, reducing the computational complexity and minimizing the breath-to-breath variability of EtCO2. 8 It is operated using a sequential gas delivery circuit which reduces alveolar inhomogeneities and the gradient between EtCO2 and PaCO2. 57
In the present study, we demonstrated an association between CVR and WMH in both patients and normal controls, an increased association between migraine and WMH, and enhanced association between CVR and WMH in patients with migraine. However, since the current study was cross-sectional, we cannot determine if the enhanced association between CVR and WMH in migraine is the cause of WMH. We are performing a longitudinal study to test the causal relationship between CVR and WMH in patients with migraine and normal controls (ClincalTrials.gov Identifier: NCT03494673).
Strengths and limitations
Our study has several strengths. Firstly, this is the first study measuring CVR and its association with WMH using the prospective CO2 targeting method and BOLD MRI in the field of migraine research. This method has been validated in several studies and its safety has been proven. 9,13 Secondly, the 3T and 0.5 mm iso-voxel size of 3D FLAIR data, which provides higher spatial resolution, was used to better detect deep WMHs than in previous studies. 21 Thirdly, deep WMHs were segmented by the method developed and validated for the purpose of deep WMH research in non-elderly patients with migraine, 11 which showed a superior performance in detecting small cortical deep WMHs compared to previous WMH segmentation techniques, which were validated among demented patients. Limitations of the present study included a relatively small sample size and a cross-sectional design, due to which determination of a causal relationship was not possible. However, we expect that our ongoing longitudinal analysis, using follow-up imaging of these study participants, will shed further light on causal factors (ClincalTrials.gov Identifier: NCT03494673). Another limitation is that we only segmented deep WMHs. Periventricular WMHs, if present, might have been classified as NAWM. When we reviewed all the scans, however, periventricular WMHs were not seen as our study subjects were young and without vascular risk factors. In addition, this study only included patients with migraine without aura. A comparison study between migraine with aura and migraine without aura is ongoing in our group. And the included patients mostly had low frequency episodic migraine. It is unclear if patients with high-frequency or chronic migraine would have yielded a different result. Lastly, we registered CVR maps with 4 mm slice thickness onto the FLAIR space with 0.5 mm thickness not to lose small deep WMHs. Thus, careful interpretations are required due to the possible errors attributed to interpolation effects. In future studies, we will collect high-resolution BOLD data so that we can precisely construct CVR maps in small deep WMH regions.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221103006 - Supplemental material for Cerebrovascular reactivity and deep white matter hyperintensities in migraine: A prospective CO2 targeting study
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221103006 for Cerebrovascular reactivity and deep white matter hyperintensities in migraine: A prospective CO2 targeting study by Mi Ji Lee, Bo-yong Park, Soohyun Cho, Seonwoo Kim, Hyunjin Park, Sung Tae Kim and Chin-Sang Chung in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Research Foundation of Korea (NRF) grants funded by the Korean government (MSIP) (Nos. 2017R1A2B2009086, 2020R1A2B5B01001826 and 2021R1F1A1052303), Institute for Information and Communications Technology Planning and Evaluation (IITP) funded by the Korea Government (MSIT) (2022-0-00448, Deep Total Recall; 2020-0-01389, Artificial Intelligence Convergence Research Center (Inha University); RS-2022-00155915, Artificial Intelligence Convergence Innovation Human Resources Development (Inha University); 2021-0-02068, Artificial Intelligence Innovation Hub; 2022-0-00448), and Institute for Basic Science (IBS-R015-D1).
Acknowledgements
We thank Ms. Sujin Moon and Ms. Jinju Lee for managing the project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Study conceptualization and design: MJL, ByP, HP, STK, C-SC. Acquisition and analysis/interpretation of data: MJL, ByP, SC, SK, HP, STK, C-SC. Drafting of manuscript: MJL, ByP. Manuscript revision: MJL, ByP, SK, HP, STK, C-SC.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
