Abstract
Cerebral autoregulation is a complex mechanism that serves to keep cerebral blood flow relatively constant within a wide range of cerebral perfusion pressures. The mean flow index (Mx) is one of several methods to assess dynamic cerebral autoregulation, but its reliability and validity have never been assessed systematically. The purpose of the present systematic review was to evaluate the methodology, reliability and validity of Mx.
Based on 128 studies, we found inconsistency in the pre-processing of the recordings and the methods for calculation of Mx. The reliability in terms of repeatability and reproducibility ranged from poor to excellent, with optimal repeatability when comparing overlapping recordings. The discriminatory ability varied depending on the patient populations; in general, those with acute brain injury exhibited a higher Mx than healthy volunteers. The prognostic ability in terms of functional outcome and mortality ranged from chance result to moderate accuracy.
Since the methodology was inconsistent between studies, resulting in varying reliability and validity estimates, the results were difficult to compare. The optimal method for deriving Mx is currently unknown.
Introduction
Cerebral autoregulation serves to keep cerebral blood flow (CBF) relatively constant within a wide range of cerebral perfusion pressures (CPP) through adaptive changes in cerebrovascular resistance. 1 Conceptually, cerebral autoregulation may be viewed as both a static and dynamic phenomenon, of which the former refers to the cerebrovascular adaptations to a steady-state change in CPP, while the latter refers to the acute cerebrovascular changes during a sudden, either spontaneous or induced, change in CPP.1,2
In 1996, Czosnyka and co-workers introduced the mean flow index (Mx) as a measure of dynamic cerebral autoregulation in the time domain. Mx can be measured at the bedside as the correlation between spontaneous fluctuations in CPP and cerebral blood flow velocity in the middle cerebral artery (MCAv) measured by transcranial Doppler. Thus, an increase in Mx indicates worsening of cerebral autoregulation, whereas a decrease indicates improvement. 2 In the initial studies, each raw recording of simultaneous values of CPP and MCAv was averaged and then split into blocks which were gathered into epochs, for which Pearson’s correlation coefficient was calculated, ranging from −1 to 1. Mx was subsequently obtained by averaging the correlation coefficients from every epoch throughout the recorded period of interest (Figure 1). CPP was later replaced with arterial blood pressure (ABP), creating an alternative to the original Mx, coined Mxa. 3
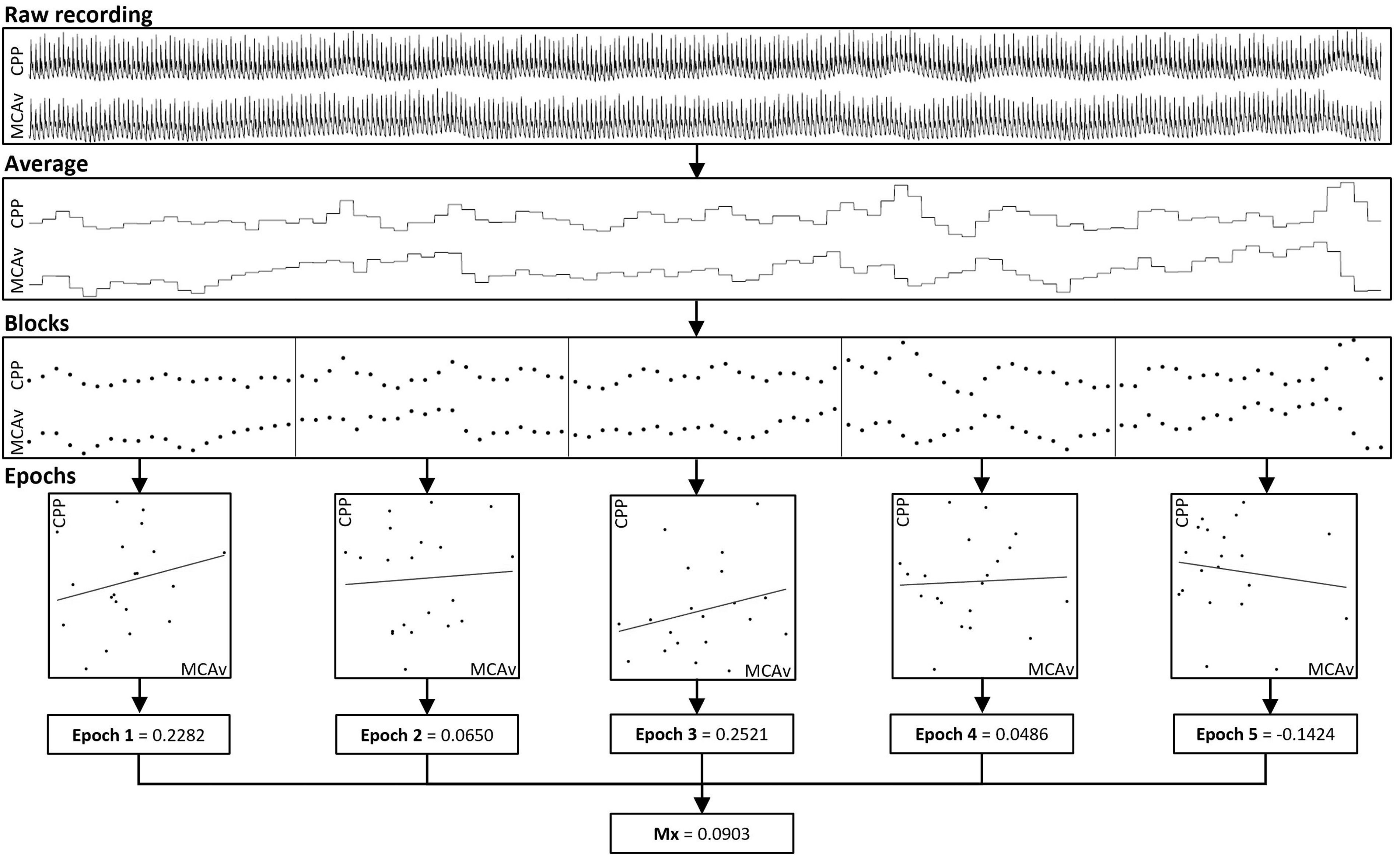
Overview of the general methodology of generating the mean flow index (Mx). The raw recording (1st row) is averaged into blocks (2nd row). The values from the blocks are then split into epochs (3rd row), and a Pearson’s correlation coefficient is calculated for every epoch (4th row), and the average of the correlation coefficient from the epochs (5th row) generates one mean flow index (6th row).
Since their inception, both Mx and Mxa have been used to assess dynamic cerebral autoregulation in several studies with a variety of patients categories, and for some of these, a poor outcome has been linked to more profoundly disturbed autoregulation. 4 However, the specific methodology varies between studies, and the reliability and validity of Mx and Mxa have never been assessed systematically. Conceivably, even small changes in the measurement approach and underlying calculations may lead to markedly different results, both for the resulting strength of autoregulation and for its association with clinical outcomes. 5
In the present study, we provide a systematic review of the existing literature on the assessment of dynamic cerebral autoregulation by Mx and Mxa, focusing on the methodological approach and measurement reliability, as well as their validity for predicting clinical outcomes.
Methods
This systematic review was carried out in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (PRISMA checklist available in Supplemental Material), 6 using the methodology outlined in the Cochrane Handbook for Systematic Reviews, 7 and was registered on PROSPERO (protocol id: CRD42020164028, Januray 8, 2020) prior to initiation. The protocol was updated on November 20, 2020 in order to specify how reliability and validity were defined and how these data were extracted. Manuscripts that reported calculations of Mx, Mxa, or both based on original data from human subjects or simulation studies were included (Supplemental Material).
Search strategy
MEDLINE, PUBMED, and EMBASE from January 1, 1996 up until February 14, 2021, were searched using commonly used synonyms of Mx. The EMBASE search string read as follows: “(Mxa OR Mx OR (Flow index)) AND (cerebral OR brain OR MCA OR (Middle cerebral artery)) AND (autoregulation OR (blood flow))”. The databases were initially searched January 31, 2020, and with a second search on February 14, 2021 to update the results. Furthermore, the reference lists of included papers and review articles were browsed for additional relevant publications.
Study selection
Two authors (MHO and CGR) screened titles and abstracts retrieved by the search strategy using the web and mobile screening app Rayyan, 8 and reports that were deemed irrelevant were excluded. Subsequently, full-text versions of potentially relevant reports were reviewed and included if they calculated Mx, Mxa, or both from original data from human subjects. At all stages of screening, any disagreement about inclusion was discussed, and if consensus was not reached, a third author (RMGB) decided whether to include the specific report. The abstracts where full-text were not available were only excluded after the primary investigator was contacted three times and did not provide a full-text manuscript published or non-published.
Data extraction
Data on methodology were extracted independently by two authors (MHO and CGR) and included patient or volunteer population, interventions if any, the method for recording arterial blood pressure, level of the transducer, insonation side (right, left or bilateral; or ipsi- or contralateral to the injured hemisphere), method of carbon dioxide measurement, terminology of the index from the article, the time resolution of recordings, recording length, number of recordings, pre-processing, whether calculations were overlapping, and the approach to creating blocks and epochs. Furthermore, measurement results from the manuscript were extracted in studies in relations to reliability, and validity.
Terminology
Mean flow index (Mx)
In the following, Mx refers to the mean flow index as a unifying concept, Mxc refers to the index calculated as the correlation between CPP (usually calculated from invasive measurement of both ABP and ICP) and mean flow velocity, Mxa to the correlation between invasively measured ABP and mean flow velocity, and nMxa to the correlation between non-invasively measured ABP and mean flow velocity.
Reliability
Reliability was divided into repeatability (ability to replicate the same results in identical settings) and reproducibility (ability to replicate the same results in the same subject under changing conditions) 9 and reported using the intraclass correlation coefficient (ICC) and a Bland-Altman plot, with limits of agreement (LOA). Repeatability also was assessed by including studies addressing the internal consistency, referring to the stability on a group level, during identical settings. 10 The ICC was interpreted using the 95% confidence interval (CI), if available, and interpreted as defined by Koo et al. to indicate poor (<0.5), moderate (0.5–0.75), good (0.75–0.9), and excellent reliability (>0.9). 11 When possible, we have reported the specific type of ICC since ICC-agreement refers to the agreement between the two ‘raters’, and ICC-consistency refers to how consistent the difference between ‘raters’ is. 12 Studies that reported Spearman’s or Pearson’s correlation coefficient only were not included in the results section, as the assumptions, requirements and pitfalls of these analyses were seldom reported.13,14
Validity
The validity of Mx was assessed by the discriminatory and prognostic ability. The discriminatory ability was defined as the ability of Mx to distinguish different patient categories from healthy volunteers, while the prognostic ability was defined as the ability to predict a defined clinical outcome or event. Validity reported using receiver operating characteristics curve with the use of the confidence limits for the area under the curve (AUC) was as toss-up (chance result, ∼0.5), low accuracy (0.5–0.7), moderate (0.7–0.9), and high accuracy (>0.9). 15 If AUC was not available, the group-specific results were presented.
Statistics
All statistical analyses were carried out using R 4.0.2 (R Core Team (2020), Vienna, Austria). Normally distributed data are presented as mean and standard deviation (SD), while non-normally distributed data are presented with median and interquartile range (IQR). If relevant raw or aggregated data were available, these were used in calculations. P-values <0.05 were considered significant.
Results
The search strategy resulted in identification of 6,580 publications, of which 312 were duplicates. After abstract and full-text screening, 128 studies reporting the Mx in humans were included (Figure 2). The authors of abstract where full-text was not available did not provide any additional published or non-published full-text manuscripts or data; hence only published peer-reviewed articles were included. Of these, 83 (65%) included patients with acute brain injury (including TBI: 45; aneurysmal subarachnoid haemorrhage (aSAH): 5; intracerebral haemorrhage (ICH): 2; ischaemic stroke or stenosis: 18; and mixed populations of acute brain injury: 11), while 32 (32%) included healthy volunteers either alone or as controls. Mxc was reported in 48 studies, Mxa in 46 studies, and nMxa in 41 studies. Forty-seven studies dichotomised Mx, with 0.3 being the predominant threshold between impaired and intact cerebral autoregulation (n = 23; Supplemental Material), while the remaining studies included Mx as a continuous measure.
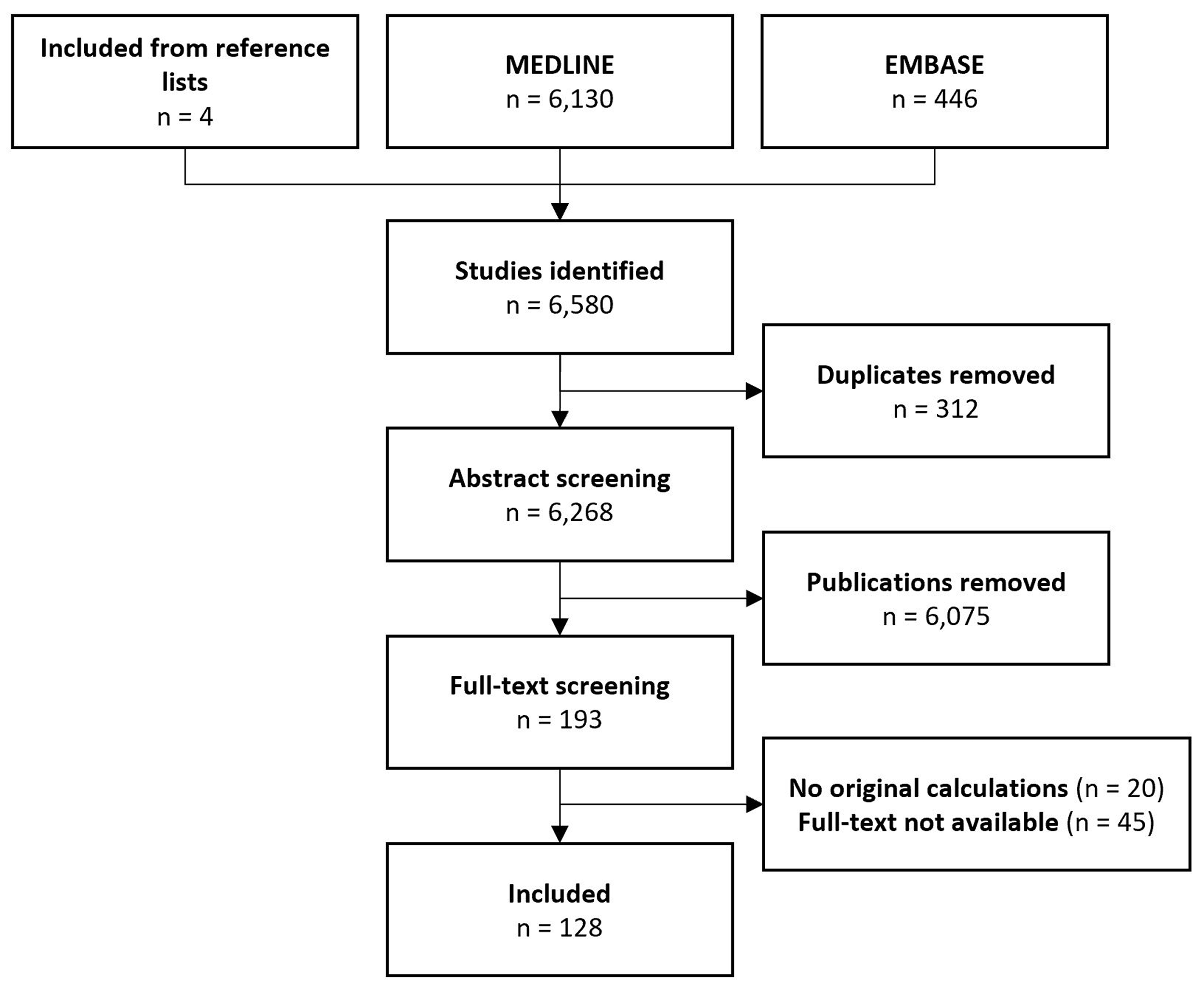
CONSORT Flow diagram. Forty-five abstracts did not have an accompanying full-text manuscript; none of the corresponding authors of these abstracts provided such full-texts after up to three attempts at contact.
Methodology
Comparisons between different measures of Mx
Five studies provided head-to-head comparisons between different measures of Mx, all in patients with traumatic brain injury (TBI). Four studies compared overall differences, where two studies reported that Mxa was higher than Mxc (mean difference, 0.15 (n = 145) 16 and 0.22 (n = 288) 17 ) one reported that Mxa was higher than nMxa (mean difference 0.08 (95%CI: 0.11–0.04)) 18 ; and one reported that nMxa was slightly higher than Mxc (mean difference, 0.01, limit of agreement, ±0.36). 19 Mxc increased significantly during so-called plateau waves of increased ICP in TBI patients, but the increase was not significant for Mxa (Mxc baseline: 0.12 ± 0.40, ICP increase: 0.47 ± 0.47; Mxa baseline: 0.21 ± 0.34, ICP increase: 0.28 ± 0.42). 20
Pre-processing
48 of 128 (38%) of the articles described an approach for pre-processing; thus, 11 studies applied low-pass filters, high-pass filters, or both to data, 8 studies used spectral filtering, two studies used a Fourier transform algorithm, and 27 studies used either manual or automatic artefact removal. Several different methods were used to remove artefacts, and none of the studies addressed the influence artefact removal have on data quality. One study, however, showed an increase in nMxa with increasing noise, 21 and one study defined an upper limit of 10% for the numbers of artefacts acceptable before exclusion 22 (Supplemental Material).
Calculation
All studies but two22,23 created blocks without overlaps. The duration of blocks varied between 3 and 10 seconds, with 10 seconds being the most predominant (Figure 3(a)). Epoch sizes ranged from 10 to 60 blocks, with 30 blocks for a total of 300 seconds being the predominant choice (n = 50) (Figure 3(b)). The epochs overlapped in 21 studies, typically by 1 to 6 blocks (for a total of 3 to 60 seconds) between each new calculation (Figure 3(c)).
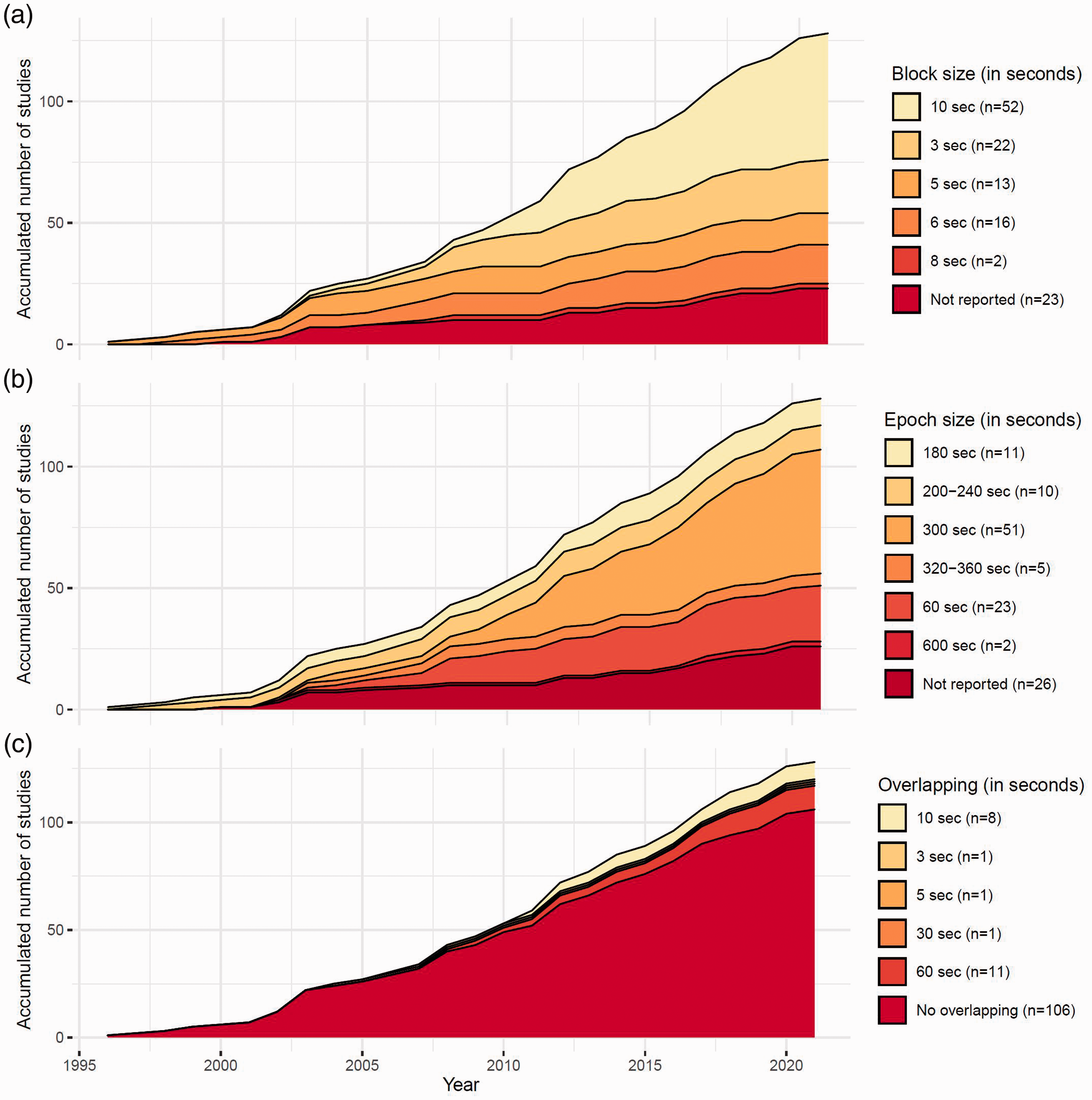
Accumulated methodology from the included studies. (a) Block size; (b) Epoch size; (c) use of overlap and interval between epochs.
Reliability
The reliability of Mx was assessed in eight studies, four of which reported data on repeatability, three on reproducibility, and one on both.
Repeatability
In one study, 10-minute recordings were obtained in 37 patients with ischaemic stroke and 51 healthy volunteers. The aim of this study was to assess the agreement between the first part of a 10-minute recording and the full recording for deriving nMxa; a difference of 0.02 ± 0.19 and an ICC-agreement of 0.93 (95%CI: 0.90–0.96) was found. 24 However, another study including 167 recordings from patients with transient ischaemic attack used a similar methodology with the comparison of the first and last half of a 10-minute recording with the full recording showed a significant difference between the full recording and the last half for both sides. 25 Comparing two consecutive 5-minute recordings, in 46 healthy volunteers, resulted in an ICC of 0.39 (95%CI: 0.08-0.63) for the native recordings. 26 In another study, repeated recordings were obtained in twenty healthy volunteers during 60 s of sitting and 60 s of free-standing. The ICC-agreement between two consecutive sessions where the participants were sitting was reported to be <0, while an ICC-agreement of ∼0.75 was reported for the standing position. 27 Recordings were obtained for 16 minutes without any interventions in 10 healthy volunteers through simulations, and nMxa was found to stabilise after 6 minutes when it was calculated by including one more minute for every calculation from 1 to 16 minutes. 28
Reproducibility
The four studies assessing reproducibility all included healthy volunteers. In a study on 19 participants, Ortega-Gutierrez et al. obtained recordings 17 (IQR 5–27) days apart and calculated ICC for nMxa based on 10-min recordings. ICC was 0.46 (95%CI: 0.02–0.75) when based on insonation of the left and 0.42 (95%CI: −0.34–0.73) when based on the right MCA. 29 Similarly, Riberholt et al. measured Mx 23 ± 3 days apart in 14 persons, both in supine position and during head-up tilt for 5 minutes each. These authors compared ICC-agreement for different block and epoch sizes and found 3-second blocks, with 20 blocks in an epoch to provide the best reproducibility in the supine position (3-second blocks: 0.55 (95%CI: 0.04–0.82); 5-second blocks: 0.22 (95%CI: −0.32–0.66); 10 second blocks: 0.21 (95%CI: −0.33–0.65)), as well as during head-up tilt (3-second blocks: 0.46 (95%CI: −0.05–0.79); 5-second blocks: 0.57 (95%CI: 0.10–0.84); 10 second blocks: 0.21 (95%CI: −0.38–0.61)). 5
Finally, Lorenz et al. assessed the reproducibility of nMxa by comparing a model of poor insonation quality, in which aluminium foil was placed between the probe and the skin to reduce the signal power, 26 with the native recording in two studies of healthy volunteers, reporting ICCs of 0.62 (n = 45; 95%CI: 0.39–0.78) 30 and 0.45 (n = 41; 95%CI: 0.16–0.67) respectively. 26
Validity
Thirty studies evaluated the validity of Mx in terms of discriminatory and/or prognostic ability.
Discriminatory ability
Fifteen studies were identified. In patients with SAH (n = 15), Mxc was higher in patients with vasospasm compared to their baseline measurement (baseline: 0.21 ± 0.24; vasospasm: 0.46 ± 0.32),31,32 and nMxa was higher than nMxa in healthy controls (patients, n = 30, 0.43 ± 0.2; controls, n = 9, 0.02 ± 0.1). 33 Higher values of Mx in different patient populations compared to healthy control groups were also obtained in patients with alcohol withdrawal syndrome (patients, n = 20, 0.16, SE: 0.05; controls, n = 20, 0.00 SE 0.04), 34 obstructive sleep apnoea (patients, n = 11, 0.41 ± 0.13; controls, n = 9, 0.23 ± 0.10), 35 schizophrenia (patients, n = 21, 0.40; controls, n = 23, 0.26), 36 and intracerebral haemorrhage (patients, n = 12, 0.41 ± 0.27; controls, n = 7, 0.17 ± 0.13), 37 whereas patients admitted to a neurorehabilitation unit showed lower values of nMxa compared to healthy controls (patients, n = 14, 0.04, SE: 0.07; healthy, n = 15, 0.35, SE: 0.07; p < 0.01). 38 In contrast, no differences were found in patients with migraine compared with healthy volunteers39,40 (19 patients vs. 75 healthy volunteers: mean nMxa 0.29 ± 0.17 vs. 0.27 ± 0.17 39 ; 22 patients vs. 22 healthy volunteers: mean nMxa 0.24 vs. 0.26) 40 ) patients with a brain tumour vs. healthy volunteers (12 patients vs. 12 healthy volunteers: mean nMxa 0.45 ± 0.10 vs. 0.36 ± 0.18 41 ) and critically ill patients with compared to without sepsis (52 septic vs. 40 non-septic: Mean nMxa 0.33 (IQR: 0.08–0.58) vs. 0.31 (IQR: 0.04–0.59) 23 )
Tang et al. reported a reduction in Mxa after compared with before stenting of the internal carotid artery (25 patients; before stent, 0.42 ± 0.16; after stent, 0.21 ± 0.09), 42 whereas successful recanalization after thrombectomy was not associated with any change (10 patients; successful recanalization: 0.50 ± 0.24; no recanalization: 0.45 ± 0.24). 43 Two studies examined nMxa as a diagnostic marker of stroke, using healthy volunteers as controls (stroke = 32 vs. healthy volunteers = 59 44 ; stroke = 37 vs. healthy volunteers = 51 24 ) reporting an AUC of 0.709 (0.604–0.799) 44 and 0.719 (0.613–0.810), respectively. 24 However, none of these studies provided further data on the performance of Mx to diagnose stroke, such as cut-off values or predictive values.
Prognostic ability
Fifteen studies were identified. Five studies of patients with severe TBI were conducted by the research group that originally introduced Mx, 2 and potentially with some overlap between patients.45–49 The authors reported lower values of Mxc in those with a favourable (Glasgow outcome scales (GOS) of 4–5) than in those with a poor outcome (GOS 1-3)45–49; in one study (n = 151) Mxa was also measured and did not differ between outcome groups (favourable 0.16 ± 0.24; unfavourable 0.23 ± 0.21; p = 0.08). 48 Four studies also from this research group calculated AUC to assess the prognostic value of Mxc for functional outcome using the same dichotomisation of GOS.50–53 In these studies an AUC between 0.593 (n = 37; p = NS 52 ) and 0.658 (n = 300; 95%CI: 0.595–0.722 50 ) was reported for Mxc, while an AUC between 0.620 (n = 300; 95%CI: 0.555-0.685 50 ) and 0.704 (n = 37; p = NS 52 ) was reported for Mxa. The AUC for the relationship between Mxc and fatal outcome, defined as 6 month all-cause mortality, also varied between 0.608 (n = 37; p = NS 52 ) and 0.628 (n = 300; 95%CI: 0.550-0.70 50 ) while that of Mxa showed an AUC from 0.565 (n = 300; 95%CI: 0.584-0.714 50 ) to 0.616 (n = 37; p = NS 52 )
In contrast to TBI, Mxc appeared not to differ between survivors and non-survivors after aSAH (alive: n = 30, 0.04 ± 0.11; dead: n = 7, 0.06 ± 0.10). 54 Two studies of overlapping cohorts with acute brain injury (approximately half with TBI and 15% with aSAH) reported a lower Mxa in those who survived (n = 6; 0.03 ± 0.21) than in those who did not survive until discharge from hospital (n = 0.28 ± 0.40), 55 with an AUC of 0.80 (n = 41). 56 Finally, a higher nMxa increased the odds of postoperative cognitive dysfunction in 82 elderly patients (>65 years of age) undergoing major non-cardiac surgery (POCD, OR: 1.44, 95%CI: 1.05–1.95), 57 and predicted sepsis-associated encephalopathy in a population of septic patients (n = 100, AUC: 0.65, 95%CI: 0.53–0.76). 22
Discussion
Based on 128 studies in healthy humans and a large variety of patient populations, the findings of this systematic review indicate that the methodology for assessing cerebral autoregulation by Mx varies markedly regarding signal processing, and the calculation of Mx. Repeatability and reproducibility varied from poor to excellent in the relatively few studies that have addressed this. Indeed, many studies provided insufficient information regarding signal processing, including artefact handling and calculations, and no attempt to generalise reporting of the findings have hitherto been made.
In terms of signal processing, the process of transforming the raw recording into Mx varies greatly in terms of removal of artefacts, pre-processing, block size, epoch size, and epoch overlaps. Only one study mentioned the maximum amount of artefacts which could be accepted, 22 even though both TCD and ABP (invasive and non-invasive) recordings are prone to artefacts. The necessary pre-processing mentioned is either manual or automated, which both could introduce bias if not standardised between studies. The actual effect of including or omitting artefacts was quantified in one study using an artificial source of noise, which showed decrease Mx with increasing amounts of noise. 21 Furthermore, the differences in block and epoch size have not been investigated in full, although one study does show a substantial variation in reproducibility when comparing different methods. 5
A consensus regarding recording length is clearly required before repeatability and reproducibility of Mx in different patient populations can be formally compared, and before its actual discriminatory and predictive ability in individual patients can be determined. This is further complicated by the lack of consensus for a threshold that identifies impaired dynamic cerebral autoregulation. 58 More than one-third of the studies mention a threshold between intact and impaired cerebral autoregulation, with the primary threshold being 0.30. However, this is primarily based on the Mxc from group-specific observations in patients with TBI. While cerebral autoregulation is widely accepted to be impaired in most of these patients, it should also predominantly be intact in healthy volunteers. One study with 56 healthy volunteers reported an average nMxa of 0.44, and the average nMxa in healthy volunteers has been reported to range from 0.00 34 to 0.44 29 in comparable cohorts.; thus, with a threshold of 0.30, it may be inferred that many healthy volunteers would also be classified as having impaired cerebral autoregulation. However, besides using non-invasive rather than invasive blood pressure recording, signal processing, notably with regard to block and epoch numbers and lengths, as well as the length of recordings, practically renders these findings incomparable. It is still possible that a threshold of impaired cerebral autoregulation can be defined, but the intrapopulation (SD of ∼0.2) and interpopulation variation in healthy volunteers is worrisome, not only in terms of reliability and validity but also in the interpretation of Mx as an index of dynamic cerebral autoregulation. The ICC in itself is affected by variation, where the same absolute difference between comparators influences the results differently, depending on the variation in the data. Thus, a difference of 0.1 might result in a ‘large’ drop in ICC in a population with small variation, while the same difference in a population with large variation might result in a negligible change in ICC. The questionable reliability could primarily be an effect of a smaller variation in the investigated populations of healthy volunteers. The variation limits the usefulness of Mx clinically, or even as an outcome in studies. If the variation of Mx reflects the actual variation of cerebral autoregulation, even standardisation might not increase its usefulness. This variation seen for Mx and other indices of cerebral autoregulation, and the fact that reference values of intact autoregulation for these indices have not yet been identified, might be due to the fact that these are merely simplified quantifications of a complex physiological mechanism. 59
Despite the inconsistent methodology, Mx was generally reported to be higher in patients than in healthy controls, thus suggesting worse dynamic cerebral autoregulation in the former. The usefulness of Mx, and other indices of autoregulation, is highly dependent on the discriminatory and prognostic ability. Only two studies, however, utilised prediction modelling, where nMxa was found to have a low to moderate accuracy at diagnosing stroke. Since the actual discriminatory accuracy was not assessed, this limits the interpretation and clinical application of Mx even as a group-based index. The interpretation of individual measures of impaired autoregulation in comparison with healthy volunteers has also not been assessed. Furthermore, the utilisation of individual thresholds for identifying impaired cerebral autoregulation requires excellent reliability, since individual variation when assessing repeatability or reproducibility might change the interpretation. The repeatability varied from poor to excellent, with one study reporting excellent 24 and the two other reporting poor to good repeatability.26,27 The reproducibility, assessed in healthy volunteers also shows variation with poor to good reliability,29,30 depending on methodology. 5 The questionable repeatability and reproducibility of Mx, might be explained partly by the length of the recordings, where one study sought to identify the potential cut-off where Mx would stabilize. 28 The actual block and epoch size are not described in the study, but the authors conclude that recordings shorter than six minutes should not be utilized. Nonetheless, all but one 29 of the studies addressing reliability used recordings shorter than these six minutes as at least one of the comparators,5,24–27,30 and one study even used 60 s recordings. 27 This may have contributed to the poor repeatability.
The above-mentioned inconsistencies and shortcomings of Mx methodology are also likely to explain the differing conclusions of the prognostic ability of both Mxc and Mxa, which has been most extensively investigated in patients with severe TBI.45–49,60 The accuracy of Mx in these studies ranges from chance-result to moderate in prediction of unfavourable functional outcome or mortality. Similar accuracy is reported in prediction of sepsis-associated encephalopathy (low to moderate accuracy), 22 and mortality in a mixed population of acute brain injury (moderate accuracy). 56 While this may simply be interpreted to reflect that impaired cerebral autoregulation is a poor prognostic marker, no firm conclusion can currently be drawn in this regard from Mx-based studies.
Apart from the importance of standardising recording length, signal processing, and Mx calculations in future studies, future systematic investigation of potential confounders and covariates is equally important. In our opinion, the most important covariates are the measures used to calculate Mx, i.e. ABP, ICP, and MCAv.16–20 Other previously highlighted confounders and covariates include age,29,61 the side of insonation relative to a given intracerebral focus,29,34,39,61–63 PaCO2,64–66 use of positive end-expiratory pressure (PEEP) in mechanically ventilated patients, 67 and posture.5,27,38,68,69 Especially PaCO2 or ETCO2 should be measured as part of a study as it is an important covariate, but their exact influence on Mx is still unclear.64–66 These should, however, be investigated using standardised methodology before any firm conclusions can be drawn.
The International Cerebral Autoregulation Research Network (CARNet) have previously addressed issues similar to those outlined in the present paper with the assessment of dynamic cerebral autoregulation by transfer function analysis. 70 No gold standard measure of dynamic cerebral autoregulation has been identified as all the hitherto identified measures have their own limitations. Transfer function analysis for instance also present with questionable reliability and no apparent reference values.24,26,29,70,71 CARNet subsequently published a white paper with the purpose of standardising the assessments in studies that use this method and provided recommendations for design, artefact handling, data reporting, and calculation. 59 We suggest a similar standardisation for future studies that assess dynamic cerebral autoregulation by Mx and related indices in the temporal domain (Table 1).
Recommendations of methodology and reporting.
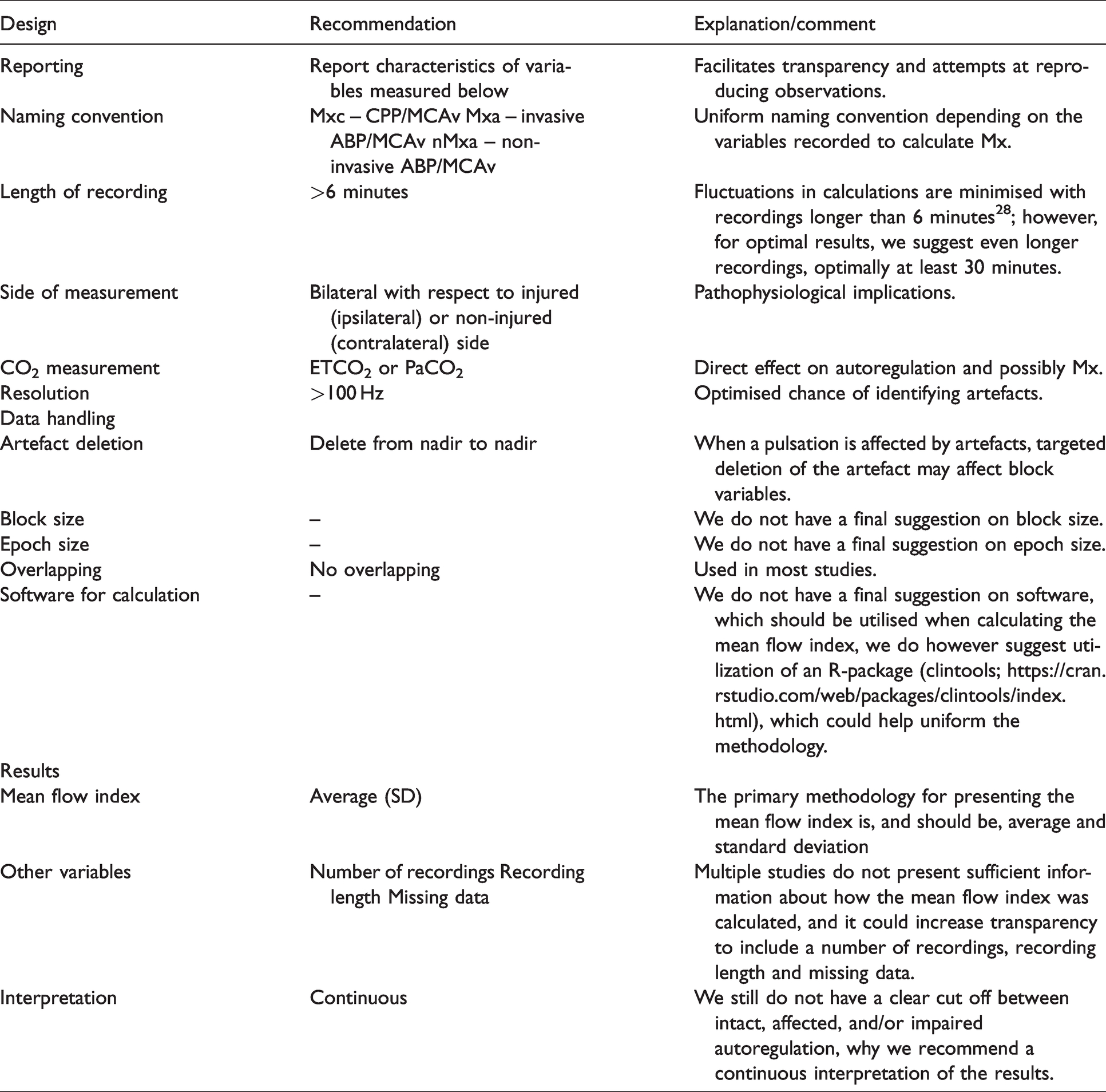
The majority of the studies investigating Mx and related indices are carried out by one research group or in collaboration with them; we are therefore aware that the current knowledge of Mx, as well as the conclusions drawn in this review, might be subject to bias.
It must, however, be noted that according to our findings, the shortcomings of Mx cannot be resolved by standardisation alone. Because the methodology varied so markedly in previous studies, the actual physiological and clinical relevance of Mx is practically unknown. As described by Colli et al., 9 phase 0 in diagnostic research is to address the validity and reliability of the test, and the currently available studies do not fully answer this question. Some knowledge of the range and variation in healthy volunteers, including potential confounders (Phase I), and the ability to distinguish patients with or without disease (Phase IIa) has been obtained, but the next feasible step should be to further investigate the reliability and validity of Mx.
Conclusion
According to this systematic review, the methodology and interpretation of Mx and related indices in previous studies is markedly inconsistent and often insufficiently reported, thus leading to highly variable reliability. Consequently, and despite being based on firm physiological principles, the optimal method for deriving Mx is currently unknown. It also remains to be established to which extent it provides meaningful clinical information in various clinical populations and healthy volunteers, both in terms of dynamic cerebral autoregulation, as well as diagnosis and prognostic stratification.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211052588 - Supplemental material for Reliability and validity of the mean flow index (Mx) for assessing cerebral autoregulation in humans: A systematic review of the methodology
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211052588 for Reliability and validity of the mean flow index (Mx) for assessing cerebral autoregulation in humans: A systematic review of the methodology by Markus Harboe Olsen, Christian Gunge Riberholt, Jesper Mehlsen, Ronan MG Berg and Kirsten Møller in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Centre for Physical Activity Research (CFAS) is supported by TrygFonden (grants ID 101390 and ID 20045).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
All authors deisgned the study; MHO and CGR carried out the selection and extraction; MHO wrote the first draft; all others revised and accepted the final version.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
