Abstract
The recent discovery of the meningeal lymphatic vessels (mLVs) and glymphatic pathways has challenged the long-lasting dogma that the central nervous system (CNS) lacks a lymphatic system and therefore does not interact with peripheral immunity. This discovery has reshaped our understanding of mechanisms underlying CNS drainage. Under normal conditions, a close connection between mLVs and the glymphatic system enables metabolic waste removal, immune cell trafficking, and CNS immune surveillance. Dysfunction of the glymphatic-mLV system can lead to toxic protein accumulation in the brain, and it contributes to the development of a series of neurodegenerative disorders, such as Alzheimer’s and Parkinson's diseases. The identification of precise cerebral transport routes is based mainly on indirect, invasive imaging of animals, and the results cannot always be applied to humans. Here we review the functions of the glymphatic-mLV system and evidence for its involvement in some CNS diseases. We focus on emerging noninvasive imaging techniques to evaluate the human glymphatic-mLV system and their potential for preclinical diagnosis and prevention of neurodegenerative diseases. Potential strategies that target the glymphatic-mLV system in order to treat and prevent neurological disorders are also discussed.
Introduction
Lymphatic vessels are considered a crucial auxiliary part of the blood circulatory system, responsible for the cleanup of interstitial fluid (ISF), constituent proteins, and other solutes not absorbed across postcapillary venules. By shuttling antigens and immune cells from peripheral tissues to lymph nodes, lymphatic vessels serve as key components of the body's adaptive immune response.1,2
The brain was thought for a long time to lack a lymphatic system, with the extracellular fluid of the brain parenchyma considered stagnant and functionally separated from the cerebrospinal fluid (CSF). 3 However, already in 1787, Mascagni provided the first evidence that lymphatics might be present in the brain. 3 Several studies in recent decades have documented the existence of CNS lymphatics, convective clearance and perivascular spaces.4 –8 Nevertheless, the scientific community has questioned the existence of a CNS “lymphatic” system because of the lack of definitive markers and imaging technologies to visualize it.
In real-time imaging studies of rodents and large mammals using two-photon laser scanning and confocal microscopy, researchers found that fluorescent tracers that have been injected intracisternally or intracerebroventricularly flow through the perivascular space (PVS), mix with the ISF, and eventually drain into the cervical lymph nodes (CLNs) through mLVs.9 –12 Mammalian brain gyrification was favorable for CSF dispersion throughout the cortical surface and the PVS influx density was 4-fold larger in the pig brain than in mice. 12 State-of-the-art imaging techniques, especially emerging noninvasive techniques that can be applied to humans, have led to the rediscovery of mLVs and the glymphatic system in the CNS, changing our understanding of fluid dynamics and brain immunity, as well as offering solid evidence of their physiological functions.
Currently, four brain lymphatic pathways have been identified: the perivascular pathway, glymphatic system, mLVs, and olfactory/cervical lymphatic drainage route. 13 In these pathways, fresh CSF flows into the brain parenchyma along arterial perivascular spaces, where it is exchanged with the extracellular fluid. Then, the mixed CSF-extracellular fluid flows towards the venous perivascular spaces and perineuronal spaces, clearing waste products and excess fluid into meningeal and cervical lymphatic vessels.9,10,14 These drainage systems also transport brain antigens and immune cells, providing a direct route for immune-related communication between the CNS and peripheral immune system. 15 These discoveries have provided novel insights into the pathology behind common neurodegenerative disorders such as Alzheimer’s disease, Parkinson's disease, and multiple sclerosis, which are driven by uncleared toxic proteins and overactive immune cells.15 –17
In this review, we describe the physiological functions of the glymphatic-mLV system and discuss evidence linking it to CNS diseases. We focus on emerging noninvasive imaging techniques for evaluating the human glymphatic-mLV system and their potential for diagnosis and prevention of neurodegenerative diseases. Finally, potential strategies that target the glymphatic-mLV system in order to treat and prevent neurological disorders are discussed.
Structure, characteristic, and organization of mLVs
Similarly to peripheral lymphatics, mLVs express all the classical lymphatic endothelial cell markers, such as vascular endothelial growth factor receptor 3, Prospero homeobox protein 1, podoplanin, lymphatic vessel endothelial hyaluronan receptor 1, CD31, and chemokine C-C motif ligand 21.9,14
Interestingly, mLVs at different anatomical sites tend to differ in structure and function. Dorsal mLVs run parallel to the dural sinuses, especially the superior sagittal and transverse sinuses, and are composed of a marginally ramified network of thin-walled vessels and loose, button-like lymphatic endothelial cell junctions.9,14 Dorsal mLVs can drain both intracerebral macromolecules and immune cells from the subarachnoid space, converge with the skull base lymphatics, and then exit the cranium into the cervical lymphatic system. 14 In contrast, mLVs from the lateral or basal parts of the skull are located around the foramina, they possess valves, and they show a mixture of button- and zipper-like junctional patterns. 11 These specialized morphologies make them the preferential route for CSF uptake and drainage into the deep cervical lymph nodes (dCLNs) and partially to the superficial cervical lymph nodes (sCLNs).9,11,14,15
In two-photon imaging studies of anesthetized mice, CSF tracers entered along the para-arterial space of the cortex and subsequently appeared in the para-venous space; this ISF/CSF flow occurred in the organized glymphatic system. 10 A functional glymphatic fluid transport system consists of three components:10,18 –20 (1) afferent flow of fresh CSF into the parenchyma along the arterial PVS; (2) mixing and exchange of new CSF with extracellular fluid (ISF), and (3) efferent flow of the mixed CSF/ISF along the venous PVS (Figure 1).
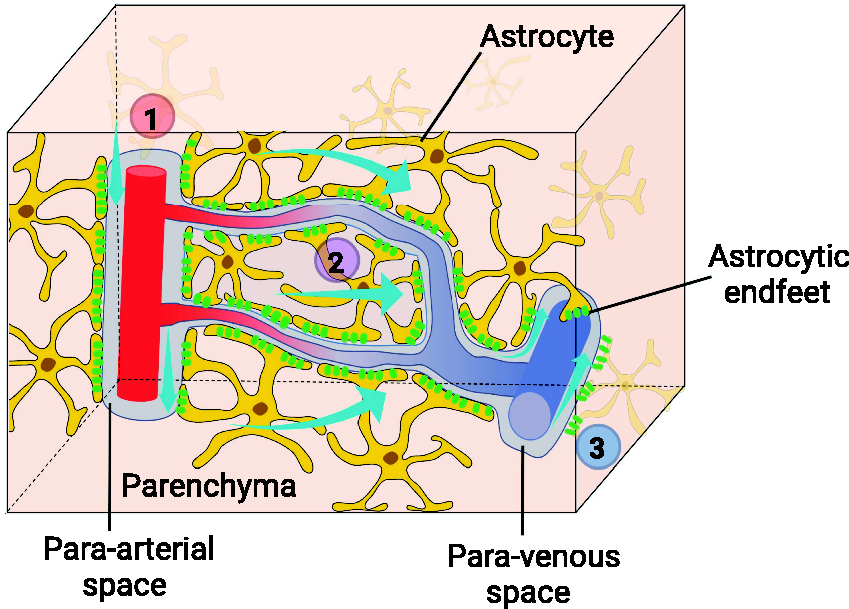
Schematic representation of the glymphatic pathway. A functional glymphatic pathway consists of three consecutive components: (1) afferent perfusion of CSF into the parenchyma along the para-arterial space, (2) mixing and exchange of new CSF with extracellular fluid, and (3) efferent flow of the mixed CSF/ISF along the para-venous space. The entire process is facilitated by AQP4 water channels expressed on the astrocytic endfeet. The figure was created using online BioRender software (BioRender.com). AQP4, aquaporin-4; CSF, cerebrospinal fluid; ISF, interstitial fluid.
The entire transport process is facilitated by aquaporin-4 (AQP4) water channels expressed on the astrocytic endfeet. The morphology and function of astrocytes and the polarized AQP4 on endfeet differ between humans and mice. Human astrocytes are larger, their arborization is more complex, and their endfeet completely envelop the vessels, forming a tighter, denser astrocytic layer than in mice. 21 Human astrocytes have a distinctly lower level of polarization, while mice have 5–10 times more AQP4 localized in the membranes of endfeet. 22 Grafting human glial progenitor cells into mice can increase their performance in certain cognitive tasks, which demonstrates that astrocytes may support our unique cognitive abilities. 23
The driving force for this fluid transport system is still under debate. The best developed theory is that CSF flow is driven by perivascular pumping due to pulsation of the arterial wall.24 –26 According to the glymphatic concept, arterial pulsation through AQP4 water channels pushes CSF from the subarachnoid or Virchow-Robin space into the brain parenchyma. AQP4 coats and fully envelops the vasculature, and it creates a perivascular space for facilitating CSF movement.10,19 The bulk flow of CSF then drains via three pathways: (1) it flows back into the subarachnoid and joins the CSF, part of which is finally drained by mLVs; (2) it joins the venous flow and enters the bloodstream; and (3) it remains in the para-venous space and finally reaches the cervical lymph nodes. Tracers injected into the CSF of adult mice are detectable in para-arterial spaces within 10 minutes of injection and reach the para-venous spaces more than 30 minutes later, which reveals the passage of CSF through the brain parenchyma and from the para-arterial to the para-venous spaces. 10 When the same tracers are injected directly into the parenchyma, they preferentially accumulate in the para-venous spaces, supporting the role of this pathway in the clearance of brain waste. 10 Macroscopic CSF flow measurement with ultra-high field MRI (7 T) also shows that the CSF pulsatility in the cerebral aqueduct is highly correlated with discrete phases of the cardiac cycle, suggesting arterial pulsations are important drivers for CSF circulation. 27
While the glymphatic hypothesis described here currently prevails, it has created controversy and needs to be comprehensively re-evaluated. First, whether CSF flow is driven mainly by arterial pulsation remains a matter of debate, as others have argued that arteriolar pulsations alone are too weak to drive clearance. 28 Second, it is unclear how and to what extent AQP4 water channels facilitate such convective flow pathways under normal conditions.29,30 because (1) convective flow from the arterial PVS to the venule PVS through the neuropil interstitium depends on the osmotic gradient; 31 (2) high hydraulic resistance in the brain extracellular space should greatly restrict such flow, but it can still support diffusion;32,33 and (3) AQP4 is incapable of transporting macromolecules, so tracers may travel through the brain neuropil primarily via size-dependent diffusive transport.34,35 Third, the relationship between diffusion (defined as the movement of solute or fluid from an area of high to low solute concentration) and convection is not entirely clear. 36 Diffusion is thought to dominate the transport of small and large molecules through the brain extracellular space and the neuropil, as well as facilitate CSF-ISF exchange at the capillary level. Even if the precise relative contributions of diffusion and convection are unclear, the combination of convective flow in perivascular spaces and diffusive flow in the brain extracellular space is plausible. 37 In other words, both types of flow most likely coexist in the brain and cooperate in solute clearance.
Characterizing the glymphatic transport system has been limited by the lack of noninvasive techniques: invasive procedures such as opening the skull and inserting a cannula effectively inactivate the transport system. 38 In addition, rigorous understanding of the glymphatic transport system requires taking into account that convective flow through the brain parenchyma is regulated by sleep,18,39 age,40,41 anesthesia,42,43 even time of day.44,45 For example, one study 34 concluded that solute transport in the mouse brain parenchyma is AQP4-independent, which conflicts with current thinking, and the discrepancy may be due to that study’s reliance on an invasive procedure, non-optimal anesthetic regimen, and use of animals at different ages. 37 Therefore, further work is needed to clarify apparent current discrepancies surrounding many aspects of the glymphatic system. In particular, more sophisticated quantitative modeling of such transport is urgently needed and will likely clarify some of these disagreements in the future. 20
Other models have been proposed to account for CSF drainage, such as the intramural periarterial drainage (IPAD) pathway.46,47 In the IPAD pathway, tracers from the CSF enter the brain parenchyma along pial-glial basement membranes on the outer face of cortical arteries, and flow out of the brain along smooth muscle cell basement membranes in the walls of arteries. Cardiac arrest or stroke abruptly initiate a pathological influx of CSF, leading to shrinkage or loss of the fluid-filled perivascular space.26,37,48,49 The IPAD model is based primarily on histological evidence that can be obtained only post mortem and is therefore vulnerable to perfusion artifacts and post mortem CSF movements.37,50 In post mortem studies, observation of CSF tracers in the smooth muscle cell layer and the basement membrane cannot be considered conclusive evidence that these layers act as an efflux pathway for ISF. Two in vivo studies have provided evidence supporting this model.51,52 but the high intracerebral injection volume and breakdown of the blood-brain barrier (BBB) in those experiments likely perturbed flow pathways. Arterial pulsations and vasomotion have been proposed as IPAD drivers,28,53 but the mechanisms underlying IPAD remain to be clarified. As a result, the hypothesis that the IPAD model may serve as an important efflux pathway for brain CSF drainage remains under intense debate.
Physiological functions of the glymphatic-mLV system
Meningeal immunity and CNS immune surveillance
For decades, lymphatic vessels were considered the missing link between the peripheral immune system and the CNS. The rediscovery of mLVs has challenged the traditional concept of CNS immune privilege and also opened the way to current research on the role of meningeal immunity and CNS immune surveillance. 15 Under steady-state conditions, meningeal compartments, especially dural meninges, are naturally populated with a rich diversity of immune cells, especially T cells but also macrophages, dendritic cells, mast cells, neutrophils, and B cells. These cells represent a crucial part of meningeal immunity, acting as the guardians of the CNS. They can detect damage and isolate pathogens before they reach the brain parenchyma (Figure 2).54,55 These immune cells in the dura mater have also been identified in mLVs. Although largely absent from the brain parenchyma, T cells are essential regulators of CNS homeostasis: meningeal T cells release the cytokines interleukin-4 and interferon-γ, which regulate spatial learning, cognitive functions, and social behavior in mice.56 –58
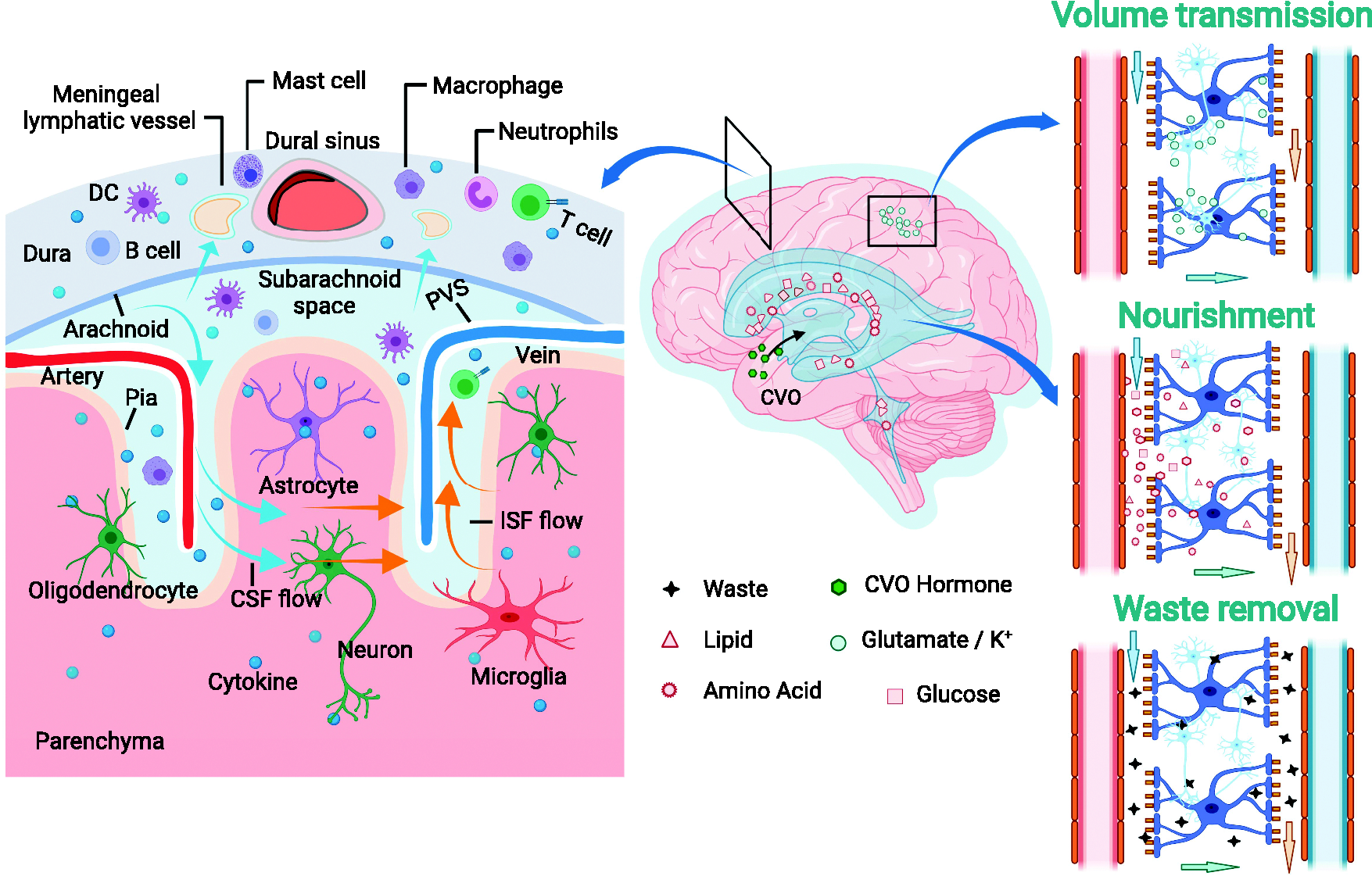
Physiological functions of the glymphatic system. By regulating extracellular fluid influx and efflux, the glymphatic system is involved in transporting immune cells and balancing brain immunity, assisting local volume transmission, transporting nutrients, and clearing extracellular metabolites and waste products from the brain parenchyma. The figure was created using online BioRender software (BioRender.com). CSF, cerebrospinal fluid; CVO, circumventricular organs; DC, dendritic cells; ISF, interstitial fluid; PVS, perivascular space.
Meningeal T cells provide immune surveillance by traveling through the CSF to ‘patrol' CNS-associated 'border' regions and detect pathogenic alterations.59,60 The large number of meningeal macrophages can make them suitable as sentinels for infection and tissue damage during monitoring of the CSF and the extracellular lumen of meningeal blood vessels. 61 When the CSF and ISF exchange in the glymphatic system, astrocytes and macrophages interact, and antigen-presenting cells and CNS-derived antigens move from the glymphatic system into the CSF. 13 The mLVs have been shown to transport T cells and dendritic cells from the CNS to CLNs.9,11,14,15 Thus, mLVs appear to transport immune cells and corresponding antigens from the brain parenchyma and meningeal compartment to the peripheral lymphatic system in a similar manner as peripheral lymphatic vessels transport antigens to the periphery, where antigens are destroyed.14 –16,62 From this perspective, mLVs and the glymphatic system are essential components of CNS immune surveillance, with mLVs playing a key role in activating immune responses (Figure 4(a)). Overall, the glymphatic-mLV system serves as a communicator between the brain and peripheral immunity, which guarantees that the CNS is not ignored by the immune system. 13 Through the glymphatic-mLV system, immune cells and CNS-derived antigens can drain from the brain parenchyma into the CSF and finally into the peripheral immune system.
Molecule clearance from the brain, nutrient delivery, and volume transmission
Removal of excess extracellular fluid, metabolites, and waste products from the brain is crucial to CNS homeostasis. For example, three mechanisms are known to clear amyloid-β protein: glymphatic efflux, local degradation, and export across the BBB (Figure 2).63,64 The BBB and glymphatic system likely rely on partially overlapping mechanisms to play complementary roles: convective flow in the glymphatic system facilitates the BBB transport pathway (Figure 4(b)). 65
The glymphatic system, on its own and in concert with other mechanisms, is a vital mediator of brain clearance.10,66 For example, the inert tracers inulin and mannitol, for which no receptors or BBB transporters are known, can be used to investigate purely glymphatic clearance. 67 Knocking out AQP4 from mice impaired glymphatic efflux, reducing the 30-min clearance of radiolabeled inulin by about 25% and the 2-h clearance of radiolabeled mannitol by about 70%.10,39
Similarly, inhibiting glymphatic function compromises clearance of neurotoxic proteins. Intraventricular injection of antisense oligonucleotides against AQP4 mRNA in a mouse model of Huntington’s disease reduced transport of mutant huntingtin protein into the CSF by 23% and transport into blood plasma by 49%. 68 Blockade of AQP4 reduced tau protein clearance from the brain by 90%, 69 and inhibition of glymphatic efflux aggravated accumulation of amyloid-β.66,70
Apart from contributing to the removal of excess protein and harmful metabolites, glymphatic influx helps deliver nutrients to the brain such as glucose, lactate, amino acids, ions, vitamins and neurotransmitters, 64 in analogy to peripheral lymphatics. For example, when the CSF is driven into the brain along the PVS of penetrating arterioles, it carries essential nutrients such as glucose and amino acids to CNS cells. In another example, the glymphatic system disperses lipids and vitamins such as folate from the choroid plexus throughout the brain. 71
The glymphatic system also plays key roles in brain function by transporting bulk volume, known as volume transmission. Such transmission can act locally, such as when ions or neurotransmitters are released into the adjacent extracellular space, from where they can reach neighboring neurons to exert parasynaptic effects. 72 Volume transmission can also act across the entire brain, 73 such as when the glymphatic system carries neuropeptides from the hypothalamus to many other parts of the brain in order to regulate eating, drinking, body temperature, neuroendocrine release, and internal circadian timing. 38
In these ways, the glymphatic system helps remove metabolic products, transport nutrients, and refresh the extracellular fluid. For the system to clear materials from the brain, the CSF can drain via mLVs and other routes, which are covered below.
Drainage routes essential for CNS homeostasis
The CSF produced by the choroid plexus fills the brain ventricles and the subarachnoid spaces, and it circulates through the drainage system to maintain the fluid homeostasis in the brain and spinal cord. In fact, the total CSF volume in the cranium is renewed about 11 times a day in young adult rats and about four times a day in healthy humans. 74 Therefore, under normal conditions, the efflux of CSF from the CNS closely matches the formation of new CSF, since any excess accumulation of CSF or other fluids could dangerously increase intracranial pressure.
CSF is thought to drain into the extra-cranial circulation via perineuronal sheets along cranial and spinal nerves, dural lymphatics, and arachnoid villi/granulations,50,63 although the relative importance of each drainage route remains under debate (Figure 3). The overall brain drainage system and the brain lymphatic drainage system in particular are depicted in Figure 4(c). CSF is produced mainly by the choroid plexus, it flows through the glymphatic pathway (Figure 1), then drains out of the brain by multiple routes.
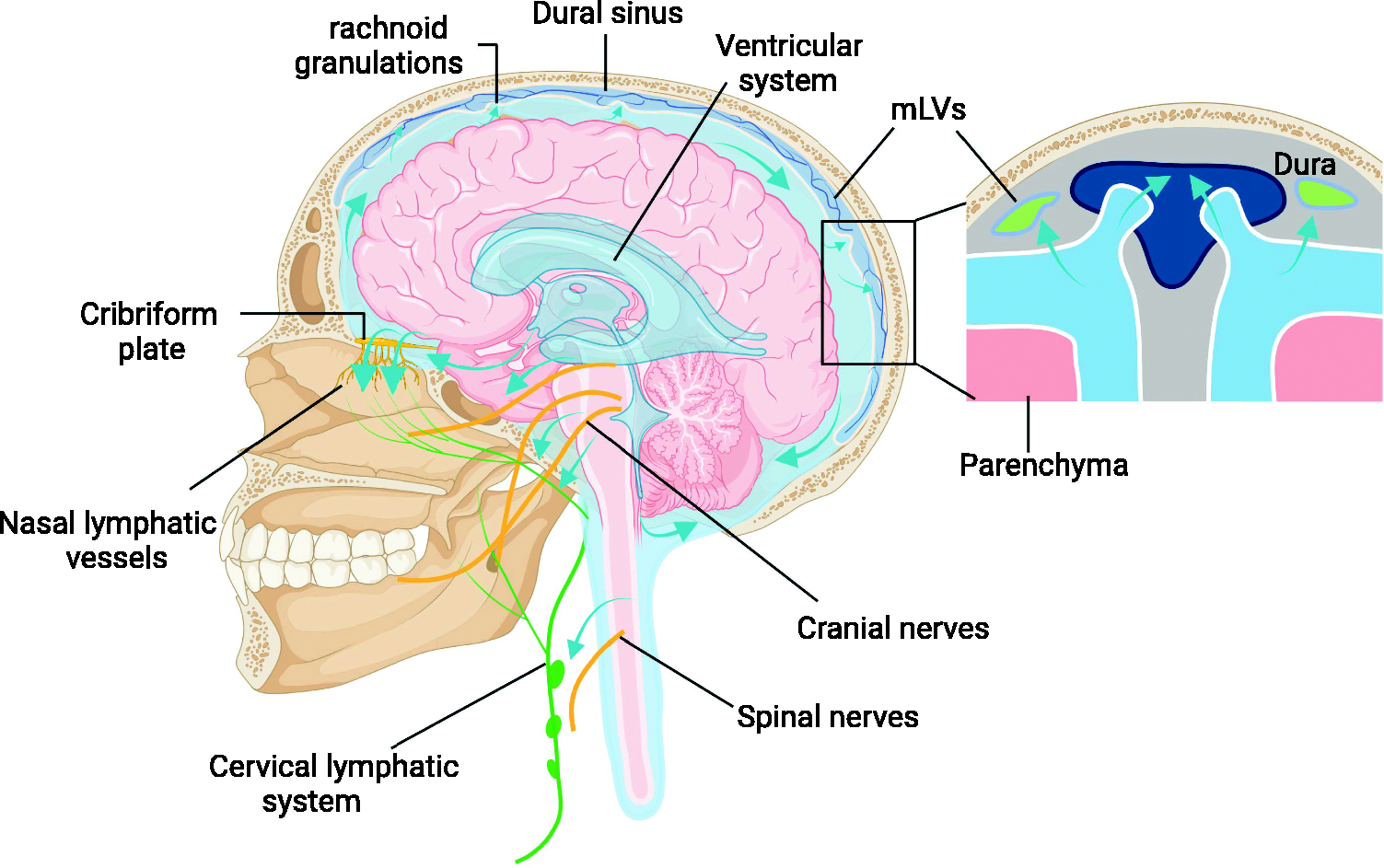
Routes of CSF efflux in humans. Outflow routes of cranial CSF (blue-green arrows) involve perineural sheaths surrounding cranial and spinal nerves, mLVs, and arachnoid villi/granulations. Experimental evidence in rodents and humans strongly supports CSF outflow along the olfactory nerve through the cribriform plate and towards nasal mucosa lymphatic vessels. Finally, the CSF drains into the dCLNs. The figure was created using online BioRender software (BioRender.com). CSF, cerebrospinal fluid; dCLNs, deep cervical lymph nodes; mLVs, meningeal lymphatic vessels.
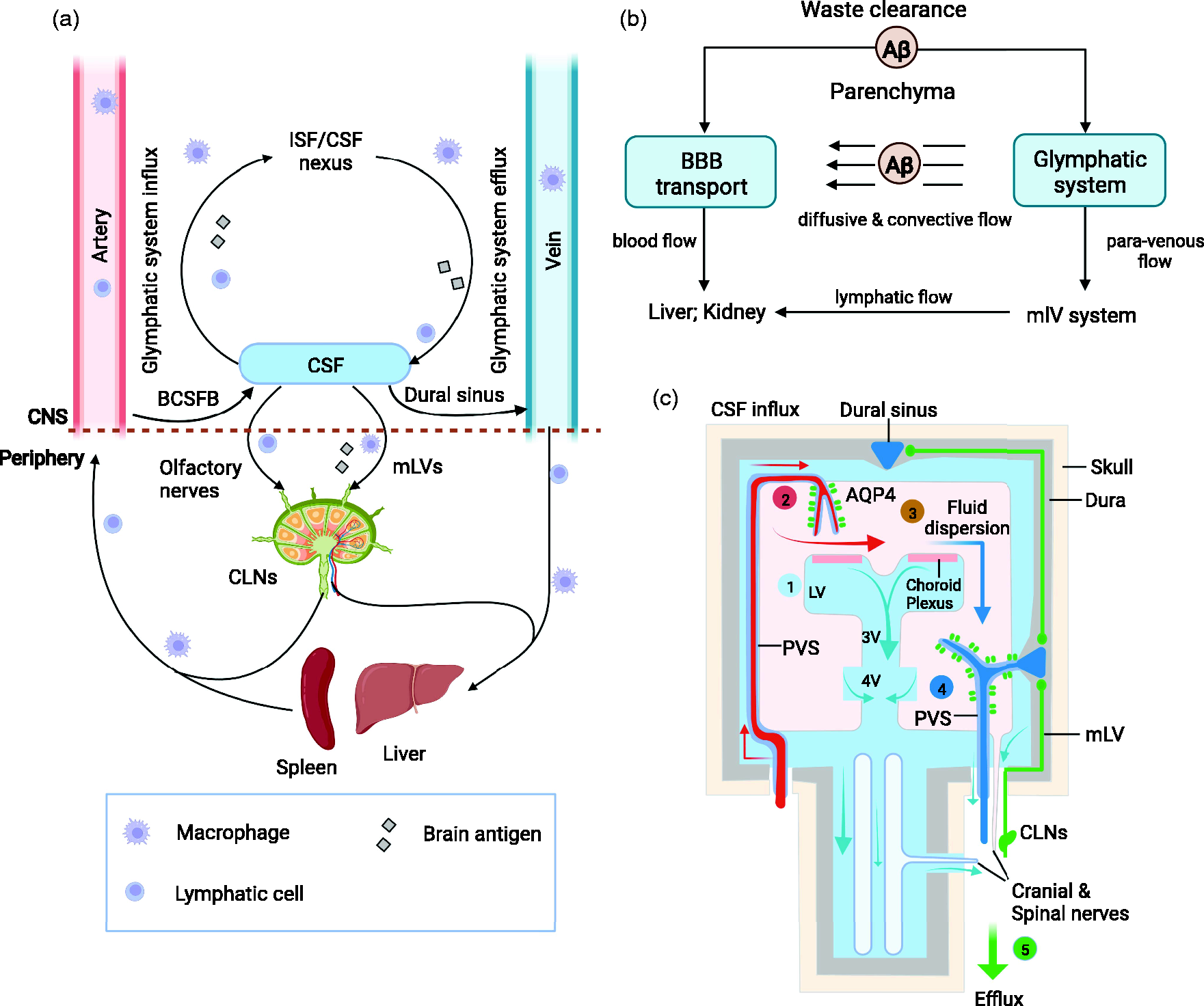
(a) In addition to the blood, the brain transport system may be a crucial component in CNS immune surveillance because it drains antigens as well as antigen-presenting and other immune cells from the brain parenchyma into CLNs for sampling and processing. (b) Convective flow of amyloid-β via the brain glymphatic-mLV system may facilitate the protein’s BBB transport. (c) Overall scheme of the brain drainage system, and especially the brain lymphatic drainage system. (1) CSF is mainly produced by the choroid plexus. (2) Arterial wall pulsations drive CSF deep into the brain along perivascular spaces. (3) CSF enters the brain parenchyma supported by AQP4 water channels and disperses within the neuropil. (4) ISF mixes with CSF and accumulates in the perivenous space, then drains out of the brain via (5) mLVs and veins, as well as along cranial and spinal nerves. Segments (2)–(4) are described in detail in Figure 1, while segment (5) is described in detail in Figure 3. The figure was created using online BioRender software (BioRender.com). Aβ, amyloid-β; AQP4, aquaporin-4; BBB, blood-brain barrier; BCSFB, blood–cerebrospinal fluid barrier; CLNs, cervical lymph nodes; CNS, central nervous system; CSF, cerebrospinal fluid; ISF, interstitial fluid; LV, lateral ventricle; mLVs, meningeal lymphatic vessels; PVS, perivascular space.
Evidence supporting the routes of CSF drainage is relatively consistent across species. 75 The greatest volume of CSF perineuronal outflow occurs along olfactory nerves through the cribriform plate into the cervical lymphatics,76,77 although CSF also drains along cranial nerves, such as trigeminal, glossopharyngeal, vagal, and spinal accessory nerves.77,78 CSF also drains along the spinal cord, especially around nerve roots from the lumbar or sacral regions.79,80 In fact, studies of intrathecal administration of tracers or gadobutrol show that CSF levels in blood peak at spinal egress sites long before they peak at cranial egress sites.81,82
Furthermore, we have previously introduced a brief concept that a functional network of lymphatics in the dural meninges also drains CSF. The mLVs are located in close proximity to the large vessels of the dura mater, such as the dural sinus and the branches of the middle meningeal artery.9,11,14 Although mLVs in the dorsal and basal dura mater collect intrathecally-delivered tracers, mLVs in the dorsal region have a smaller diameter than those in the basal region, they are discontinuous and they lack valves. 11 Ablation of the dorsal mLVs decreases drainage of CSF to CLNs, suggesting that mLVs are vital routes of CSF clearance.9,11,14,16 Arachnoid villi/granulations are protrusions of the arachnoid membrane 83 that are especially prominent at the superior sagittal sinus and are also observed at the transverse sinuses, confluence of sinus, and cavernous sinus. 84 CSF flows via the arachnoid villi/granulations into the bloodstream or lateral lacunae of the dura mater. The traditional view was that arachnoid villi/granulations are the main CSF egress sites in humans, but we now know that this is true only during the late fetal and early neonatal periods; with aging, arachnoid villi degenerate and the arachnoid membrane thickens, so this drainage route loses its efficiency. 36 At physiological intracranial pressure in adult, no CSF exits via this route into the bloodstream, 63 but this route may become recruited when pressure increases.50,75 Parasagittal spaces in the dura 85 and adventitia of major cerebral blood vessels 86 also contribute to CSF drainage.
Although numerous studies provide evidence for the nasal lymphatic pathway as the major route for CSF drainage into the CLNs, it is still difficult to rank the various pathways by importance and to quantify the absolute volume of fluid leaving the CNS through any particular outflow path. A recent review 75 concluded that evidence for CSF outflow from the cribriform plate was very strong, while it was moderate for outflow from the spinal nerve roots, optic nerves, and base of the skull. In contrast, evidence remains limited for CSF outflow via the mLVs and arachnoid villi/granulations under normal conditions. Regardless of which pathway is dominant, all these pathways must work in concert to ensure the effective removal of toxic waste products from the brain and ensure healthy neuronal functioning.
In vivo imaging for the evaluation of the glymphatic-mLV system and use in humans
State-of-the-art imaging techniques have provided definitive structural and functional characterization of the glymphatic-mLV system. Imaging the CSF-mediated brain removal pathways is an emerging field of great importance for understanding how drainage pathways degrade during aging and in neurodegenerative disorders. Table 1 shows in vivo imaging approaches currently used in rodents, mammal and/or humans. Analyzing the glymphatic-mLV system is quite difficult, so most imaging techniques so far have required brain surgery, and some techniques have yet to be applied to humans because of safety concerns.
Current in vivo approaches for imaging glymphatic pathways in rodents and humans.
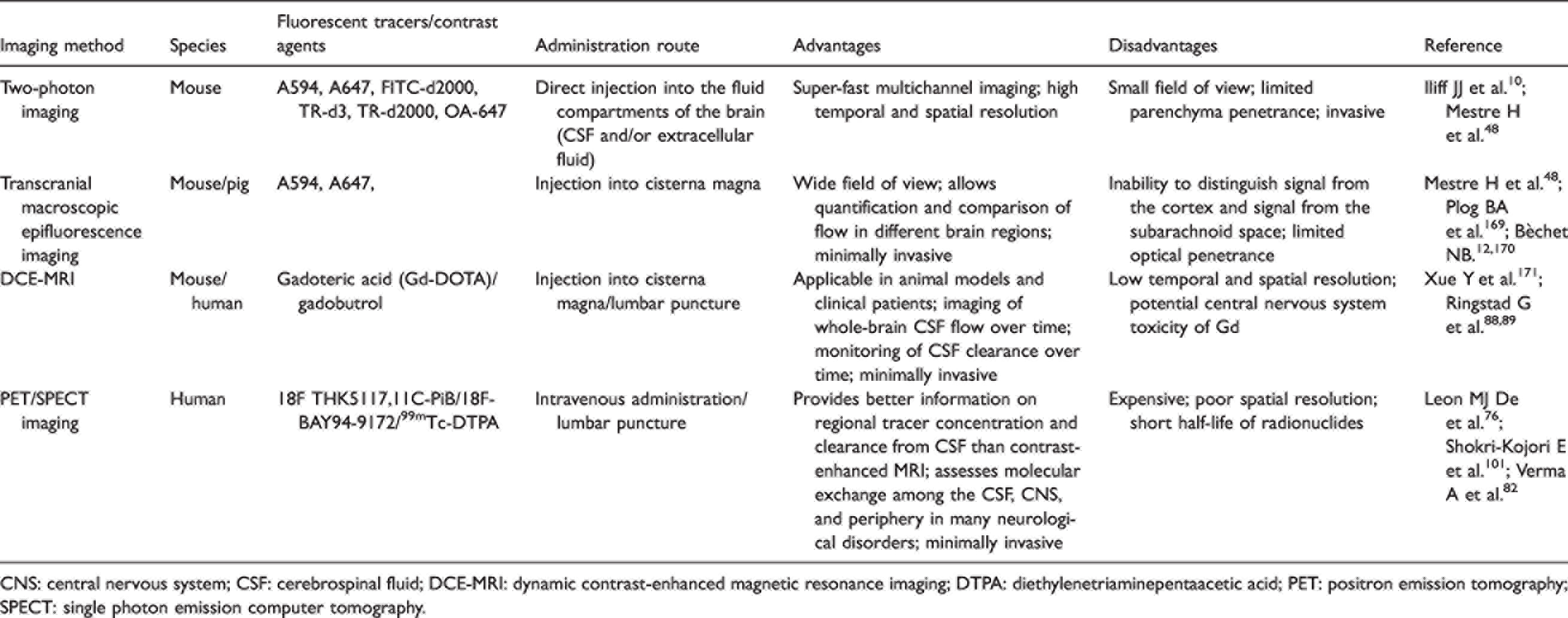
CNS: central nervous system; CSF: cerebrospinal fluid; DCE-MRI: dynamic contrast-enhanced magnetic resonance imaging; DTPA: diethylenetriaminepentaacetic acid; PET: positron emission tomography; SPECT: single photon emission computer tomography.
Magnetic resonance imaging (MRI) has recently gained momentum as a technique to investigate the glymphatic system in humans. Notably, dynamic contrast-enhanced (DCE) MRI has proven feasible as a minimally invasive method to visualize CSF flow in the human brain. 87 For instance, DCE-MRI studies involving intrathecal administration of gadobutrol revealed that CSF is transported along the glymphatic pathway with an anterograde flow, and that clearance is slowed in patients with idiopathic normal pressure hydrocephalus.88 –90 DCE-MRI studies have also shown that aging and sleep deprivation degrade glymphatic function and impair clearance in humans.91 –93
Other MRI methods have been applied to visualize the glymphatic system in humans. For example, diffusion tensor imaging can assess perivascular fluid movement and diffusion properties of the glymphatic system without the need for gadolinium-based contrast agents. 94 Three research groups independently reported that the method “diffusion tensor analysis along the perivascular space” can be applied to evaluate the glymphatic system, with lower diffusivity along the PVS indicating impairment of the glymphatic system in Alzheimer's disease, type 2 diabetes mellitus, and idiopathic normal pressure hydrocephalus.95 –97
T2-weighted-fluid-attenuated inversion recovery MRI and T1-weighted black-blood MRI have been used to visualize mLVs in human and nonhuman primates after intravenous injection of radiolabeled tracers. One study verified the existence of a dural lymphatic network and described the aforementioned two MRI techniques that can visualize it in vivo. 98
Several noninvasive MRI approaches are under development. For example, sleep- or resting-state functional MRI can track the dynamics of CSF flow in parallel with the global blood-oxygen-level-dependent (gBOLD) signal, providing insights into the relationship between glymphatic function and global brain activity.99,100 Interestingly, the gBOLD signal and CSF flow in those studies were coupled in healthy humans and patients with Parkinson's disease without cognitive dysfunction, but not in patients with Parkinson's disease and cognitive impairment.
Positron emission tomography (PET) and single photon emission computer tomography (SPECT) are minimally invasive methods that can investigate CSF flow in the human brain. In fact, whole-body dynamic SPECT is superior to MRI for providing regional information on tracer exchange between the CSF and brain ISF, as well as on tracer clearance from CSF into the periphery. 82 Dynamic PET can also measure CSF clearance, and PET studies showed lower ventricular tracer clearance in Alzheimer's patients than in normal controls, and the lower clearance was positively associated with brain amyloid-β deposition. 76 PET was also able to detect amyloid-β accumulation in the human brain after a night of sleep deprivation. 101
In summary, MRI and PET allow visualization of the human brain glymphatic system. These techniques allow detailed studies of how the brain removes toxic proteins or small molecules, which may significantly improve our understanding and treatment of disease. As minimally invasive techniques, MRI and PET may also allow the identification of reliable early biomarkers to screen for patients at risk of waste accumulation in the brain due to inadequate clearance.
Neurological diseases related to the glymphatic-mLV system
So far in this review, we have outlined the important roles of mLVs and the glymphatic pathway in CNS physiology. Dysfunction of mLVs and/or the glymphatic pathway has been implicated in a variety of neurologic diseases, and further study of those disease associations may help clarify pathogenesis and guide treatment.
Aging and neurodegenerative disorders
Aging is the greatest risk factor for numerous neurological disorders. A DCE-MRI study found that both the glymphatic pathway and mLVs might be impaired in the aging human brain. 91 Glymphatic function declines with age because the cerebral arteries become stiffer and the amplitude of pulsations decreases. More fluid and metabolites remain in dilated PVS and obstruct the perivascular pathways.24,26,40,41,102 Aging also dysregulates glymphatic function because AQP4 mislocalizes during astrogliosis (Figure 5).
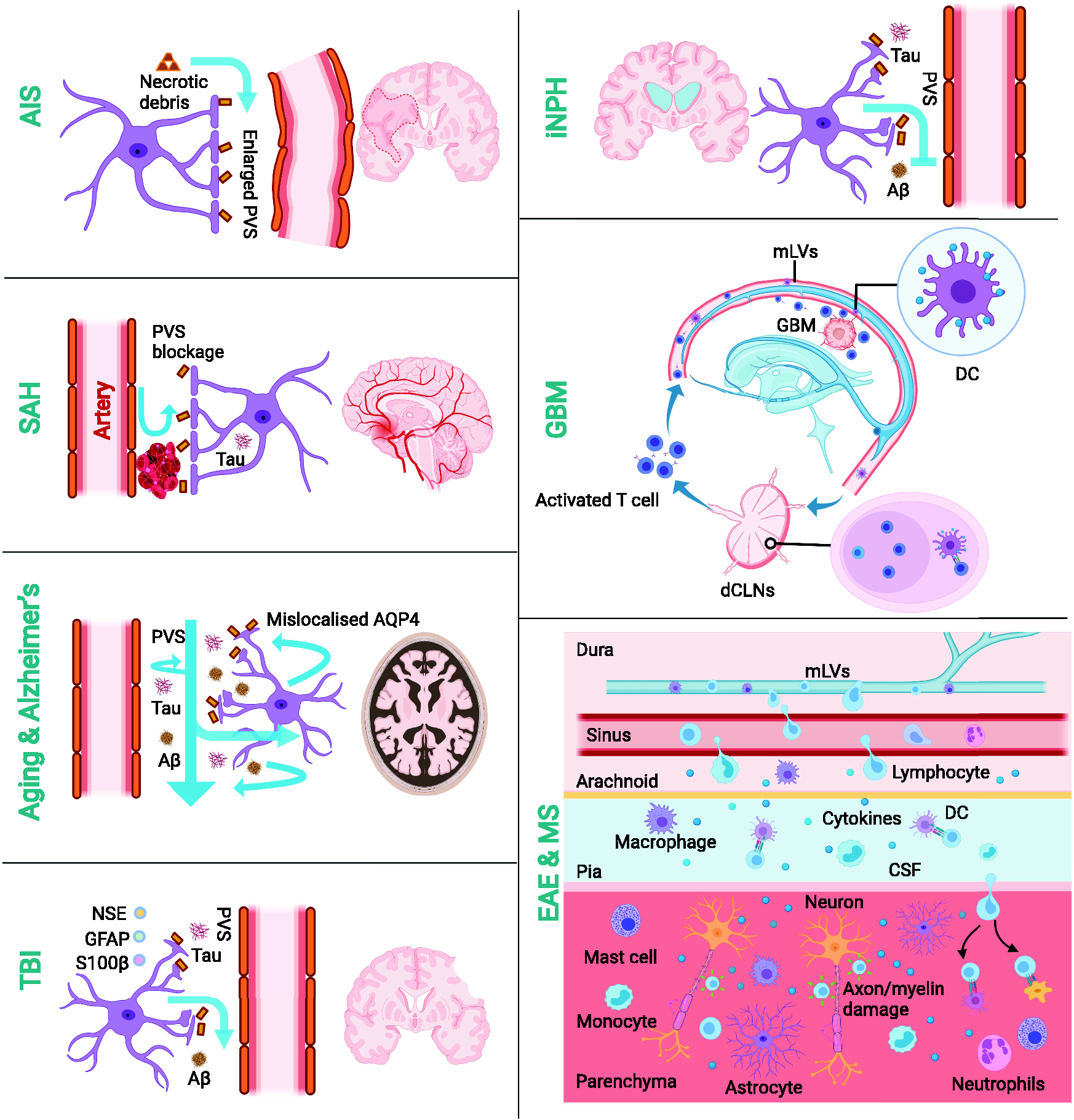
Pathological changes in the glymphatic-mLV system and related neurological diseases. A reduction in glymphatic fluid transport has been documented during healthy aging and in some neurological disorders, such as acute ischemic stroke (AIS), subarachnoid hemorrhage (SAH), traumatic brain injury (TBI), Alzheimer's disease, and idiopathic normal pressure hydrocephalus (iNPH). In addition, mLVs have been implicated in glioblastoma (GBM) and multiple sclerosis (MS). The mLVs act as conduits connecting the central nervous system and peripheral immunity: they transport numerous immune cells and antigens to dCLNs or to the CNS. The figure was created with online BioRender software (BioRender.com). Aβ, amyloid-β; CSF, cerebrospinal fluid; EAE, experimental autoimmune encephalitis; DC, dendritic cells; dCLNs, deep cervical lymph nodes; GFAP, glial fibrillary acidic protein; mLVs, meningeal lymphatic vessels; NSE, neuron-specific enolase; PVS, perivascular space.
Dysfunction of the brain glymphatic-mLV drainage system contributes to the deposition of toxic proteins such as amyloid-β and α-synuclein in the brain parenchyma, and it can cause neuropathological changes in neurodegenerative disorders such as Alzheimer's and Parkinson's diseases.103,104 In the healthy brain, the removal system efficiently eliminates amyloid-β and α-synuclein from the extracellular space, but clearance is impaired in those two neurological disorders.16,105,106 Older mice showed 40% less clearance of injected amyloid-β than younger animals, 40 and knocking out AQP4 increased brain amyloid-β aggregates in mice overexpressing amyloid precursor protein and presenilin.107,108 Knocking out AQP4 or blocking its activity in mice nearly abolished clearance of pathogenic tau 69 or α-synuclein. 109 These results are consistent with the idea that AQP4 mislocalization sets the stage for glymphatic failure, increasing the risk of Alzheimer's and Parkinson's diseases. In postmortem frontal cortex analysis, intense AQP4 expression was maintained near the cortical surface in Alzheimer's patients and the AQP4 expression became discontinuous below cortical layer. Further, perivascular AQP4 localization in Alzheimer's patients was reduced when compared to young and aged cognitively intact individuals; loss of AQP4 localization to perivascular astrocytic endfeet was associated with increasing Aβ plaque density and worsening Alzheimer's status. As a result, the mislocalization of AQP4 may slow glymphatic function and promote protein aggregation and neurodegeneration. 105
CSF egress via dural lymphatics also appears to be vital for waste clearance. In mouse models of Alzheimer's disease, higher lymphatic coverage can lead to better brain clearance, while ablation of dorsal dural lymphatics or ligation of deep CLNs increases the burden of amyloid-β and tau in the brain and meninges.16,110 Similarly, studies in humans also implicate functional impairment of the glymphatic-mLV system in Alzheimer's and Parkinson's diseases.105,106,111
Sleep disturbances
Our brain can produce large amounts of toxins, and sleep promotes neuroprotection.18,112 During sleep, the glymphatic pathway is highly active in removing waste products. 113 During sleep, especially the nonrapid eye movement period, the extracellular space increases in volume and both blood vessel diameter and blood volume fluctuate more, all of which helps drive bulk flow of glymphatics.18,99,114 During sleep, ISF volume increases by 60%, and up to 60% of neurotoxic proteins and solutes are removed via the glymphatic pathway.10,18 One night of sleep deprivation in human patients results in decreased clearance of the CSF tracer gadobutrol and significant amyloid-β accumulation in the brain parenchyma. After one night of normal sleep, sleep-deprived patients still show higher levels of gadobutrol in the brain than patients who are not deprived.92,101 Similar to these effects, chronic insomnia patients had higher CSF levels of amyloid-β than normally sleeping individuals. 115 Interestingly, poor sleep quality is associated with the enlargement of PVS, 116,117 sleep deprivation induces reactive astrocytes and reduces perivascular flow.118,119 Taken together, these data suggest that sleep deficiency may lead to structural changes in PVS. Glymphatic function is increased during sleep and impaired by sleep disturbances. Thus, glymphatic system dysfunction may help explain why chronic sleep deficiency is associated with a variety of neurological disorders, including Alzheimer's and Parkinson's diseases. Sleep disturbance is an early feature of the two diseases, often preceding the onset of classic symptoms. 120 Interventions targeting sleep disturbances may improve glymphatic efficiency and thereby exert neuroprotective effects.
Multiple sclerosis and neuroinflammation
Multiple sclerosis is a chronic neuroinflammatory disease characterized by periodic infiltration of immune cells into the CNS, leading to neuronal demyelination and dysfunction as well as progressive disability. 121 We have introduced that mLVs are important components of CNS immune surveillance and connect directly with CLNs, thereby mediating the communication between the CNS and peripheral immunity. Lymphoid clusters of T cells in mLVs have been identified in mice with experimental autoimmune encephalitis (EAE), providing the first clues about how CNS-infiltrating T cells drain into CLNs through the meningeal lymphatics. The brain parenchyma in those mice is infiltrated by many immune cells, including T cells, B cells, macrophages and dendritic cells, which damage nerves (Figure 5). Ablation of mLVs or removal of components of their downstream pathways attenuates inflammation in the cerebral hemispheres and/or spinal cord.15,122,123 EAE-induced neuroinflammation also promotes lymphangiogenesis near the cribriform plate, 17 which aids recovery in late EAE by promoting the drainage of CNS-derived antigens and immune cells, thereby stimulating the proliferation of antigen-specific T cells in the draining lymph nodes. 17 At the same time, lymphangiogenesis near the cribriform plate helps drain neuroinflammation-induced excess fluid in order to maintain homeostasis. 17 This lymphangiogenesis appears to depend on vascular endothelial growth factor C (VEGF-C). 17
Stroke and traumatic brain injury
Hemorrhagic or ischemic stroke as well as traumatic brain injury are the most common adult brain injuries and are associated with extensive damage and limited recovery. 124 These events can severely disrupt the cerebral glymphatic drainage system (Figure 5).125 –127 For example, in mouse and non-human primate models of subarachnoid hemorrhage, erythrocytes extended into the perivascular spaces of parenchymal arterioles, 128 the glymphatic influx of CSF was blocked by fibrin and fibrinogen accumulation in the PVS, severely reducing ISF perfusion (Figure 5). Perivascular macrophages were involved in erythrocyte clearance, contributing to perivascular inflammation and neuronal cell death after subarachnoid hemorrhage. 128 CSF clearance through the glymphatic pathway could be rescued by administering tissue plasminogen activator, a specific serine protease.125,129 In a mouse model of acute ischemic stroke, CSF inflow was reduced in the ipsilateral cortex after 3 hours of middle cerebral artery occlusion, while spontaneous arterial recanalization increased glymphatic perfusion within 24 h. 129 In fact, the brain clearance system also contributes to later recovery from ischemic stroke: even at seven weeks after hypoxic middle cerebral artery occlusion in mice, neurotoxic materials from liquefactive necrosis were found to be leaking across the glial scar and were being removed from the brain along the PVS. 130 Meningeal lymphatic hypoplasia could exacerbate stroke severity after transient middle cerebral artery occlusion. 131 Traumatic brain injury and multiple microinfarcts have been shown to trigger structural destruction of glymphatics, reactive gliosis and AQP4 mislocalization, reducing brain drainage and ultimately allowing the accumulation of waste products in the parenchyma.63,132 –134
The meningeal lymphatic drainage system disposes of erythrocytes generated following subarachnoid hemorrhage and hematoma, and it transports neurotoxic material following stroke and traumatic brain injury. This helps prevent chronic inflammatory responses and secondary brain damage.135 –137 The mLVs also serve as a route for immune cell infiltration and post-stroke inflammation, and they help eliminate damage/danger-associated molecular patterns that are released in the early phase of traumatic brain injury.136,138 –140
Glioblastoma
In the past, CNS immune surveillance against brain pathogens and tumors was thought to be limited due to the lack of lymphatic drainage. The discovery of functional lymphatic vessels in the meninges has changed this view (Figure 5). For example, mLVs appear to play a crucial role in the immune response to glioblastoma, 141 the most common malignant primary brain tumor. 142 Treating mLVs with VEGF-C resulted in lymphangiogenesis and enhanced priming and migration of CD8+ T cells, causing rapid clearance of the established glioblastoma. Further work has confirmed the contribution of mLVs to brain tumor immunity, 143 and found that increased expression of VEGF-C could strengthen the antitumor effect of anti-PD-1/CTLA-4 antibodies. VEGF-C-treated mice showed many more glioblastoma-specific cytotoxic CD8+ T cells in tumors and dCLNs than untreated mice. These studies suggest novel therapeutic options against glioblastoma that are worth investigating.
Idiopathic normal pressure hydrocephalus
Idiopathic normal pressure hydrocephalus is a rare neurological disorder characterized by cognitive impairment, gait ataxia, and urinary incontinence. Other clinical features include progressive enlargement of the cerebral ventricles without an associated increase in intracranial pressure. Approximately 10% of dementia patients have this condition, and an impaired glymphatic system has been identified as an important contributor to the pathogenesis.144,145 An MRI study found that, after intrathecal injection of the CSF tracer gadobutrol at the lumbar level, patients with idiopathic normal pressure hydrocephalus showed slower tracer influx and subsequent clearance than healthy controls (Figure 5). 88 Similar impairments in glymphatic flux were observed in a four-week study of patients and controls, 89 and in a study using the “diffusion tensor image-analysis along the perivascular space” method. 97 Patients show decreased expression of AQP4 in astroglial endfeet in cortical brain, further linking this disorder to a compromised glymphatic system. 146
Brain edema
Swelling of the brain is a result of abnormal accumulation of fluid caused by a range of neurological conditions, mainly stroke, trauma, and tumors.147,148 Traditionally, brain edema is divided into two phases: an early cytotoxic phase and a later vasogenic phase. 149 The cytotoxic phase is predominant in the early stages of acute ischemic stroke and traumatic brain injury, and it is related to cellular energy depletion, failure of membrane integrity, and cellular swelling. Glymphatic inflow of CSF appears to be the primary event in the early phase of acute ischemic stroke: in one study, CSF flow into the brain along the PVS doubled within a few minutes after middle cerebral artery occlusion, driving acute brain swelling. 48 The increase in flow was attributed to pathological constriction of cerebral blood vessels following spreading depolarization. In addition, the hypoxia can trigger an increase in AQP4-mediated flux of water into astrocytes, and translocation of AQP4 to the astrocyte cell surface increases water flux. 150 Consistently, knocking out AQP4 in mice reduces cerebral ischemic volume after stroke and improves prognosis.151 –153 These studies suggest that unregulated glymphatic function in the early stage of acute ischemic stroke may contribute to brain edema. Thus, targeting APQ4 may be able to prevent cytotoxic edema. 150
However, simply inhibiting AQP4 may not always be desirable. Vasogenic edema occurs mainly in subjects with brain tumors or subacute stroke. In vasogenic edema, the glymphatic drainage system can help clear extravasated fluid and serum proteins into the CSF, and knocking out AQP4 impairs this process in mice.154,155 These studies indicate that targeting AQP4 may be a way to prevent CNS edema during the acute phase, but whether that means activating or inhibiting AQP4 should be considered carefully.150,156
Impairments in the glymphatic system are also associated with other diseases such as small vessel disease,67,157 hypertension 158 and diabetes. 159 In small vessel disease, 160 damage to the small end arteries, arterioles, capillaries, and venules, along with the expansion of the perivascular spaces, adversely affects glymphatic system function and ultimately leads to dementia. Hypertension and diabetes as the conditions affecting vascular compliance likely exacerbate disturbances in CSF flow and CSF-mediated brain clearance.158,159 However, it is unclear whether impairments in the glymphatic system are a cause or effect of the disease, or both. For example, it has been proposed that Alzheimer's disease is caused by the accumulation of toxic proteins (tau and amyloid-β), and that the primary cause may be the impairment of the glymphatic system. Therefore, further studies are needed to investigate the link between glymphatic system impairment and correlated diseases.
Future directions
Clinical imaging
Numerous studies have demonstrated the usefulness and acceptable toxicity of intrathecal injection of gadolinium-based contrast agent to assess glymphatic-mLV drainage using DCE-MRI.87 –91,93,101 In the near future, intrathecal administration of small doses of MRI contrast agent may allow accurate evaluation of glymphatic system function. This relatively noninvasive technique may allow the study and evaluation of waste clearance and load of toxic proteins such as amyloid-β, α-synuclein, and tau in Alzheimer's disease, Parkinson's disease, and aging. Such work may improve early diagnosis of neurodegenerative diseases, monitoring of their progression, and even their treatment. 161
Preventive and therapeutic strategies for neurological diseases
Numerous factors can affect glymphatic function, including sleep, chronic stress, and high alcohol intake.162,163 Impaired glymphatic drainage has been shown to promote waste deposition: glymphatic failure is considered the point of convergence for various pathways leading to neurodegenerative dementia. Thus, it may be possible to slow or prevent neurodegeneration though healthy lifestyle choices, such as reducing alcohol intake, increasing intake of omega-3, engaging in intermittent fasting and physical exercise164 –166 as well as improving sleep. 113
Another therapeutic option is suggested by studies in which giving VEGF-C to aged mice safely increased the diameter of mLVs and enhanced meningeal lymphatic drainage, improving brain perfusion, learning, and memory.9,16 In another study, treating aged mice with VEGF-C and amyloid-β immunotherapy synergistically enhanced glymphatic function, cleared amyloid-β plaques, and improved cognition. 110 Local delivery of VEGF-C also boosted the effectiveness of immune checkpoint inhibitors, 143 highlighting the importance of meningeal lymphatics in regulating brain tumor immunity. These studies imply the therapeutic potential of mLVs.
Beyond clearance, the glymphatic pathway is also indispensable for nutrient distribution. 167 By taking advantage of the glymphatic exchange between CSF and ISF, administering drugs directly into the CSF may bypass the BBB and enhance convective drug delivery from the CSF into the parenchyma. Indeed, the glymphatic system can mediate the brain-wide delivery of adeno-associated viruses 168 and the transport of intrathecally administered drugs into the parenchyma. 42 These studies indicate that the CSF-ISF pathway may be an efficient delivery route for gene and drug therapies against neurological diseases.
AQP4 in astrocytes in the early stage of acute ischemic stroke may contribute to cytotoxic edema in the CNS. 48 Reversible inhibition of AQP4 using the licensed drug trifluoperazine can eliminate such edema. 150 Thus, modulation of AQP4 may be a powerful strategy for treating brain and spinal cord edema.
Conclusions and open questions
The glymphatic pathways and mLVs are internally connected, participating in material transport, waste removal, and meningeal immunity in the brain. They maintain CNS homeostasis, and their dysfunction has been linked to neurodegenerative disorders. Several imaging methods have been applied for the dynamic evaluation of glymphatics in humans, and research with mice has identified mLVs as potential therapeutic targets in the treatment of neurological diseases.
Future work should explore imaging-based assessment of the glymphatic-mLV system and the development of strategies to compensate for its dysfunction. We and other groups are trying to address several research questions that remain open:
Will there be a noninvasive, effective, and standardized clinical imaging examination for evaluating glymphatic system function in the near future? Will it be DCE-MRI? To what extent can improved sleep enhance glymphatic function? Should we routinely take measures to improve sleep to slow the progression of Alzheimer's and Parkinson's diseases? Can local delivery of VEGF-C improve human mLV function? Can Alzheimer's disease and glioblastoma be treated using the combination of VEGF-C, antibodies against amyloid-β or the immune checkpoint inhibitors PD1 or CTLA4, and/or other therapeutic drugs? How does the glymphatic-mLV system work together with other waste clearance routes, such as the bloodstream or channels running along the blood vessels or nerves, in order to deliver molecules to the lymphatic system outside the CNS? Can the CSF-ISF be better exploited as a route for widespread delivery of drugs injected intrathecally or intraventricularly? Are AQP4 inhibitors safe and effective to prevent or treat cerebrospinal edema in the early stage of acute ischemic stroke?
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81772693), Double Top Construction and Innovation Spark Item of Sichuan University (2082604401047), Incubation project of clinical study, West China Hospital, Sichuan University (2019HXFH020).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
GWL, YC, XT, JHH and LJC contributed to the conceptualization and writing of the final manuscript. GWL contributed to the literature search of the original draft. YC, JHH and LJC designed the figures. XT and LXZ reviewed, modified and edited the manuscript. All authors have read and approved the final version of the manuscript.
