Abstract
The effects of upright postures on the cerebral circulation early post-ischemic stroke are not fully understood. We conducted a systematic review and meta-analysis to investigate the effects of head positioning on cerebral haemodynamics assessed by imaging methods post-ischemic stroke. Of the 21 studies included (n = 529), 15 used transcranial Doppler. Others used near-infrared, diffuse correlation spectroscopy and nuclear medicine modalities. Most tested head positions between 0° and 45°. Seventeen studies reported changes in CBF parameters (increase at lying-flat or decrease at more upright) in the ischaemic hemisphere with position change. However, great variability was found and risk of bias was high in many studies. Pooled data of two studies ≤24 h (n = 28) showed a mean increase in cerebral blood flow (CBF) velocity of 8.5 cm/s in the ischaemic middle cerebral artery (95%CI,−2.2–19.3) from 30° to 0°. The increase found ≤48 h (n = 50) was of 2.3 cm/s (95%CI,−4.6–9.2), while ≤7 days (n = 38) was of 8.4 cm/s (95%CI, 1.8–15). Few very early studies (≤2 days) tested head positions greater than 30° and were unable to provide information about the response of acute stroke patients to upright postures (sitting, standing). These postures are part of current clinical practice and knowledge on their effects on cerebral haemodynamics is required.
Introduction
Head positioning has been discussed as a non-pharmacological therapy in ischaemic stroke over many years. In theory, lying-flat in bed would increase blood flow to the ischaemic penumbra in the first hours or days after stroke, limiting damage and improving outcome. The large (n = 11,093) multi-site HeadPoST trial 1 was conducted to test this theory. Investigators reported no differences in 90-day disability outcomes in people with acute minor stroke who were nursed for 24 h after hospital admission in either a lying-flat position or with the head elevated to 30°.
There remains no consensus about the optimal head position for people with ischaemic stroke during bed rest, or the time course over which head position should be monitored. At the same time, recommendations to start out of bed activities early post-stroke appear in many stroke guidelines.2–9 In the large (n = 2104) multi-site AVERT trial, very early (started <24 h) and intensive upright activity reduced the odds of good recovery (mRS 0–2) compared to lower dose usual care. 10 It is plausible that reduced cerebral blood flow (CBF) in upright postures could lead to enhanced conversion of ischaemic penumbra to core and therefore increased tissue damage or interfered with recovery processes. 11 This effect could be even more detrimental in specific stroke populations, such as those with severe stroke or those who were haemodynamically impaired, for instance having persistent large vessel occlusions and/or intra or extracranial arterial high-grade stenosis with poor collaterals.12,13
A limitation of these two major trials was that CBF and the presence of salvageable tissue were not studied. Cutting-edge brain imaging studies suggest that parts of penumbra may be present up to 48 h post-stroke in some patients and an extended window of viable tissue might be present. 14 Given ongoing uncertainties, and that diametrically opposed approaches to acute ischemic stroke management exist, better understanding of the effect of upright postures (sitting, standing) on brain circulation and patient outcomes is warranted. Therefore, in this literature review, we aimed to explore the dynamics of blood flow to the brain with changes in positions of the head, assessed with any imaging method after ischaemic stroke.
Material and methods
Protocol and registration
A protocol of this systematic review was registered in advance on PROSPERO on 30 August 2018 (CRD42018105565).
Eligibility criteria
The search strategy was designed to identify studies examining cerebral haemodynamics using any imaging tools, across a range of head positions, in people with ischaemic stroke. Response to head position changes in the first day(s) after stroke, when cerebral haemodynamics may be most impaired, was of particular interest, but no time restriction was set in this review. All study designs, except single case reports, were eligible. No language or publication date restrictions were applied.
Participants
Participants over 18 years old with a diagnosis of ischaemic stroke were the target population. Mixed populations were considered if specific ischaemic stroke data could be extracted from the paper or by contacting the authors.
Outcome measures
Any imaging method and related outcome measures were considered, provided they assessed cerebral haemodynamics.
Head positioning
Studies that compared two or more head positions with different angles of elevation (e.g. lying flat and 30°) were eligible. Changes in head positioning could be passive (e.g. changing the head of bed angle or using a tilt-table) or active (e.g. sitting on a chair or standing).
Information sources and search
The following electronic databases were searched from inception to 25 July 2019: MEDLINE, Embase, CINAHL, Cochrane Library and Emcare; with reviews checked for potential additional eligible studies. The search strategy, peer reviewed by a librarian, can be found in Supplementary Appendix A. Keywords used to identify studies included: stroke, cerebral infarction, brain infarction combined with posture, head position, body position, head tilt, and combined with terms that could retrieve any type of measurement of cerebral circulation such as cerebral blood flow, cerebral blood flow velocity, cerebral haemodynamics, cerebral monitoring, autoregulation, transcranial Doppler ultrasound, near-infrared spectroscopy, diffuse correlation spectroscopy, computed tomography, magnetic resonance imaging, perfusion imaging.
Study selection
Titles and abstracts were screened independently by two authors (LC, KB). Both independently assessed full-text versions of potential studies for eligibility. Consensus was reached by discussion.
Data extraction process
Data from included studies were extracted into excel spreadsheets by the first author (LC) and checked by two other authors (SK, BC). We contacted nine authors for further information. Responses with further data were received from two authors. One author provided data that were specific for ischaemic stroke patients and an English translation. 15 The other author provided additional data on participant characteristics. 16
Risk of bias assessments
Risk of bias was assessed independently by two authors (LC, SK). Any disagreements were resolved through discussion.
Cohort studies were assessed using Joanna Briggs Institute Critical Appraisal tools for use in Systematic Reviews 17 with additional criteria added to address study methodologies not covered by this tool. Each item was assessed as low risk, unclear or high risk of bias. The overall bias of each cohort study was then classified as high or low/unclear risk of bias (Supplementary Appendix B).
Randomized controlled trials (RCT) were assessed using the Revised Cochrane risk of bias tool for randomized trials (RoB2). 18 Each item was assessed as low risk, some concerns or high risk of bias. The RoB2 algorithms were followed to judge the overall risk of bias of each study.
Synthesis of results
Data for all studies were clustered by imaging method, time post-stroke and head positions tested in each study with summaries on key outcomes created.
Time of assessment post-stroke onset was grouped into one of four time-points: ≤24 h, ≤48 h, ≤7 days and >7 days. More precise classification of time was not possible as the exact time of assessment was often not reported. Each time point could therefore include patient’s assessment between, for example, 0–48 h or 0–7 days. Data from studies that included one assessment time-point were not pooled across time-points (i.e. data from ≤24 h group is not included in ≤7 days group). Where possible, we analysed data from studies in the ≤24 h, ≤48 h, ≤7 days groups. Studies with assessments >7 days comprised a wide range of time-points: few weeks to many years post-stroke, making analysis less meaningful. If assessments were performed more than once, data were separated into the corresponding time-point categories where possible.
Studies with similar protocols, head positions and outcome measures tested within seven days were included in the meta-analysis if enough data were provided. CBF outcomes are continuous. We used means and standard deviations to estimate the pooled mean difference and 95%CI using an inverse-variance method and random-effects model, 19 which assumes the studies estimate different intervention effects that are related. We used RevMan software 5.3 to perform the meta-analysis and generate forest plots.
Statistical heterogeneity was assessed using I2. I2 shows the percentage of the variability in effect estimates that is due to heterogeneity rather than change. 19
Results
Study selection and characteristics
The search strategy identified 1893 records after duplicates were removed. After screening titles and abstracts, 78 records were selected for full text assessment of eligibility. Twenty-one studies met the inclusion criteria (see Figure 1 for flow chart and exclusions).
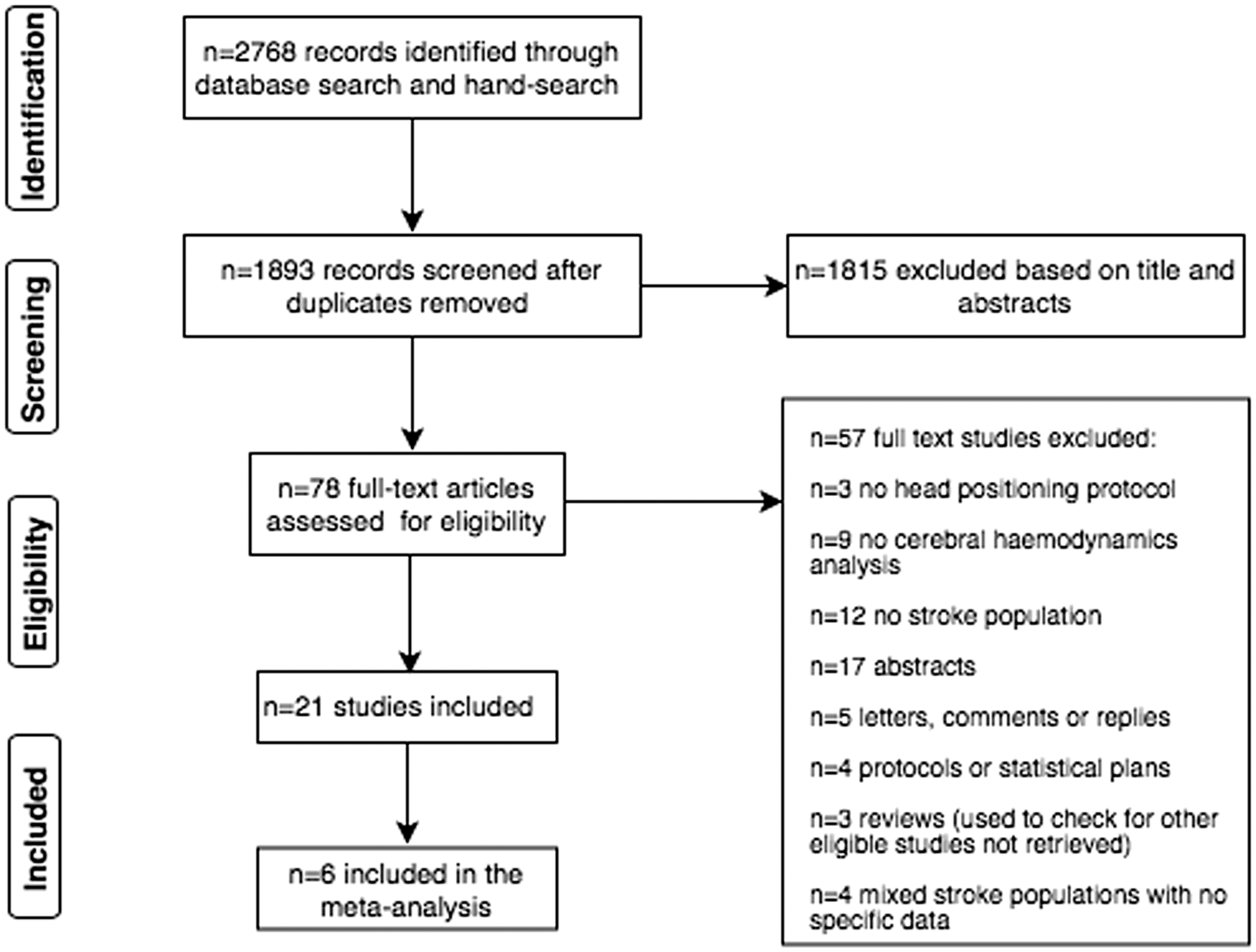
Flow of information through the different phases of the systematic review.
Included studies were published between 2001 and 2019. Study characteristics, design and outcomes of the 21 studies are summarised in Table 1. Most (n = 19) were cohort studies, of which 10 included a control group and 4 reported patient-related outcomes at follow-up;16,20–22 two of these were RCTs.21,22
Description of studies assessing cerebral haemodynamics using different imaging techniques with changes in head positions post-ischaemic stroke.
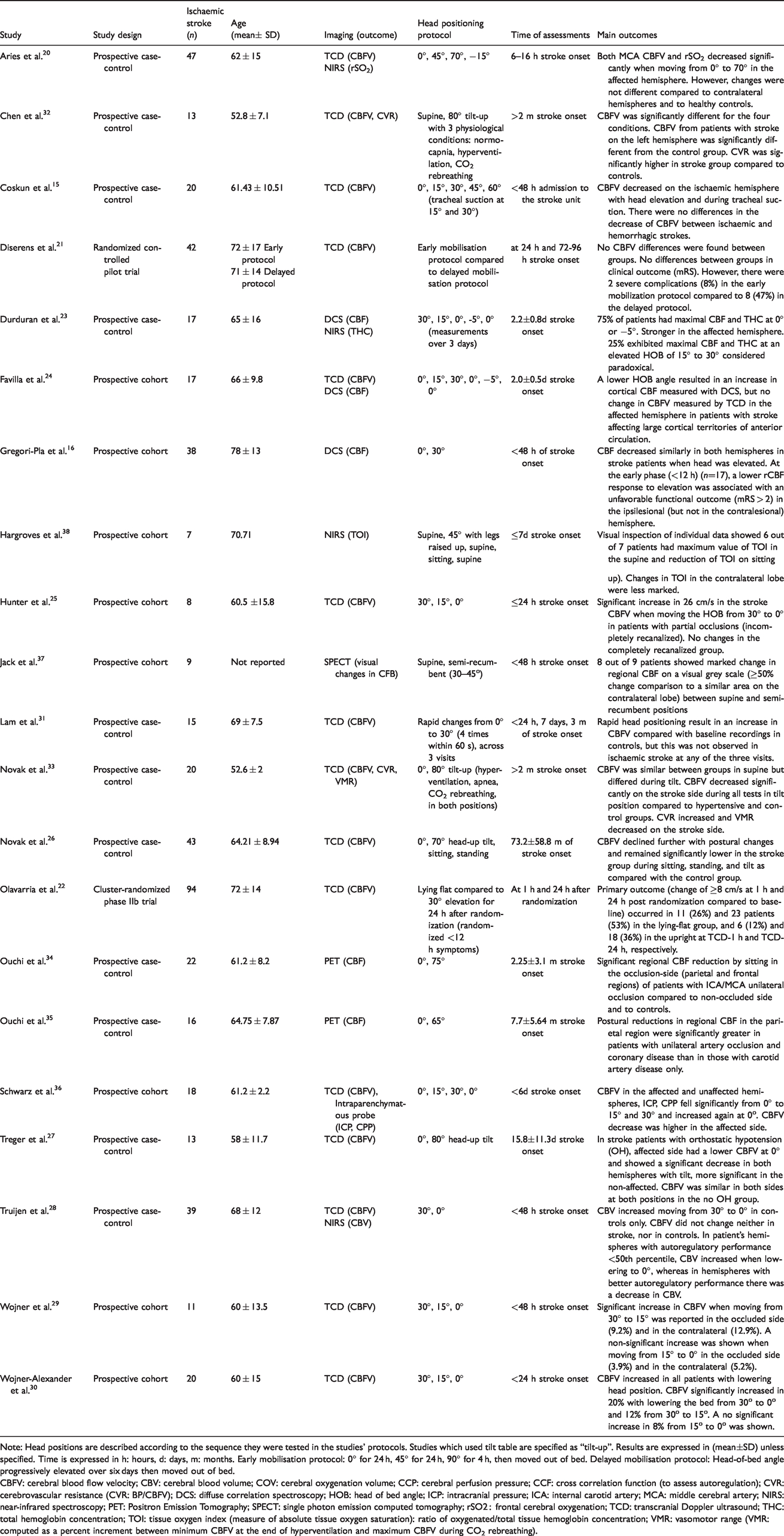
Note: Head positions are described according to the sequence they were tested in the studies’ protocols. Studies which used tilt table are specified as “tilt-up”. Results are expressed in (mean±SD) unless specified. Time is expressed in h: hours, d: days, m: months. Early mobilisation protocol: 0° for 24 h, 45° for 24 h, 90° for 4 h, then moved out of bed. Delayed mobilisation protocol: Head-of-bed angle progressively elevated over six days then moved out of bed.
CBFV: cerebral blood flow velocity; CBV: cerebral blood volume; COV: cerebral oxygenation volume; CCP: cerebral perfusion pressure; CCF: cross correlation function (to assess autoregulation); CVR: cerebrovascular resistance (CVR: BP/CBFV); DCS: diffuse correlation spectroscopy; HOB: head of bed angle; ICP: intracranial pressure; ICA: internal carotid artery; MCA: middle cerebral artery; NIRS: near-infrared spectroscopy; PET: Positron Emission Tomography; SPECT: single photon emission computed tomography; rSO2 : frontal cerebral oxygenation; TCD: transcranial Doppler ultrasound; THC: total hemoglobin concentration; TOI: tissue oxygen index (measure of absolute tissue oxygen saturation): ratio of oxygenated/total tissue hemoglobin concentration; VMR: vasomotor range (VMR: computed as a percent increment between minimum CBFV at the end of hyperventilation and maximum CBFV during CO2 rebreathing).
Participant characteristics
The studies included 529 people with ischaemic stroke and 222 healthy controls. Mean age reported across different studies ranged from 52.6 to 78 years for people with stroke and 43.4 to 64.5 years for healthy controls. Stroke severity was reported using the National Institutes of Health Stroke Scale (NIHSS) in 13 studies.16,20–31 Mean NIHSS range was 3–19. Lesion side was reported in 14 studies,15,20,23–26,28–30,32–36 while only 4 reported thrombolytic therapy.16,20,22,25
Time of assessments post-stroke onset
In seven studies (n = 243), participants were assessed within 24 h of stroke onset.16,20–22,25,30,31 In four studies (n = 80), they were assessed within 48 h,16,28,29,37 while in seven studies (n = 136), it was within seven days.15,21,23,24,31,36,38 In the final seven studies (n = 142), patients were assessed over seven days;26,27,31–35 months32–34 or years26,35 post-stroke (16 days to 6 years, Table 1).
Head positioning protocols
Head positioning protocols varied across studies (Table 1, Figure 2). All cohort studies included a 0° head position (lying-flat). In three studies, head of bed angles below zero (−15°, −5°) were also tested.20,23,24 Passive head of bed angle elevation varied from 15° to 75°. In two studies, active sitting was included,26,38 but only in one study was active standing tested. 26 Passive standing using a tilt-table (70°–80°) was tested in four studies.26,27,32,33 In one study, a specific rapid head positioning protocol was used (0° to 30° four times within 60 s). 31
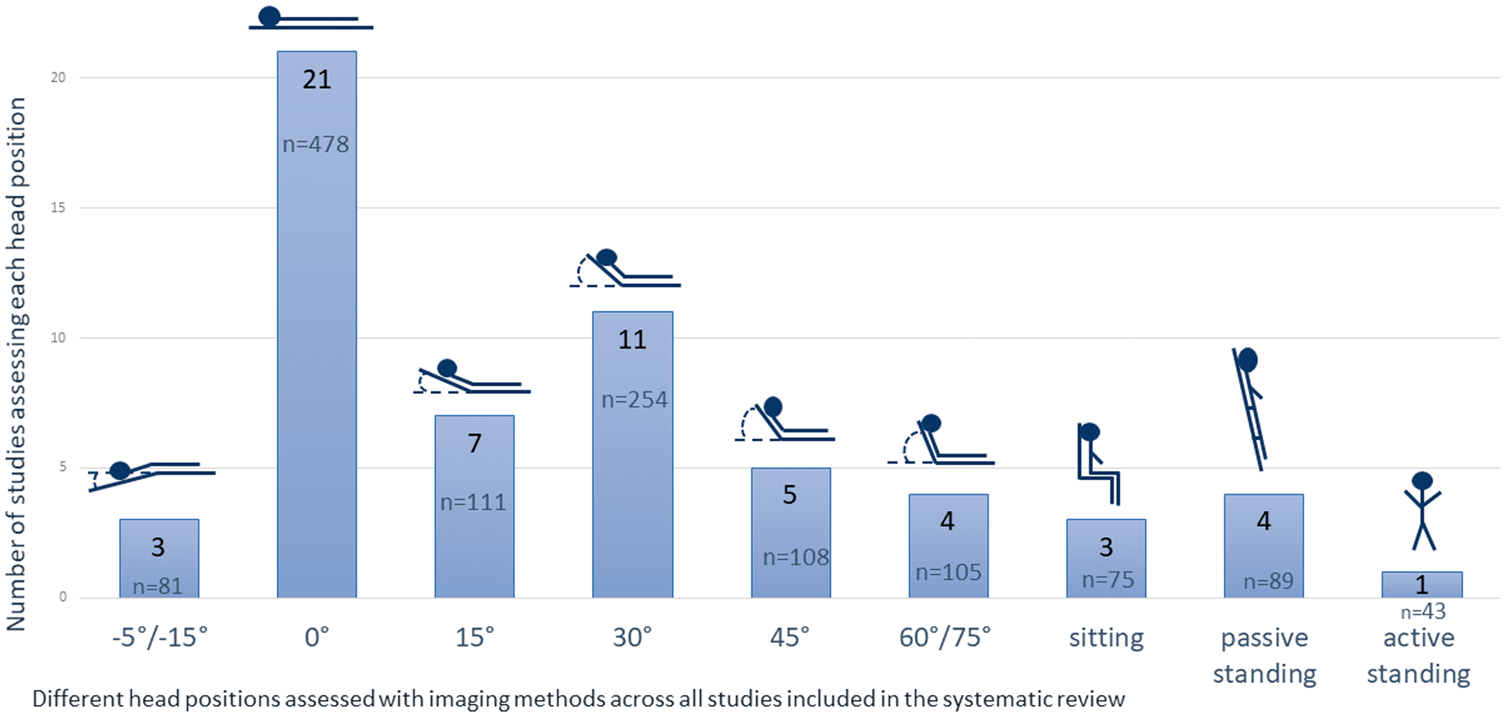
Number of studies assessing the specified head positions. Total number of patients tested in each position across the studies is reported below the number of studies. Diserens et al. 21 reported the early mobilisation protocol as 0° for 24 h, 45° for 24 h, 90° for 4 h, then patients were moved out of bed. Therefore, it was counted in 0°, 45° and sitting positions. As for the delayed mobilisation protocol any specific head position angle was reported, it is not represented in this figure. Jack et al. 37 reported the position as 30° to 45° semi-recumbent. It is represented here in the 45° head position only.
In one RCT, CBF was measured with patients recumbent at 24 h and 72–96 h post-stroke, following progressive head elevation (more rapid elevation to upright versus less rapid) over the first six days post-stroke. 21 The second RCT compared lying-flat to a 30° elevation 22 with assessments performed within 13 h and 36 h of stroke.
Imaging methods and cerebral blood flow parameters
In the majority of studies, transcranial Doppler (TCD) was used to assess real-time changes in the cerebral blood flow velocity (CBFV) (15 studies, n = 420).15,20–22,24–30,32,33,36 CBFV in the middle cerebral artery (MCA) is measured by low-frequency ultrasound probes which transmit Doppler waves through the skull.39–43 In six studies, cerebral haemodynamics was assessed using optical modalities such as near-infrared spectroscopy (NIRS) (four studies, n = 110)20,23,28,38 and diffuse correlation spectroscopy (DCS) (three studies, n = 72).16,23,24 Those methods measure cerebral oxygenation (oxy, deoxy-hemoglobin) and CBF using temporal intensity fluctuations of scattered light in the frontal cortex.24,44,45 CBF was assessed using positron emission tomography (PET) in two studies (n = 38),34,35 and single photon emission computed tomography (SPECT) in one study (n = 9). 37
Arterial vessel occlusions and stenosis status
In only eight studies (Table 2), was arterial occlusion or stenosis noted or included in analyses (total n = 109).22–25,29,30,34,35 In one study, patients with complete recanalization were compared to patients with incomplete recanalization. 25 In four studies, only patients with occlusive artery disease were included,29,30,34,35 with no comparison stroke group. Imaging methods to identify arterial occlusions varied. Definitions also varied and were often not clear. Although significant differences in response to head position change when patients had an occlusion compared to those without was found in several studies,23–25 this was not consistent across studies. 22
Studies of cerebral haemodynamics with changes in head positions post-ischaemic stroke which reported subgroup analysis of patients with arterial occlusions or stenosis.
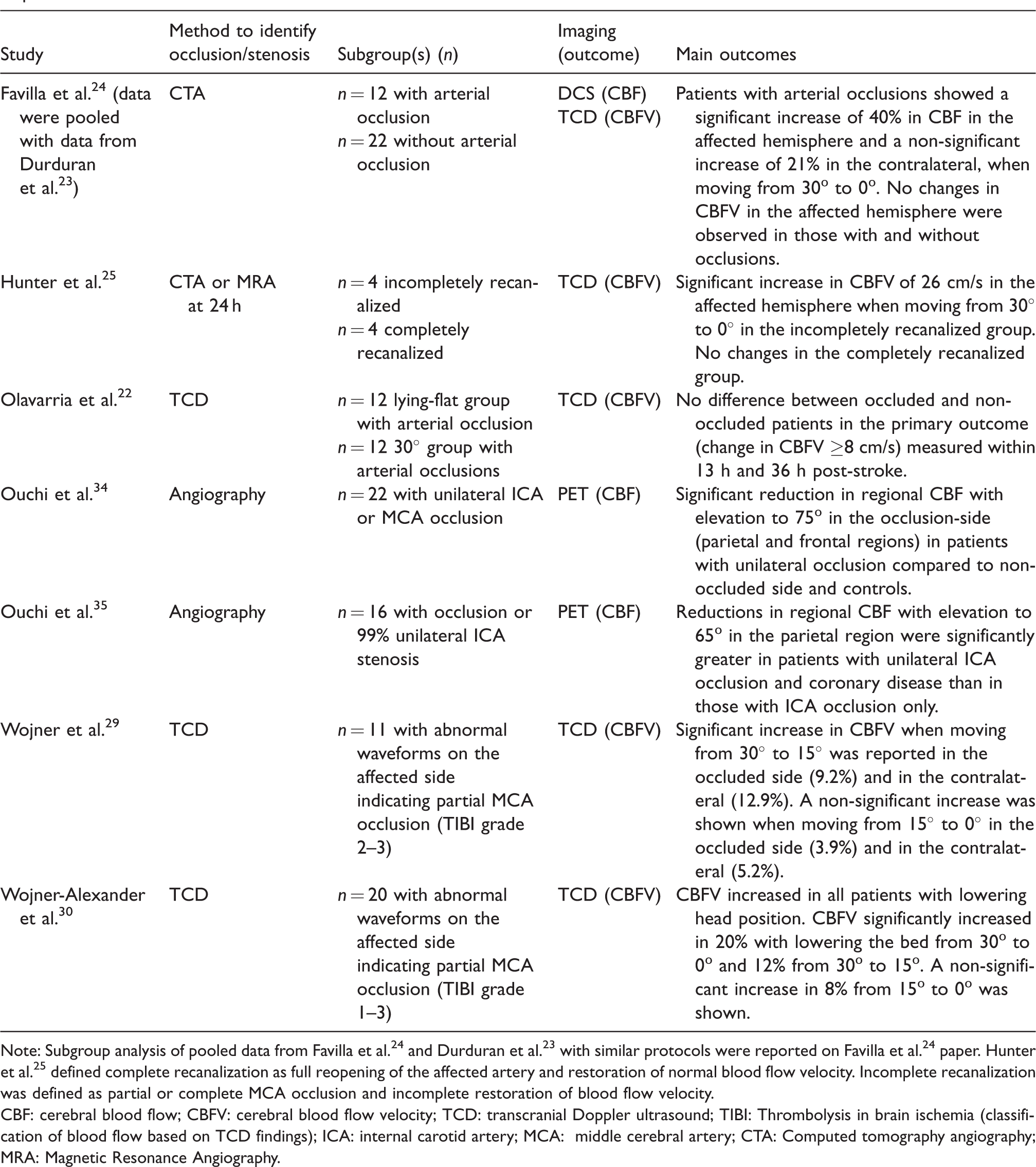
Note: Subgroup analysis of pooled data from Favilla et al. 24 and Durduran et al. 23 with similar protocols were reported on Favilla et al. 24 paper. Hunter et al. 25 defined complete recanalization as full reopening of the affected artery and restoration of normal blood flow velocity. Incomplete recanalization was defined as partial or complete MCA occlusion and incomplete restoration of blood flow velocity.
CBF: cerebral blood flow; CBFV: cerebral blood flow velocity; TCD: transcranial Doppler ultrasound; TIBI: Thrombolysis in brain ischemia (classification of blood flow based on TCD findings); ICA: internal carotid artery; MCA: middle cerebral artery; CTA: Computed tomography angiography; MRA: Magnetic Resonance Angiography.
Risk of bias within studies
Randomized controlled trials
One study 22 was assessed as low risk of bias in all domains. The second RCT 21 was assessed as having an overall high risk of bias due to deviations from intended interventions, missing outcome data, and some concerns in the selection of the reported results (Supplementary Appendix C).
Prospective studies
Eleven out of 19 studies were rated as having an overall high risk of bias,23,24,29,30,32–38 generally due to inadequate reporting of confounders, a lack of offline blinded assessments of outcomes, limited reporting of drop-outs and missing data. Eight prospective studies were assessed as low/unclear overall risk of bias (Supplementary Appendix C).15,16,20,25–28,31
Main outcomes
Prospective studies
The results from 18 prospective studies are summarized in Tables 1 and 3. Results of the remaining three studies (one prospective, two RCTs)21,22,31 with significantly different protocols are described below. Table 3 summarises main changes in the ischaemic hemisphere. Comparisons to the contralateral hemisphere and healthy controls are reported in Supplementary Appendices D and E. In the following section, results for the prospective studies are described according to time post-stroke.
Changes in CBF parameters in the ischaemic hemisphere at different head positions in relation to a lying-flat (0°) position reported in each study (n = 18 a ).
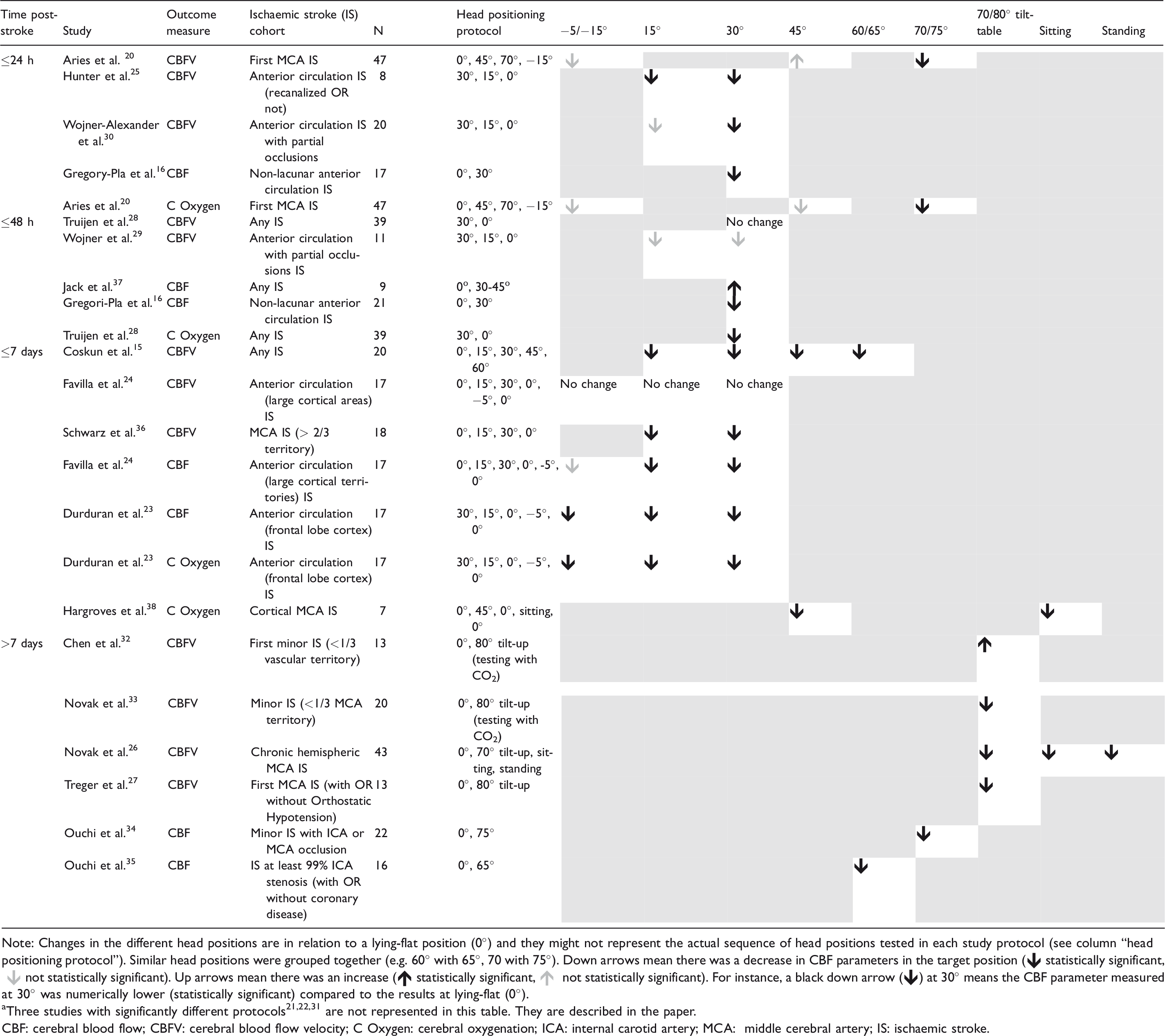
Note: Changes in the different head positions are in relation to a lying-flat position (0°) and they might not represent the actual sequence of head positions tested in each study protocol (see column “head positioning protocol”). Similar head positions were grouped together (e.g. 60° with 65°, 70 with 75°). Down arrows mean there was a decrease in CBF parameters in the target position ( statistically significant,
statistically significant,  not statistically significant). Up arrows mean there was an increase (
not statistically significant). Up arrows mean there was an increase ( statistically significant,
statistically significant,  not statistically significant). For instance, a black down arrow (
not statistically significant). For instance, a black down arrow ( ) at 30° means the CBF parameter measured at 30° was numerically lower (statistically significant) compared to the results at lying-flat (0°).
) at 30° means the CBF parameter measured at 30° was numerically lower (statistically significant) compared to the results at lying-flat (0°).
aThree studies with significantly different protocols21,22,31 are not represented in this table. They are described in the paper.
CBF: cerebral blood flow; CBFV: cerebral blood flow velocity; C Oxygen: cerebral oxygenation; ICA: internal carotid artery; MCA: middle cerebral artery; IS: ischaemic stroke.
≤24 h post-stroke
Studies of head positioning in the hyperacute phase are limited. Five prospective studies tested patients within 24 h of stroke symptoms.16,20,25,30,31 TCD was the main imaging device used and CBFV was the main outcome (n = 4).20,25,30,31 Overall, when comparing a flat or below-zero to a more elevated position, a decrease in blood flow parameters in the ischaemic hemisphere was reported in an elevated head position, except for Aries et al. 20 (n = 47) who reported a non-significant increase in CBFV at 45°. The highest head position assessed ≤24 h was 70° with a significant decrease reported in the ischaemic hemisphere in both CBFV and cerebral oxygenation. 20 However, no difference was shown compared to the contralateral hemisphere or to controls (Table 3, Appendices D and E). 20
In two small studies (total n = 28), patients with large vessel occlusions were assessed within 24 h. Hunter et al. 25 reported a significant increase in MCA velocities in the incompletely recanalized group, identified through CT or MR angiography at 24 h, when moving from 30° to 0°. At the same head of bed movement, Wojner-Alexander et al. 30 also reported a significant CBFV increase by 20% at 0° in patients with partial MCA occlusions identified using TCD. 46
Gregori-Pla et al. 16 showed a decrease in CBF in the ischaemic hemisphere, however not different to the contralateral, measured by DCS when moving from 0° to 30°. Results were similar at later time-points (Tables 1 and 3). For patients tested at a very early phase (<12 h) (n = 17), a lower change in CBF in the affected hemisphere was associated with an unfavourable functional outcome (mRS > 2). The association was not found >12 h (n = 21). 16 Lam et al. 31 found no difference in CBFV in stroke patients compared to controls assessed with a repeated rapid head position change protocol (0° to 30° 4 times within 60 sec).
Overall, results demonstrate a trend for CBF parameters to decrease in elevated positions compared to lying-flat (in the range of 0–70°). However, most studies tested head positions between 0° and 45°. The effects of sitting or standing (90°), a common activity undertaken in clinical setting, have yet to be tested.
≤48 h post-stroke
In four studies, protocols were applied ≤48 h of symptom onset.16,28,29,37 Truijen et al. 28 (n = 39) reported no change in CBFV when lowering the head of bed from 30° to 0°. However, for those with autoregulatory performance <50th percentile (phase difference of the arterial pressure-to-CBFV transfer function), an increase in cerebral oxygenation at 0° was shown using NIRS, suggesting that cerebral autoregulatory performance affects cerebrovascular response with head position changes.
In the second study by Wojner et al. 29 (n = 11), a significant CBFV increase was found moving from 30° to 15° in both hemispheres for patients with partial arterial occlusions. In contrast, Jack et al. 37 reported an increase in perfusion by visual analysis using SPECT in eight out of nine patients when moved from lying-flat to 30°–45°.
Similar to studies conducted ≤24 h of symptoms, lower CBF parameters were observed in elevated head positions compared to lying-flat (0°) in three out of four studies performed ≤48 h.16,28,29 However, no head positions higher than 45° were assessed.
≤7 days post-stroke
In the six prospective studies that included assessments ≤7 days of onset,15,23,24,31,36,38 TCD (n = 4),15,24,31,36 DCS (n = 2)23,24 and NIRS (n = 2)23,38 were used. Passive head of bed angles ranged between −5° and 30°. One study tested a sitting-up position (90°). 38
Coskun et al. 15 reported a decrease in CBFV when moving from lying-flat to 15°, 30°, 45° and 60° (n = 20). Similarly, Schwarz et al. 36 showed that CBFV, intracranial pressure (ICP), and cerebral perfusion pressure all fell significantly from 0° to 15° and 30° and increased again at 0° in patients with large ischaemic MCA strokes (n = 18). CBFV decreased bilaterally but was more pronounced in the affected side. Hargroves et al. 38 also reported a reduction in oxygenation index in the affected hemisphere in six out of seven patients through visual inspection using NIRS between lying-flat and 45° and 90°. This was the only study to analyse cerebral haemodynamics in upright postures (90°) in acute ischaemic stroke.
While Favilla et al. 24 reported no change in CBFV on head elevation to 30° measured using TCD, they found significant reduction in cortical CBF measured with DCS (n = 17). Varying responses across patients for both methods were reported, including unexpected changes in CBF, i.e. an increase in blood flow parameters when moving from lying-flat to a more elevated position. Similarly, between-subject variability was found in the study by Durduran et al. 23 (n = 17) using both DCS and NIRS. While 75% of patients had maximal CBF and total haemoglobin concentration at 0° or −5°, 25% had maximal CBF at more elevated positions (15° or 30°).
>7 days post-stroke
In the post seven days period, seven studies were performed months or even years after stroke.26,27,31–35 Five used TCD26,27,31–33 and two used PET.34,35 In contrast to studies conducted earlier, more elevated positions ranging from 65° to 90° were tested, with most (n = 4 studies) using a tilt table (70–80°).26,27,32,33 Study protocols in this time range were highly variable. Novak et al. 26 (n = 43) tested whether CBFV was dependent on perfusion pressure in older patients with chronic MCA infarcts using a protocol of hyperventilation followed by CO2 rebreathing. Significantly, lower CBFVs in both hemispheres were found in stroke patients at baseline and with postural challenges compared to age and sex-matched controls. Previously, Novak et al. 33 (n = 20) showed a decline in CBFV in the affected hemisphere during tilt-up at two months after minor ischaemic stroke. Within a month post-stroke, Treger et al. 27 (n = 13) showed a significant decrease in CBFV in both hemispheres (greater in the unaffected) at 80° in those with known orthostatic hypotension compared to those without.
Studies by Ouchi et al.34,35 using PET, including stroke patients with occlusive artery disease, revealed a decrease in CBF at 65° and 75° of head elevation compared to 0°, with greater change observed in those with concomitant coronary disease. 35
Blood pressure and head position changes
In most studies, the monitoring of physiological parameters such as blood pressure (BP) during head positioning protocols was noted (n = 19),15,20–22,24–38 however not all report BP as an outcome. In those that reported, no difference in BP with position change was found in the majority of studies,24,29,30,32,34,35,37 or there was small and/or not significant variability.15,25,28,38 Of the studies in which CBF parameters changed in the ischaemic hemisphere (i.e. increase in CBF parameters at lying-flat or decrease at more elevated head positions) (n = 17),15,16,20,22–30,33–36,38 in only five studies did BP change significantly with position change.20,26,27,33,36 In most studies, both CBF parameters and BP decreased at more elevated head positions,20,27,36 while in one study stroke hypertensive patients showed a decrease in CBFV at tilt-up and an increase in BP. 33 BP increment with tilt-up was much smaller in stroke normotensive patients. 33
Meta-analyses
Six out of 21 studies, all using TCD (assessing CBFV in the MCA), could be included in the meta-analysis (Figure 3). Pooled data of two studies within 24 h post-stroke (n = 28)25,30 showed a mean increase of 8.5 cm/s in the ischaemic hemisphere (95% CI, −2.2–19.3) when the head of bed was moved from 30° to 0°. Within 48 h, a lower mean increase of 2.3 cm/s (95% CI, −4.6–9.2) in the ischaemic hemisphere was shown from 30° to 0° (2 studies, n = 50).28,29 For those ≤7 days (two studies, n = 38)15,36 a mean increase of 8.4 cm/s (95% CI, 1.8–15) was observed in the affected hemisphere when moving to 0° from 30°. Some heterogeneity within this last comparison was identified (I2 = 12%; χ 2 2.26, df 2; P= 0.32). According to the Cochrane classification of heterogeneity, 19 this might not be important. We think this could be attributed to a very different cohort of patients in the study by Schwarz et al. 36 (large strokes (> 2/3 MCA territory) treated in intensive care, n = 18).
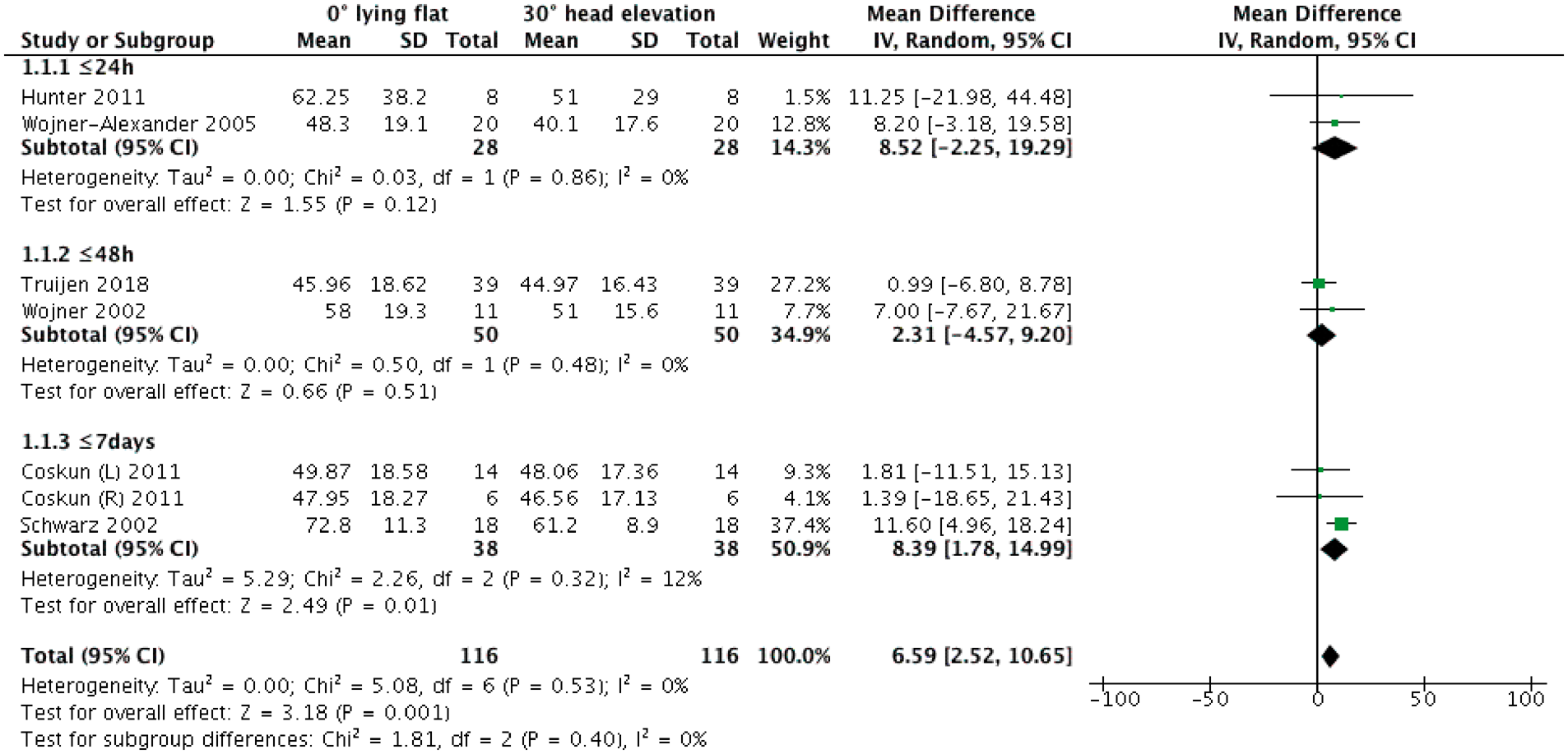
Forest plot of mean changes in MCA CBFV in the ischaemic hemisphere between 0° and 30° of head elevation for each time-point within the first week post-stroke. Six prospective studies were included (n = 2 ≤ 24 h, n = 2 ≤ 48 h, n = 2 ≤ 7 days). For each study and time-points, mean differences in CBFV were estimated. IV: inverse variance; CI: confidence interval.
In four out of six studies, data on the contralateral hemisphere were included (Figure 4). When the head of bed was moved from 30° to 0°, mean changes estimated from one study (n = 8) 25 showed an unexpected decrease of 2.1 cm/s (95%CI, −21.8–17.5) ≤24 h. For the same head of bed movement, within 48 h there was an increase of 3.2 cm/s (95%CI, −5.8 – 12.1, two studies, n = 50),28,29 while a mean increase of 3.7 cm/s was reported (95%CI, 1.7–5.7, one study, n = 18) ≤7 days. 36
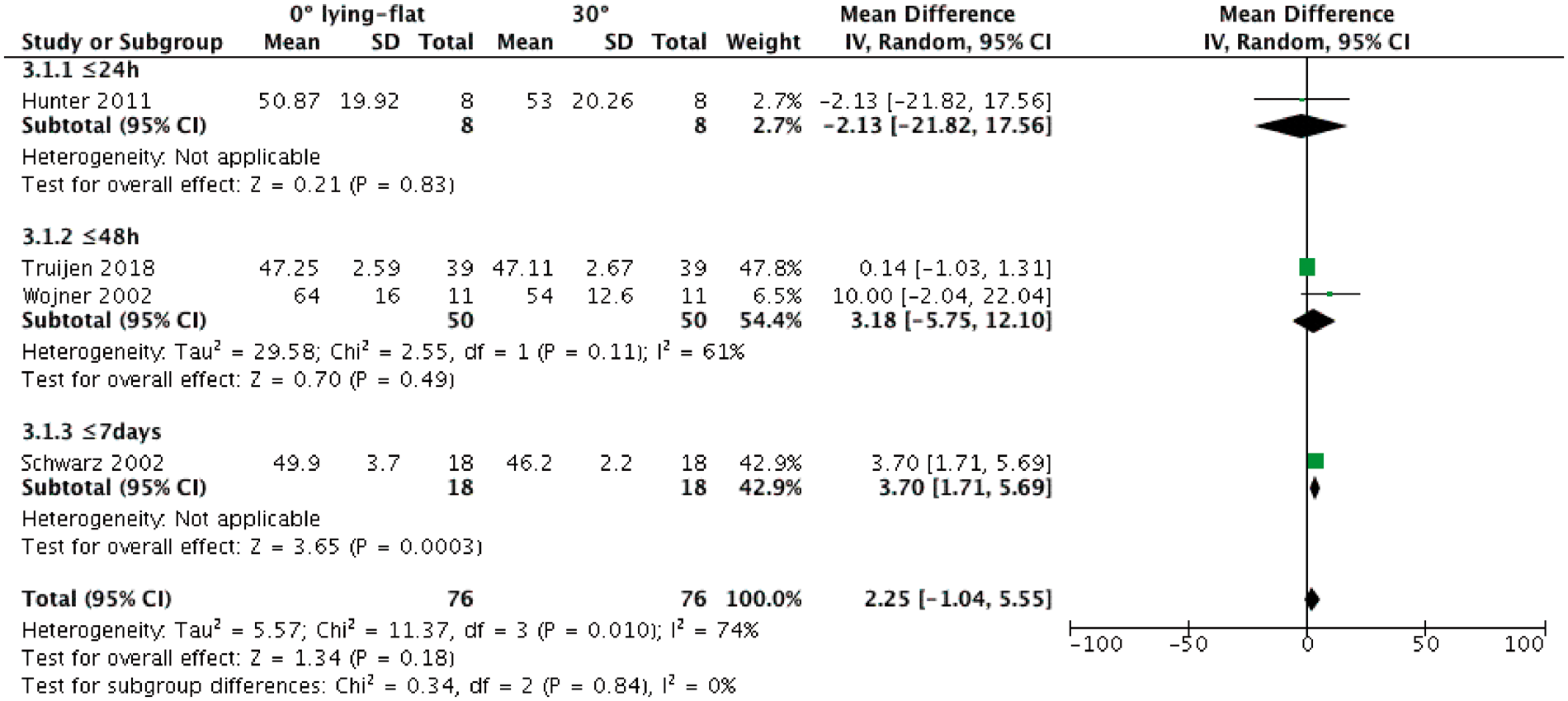
Forest plot of mean changes in MCA CBFV in the contralateral hemisphere between 0° and 30° of head elevation for each time-point within the first week post-stroke. Four prospective studies were included (n = 1 ≤ 24 h, n = 2 ≤ 48 h, n = 1 ≤ 7 days). For each study and time-points, mean differences in CBFV were estimated. IV: inverse variance; CI: confidence interval.
Randomized controlled trials
The protocols of the two RCTs differed substantially from each other. Olavarria et al. 22 showed that patients randomised to lying-flat had higher CBFV measured with TCD in the affected hemisphere within 13 and 36 h post-stroke compared to patients randomised to 30°. No differences in functional outcome were found between groups at 90-days. 22 In the trial by Diserens et al., 21 no difference in CBFV was found at 24 h and 72–96 h post-stroke between patients randomised to rest in bed (slow, progressive head elevation to standing over six days) or an ‘early mobilisation’ (more rapid progressive head elevation to standing by day 3). In terms of clinical outcomes, severe complications and drop-outs were significantly higher in the delayed mobilisation group. 21
Discussion
Knowledge of cerebral haemodynamics continues to grow as new measurement methods, both invasive and non-invasive, are developed and refined. Measuring cerebral blood flow in a range of head positions is possible but challenging. This review provides an up to date summary of the evidence in this field. We acknowledge a number of limitations. Notably, included studies were characterized by variability in methods and outcome measures, were often small in size and exhibited inconsistent reporting, even for studies using similar imaging methods, which restricted the synthesis and interpretation of results. While we included studies published in languages other than English, some non-English publications may not have been indexed in the major databases searched. Additionally, we did not include grey literature or perform a formal publication bias assessment. Our results should be considered in this light.
In this review, we found five different imaging methods of assessing post-stroke cerebral haemodynamics. PET remains the gold-standard for tissue CBF mapping at steady state. 47 In studies by Ouchi et al.,34,35 a research PET scanner with a mobile gantry was used. This imaging device, however, is not commonly available in the clinic. TCD, NIRS and DCS on the other hand allow non-invasive real-time monitoring of head position changes. NIRS and DCS have been increasingly used in critical care as they assess perfusion of parenchymal microvasculature;23,44,48–50 however, measurement is restricted to the frontal cortex and does not reach deeper subcortical areas. 24 TCD has good penetration to allow access to most large intracranial arteries. It has proved to be a reliable method giving accurate diagnostic and prognostic information post-acute stroke, 51 yet is operator dependant and cannot be performed in some people due to poor temporal windows. 52 In our review, TCD was the most used technique to assess dynamic CBFV inside intracranial conduit vessels. However, it does not reflect the direct flow within parenchymal tissue. Previous studies showed CBFV can be used as a surrogate measure of CBF provided the artery diameter does not change,41–43 yet this may not be guaranteed in such a dynamic condition as acute ischaemic stroke. While TCD has limitations, it remains the current best method for indirectly measuring CBF with position change in ischaemic stroke.
Given the challenges of imaging techniques it would be helpful if other physiological measures could provide in-proxy markers of CBF. However, systemic BP, a logical candidate, showed little consistent relationship with CBF in this review. In studies showing change in CBF parameters with position change, only 30% did BP change. Assessments of BP may help monitor symptomatic patients such as those with orthostatic hypotension but does not appear to substitute cerebral assessments of blood flow.
We found evidence suggesting that compared to elevated head positions, lying-flat increases CBF, CBFV and cortical oxygenation in the affected hemisphere. However, these findings were not uniform across studies. We saw significant between-patient variability. The risk of bias in many studies was high. Further, few very early studies (≤48 h) tested head positions greater than 30°. Therefore, the response of acute patients to more upright postures is uncertain.
Despite large trials1,10 uncertainty remains about the benefit or risk of maintaining flat or low positions or alternatively rapid upright activity in individuals early after ischaemic stroke. The mechanism by which risk or benefit may occur is currently unknown and this requires further more mechanistically informed high-quality studies. They should gather not just immediate effects of position change, but longer term assessment of functional outcome.
The presence of persistent arterial occlusion and the response to position change was assessed in a handful of studies in this review. Persistent arterial occlusion is recognised as an independent predictor of worse outcomes in patients treated with intravenous thrombolysis.51,53–55 Most patients with atherothrombotic cerebral disease rely on collateral blood flow. However, not all patients will have adequate collaterals. The presence of collateral flow has great significance, as leptomeningeal collaterals identified on CT angiography predict better clinical outcomes. 12 Having persistent arterial thrombus or severe arterial stenosis could reduce blood flow to the penumbra, which could potentially be exacerbated by orthostatic challenge in acute stroke. However, whether stroke patients with arterial occlusions or high-grade stenosis are more susceptible to neurological worsening upon orthostatic challenge is currently unknown. Studies comparing stroke patients with and without arterial occlusions or stenosis with head position changes are needed. This may help formulate clinical guidelines concerning head positioning and early upright activity and clearly identify best protocols in specific groups of stroke patients.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20922457 - Supplemental material for Cerebral haemodynamics with head position changes post-ischaemic stroke: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20922457 for Cerebral haemodynamics with head position changes post-ischaemic stroke: A systematic review and meta-analysis by Lilian B Carvalho, Sharon Kramer, Karen Borschmann, Brian Chambers, Vincent Thijs and Julie Bernhardt in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LBC is supported by PhD scholarship of the Centre for Research Excellence in Stroke Rehabilitation and Brain Recovery (NHMRC Grant 1077898).
Acknowledgements
We would like to thank the Austin Health Sciences Library, particularly Helen Baxter, for helping with the development of the electronic search strategy. We acknowledge Assoc/Prof Halise ÇOSKUN for kindly providing the English translation of their paper and some additional unpublished data.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JB and VT have competitive grants to study the effect of early mobility training within a clinical trial (N1139712).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
