Abstract
Acute stroke is associated with high morbidity and mortality. In the last decades, new therapies have been investigated with the aim of improving clinical outcomes in the acute phase post stroke onset. However, despite such advances, a large number of patients do not demonstrate improvement, furthermore, some unfortunately deteriorate. Thus, there is a need for additional treatments targeted to the individual patient. A potential therapeutic target is interventions to optimize cerebral perfusion guided by cerebral hemodynamic parameters such as dynamic cerebral autoregulation (dCA). This narrative led to the development of the INFOMATAS (Identifying New targets FOr Management And Therapy in Acute Stroke) project, designed to foster interventions directed towards understanding and improving hemodynamic aspects of the cerebral circulation in acute cerebrovascular disease states. This comprehensive review aims to summarize relevant studies on assessing dCA in patients suffering acute ischemic stroke, intracerebral haemorrhage, and subarachnoid haemorrhage. The review will provide to the reader the most consistent findings, the inconsistent findings which still need to be explored further and discuss the main limitations of these studies. This will allow for the creation of a research agenda for the use of bedside dCA information for prognostication and targeted perfusion interventions.
Introduction
Stroke is the second major cause of death after ischemic heart disease in the general population and one of the highest economic-impact diseases due to the high prevalence of disability in survivors.1,2 In the last decade, new therapies have been investigated with the aim of improving clinical outcomes after acute ischemic (e.g., intravenous and intra-arterial thrombolysis) and haemorrhagic stroke (e.g., targeted arterial blood pressure (ABP) management).2–10 However, even with these new investigational and/or approved therapies, a selection of patients (10-40%)11–13 deteriorate after admission and subsequently have poor clinical outcome.14,15 Improving global cerebral perfusion in order to save salvageable brain tissue in the area surrounding the ischemia (referred to as the ‘penumbra’) and the area surrounding haemorrhagic lesion (area with ongoing inflammatory processes) is a promising option. In addition, impaired protective mechanisms like cerebral autoregulation have been associated with clinical deterioration.14,16,17 Dynamic cerebral autoregulation (dCA) is an active process, maintaining cerebral perfusion at an appropriate level via regulation of the cerebral vasculature. In recent years, non-invasive and invasive techniques have been improved, allowing clinicians to assess dCA function at the bedside, via spontaneous or induced changes in ABP. Furthermore, to record changes in cerebral perfusion during the acute phase of stroke, most researchers have used different estimates of cerebral blood flow (CBF) (e.g. transcranial Doppler, TCD). Phase II studies to improve dCA capacity by specific interventions in acute stroke are underway18,19 and ideas to incorporate hemispheric dCA status into treatment decisions during the acute phase of stroke have been proposed.19–22
In 2011 the Cerebral Autoregulation Network (CARNET, www.car-net.org) was initiated to foster collaboration between researchers and prioritize certain dCA research areas. The group comprises experienced clinicians and researchers in different areas such as vascular neurology, cardiology, cardiovascular physiology, biomedical engineering, and neuroscience. Since then, regular meetings have been organised and one of the key achievements is the publication of a “White Paper” designed to harmonise dCA studies and provide a homogeneous and uniform method of data collection and analysis. 23 In addition, CARNET members conducted two Bootstrap projects to investigate the variability of data collection, the reproducibility of analysis, and the physiological variability that influences dCA studies.24–27 In line with that, in 2016 a new project with the acronym INFOMATAS (Identifying New targets FOr Management And Therapy in Acute Stroke) was launched to introduce dCA measurement as a tool to guide therapeutic decisions in clinical practice.
The first step of INFOMATAS project would be to summarize the current dCA literature in acute stroke patients. A special issue in this journal is dedicated to covering different aspects of dCA research in six separate articles. The aim of this article is to perform a narrative review of observational studies that assessed dCA in acute ischemic and haemorrhagic stroke patients, with a special focus on the relationship between dCA status and clinical outcome measures. Although, in the literature, there are several publications reviewing dCA in stroke,20,28–35 the present review is distinct from the others as the focus is on the studies that evaluated patients in the acute phase (<48hs) after stroke onset. The INFOMATAS group believes that one of the core issues to be understood is whether cerebral hemodynamics in the acute phase is impaired and consequently impacts clinical outcome.
Methodology
Studies were identified using a search strategy in two English language databases up to July 2020 (PubMed and Web of Science) with pre-defined search terms (see supplemental material). The specific subsections and search terms were discussed between selected members from the CARNET consortium with clinical and research expertise in the field of acute stroke. Each co-author was responsible for one of the predefined subsections. References of the selected articles were screened for additional relevant articles. The inclusion criteria for all subsections were: (1) clinical study in adult patients (>18 years old); (2) reporting results of an established dCA autoregulation method using a continuous bedside brain monitoring technique (invasive or non-invasive) in combination with continuous (invasive or non-invasive) ABP monitoring; and (3) investigations had to be performed in the acute phase (defined as < 48 hours) of ischemic stroke (IS), intracerebral haemorrhage (ICH), and subarachnoid haemorrhage (SAH). The formulated exclusion criteria were: non-human studies and non-English language publications. The data extracted from the retrieved studies were: 1) specific research objectives; 2) type and number of stroke patients included; 3) selected method of dCA measurement with reported values and/or thresholds for impaired DCA; 4) main results and conclusions; 5) relationship between dCA and clinical outcomes; and 6) important limitations of the study. All the data were summarized in structured tables both in the main document and as an extended version in the supplemental material. In addition, we included, in the main tables, a summary of the most important findings regarding dCA divided according to their consistency: consistent (findings that were common to more than one study without any conflicting results) or inconsistent (findings from a single study or with conflicting results). At the end of the review the INFOMATAS panel grades the main findings (please refer to supplemental material for the grading system) and gives a recommendation based on it.
In order to orientate the reader as to the breadth of the data included, we will briefly discuss the main monitoring techniques and associated dCA calculation methods used in the reported studies. We refer to chapter 3 in this journal’s special issue for in-depth information. The studies retrieved will be reported in sections divided by the three medical conditions (IS, ICH, and SAH). Each section will contain separate subsections to report the studies that evaluate CA status during the acute phase of stroke and the correlation of dCA with clinical outcomes; at the end of each section, the main limitations of the studies are summarized, as are the main knowledge gaps and directions for future studies.
Monitoring techniques used in stroke studies
When considering the methodology used to investigate dCA, the most common bedside cerebral technique used is non-invasive TCD. The advantage of applying this technique in stroke patients is its portability, excellent temporal resolution, and low cost.28,35,36 While measurements can be repeated daily to track dCA function over time, the duration of individual monitoring sessions is usually limited to less than 30 minutes. Furthermore, TCD is operator-dependent with a long individual learning curve, and some patients will have an insufficient acoustic bone window.30,37,38 Non-invasive NIRS was used in just a few studies.39,40 NIRS is a promising technique because it is operator independent and allows extended periods of monitoring with little infringement on clinical care. However, the method only provides a regional measurement. There are also concerns about potential contamination by the extracranial circulation or artefacts caused by intracranial blood.41–44 Other methods used in the retrieved studies are invasive brain tissue oxygen pressure (PtO2) and intracranial pressure (ICP),45–48 Although these techniques provide robust parameters, they are invasive techniques that can be applied only in critically ill patients.47,49 PtO2 measures changes in the local oxygen tension in the tissue surrounding the probe, which can be used as a surrogate for CBF.47,48 Measuring ICP is very interesting because not only is it possible to evaluate dCA globally but also cerebral compliance and cerebral perfusion pressure (CPP) can be determined.45,46,50
dCA assessment methodologies used in stroke studies
dCA refers to the dynamic response of the cerebral vasculature to changes in ABP. It measures the dampening effect and the rate of CVR change. Moreover, it can be assessed spontaneous ABP oscillations making it the preferred approach for studying cerebral autoregulation in acute stroke.28,30,51,52
The two most common mathematical modeling techniques used in dCA studies are transfer function analysis (TFA) and associated autoregulation index (ARI), which are suitable models to be applied in acute conditions.23,36,53 TFA expresses the ABP-CBF relationship at each frequency that uses the spontaneous (or induced) oscillations in ABP as input signals. At each frequency, this relationship can be expressed by the measures of gain (amplitude of oscillations) and phase (time delay between oscillations) if enough coherence between the signals is present. The ARI index is the product of gain and phase and may represent a more sensitive method of dCA assessment. 52 Both methods are frequently used in the stroke population because the parameters can be extracted by (periods with enough) spontaneous fluctuations of ABP over a relatively short period, avoiding the risk of more intense and uncontrolled ABP fluctuations. 52 Another modeling technique used in acute stroke studies is time correlation methods48,54,55 (e.g. PtO2 oxygen reactivity index, ORx; TCD mean flow index, Mx; TCD systolic (arterial) velocity autoregulation index, S(a)x; TCD diastolic (arterial) velocity autoregulation index, TCD D(a)x; NIRS tissue oxygenation derived autoregulation index; TOx) and project pursuit regression analysis. 56 , 57 The correlation analysis calculates the relation of ABP with the TCD cerebral flow surrogate (e.g. CBFV in Mx); 58 it has an attractive advantage in that the associated software algorithm analyzes trends of dCA in real time, allowing for measurement of the influences of coupled therapeutic interventions. This is currently under investigation for severe traumatic brain injury patients with the invasive signal ICP.59,60 The problem with the TCD version is the need for longer recordings to reduce the signal-noise ratio, which is possible in critical care patients but requires automatic and continuous correction of the insonation angle. In future studies, the possibility of monitoring dCA should be explored in real time using other methodological approaches (TFA, ARI, wavelets, etc.) and robotic TCD probes.
Finally, some methodologies applied to dCA calculation used invasive monitoring; one of these methods is the pressure reactivity index (PRx), which calculates the moving correlation between slow waves of ABP and ICP. Calculation of PRx has an attractive therapeutic application, which is the calculation of the ‘optimal’ cerebral perfusion pressure (CPPopt) value at which dCA functions best for an individual patient according to cerebrovascular reactivity.61,62 At the moment, a phase II study recently reported, assessing the feasibility and safety of autoregulation guided CPP management in severe traumatic brain injury (TBI) patients with ICP monitoring. 63 The idea of individualizing treatment guided by dCA, like that applied in the CPPopt feasibility study, is one of the major objectives of the INFOMATAS project. 64
dCA in IS studies
Dynamic cerebral autoregulation status
The search retrieved 11 suitable articles comprising the analysis of 631 patients 17 , 54 , 55 ,65–73 (Figure 1(a)) (Table 1 and Supplemental Table 1). The only method used to monitor CBF was TCD, and the modelling used to calculate dCA were TFA, Mx, and Dx.17,54,55,65–72
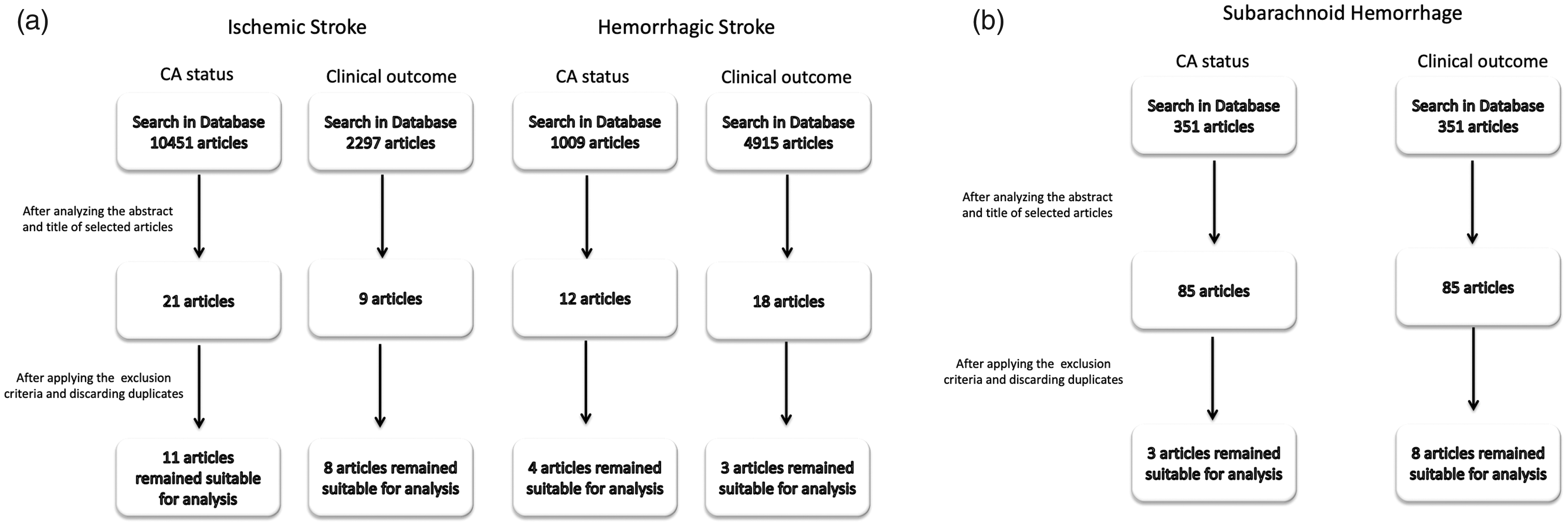
(a & b) a: Flow diagram of studies identification for ischemic and intracerebral haemorrhage stroke. b: flow diagram of studies identification for subarachnoid haemorrhage stroke.
Studies that evaluated dynamic cerebral autoregulation status in the acute phase of ischemic stroke.
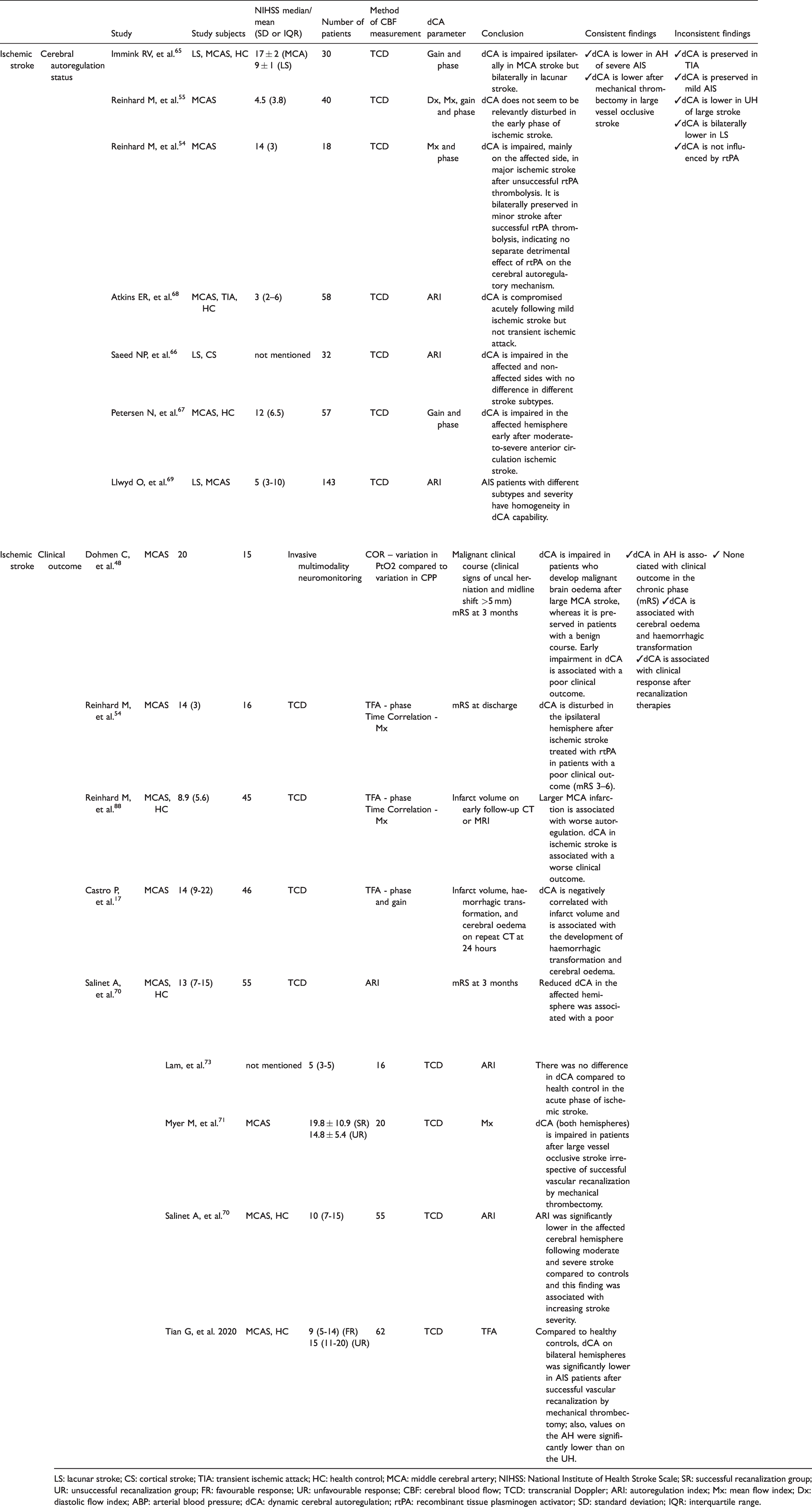
LS: lacunar stroke; CS: cortical stroke; TIA: transient ischemic attack; HC: health control; MCA: middle cerebral artery; NIHSS: National Institute of Health Stroke Scale; SR: successful recanalization group; UR: unsuccessful recanalization group; FR: favourable response; UR: unfavourable response; CBF: cerebral blood flow; TCD: transcranial Doppler; ARI: autoregulation index; Mx: mean flow index; Dx: diastolic flow index; ABP: arterial blood pressure; dCA: dynamic cerebral autoregulation; rtPA: recombinant tissue plasminogen activator; SD: standard deviation; IQR: interquartile range.
Consistent findings
There is only one consistent finding concerning dCA status in the affected hemisphere (AH) of severe IS.
Six studies disclosed that dCA is lower in the affected hemisphere (AH) of severe IS, as measured by the National Institute of Health Stroke Scale (NIHSS).54,65,67,70–72 Two of them, investigated patients with large vessel occlusion who underwent recanalization therapies by mechanical thrombectomy.71,72 Interestingly, both studies disclosed disturbed dCA in the AH of patients with successful recanalization. However, it should be noted that one of the studies defined dCA status by a cut-off value of the Mx index. The Mx index has been extensively validated for traumatic brain-injured patients but not for IS patients. 74
The above findings suggest that close monitoring of dCA status may be particularly important during the early stages of clinically severe stroke and/or the in the reperfusion phase in large-vessel occlusion strokes. ABP fluctuations occur mostly in this acute stage, 75 and in combination with dCA changes, this might have provided therapeutic interventions for salvaging the ischemic penumbra. What is still unclear is whether dCA impairment is the cause or consequence of stroke severity in the acute phase. Further studies should address this issue for clinical purposes.65,70
Inconsistent findings
There are four inconsistent findings concerning dCA in unaffected hemisphere (UH) of severe IS, dCA in mild-to-moderate stroke, dCA in different stroke subtypes and dCA after intravenous recanalization treatment.
Two studies found significantly impaired dCA in the unaffected hemisphere (UH) after severe IS.71,72 However, this was not confirmed by Salinet et al., who did not find a significant difference in UH dCA status as compared to a healthy control (HC) group. 70 In the case of mild-to-moderate stroke, there are more contradictory findings; from one side, some results (3 studies) demonstrated no dCA difference.54,69,73 On the other hand, Atkins et al found significant impaired dCA in the AH as compared to HC subjects, leaving unanswered the question of whether dCA is changed in the sub-population of mild-to-moderate stroke patients. 68 As in the case of UH one possible reason for this conflicting findings is that our review considered dCA measurements taken in the first 48 hours of stroke onset, while there are studies that demonstrated a compromise of dCA in later stages of acute IS patients.76,77 Thus, the hypothesis would be that severe IS patients have immediate dCA disturbance mainly in the AH while mild-to-moderate stroke patients have more subtle dCA change or even no impairment. The rationale for this hypothesis is that during the ischemia there is a global autonomic and inflammatory response and factors like lactate and free oxygen radicals contribute to the dCA failure by direct vasoparalysis in the occluded, reperfused vascular territory and also in the UH.54,75,78,79 In this line of thought, the dCA change would be earlier in the territory of large vessel occlusion due to more injured tissue releasing cellular substances. This reinforces the need for future studies to look at individual patients and separate the IS population under study, not just for admission stroke severity but also for the moment when the dCA result was collected and reperfusion was established.
Regarding stroke subtype, one study demonstrated that lacunar stroke (LS) had a bilateral compromise of dCA. Immink et al. compared measurements in LS patients (both hemispheres) with the AH of severe large artery stroke and concluded that in both patient populations dCA was compromised. However, it seems that dCA is lower – although not significantly so – in more severe stroke patients. 65 On the other hand, Saeed et al., using the method of autoregulation index (ARI) with spontaneous or induced ABP fluctuations, found that dCA status 66 was not related to stroke subtype (classified as total/partial anterior circulation syndrome, lacunar syndrome, and posterior circulation syndrome); 66 however, it should be pointed out that in this work, the stroke severity was not reported. The finding was corroborated by Llwyd et al. that did not find a relationship amongst different stroke subtypes and dCA. These contradictory findings raises the question of whether large vessel occlusion stroke leads to direct change of the affected vascular territory (and perhaps the failure of collaterals), while lacunar stroke has a more global and less severe dCA reduction caused by long standing microvascular compromise of both hemispheres. As lacunar strokes are the result of cerebral small-vessel disease,80,81 it is hypothesized that the reduction of CA is not the result of acute vessel blockage per se, but more the result of a chronic condition like hypertension.65,82 This hypotheses reinforces the idea to further explore different stroke groups according to aetiology of stroke.
Interestingly, just one study evaluated dCA changes in patients with transient ischemic attack (TIA) in the acute phase. 68 TIA increases the risk of an ischemic event, and preventive strategies can limit the burden of stroke in the population. 83 Although Atkins et al. did not find a change in dCA in their TIA population, their cohort was small and no follow-up of their patients was done. In one study, which evaluated patients in the subacute phase of TIA, there was a marked elevation of ABP variability but normal dCA status. 84 Thus, we propose to further evaluate the dCA status and other hemodynamic variables in the acute phase of TIA, as it represents a potential opportunity for secondary prevention and understanding stroke pathology. 84
The dCA status during recanalization treatment with recombinant tissue plasminogen activator (rtPA) was investigated in one study. Reinhard et al. found intact dCA in nine patients with stroke receiving IV thrombolysis compared to rtPA-naïve and HC. 54 The authors concluded that there is no impact of the rtPA treatment on autoregulation status. 54 Investigating the hemodynamic changes during pharmacologic recanalization treatment is an important issue that should be further explored. Although recanalization therapies with mechanical thrombectomy have been increasing lately and constitute the most promising treatment for large vessel occlusion, this therapy remains in limited use in both developed and developing countries.85,86 The rationale for studying dCA in rtPA treated patients is that in this population, dCA could be diminished not just by the ischemic process but also by the pharmacological toxic properties of the drug itself, as demonstrated in animal studies. 87 Although Reinhard et al. did not find an association between rtPA treatment and dCA change, it should be pointed that they investigated a small group of patients in whom dCA was collected later (average 10-20 hours) after therapy, and a transient short-lived period of dysautoregulation could not be excluded immediately after rtPA application. 54
Correlation of dCA and clinical outcomes
The search retrieved eight studies that evaluated the relationship between dCA and clinical outcome, comprising a total of 367 patients (Figure 1(a); Table 2 and Supplemental Table 2).16,17,39,48,54,70,72,88 Regarding the methodology to estimate CBF, most studies (six) used TCD, one used invasive monitoring, and one used NIRS. The modelling most commonly used to analyse dCA was TFA (including ARI) and correlation analysis (Mx, TOx, ORx).
Studies that evaluated the correlation of dynamic cerebral autoregulation with outcome in the acute phase of ischemic stroke.
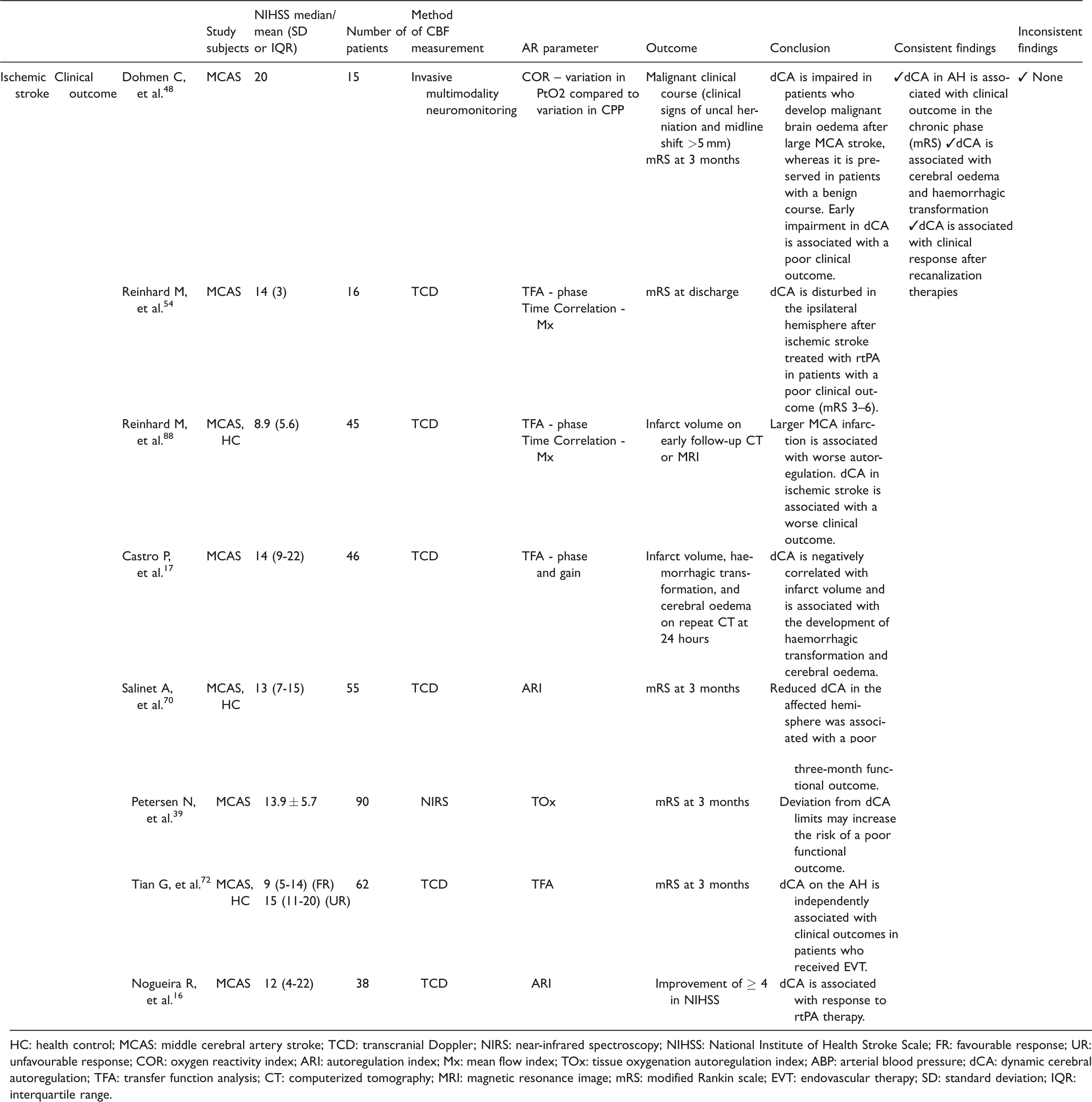
HC: health control; MCAS: middle cerebral artery stroke; TCD: transcranial Doppler; NIRS: near-infrared spectroscopy; NIHSS: National Institute of Health Stroke Scale; FR: favourable response; UR: unfavourable response; COR: oxygen reactivity index; ARI: autoregulation index; Mx: mean flow index; TOx: tissue oxygenation autoregulation index; ABP: arterial blood pressure; dCA: dynamic cerebral autoregulation; TFA: transfer function analysis; CT: computerized tomography; MRI: magnetic resonance image; mRS: modified Rankin scale; EVT: endovascular therapy; SD: standard deviation; IQR: interquartile range.
Consistent findings
There are two consistent findings concerning dCA correlation with clinical outcome and with acute complications such as infarct size, cerebral edema and haemorrhagic transformation.
Regarding clinical outcome, the most consistent finding was that impaired dCA status in the AH is associated with worse clinical outcome as measured by the modified Rankin scale (mRS).39,48,54,70,72,88 The observed relationship was more consistent in more severely affected stroke patients.39,48,54,70,72,88 Another consistent finding from the retrieved articles (4 studies) was related to the relationship between dCA and clinical outcome after recanalization therapies (rtPA treated patients and mechanical thrombectomy).16,39,54,72 Both rtPA studies found that patients who had lower dCA evolved to a worse outcome (NIHSS and/or mRS at hospital discharge).16,54 Although the findings are consistent, the conclusions are limited by the fact that no multivariate correction was applied for outcome with known prognostic factor including recanalization status.16,54 From the thrombectomy studies, Petersen et al., using the NIRS method, demonstrated that the long-term monitoring of dCA may directly impact therapeutic strategies towards individualized management of ABP, while Tian et al. demonstrated that dCA is an important predictor of unfavourable outcome.72,89 This relationship of dCA and outcome during recanalization therapies is an important step towards understanding why a significant percentage of patients do not have good neurological outcomes despite successful recanalization, a so-called ‘futile recanalization’ outcome.14,90 Different hypotheses have been put forward to explain this phenomenon, such as poor collateral and/or microvascular occlusion; 14 however, it also seems plausible to consider the role of dCA impairment. In this scenario, dCA reduction could nullify penumbral recovery by reperfusion injury, as demonstrated by Castro et al. 17
dCA status was also related to final infarct core size17,88 and early complications such as haemorrhagic transformation and cerebral oedema (Figure 2).17,48 Using invasive monitoring, Dohmen et al. measured dCA in intensive care patients with large MCA territory infarction via ORx. 48 Patients who developed malignant cerebral oedema and, consequently, poor clinical outcome showed a high degree of ipsilesional dCA reduction and metabolic derangement (a higher lactate-pyruvate ratio as measured by cerebral microdialysis) compared to patients with a more benign stroke course. This finding was corroborated by Castro et al., who used a non-invasive monitoring technique to show a positive relationship between dCA change and final infarct core volume. 17 The reduction of dCA was an independent predictor for the development of haemorrhagic transformation and cerebral oedema evaluated by brain CT (Figure 2). 17 The pathophysiological process that might contribute is associated with the reduced capacity of the resistance vessels to buffer ABP oscillations and any elevation of hydrostatic pressure across the vascular bed will lead to extravasation of fluid into the brain tissue or to capillary collapse.17,48 Additionally to this process, the cerebral edema may worsen tissue perfusion in regions surrounding the ischemic core with the increased risk of additional regional ischemia and cell death. 48 The knowledge of the influence of dCA in clinical and radiological outcomes is particularly important for developing individual treatments targeted toward optimizing ABP fluctuations and stabilizing the blood-brain barrier.91,92 In addition, the relationship of dCA with cerebral edema could be further explored to guide the ideal time window for indication of last tier therapies such as decompressive craniectomy. 93
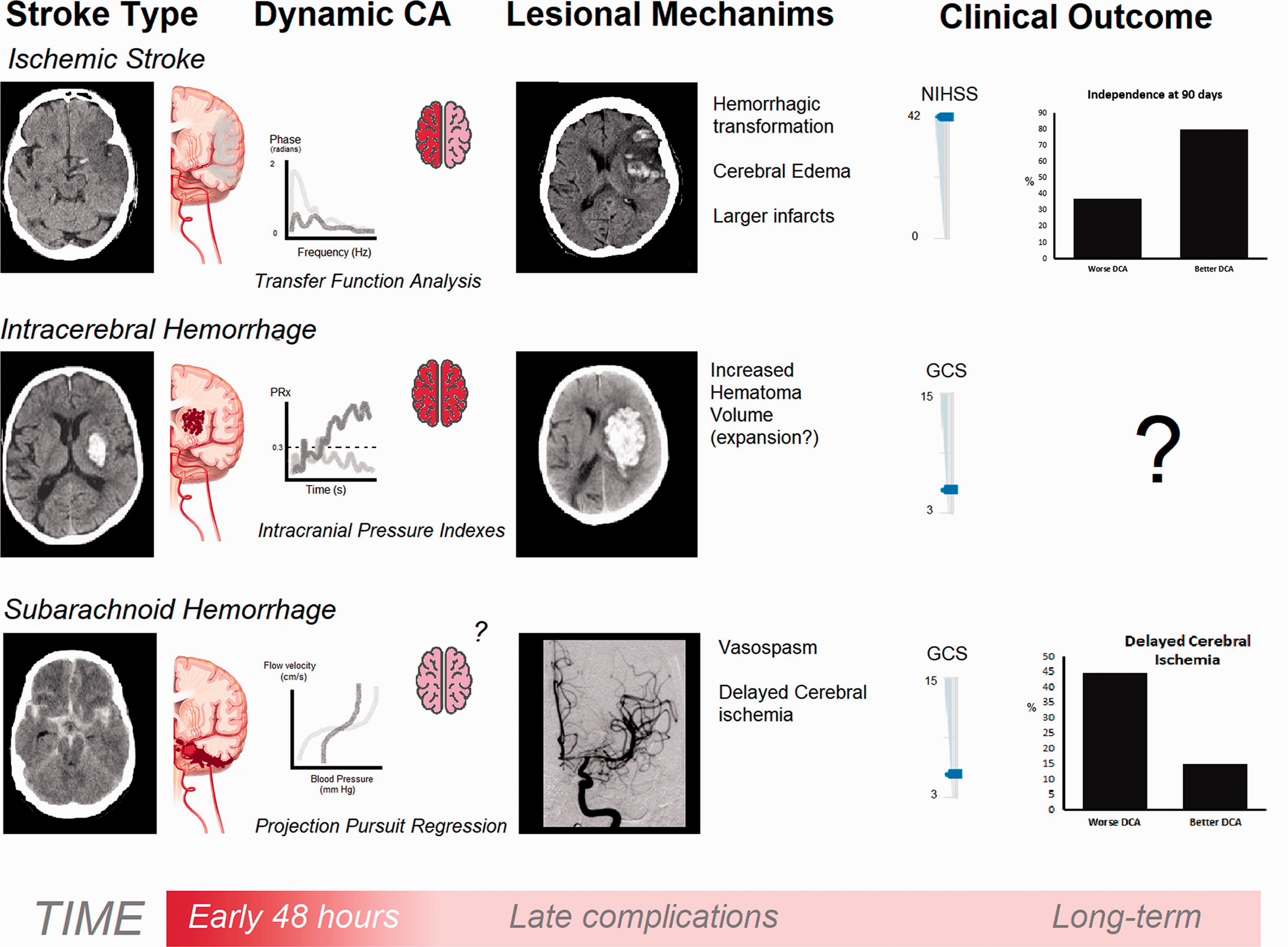
Schematic representation of the main findings concerning dynamic cerebral autoregulation (CA) status at the acute stage of stroke and clinical outcome. From the upper to bottom rows of figures are represented ischemic stroke, intracerebral hemorrhage, and subarachnoid hemorrhage. From left to right, we depicted, sequentially, head computed topographies representative of each stroke type, dynamic CA impairment pattern measured by three methods (just for exemplification), and proposed lesion mechanisms linking CA impairment to the clinical outcome, which is in the last position. In ischemic stroke, a patient with large vessel occlusion presents with reduced phase (impaired CA) in the affected hemisphere but not in the contralateral one. 17 These patients developed hemorrhagic transformation and cerebral edema causing midline shift, hampering the clinical recovery with severe neurological deficits as measured by the National Institutes of Health Stroke Scale (NIHSS). 17 In the intracerebral hemorrhage patient, dynamic CA was assessed invasively with an intracranial pressure probe to measure the pressure-reactivity index (PRx). As compared to patients with better clinical course, these patients showed a PRx plateau pattern mostly near the threshold of 0.3 (darker line), which is believed to be the limit of a competent CA. This occurs in patients with large hematoma volumes and it is not known if CA is a reflex of stroke severity. This has also not been proved to be an independent prognostic factor in the long term. 47 Finally, in subarachnoid hemorrhage, a patient is represented in whom there is a steeper response of cerebral flow velocity to pressure oscillations (darker line); this predicted the development of vasospasm and delayed cerebral ischemia.. 103
Inconsistent findings
There were no inconsistent findings regarding studies that investigated dCA and its association with outcome.
Limitations/knowledge gaps and future perspectives
The most common factor inhibiting our ability to draw firm conclusions is the heterogeneity in included patient numbers across the different studies (Supplemental Tables 1 and 2). Most studies included fewer than 50 patients (10 out of 18 studies) and only one study included more than 100 patients. 69 IS stroke patients comprise a heterogeneous population in which different individual patient, stroke, and treatment characteristics may influence the final dCA status. We propose that future studies report at least the following important variables, which were identified in this review to influence dCA: stroke severity, stroke subtype, side of measurement, vascular comorbidities, applied and result of recanalization therapies, collateral vessel status, and radiological variables like penumbra and infarct core.
In addition, interventional studies with ABP manipulation in the acute phase of IS should include dCA monitoring. There is a lack of clear guidelines concerning individual ABP management during the acute stroke phase 94 with studies pointing towards a clear influence of this variable on clinical outcome. 75
dCA in ICH studies
Dynamic cerebral autoregulation status
The search retrieved four studies with a total of 113 patients with deep or lobar ICH (Figure 1(a); Table 3 and Supplemental Table 3).47,95–97 All studies used spontaneous ABP fluctuations as input and used ICP (Pressure Reactivity Index, PRx) 47 or TCD (TFA or Mx) monitoring derived dCA indices.95–97
Studies that evaluated dynamic cerebral autoregulation status in the acute phase of intracerebral haemorrhage.
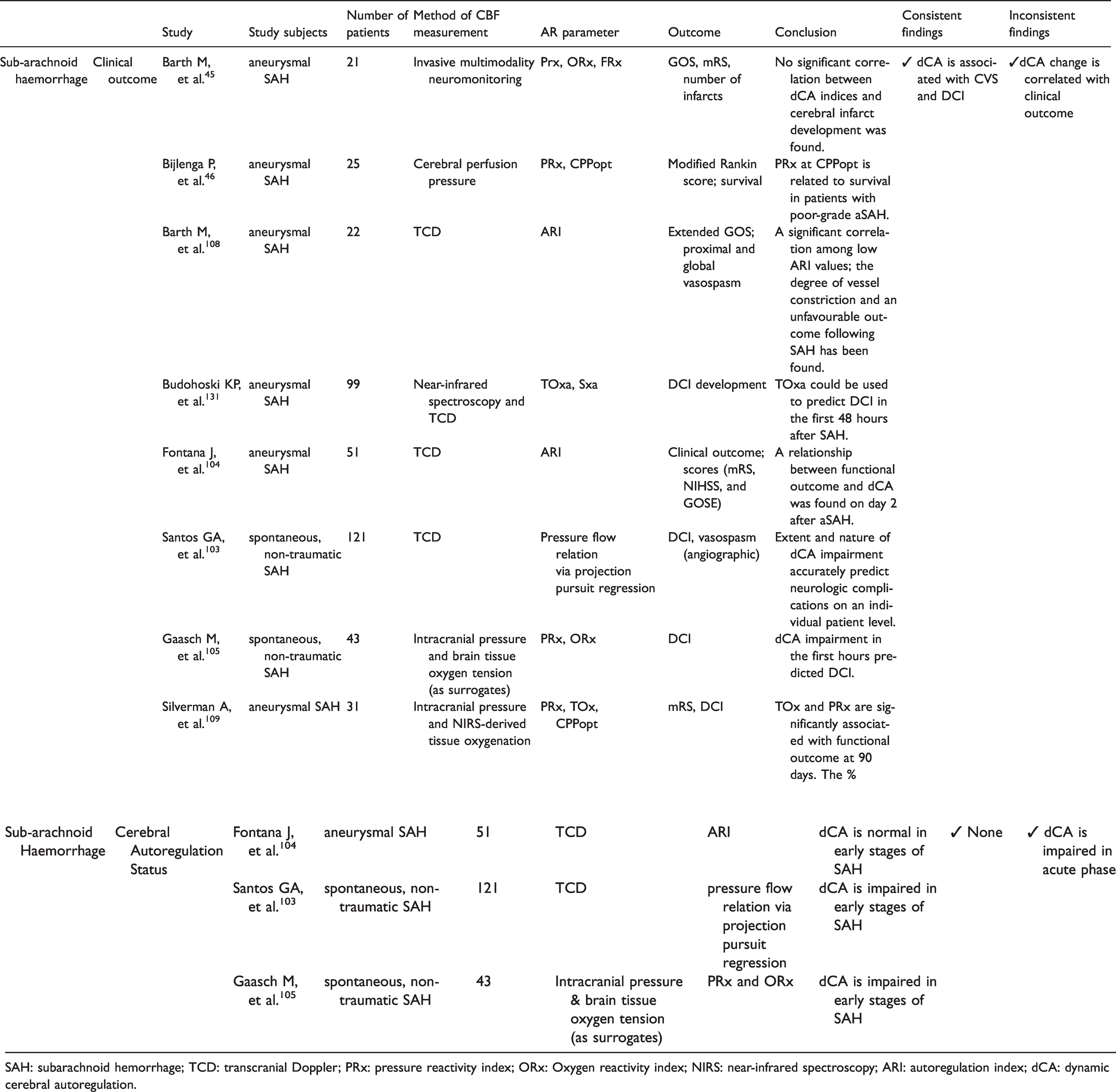
ICH: intracerebral hemorrhage; HC: healthy controls; NIHSS: National Institute of Health Stroke Scale; TCD: transcranial Doppler; PRx: pressure reactivity index; Mx: mean flow index; ORx: oxygen pressure reactivity index; TFA: transfer function analysis; dCA: dynamic cerebral autoregulation.
Consistent findings
There were no consistent findings regarding dCA status from the retrieved studies.
Inconsistent findings
The only inconsistent finding disclosed in our review concerns the dCA status in the acute phase of ICH.
Two studies concluded that dCA was changed bilaterally or ipsilateral to the hematoma.47,95 One of the studies evaluated patients with severe deep ICH stroke (high NIHSS and on mechanical ventilation) submitted to neurosurgery for ventricular drainage placement. The authors used invasive multimodal neuromonitoring with ICP and PtO2 to measure dCA (PRx and ORx). Although the study disclosed that dCA was impaired in the perihaemorrhagic region, the authors could not demonstrate a global reduction (Figure 2). 47 It should be noted that the study classified impairment of dCA based on a cut-off value that is not largely validated for stroke. 47 The other study evaluated a large number of patients with mild to moderate lobar and deep ICH stroke using a non-invasive method of dCA measurement, disclosing diminished dCA compared to healthy control patients. 95 On the other hand, two studies did not find any dCA change compared to HC.96,97 The two studies used the same cohort and included 26 patients with mainly deep ICH (81%). Thus, based on only four studies, it is still not clear whether dCA is compromised in the acute phase of ICH. The only unrefuted finding was that dCA is probably diminished in the peri-haemorrhagic region. 47
Interestingly, but not part of our review inclusion criteria, of the two studies showing contradictory results in the acute phase, both report diminished dCA at later stages (> 48hs) of ICH.95–97 This points towards the importance of comparing dCA during different periods of the stroke phase.
The haemorrhage causes an acute inflammatory process by either compression of brain structures or irritation caused by blood, eliciting a metabolic response mainly in the regions adjacent to brain haemorrhage with hyperglycolysis, lactate acidosis, release of glutamate and other vasotoxic substances, which will culminate in a metabolic crisis. The brain region affected by the metabolic crisis is also known as the metabolic penumbra.95,98 This metabolic crisis usually starts between two to four days and resolves by five to eight days after the stroke onset. 98 Diedler et al. demonstrated a dCA reduction in this region but a preserved global dCA over the first 48 hours. The concept of a ‘dynamic’ metabolic penumbra which affects dCA should be kept in mind when planning therapeutic strategies such as aggressive ABP control in the early stages of ICH.
Correlation of dCA and clinical outcomes
The search retrieved only three studies, with a total of 95 patients, that investigated the clinical impact of dCA in ICH (Figure 1(a); Table 4 and Supplemental Table 4).95–97 Of these, two studies used TFA and one used the time correlation index Mx.
Studies that evaluated the correlation of dynamic cerebral autoregulation with outcome in the acute phase of intracerebral haemorrhage.
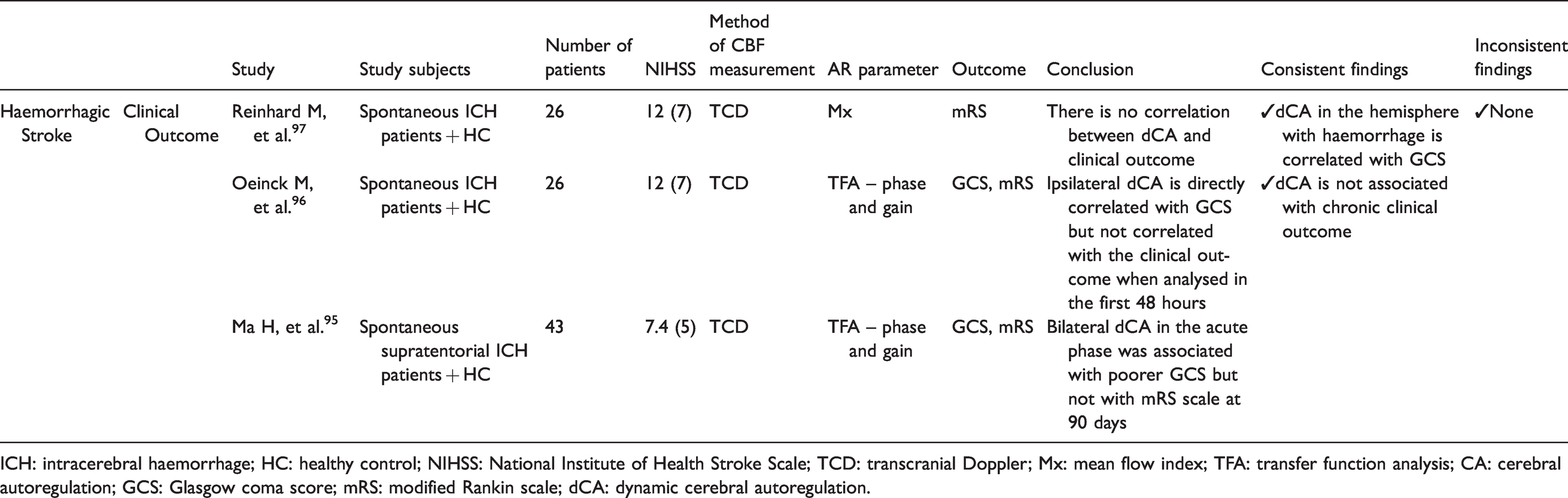
ICH: intracerebral haemorrhage; HC: healthy control; NIHSS: National Institute of Health Stroke Scale; TCD: transcranial Doppler; Mx: mean flow index; TFA: transfer function analysis; CA: cerebral autoregulation; GCS: Glasgow coma score; mRS: modified Rankin scale; dCA: dynamic cerebral autoregulation.
Consistent findings
The consistent finding concerns the association of dCA with early and late clinical outcome.
Oienck et al. 96 and Ma et al. 95 found a significant correlation between diminished dCA (measured by TFA) in the affected hemisphere and the admission Glasgow Coma Scale (GCS). In both studies, no multivariate correction for known prognostic factors was applied. The relationship between admission GCS – as an indicator for bleeding volume – and dCA needs to be confirmed in future studies. Interestingly, the temporal profile of dCA and the relationship with global brain (dys)function could give insights into the inflammatory cascade that evolves over time. We propose to study dCA in patients with similar haemorrhages (same volume, location, age, etc.) correlating this with clinical presentation and radiological diagnostics.
Two studies reported no association between dCA status in the first 48 hours and functional outcome at three months, measured by mRS (Figure 2).96,97
Inconsistent findings
There were no inconsistent findings regarding dCA and outcome from the retrieved studies.
Limitations/knowledge gaps and future perspectives
As with IS, the most common limitations reported by the literature were the small sample size with the heterogeneity of the studied population, the lack of validated thresholds for impaired dCA, and the lack of the reproducibility of data.47,95–97 Two studies used the same cohort of patients.96,97
Studies did not separated patients according to the type or location of the ICH (primary vs secondary haemorrhage and lobar vs deep haemorrhage). There are a few reasons why this should be taken into consideration. Firstly, the location of the hematoma is important because deep haemorrhage is associated in a majority with hypertension, while lobar haemorrhage is often associated with cerebral amyloid angiopathy (CAA).99,100 Although it is not clear whether hypertension has a direct impact on dCA, there is a recent study linking CAA to significant dCA impairment. 101 In addition, most secondary ICH causes (e.g. cerebral malformation, cerebral venous thrombosis, etc.) present as a lobar hemorrhage, and there might be a direct link between brain abnormalities, dCA change, and ICH specially in the young and healthy population.99,100 Finally, patients with deep ICH (the most common cause reported by the studies in this review) usually have multiple comorbidities (e.g. hypertension, diabetes, etc.) and the change of dCA could be a consequence of comorbidities and not the haemorrhage itself. Thus, reporting of the ICH location is important for the interpretation of the dCA results.95,99,100
Another important factor to take into account in future studies is the influence of anti-hypertensive drugs in the acute phase. Hypertension is the most common cause of (deep) ICH, 99 but nearly all patients present with high ABP at hospital admission, which decreases over the days. Whether aggressive treatment of hypertension is needed and to what extent in individual patients is still a matter of debate but most guidelines advise lowering ABP in the acute phase to limit hematoma growth.99,100,102 This therapy, which could potentially influence cerebral perfusion negatively, and the incorporation of dCA status might guide individual ABP targets over time (please see chapter 5 of this special issue for more information).
dCA in SAH
Dynamic cerebral autoregulation status
The search retrieved three articles with a total of 215 patients evaluated for dCA during aneurysmal SAH. All studies used TCD as the monitoring technique (Figure 1(b); Table 5 and Supplemental Table 5).103–105 One study used PtO2 and ICP-derived indexes. 105 The articles used different methodologies to evaluate dCA (project pursuit regression, 103 correlation analysis, 105 and ARI method 104 ).
Studies that evaluated dynamic cerebral autoregulation status in the acute phase of subarachnoid haemorrhage.
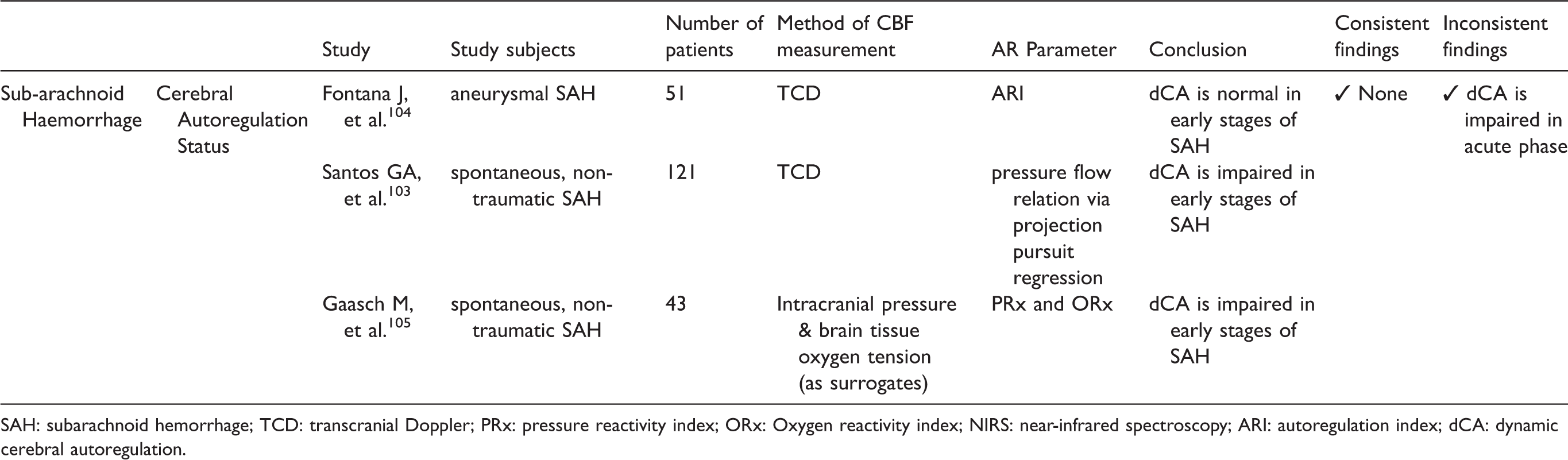
SAH: subarachnoid hemorrhage; TCD: transcranial Doppler; PRx: pressure reactivity index; ORx: Oxygen reactivity index; NIRS: near-infrared spectroscopy; ARI: autoregulation index; dCA: dynamic cerebral autoregulation.
Consistent findings
There were no consistent findings regarding dCA status from the retrieved studies.
Inconsistent findings
The inconsistent finding disclosed in our review concerns the dCA status in the acute phase of ICH.
Of the three articles, two disclosed impaired dCA in the acute (< 48 hours) phase of SAH.103–105 All three articles classified impairment of dCA using cut-off values that were not validated for the SAH population.
During the early stages of SAH, there is an inflammatory process caused by subarachnoid blood. This might trigger macro- and microvascular vasospasm. 106 This is often accompanied by intracranial hypertension – sometimes due to hydrocephalus – and the risk of cerebral hypoperfusion. 107 Whether all these changes lead to dCA change or whether dCA is reduced by the bleeding event directly is not clear. 107 However, there is some clinical evidence that dCA is impaired in the early phase of SAH, which might be an important factor to follow over time.
Correlation of dCA and clinical outcomes
The search retrieved eight articles with a total of 413 patients, evaluating the correlation of clinical outcome and dCA in SAH (Figure 1(b); Table 6 and Supplemental Table 6).40,45,46,103,104,108 Methods to assess dCA varied widely. Most studies used TCD to compute the ARI,104,108 or the index Sx (representing the correlation between slow waves in CPP and TCD systolic cerebral blood flow velocity).40,108 In three studies, ICP signals or PtO2 monitoring signals were used to calculate two time correlation indices: PRx and associated CPPopt and ORx, respectively.45,46 One study used NIRS to calculate the time correlation index, TOx. 40
Studies that evaluated the correlation of dynamic cerebral autoregulation with outcome in the acute phase of subarachnoid haemorrhage.
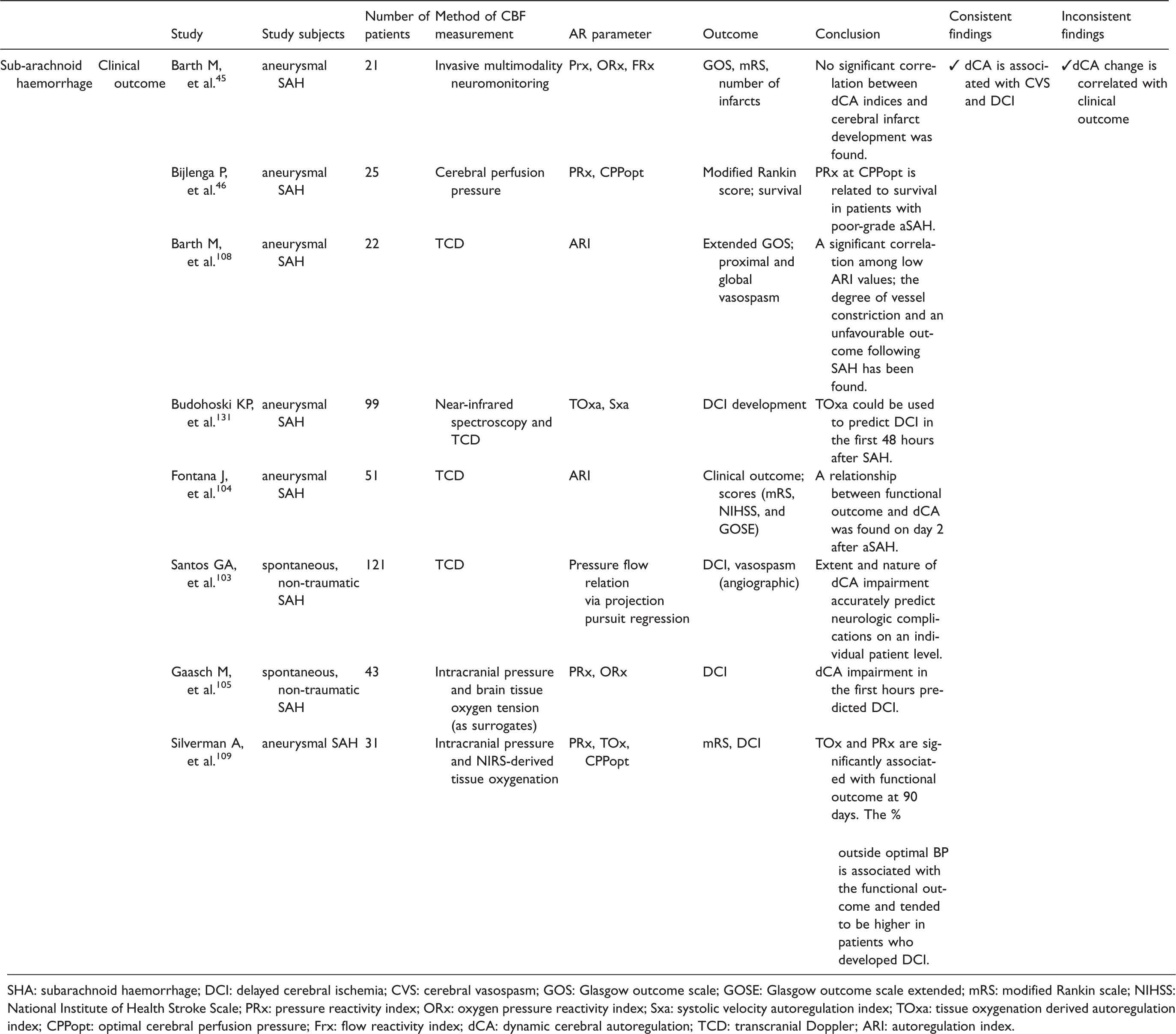
SHA: subarachnoid haemorrhage; DCI: delayed cerebral ischemia; CVS: cerebral vasospasm; GOS: Glasgow outcome scale; GOSE: Glasgow outcome scale extended; mRS: modified Rankin scale; NIHSS: National Institute of Health Stroke Scale; PRx: pressure reactivity index; ORx: oxygen pressure reactivity index; Sxa: systolic velocity autoregulation index; TOxa: tissue oxygenation derived autoregulation index; CPPopt: optimal cerebral perfusion pressure; Frx: flow reactivity index; dCA: dynamic cerebral autoregulation; TCD: transcranial Doppler; ARI: autoregulation index.
Consistent findings
The consistent finding concerns the association of dCA status with complications such as cerebral vasospasm (CV) and delayed cerebral ischemia (DCI).
The most consistent finding disclosed by six studies is that dCA is associated with the development of the major complications of SAH, which are CV and DCI (Figure 2).40,103–105,108,109 Most authors considered dCA impairment as an “early” warning marker before major complications are diagnosed around days 4–6 after ictus. Santos et al. observed that patients who developed DCI had dCA impairment resulting in a significant reduction in cerebral perfusion following transient reductions in ABP. 103 Few studies also included non-aneurysmal SAH patients, which might have influenced the findings.103,105
CV is a common and feared complication of SAH presenting in about 70% of cases. 110 Its pathophysiological mechanisms are attributed, but not limited, to the amount of inflammatory response caused by the blood. Large vessel CV has been a target for many medical and mechanical therapeutic interventions but, surprisingly, the improvement of CV does not directly translate into better clinical outcomes. 111 Authors have reported metabolic responses with the release of inflammatory cytokines that lead to microvascular dysfunction and/or thrombosis causing tissue hypoxia and ischaemia independent of large vessel diameter. 107 In this context, dCA might be a sensitive method to detect the failure of microvascular function. This might be the reason why dCA is highly correlated with DCI (Figure 2) and might be used as an early cerebral monitor or even as a target to guide focal interventions.
Inconsistent findings
The inconsistent finding concerns the association of dCA status with clinical outcome.
Four out of five studies disclosed a correlation of dCA and clinical outcome (measured with mRS, NIHSS, and Glasgow Outcome Scale).45,46,104,108,109 There was no multivariate correction for known prognostic variables and complications. We suggest that future dCA studies aimed to correlate with clinical status should include important complications like CV and DCI, 107 as they have a direct relationship with dCA and also with clinical outcome.
Limitations/knowledge gaps and future perspectives
The main limitations are the small number of studies and patients included. In addition, one study used a methodology that is not widely applied to measure dCA. 103 Common limitations are the use of group threshold values to define dCA impairment and the poor reproducibility of data. In general, the studies that used similar modelling to evaluate dCA had the same results, reinforcing the idea that homogeneous methods should be implemented to get more robust results. 23 The majority of patients studied were poor-grade SAH, biasing the results to this population (Supplementary Tables 5 and 6). In addition, in future studies, the definitions for complications such as DCI and clinical outcome should be more uniform.
Discussion
It seems plausible to assume that knowledge of the cerebral hemodynamic status during the acute phase of a stroke is very helpful for evaluating individual therapeutics in the critical phase. 22 With the current bedside dCA monitoring methods, there are possibilities for monitoring the intermittent (mainly non-invasive) or continuously (mainly invasive) hemodynamic consequences of therapeutic interventions like recanalization therapies and active ABP lowering.18,36,58,112 Sheriff at al, in a recent paper demonstrated the importance of monitoring dCA after EVT and reinforces the need to include variables such as end-tidal CO2 (ETCO2) as a monitoring parameter. 112 In addition, another recent study demonstrated that interventional protocols based on dCA status is safe and feasible. 18 Guiding therapeutic decisions based on dCA measurement is the fundamental background of the INFOMATAS initiative. Our review of dCA studies in acute IS, ICH, and SAH demonstrated that despite some consistent findings, dCA is still mainly in the conceptual and explorative phase. At the moment, the heterogeneity of studies (with different populations, broad methodology options, short measurements, and inconsistent short and long outcome relationships) limits the practical application and validation of dCA to guide treatment.30,64 Based on the review, the next step of the project, which is already in course, will be to gather dCA databases from different centres to perform a meta-analysis of the already published stroke data. 64 The objective of the large meta-analysis is to confirm or refute the findings of this review. If the relationship of dCA and clinical outcome is confirmed, the group will proceed to analyse the original data using a homogeneous methodology, which will form the basis of an intervention study to optimize blood pressure in the acute phase of stroke to improve outcome or reduce complications. .
In the following section, we will summarize the main findings, limitations/gaps (Figure 3(a) and (b)) and recommendation for future trials and/or observational studies.
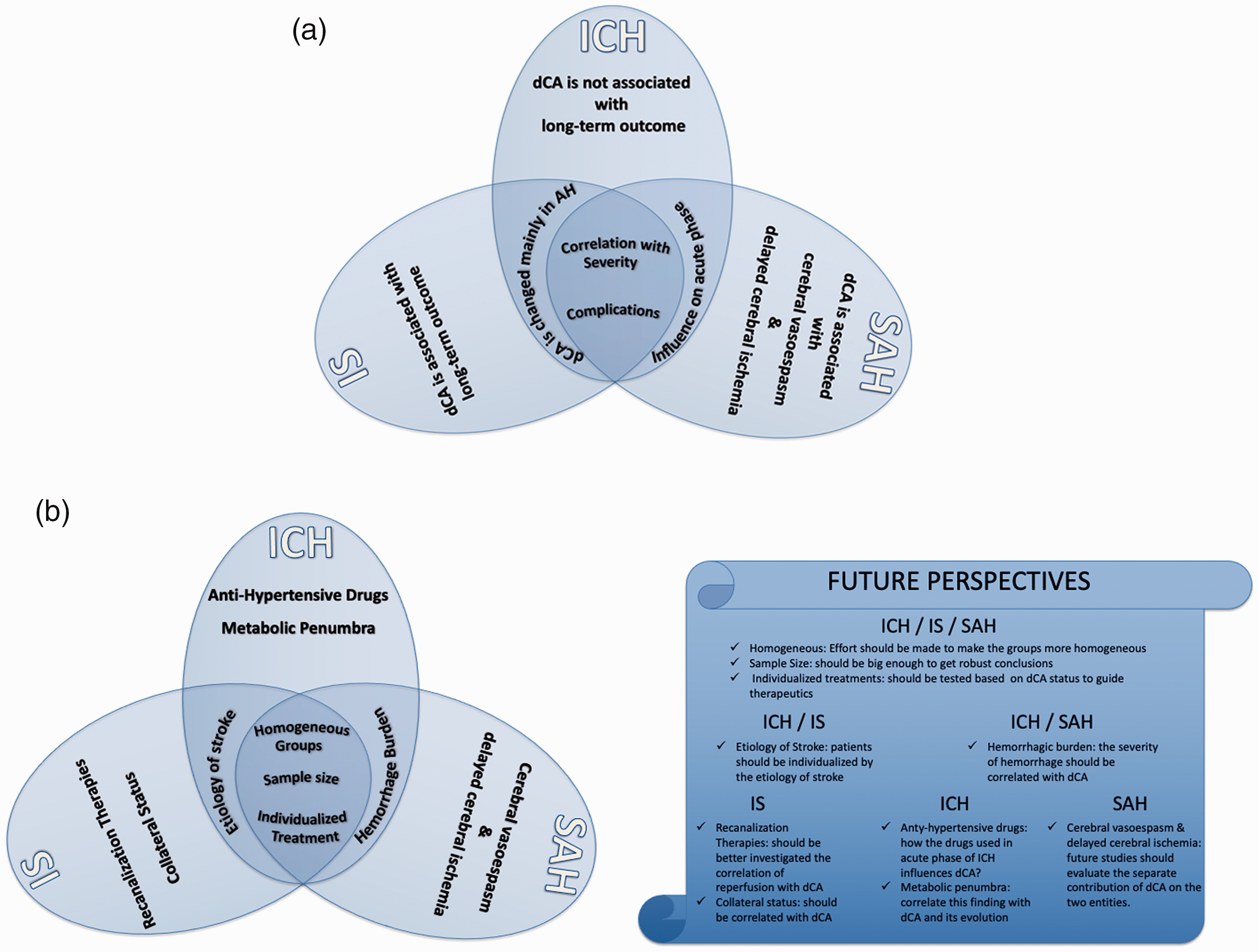
(a & b). a: main findings from the articles; dark blue, common findings for all three clinical conditions (IS, ICH and SAH); intermediate blue, common finding for IS/ICH and ICH/SAH; light blue, findings for each clinical condition. b (left): main limitations from the articles; dark blue, common limitations for all three clinical conditions (IS, ICH and SAH); intermediate blue, common limitations for IS/ICH and ICH/SAH; light blue, limitations for each clinical condition. b (right): perspectives for future studies based on the limitations.
Summary of main findings
This review retrieved 30 studies involving a total of 1700 acute stroke patients. An important consistent finding is that stroke severity is positively correlated with dCA. This is most obvious for the AH. Patients most at risk for impaired dCA are those developing complications like (malignant) cerebral oedema after large vessel IS, those who need intensive care after large ICH, and the poor-grade SAH patients who develop VS and/or DCI (Figure 3(a)).
In ICH and SAH, a relationship between dCA and clinical presentation was found, whereas in IS patients a correlation between poor long-term clinical outcome (e.g. mRS at three months) and impaired dCA was demonstrated (Figure 3(a)). This might indicate different pathophysiological and disease-specific processes leading to dCA impairment but conclusions are hampered by the amount of studies with specific aims. Therefore, at present, dCA results in ICH and SAH might be helpful in the detection and early treatment of complications, whereas in IS, dCA results might assist the clinicians in outcome prediction and evidence of effectiveness of interventions (Figure 3(a)).
Summary of main limitations and perspectives for the future
The small number of patients included in individual studies was the most common consistent overall limitation. As a consequence of this, no studies fulfilled the criteria for reporting on homogenous groups of stroke patients in terms of stroke severity, stroke mechanism, vascular comorbidities (e.g. pre-morbid hypertension), recanalization therapies, complications, and clinical outcome (Figure 3(b)). In addition, despite most of the studies controlled for CO2 changes, a few did not report this variable39,67,105,109 which is very important and recommended to be included in dCA studies. 23
Another common limitation observed was the paucity of studies that investigated individualized treatment based on dCA findings (Figure 3(b)). 39 In the acute phase of stroke, it is common to have hemodynamic instability99,107,113 and the monitoring of multiple parameters would be helpful to guide clinical decisions. Autoregulation-guided ABP management is a promising advance over the classical approach of maintaining ABP within a fixed, pre-determined range.39,46 After an acute stroke, the loss of dCA increases the risk of neurological worsening from hypo- or hyperperfusion. 39 Only a few studies have demonstrated that continuous dCA monitoring can identify the ABP range in individual patients at which cerebral autoregulation is most preserved.89,109 Furthermore, exceeding this ABP range has been associated with an increased risk of worse functional outcomes. 39 However, despite good conceptual reasons and observational data, we currently lack rigorous clinical evidence to support autoregulation-guided ABP management. Continuous monitoring of dCA parameters derived from robust ICP and CPP signals has been proven to be a very interesting and potentially helpful tool in comatose patients with severe TBI. 114 In retrospective studies, individual deviation from autoregulation guided CPP (CPPopt) levels was associated with poor clinical outcome. Hypoperfusion was associated with mortality and hyperperfusion with severe disability at six months. 114 In the same line of thought, it would be reasonable to think that evaluating dCA in IS, ICH, and SAH patients using the principle of individualized autoregulation guided treatment could overcome complications, adjust cerebral perfusion needs and improve the clinical outcome. However, as pointed out in our review, the main method used in the studies is TFA, which is a method that, at present, is so far not tested to monitor and guide clinical management using dCA information in real time at the bedside. 23 Correlation methods like PRx and Mx, which are used to construct treatment targets (like CPPopt and ABPopt), have limitations in terms of sensitivity and specificity for short periods of data acquisition, which is commonly used for the non-sedated patient like most stroke patients. 23 Thus, there is a need to investigate the feasibility of incorporating different modelling methods, with less invasive monitoring techniques other than ICP, to measure or challenge dCA in a way such that they can be applied in the clinical setting to provide reliable dCA data in a short period of time.
In most studies, the aetiology of stroke (e.g. mechanism of IS, primary, secondary ICH, etc.) was not accounted for in the analysis. As pointed out in each section, there is a need to take into account the aetiology of stroke.22,115 Additionally, for IS patients, patients submitted to recanalization therapies should be further studied, and the collateral circulation should also be accessed in dCA studies (Figure 3(b)). The collateral status has been put in evidence in the last years because not only does it enhance the response to recanalization therapies but it also may have an influence on dCA status116,117 (please refer to chapter 1 of this special JCBFM issue for more information). Still regarding recanalization studies, there is no study that included dCA evaluation before the therapy, although this might be unfeasible and unethical due to the delay to start the treatment. On the other hand, for ICH stroke patients, special attention should be paid to the interaction of dCA and anti-hypertensive treatment and the metabolic penumbra, which is the region surrounding the hematoma area with metabolic derangement (Figure 3(b)), 98 as this may be the region where the dCA deterioration starts., It would be interesting to see whether the metabolic derangement has similar temporal course as dCA.
One limitation common to patients with ICH and SAH was that all the studies did not consider the known prognostic features associated with severe haemorrhage (ex. hematoma volume, SAH extension, etc,) in the dCA evaluation. Adverse prognostic features are associated with poorer outcome and greater complications (ex. DCI, re-bleeding, etc.),99,107 and future studies should consider evaluating dCA during therapeutic interventions including ABP lowering treatment, aneurysm interventions, hematoma evacuation or treatment of hydrocephalus (Figure 3(b)). The burden of haematoma could be the cause or consequence of dCA impairment, and interventions towards cerebral hemodynamic status could prevent or ameliorate this complication. Finally, in SAH studies, although a high association was demonstrated amongst dCA, DCI, and CV, it was not possible to dissociate the separate influence of this association. DCI and CV are probably separate complications with different pathophysiology mechanisms and possible different influences in clinical outcome (Figure 3(b)). 107
INFOMATAS recommendations for future trials and/or observational studies
In this section the group defined specific recommendations based on the evaluation (grading) of main findings disclosed in this review (Table 7). It is paramount that all the recommendations for planning future studies consider the limitations/gaps discussed above.
Specific recommendations with grade of evidence based on the main findings disclosed in this review.

IS: ischemic stroke; ICH: intracranial hemorrhage; SAH: subarachnoid haemorrhage; dCA: dynamic cerebral autoregulation; AH: affected hemisphere; UH: unaffected hemisphere; TIA: transient ischemic attack; GCS: Glasgow coma scale; DCI: delayed cerebral ischemia; CV: cerebral vasospasm; NIHSS: National Institute of Health Stroke Scale; mRS: modified Rankin scale.
AIS recommendations (Table 7)
Six studies (242 patients) confirmed that dCA is altered in AH of severe AIS..54,65,67,70–72 In addition, six studies (306 patients) correlated dCA status with short and long term clinical outcome in severely affected stroke patients.39,48,54,70,72,88 Thus, the group strongly recommends that dCA status should be corrected for in prognostic or interventional studies with clinical outcome endpoints. This recommendation also applies to patients submitted to recanalization therapies. Finally, three studies (106 patients) found a significant correlation of dCA with AIS complications such as cerebral edema and hemorrhagic transformation17,48,88 and it is strongly recommended that this association is of study priority in the near future (Figure 3(b)).
There is only moderate/weak evidence (2 studies supporting (82 patients)71,72 and 1 study against (232 patients) 70 ) to recommend focusing on UH dCA status, This is also valid for mild/moderate stroke patients (1 study supporting (58 patients) 68 and 3 studies against (177 patients)54,69,73) different stroke subtypes (e.g. TIA, lacunar, etc) (1 study supporting (30 patients) 65 and 3 against (233 patients)66,68,69) and after intravenous recanalization treatment (1 study supporting 54 ) . Thus, in order to investigate better these issues we strongly recommend that future observational dCA studies take into account: stroke severity, stroke subtype, side of measurement, vascular comorbidities, applied and result of recanalization therapies, collateral vessel status, and radiological variables like penumbra and infarct core.
ICH recommendations (Table 7)
Four studies (2 supporting (58 patients)47,95 and 2 against (55 patients)96,97) with contradictory results could not determinate if there is an impairment in dCA in the acute phase of ICH. In addition, just two studies (69 patients) showed a significant correlation of dCA status and initial GCS.95,96 As there is moderate/weak evidence of these findings the group recommends that further studies should be conducted observationally to be able to focus any further investigation on interventional research. For the design of studies, the group strongly recommends that the patients are selected by the location of haemorrhage and, if possible, that the effects of medications in dCA be explored (Figure 3(b)).
SAH recommendations (Table 7)
Six studies (367 patients) demonstrated strong correlation of dCA status with SAH complications such as DCI and CV.40,103–105,108,109 The group strongly recommends that autoregulation guided interventions should take these complications as endpoints (Figure 3(b)).
There is not sufficient data to demonstrate a change of dCA status in acute phase of SAH (2 studies supporting (164 patients)103,105 and 1 study against (51 patients) 104 ) in addition, only a moderate correlation between dCA and clinical outcome was found (4 studies supporting (129 patientts)46,104,108,109 and 1 study against (25 patients). 45 Thus, the group suggest that this should be further investigated in observational studies which should include different SAH severities (Figure 3(b)).
Limitations of the review
Our review has several limitations: 1) we did not perform a meta-analysis with the data retrieved; though the studies are very heterogeneous, it would be interesting to perform an individual patient data meta-analysis (IPDMA) for all the sub-sections, as is planned for acute IS in the INFOMATAS initiative; 64 2) we did not perform a systematic review, although we intended to provide a comprehensive view of the state of the art of the issue; 3) several articles had to be excluded because they included data from patients > 48 hours after the ictus or the time of measurement was unclear or not reported;118–130 and 4) our literature search was limited to the two most commonly used literature databases. However, in all the articles retrieved, the bibliographic reference was checked for further studies.
Conclusion
In conclusion, dCA has the potential to guide clinical practice in the acute phase of stroke with individualized strategies aimed at achieving the best individual CBF. This review points out the strengths and weaknesses of the studies evaluating dCA in the acute phase of stroke and provides a road map for future study design and delivery. There is an urgent need to gather large samples of data, which will be achieved only through multicentre collaborations and data sharing of AIS, ICH, and SAH patient haemodynamic measurements. This is the main goal of the INFOMATAS group to finally introduce into clinical practice dCA measurement as a useful tool to guide therapeutics.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211045222 - Supplemental material for Review of studies on dynamic cerebral autoregulation in the acute phase of stroke and the relationship with clinical outcome
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211045222 for Review of studies on dynamic cerebral autoregulation in the acute phase of stroke and the relationship with clinical outcome by Ricardo C Nogueira, Marcel Aries, Jatinder S Minhas, Nils H Petersen, Li Xiong, Jana M Kainerstorfer and Pedro Castro in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Jatinder S. Minhas is funded by an NIHR Clinical Lectureship in Older People and Complex Health Needs. The views expressed in this publication are those of the author(s) and not necessarily those of the NHS, the National Institute for Health Research, or the Department of Health, or the authors’ respective organisations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
