Abstract
Postoperative pediatric cerebellar mutism syndrome (pCMS), characterized mainly by delayed onset transient mutism is a poorly understood complication that may occur after pediatric medulloblastoma (MB) resection. Our aim was to investigate postoperative changes in whole-brain cerebral blood flow (CBF) at rest in pCMS patients using arterial spin labeling (ASL) perfusion imaging. This study compared preoperative and postoperative T2-weighted signal abnormalities and CBF using a voxel-wise, whole-brain analysis in 27 children undergoing MB resection, including 11 patients who developed mutism and 16 who did not. Comparison of postoperative T2 signal abnormalities between patients who developed pCMS (mean age 7.0 years) and those who did not showed that pCMS (mean age 8.9 years) patients were significantly more likely to present with T2-weighted hyperintensities in the right dentate nucleus (DN) (p = 0.02). Comparison of preoperative and postoperative CBF in patients with pCMS showed a significant postoperative CBF decrease in the left pre-supplementary motor area (pre-SMA) (p = 0.007) and SMA (p = 0.009). In patients who did not develop pCMS, no significant differences were observed. Findings provide evidence of an association between pCMS, injury to the right DN, and left pre-SMA/SMA hypoperfusion, areas responsible for speech. This supports the relevance of CBF investigations in pCMS.
Introduction
Medulloblastoma (MB) is the most common brain tumor in children, accounting for 15 to 20% of all childhood primary central nervous system neoplasms. About 50% of MB are diagnosed in children younger than 5 years of age. 1 MB arises in the posterior fossa, mostly in the fourth ventricle, with potential adhesion to the brainstem, the cerebellar peduncles (CP) and the dentate nuclei (DN). Clinically, MB symptoms generally progress over a period of weeks to months and include a combination of signs and symptoms linked to cerebellar dysfunction, as well as increased intracranial pressure due to frequent hydrocephalus. Standard of care therapy for MB includes surgical resection, chemotherapy and craniospinal irradiation. However, aggressive resection may be associated with more frequent surgical complications such as cerebellar syndrome, cranial neuropathies, and postoperative pediatric cerebellar mutism syndrome (pCMS). 2
Postoperative pediatric cerebellar mutism syndrome (pCMS) is characterized by delayed onset transient mutism/reduced speech, emotional lability and oropharyngeal dysfunction/dysphagia after cerebellar or fourth ventricular tumor surgery in children. Additional commonly associated symptoms include cerebellar syndrome and cranial neuropathies. The onset of pCMS is delayed by hours to several days after surgery, and symptoms may last from a few days to several months. 3 Although defined as a transient phenomenon, pCMS has been reported as a risk factor for poor long-term outcome following childhood posterior fossa tumor surgery, with more serious neurological, fine-motor, speech and cognitive deficits in patients who develop pCMS compared to those who do not.4,5
The physiopathology and the risk factors of this syndrome are not yet well established, but there is evidence suggesting that MB tumor histology 6 and damage to the DN 7 and the superior cerebellar peduncles (SCP) 8 increase the risk of pCMS. Brain imaging studies investigating the neural basis of pCMS have reported anatomical abnormalities in the dentato-thalamo-cortical (DTC) 9 tract, an efferent pathway comprising axons originating in the DN and passing, via the red nucleus, to the ventrolateral nucleus of the thalamus before projecting to the cortex, including sensory, primary motor, supplementary motor areas and dorsolateral prefrontal cortex (DLPFC). 10 Damage to the DTC tract, caused by a tumor localized in the fourth ventricle and/or its resection may impact the connections between the cerebellum and supratentorial areas inducing cognitive or affective impairments. 11 In this context, perfusion MRI techniques could help to better understand the pathophysiology of pCMS. Indeed, single photon emission computed tomography and PET studies have reported cerebellar 12 and cortical 13 hypometabolism and/or hypoperfusion in patients with pCMS. To the best of our knowledge, only two studies have used ASL-MRI to investigate CBF abnormalities at rest in pCMS and no study has used a voxel-wise, whole brain approach to compare preoperative and postoperative ASL-CBF at rest.
The aim of this study was to investigate postoperative changes in whole-brain CBF at rest in pCMS patients using ASL perfusion imaging which permits quantification of cerebral blood flow (CBF) at rest non-invasively using arterial water as an endogenous tracer. 14 We first compared preoperative and postoperative ASL-CBF in 11 patients who developed mutism after MB resection. We then compared preoperative and postoperative ASL-CBF in 16 patients who did not develop mutism after MB resection. We further compared preoperative and postoperative ASL-CBF between patients who developed mutism and those who did not. In addition, we also studied T2-weighted hyperintensities on preoperative and postoperative MRIs in the different cerebellar regions surrounding the tumor bed between patients who developed mutism and those who did not.
Materials and methods
Participants
This retrospective study included 32 children out of 67 children who underwent surgical resection of fourth ventricular medulloblastoma at our institution between 2012 and 2015. Children who fulfilled all of the following criteria were included in this study: i) age between 2 and 16 years; ii) midline MB localized in the fourth ventricle (with pathology confirmation); iii) surgery performed with the aim of achieving maximal tumor resection iv) no signs of cytotoxic edema in the postoperative scans; v) native French speaker; vi) no speech impairment prior to surgery according to parents’ report; vii) sufficient retrospective data to evaluate for pCMS according to the criteria detailed hereafter; viii) availability of ASL, 3D T2-weighted and 3D T1-weighted sequences with and without gadolinium injection before and after surgery on a 1.5 Tesla MRI scanner.
Ethical approval
This study has been approved by the local Ethical Committee of French Public Hospitals “Ethic Committee Ile de France II”, (N° EUDRACT 2014A00541-46) in agreement with the Declaration of Helsinki of 1975 (as revised in 1983). Written informed consent was obtained from parents or legal guardians for all children participating in this study.
Postoperative clinical assessment
Patients were divided into two groups based on a retrospective review of postoperative clinical notes: patients who developed pCMS, versus those who did not (no-pCMS). The clinical status of children was assessed at different postoperative stages (day 2 in the intensive care unit, day 5 in the neurosurgery unit, day 10 in the oncology unit) and independently reviewed by a senior neurosurgeon (S.P) and a senior pediatric oncologist (C.D) for evidence of pCMS.
Due to the lack of a standardized quantification scale for pCMS symptoms, children were classified according to the main symptoms described in the literature. We used these criteria as follows: (1) primary symptoms including the degree of speech impairment (none, mild or mutism) and the degree of emotional lability (none, mild, or severe); (2) secondary symptoms including the presence of cerebellar syndrome, cranial neuropathies and motor disorders.
Children were then categorized into 2 groups: (1) pCMS with mutism: children with mutism and mild or severe emotional lability for a minimum of two weeks following surgery, and at least two secondary symptoms; (2) no-pCMS: children without mutism (no or mild speech impairment), no emotional lability and only one secondary symptom following surgery. In order to improve text readability, we referred to patients who developed mutism as “pCMS” instead of “pCMS with mutism”. In total, 12 children were classified as pCMS and 20 as no-pCMS. Detailed clinical data for all patients is provided in Supplementary Table 1.
Imaging
ASL-MRI method
Arterial Spin Labeling is a non-invasive perfusion MRI method allows reliable absolute quantification of rest CBF by using magnetically labeling water protons in the arterial blood as an endogenous and a freely diffusible tracer. In ASL, the diffusible tracer is a magnetic label applied to blood water molecules, produced by saturating or inverting the longitudinal component of the MR signal. If all the label arrives at the capillary bed or tissue at the time of imaging, this results in a 3D T1-weighted signal reduction proportional to CBF, called the tagged image, which is compared to a control image, in which the blood water molecules are not perturbed. 15 The quantified CBF is expressed in physiological unit of millilitre per minute per a hundred grams of brain tissue (ml/100 g/min). Because ASL-MRI do not request exogenous contrast agents and has a short acquisition time, it is a promising perfusion imaging method for assessing rest CBF in pediatric patients.
MRI data acquisition
All patients underwent preoperative and postoperative MRI scans on a 1.5 Tesla MRI scanner (Signa General Electric) at Necker Children’s Hospital (Paris, France). The MRI protocol included a 3D T1-weighted fast spoiled gradient-echo sequence (TR/TE 16.4/7.2 ms; FOV 22 × 22 cm2; number of axial slices 240; slice thickness 1.2 mm; in-plane resolution 0.47 × 0.47 mm2; flip angle = 12° and matrix 512 × 512), a 3D pseudo-continuous ASL perfusion sequence (TR/TE 4554/10.5 ms; FOV 24 × 24 cm2; number of slices 40; in-plane resolution 1.82 × 1.82 mm2; post labeling delay (PLD) = 1025 ms; slice thickness = 4 mm; labeling duration = 1500 ms and matrix 512 × 8) and a 3D T2-weighted Fast Spin Echo (FSE) sequence in 3 planes (TR/TE = 2500/102 ms, FOV = 512 × 512 mm2, slice thickness = 1 mm, flip angle = 90°, Echo train length = 127). CBF quantification from ASL perfusion sequence was performed automatically by General Electric Medical 3D ASL software. Details on this process can be obtained in Zaharchuk et al. 16 Only eight patients (3 in the pCMS group, 5 in the no-pCMS group) received sedation with intrarectal pentobarbital (5 mg/kg under 5 years of age) approximatively 30 minutes prior to the scans, administered by an experienced nurse or MRI technologist, for both preoperative and postoperative scans to prevent motions artefacts.
MRI analysis
All preoperative and postoperative MRI scans, including 3D T1-weighted sequences with and without gadolinium injection and 3D T2-weighted sequences, were reviewed retrospectively. A senior pediatric neuroradiologist (N.B) and a senior pediatric neurosurgeon (S.P) blinded to the diagnosis of pCMS independently analyzed MRI scans for T2-weighted hyperintensities in the different cerebellar regions surrounding the tumor bed: vermis, dentate nuclei, cerebellar peduncles and cerebellar hemispheres. High signal changes on T2-weighted images were subjectively classified as either normal or abnormal (invaded, compressed or resected).
Any discrepancies were resolved by consensus with other pediatric neuroradiologists (R.L/V.D.R/C.J.R/D.G). Areas of T2 hyperintensity in the different cerebellar regions were compared between pCMS and no pCMS patients on both preoperative and postoperative imaging. Other imaging characteristics that were recorded included: maximum tumor width; tumor volume; maximum brainstem width (at the pontine level); degree of hydrocephalus evaluated using Evans’ index (defined as the ratio of the maximum width of the frontal horns divided by the maximum width of inner skull); 17 and extent of tumor resection (complete, near total or subtotal) on postoperative MRI scans.
Whole-brain CBF at rest analysis
Quality control
Quality control was performed before and after preprocessing of both 3D T1- and CBF images as a three-steps procedure. First, all images were qualitatively checked for major artefacts such as motion, aliasing, ghosting, spikes, low signal to noise ratio by a senior pediatric neuroradiologist (N.B). Second, a quality control tool implemented in the CAT12 software (http://www.neuro.uni-jena.de/vbm/check-sample-homogeneity/) which automatically check data quality, was applied to anatomical images after 3D T1 segmentation to ensure high quality of the segmentation of GM from WM. Finally, all coregistered ASL images were visually inspected by two experts imaging processing expert engineers (LF) and (HL).
Data preprocessing
Preoperative and postoperative MRI images were preprocessed using Statistical Parametric Mapping 12 (SPM12; Welcome Department of Cognitive Neurology. London, UK, www.fil.ion.ucl.ac.uk/spm).
For the MRI preprocessing, the quantified CBF images (ml/100 g/min) was used.
After a DICOM to NIFTI conversion, 3D T1-weighted images were spatially normalized into the Montreal Neurological Institute (MNI) space and segmented into grey matter (GM), white matter and cerebrospinal fluid using the CAT12 software and the tissue probability maps in SPM12. In addition, preoperative and postoperative CBF images were co-registered to their native GM images. Then, the deformation fields obtained during the spatial normalization of the 3DT1-weighted images were applied to the CBF images to put all images in the same spatial reference. Finally, the resulting images were smoothed using a Gaussian kernel of 6-mm full-width at half-maximum.
Statistical analysis
Clinical and imaging data
Continuous variables were compared using Fisher’s exact test or Student’s t-test after having checked normality distribution of variables. Categorical variables were compared using a Chi-Square test. All statistical analyses were performed with R software (R 3.1, www.r-project.org).
ASL perfusion imaging
Comparison of preoperative and postoperative ASL-CBF at rest in each group
Comparison of preoperative and postoperative ASL-CBF in each groups (pCMS, no-pCMS) was performed using voxel-wise, whole-brain analysis within the framework of general linear model (GLM) in SPM12 with a flexible factorial model considering three factors: subject, time point (preoperative, postoperative) and group (pCMS, no-pCMS). Gender, age, handedness, tumor volume and postoperative degree of hydrocephalus were a priori selected as covariates since these factors may influence ASL-CBF measures. Statistical analyses were performed on the smoothed and normalized CBF images within a binary GM mask excluding the cerebellum. This mask was built from the mean image of the GM maps of all children with a threshold of 0.2. The significance threshold was set at 0.05 with a Family-Wise-Error (FWE) correction for multiple comparisons.
Comparison of preoperative ASL-CBF at rest between groups
Comparison of preoperative and postoperative ASL-CBF between the two groups (pCMS versus no-pCMS) was performed with voxel-wise, whole-brain analysis within the GLM framework in SPM12. Gender, age, handedness, tumor volume and preoperative degree of hydrocephalus were entered as covariates. Statistical analyses were conducted on the smoothed and normalized preoperative CBF images within a binary GM mask excluding the cerebellum.
Comparison of postoperative ASL-CBF at rest between groups
Comparison of postoperative ASL-CBF between the two groups (pCMS versus no-pCMS) was performed with voxel-wise, whole-brain analysis within the GLM framework in SPM12. Gender, age, handedness and postoperative degree of hydrocephalus were entered as covariates. Statistical analyses were conducted on the smoothed and normalized postoperative CBF images within a binary GM mask excluding the cerebellum.
Results
Cohort characteristics
Out of the 32 children who had undergone MB resection, 5 children (1 pCMS, 4 no-pCMS) were excluded after MRI quality control due to motion artifacts (n = 1) or poor image quality causing faulty segmentation or co-registration (n = 4). Thus, a total of 27 children were included: 11 who developed pCMS (36% male, mean age of 7.0 ± 3.5 years) and 16 who didn’t develop pCMS (68.5% males, mean age of 8.9 ± 5.1 years). There were no statistically significant differences between the two groups regarding gender, age at diagnosis and preoperative and postoperative tumor characteristics (Table 1).
Patient and tumor characteristics.
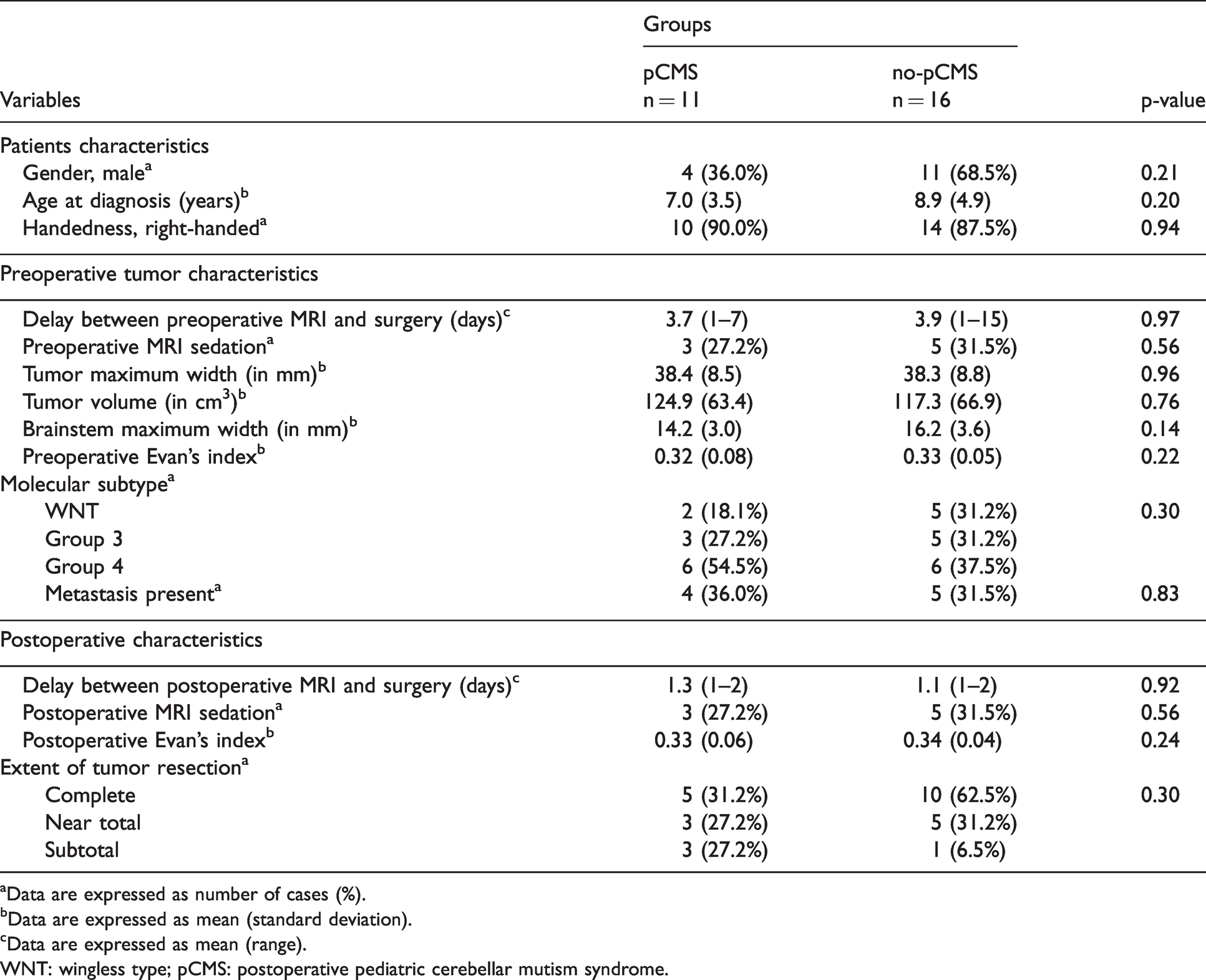
aData are expressed as number of cases (%).
bData are expressed as mean (standard deviation).
cData are expressed as mean (range).
WNT: wingless type; pCMS: postoperative pediatric cerebellar mutism syndrome.
T2-weighted imaging
Comparison of preoperative T2 signal abnormalities between groups
Preoperative MRI scans were performed with a mean delay of 3.8 days (range, 1–15 days) prior to surgery. Preoperatively, there were no statistically significant differences between the two groups regarding T2-weighted hyperintensities in all locations (superior, middle and inferior CP, vermis, DN, cerebellar hemispheres), maximum tumor width, tumor volume, maximum brainstem width and Evans’ index.
Comparison of postoperative T2 signal abnormalities between groups
All postoperative MRI scans were acquired 1 to 2 days after surgery. Postoperatively, patients who developed pCMS were significantly more likely to present with T2-weighted hyperintensities in the right DN (p = 0.02) (pCMS: n = 11/11; no-pCMS: n = 9/16) than those who did not (Figure 1). There were no statistically significant differences between the two groups regarding T2-weighted hyperintensities in other locations (superior, middle and inferior CP, vermis, left DN, cerebellar hemispheres) and the extent of surgical resection. Results of preoperative and postoperative T2-weighted sequence analysis for both groups are shown in Table 2.
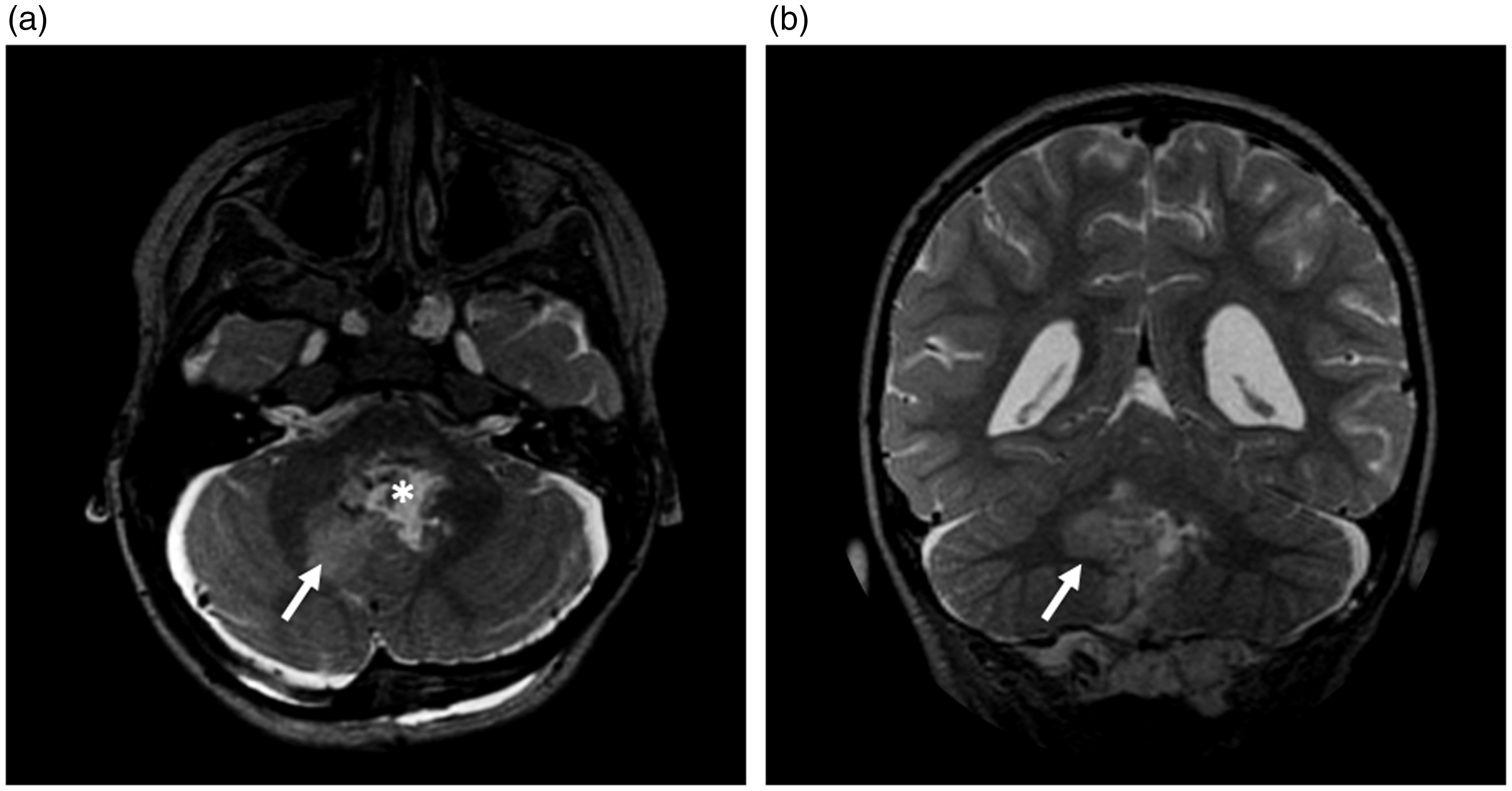
Postoperative axial (a) and coronal (b) T2-weighted MRI images of a 4-year-old patient with pCMS showing obvious damage to the right dentate nucleus (arrow) surrounding the resection cavity (asterisk).
Comparison of preoperative and postoperative high T2 signal abnormalities in the different cerebellar regions surrounding the tumor bed between pCMS and no-pCMS patients.
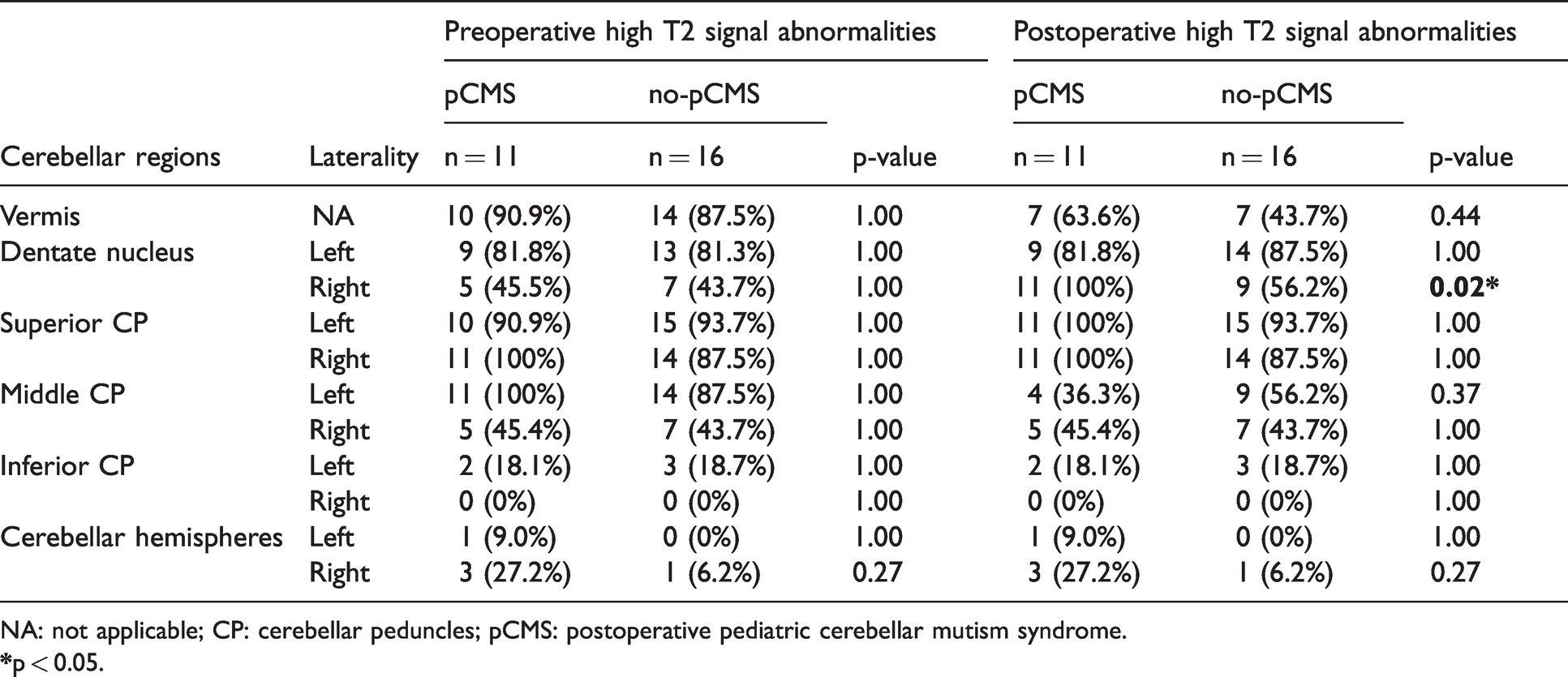
NA: not applicable; CP: cerebellar peduncles; pCMS: postoperative pediatric cerebellar mutism syndrome.
ASL perfusion imaging
Comparison of preoperative and postoperative ASL-CBF at rest in each group
In the pCMS group, voxel-wise, whole-brain comparison of preoperative and postoperative ASL-CBF revealed a significant postoperative decrease in CBF at rest in the left pre-SMA (t = 6.78; z(score) = 5.02; p(corr)=0.007; MNI coordinates: x = −3 y 32 z = 51) and the left SMA (t = 6.53; z(score)=4.94; p(corr)=0.009; MNI coordinates: x = −4 y = 9 z = 64) (Figure 2).
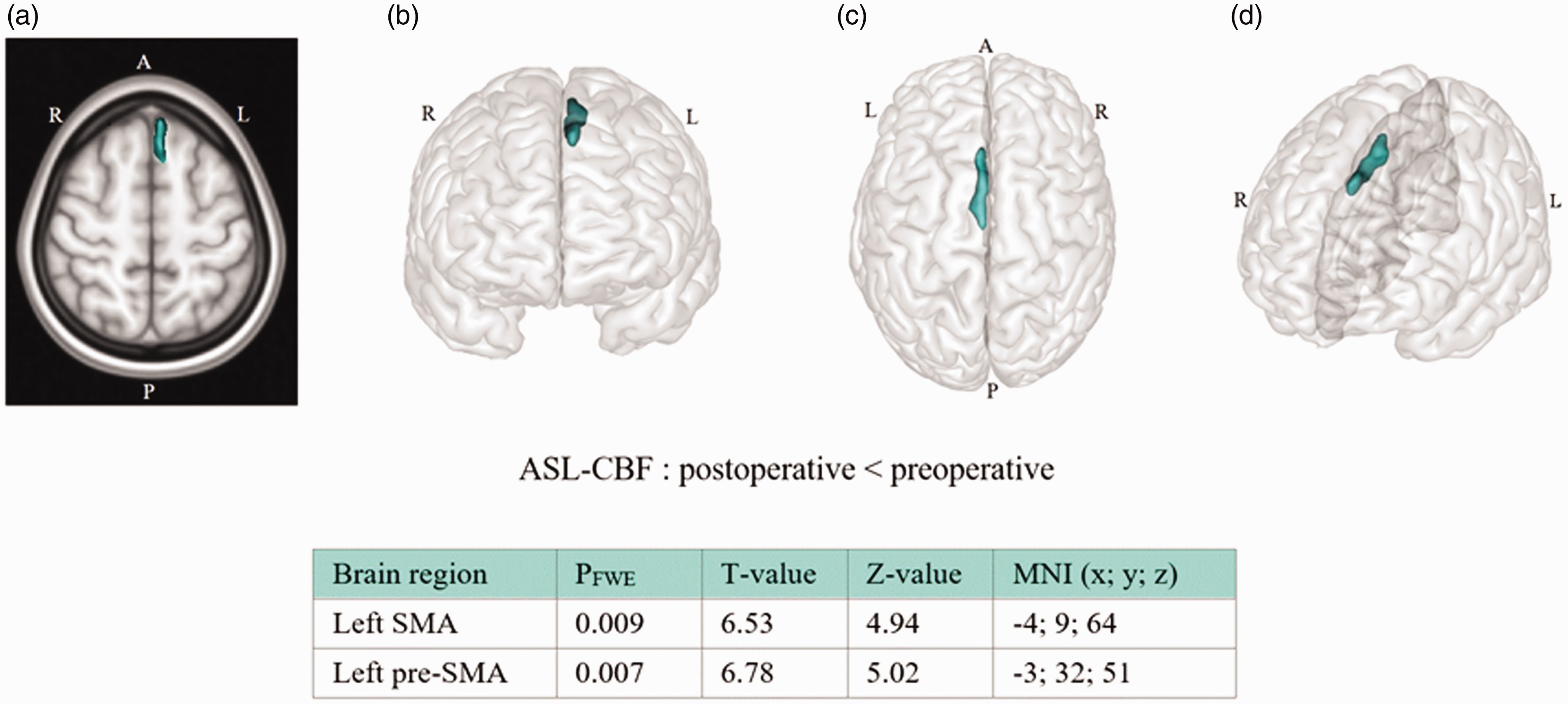
Regional differences between preoperative and postoperative ASL-CBF in patients who developed pCMS following medulloblastoma resection. Maximum intensity projections of T statistic clusters that are significantly different between preoperative and postoperative ASL-CBF were superimposed on a 2D rendering of T1 weighted images in Montreal Neurological Institute (MNI) space in an axial view (a) and on 3D volume rendering of GM in MNI space: front view (b), top view (c), and left oblique view (d). Characteristics of significant clusters are summarized in the table: PFWE is the family wise error (FWE) corrected cluster p-value and MNI refers to the MNI coordinates. Abbreviations: R: right; L: left; A: anterior; P: posterior; CBF: cerebral blood flow; pre-SMA: pre-supplementary motor area; SMA: supplementary motor area; ASL: arterial spin labeling; pCMS: postoperative pediatric cerebellar mutism syndrome; FWE: family wise error; MNI: Montreal Neurological Institute.
In the group that did not develop pCMS, no significant differences were observed between preoperative and postoperative CBF.
An example of axial CBF maps in MNI space in immediate postoperative period from one patient from the pCMS group and one patient from the no-pCMS group is provided in Supplementary Figure 1.
Comparison of preoperative ASL-CBF at rest between groups
On preoperative CBF images, there were no significant differences in CBF at rest between the two groups (pCMS versus no-pCMS) in any brain regions.
Comparison of postoperative ASL-CBF at rest between groups
On postoperative CBF images, there were no significant differences in CBF between both groups in any brain regions using a FWE-corrected threshold of p = 0.05. However, there was a significant decrease in CBF at rest in several brain regions in patients who developed postoperative pCMS compared to those who did not using an uncorrected threshold of P < 0.001: left pre-SMA (t = 3.44; z(score) = 3.07; p(uncorr) = 0.001; MNI coordinates: x = −9 y = 20 z = 58), left SMA (t = 3.45; z(score)=3.08; p(uncorr) = 0.001; MNI coordinates: x = −6 y = 0 z = 70), left thalamus (t = 3.14; z(score) = 2.85; p(uncorr) = 0.002; MNI coordinates: x = −12 y = −14 z = 14) left primary motor cortex (t = 1.84; z(score) = 1.77; p(uncorr) = 0.038; MNI coordinates: x = −16 y = −20 z = 74) and left dorsolateral prefrontal cortex (t = 2.68; z(score) = 2.48; p(uncorr) = 0.006; MNI coordinates: x = −34 y = 57 z = 16).
Discussion
To the best of our knowledge, this is the first study using a voxel-wise, whole-brain analysis to investigate postoperative changes in ASL-CBF at rest in children who developed pCMS after MB resection. Comparison of preoperative and postoperative CBF in patients with pCMS showed a significant postoperative decrease in CBF at rest in the left pre-SMA and left SMA. In patients who did not develop pCMS, no significant differences were observed between preoperative and postoperative CBF. Importantly, there were no statistically significant differences in preoperative CBF between patients who developed pCMS and those who did not. Furthermore, analysis of T2-weighted signal abnormalities showed that patients with pCMS were significantly more likely to present with T2-weighted hyperintensities in the right DN postoperatively compared to those who did not.
The left pre-SMA and SMA hypoperfusion observed in children who developed mutism after MB resection is consistent with the evidence that these areas are involved in speech production. Interestingly, since 10 out of 11 patients with pCMS were right-handed, results observed in left brain areas associated with language (pre-SMA/SMA) join evidence of left lateralization for language in right-handed subjects.18,19 Traditionally the pre-SMA, an area connecting the pre-frontal cortex to the anterior striatum, 20 is associated with the production of volitional movements. 21 Functional neuroimaging studies have also demonstrated that the pre-SMA plays a key role in speech processing, involving internally guided word generation 22 and sentence production. 23 Particularly, a functional MRI study on speech production reported a division of the left pre-SMA into two distinct functional regions: anterior pre-SMA and posterior pre-SMA, respectively implicated in word selection and word form encoding. These results suggest that the left pre-SMA plays a key role in speech initiation. 24 In our study, the left pre-SMA and SMA hypoperfusion observed in pCMS patients suggests that this condition may be associated with perfusion abnormalities in not one but two complementary regions. According to the GODIVA (Gradient Order Directions Into Velocities of Articulators) model, a neural network model of speech production, describing speech planning through the stages of articulation. the pre-SMA would start the encoding word selection process and the SMA would initiate the motor program. 25 The axonal projections between the pre-SMA and the SMA would facilitate the interactions between the basal ganglia, motor, premotor cortex and the SMA to activate the “initiation map” in the SMA starting the pre-articulatory motor program at the appropriate timing. 26 Thus, the GODIVA model provides a relevant framework to interpret our findings and to argue a possible cause for mutism. Indeed, pre-SMA hypoperfusion in children with postoperative mutism would impair speech planning, and subsequently impact the pre-articulatory motor program, which would prevent proper initiation of the cortical motor program responsible for producing sounds at the right moment. We surmise that pre-SMA/SMA hypoperfusion may be the first step of the cortical processes involved in mutism.
Comparison of postoperative CBF images, acquired 1 to 2 days after surgery from both groups, also showed a significant decrease in CBF at rest, at uncorrected threshold, in several brain regions in children who developed pCMS compared to those who did not. Indeed, the hypoperfusion was observed in left brain areas including not only pre-SMA/SMA but also the thalamus, primary motor and dorsolateral prefrontal cortex, which are cortical and subcortical components of the DTC tract. It’s worth noticing that this hypoperfusion does not seem associated with any cerebral vascular effect or impairment, since it was not observed in areas with the same blood supply territory. Importantly, even if described at threshold of 0.001 uncorrected for multiple comparison, hypoperfusion in areas localized along the path of the proximal DTC tract corroborates the current hypothesis of an association between proximal DTC tract injury and pCMS. 9 Moreover, we may assume that damage to the proximal DTC tract caused by the tumor and/or its surgical resection results in hypoperfusion of subcortical and cortical brain areas. The hypoperfusion observed in supratentorial cortical brain areas in pCMS may be associated with a “Crossed Cerebello-Cerebral-Diaschisis” phenomenon. This is supported by the only two previous studies investigating cortical CBF abnormalities at rest in pCMS using ASL-MRI. Firstly, results from a case study showed a significant bilateral decrease in ASL-CBF at rest in the cerebellum, thalami and frontal lobes in a 2-year old patient with pCMS following atypical teratoid-rhabdoid tumor resection. 27 More recently, this “phenomenon” was also highlighted by Yecies et al., identifying a significant postoperative decrease in rest-CBF in the right frontal lobe in 11 patients who developed pCMS following MB resection compared to 10 patients who did not develop pCMS. 28 These results support the robustness of ASL-MRI as a valuable method for identifying Crossed-Cerebello-Cerebral-Diaschisis after stroke,29,30 but also within the context of brain surgery as presented here. In this present study, posterior fossa injury caused by surgery may result in supratentorial cortical hypoperfusion of brain areas responsible for higher-order linguistic, emotional and cognitive functions by a “diaschisis” phenomenon, resulting in functional disabilities. 31 Interestingly, this is consistent with the extent of the hypoperfused areas we observed in this analysis, beyond left pre-SMA and SMA hypoperfusion, which could explain and cover the broader constellations of symptoms (cognitive, emotional and behavioral disturbances) associated with mutism in pCMS.
Comparison of postoperative T2 signal abnormalities between patients who developed pCMS and those who did not showed that pCMS patients were significantly more likely to present with T2-weighted hyperintensities in the right DN. Postoperative damage to the DN has long been supposed to be crucial for the development of pCMS, although findings remain debated with studies reporting either unilateral 9 , bilateral 7 or no damage to the DN. 32 Our results showing postoperative T2 signal abnormalities in the right DN also in 56% of patients who did not develop pCMS support that damage to the DN alone is not sufficient to induce pCMS. Therefore, the mutism would be triggered when the DN damage is associated with other brain abnormalities, such as cortical brain abnormalities as described in this study. In addition, given its anatomical location, the DN is susceptible to be compressed or invaded by the medulloblastoma located in the fourth ventricle. Thus, T2 signal abnormalities in the DN could be associated with the tumor itself or with surgical resection, regardless of the outcome.
Finally, the laterality of the observed T2-weighted hyperintensities, in the right DN, relative to the contralateral, left-sided, cortical hypoperfusion, is consistent with the known decussation pattern of the DTC pathway which terminates in the left cerebral hemisphere. 33 Indeed, given that the DN is the origin of the DTC tract, one may speculate that injury to the right DN caused by the tumor and/or its surgical resection damages the right DTC tract, leading to left pre-SMA/SMA hypoperfusion and mutism (Figure 3).
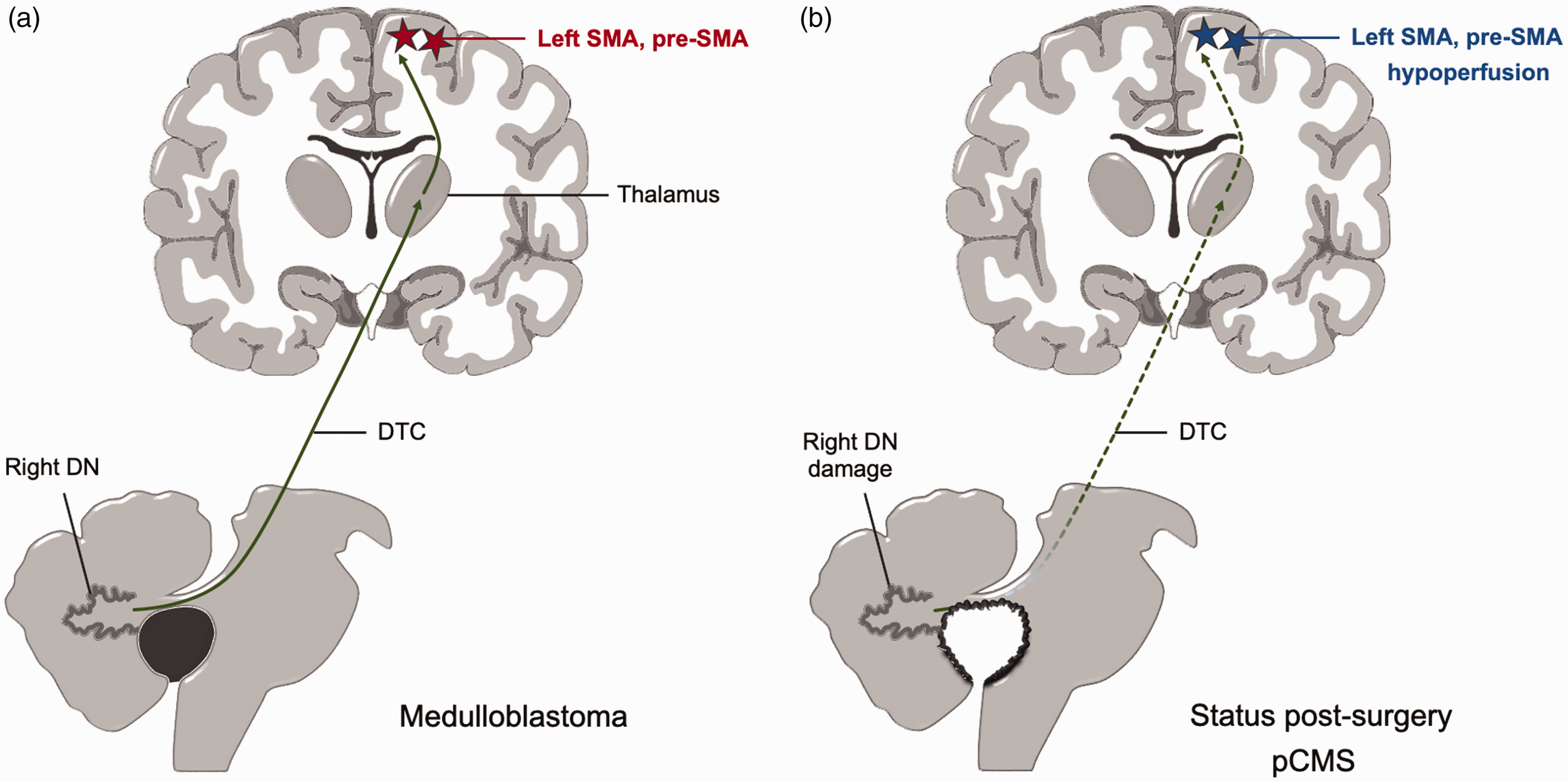
Schematic illustration summarizing the hypothesis for postoperative pediatric cerebellar mutism syndrome following medulloblastoma resection. Preoperative (a) and postoperative (b) figures illustrate the proximal damage to the ascending cerebellar fibers of the dentato-thalamo-cortical tract (DTC) following medulloblastoma resection. The neuronal injury would result in deafferentation and hypoperfusion of the supplementary motor and pre-supplementary motor areas in the superior frontal gyrus, as demonstrated with ASL-MRI perfusion in pCMS patients. Abbreviations: DN: dentate nucleus; DTC: dentato-thalamo-cortical; pCMS: postoperative pediatric cerebellar mutism syndrome; SMA: supplementary motor area; pre-SMA: pre-supplementary motor area.
Several limitations of this study need to be acknowledged, the first being its retrospective observational design. Although mutism is considered the core clinical symptom of pCMS, the diagnosis of pCMS can be controversial since no objective clinical tests are available. To address this issue, we limited our analysis to children who had developed postoperative mutism. It is important to mention that 8 patients younger than 5 years of age (3 in the pCMS group, 5 in the no-pCMS group) were administered intrarectal pentobarbital for MRI sedation as part of our institution’s routine standard protocol. Even though the effect of pentobarbital on brain perfusion has not been precisely established, it has been shown that regional CBF measured using ASL sequences is not influenced by the use of general anesthesia or sedation in a pediatric population. 34 In our study, these 8 children were sedated for both preoperative and postoperative MRI scans, which limits the potential intra-individual variability. Furthermore, since the assessment of T2-weighted signal abnormalities on postoperative MRIs can be challenging, as supported by other authors,9,32 discrepancies in interpretation were resolved through discussion and reaching consensus among the two main readers and four other pediatric neuroradiologists. Importantly, even though our results support that the damage to the right DN, the origin of the DTC tract, may be associated with left pre-SMA/SMA hypoperfusion, specific damage to the DTC was not measured since diffusion tensor imaging sequences were not available. A prospective study investigating DTC damage in pCMS, particularly based on tractography investigation, is currently performed in our lab to further clarify the link between right dentate nucleus and the dentato-thalamo-cortical-tract. Finally, methodological considerations related to CBF measures include the fact that it can be sensitive to variations in arterial transit time and also to changes in velocity of the inflowing blood in the labeling plane. For instance, since it could be argued that T1 of blood relaxation might be imprecise for pediatric population, its effects on M0 of blood equilibrium magnetization estimation, and therefore on CBF quantification, would not be completely addressed by optimizing the PLD alone, which could potentially partially affect our results. However, Alsop et al. support the use of single post labeling delay pseudo continuous ASL for clinical investigations. 35
In summary, this study provides evidence of an association between pCMS, right DN and left pre-SMA/SMA hypoperfusion. These findings support the hypothesized link between right proximal DTC tract injury and pCMS, whereby damage to the right DN, the origin of the dentato-thalamo-cortical pathway, results in left pre-SMA/SMA hypoperfusion, key areas involved in speech production. This suggest that a special care should be taken during surgery to prevent damage of the dentato-thalamo-cortical tract. Brain perfusion abnormalities detected by ASL imaging appear to be a robust biomarker for mutism in pCMS and could be used to monitor the effectiveness of potential treatment strategies. Finally, resting state fMRI studies could help to clarify the functional connectivity abnormalities in patients with this puzzling and intriguing syndrome.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211031321 - Supplemental material for A CBF decrease in the left supplementary motor areas: New insight into postoperative pediatric cerebellar mutism syndrome using arterial spin labeling perfusion MRI
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211031321 for A CBF decrease in the left supplementary motor areas: New insight into postoperative pediatric cerebellar mutism syndrome using arterial spin labeling perfusion MRI by Jennifer Boisgontier, Ludovic Fillon, Caroline Rutten, Ana Saitovitch, Christelle Dufour, Hervé Lemaître, Kévin Beccaria, Thomas Blauwblomme, Raphaël Levy, Volodia Dangouloff-Ros, David Grévent, Charles-Joris Roux, Jacques Grill, Alice Vinçon-Leite, Lila Saidoun, Franck Bourdeaut, Monica Zilbovicius, Nathalie Boddaert and Stéphanie Puget in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X211031321 - Supplemental material for A CBF decrease in the left supplementary motor areas: New insight into postoperative pediatric cerebellar mutism syndrome using arterial spin labeling perfusion MRI
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X211031321 for A CBF decrease in the left supplementary motor areas: New insight into postoperative pediatric cerebellar mutism syndrome using arterial spin labeling perfusion MRI by Jennifer Boisgontier, Ludovic Fillon, Caroline Rutten, Ana Saitovitch, Christelle Dufour, Hervé Lemaître, Kévin Beccaria, Thomas Blauwblomme, Raphaël Levy, Volodia Dangouloff-Ros, David Grévent, Charles-Joris Roux, Jacques Grill, Alice Vinçon-Leite, Lila Saidoun, Franck Bourdeaut, Monica Zilbovicius, Nathalie Boddaert and Stéphanie Puget in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Part of this work has been supported by Lisa Forever association (https://www.lisaforever.org/), and by Canceropole “Ile de France” (![]() ) and Institut National du Cancer (InCa).
) and Institut National du Cancer (InCa).
Acknowledgements
The authors would like to thank Dr Olivier Ayrault for his contribution to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contribution
Conception and design: JB, NB, SP, CD, AS. Collection and assembly of data: JB, NB, SP, CD, RL, VDR, DG, CJR, LS. Data analysis: JB, LF, HL, RL, VDR, DG, CJR. Results interpretation: JB, NB, SP, CD, CR, LF, AS, HL, JG, AVL, KB, TB, FB, MZ. Manuscript writing: JB, CR, NB, SP. Critical and intellectual revision of manuscript: NB, SP, CR, CD, LF, AS, AVL, TB, JG, MZ. Final approval of manuscript: All authors. Accountable for all aspect of the study: All authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
