Abstract
It is commonly believed that cerebrospinal fluid (CSF) movement is facilitated by blood vessel wall movements (i.e., hemodynamic oscillations) in the brain. A coherent pattern of low frequency hemodynamic oscillations and CSF movement was recently found during non-rapid eye movement (NREM) sleep via functional MRI. This finding raises other fundamental questions: 1) the explanation of coupling between hemodynamic oscillations and CSF movement from fMRI signals; 2) the existence of the coupling during wakefulness; 3) the direction of CSF movement. In this resting state fMRI study, we proposed a mechanical model to explain the coupling between hemodynamics and CSF movement through the lens of fMRI. Time delays between CSF movement and global hemodynamics were calculated. The observed delays between hemodynamics and CSF movement match those predicted by the model. Moreover, by conducting separate fMRI scans of the brain and neck, we confirmed the low frequency CSF movement at the fourth ventricle is bidirectional. Our finding also demonstrates that CSF movement is facilitated by changes in cerebral blood volume mainly in the low frequency range, even when the individual is awake.
Introduction
The glymphatic system plays an important role in the clearance of metabolic waste products in the extracellular, interstitial space1,2 and the distribution of homeostasis-sustaining compounds (e.g., glucose) in the brain. 3 Recent advancements in understanding the glymphatic system highlight its role in the development of neurodegenerative diseases.4–6 The ventricles of the brain are a connecting network of cavities filled with cerebrospinal fluid (CSF). The ventricular system is composed of 2 lateral ventricles, the third ventricle, the cerebral aqueduct, and the fourth ventricle. The choroid plexuses of the ventricles produce CSF, which fills the ventricles and subarachnoid space. The circulation of CSF in the brain involves constant production and reabsorption. 7 Unlike blood flow, which is driven by the heart pumping, CSF does not appear to have a single engine to generate flow. 8 CSF movement has been assumed to be mostly facilitated by the mechanisms associated with vessel wall movement, including vasomotion,9,10 pulsation, 11 inspiratory thoracic pressure changes,12,13 and/or osmotic pressure. 14
Several magnetic resonance imaging (MRI) techniques have been used to investigate CSF flow in the brain, including 1) phase-contrast imaging; 15 2) velocity density imaging; 16 3) time-spatial labeling inversion pulse imaging; 17 and 4) 4D flow MRI. 18 These imaging techniques were often used to detect the direction of CSF motion within a single cardiac/respiration cycle. However, only a few studies reveal real-time CSF movement over a longer period or bulk flow in a wakeful state. 11
Recently, Fultz et al. used a fast echo-planar imaging (EPI) sequence (TR < 400 ms) to study CSF flow. 19 They ingeniously placed the edge of the imaging volume (the first slice) at the fourth ventricle, which allows them to measure the flow of CSF using the inflow effect in the fMRI signal (rather than blood-oxygen-level-dependent (BOLD) contrast). They discovered a coherent pattern of oscillating electrophysiological, hemodynamic, and CSF dynamics that appears during non–rapid eye movement (NREM) sleep. Their results demonstrated the couplings between CSF movement and the hemodynamic signal, and low frequency electroencephalogram (EEG) during sleep. While still a qualitative approach to the measurement of CSF flux, the advantages of the fast EPI approach are obvious. First, the fast EPI sequence is widely available, and the data processing is straightforward. Second, through simultaneous recording, coupling between brain hemodynamics and CSF signal, which may be crucial in understanding the fundamental driving mechanism of CSF movement, can be examined. In this case, hemodynamics and CSF signal can be assessed by BOLD and in-flow contrast of fMRI, respectively.
While effective and innovate, the study by Fultz et al. left crucial questions unanswered. First, they reported correlations between the derivative of the averaged fMRI signal in gray matter with CSF inflow signal (into the brain), assuming CSF dynamics in the fourth ventricle are driven by changes in cerebral blood volume (CBV). However, due to the limitation of the scan parameters, their method could only assess the inflow of CSF to the brain and not the outflow from the fourth ventricle (i.e., to the neck), We wondered if the same CBV-based mechanism could be demonstrated for CSF outflow. Second, the existence of CBV-CSF coupling was not fully explored during the awake state, when CBV changes may be driven primarily by non-stationary blood gases rather than with widespread electrical activity. 20
The present study was designed to address these questions. First, we proposed a simple mechanical model to explain the relationships between CSF movement and hemodynamic fluctuations in the fMRI signal. Second, we investigated the coupling between brain hemodynamics and CSF movement during the awake state by an fMRI scan. Last, an additional fMRI scan was employed to assess CSF outflow separately, and compare it with the brain hemodynamics. In the rest of the manuscript, we use “CSF movement” to represent the oscillations in CSF through the fourth ventricle, distinct from “CSF flux” which represents the bulk flow. We use the terms inflow and outflow to indicate the relative CSF movement towards brain and neck, respectively, with periodic events such as the cardiac and respiratory cycles, while remaining agnostic on the directionality of the CSF flux.
Material and methods
Model
Even though the brain is not rigid, the skull forms a rigid “container” for the brain. As a result, the volumes of the constituents (blood, CSF, and brain parenchyma) consistently fluctuate to maintain the total volume needed for proper internal pressure—i.e., the Monro-Kellie doctrine. 21 According to this doctrine, the transfer of movements from the arterial walls into the surrounding tissue will eventually cause CSF to flow into and out of the spinal canal.22–24 Multiple mechanisms cause arterial wall oscillations at various frequencies, such as cardiac pulsation (∼1 Hz) and vasomotion (<0.1 Hz). They have all been shown to produce CSF movements.9–13 However, the interpretation of the coupling between hemodynamic and CSF movement using fMRI signal is still not clear.
Here, we offer a general model that explains the coupling between fMRI hemodynamic signals and CSF movement. The model is based on the hypothesis that the cumulative effects of vessel dilations and contractions (pulsation, vasomotion) will exert force on the walls of ventricles (in which there are no blood vessels), especially the lateral ventricles, forcing CSF in and out of the fourth ventricle at the bottom (see Figure 1). In this model, the global mean of fMRI signals (GMS) is used as a surrogate signal to indirectly assess the cumulative effects of vessel dilations and contractions in the brain.20,25,26 Notably, several previous studies had used this method for blood tracking.27–30 In Figure 1, we illustrate volume changes in blood vessels and their consequential CSF movement observed at the fourth ventricle. In detail, the blood-rich region will expand because of vessel dilation. Cerebral arterial tone is mediated by CO2 partial pressure, autoregulatory mechanisms, vasomotion, sympathetic nervous activity, and neurovascular coupling. While veins are not actively controlled, their volume can be affected by a Windkessel effect, or ballooning, caused by dilation of the upstream arteries. 31 Together, these blood volume changes will compress the volume of the lateral ventricle, forcing CSF out of the brain. The reverse happens when the blood vessel volume contracts. However, the key point here is that CSF movement only happens at the transitions, during which the cerebral blood volume (CBV) changes (as shown by the arrows in Figure 1)19. When CBV is stable, no force is exerted on the ventricular wall, thus no flow of CSF. Therefore, the model predicts that CSF moves towards the neck through the fourth ventricle during CBV transition from low to high, and CSF move towards the brain during CBV transition from high to low.
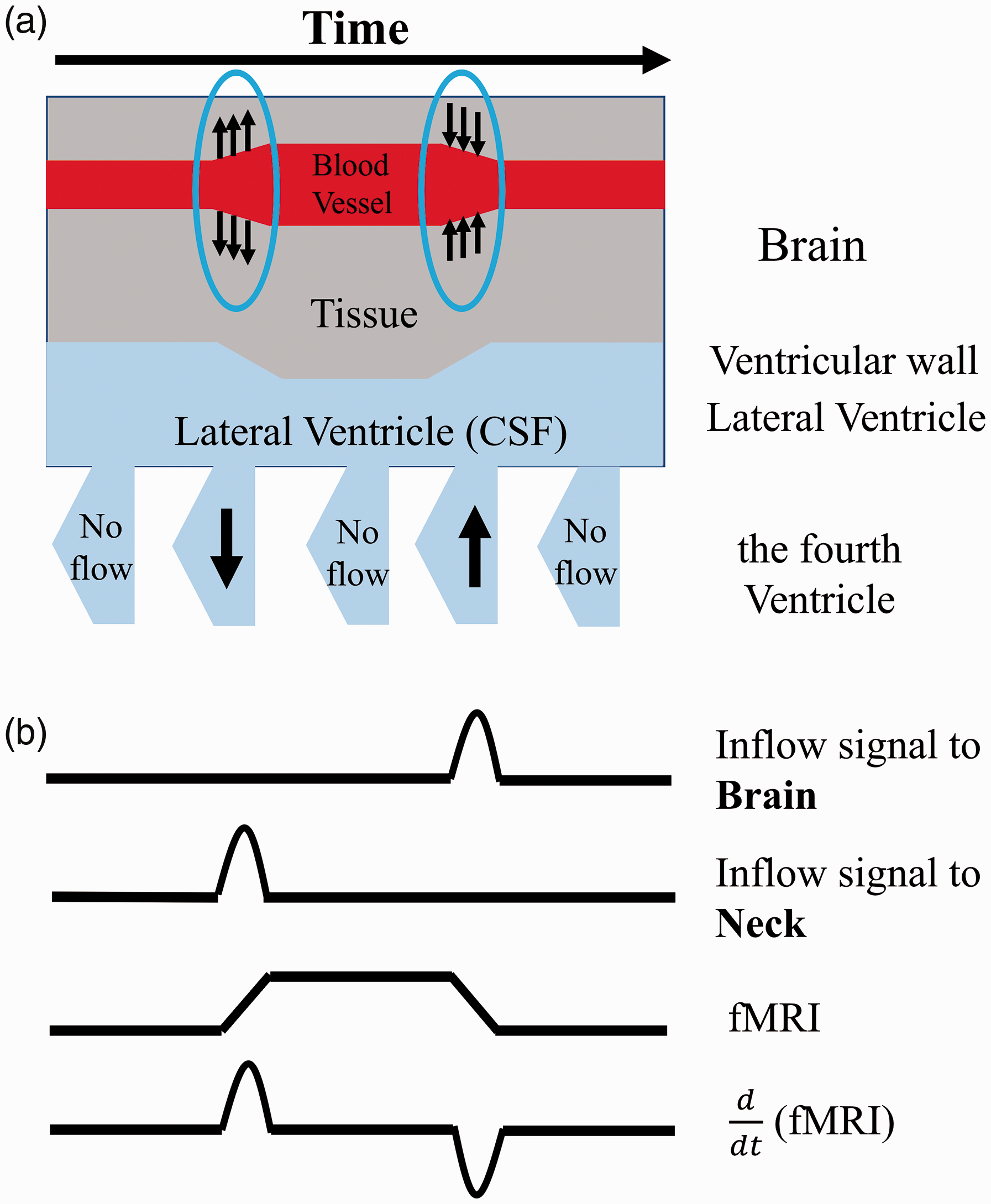
Model describes the derivative of fMRI signals are coupled with CSF Inflow and outflow fluctuations in the fourth ventricle. (a) Main structural of the model with relative brain (blood vessel and tissue) and ventricle structural (lateral and the fourth ventricle). Arrows circled with blue in brain show the vasodilation (arrows going out from blood vessel) and vasoconstriction (arrows going into blood vessel). Arrows in the fourth ventricle indicate the inflow and outflow fluctuation. (b) CSF inflow and outflow signal in the fourth ventricle affected by
Based on this model, we have several predictions (Figure 1): 1) CSF inflow (toward the brain) fluctuation should be negatively correlated with
Experimental design
Structural scans
The study was approved by Purdue University's Human Research Protection Plan (IRB-2019-303) and was conducted in accordance with application of Belmont Report principles (Respect for Persons, Beneficence, and Justice) and federal regulations at 45 CFR 46 and 21 CFR 50 and 56. Informed consent was obtained from all participants. Ten healthy individuals (5 female, 5 male, age range 23–30 years) were recruited. MRI data were obtained using a 3 T SIEMENS MRI scanner (Magnetom Prisma, Siemens Medical Solutions, Erlangen, Germany) and a 64-channel head coil. Each participant underwent one T1-weighted, one T2-weighted, and two functional scans. For T1-weighted scans, Magnetization Prepared Rapid Acquisition Gradient Echo (MPRAGE) structural images were acquired with the following parameters (TR/TE: 2300/2.26 ms, 192 slices per slab, 321 s, flip angle: 8 deg, resolution: 1.0 mm
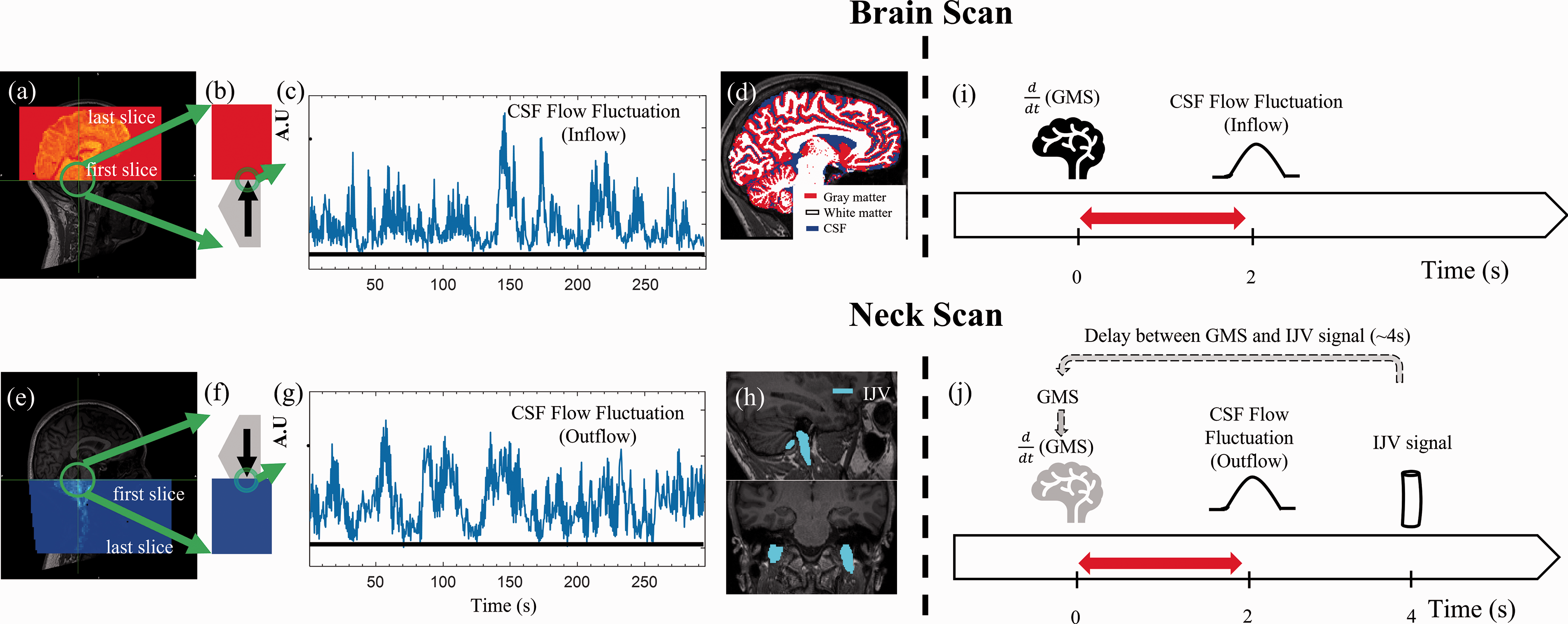
Low frequency oscillations in CSF signals in the fourth ventricle during wakefulness. Example scan positioning for (a) CSF brain scan and (e) CSF neck scan. The red shaded area in (a) and blue shaded area in (e) are the functional image coverages relative to the anatomy. The green circles point out the fourth ventricle. the fourth ventricle was enlarged as gray pentagons in (b) and (f) with arrows indicated CSF flow fluctuation direction and displayed with functional image (red and blue areas). Time series of a single CSF voxel from (c) brain scan and (g) neck scan. Both show large slow fluctuations at rest. (d) Brain segmentations (Gray matter, White matter, and CSF). (h) Blood vessels segmentations (IJV). (i) In the brain scan, CSF flow fluctuation (inflow to the brain) occurs ∼2 s after
EPI scans
The functional resting state (RS) scans were acquired using a multiband echo-planar imaging sequence (FOV = 230 mm, acquisition matrix =92 × 92, 48 slices, voxel size = 2.5 × 2.5 × 2.5 mm3, TR/TE = 440/30.6 ms, echo-spacing = 0.51 ms, flip angle = 35°, hyperband acceleration factor = 8, multi-slice mode: interleaved). Participants were instructed to stay awake during the RS scans, however vigilance was not monitored. CSF inflow fluctuation captured by a fast EPI sequence has been discussed in detail in Fultz et al. 19 In short, as fresh fluid (that has not experienced radiofrequency pulses) move into the imaging volume, it induces higher signal intensity via apparent T1 shortening (i.e., inflow effect). To capture the inflow effect in both directions in the fourth ventricle, two 5-minute RS scans with different regions of interest were included. Both scans are positioned with the edge of the volume placed at the bottom of the fourth ventricle (see Figure 2(a) and (e)), with one scan extending upward toward the top of the head (i.e., “brain scan”) and the other scan extending downward into the neck (i.e., “neck scan”). As a result, CSF inflow to the brain can be assessed by the brain scan, while CSF outflow from the brain can be assessed by the neck scan under the assumption that flow sensitivity in the opposite directions can be ignored due to steady-state excitation in those slices either above or below the slice of interest. The slice of interest, at the fourth ventricle, was consistently acquired first in the TR. The fourth ventricle was chosen for the following reasons: 1) From a physiological standpoint, it represents the conduit for CSF movement between the midbrain and neck. 2) Unlike the lateral ventricles, the fourth ventricle has a narrow shape which restricts the direction of the CSF flow (only allowing vertical movements). Therefore, the first EPI slice can be placed perpendicular to the flow direction to maximize the inflow effect. 3) We chose the middle part of the fourth ventricle over the other narrower parts to increase the likelihood of having some voxels (2.5 mm3) that are fully immersed in the CSF, with low partial volume from surrounding tissues.
Data processing
CSF signals
All MR data were processed using FSL (FMRIB Expert Analysis Tool, v6.01; Oxford University, UK) and MATLAB. For analysis of CSF flow dynamics, a voxel in the fourth ventricle in the edge slice of the EPI data was identified with the help of the T1-weighted image (registered onto the EPI data). The time series of the voxel was extracted to represent CSF flow fluctuation in a certain direction. For CSF inflow fluctuation, the signal was extracted from the bottom EPI slice of the head scan as shown in Figure 2(a) and (b). For CSF outflow fluctuation, the signal was extracted from the top slice of the neck scan as shown in Figure 2(e) and (f). We hereafter refer to these two signals as “CSF inflow fluctuation” and “CSF outflow fluctuation”, even though they both stem from the inflow effect in fMRI.
Preprocessing
Since motion correction cannot be performed accurately on edge slices (given that tissue moves in and out of the imaging volume), only the slice-timing correction was performed prior to CSF signal extraction. 19 To make sure our observations were not due to motion artefacts, validations were performed which can be found in the supplemental materials (supplemental Table S5–7). Then, RS-fMRI data were preprocessed with the steps recommended by Power et al.: 1) slice-timing correction (FSL slicetimer) 2) motion correction (FSL mcflirt) and 3) spatial smoothing with a full width at half maximum (FWHM) of 5 mm isotropic Gaussian kernel was applied to the brain scan. 32 Spatial smoothing was not performed on the neck scan to avoid smoothing effect around the veins in the neck.
Data and statistical analysis
Segmentation was performed on the structural image. Several masks were made to investigate the coupling between CSF inflow fluctuation and fMRI signal from these segmentations (Figure 2(d)). First, anatomical masks for gray matter, white matter, and CSF were made using an automated segmentation program (FSL fast). 33 Second, we identified large veins in the neck (i.e., internal jugular veins (IJVs)) and made corresponding masks (Figure 2(h)). In a previous RS-fMRI study, we found consistent positive correlation between the GMS of the brain and the fMRI signal from the IJVs (IJV signal lags the GMS by 4 seconds). 34 Therefore, here we used the fMRI signal from the IJVs as a surrogate signal of the GMS (i.e., a delayed version) in the neck scan (Figure 2(j)) Signals in IJVs were averaged as one joint IJV signal to improve the signal-to-noise ratio. More information can be found in the discussion.
After extraction, the fMRI signals including the CSF signal (from the fourth ventricle), the GMS from gray matter, white matter, CSF-regions (Figure 2(d)), and signals from big veins (IJV, Figure 2(h)) - were first linearly detrended and then band-pass filtered (0.01 to 0.1 Hz) using a zero delay, fourth-order Butterworth filter to extract the low frequency oscillations (LFO). Cross correlations (MATLAB xcorr, lag range =
Although we used a fast EPI scan with TR = 440 ms, it is possible that the dynamics we observe are caused by aliasing of the cardiac cycle. To investigate pulsatile oscillations, we extracted the full cardiac pulsation waveform from the CSF signal and the brain fMRI scan from an example participant using Happy, which is a method to estimate cardiac pulsation from raw fMRI without additional physiological measurements. 37 The coupling between the CSF and the brain signals at cardiac frequency were assessed. Lastly, the CSF signal powers in cardiac and low frequencies were compared.
Results
CSF flow fluctuation during wakefulness
Figure 2(c) shows an example of CSF flow fluctuation obtained from the brain scan. First, it shows that during an awake state, CSF inflow fluctuation is clearly detectable in agreement with Fultz et al. for deep sleep. 19 Second, similar fluctuation patterns were detected in the neck scan (Figure 2(g)), which indicates the existence of a similar CSF outflow fluctuation.
CSF flow fluctuation and
Figure 3 displays example low frequency data from one research participant. It shows
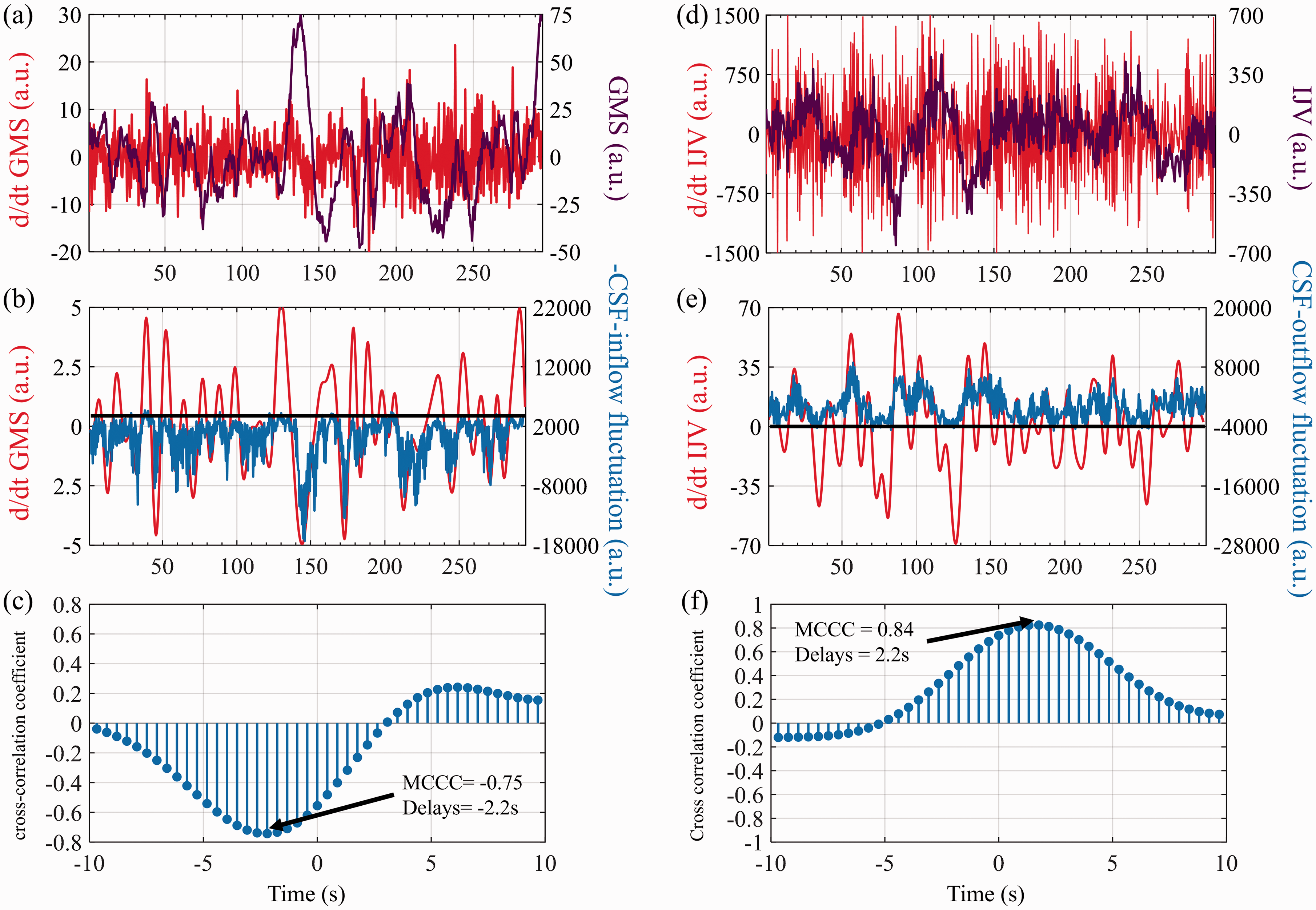
The relationships between global mean signal (GMS) of the brain and CSF inflow fluctuation, and IJV signal (neck) and CSF outflow fluctuation from one research participant. (a) Global mean signal (GMS) and derivative of global mean signal (
Figure 4(a) shows the individual and average results of MCCCs and delays calculated between
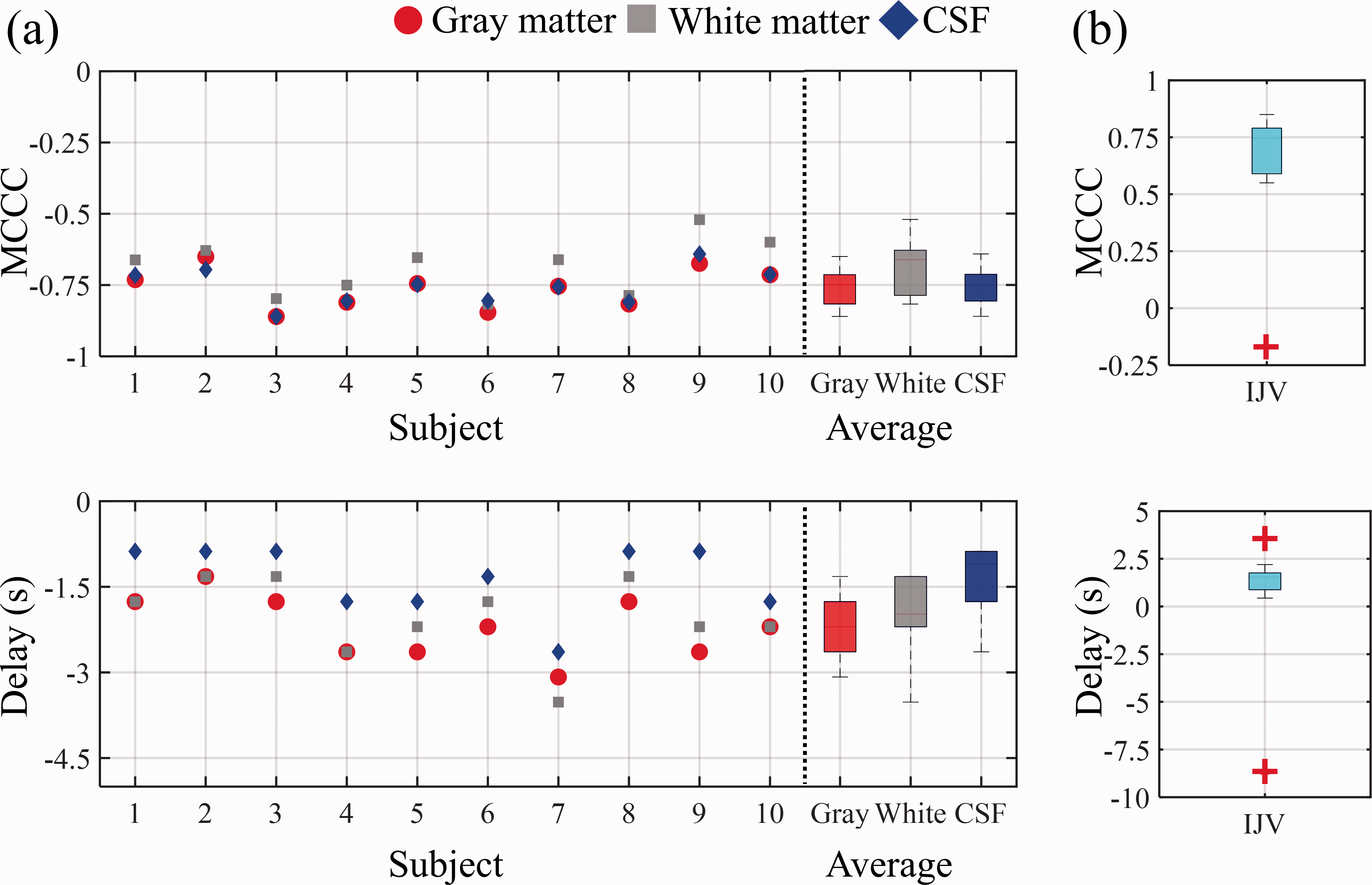
The summary of cross-correlations between CSF inflow fluctuation and
Cross-correlation results between
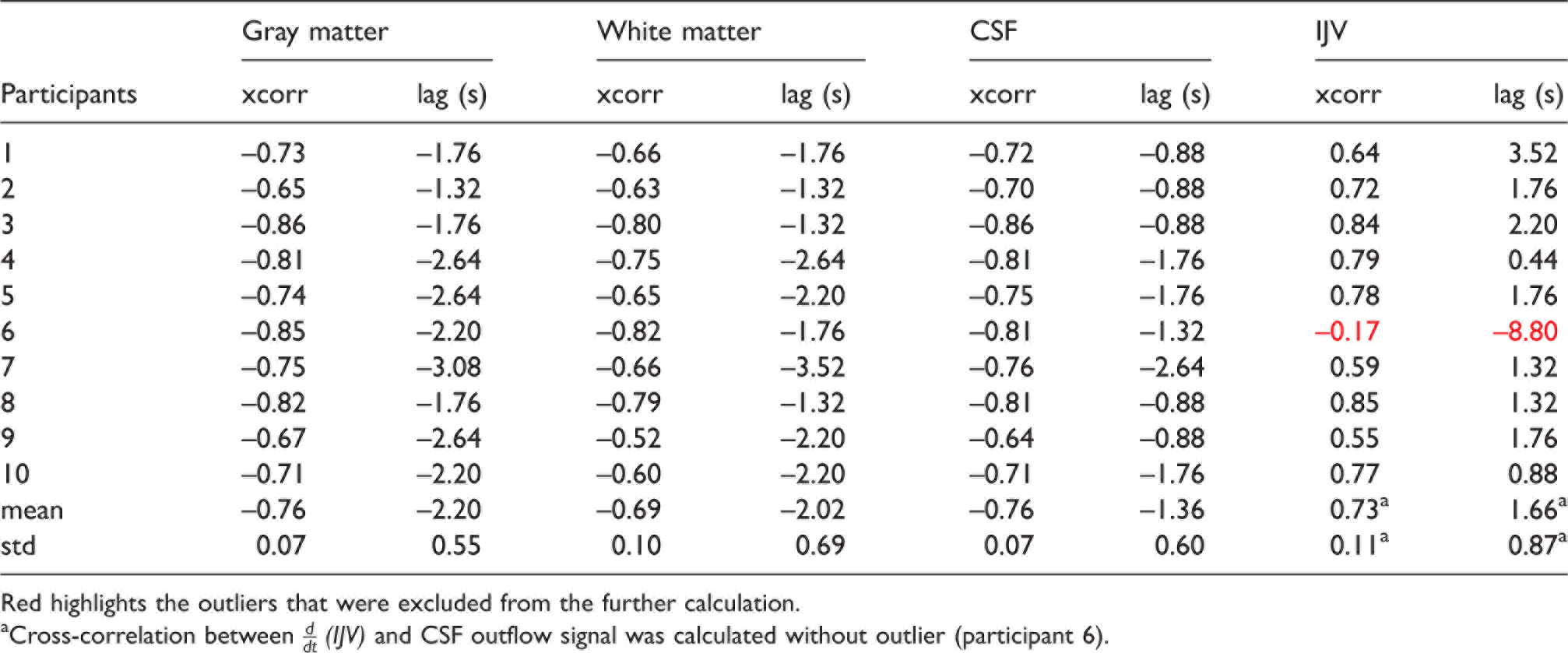
Red highlights the outliers that were excluded from the further calculation.
aCross-correlation between
Spatial-temporal information of the coupling of CSF inflow with the brain signal
Figure 5 shows the averaged MCCCs and delay maps derived from the voxel-wise cross correlation between CSF inflow fluctuation and
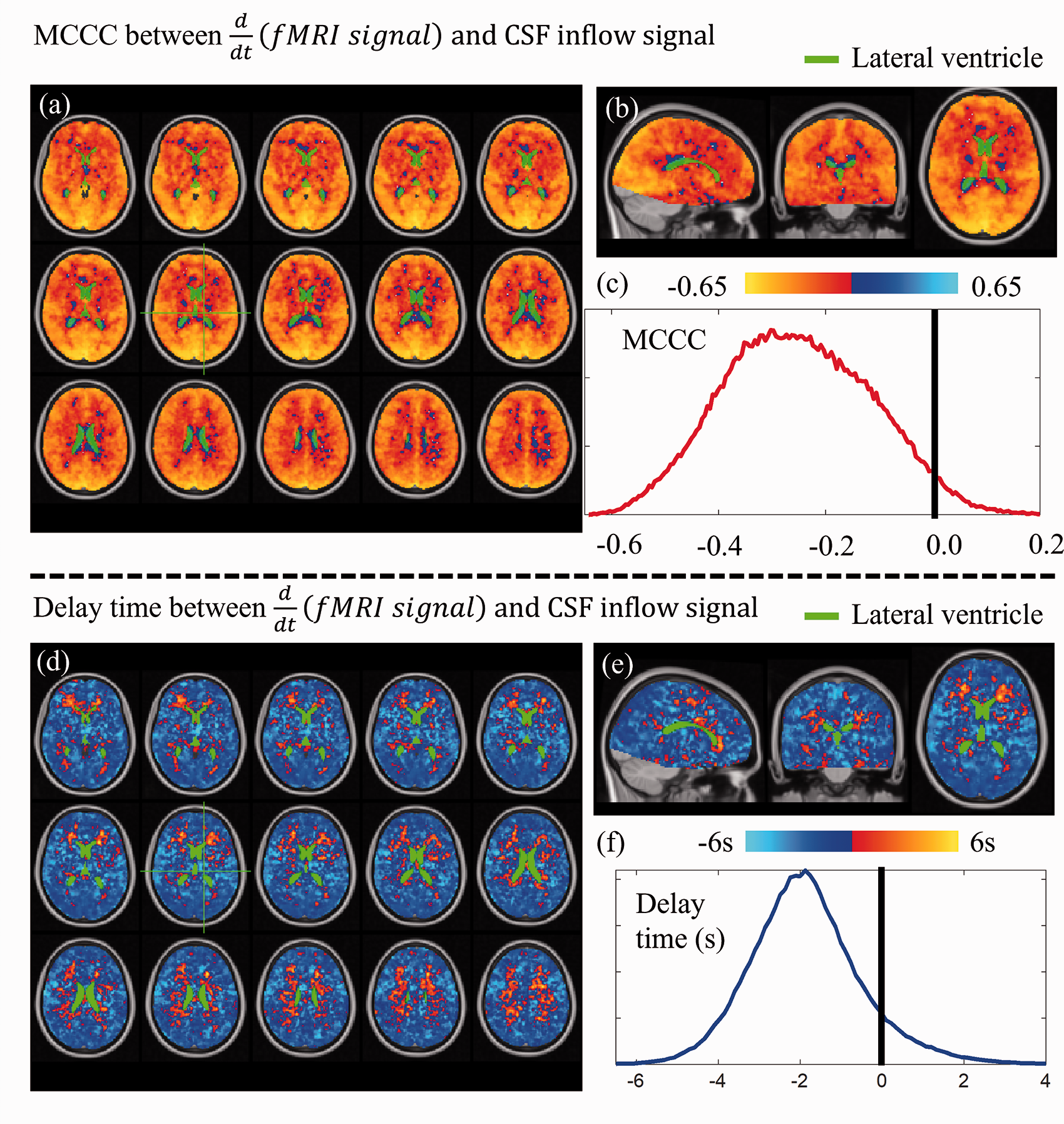
Group average of voxel wise cross-correlation between
Negative delays were found in most of the voxels (cold colors in Figure 5(d) and (e)) with distributions shown in Figure 5(f). Negative delays indicate that
Cardiac pulsation signal found in CSF fluctuation and the brain signal
Figure 6(a) is example data of CSF inflow fluctuation taken from one participant’s head scan and its cardiac component extracted from fMRI data without additional physiological measurement 37 (lower panel). To compare the cardiac components extracted from CSF fluctuation to that from the brain, small sections of both time series were displayed in Figure 6(b). There is no clear time delay between cardiac components extracted from CSF fluctuation and that from the brain. To compare the effect of pulsation and slow oscillation on CSF flow fluctuation, power spectrum of the signal was shown in Figure 6(c), in which the heartbeat signal (∼0.87 Hz) is much smaller than that of LFOs, suggesting negligible contribution of the cardiac cycle on our data.
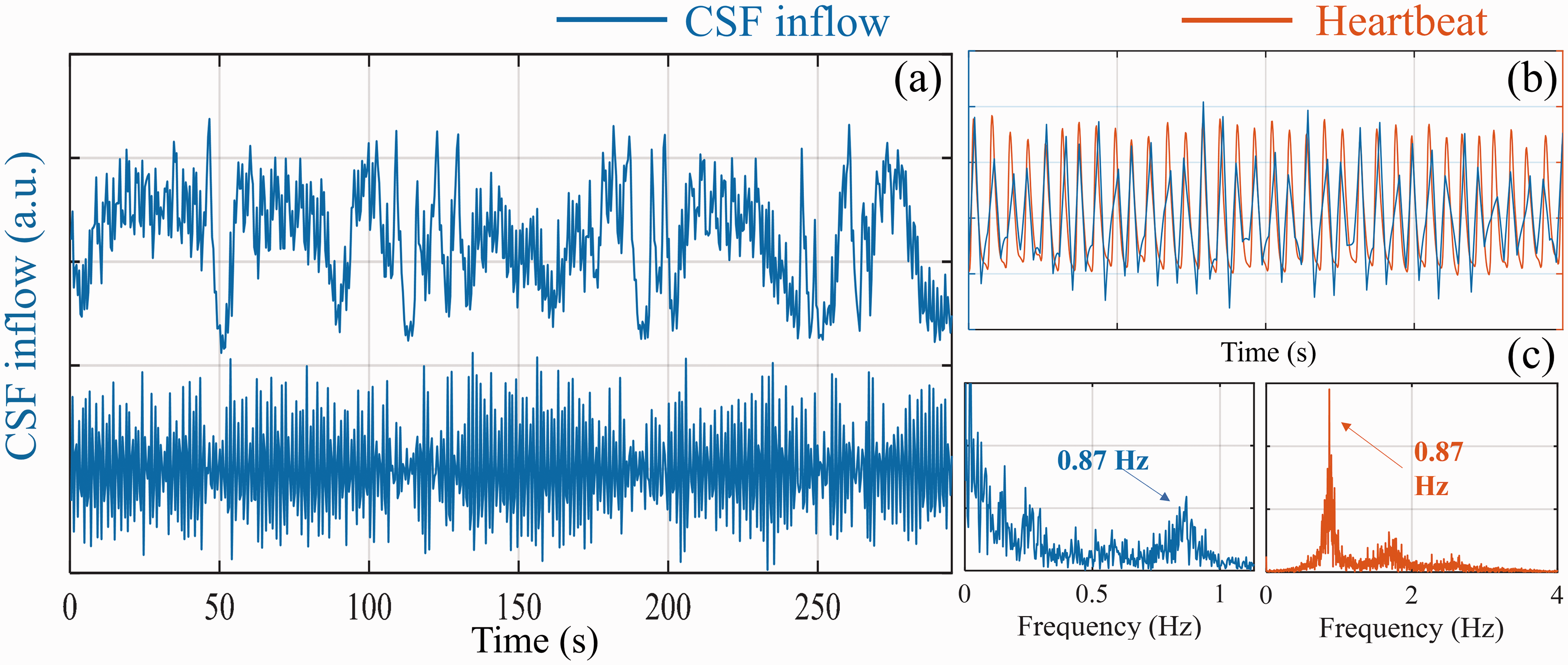
Cardiac pulsation signal found in CSF fluctuation. (a) CSF inflow fluctuation (upper panel: raw data of CSF inflow; lower panel: high-passed CSF inflow fluctuation (>0.6 Hz)). (b) Segment of High passed CSF inflow fluctuation (>0.6 Hz, blue) and heartbeat signal (orange) derived by Happy, which is a method to estimate cardiac pulsation. 37 (c) power spectrum of high frequency CSF inflow fluctuation (>0.6 Hz, left) and power spectrum of heartbeat signal estimated by Happy (right). 37
Discussion
This study used a simple model to explain the correlation and delays between
CSF flow fluctuation detected during wakefulness
Figure 2 shows that CSF inflow and outflow fluctuations were captured by the brain and the neck scans, respectively, during wakefulness. Since the CSF signal originates from an inflow effect that is sensitive to CSF flow in one direction (i.e., into the scan volume), a brain scan can only capture the CSF inflow fluctuations while a neck scan can only capture the CSF outflow fluctuations. The flat sections found in both Figure 2(c) and (g)) indicate the time periods when the CSF flow was in the undetectable direction (e.g., CSF outflow during the brain scan). Two important things can be derived from this observation. First, low frequency CSF movement is bi-directional, like pulsatile CSF movement.2,9,38 Therefore, our measurement only reflects the flow fluctuation, instead of CSF flux or bulk flow. Second, as stated by the model, the
CBV and GMS (IJV signals)
It has been shown that during resting state, ongoing dynamics in the vasculature which are independent of neural activity are likely due to vessel-autonomous or respiratory-related oscillations in the arterial diameter (∼30%).32,39,40 CBVa (arterial CBV) fluctuations have been found in arteries throughout the brain.41–44 In our previous research, these oscillations were even found in the internal carotid arteries (ICA) earlier than their appearances in the brain.34,35 These upstream oscillations move along the arterial tree into the brain and cause corresponding CBF changes in the downstream capillaries, venules, and veins, following the path of blood flow. Animal studies have confirmed that these vasomotor oscillations in the arteriole diameter drive oscillations in the rate of brain tissue oxygenation,45,46 which then leads to delayed passive dilations in the draining venules.47,48 These CBF related oxygenation changes in capillaries, venules, and veins, induce the BOLD contrast that we observed as the GMS. The same CBF changes also lead to passive dilations in the draining venules and veins 49 with smaller scale in vessel diameters.50,51 Therefore, the GMS reflects the low frequency CBV changes in both arterioles and venules, primarily by modulation of CBF that results in the well-known dilution model of BOLD. 20
The same CBF changes will later be observed via BOLD signal in large draining veins, such as the superior sagittal sinus (SSS) and IJV, as the blood moving out of the brain. For example, a previous study with 90 resting-state scans showed the fMRI signal in IJV lagged that of SSS by 1 s, while the fMRI signal in SSS lagged the GMS by 3 s. 34 This is consistent with a blood flow phenomenon mediated upstream by changes in CBV. For further validation, we rescanned two participants using a similar protocol but with slices covering from the brain to the neck (TR increased to 1 s). We confirmed that the LFO in the IJV is correlated with the GMS (participant 1: 0.46; participant 2: 0.69) with few second delay (participant 1: 3.39 s; participant 2: 4.52 s). The results and scan protocols can be found in the supplemental material (Supplemental Figure S3). Based on these findings, we also confirmed that the IJV signal can be used as a delayed surrogate signal of the GMS in the neck scan, as illustrated in Figure 2(j), in which the actual GMS could not be obtained.
Time delays
Based on the results of the brain scan (Figure 4(a)),
Similar results in delay were also shown in Figure 4(b). However, the CSF outflow fluctuation leads
The time delays differ from the results obtained from the cardiac pulsation (Figure 6(b)). As cardiac pulsation travels through the brain (i.e., fast pressure wave), it affects all the brain regions almost simultaneously. This synchronized oscillation will place pressure on the ventricular wall and leads to instantaneous CSF flow fluctuation at the fourth ventricle, as first predicted by the Monro-Kellie doctrine. 21 Adams et al. has measured the intracranial volume dynamics using Displacement Encoding with Stimulated Echoes (DENSE). 52 They demonstrated the dynamic balance between CSF flow and brain tissue volume over one cardiac cycle. Interestingly, they also found a small delay between peak gray and white matter volumetric strain (within one cardia cycle), which, they attributed to the perfusion from gray matter to white matter following the cerebrovasculature. From our study, we believe CBV also oscillates at a much lower frequency (0.01–0.1 Hz), which leads to the low frequency CSF flow fluctuations. Importantly, the low frequency CBV changes do not move as fast as cardiac pulsation. The pressure induced by CBV propagates through the vasculature as blood flow, which takes seconds. The spatial-temporal pattern of the low frequency wave moving through the brain has been validated in several groups of human subjects.30,53 Similarly, various delays (0.5-6 s) were also found between the CBV signal primarily from arterioles and the BOLD signal from individual venules49,54 in animal studies.
Figure 5 shows regional heterogeneity in the CBV changes. Most voxels show negative delays (Figure 5(d) and (e)), which is consistent with the results obtained between
Various physiological mechanisms for CSF flow or flow fluctuation
Previous studies have proposed several driving forces for CSF flow, which included 1) cardiac pulsation, 56 2) vasomotion, 10 and 3) respiration.13,38 Some studies demonstrate that pulsation is a driving force for CSF.2,56,57 Others studies used mathematical model to argue that arteriolar pulsation alone are too weak to drive CSF circulation or flux.58,59 However, the pulsation might still contribute to the fast exchange of CSF-interstitial fluid (ISF) through mixing and diffusion. 52 This could be the crucial first step in removal of the waste products in the brain.
Figure 6 shows that, compared to cardiac pulsation, LFOs are dominant in the CSF signal. However, in this study, we can only measure CSF flow fluctuations in one direction at a time in each scan and cannot determine the CSF flux directly. We estimated the CSF flux combining two consecutive scans (brain and neck scans) and found no clear flux during the short scan time (see Supplemental Figure S1). Many animal studies have tried to understand the CSF flux using contrast agents. However, the pathways for the lymphatic drainage of CSF are still not clear. 60
We showed that low frequency CSF movement was detected in both directions. Like the effect of cardiac pulsation,9,52 the low frequency CSF flow fluctuations might also contribute to the fast exchange of CSF-ISF through mixing and diffusion. It would be of great interests to understand the distinct roles played by bi-directional CSF flow fluctuation and CSF flux in the clearance of the wastes in the brain.1,8,61 Nevertheless, increased low frequency vasomotion (0.1 Hz) in arterioles has been found to increase clearance rate from perivascular space. 10
In addition to pulsation and LFOs, several lines of work have shown that respiration is instrumental in driving CSF through the aqueduct by using real-time phase contrast MRI measurements.11–13,38,62 Furthermore, Dreha-Kulaczewski et al. proposed that increased CSF flow from caudal to cranial during inspiration was considered compensation of venous blood leaving from head due to lower intrathoracic pressure. 13 Based on this theory (i.e., pressure), the CSF response to respiration should be almost instantaneous. However, no respiration belt was used in this study, which limited us from assessing the respiratory effect on the CSF flux or flow fluctuation (see limitations of the study).
Our study focused on the LFO of the signal, not the respiration frequency. The natural breathing frequencies of our participant were from 0.2–0.4 Hz (The power spectra of the CSF inflow/outflow and the GMS signals of all the participants can be found in the Supplemental Table S1, S2). Like cardiac pulsation, the power of the respiration signal is much less than that of the LFOs, indicating that the arterial LFO is the dominant force in CSF flow fluctuation, at least in resting state without using any forced/paced breathing protocol. In addition, forced or paced breathing as used in Dreha-Kulaczewski’s study might invoke different physiological responses/mechanisms, other than the ones under regular breathing. 13
Sleep vs. awake states
Previous research found increases in the cortical interstitial space by more than 60% during sleep when compared with an awake condition, resulting in efficient convective clearance of
Limitation and future studies
There are several limitations to the study which we hope to address in future experiments. First, physiological parameters (e.g., respiratory, cardiac and PETCO2 measurements) have not been recorded, which limited us from assessing their relationships with LFOs in CSF and BOLD fluctuations. Second, we did not fully sample the cardiac signal in the fMRI data due to the low temporal resolution, thus we cannot systemically assess the relationship between the GMS and the CSF signals associated with cardiac pulsation. Third, we did not collect simultaneous EEG data. Thus, we could not confirm if the neuro-vascular coupling (demonstrated in the Fultz et al 19 ) works in the wakeful state.
Conclusions
A model, based on the Monro-Kellie doctrine,
21
was proposed in this study to interpret fMRI signal and link fMRI signal with certain brain dynamics. Two fMRI scans were conducted to validate the model and several predictions. We found 1) coupling existed between LFOs of
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221074639 - Supplemental material for Coupling between cerebrovascular oscillations and CSF flow fluctuations during wakefulness: An fMRI study
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221074639 for Coupling between cerebrovascular oscillations and CSF flow fluctuations during wakefulness: An fMRI study by Ho-Ching (Shawn) Yang, Ben Inglis, Thomas M Talavage, Vidhya Vijayakrishnan Nair, Jinxia (Fiona) Yao, Bradley Fitzgerald, Amy J Schwichtenberg and Yunjie Tong in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health, Grants S10 grant (S10 OD012336 - 3T MRI Scanner dedicated to Life Sciences Research PI: Ulrike Dydak). Indiana CTSI Core Facility Grant.
Acknowledgements
We would like to thank Drs. Blaise B. Frederick, Ahmed A. Khalil, Lia M. Hocke, Ioannis Pappas, and Xiaopeng Zhou for useful discussion. We would also like to thank Ms. Antonia Susnjar for data collection. Lastly, we would like to thank two anonymous reviewers. The manuscript has been improved significantly with their comments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Yunjie Tong, Ho-Ching (Shawn) Yang, and Ben Inglis conceived of the presented idea and developed the theory. Ho-Ching (Shawn), Vidhya Vijayakrishnan Nair and Jinxia (Fiona) Yao performed the computations and verified the analytical methods. Ho-Ching (Shawn) Yang, Ben Inglis, Thomas M. Talavage, Vidhya Vijayakrishnan Nair, Jinxia (Fiona) Yao, Bradley Fitzgerald, Amy J. Schwichtenberg, and Yunjie Tong provided critical feedback and helped shape the research, analysis and manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
