Abstract
Stroke remains a major cause of serious disability due to the brain’s limited capacity to regenerate. Current treatments focus on acute recanalization of the occluded blood vessels; however, currently there are no approved therapy options to regenerate neural circuits and reduce stroke-related disability. To promote recovery, therapeutic angiogenesis has been proposed as a promising target. Although restoration of blood vessels providing oxygen and nutrients to the peri-infarct regions may be beneficial, newly generated capillaries may also carry pathophysiological risk factors that need to be considered. One major concern are adverse effects including edema formation and haemorrhagic transformation due to the comprised endothelial barrier function during vascular remodelling. This brief opinion article will discuss the challenges and the newest advancements of angiogenesis as a therapeutic strategy for ischemic stroke.
Introduction
Angiogenesis, the formation of new capillaries from existing blood vessels, is a widely conserved response to hypoxia. It is involved in many human physiological functions such as wound healing and the maturation of the female reproductive system but also plays a central role in the pathological regulation of tumour growth, retinopathies and ischemic diseases. 1 In many ischemic diseases, e.g. myocardial infarction and limb ischemia, angiogenesis is a key restorative process and has been clearly shown to be beneficial.2,3 In the ischemic brain, however, the responses and regulatory mechanisms underlying angiogenesis after injury may be more complex and less clear. 4
Following ischemic stroke, angiogenesis occurs in the peri-infarct regions and has been shown to positively correlate with survival and recovery in laboratory animals5,6 and patients.7,8 Clearly, restoration of the blood supply providing oxygen, nutrients and trophic factors is beneficial for stroke recovery. However, it is debated whether physiological post stroke angiogenesis truly contributes to functional recovery.
Moreover, although physiological post-stroke angiogenesis is very limited, it is strongly debated if therapeutic enhancement of this process might be beneficial for patients due to safety concerns. Aberrant growth of new capillaries poses the risk for immature vessel formation that may enhance edema formation, haemorrhagic transformation and blood–brain barrier damage.9,10
In this brief opinion article, both aspects, the potential benefits and dangers of angiogenic therapies, are carefully summarized and compared with reference to the newest development in the field (Figure 1).
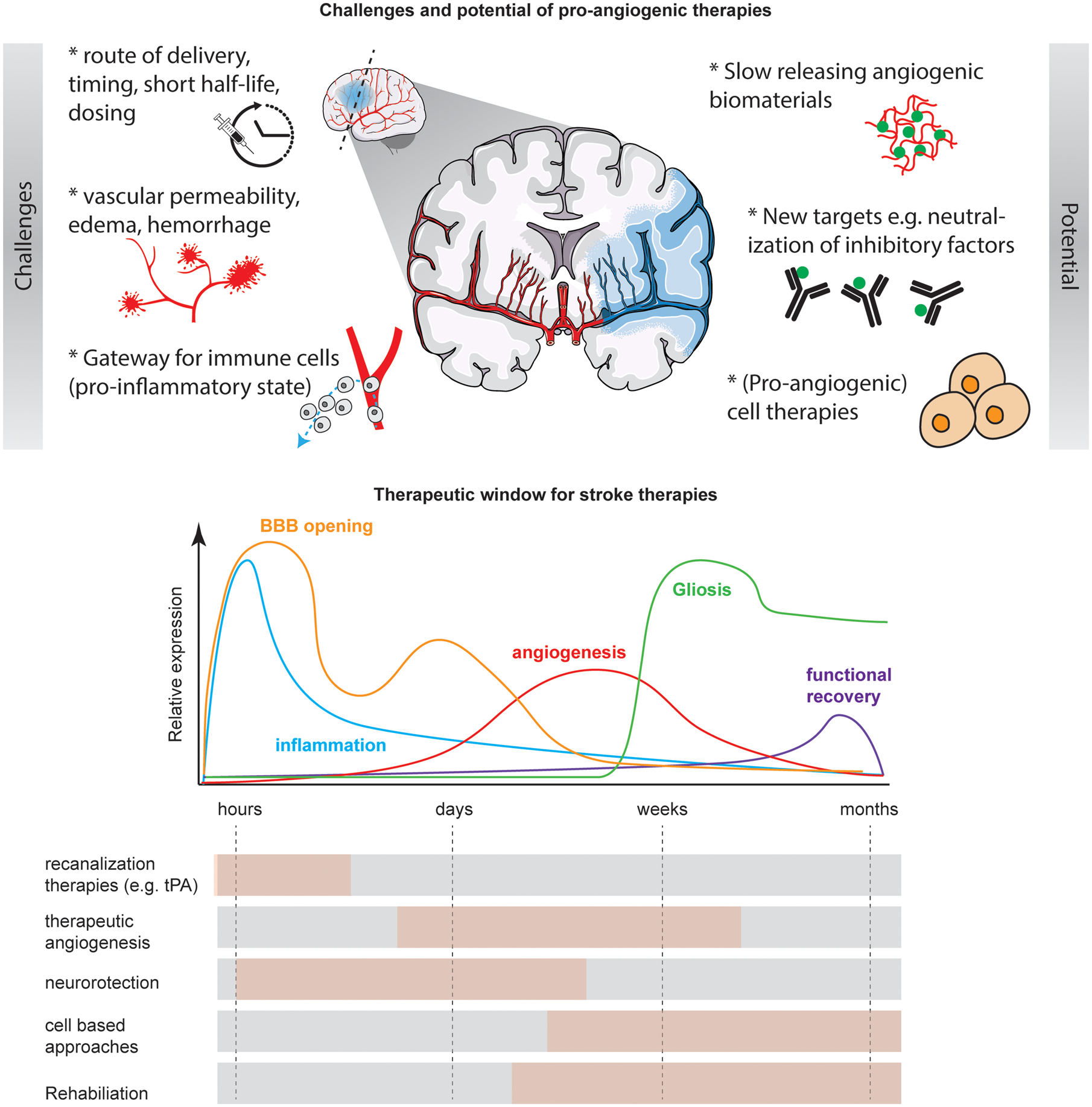
Challenges and potential of pro-angiogenic therapies. Therapeutic restoration of the vascular network is an interesting regenerative target following stroke. The major challenges of this approach are the correct timing, route of administration and dosing of the pro-angiogenic substance. Moreover, newly formed immature vessels may be leaky and cause bleedings. New advancements in pre-clinical research suggest slow-release biomaterials, inhibitory vascular molecules or the use of pro-angiogenic cell therapies. The therapeutic window of pro-angiogenic therapies ranges from days to multiple weeks and is compared with other treatment options. BBB: blood–brain barrier; tPA: tissue plasminogen activator.
What are the potential dangers and limitations of pro-angiogenic therapies following cerebral ischemia?
The formation of blood vessels in the CNS is a complex and highly ordered process that relies upon extensive signalling between vascular cells and the other components of the BBB. Nonetheless, therapeutic approaches have been mostly very simplistic almost exclusively focusing on the master regulator of angiogenesis the vascular endothelial growth factor (VEGF). Within the first in vivo stroke animal models, it has become clear that although VEGF may increase angiogenesis, it also enhances vascular permeability. 11 The latter is associated with edema formation and an increased haemorrhagic risk, which may have fatal consequences for the patient. 11 Administration of VEGF in the hyperacute phase following stroke has been shown to enhance edema formation and its pharmacological blockage led to improved blood–brain barrier integrity in a preclinical rat stroke model.12,13 Administration of high concentrations of VEGF has been shown to fail at the recruitment of other BBB components including peri-vascular cells (pericytes and smooth muscles), extracellular matrix (ECM) components and tight junctions that are necessary for a functional neurovascular unit. 14 Moreover, conflicting results have been reported potentially due to the short half-life time and limited spatiotemporal precision in its application. 15 VEGF is known to also have non-angiogenic functions such as being a neurotrophic and neuroprotective factor by modulation of glutamatergic synaptic excitability and interference with ion-channels physiology. 16 Similar effects have also been observed when interfering with other vascular growth stimulators and receptors. 17
Recent interest in the impact of comorbid vascular diseases such as diabetes following stroke has indicated detrimental effects of pathological vascularization on stroke outcome and recovery. Moreover, potential targets for intervention differ in those pre-clinical animal models of comorbid diabetic vascular disease and their response to therapeutic intervention might be altered. 18
There is also some evidence that post-stroke angiogenesis is transient and rather functions as a gateway for macrophage infiltration following stroke. It has been observed that there is a higher accumulation of immune cells in areas with increased vascular density. Although this hypothesis needs to be further tested, therapeutic enhancement of angiogenesis may implement a more pro-inflammatory environment that may increase neuronal damage within the peri-infarct regions.
These observations need to be closely monitored in future pre-clinical studies to ensure a safe and effective application of the pro-angiogenic therapeutics following ischemic injury in the CNS.
What is the potential of pro-angiogenic therapies?
In the ischemic brain, treatment options are very limited and exclusively focus on acute reperfusion therapies including recombinant tissue plasminogen activator (tPA) and/or mechanical thrombectomy (MT).19,20 Although these methods are very effective, they have a very short time window and are therefore not applicable for many patients.
There are pre-clinical strategies to delay the therapeutic time window of tPA that include co-administration of granulocyte colony-stimulating factor (G-CSF) 21 or cell-based strategies 22 to enhance endothelial cell survival and avoid haemorrhagic transformation. Nonetheless, there is a high demand for regenerative therapies to reverse neurological damage once a stroke has occurred.
Preclinical studies suggest that restoration of vascular circuits may be an effective strategy for reducing neurological damage and improve functional outcome.5,6 Angiogenesis has been shown to occur in stroke patients as documented in post-mortem tissue. Several studies have pointed out a positive relationship between angiogenesis and stroke outcome.7,8 Patients with higher vascular densities in the peri-infarct regions following stroke showed higher survival and higher recovery rates. 8 Also, high levels of VEGF and other pro-angiogenic factors in the serum correlated with favourable functional outcome.23,24 Some clinical data from ischemic diseases outside the CNS such as coronary heart disease or limb ischemia have shown positive effects of pro-angiogenic factors including VEGF, FGF and HGF that were applied as a gene therapy or on a protein level. 15 However, bigger trials did not confirm the results, most likely because of the complexity in the route of administration dosing and the complex biology of these substances.
Consequently, new pre-clinical approaches focus on biodegradable biomaterials or balanced vector delivery as a slow release source of growth factors. In animal models, it has been demonstrated that slow release of growth factors may lead to improved angiogenesis, functional recovery with integrating neural networks. These slow release strategies seem also to provide more natural maturation of blood vessels as documented by higher coverage of peri-vascular cells and no indication of leakage.5,25
Alternative pre-clinical approaches target inhibitory factors within the scar-forming peri-infarct regions such as guidance molecules (netrins, ephrins, semaphorins, slits, nogos), many of which act on the vascular and nervous system. Neutralization of these proteins has been shown to improve angiogenesis, tissue recovery without the risk of enhanced vascular permeability.6,26
Similar observations have been made with cell-based approaches in different pre-clinical models of stroke. The preferred cell type is bone-marrow-derived stem cells or neural stem/progenitor cells that have been shown to enhance functional outcome. Cells have been largely infused into the infarcted area but have also been applied systemically. Mechanistically, transplanted cells have been shown to contribute through the release of trophic factors including pro-angiogenic and neurogenic substances but also through direct integration into the neural network. These results have been confirmed in smaller clinical trials using both mesenchymal and neural stem cells.27–29 However, it is unknown (especially in human stroke patients) to which extent angiogenesis contributed to the overall functional recovery following cell therapy.
Conclusion
The ultimate role of angiogenesis-based treatment for stroke cannot be determined yet. Although there is strong evidence from pre-clinical research that restoration of the vascular anatomy and blood flow increases the neuronal and functional recovery following stroke, potential therapies require in-depth quality control and safety assessment. Today, research in the field of pro-angiogenic therapies does not focus solely on single growth factors anymore. The extended range of tested pro-angiogenic therapeutics including slow-release biomaterials, neutralization of inhibitory factors and cell transplants raises new hopes for future bench-to-bedside transitions and clinical trials. Further investigation is needed to identify the most promising therapy regarding safety, delivery method, dosage, timing of delivery, infarct size and location for ischemic stroke.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
The brain cross section in Figure 1 was modified from Servier Medical Art, licensed under a Creative Common Attribution 3.0 Generic License. ![]() .
.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
