Abstract
The feasibility of transcranial sonothrombolysis has been demonstrated, although little is known about the relationships between thermal or mechanical mechanisms and thrombolytic outcomes. Therefore, the present study aims to reveal the effect and safety of temperature and ultrasound through in vitro and in vivo thrombolysis models. Artificial clots in microtubes were heated in a water bath or sonicated by ultrasound irradiation, and then clots weight decrease with rising temperature and sonication time was confirmed. In the in vitro thrombotic occlusion model, based on spot heating, clot volume was reduced and clots moved to the distal side, followed by recanalization of the occlusion. In the in vivo study, the common carotid artery of rats was exposed to a spot heater or to sonication. No brain infarct or brain blood barrier disruption was shown, but endothelial junctional dysintegrity and an inflammatory response in the carotid artery were detected. The present spot heating and ultrasound irradiation models seem to be effective for disintegrating clots in vitro, but the safety of the in vivo model was not fully supported by the data. However, the data indicates that a shorter time exposure could be less invasive than a longer exposure.
Introduction
Thrombolysis with recombinant tissue-type plasminogen activator (rt-PA) and endovascular therapy have been used to treat acute strokes. However, rt-PA thrombolysis is limited by various exclusion criteria such as a narrow time window and a significant risk of bleeding.1–4 Endovascular therapy is also currently limited because of its limited availability 5 and high costs. Hence, there is a strong demand for novel treatment options for strokes.
Focused ultrasound (FUS) surgery, such as high-intensity focused ultrasound (HIFU) and MRI-guided focused ultrasound (MRgFUS), is a non-invasive thermal ablation method used to treat various tumors6,7 and intracranial focal brain structures.8,9 FUS can produce both thermal and mechanical effects in tissue. Thermal effects result from the generation of heat at the focus point due to absorption of acoustic energy by tissue. 10 The underlying mechanical effects include acoustic cavitation, shear stress and acoustic streaming. 11
The feasibility of transcranial sonothrombolysis by FUS has been demonstrated both in vitro12–14 and in vivo.15–18 Clinical trials for sonothrombolysis that employed diagnostic transcranial ultrasound have been conducted.19–21 A common mechanism of sonothrombolysis are mechanical effects such as the displacement and cavitation of clots. 22 While temperature during exposure to ultrasound is assumed to potentially contribute to enhanced thrombolysis, 22 most studies have not found a major relationship between thermal mechanisms and thrombolytic outcomes,23–26 probably due to the use of different measurements during exposure to ultrasound.24,27
Therefore, in the present study, we examined thermal and mechanical effects on thrombolysis by using a photic heater and ultrasound irradiation in vitro, and evaluated their safety on the cervical endothelium of an in vivo animal.
Materials and methods
Data availability statement
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
In vitro study
Clot preparation
Artificial clots were prepared by using fibrin glue, Bolheal (Chemo-Sero Therapeutic Research Institute, Kumamoto, Japan). It is composed of solutions A and B. Solution A contains human fibrinogen (80 mg/mL), human coagulation-factor XIII (75 U/mL), and bovine aprotinin (1000 ∴ KIE/mL). Solution B contains human thrombin (250 U/mL) and calcium hydrochloride (5.9 mg/mL) with supplemental red ink for coloring. First, each solution was drawn by a syringe (Figure 1(a)) and simultaneously applied to a drinking straw with a 3.0 mm diameter (Figure 1(b)). Next, the straw was removed (Figure 1(c)), and each clot was finally cut into a section 8.0 mm long (Figure 1(d)). Clot size was determined based on our own data, as explained next. Based on an analysis of clots that were retrieved from 52 acute ischemic stroke patients, 28 clot sizes for the terminal internal carotid artery (ICA), proximal M1 segments, and distal M1 segments of the middle cerebral artery (MCA) were 5.0 × 11.2 mm (short axis × long axis), 2.8 × 6.6 mm, and 2.0 × 7.6 mm, respectively. On the other hand, several reports have indicated the frequency of intracranial large vessel occlusion during acute ischemic stroke. Of all occlusions found in anterior circulation, about 20% of patients had an ICA occlusion, with 40% for M1 segments, and 40% for M2 segments.29–31 By considering each clot size and the occurrence of occlusions for ICA, M1, and M2 based on previous papers, we determined that the average clot size in the present study was 3.0 × 8.0 mm.
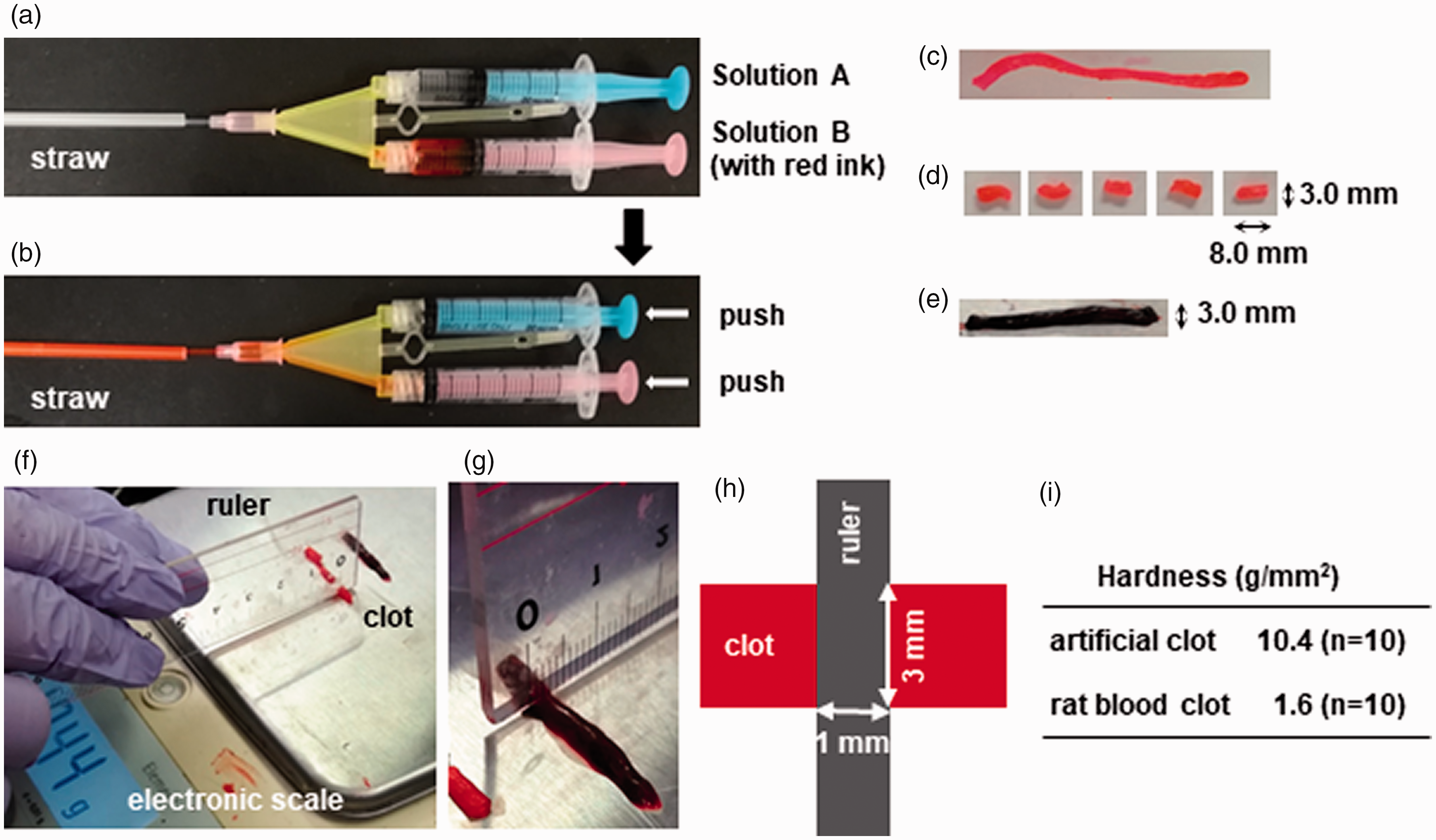
Clot preparation. Artificial clots were prepared by using the fibrin glue, Bolheal. It is composed of solution A containing human fibrinogen and solution B containing human thrombin, and red ink is added for coloring. Each solution was loaded to a syringe (a) and simultaneously applied to a drinking straw with a 3.0 mm diameter (b). The straw was removed (c) and each clot was finally cut into an 8.0 mm long section (d). (e) The clot made from rat arterial blood. Artificial clots and rat blood clots were weighed on an electronic scale until the weight of the clot cut by a ruler was measured (f and g). Clot hardness was obtained by dividing the weight by the area of the clot in contact with the ruler (h and i).
Comparison of hardness between arterial clots and traditional blood blots
We prepared traditional blood clots according to a previously reported method.32,33 In brief, arterial blood from a rat was withdrawn into a 2.5 mL syringe and retained for 2 h at room temperature, then subsequently retained for 22 h at 4 °C. The clot was cut when it was 3 mm in diameter (Figure 1(e)). The weights of both artificial clots and traditional rat blood clots were assessed on an electronic scale (ELT602; Sartorius Mechatronics Japan, Tokyo, Japan) and continued to be assessed until the clot was cut by a ruler (Figure 1(f) and (g)). Clot hardness (g/mm2) was obtained by dividing the weight (g) by the area (mm2) of the clot in contact with the ruler (Figure 1(h)).
Heating clots in Eppendorf tubes
Artificial clots in 1 mL saline were added to a 1.5 mL Eppendorf microtube, which was heated in a constant temperature bath of 45 °C, 55 °C, or 65 °C (n = 3 for each) for 30 min. Clot weight was measured before and after heating. Total protein eluted from heated clots was estimated with the BCA protein assay kit (T9300A; Takara, Kusatsu, Japan).
Recanalization of thrombotic occlusions by photic heating
Artificial clots were placed in micropipette tips (one/tip) connected to an infusion bottle. Bottles were hung at three heights to mimic a blood pressure (BP) of 100, 150, and 200 mmHg (Figure 2(a-3)). A pressure which supports a 1 mm column of mercury will support a 1.36 cm column of water. Therefore, to mimic a BP of 200 mmHg, the bottle was hung 272 cm above the clot level (272 cmH2O). Similarly, BPs of 150 mmHg and 100 mmHg were mimicked by heights of 204 cm and 136 cm, respectively. Clots were then exposed to a halogen point heater (LCB-50, 2 mm focal diameter, 35 mm focal distance; Inflidge Industrial Ltd., Yokohama, Japan) until clots moved and then recanalization with a water flow was observed. A thermometer probe (HD-1100E; Anritsu Meter, Tokyo, Japan) was placed on the micropipette tip at the rear side of the heater and changes in temperature were continuously recorded.
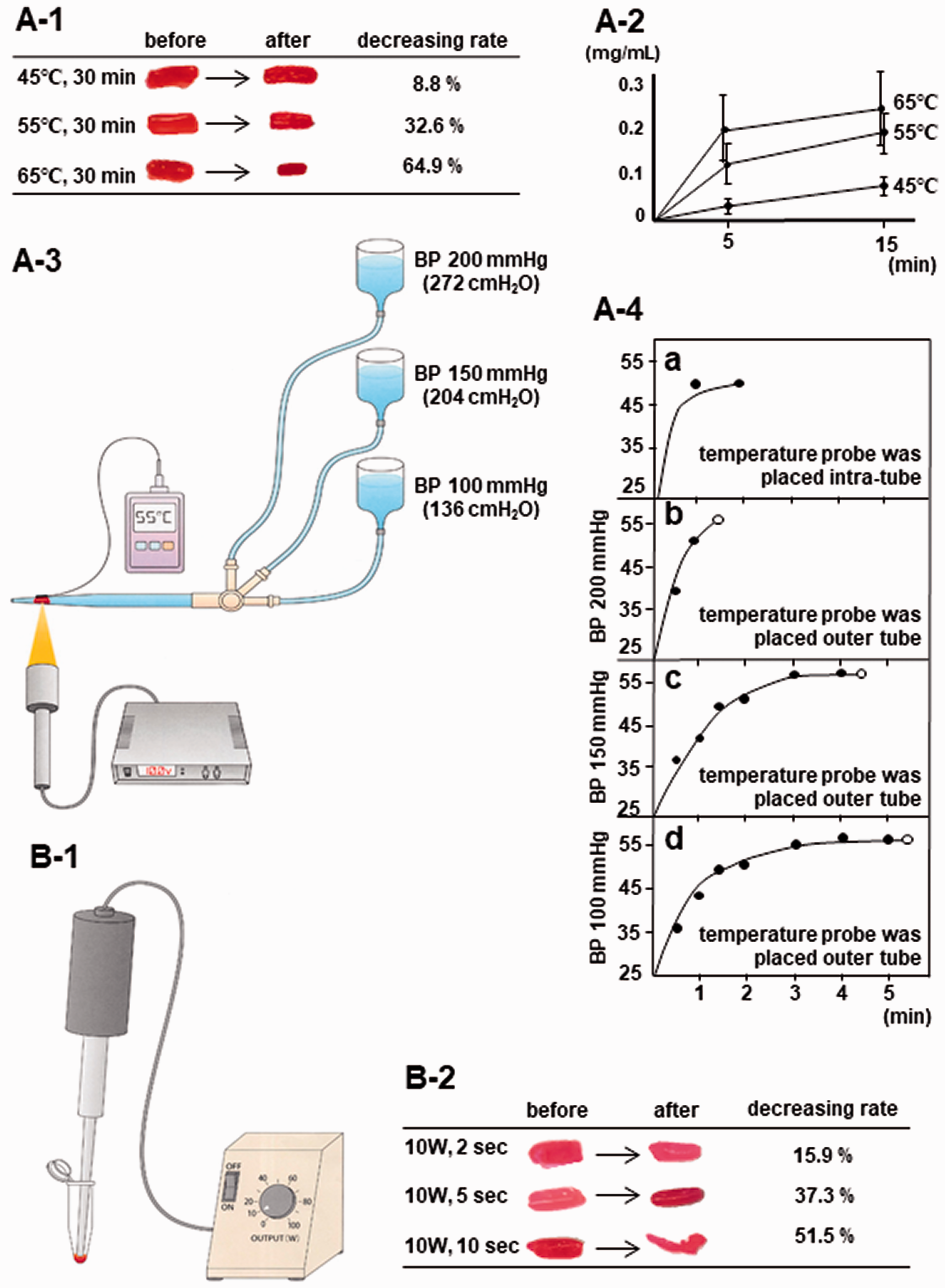
Clots lysis after heating or sonication, and recanalization of in vitro thrombotic occlusions by photic heating. The clot in saline was heated in a 1.5 mL microtube in a constant temperature bath. A reduction in clot weight (a-1) and increased eluted protein from clots (a-2) were observed, proportional to the increase in temperature. (a-3) The clot was placed in a micropipette tip connected to an infusion bottle which was hung at three heights to mimic 100, 150, or 200 mmHg blood pressure. The clot was then exposed to a halogen point heater until recanalization with water flow was observed. A thermometer probe was placed on the micropipette tip at the rear side of the heater and changes in temperature were continuously recorded. (a-4) Recanalization time was shortened as water pressure increased, i.e., 1 min 25 sec on average for BP of 200 mmHg (n = 2; b), 4 min 16 sec for BP of 150 mmHg (n = 3; c), and 5 min 34 sec for BP of 100 mmHg (n = 2; d). Note that temperature reached about 55 °C within 1 to 3 min and was maintained at 55 °C until recanalization occurred (b-d). The curve of the rise in temperature was similar for the intratube probe (a) and on the rear side of the tube (b-d). (b-1) Schematic drawing of the in vitro clots sonication model. The clot was placed in a 1.5 mL microtube, then sonicated at minimum output (10 W) for 2, 5, or 10 sec by ultrasound irradiation. (b-2) Based on sonication, clot weight decreased in a manner proportional to irradiation time.
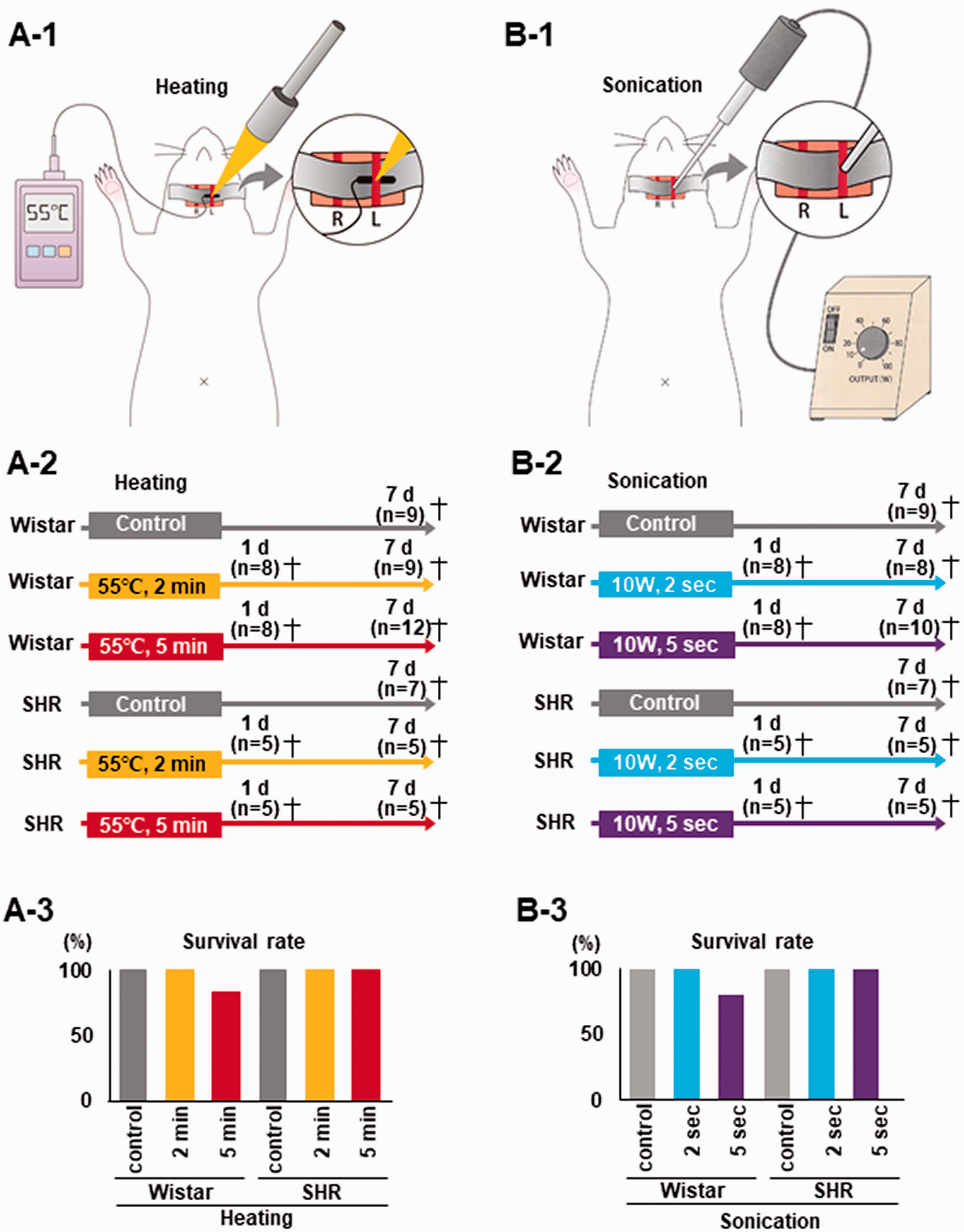
In vivo schematic procedure, experimental design, and survival rate. (a-1) The left CCA was irradiated with a halogen point heater at 3 mm proximal to the bifurcation. The thermometer probe was place on the rear side of the left CCA, and temperature was kept constant at 55 °C. To minimize heat stress, other parts were covered with aluminum foil. (a-2) Experimental design showing the control and heating at 55 °C for 2 and 5 min groups for both Wistar rats and SHR. After heating, rats were sacrificed on day 1 and 7. (a-3) Two out of 12 (16.7%) Wistar rats died in the 5 min heating group. (b-1) The left CCA was sonicated by ultrasound irradiation at 3 mm proximal to the bifurcation. (b-2) Experimental design showing the control and sonication for 2 and 5 sec groups for both Wistar rats and SHR. After sonication, rats were sacrificed on day 1 and 7. The same animals were used as the sham control in the heating and sonication experiments. (b-3) Two out of 10 (20%) Wistar rats died after 5 sec sonication. The same animals were used as the sham control in the heating and sonication experiments.
Sonicating clots in Eppendorf tubes
Artificial clots were placed in 1.5 mL Eppendorf microtubes (one/tube) to avoid scatter. Microtubes were sonicated at minimum output (10 W) for 2, 5, or 10 sec (n = 3 for each) by ultrasound irradiation (Figure 2(b-1)) (200 W for maximum output, 20 kHz, UD-200; Tomy Digital Biology, Tokyo, Japan). Clot weight was measured before and after sonication.
In vivo study
Animals
All animal experiments were approved by the Animal Committee of the Graduate School of Medicine and Dentistry, Okayama University (OKU-2021408) and conducted in accordance with ARRIVE guidelines (https://www.nc3rs.org.uk/arriveguidelines) and the Okayama University Guidelines for the Care and Use of the Laboratory Animals. Male Wistar rats (11 weeks old, body weight 250–330 g; SLC, Shizuoka, Japan) and male spontaneously hypertensive rats (SHR) (11 weeks old, body weight 260–310 g; SLC) were acclimatized in standard rat cages under conventional laboratory conditions with a 12/12 h light-dark cycle and constant humidity and room temperature for 2 weeks. The animals were fed rat pellets (MF, Oriental Yeast, Tokyo, Japan). Water was provided ad libitum in bottles. Cages were cleaned weekly and lined with paper pulp bedding (Oriental Yeast).
Each rat was anesthetized with a mixture of nitrous oxide/oxygen/isoflurane (69%:30%:1%) using an inhalation mask, and then a burr hole with a diameter of 2 mm was carefully made in the skull to measure regional cerebral blood flow (rCBF). The dura mater was preserved at this time. The location of the burr hole was 3 mm dorsal and 4 mm lateral to the right from the bregma, which is located in the upper part of the MCA territory.
Heating common carotid artery experiment
The present study included two in vivo experiments: the heating common carotid artery (CCA) model and the sonicating CCA model. On the day following burr hole surgery, bilateral CCAs were exposed after induction by anesthesia. The left CCA was irradiated by a halogen point heater at 3 mm proximal from the bifurcation. The thermometer probe was place on the rear side of the left CCA and temperature was kept constant at 55 °C. To minimize heat stress, other parts were covered with aluminum foil (Figure 3(a-1)). During the surgical procedure, body temperature was monitored and maintained at 37 ± 0.3 °C using a heating pad (BWT-100; Bio Research Center, Nagoya, Japan). In this experiment, Wistar rats and SHR were randomly divided into three groups of each by a block randomization method: sham control (n = 9, 7 for Wistar and SHR, respectively), heating at 55 °C for 2 min (eight Wistar rats sacrificed after 1 day and nine rats sacrificed after 7 days; five SHR sacrificed after 1 day and five rats sacrificed after 7 days) and 5 min (eight Wistar rats sacrificed after 1 day and 12 rats sacrificed after 7 days; five SHR sacrificed after 1 day and five rats sacrificed after 7 days) (Figure 3(a-2)). Sham control rats not exposed to heating were incised similarly and were sacrificed only at 7 days after the sham operation.
Sonicating common carotid artery experiment
On the day following burr hole surgery, bilateral CCAs were exposed after induction by anesthesia. The left CCA was sonicated by ultrasound irradiation at 3 mm proximal from the bifurcation. To minimize stress from the sonicator, other parts were covered with aluminum foil (Figure 3(b-1)). In this experiment, Wistar rats and SHR were divided into three groups of each: sham control (n = 9, 7 for Wistar and SHR, respectively), sonicating with 10 W for 2 sec (eight Wistar rats sacrificed after 1 day and eight rats sacrificed after 7 days; five SHR sacrificed after 1 day and five rats sacrificed after 7 days) and 5 sec (eight Wistar rats sacrificed after 1 day and 10 rats sacrificed after 7 days; five SHR sacrificed after 1 day and five rats sacrificed after 7 days) (Figure 3(b-2)). The sham control were the same animals used in the heating experiment. Since 4 out of 34 Wistar rats and 3 out of 20 SHR showed bleeding from CCA immediately after sonication, direct pressure was applied to the bleeding site until bleeding stopped. We calculated sample size based on a previous study. 34 To detect an increase of carotid wall thickness between the sham control group and the heating or sonication group with a two-sided 5% significance level and 80% power, a sample size of eight mice per group was necessary, given an anticipated dropout rate of 20%. rCBF of the left frontoparietal cortex region was measured before, immediately after, and 1, 2, 3, 4, 5, 6, 7 days after heating or sonicating through the burr hole by using a laser blood flowmeter (Flo-C1; Omegawave, Tokyo, Japan).
Tissue preparation
A lot of preclinical studies have demonstrated that female rats and mice sustain less tissue damage compared to males under an equivalent insult from cerebral ischemic injury. 35 Therefore, we used only males to minimize individual variation. We used 132 rats in this study, five of which were excluded based on the following exclusion criteria: three rats that died as a result of a procedural problem during surgery, one that failed to reach 55 °C with the thermometer, and one whose bleeding from CCA after sonication could not be stopped.
At 1 day or 7 days after heating or sonicating procedures, blood samples were collected by cardiac puncture under deep anesthesia with a mixture of nitrous oxide/oxygen/isoflurane (69%:30%:1%). Each rat was transcardially perfused with ice-cold phosphate-buffered saline (PBS), then with ice-cold 4% paraformaldehyde (PFA) in 0.1 mol/L phosphate buffer. The whole brain and bilateral CCAs were removed and immersed in the same fixative for 24 h at 4 °C. After washing with PBS, tissues were sequentially transferred into 10, 20 and 30% (w/v) sucrose solutions. Each CCA was embedded in Optimal Cutting Temperature Compound (Tissue Tek 4583, Finetek USA, Inc., Torrance, CA, USA) in powdered dry ice and stored at −80°C. Sections (10 µm thick) were prepared using a cryostat (HM525 NX; Thermo Fisher Scientific, Waltham, MA, USA) at −25°C and mounted on silane-coated glass slides (MAS-GP, typeA; Matsunami Glass Ind., Ltd., Osaka, Japan).
Histology and single immunohistochemistry analysis
CCA sections were cut at 6, 5, 4, 3, 2, 1, and 0 mm proximal from the bifurcation, then hematoxylin and eosin (HE) staining was performed to evaluate the vessel wall structure of the CCA. In addition, brain sections were cut and stained with cresyl violet and Nissl, and the ischemic lesion area was examined by microscopy (SZX-12; Olympus Optical, Tokyo, Japan).
It has been reported that endogenous rat IgG is immunolocalized in areas where blood-brain barrier (BBB) breakdown occurs. 36 To estimate levels of rat IgG, sections were incubated with biotin-labeled rabbit anti-rat IgG antibody (1:500; Vector Laboratories, Burlingame, CA) followed by horseradish peroxidase streptavidin–biotin complex solution (Vectastain ABC kit; Vector Laboratories) and diaminobenzidine tetrahydrochloride (DAB tablets; Wako Chemicals, Richmond, VA; 045-22833).
For immunohistochemistry of the brain and CCA, we used the following commercial primary antibodies: goat anti-glial fibrillary acidic protein (GFAP) antibody (1:1000; Abcam; Cambridge, UK; ab53554), rabbit anti-ionized calcium-binding adaptor molecule-1 (Iba-1) antibody (1:1000; Wako Chemicals; 019-19741), rabbit anti-PECAM-1 (CD31) antibody (1:2000; Abcam; ab182981), rabbit anti-von Willebrand factor (vWF) antibody (1:200; Abcam; ab6994), mouse anti-HSP72 antibody (1:100; Abcam; ab2728), and rabbit anti-MCP-1 antibody (1:100; Abcam; ab246795). Sections were then incubated with an appropriate biotin-labeled secondary antibody (1:500, Vector Laboratories) followed by the ABC kit and DAB. A set of negative control sections were also stained using the same protocol but without the primary antibody.
Semiquantitative analysis
For semi-quantitative evaluation of rat IgG, GFAP, and Iba-1, stained sections of the caudate putamen (0.2 mm rostral to the bregma) 37 were selected, and the area of ipsilateral (left) cerebral cortex in each section was chosen randomly. To evaluate CD31, vWF, HSP72, and MCP-1 staining intensity, stained sections of the left CCA cut at 3 mm proximal to the bifurcation were selected from each rat. Then images were captured at ×200 or ×400 magnification by light microscopy (BX51, Olympus, Tokyo, Japan). Staining intensity was measured with image processing software (Scion Image, Scion Corp., Frederick, MD, USA).
Statistical analysis
The investigators who conducted the operations and immunohistochemical analysis were blinded to the treatment assignment. Data were analyzed using statistical software (SPSS v.22.0; IBM Corp., Armonk, NY, USA). All data are expressed as the mean ± standard deviation (SD). Based on the Shapiro–Wilk normality test, differences between three or more groups were assessed using one-way analysis of variance (ANOVA) followed by Dunnett's test for normally distributed data, and with the Kruskal–Wallis variance analysis test for non-normally distributed data. P < 0.05 was considered statistically significant.
Results
In vitro studies
Heating clots in Eppendorf tubes
Clot hardness was 10.4 ± 2.4 (mean ± SD) g/mm2 for the artificial clot and 1.6 ± 0.50 g/mm2 for the rat blood clot (Figure 1(i)).
After heating artificial clots in a constant temperature bath for 30 min, the weight of clots decreased from 30.3 ± 8.0 to 27.7 ± 6.2 mg at 45 °C (n = 3, 8.8% reduction), from 29.7 ± 6.5 to 20.0 ± 2.9 mg at 55 °C (n = 3, 32.6% reduction), and from 31.3 ± 5.2 to 11.0 ± 1.6 mg at 65 °C (n = 3; 64.9% reduction) (Figure 2(a-1)). The total amount of eluted protein increased from clots by heating in accordance with temperature and time (n = 3, 45 °C; n = 7, 55 °C; n = 7, 65 °C; Figure 2(a-2)).
Recanalization of thrombotic occlusions by photic heating
In the in vitro thrombotic occlusion model, based on spot heating, we observed a reduction in clot volume and clots moved to the distal side, followed by recanalization of the occlusion (Figure 2(a-3), Video S1). Recanalization time was shortened when water pressure increased, i.e., 1 min 25 sec on average for BP of 200 mmHg (n = 2), 4 min 16 sec for BP of 150 mmHg (n = 3), and 5 min 34 sec for BP of 100 mmHg (n = 2) (Figure 2(a-4) and (b) to (d)).
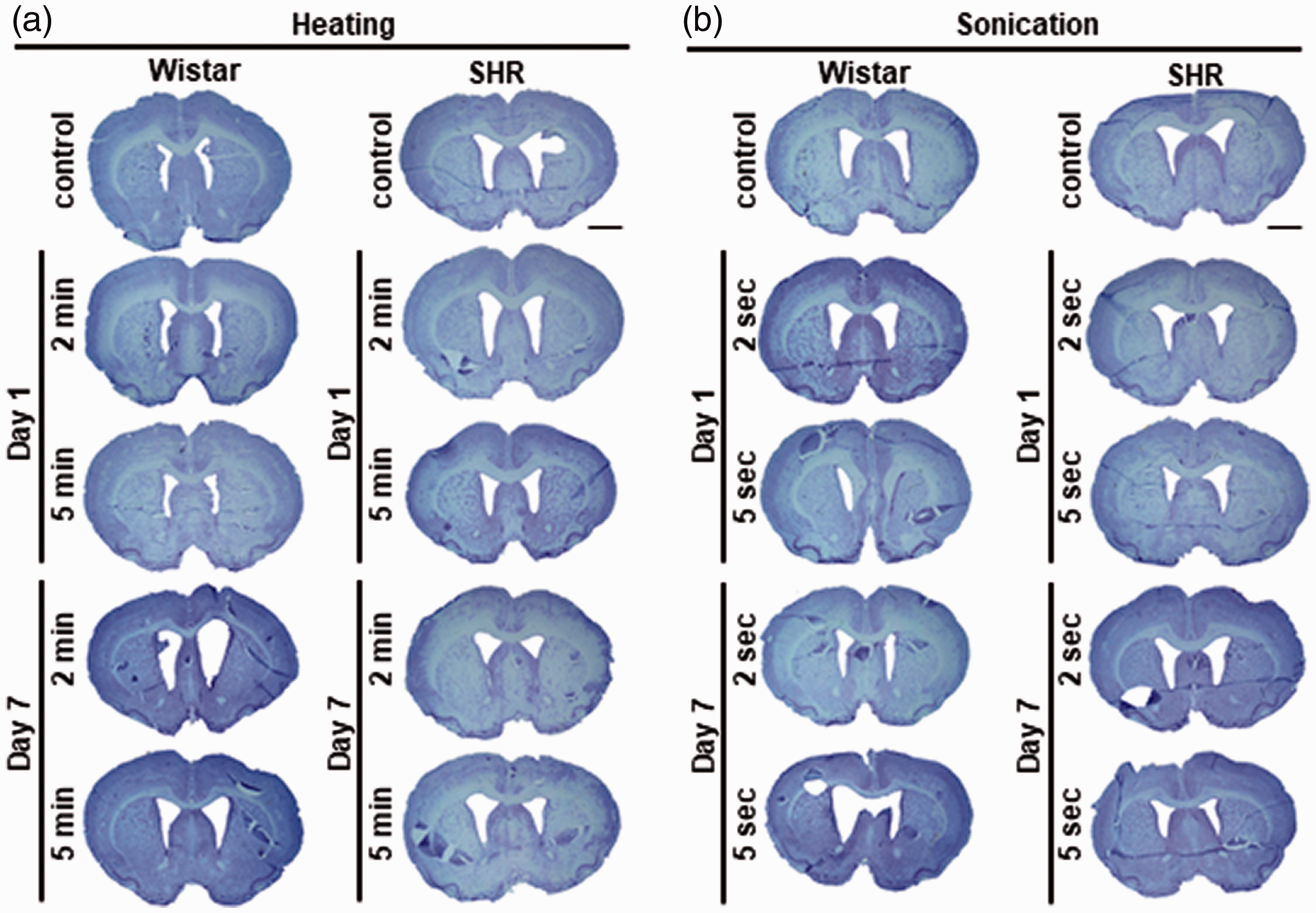
No strokes or hemorrhages in surviving rats. No brain infarct lesions were detected by Nissl staining in both heating (a) and sonication models (b). The same animals were used as the sham control in the heating and sonication experiments. Scale bar: 2 mm.
We compared and confirmed that the curve in the temperature rise was similar for the probe placed within and at the rear of the tube (Figure 2(a-4) and (a) to (d)). Temperature reached about 55 °C within 1 to 3 min, and was maintained until recanalization occurred. Thus, the thermometer probe was placed on the rear side of the tube in order to apply correct gravitational pressure (Figure 2(a-3)). Based on this result, we adopted a temperature of 55 °C for 2 or 5 min in the following in vivo heating CCA model in animals.
Sonicating clots in Eppendorf tubes
By sonicating at minimum output using 10 W of ultrasound irradiation (or a maximum of 200 W), the weight of artificial clots decreased from 33.3 ± 0.5 to 28.0 ± 2.2 mg for 2 sec sonication (n = 3, 15.9% reduction), from 33.0 ± 2.4 to 20.7 ± 4.2 mg for 5 sec (n = 3, 37.3% reduction), and from 33.0 ± 4.1 to 16.0 ± 4.3 mg for 10 sec (n = 3, 51.5% reduction) (Figure 2(b-2)). Based on this result, we adopted an output of 10 W for 2 or 5 sec in the following in vivo sonication CCA model in animals.
In vivo studies
Physiological parameters in the rat heating CCA model
Most physiological parameters did not show significant differences between the control and heating for 2 and 5 min rat groups sacrificed on day 7 except for the following results (Fig. S1A): Body temperature (BT) on days 2 and 3 for SHR (p = 0.045, p = 0.0019, respectively) and on day 5 for Wistar rats (p = 0.033; Fig. S1A-b); systolic blood pressure (SBP) before heating, on days 1, 5 and 7 for Wistar rats (p = 0.021, 0.002, 0.008, 0.003, respectively; Fig. S1A-c); diastolic blood pressure (DBP) before heating for SHR (p = 0.024; Fig. S1A-d); heart rate (HR) on days 1 and 2 for SHR (p = 0.027, p = 0.012, respectively; Fig. S1A-e); CBF before heating, on days 4 and 7 for SHR (p = 0.031, p = 0.0074, p = 0.034, respectively; Fig. S1A-f).
Survival rates of rats sacrificed on day 7 in the control and heating for 2 and 5 min rat groups were 100% (n = 9), 100% (n = 9), and 83.3% (n = 10 out of 12) for Wistar rats, and 100% (n = 7), 100% (n = 5), and 100% (n = 5) for SHR (Figure 3(a-1)). We dissected the brains of rats that died after 5 min heating in the period leading up to sacrifice (n = 2) and detected edema (Fig. S2A-1, a, b, arrowheads). There was no significant difference in white blood cell (WBC) count between the control and heating groups that were sacrificed on 1 or 7 days for both Wistar rats and SHR (Fig. S2A-2).
Physiological parameters in the rat sonicating CCA model
Most physiological parameters did not show significant differences between the control and sonicating for 2 and 5 sec rat groups sacrificed on day 7 except for the following results (Fig. S1B): BT on day 3 for SHR (p = 0.0080; Fig. S1B-b); SBP on days 1, 3, and 7 for Wistar rats (p = 0.005, p = 0.0091, p = 0.003, respectively; Fig. S1B-c); DBP before sonication for SHR and on day 1 for Wistar rats (p = 0.021, p = 0.00014, respectively; Fig. SIB-d); HR on day 2 for Wistar rats (p = 0.047; Fig. S1B-e); CBF before sonication and on day 4 for SHR (p = 0.011, p = 0.020, respectively; Fig. S1B-f).
Survival rates of rats sacrificed on day 7 in the control and sonicating for 2 and 5 sec rat groups were 100% (n = 9), 100% (n = 8), and 80% (n = 10 out of 12) for Wistar rats, and 100% (n = 7), 100% (n = 5), and 100% (n = 5) for SHR (Figure 3(b-1)). We dissected the brains of rats that died after 5 sec sonication in the period leading up to sacrifice (n = 2) and detected findings suggestive of cerebral hemorrhage (Fig. S2B-1, a) and subarachnoid hemorrhage (Fig. S2B-1, b, arrows). There was no significant difference in WBC between the control and sonication groups that were sacrificed on 1 or 7 days for both Wistar rats and SHR (Fig. S2B-2).
Nissl staining and immunohistochemical analysis of the brain
No brain infarct lesions were detected by Nissl staining in both heating and sonication models (Figure 4(a) and (b)). BBB damage and breakdown leads to endogenous rat IgG leaking from blood vessels into brain parenchyma. 36 There was no difference between the control and heating groups or among the sonication groups for both Wistar rats and SHR regarding IgG DAB staining (Figure 5(a) and (b)). Similarly, no significant differences in GFAP and Iba-1 immunoreactivity could be found between the experimental groups (Fig. S3 and S4).
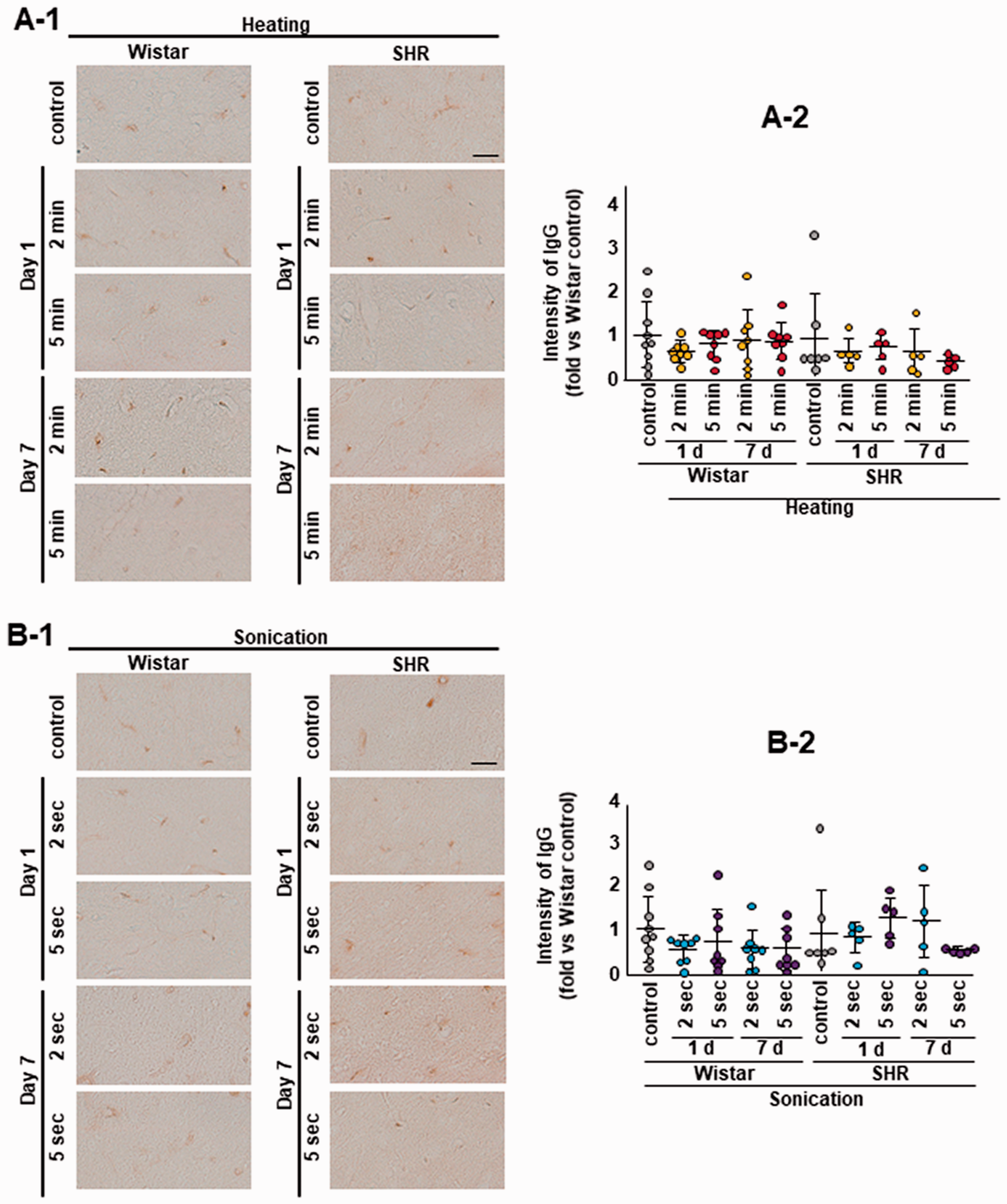
No BBB breakdown after heating or sonication. There was no difference in IgG DAB staining between the control and heating groups (a) or among the sonication groups (b) for both Wistar rats and SHR. The same animals were used as the sham control in the heating and sonication experiments. Scale bar: 50 µm.
HE staining and immunohistochemical analysis of CCA
We then examined HE staining of seven CCA cross sections at 6, 5, 4, 3, 2, 1, and 0 mm proximal to the bifurcation (Figure 6(a-1) and (b-1)). The biggest change in vessel wall structure was observed in the section 3 mm proximal to the bifurcation. Consequently, the average maximum thickness of vessel walls was measured in three cross sections, 4, 3, and 2 mm proximal to the bifurcation. One animal showed neointimal hyperplasia after sonication (Figure 6(b-1), arrows), although there were no significant differences in vessel thickness between the control and heating groups or among the sonication groups for both Wistar rats and SHR (Figure 6(a-2) and (b-2)).
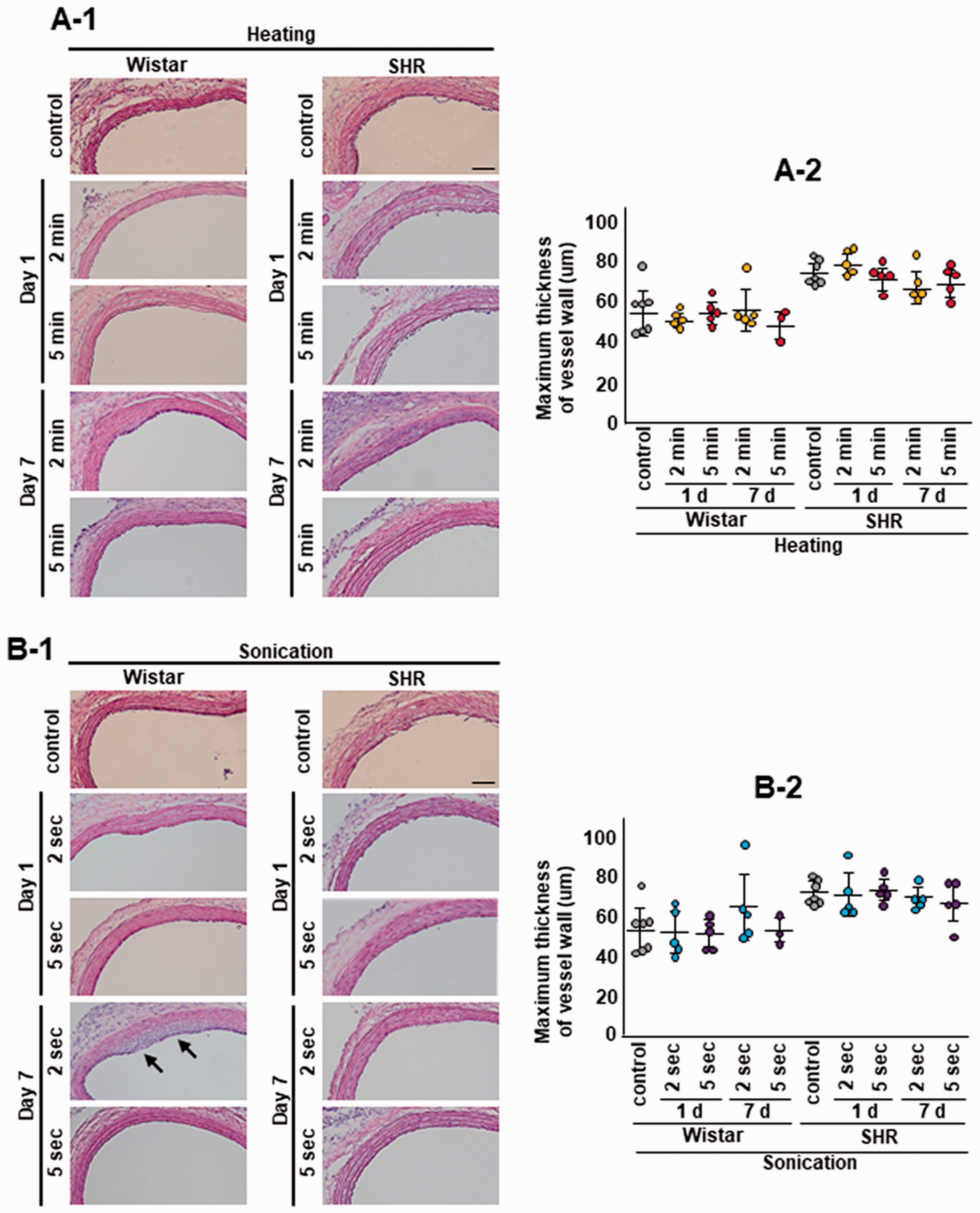
Carotid wall thickness after heating and sonication. One animal showed neointimal hyperplasia after sonication (b-1, arrows) but there was no significant difference in vessel thickness between the control and heating groups or the sonication groups for Wistar or SHR (a-2 and b-2). The same animals were used as the sham control in the heating and sonication experiments. Scale bar: 200 µm.
After CD31, vWF, HSP72, and MCP-1 staining, no positive cells were detected in samples without primary antibody (negative controls; data not shown). CD31 immunoreactivity for the endothelium of the luminal wall tended to be lower after heating in SHR (Figure 7(a)) and was considerably reduced and distributed discontinuously after sonication in SHR (p = 0.039, control vs 2 sec sonication sacrificed on day 1; p = 0.0042, control vs 5 sec sonication sacrificed on day 1; Figure 7(b)). vWF immunoreactivity was significantly lower in the 5 min heating Wistar rat group compared with the control group (p = 0.034; Fig. S5A). HSP72 immunoreactivity tended to be higher in the heating SHR group sacrificed on day 1 (Fig. S6A) and was significantly higher in the 5 sec sonication SHR group sacrificed on day 1 compared with the control group (p = 0.043; Fig. S6B). There were no differences in MCP-1 staining between the control and heating groups or among sonication groups for Wistar rats or SHR although MCP-1 expression tended to be higher in the heating SHR group sacrificed on day 1 (Fig. S7A).
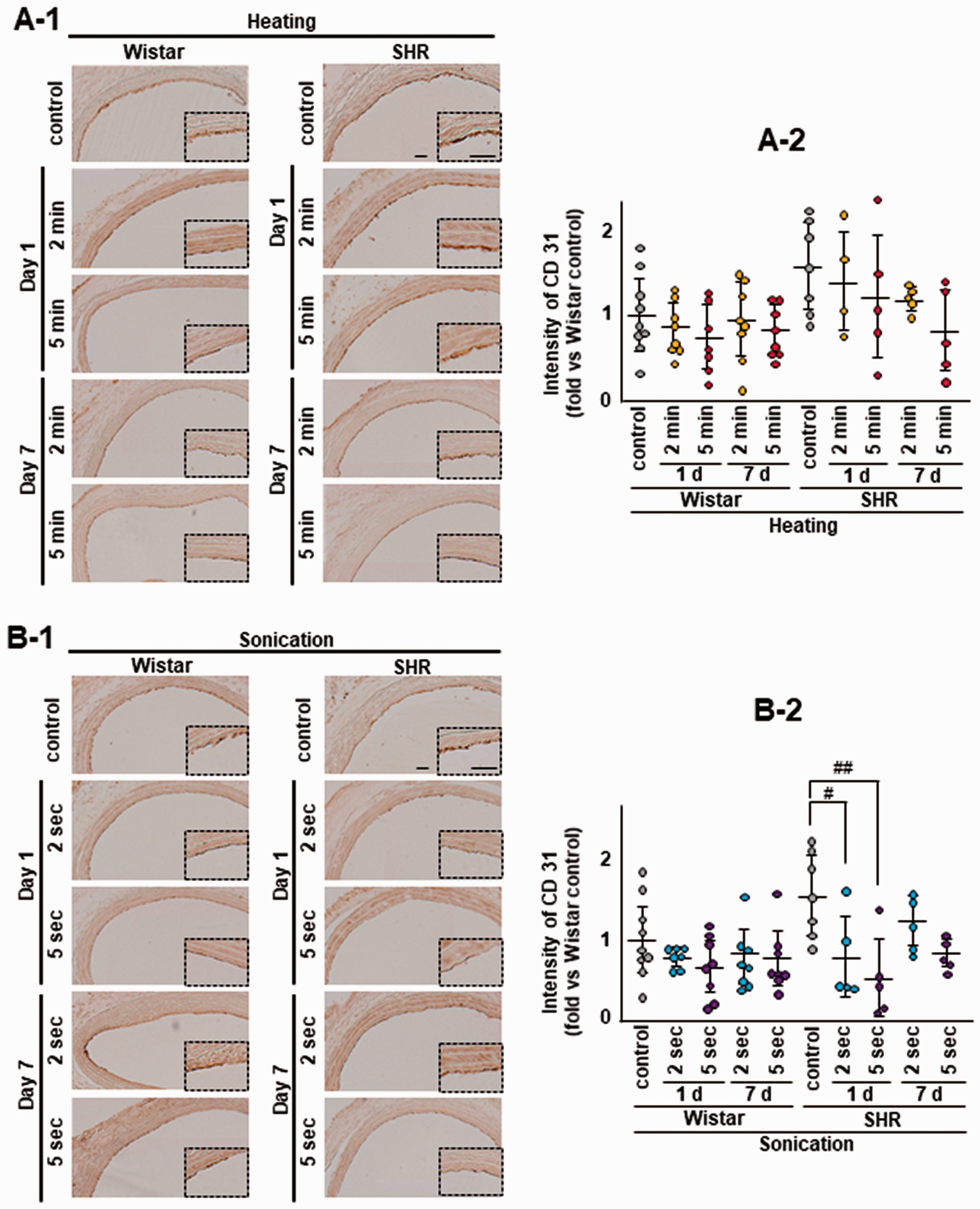
CD31 reduction after heating and sonication. CD31 immunoreactivity for the endothelium showed a decreasing trend for CD-31 (without significance) after heating in SHR (a-1 and a-2). CD31 immunoreactivity was remarkably reduced and distributed discontinuously after sonication in SHR (#p = 0.039, control vs 2 sec sonication sacrificed on day 1; ##p = 0.0042, control vs 5 sec sonication sacrificed on day 1; b-1 and b-2). The same animals were used as the sham control in the heating and sonication experiments. Scale bar: 100 µm.
Discussion
In the present study, we introduced a novel protocol of blood clot formation by using fibrin glue, Bolheal, whose compounding ratio was determined by an optimization study that reported that adhesion and cross-linking of fibrin was maximized by more than 8% fibrinogen, 30–500 U/mL of thrombin, 50–100 U/mL of coagulation-factor XIII, 1000–5000 KIE/mL of aprotinin, and 40 mM calcium hydrochloride. 38 Therefore, our artificial clot can introduce maximum clot stability and stiffness compared to a traditionally prepared blood clot. In the present study, hardness of this artificial clot was much stronger than the traditional rat blood clot (Figure (1i)), indicating that this artificial clot is more resistant to thrombolysis. It also has additional advantages: simple preparation, long lasting, and easy to make thrombotic occlusions because of its morphological stability.
Although several studies observed temperature-dependent clot lysis during ultrasound exposure,23–26 the thermal effect was not measured directly or was not measured solely in the absence of other effects such as cavitation and acoustic streaming. The present study showed a relationship between lysis of artificial clots and temperature (Figure 2(a-1) and (a-2)) and recanalization of mimicked thrombotic occlusion by spot heating (Figure 2(a-4) and Video S1) probably due to fibrin denaturation at high temperature. 39 In fact, heating at various temperatures disaggregated fibrin clot strands, as observed by scanning electron microscopy. 40
In the present in vivo heating experiment, two out of 12 Wistar rats exposed to 5 min heating died in the period leading up to sacrifice (Figure 3(a-3)). The surviving rats did not show any brain infarct (Figure 4(a)), BBB damage (Figure 5(a)), astrocyte responses (Fig. S3A), microglial activation (Fig. S4A), or carotid wall thickness (Figure 6(a)). Although a significant decrease was not observed, the heating model in SHR showed a decreasing trend for CD-31 as a marker of the loss of endothelial junctional integrity (Figure 7(a-2)). vWF decreased significantly with heating in the heating Wistar rats group (Fig. S5A), suggesting that less activity or lower production of this clotting factor occurs in response to the loss of endothelial cell function. Similarly, although there were no significant differences, the heating model showed an increasing trend for HSP72 as a marker of heat stress (Fig. S6A-2) and for MCP-1 as a marker of the inflammatory response (Fig. S7A-2). These results suggest that 55 °C heating may effectively induce thrombolysis, but safety of the heating treatment was not fully supported by the data.
For the present in vitro thrombolysis with ultrasound sonicator, we observed a loss in weight of clots after 2 or 5 sec sonication, even with the lowest output, i.e., 10 W (Figure 2(b-2)). In the in vivo sonication experiment, two out of 10 Wistar rats exposed to 5 sec sonication died in the period leading up to sacrifice (Figure 3(b-3)). The surviving rats did not show any infarct lesions (Figure 4(b)), BBB damage (Figure 5(b)), astrocyte responses (Fig. S3B), or microglial activation (Fig. S4B). The carotid wall thickened in one animal (Figure 6(b-1), arrows), endothelial junctional integrity significantly decreased (Figure 7(b-2)), and HSP72 immunoreactivity was induced (Fig. S6B-2). These results suggest that 20 kHz sonication may effectively induce thrombolysis, but it could be invasive to the cervical endothelium and brain. In previous studies, therapeutic ultrasound for thrombolysis utilized a transmission frequency of around 220 kHz to 2 MHz.12,19,20 On the other hand, for the present in vivo thrombolysis, we used a 20 kHz ultrasound sonicator. Lower frequency produces larger cavitation bubbles and their resulting implosions become more energetic. Therefore, a lower frequency may be a more effective way to induce thrombolysis, but it may also be more invasive to blood vessels.
The present study has other limitations. First, the heating and sonication models in the present study can cause thermal or mechanical injury to non-targeted brain tissue because of wide directionality of the spot heater and the 20 kHz sonicator. Second, heating itself could damage brain tissue. In patients having suffered an acute ischemic stroke, the benefit of treatment with induced hypothermia is uncertain, 41 but a recent study reported that intra-arterial brain cooling improved outcomes in an animal stroke model. 42 Third, the sample size of SHR was small, which probably reduces the power of the study. Although there was no significant difference, CD31 tended to be lower (Figure 7(a-2)), HSP-72 tended to be higher (Fig. S6A-2), and MCP-1 tended to be higher (Fig. S7A-2) in the SHR heating model. Since small sample size increases the margin of error, it cannot be concluded that there are no difference as for these endothelial cells marker. Moreover, clot fragmentation and delayed neointimal hypertrophy are still safety concerns.
In conclusion, the present spot heating and ultrasound irradiation models seem to be effective for disintegrating clots in vitro, but safety of the in vivo model was not fully supported by the data. However, the data indicates that a shorter time exposure could be less invasive than a longer exposure. ExAblate 4000 type transcranial MRgFUS can be applied to treat thrombosis.12,13,15 It is comprised of 1000-array element transducers that focus ultrasound waves into a designated lesion, producing a sharp focus within a 2–3 mm radius. In addition, 200 kHz to 1.5 MHz frequency of MRgFUS allows a higher thermal effect, sharper directionality (reduction of focal spot) of ultrasound, and is less invasive to an organism because of weaker cavitation. In a future experiment, we plan to investigate the efficacy and safety of both in vitro and in vivo sonothrombolysis by MRgFUS by monitoring changes in temperature.
Early reperfusion therapies using rt-PA and endovascular therapy have become a standard treatment for acute ischemic stroke, but there are still several limitation and problems. First, reperfusion of the occluded arteries by rt-PA induces disruption of the neurovascular unit and BBB injury which cause intracranial hemorrhage.3,43,44 Many studies have assessed the factors that are associated with BBB integrity and hemorrhagic transformation after rt-PA.45–48 Second, data on the efficacy of thrombolysis with rt-PA in mild patients with a non-disabling deficit 49 or disability-producing symptoms 50 are limited. Third, mechanical thrombectomy may also cause direct vessel damage and BBB disruption.51,52 Fourth, it was reported that overestimated cases of a modified Thrombolysis in Cerebral Infarct (mTICI) score may have been amenable to further thrombectomy. 53 Sonothrombolysis can cover the shortcomings of r-tPA thrombolysis and endovascular therapy and may provide another option to thrombobusters, having the advantage of being faster, and having lower costs and smaller side effects.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221079127 - Supplemental material for Efficacy and safety of spot heating and ultrasound irradiation on in vitro and in vivo thrombolysis models
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221079127 for Efficacy and safety of spot heating and ultrasound irradiation on in vitro and in vivo thrombolysis models by Ryuta Morihara, Toru Yamashita, Yosuke Osakada, Tian Feng, Xinran Hu, Yusuke Fukui, Koh Tadokoro, Mami Takemoto and Koji Abe in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly supported by a Grant-in-Aid for Scientific Research (C) 20K09370, 20K12044, Challenging Research 21K19572, Young Research 20K19666, 21K15190, and by Grants-in-Aid from the Research Committees (Toba K, and Tsuji S) from the Japan Agency for Medical Research and Development.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
KA designed the research; RM, YO, TF, XH, YF, and KT performed the experiments; RM analyzed the data; RM and KA wrote the manuscript; RM and TY revised the manuscript, RM, YF, MT, TY, and KA obtained funding.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
