Abstract
Targeted temperature management (TTM) is a recommended neuroprotective intervention for coma after out-of-hospital cardiac arrest (OHCA). However, controversies exist concerning the proper implementation and overall efficacy of post-CA TTM, particularly related to optimal timing and depth of TTM and cooling methods. A review of the literature finds that optimizing and individualizing TTM remains an open question requiring further clinical investigation. This paper will summarize the preclinical and clinical trial data to-date, current recommendations, and future directions of this therapy, including new cooling methods under investigation. For now, early induction, maintenance for at least 24 hours, and slow rewarming utilizing endovascular methods may be preferred. Moreover, timely and accurate neuro-prognostication is valuable for guiding ethical and cost-effective management of post-CA coma. Current evidence for early neuro-prognostication after TTM suggests that a combination of initial prediction models, biomarkers, neuroimaging, and electrophysiological methods is the optimal strategy in predicting neurological functional outcomes.
Introduction
The morbidity and mortality surrounding out-of-hospital cardiac arrest (OHCA) remain a global challenge. In the United States, more than 350,000 people experience an OHCA every year, and about 10.8% of them survive to hospital discharge. 1 Although timely and advanced emergency aid helps to improve the probability of return of spontaneous circulation (ROSC) and subsequent survival, only about 9% of OHCA patients achieve good neurological function at discharge. 1 Notably, survivors with good neurological function can still suffer from cognitive impairment 2 and encounter problems while returning to work. 3
Currently, targeted temperature management (TTM), previously called therapeutic hypothermia, is the sole intervention for improving neurological outcomes in the post-ROSC phase of care. 4 TTM has been considered a standard of care treatment for over almost two decades and has been included in the International Post-Cardiac Arrest Guidelines since 2005. 5 With a growing literature base, confusion regarding the optimal application of TTM has also grown (briefly summarized in Figure 1). Hundreds of new clinical and preclinical publications have focused on TTM after cardiac arrest (CA) in recent years, yet extensive and updated reviews of both pre-clinical and clinical evidence are limited, as is the level of evidence in current recommendation guidelines. In particular, the 2013 TTM study sparked the heated debate of optimized target temperature. 6 Given the global health burden of this disease process, it is vital that we critically evaluate the current level of evidence and recommendations and identify gaps in knowledge that require further investigation. This review investigates the literature on crucial aspects in the application of TTM, presenting the evidence and defining recommendations on ideal timing, the target depth, and available methods of cooling. We dissect areas of ongoing controversy and the up-to-date supporting evidence for current guidelines, as well as areas warranting critical attention for further investigation and advancement in providing high-quality, patient-centered care.
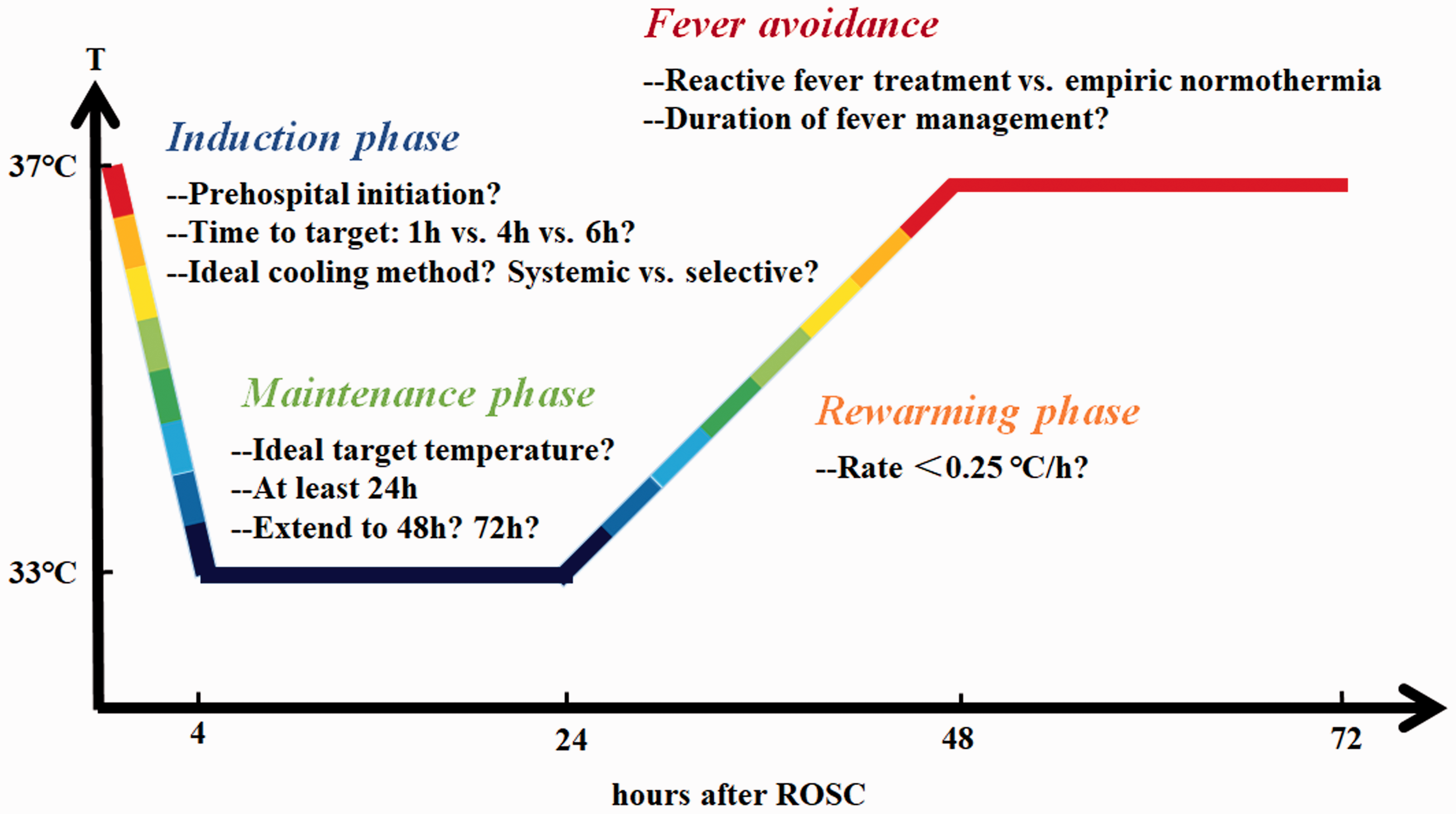
Unanswered questions concerning the TTM of OHCA patients.
In addition to providing optimal therapy including TTM, critical care providers are challenged with providing families with prognostic information used in goals of care decision making. However, implementation of TTM requires the use of sedative, and potentially paralytic, medications. These adjunct therapies can confound neurological exam and electrophysiological testing, which has complicated the early prognostic information acquired. 7 To our knowledge, few reviews have focused implicitly on the application of prognostic tools in the early phases of post-CA care with TTM. We present a review of the validity of currently available neuro-prognostic modalities primarily within the first 72 hours after admission and potential future directions for this field.
Materials and methods
We searched MEDLINE for evidence on the use of TTM for neuroprotection in OHCA and in animal models from January 2017 to March 2020. Specifically, search details included ‘hypothermia OR targeted temperature management AND cardiac arrest’. 1199 abstracts were retrieved and screened. 115 met the scope of this review and were included. In addition, 25 articles published prior to 2017 were included because of their influence on this field. The studies included should be not only relevant to the current theme, but also scientifically vigorous. The included clinical studies were required to be randomized clinical trials (RCTs) and prospective corhort studies, or retrospective studies with large sample sizes and matched controls. Cerebral Performance Category (CPC) score of 1–2 is considered a good neurological outcome unless otherwise specified.
The depth of TTM
Clinical evidence
The classical target range of 32–34°C has been based on preclinical data derived from young healthy animals. 8 Initial randomized clinical trials showed improved neurological outcomes (ability to be discharged home or to a rehab facility 9 or CPC 1–2 10 ) by cooling the body temperature to 32–34°C for 12–24 hours post-ROSC, and this target temperature range remained the standard for decades. However, new data has challenged this standard and raised questions regarding the ideal target temperature.
A recent meta-analysis pooled data from RCTs and suggested that moderate TTM (32–34°C) does not improve survival or neurological outcome when compared to mild TTM (>34°C). In contrast, observational data has favored a lower target temperature. 11 The landmark TTM trial reported that moderate TTM (33°C) did not confer a benefit over mild TTM (36°C) after CA, though equivalence between the two interventions was not concluded. 6 Additionally, there has been criticism surrounding the study quality, including the prolonged time to target temperature (average 10 hours), inadequate temperature maintenance during the maintenance phase, and rapid rewarming (about 0.5°C/h). 12 A brief summary of the early, landmark studies in TTM is listed in Table 1. 6 ,9,10,13
Early and landmark studies investigating targeted temperature management after cardiac ARREST.
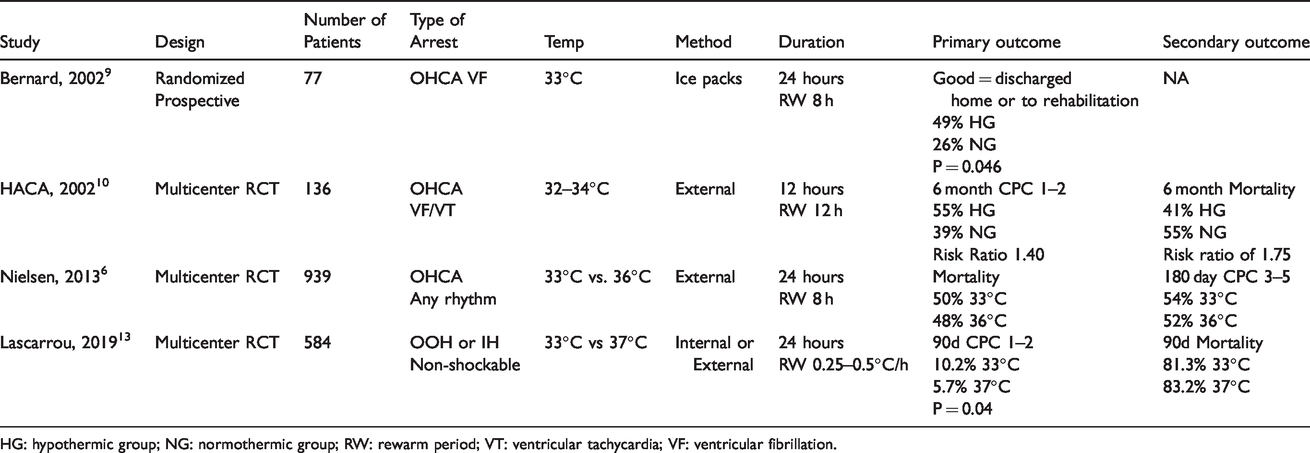
HG: hypothermic group; NG: normothermic group; RW: rewarm period; VT: ventricular tachycardia; VF: ventricular fibrillation.
One of the leading centers in the ongoing TTM-2 trial (NCT02908308) observed a significant reduction in time at the target temperature and an increase in fever rates using a target of 36°C, which might result in a trend toward worse outcome. 14 A retrospective before-and-after study including 453 patients treated with TTM showed that 33°C improved discharge with good neurological outcome compared with 36°C, and hospital mortality was similar. 15 In the post-TTM trial era, soaring frequency of fever and non-significantly increased mortality were also observed in Australian and New Zealand ICUs after the introduction of the 36°C target. 16 In the post-hoc analysis of the TTM trial, there was no difference in documented bleeding 17 complications when comparing mild to moderate hypothermia. In addition to similar safety profiles, moderate TTM may protect tissues from ischemia/reperfusion injury. Compared with mild TTM, moderate TTM reduced blood markers of oxidative damage, increased the level of antioxidants, such as glutathione, 18 and reduced circulating mitochondrial DNA. 19 In addition, a recent high-quality RCT showed that for OHCA and in-hospital CA patients with non-shockable rhythm, TTM (33°C) resulted in better 3-month neurological outcomes when compared to normothermia (37°C), 13 and retrospective reports have supported the notion that the outcome of moderate TTM might be better.14,16 As for lower targets, the ongoing CAPITALCHILL (Mild Versus Moderate Therapeutic Hypothermia in Out-of-hospital Cardiac Arrest Patients) trial (NCT02011568) is now comparing TTM at 31°C and 34°C.
Importantly, we shall bear in mind that in addition to methodological variations, CA itself has diverse etiologies and comorbidities, hence pooled data of this heterogeneous patient population could be misleading. Ultimately, recent meta-analyses report that both adult and pediatric populations lack conclusive evidence to support either a moderate or mild temperature target.20,21 Prospective, as well as retrospective, studies revealed no significant difference concerning the targets of 34°C and 36°C.22,23 A recent international database analysis including 1710 OHCA patients who received TTM showed comparable functional outcome at discharge between 32–34°C and 35–37°C. 24 An RCT also found no difference of 32, 33, and 34°C for witnessed OHCA patients with initial shockable rhythm. 25 Results of recent studies comparing different depths of TTM are briefly summarized in Table 2.6,13–16,22–27
Brief summary of recent studies comparing different depths of TTM.
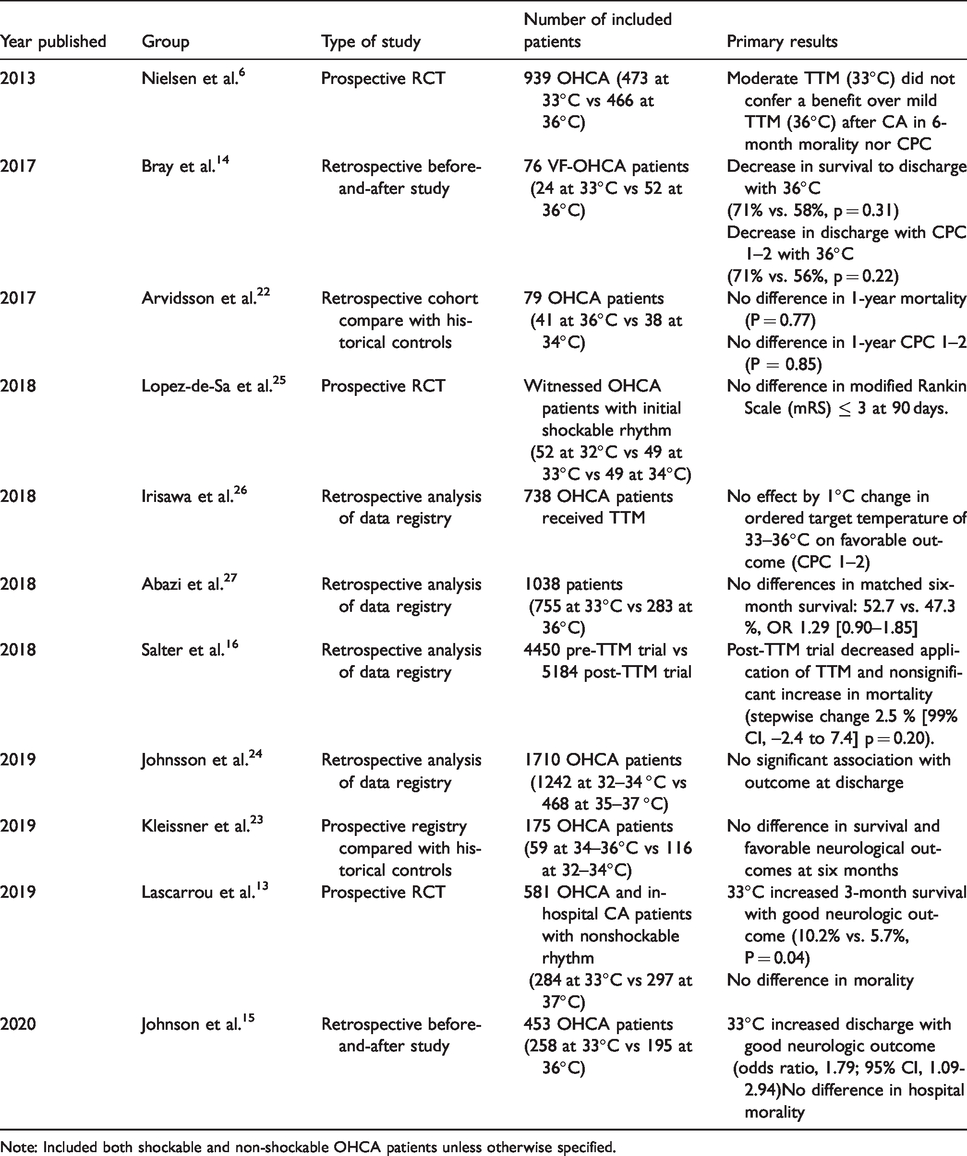
Note: Included both shockable and non-shockable OHCA patients unless otherwise specified.
The latest guidelines, including the 2017 Neurocritical Care Society guidelines 28 and 2017 French guidelines, 29 do not specify or recommend an ideal target temperature, given current debate. Rather, target temperature between 32°C and 36°C is left to the discretion of the treating physician, reinforcing the fact that a tailored and ideal temperature target for each individual patient, taking into consideration age, sex, comorbidities, and genetic backgrounds, is poorly understood.
The duration of TTM
Clinical evidence
Current recommendations suggest TTM for at least 24 hours after ROSC, though this recommendation is conditional and comes with only moderate quality of evidence according to the Neurocritical Care Society. 28 TTM of 33°C for 24 hours has been shown to improve the recovery of post-CA left myocardial dysfunction. 30 These studies could be used to infer that TTM of at least 24 hours duration could improve the perfusion of vital organs, including the brain.
Improved outcome with prolonged cooling, beyond 24 hours, has been suggested. However, these reports are confounded by the earlier implementation of TTM in addition to prolonged duration.31,32 A recent RCT, the Time differentiated Therapeutic Hypothermia (TTH48) trial, reported a trend toward better outcomes accomplished by 48 hours of TTM targeting 33°C. Six-month mortality was reduced from 34% to 27% with prolonged TTM, and 69% in the 48-hour group (vs. 64% in the 24 h group) achieved a favorable outcome at 6 months. 33 Further, a sub-study of the TTH48 trial revealed that the 24-hour group was almost three times more likely (p = 0.02) to have cognitive impairment compared with the 48-hour group. 34 Currently, a multicenter randomized trial titled Influence of Cooling Duration on Efficacy in Cardiac Arrest Patients (ICECAP, NCT04217551) is investigating 90-day functional outcomes after varying durations of TTM at 33°C, ranging from 6 hours of TTM post-ROSC to 72 hours of TTM post-ROSC.
Data from a multi-center prospective cohort study including 2,579 patients found that a larger hypothermia/ischemia ratio (calculated as total TTM duration [in hours]/ischemia duration [in minutes]) was associated with favorable functional outcome after OHCA (OR 2.01). 35 However, prolonged TTM is not without risks. Although platelet aggregation between standard (24 hours) and prolonged (48 hours) TTM was not substantially different, 36 prolonged TTM at 33°C impaired thrombin generation, which may cause additional bleeding complications. 37 The TTH48 investigators 38 speculated that a subgroup of patients with younger age (under 60 years) and/or a longer “no-flow” period might benefit from prolonged TTM, though the latter was not supported by a post-hoc analysis of the trial. 39 Further clinical investigation and evidence are required to elucidate the optimized TTM duration. Figure 1 summarizes the controversies surrounding when and how to apply TTM.
The timing of TTM
Preclinical evidence
Preclinical data supports the belief that rapid cooling should be started as soon as possible for CA patients in order to protect the brain. Jia lab demonstrated that in rats, hypothermia (33°C) started immediately after asphyxial CA attained better neurologic outcome [higher serial Neurological Deficit Score (NDS)] compared with delaying hypothermia for 1 hour. 40 The Neumar group discovered that longer delays (1/4/8 hours) of hypothermia (33°C) resulted in higher mortality and worse neurological function scores in a rat asphyxial-CA model. 41 In a mouse CA model induced by hyperkalemia, TTM (brain temperature 28–32°C) initiation before resuscitation improved survival, reduced hippocampal neuronal injury, and ameliorated spatial memory impairment and anxiety-like behaviors. 42 TTM (33°C) initiated during cardiopulmonary resuscitation (CPR) in a ventricular fibrillation rat CA model achieved better preservation of cerebral microcirculation and reduced apoptosis. 43 However, it should be noted that most of the earlier rodent data are acquired using male animals that are relatively young and healthy, which may limit translation. A new extracorporeal perfusion system was tested in a female swine model of ventricular fibrillation induced CA, and 30 minutes of moderate TTM (32°C) started with extracorporeal resuscitation achieved a trend toward better neurological recovery (nonsignificant secondary to the sample size) measured by pig-specific NDS. 44
Clinical evidence
The translation of these preclinical findings has remained inconsistent. Early initiation of in-hospital TTM (within 2 hours after admission) has been shown to be generally safe and associated with improved survival, but not with better neurological outcome. 45 As for pre-hospital cooling, a wide gap exists between preclinical research and clinical practice. Previous studies, for example the 2016 RINSE (Rapid Infusion of Cold Normal Saline) trial, 46 and a 2017 RCT, 47 applied endovascular cooling such as infusion of cold saline during CPR or immediately after ROSC. These studies did not show improved survival nor improved neurological outcomes with pre-hospital cooling. A recent meta-analysis 48 showed that pre-hospital TTM after OHCA effectively decreases body temperature during patient transfer, but it does not improve overall survival or survival with good neurological outcome, regardless of whether the TTM was started intra-arrest or post-arrest, or rhythms were shockable or non-shockable. In addition, this pre-hospital TTM may increase rates of early re-arrest and pulmonary edema. 47 The results of a two-phase, prospective, observational study conducted in New York also showed that intra-arrest application of cold saline had no survival benefit. 49 The Pre-Hospital Resuscitation Intra-Nasal Cooling Effectiveness Survival Study (PRINCESS) trial 50 focused on pre-hospital trans-nasal evaporative intra-arrest (i.e. during CPR) cooling in an effort to reduce the side effects of systemic cooling. Compared with usual care, pre-hospital trans-nasal evaporative intra-arrest cooling did not lead to a significant improvement in survival with good neurologic outcome. Notably, subgroup analyses showed that pre-hospital cooling had a tendency towards higher rates of good neurologic outcome at 90 days for patients with a shockable rhythm (34.8% vs. 25.9%, p = 0.11). At present, pre-hospital cooling is not standard practice. However, it seems that pre-hospital trans-nasal cooling may have a role in OHCA patients with ventricular tachycardia or fibrillation arrest, which warrants further investigation. In addition, no study has identified an ideal time to target temperature following ROSC. While most studies agree that rapid cooling to the target temperature as quickly as possible reduces reperfusion injury, there is a lack of literature evidence identifying an exact time-point at which target temperature should be reached post-ROSC or time point after which TTM may not have net benefit.
The cooling methods of TTM
Systemic cooling
Preclinical evidence
Preclinical advances in systemic cooling continue to add new potential avenues for TTM. Xu et al. applied continuous renal replacement therapy in a porcine model, achieving rapid hypothermia within 30 minutes, reducing both brain injury and systemic inflammation. 51 New systemic cooling devices are also currently under investigation. The effect of an Esophageal Cooling Device was similar to traditional methods of endovascular cooling to a target of 33°C with no additional adverse effects, and these findings are further confirmed in swine models.52,53 Another preclinical technique using breathable perfluorocarbon liquid, termed “total liquid ventilation”, accomplished rapid cooling of the brain in less than 3 minutes (−72°C/h) in a sheep model. 54 However, to our knowledge, these new techniques have not been verified in any large prospective clinical studies. Hence, whether they could effectively and safely improve post-CA outcomes requires further investigation.
Clinical evidence
Systemic cooling, primarily achieved through external (surface) cooling or endovascular cooling, is the traditional choice with relatively abundant clinical evidence. A recent meta-analysis focusing on the clinical application of advanced surface cooling using hydrogel found no significant difference compared to endovascular cooling in terms of survival and neurological outcome. 55 However, other detailed analyses seemed to favor endovascular cooling. A retrospective cohort of 132 patients showed that endovascular cooling with cold saline was associated with faster time to target temperature and better temperature maintenance. 56 Despite a higher incidence of pulmonary edema and diuretic utilization with endovascular cooling, there was no significant difference in cardiac re-arrest, arterial oxygenation, and CPC scores. 56 An additional retrospective study of 34 patients showed that computer-assisted endovascular cooling achieved a faster time to target temperature and better thermic stability when compared to traditional infusion of cold saline. 57 From a nursing perspective, endovascular cooling had the best rating with regard to ease of temperature monitoring and least additional workload. 58 In terms of adverse effects, endovascular cooling may be a risk factor for venous catheter-related thrombosis. 59 However, a post-hoc analysis of the TTM trial 60 showed a tendency towards a better neurologic outcome, and a recent meta-analysis revealed that endovascular cooling was associated with improved neurological outcomes at discharge. 61 At present, endovascular cooling may be the preferred method of systemic cooling endorsed by the NCS guidelines. 28 Translation of esophageal cooling was explored in a 2017 published study by applying a new semi-invasive “Esophageal Cooling Device”, which revealed feasibility, safety, and accuracy of this device in a chort of 17 OHCA patients 62 and opened a new potential mechanism for systemic cooling.
Selective brain cooling
On average, brain temperature can be 0.34°C higher than core temperature and lags behind core temperature by about half an hour. 63 TTM using selective brain cooling (e.g. 33°C for the brain and 36°C for the body) might have better outcomes by reducing the complications of prolonged systemic cooling. 64
Preclinical evidence
A high-flow (250 L/min) cold air (−10°C) cooling device achieved effective cooling of the anterior portion of the brain in a porcine model. 65 However, by MRI estimation, the temperature of the posterior brain was largely unaffected, and the rapid cooling (an average of −0.33°C during 5 minutes) caused damage to the nasal mucosa. 65 Conversely, a swine model did demonstrate the safety and efficacy of a new nasopharyngeal cooling catheter, with no reported device-related adverse effects. 66 Further research is required to confirm the advantage of cooling the brain (as well as the body) through the upper airway over other cooling methods.
Of note, with the help of the placement of intraventricular catheters, intraventricular cooling based on cerebrospinal fluid effectively reduced temperature in all identified brain regions a sheep model, while systemic normothermia was well maintained. 67 For post-CA patients with the need for advanced invasive brain monitoring, intraventricular catheters might be a potential avenue for TTM.
Clinical evidence
In humans, non-invasive methods of selective brain cooling showed that a specialized neck collar can cool the carotid arteries and is easy to apply. However, a computer model showed that neck collars may not be effective unless under a low blood flow rate condition. 68 The upper airway, particularly the nasal and pharyngeal cavities, contains anatomic structures proximal to the brain, which make them attractive sites for heat exchange. The PRINCESS trial 50 further investigated trans-nasal evaporative cooling and demonstrated that the cooling rate was relatively slow and may not be effective for cooling the whole brain.
Pharmaceutical-induced hypothermia
Preclinical evidence
Pharmaceutical-induced hypothermia is another appealing option for TTM.69,70 A neurotensin receptor agonist, ABS 201, both induced and maintained TTM in a ventricular fibrillation induced CA rat model, improving neurologic outcome and survival duration. 71 Dihydrocapsaicin, an agonist of transient receptor potential vanilloid type 1 cation channel (TRPV1), also achieved similar results in an asphyxial-CA rat model. 69 However, drugs like dihydrocapsaicin are not without risks, including cardiovascular side effects. 72 To date, post-CA pharmaceutical-induced hypothermia is mainly studied in rodent preclinical models. Pending further discovery about the pharmacological characteristics and possible synergistic effects with physical cooling, studies may move from rodents to larger animals, with an eventual goal of human implementation.
Confounding factors influencing outcome after CA and TTM
Age
Advanced age is a known factor for worse neurologic outcome in OHCA patients treated with TTM. 73 In a registry-based retrospective study including 1357 OHCA patients (45.5%) received TTM, for patients aged ≥75 years, TTM seemed to be associated with increased morality (OR = 1.22). 74 Another similar study included 1885 OHCA patients, 921 were treated with TTM (32–34°C), and determined that TTM was not associated with good outcome in patients over the age of 65 (OR < 1.5). 75 Conversely, a Korean registry-based retrospective analysis including 161 matched pairs of OHCA patients found that endovascular cooling was associated with good neurological outcomes for patients 65 or older. 76
Sex
Limited preclinical studies have focused on the possible influcnce of sex in outcome after CA and TTM. A juvenile mouse CA/CPR study suggested that a lower temperature (30°C) is required for male animals to achieve similar neuroprotective effects compared with female counterparts (32°C), 77 suggesting that sex-specific personalization of TTM may be needed.
In humans, studies have indicated that men are more likely to receive TTM than women, even when controlled for initial rhythm.74,78 A recent single-center retrospective study including both OHCA and in-hospital CA patients showed that 31% of the 167 paitents included underwent TTM, and females were equally likely to receive TTM as males. 79 However, females were more likely to have unfavorable cognitive outcomes. 79
Estrogen has been shown to have neuroprotective effects after cerebral ischemia, 80 whereas testosterone has shown to exacerbate neuronal injury. 81 These findings contrast currently reported prognostic information for females vs. males, and it has been suggested that TTM may counter this beneficial estrogen effect. However, in a Korean registry-based retrospective multicenter study, sex did not alter the neurologic outcomes of 930 Korean OHCA patients who underwent TTM. 73 Although the literature base shows varying results in terms of sex differences and the effect of TTM and prognosis post-CA, a sex-targeted approach in the management of post-CA patients is worthy of future investigations.
Early neuro-prognostication under TTM
Neuro-prognostication after successful resuscitation is a fundamental, yet challenging, component of post-CA management. Clinician prediction of prognosis has been shown to greatly affect family decisions regarding withdrawal of life-sustaining therapies (WLST). 82
Post-CA brain injury remains the most significant independent predictor of 6-month mortality in OHCA patients. 83 Approximately 10% of these deaths meet the clinical criteria for brain death, and a majority of deaths result from WLST in light of poor predicted outcome. 84 A recent report showed that early WLST (within 3 days after resuscitation) is prevalent in western countries, which raises concerns regarding premature WLST in patients who may have the potential for a good outcome. 85 In this study, 19% of patients in the early WLST group were predicted to have a good neurologic outcome, however, they all died. 85 Further, in a matched cohort for the early WLST group, 21% of patients had a good neurologic outcome. 85 This pattern raises concern for a self-fulfilling prophecy, as clinicians are often not blinded to the results of neuro-prognostic tests. On the other hand, pursuing futile and expensive critical care in patients destined for death and/or severe neurologic disability is also undesirable. The 2015 European Resuscitation Council (ERC)/European Society of Intensive Care Medicine (ESICM) guidelines 86 recommend waiting for at least 72 hours post-TTM, after a patient has returned to normothermia, to provide neuro-prognostic information, in an effort to reduce possible confounders such as sedatives required for TTM. However, given the desire to dedicate medical resources appropriately and provide families with valuable information as soon as possible after acute injury, efforts continue to identify a prognostic tool with high sensitivity and specificity that can be used early in the post-CA course to identify which patients will have a good or poor outcome. We summarize recent literature concerning early neuro-prognostic methods under TTM, conducted 48 hours or less after admission unless otherwise specified.
Pupillary light reflex
Early quantitative assessment of pupillary light reflex has growing supportive data. Pupillary light reflex measured both at 0 and 6 hours after ROSC showed promising predictive value in a prospective multicenter study. 87 In another prospective multicenter study, quantitative pupillometry [using the neurological pupil index (NPi)] performed better than standard manual method. 88 Briefly, the NPi is based on the reflex amplitude, the velocity of constriction (CV) and dilation (DV), and latency; the algorithm appears to be independent of the size of the pupil and turns out a continuous variable from 0 (nonreactive) to 5 (normal). An NPi ≤ 2 achieved a 100% specificity for the prediction of poor outcome at any time from day 1 to 3. 88 Likewise, sluggish or nonreactive pupils (NPi < 3) at the start of TTM or during TTM were associated with poor outcome; while the outcome was not correlated with dilated pupils. 89 A more recently published prospective multicenter study showed that maximal constriction velocity, dilation velocity, and NPi were statistically greater in patients with favorable neurologic outcomes compared to those with poor outcome, and the poor outcome predictive area under curve (AUC) improved to 0.95 when admission pupillary light reflex, constriction velocity, dilation velocity, or NPi was combined with clinical factors such as witness status, bystander CPR, initial shockable rhythm, and implementation of targeted temperature management. In this study, the use of quantitative values such as maximal constriction velocity and NPi at admission displayed equivalent predictive value compared to pupillary light reflex alone or in combination with clinical factors at the time of admission. 90
Electrophysiological outcome predictors
Electroencephalogram (EEG)
The utility of continuous EEG (cEEG) post-CA has been an ongoing discussion in the literature, particularly since the introduction of TTM to the standard of care. 91 Up to 30% of comatose post-CA patients will have electrographic seizures, but this is not consistently congruent with poor neurological outcome. 92 Though many recent reviews and studies question the utility of EEG in conjunction with TTM due to confounding variables, the evidence does exist for the early use of this modality in predicting neurological outcome. Specifically evaluating the use of EEG early in the post-CA phase, a retrospective study with most patients undergoing TTM showed that routine EEG with “highly malignant” features, defined by the American Clinical Neurophysiology Society as suppressed background without discharges, suppressed background with continuous periodic discharges, and burst-suppression background with or without discharges, 93 at day 1 could predict poor prognosis with better accuracy (85.7%) compared with other prognostic predictors. 94 Another multicenter prospective study that included 850 patients discovered a 100% specificity for poor outcome if cEEG, measured 12 or 24 hours after CA, showed generalized suppression (all activity <10 µV) or synchronous patterns with ≥50% suppression, though sensitivity was less than 50%. 95 A prospective trial using cEEG found that malignant EEG patterns at 24 hours predicted a poor outcome, not including WLST, with a specificity of 100% and sensitivity of 29%. 96 This same study found that favorable EEG patterns, such as continuous patterns with reactivity, predicted good outcome at 6 months with a specificity of 88% and sensitivity of 51%. A multicenter prospective investigation revealed that an early (within 24 h) epileptiform EEG was associated with poor outcome (specificity 89%), while an early recovery of background achieved a predictive value of 76% for good outcome. 97 Burst suppression pattern, previously believed to represent a malignant pattern, has been shown to have limited early predictive value in a recent retrospective report. 98 A recent single-center prospective study demonstrated that a highly malignant EEG pattern on day 2 was fairly specific (99.5%) for poor prognosis. 99
To avoid the time-consuming and expertise-demanding interpretation of EEG, quantitative EEG is a promising, yet premature, alternative. Amplitude-integrated electroencephalography (aEEG), a type of qEEG that can be easily interpreted by a non-neurologist, was shown to successfully categorize post-CA comatose survivors into different categories based on aEEG findings post-ROSC. If recovery of continuous normal voltage was not detected within the first 36 hours or if burst suppression was detected, the patient would have a poor neurological outcome at hospital discharge. 100 Computer-assisted interpretation of early EEG (9–30 hours after CA) during TTM using a convolutional neural network achieved an AUC of 0.885 in discriminating good and poor neurological outcome. 101
Nonetheless, confounding variables such as the effect of hypothermia on EEG or sedatives used during TTM must be considered, 102 although recent studies suggested that sedation required for TTM did not alter the predictive value of standardized EEG interpretation, 93 and EEG patterns were not significantly changed in propofol-sedated patients undergoing TTM (both 33°C and 36°C targets). 103 Computer-assisted interpretation of EEG is a promising option and warrants further investigation.
Bispectral index (BIS)
BIS is another quantitative measurement of EEG with a relatively large body of evidence. BIS value obtained at a mean of 2.3 hours after ROSC was predictive of outcome, and a cut-off value of 10.5 achieved a specificity of 100%. 104 A mean BIS of 25 or less at hour 12 [AUC: 0.722 (0.570–0.875); p = 0.006] and a mean suppression ratio ≥ 3 at hour 23 [AUC: 0.836 (0.717–0.955); p < 0.001] might be applied to predict poor neurologic outcome in OHCA patients with a presumed cardiac cause. 105 A total duration of 5 hours with a BIS value of 0 over the first 48 hours could predict a poor neurologic outcome with a sensitivity of 63% and specificity of 100%. 106 Similarly, epileptic activity via BIS monitoring within the first 6–12 hours predicted poor outcome with a positive predictive value (PPV) of 100%. Epileptic activity between 18–24 hours and 36–48 hours predicted negative outcomes with PPV 90% and 93% respectively. 107 The use of neuromuscular blockade to improve the quality of BIS data is under debate, and results should be interpreted with caution.
Evoked potentials
EEG reactivity and/or electromyography reactivity to repetitive stimuli, such as auditory and somatosensory evoked potentials, during TTM revealed a sensitivity of 82% for prediction of good outcome. 108 The use of somatosensory evoked potentials (SSEPs), which are usually obtained by stimulation of the median nerves, has a growing body of evidence. The classic bilateral absence of cortical SSEP responses (the N20 potentials) usually suggests the destruction of the integrity of thalamocortical projections and has been regarded as a reliable predictor, at day 1–3 post-CA, of poor neurological outcome, with or without TTM.109–111 Meanwhile, the absence of N20–P25 amplitude, defined as the largest difference in amplitude between the N20/P25 peaks bilaterally, measured as soon as completion of rewarming, had a specificity of 100% (93.0–100.0%) and sensitivity of 71.6% in predicting poor outcome. This outperformed the classic absence of N20 potentials. 112 However, a recent systematic review discovered that the bilateral absence of N20 potentials might have a false positive rate of about 7.7% in predicting poor neurologic outcome after taking into account WLST. 113
Biomarkers
Biomarkers obtained from peripheral blood are overall non-invasive and easy to obtain. A prospective cohort study found that higher S100B (a serum marker for glial and blood-brain barrier damage) at 24 h was associated with poor neurologic outcome at 3 months, and higher procalcitonin (PCT) at 24 h was associated with mortality at 14 days. When combined, S100B and PCT at 24 h had sensitivity 77.6% and specificity 100% for poor neurologic outcome at 3 months. 114 However, S100B measured at 24 h alone was not an independent predictor of neurologic outcome in a post-hoc analysis of the TTM trial. 115 Elevated neurofilament light chain (NFL), which reflects axonal damage, at 24 hours after CA has been shown to have 69% sensitivity and 98% specificity for poor neurologic outcome at 6 months. 116 Although neuron-specific enolase (NSE) can be confounded by factors such as hemolysis, it remains the most supported biomarker for neural damage in the literature, and has been shown to remain reliable under TTM. 109 Patients undergoing TTM targeting 33°C had poor outcomes if the NSE at 72 h post-CA was > 90 ng/mL. 117 A recent study targeting the same temperature also showed that elevated NSE at 48 h post-CA had the best AUC of 0.871 for predicting poor outcome compared with other time points. 118 However, we shall bear in mind that NSE values vary among different labs, and a generalizable cut-off point is difficult to identify. 119 A recent study pointed out that the permeability of the blood-brain barrier may not be enough at 24 h, and serum NSE tested at 48 h is more accurate for early neuro-prognostication. 120 Another recent report revealed that the elevation of NSE between hospital admission and 72 hours after ROSC was not only associated with poor neurological outcome, with an AUC of 0.90 (95%CI, 0.85–0.95), but was also an independent predictor of poor outcome in a multivariate analysis (P < .001). 121 Present data suggest that NFL is a promising neural marker and warrants further study and that NSE is the most generally supported biomarker in predicting outcome to date. Clinicians shall take other prognostic assessments into account when interpreting biomarker results, which require further standardization and validation in large prospective cohorts.
Neuroimaging
Brain CT is the first imaging choice to rapidly exclude major intracranial lesions such as hemorrhage. The use of CT has been investigated in early outcome prediction after cardiac arrest, and although cut-off values vary among studies, decline of the gray-white ratio (GWR) remains a classic indicator of cerebral edema due to ischemic insult, particularly in the basal ganglia and thalamus. 122 On the other hand, a scoring system based on parameters such as gray-white matter effacement prior to TTM showed a significant relationship with the discharge destination, mortality and neurologic outcome. 123 Of note, the interpretation of early post-CA CT scans by emergency physicians and radiologists showed acceptable inter-rater reliability (Kappa statistics: 0.66). 124 In contrast, another study showed that interobserver variability when identifying ischemic brain injury was moderate to high (kappa statistics: 0.0–0.48) on head CT scans obtained from adults within two hours of resuscitation from out-of-hospital cardiac arrest. 125 In addition, using specialized scoring systems, extensive anoxic brain injury revealed by early CT (performed before the initiation of TTM) was associated with unfavorable outcome.123,126 Despite the lack of uniformly recognized quantitative interpretation methodology, brain CT performed at least 24 hours after ROSC might be better if an evaluation of cerebral edema is required for early neuro-prognostication.
A recent retrospective report revealed that surogate parameters, such as the proportion of cerebrospinal fluid volume, seemed to be better than GWR when CT was performed within 2 hours after ROSC. 127 Combining the 3 parameters of cortical necrosis, cytotoxic edema, and cerebral edema derived from ultra-early (within 6 hours after ROSC) diffusion-weighted/apparent diffusion coefficient imaging achieved an impressive AUC of 0.956 in differentiating poor neurologic outcome with 100% specificity using MRI. 128 However, the performance of ventricular characteristics and early MRI still need validation in multicenter prospective studies, and the availability of MRI in the acute phase after ROSC may limit this imaging method in early prediction of outcome.
Perfusion examinations, such as computed tomographic perfusion, can semi-quantify cerebral perfusion and have achieved promising results. A pilot study with 10 comatose patients found that perfusion data acquired after TTM is feasible, with AUC of 0.83 for predicting immediate clinical assessment, but AUC of only 0.63 for predicting neurological outcome at discharge. 129 Another powerful, yet less widespread, tool is positron emission tomography, and its application in outcome prediction of CA has been reviewed recently. 130
Measurements of intracranial pressure (ICP)
The value of ICP monitoring and aggressive treatment of increased ICP secondary to global cerebral edema post-CA remains unclear. A recent pilot study including 10 patients revealed that invasive intraparenchymal ICP monitoring is generally safe in post-CA patients, but neither the ICP nor brain compliance could differentiate neurologic outcomes. 131 In addition to a limited sample size, the study may be influenced by inherent bias because the patients had either a 6-month GOS of 1 or 4/5. On the contrary, a recent retrospective analysis including 92 patients revealed that higher ICP, estimated by lumbar puncture, was associated with worse 6-month neurological outcome. 132 Measurement of ICP by lumbar puncture remains controversial given concerns for a cerebral herniation in these patients and is not currently considered standard practice. 132 Higher optic nerve sheath diameter (ONSD) within 2 days after CA was predictive of poor neurologic outcome, and an ONSD of 5.75 mm or more had a specificity of 100% for mortality. 133 ONSD findings did not differ significantly with TTM, and an ONSD of 4.90 mm or more measured 24 hours after ROSC achieved an AUC of 0.91 in predicting poor neurologic outcome, and 24 h was a better time point compared with 48 and 72 hours after ROSC. 134
Multimodal methods
Given the lack of a single perfect test for neuro-prognostication after CA, current recommendations encompass a multimodal approach, utilizing a combination of clinical exam, neuroimaging, biomarkers, and electrophysiologic tests to aid the clinician in providing prognostic information. 109
By combining BIS values with clinical variables at hour 24 post-CA, a prediction model predicted favorable neurological outcome with a sensitivity of 75% and specificity of 82%. 135
The combination of unfavorable findings on SSEPs (bilaterally absent or unilaterally combined with pathologic on the contralateral side) and GWR of the basal ganglia < 1.07 on brain CT within 24 hours after CA achieved a specificity of 94.9% for poor outcome. 136 Prospectively collected data demonstrated that bilaterally absent/absent-pathological amplitude SSEPs, a GWR < 1.21 (calculated using multiple sites of the basal ganglia) and isoelectric/burst-suppression EEG within 24 hours predicted poor neurologic outcome with 100% specificity. 137 In addition, a recent report showed that in the setting of different TTM targets (33°C or 36°C), the predictive value of two or more early predictors including the neurological examination (absent pupillary and/or corneal reflex, early myoclonus), electrophysiological tests (unreactive/epileptiform EEG, bilaterally absent SSEPs), and NSE > 75 µg/L was comparable between temperature targets (PPV 1.00). 138 Figure 2 illustrates early validated timing of neuroprognostic tools post-CA.
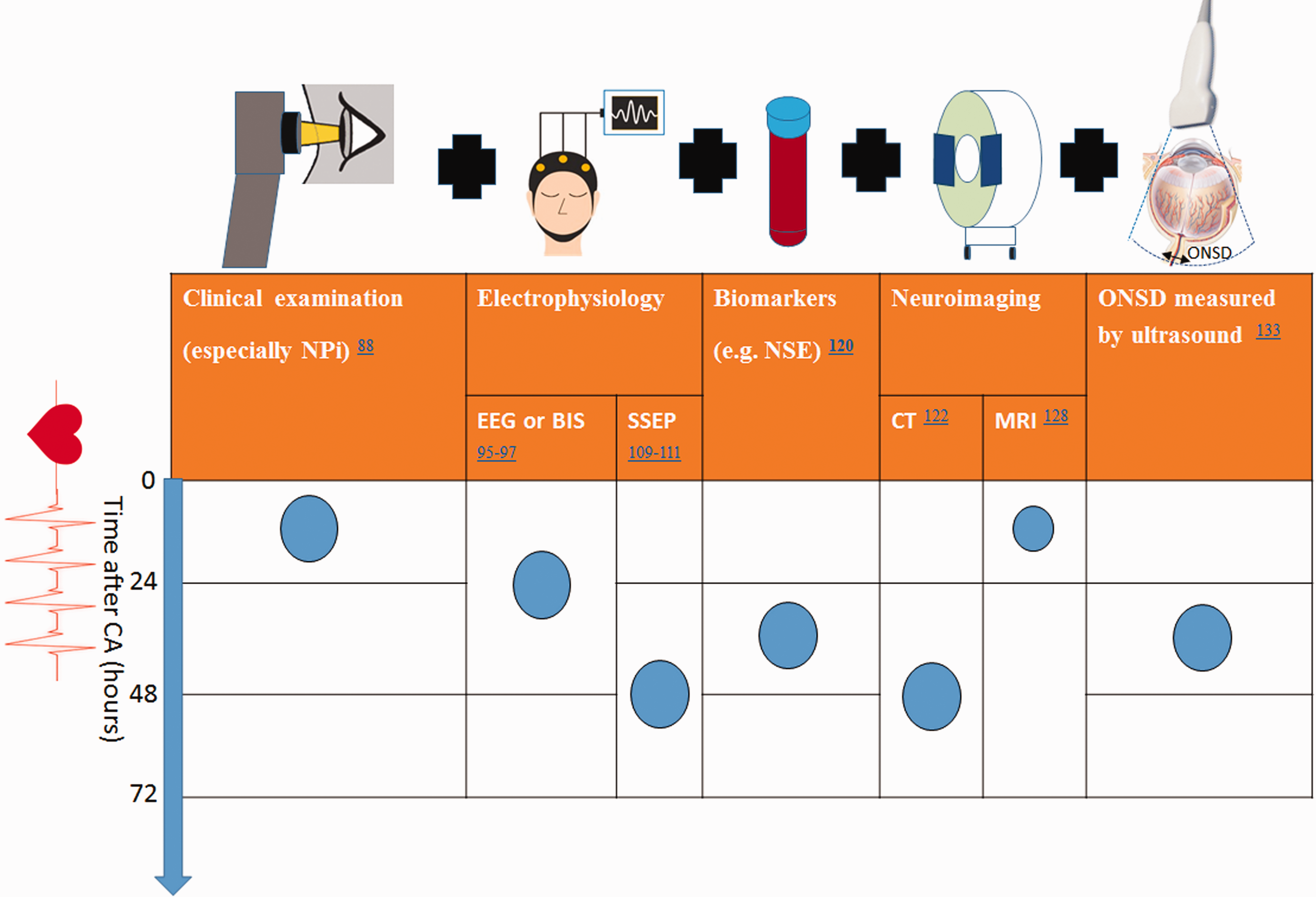
Earliest Time Points for Validated Multimodal Prognostication Tools. Blue bullet points represent the earliest validated time-point for each modality in predicting outcome in the first 72 hours after CA. Black ‘+’ symbol reinforces the concept that current recommendations suggest a multimodal approach, utilizing multiple modalities for neuroprognostication. Abbreviations: BIS, bispectral index; CT, computed tomography ; EEG, Electroencephalography; MRI, magnetic resonance imaging; NPi, neurological pupil index; NSE, neuron-specific enolase; ONSD, optic nerve sheath diameter; SSEP, somatosensory evoked potential.
Challenges and future perspective
RCTs are the cornerstone of evidence-based medicine. Due to the troublesome recruitment of large cohorts of CA patients, time-consuming and labor-intensive RCTs are inclined to be underpowered. Very few interventions in the modern era of neurocritical care have enough power to make a statistically significant difference in classical RCTs. Though patients are treated according to similar protocols, complex ancillary care remains an overall uncontrollable confounder. However, it is challenging to quantify the respective effects of TTM and ancillary care. In addition, the individualized titration and varying applications of TTM remains an uncontrolled variable between patients, between ICUs, and between institutions. 4
CA itself has various causes, leading to a heterogeneous subject pool. Further, the induction, as well as duration of CA, varies in different animal models, as does the implementation of TTM (depth, duration) in the preclinical setting. The experience of Operation Brain Trauma Therapy 139 may be adopted to improve the standardization and translation of preclinical CA research. With a better understanding of the pathophysiology of CA, particularly the evolution of brain injury with TTM, early and accurate neuro-prognostication could be available via multimodal approaches in the future.
Conclusions
While TTM has been a mainstay of treatment for comatose post-CA patients for almost two decades, there still exists no globally accepted, evidence-based consensus regarding the proper application of this therapy. The ideal depth, timing, and duration of TTM remain unknown, and thus the use of this therapy varies from patient to patient and institution to institution. At present, general recommendations allow for a wide target temperature range, from 32–36°C, for a duration of at least 24 h by the method of endovascular or surface cooling devices, but this recommendation is far from standardized. An individualized approach based on cardiac arrest details (e.g. etiology, duration, age, and comorbidities), and more importantly, based on the end-organs’ response to the TTM intervention, may be the optimal approach for this heterogeneous patient population, and future studies should focus on how to best tailor this therapy for the optimal neurological outcome.
Clinician prognostication post-CA is a primary factor in family decision making and WLST. A standardized prognostic algorithm does not yet exist, but current recommendations suggest that a multimodal approach encompassing physical exam, neuroimaging, electrophysiological testing, and serum biomarkers holds the highest sensitivity and specificity.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was partially supported by R01HL118084 and R01NS110387 from the United States National Institutes of Health (both to Xiaofeng Jia).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Songyu Chen contributed with data acquisition and analysis, searched and reviewed the literature, drafted and revised the manuscript; Brittany Bolduc Lachance contributed with data acquisition and analysis, and revised the manuscript; Liang Gao provided critical appraisal; Xiaofeng Jia conceived the idea, designed and formulated the review theme, viewed the literature, data analysis, and revised and finalized the manuscript.
