Abstract
Noradrenaline (NA) has been shown to influence astrocytic and vascular functions related to brain homeostasis, metabolism, local blood flow, and blood-brain barrier permeability. In the current study, we investigate the possible associations that exist between NA-immunoreactive nerve terminals and astrocytes and intraparenchymal blood vessels in the rat frontoparietal cortex, both at the light and electron microscopic levels. As a second step, we sought to determine whether the NA innervation around intracortical microvessels arises from peripheral or central structures by means of injections of N-(2-chloroethyl-N-ethyl-2-bromobenzylamine) (DSP-4), a neurotoxin that specifically destroys NA neurons from the locus ceruleus. At the light microscopic level, 6.8% of all NA-immunoreactive nerve terminals in the frontoparietal cortex were associated with vascular walls, and this perivascular noradrenergic input, together with that of the cerebral cortex, almost completely disappeared after DSP-4 administration. When analyzed at the ultrastructural level in control rats, NA terminals in the neuropil had a mean surface area of 0.53 ± 0.03 µm2 and were rarely junctional (synaptic incidence close to 7%). Perivascular terminals (located within a 3-µm perimeter from the vessel basal lamina) counted at the electron microscopic level represented 8.8% of the total NA terminals in the cortical tissue. They were smaller (0.29 ± 0.01 µm2, P < 0.05) than their neuronal counterparts and were located, on average, 1.34 ± 0.08 µm away from intracortical blood vessels, which consisted mostly of capillaries (65%). None of the perivascular NA terminals engaged in junctional contacts with surrounding neuronal or vascular elements. The primary targets of both neuronal and perivascular NA nerve terminals consisted of dendrites, nerve terminals, astrocytes, and axons, whereas in the immediate vicinity (0.25 µm or less) of the microvessels, astrocytic processes represented the major target. The results of the current study show that penetrating arteries and intracortical microvessels receive a central NA input, albeit parasynaptic in its interaction, originating from the locus ceruleus. Particularly, they point to frequent appositions between both neuronal and perivascular NA terminals and astroglial cells and their processes. Such NA neuronal-glial and neuronal-glial-vascular associations could be of significance in the regulation of local metabolic and vascular functions under normal and pathologic situations.
The cerebral cortex receives a rich input from brainstem noradrenaline (NA) neurons located in the locus ceruleus (LC) (Mason and Fibigier, 1979). This cortical afferent pathway has been associated with multiple functions such as sleep-wake cycle and motor activity while in the hypothalamus, NA is involved in the regulation of temperature control as well as eating and drinking behaviors (Kramarcy et al., 1984; Lin et al., 1984; Hilakivi, 1987; Towell et al., 1989). Evidence also shows that NA and astrocytes work in tandem to modulate several cellular functions that include neurotransmission (Kimelberg, 1986), synthesis and release of neurotrophic factors (Schwartz and Mishler, 1990), and neuroprotection against ischemic insults (Blomqvist et al., 1985). Recently, NA has been shown to influence astrocytic properties such as intercellular communication, glycogenolysis, and glucose uptake (Giaume et al., 1991; Tsacopoulos and Magistretti, 1996).
A role for NA in vascular-related functions such as regulation of blood-brain barrier (BBB) permeability and CBF also has been suggested. Stimulation of the LC can increase or decrease BBB permeability, depending on the experimental paradigm (Raichle et al., 1975; Harik, 1986; Borges et al., 1994), and it also induces small decreases in local CBF (Raichle et al., 1975, De la Torre, 1977; Goadsby and Duckworth, 1989; Adachi et al., 1991) that can be blocked by α2-adrenoceptor antagonists (Goadsby et al., 1985). Conversely, pharmacologic manipulations that selectively destroy central noradrenergic neurotransmission (Yokote et al., 1986; Kobayashi et al., 1991) increase CBF-a response that can be reversed by addition of NA.
Overall, these observations suggest that NA might directly interact with brain astrocytes and microvasculature. In this respect, NA fibers coursing along or approaching small blood vessels have been observed in various deep brain nuclei and in the cerebral cortex (Edvinsson et al., 1973; Jones, 1982), a region where NA neuronal-astroglial interactions have been documented (Séguéla et al., 1990). Nerve endings (presumably noradrenergic) were found to be intimately associated with intraparenchymal blood vessels in the richly vascularized paraventricular nucleus of the hypothalamus (Swanson et al., 1977), and NA terminals in the vicinity of local microvessels have been incidentally reported in the medulla oblongata and cerebral cortex (Milner et al., 1989, Papadopoulos et al., 1989).
To establish a morphologic substrate for the effects of NA on brain astrocytes and microvessels, we performed a quantitative and morphometric analysis of the neuronal-glial and/or vascular associations of NA nerve terminals by immunocytochemistry at the light microscopic (LM) and electron microscopic (EM) levels. The central origin of the NA fibers associated with cortical microvessels was assessed with the neurotoxin N-(2-chloroethyl-N-ethyl-2-bromobenzylamine) (DSP-4), which is highly selective for NA terminals originating from the LC (Grzanna et al., 1989; Fritschy et al., 1990). Parts of these results have been presented as an abstract (Cohen et al., 1994).
MATERIALS AND METHODS
Tissue preparation
Adult male Sprague Dawley rats (Charles River, 250 g, n = 7) were deeply anesthetized with sodium pentobarbital (Somnotol, 65 mg/kg body weight intraperitoneally). They were perfused intracardially first at low speed (100 mL/minute) with 50 mL phosphate-buffered saline (0.1 mol/L, pH 7.4) and then rapidly (300 mL/minute) with 600 mL of 5% glutaraldehyde in 0.1 mol/L sodium phosphate buffer containing 0.2% of sodium metabisulfite (PBSM). After removal, the brains were immersion-fixed in the glutaraldehyde solution for 2 hours at room temperature. Brains processed for LM study were cryoprotected with 30% sucrose overnight, frozen in isopentane, and then coronal sections (30 or 60 µm) at the level of the frontoparietal cortex were obtained on a freezing microtome and collected in PBSM. Brains processed for EM analysis were immediately cut (thick sections of 60 µm) on an Oxford vibratome after the postfixation period and collected in PBSM. Before incubation with primary antibodies, the sections were reacted with 0.5% sodium borohydride and rinsed in PBSM, as previously documented (Séguéla et al., 1990). All experiments were approved by the Animal Ethics Committee based on the guidelines of the Canadian Council on Animal Care.
DSP-4 lesion of NA terminals
For noradrenergic denervation, adult rats (n = 4) received a first intraperitoneal injection (60 mg/kg; 7 to 10 days before perfusion) followed 3 to 4 days later by a second injection (50 mg/kg) of DSP-4 in sterile saline, whereas control rats (n = 5) received injections of saline alone (Berger et al., 1988). Both DSP-4-injected and control rats were perfused as just described and processed simultaneously for immunocytochemical examination at the LM level (see later).
Immunocytochemical study of NA
Free-floating sections were incubated overnight with an antiserum directed against a NA-glutaraldehyde-protein conjugate, diluted 1/5000 in PBSM and 1% normal goat serum. The production and specificity of this antibody has been previously described (Geffard et al., 1986; Mons and Geffard, 1987). The sections then were thoroughly rinsed in PBSM, incubated sequentially with a biotinylated goat anti-rabbit immunoglobulin G (1/100, Vector Labs, Burlingame, CA, U.S.A.), and the avidin-biotin-peroxidase complex (1/50, ABC, Vectastain Elite kit, Vector Labs). Rinses of 0.1 mol/L phosphate-buffered saline were carried out before and after each antibody incubation. The immunocytochemical product was revealed with 0.05% 3.3-(±) diaminobenzidine tetrahydrochloride (Sigma Chemical Co., St. Louis, MO, U.S.A.) containing 0.01% hydrogen peroxide in 0.1 mol/L Tris-HCl (6 minutes).
Only sections prepared for LM analysis were incubated in solutions supplemented with 0.1% Triton X-100. The 30-µm thick immunostained sections were mounted on gelatin-coated slides, dehydrated, and defatted before observation and photography under a Leitz Aristoplan light microscope. The 60-µm thick sections were used either for high-resolution LM (sections from cryoprotected brains) or ultrastructural (vibratomecut sections) analysis. All were postfixed with 2% osmium tetroxide in 0.4 mol/L sodium phosphate buffer containing 7% dextrose and then processed for flat embedding in Araldite 502 resin (for details, see Cohen et al., 1995). After polymerization, small blocks comprising the region of interest were trimmed and reembedded, and semithin (2-µm) sections comprising all six layers of the frontoparietal cortex or thin (90 to 120 nm, straw color) sections of the cortical upper layers were obtained using a Reichert ultramicrotome for LM and EM study. Semi-thin sections were observed and photographed under a Leitz Aristoplan light microscope. Thin sections were recovered on copper grids, double stained with uranyl acetate and lead citrate, and examined with a JEOL CX100II electron microscope at a working magnification of ×8000 to 14,000.
Analysis at the light microscopic level
Analysis of NA-immunoreactive nerve fibers associated with local microvessels within the frontoparietal cortex was done on photomicrographs of semithin sections. For this purpose, all NA-immunopositive nerve endings in 10 different cortical slices (n = 3 rats) were counted, including those (hereafter referred to as perivascular) that were directly apposed to blood vessel walls. The perivascular terminals then were expressed as a percentage of total NA terminals. The efficacy of the DSP-4 lesion also was verified on thick and semithin sections.
Ultrastructural analysis
Neuronal terminals.
All cortical NA terminals that were not perivascular (see later) were treated as neuronal. Their immediate microenvironment, surface area, and synaptic frequency were determined in single thin sections. The NA immunopositive axonal varicosities (n = 130) were randomly chosen (every fifth terminal encountered) and photographed. The cellular elements (n = 712) apposed to these NA nerve endings were identified (an average of six appositions were observed for each terminal) and classified as dendrites (dendritic trunks, shafts, or spines), nerve terminals, axons, myelinated axons, astrocytic cells, and nerve cell bodies. The prints also were used to measure the surface area of each varicosity using a Bioquant II analysis program and a MTI 65 camera, and the proportion of NA terminals engaged in synaptic contacts. Varicosities were considered to be synaptic when at least one of the juxtaposed membranes exhibited a straightening or thickening with an expanded intercellular space with or without a postsynaptic density (Peters et al., 1991). The synaptic incidence, which yields a precise estimate of the overall proportion of cortical NA terminals engaged in synaptic junction, was calculated using the extrapolated stereologic formula of Beaudet and Sotelo (1981).
Perivascular terminals.
The NA-immunoreactive terminals were defined as perivascular when located within a 3-µm perimeter from the basal lamina of a blood vessel, a distance corresponding to that previously reported for functional perivascular nerve fibers (Lee, 1981; Dodge et al., 1994; Chédotal et al., 1994). This interval was defined as the largest distance within which axon terminals might affect vascular functions either directly or indirectly through interactions with other neuronal or nonneuronal elements within the perivascular perimeter. The percentage of perivascular NA terminals in a given area of frontoparietal cortex was determined. For this purpose, from a total of 458 NA immunolabeled terminals observed directly on the EM screen, the population of perivascular terminals was identified with the help of scale bars to determine whether the terminal was within 3 µm from the vessel. They were then expressed as a percentage of the total amount of NA terminals counted within a given area. In addition, the first 125 perivascular NA terminals encountered were photographed and their distance from the vessel wall, surface area, synaptic incidence, and immediate microenvironment determined. The cellular elements (n = 643) contacted by NA perivascular terminals were divided as described earlier for the population of neuronal terminals. Particular attention was given to the micro-environment of nerve terminals located within the first 0.25 µm from the vessel basal lamina. The type of vessel associated with each terminal was identified directly on the EM screen and recorded for analysis. Capillaries were defined as small blood vessels (<10 µm) comprising a layer of endothelial cells within a basal lamina with or without associated pericytes. Arterioles were larger vessels (>10 µm) consisting of one or two layers of smooth muscle cells enclosed by the basal lamina (Peters et al., 1991).
RESULTS
Light Microscopy
The NA immunostaining in the frontoparietal cortex, as observed in 30-µm thick sections, corresponded to that previously described by 3H-NA radioautography, dopamine-β-hydroxylase- and NA-immunocytochemistry (Audet et al., 1988; Olschowska et al., 1981; Séguéla et al., 1990). Long varicose fibers, running vertically and some sideways, were scattered throughout the neocortex with an increased density in the most superficial layer (Fig. 1). Some fibers were seen to approach and run closely to intraparenchymal blood vessels. In semithin sections (Fig. 1B and C), the quantitative analysis showed that 412 of the 6074 immunoreactive NA terminals in the cerebral cortex were directly apposed to blood vessel walls, corresponding to 6.8% of all cortical NA nerve terminals.
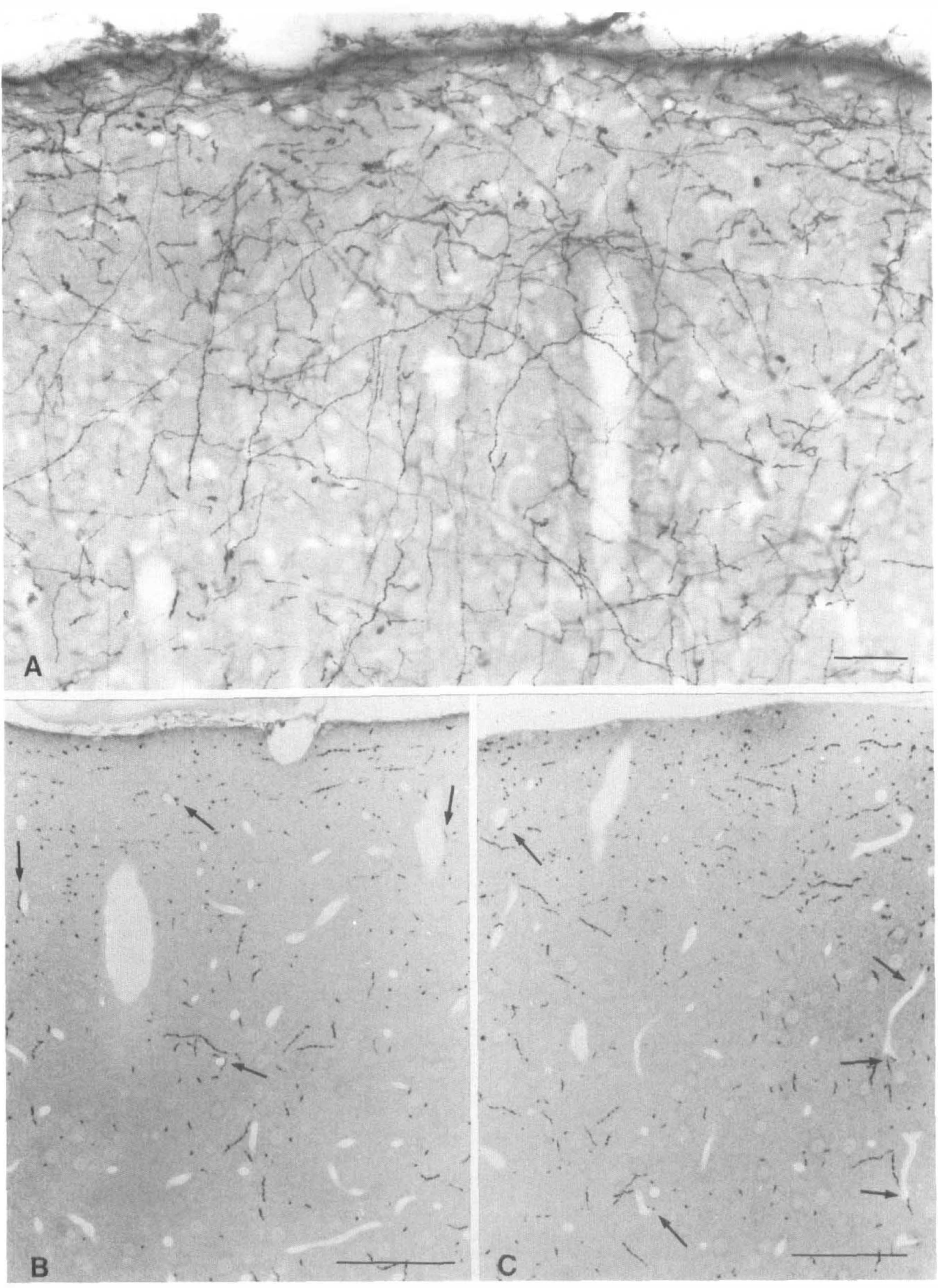
Photomicrographs depicting the distribution of NA-immunoreactive nerve fibers in the frontoparietal cortex in 40-µm thick sections
Photomicrographs of thick
Electron Microscopy
Neuronal terminals.
At the ultrastructural level, the immunostained NA profiles were found to correspond primarily to round or ovoid varicosities as well as to longitudinal axon fibers. They contained numerous small synaptic and a few dense core vesicles, with one or more mitochondria (Fig. 3). The immunolabeled nerve endings were juxtaposed mostly on dendritic processes, unlabeled axonal varicosities, astroglial processes, less so on axons, and only rarely on cell soma and myelinated axons (Fig. 3; Table 1). Overall, neuronal NA terminals had a mean surface area of 0.53 ± 0.03 µm2 (calculated diameter of about 0.82 µm) and rarely exhibited synaptic junctions in single thin sections, exclusively asymmetrical and with dendritic elements (Table 1). When extrapolated to whole volume, a synaptic incidence of about 7% was calculated.
Morphological characteristics and microenvironment of neuronal and perivascular NA-immunoreactive nerve terminals in the rat frontoparietal cortex
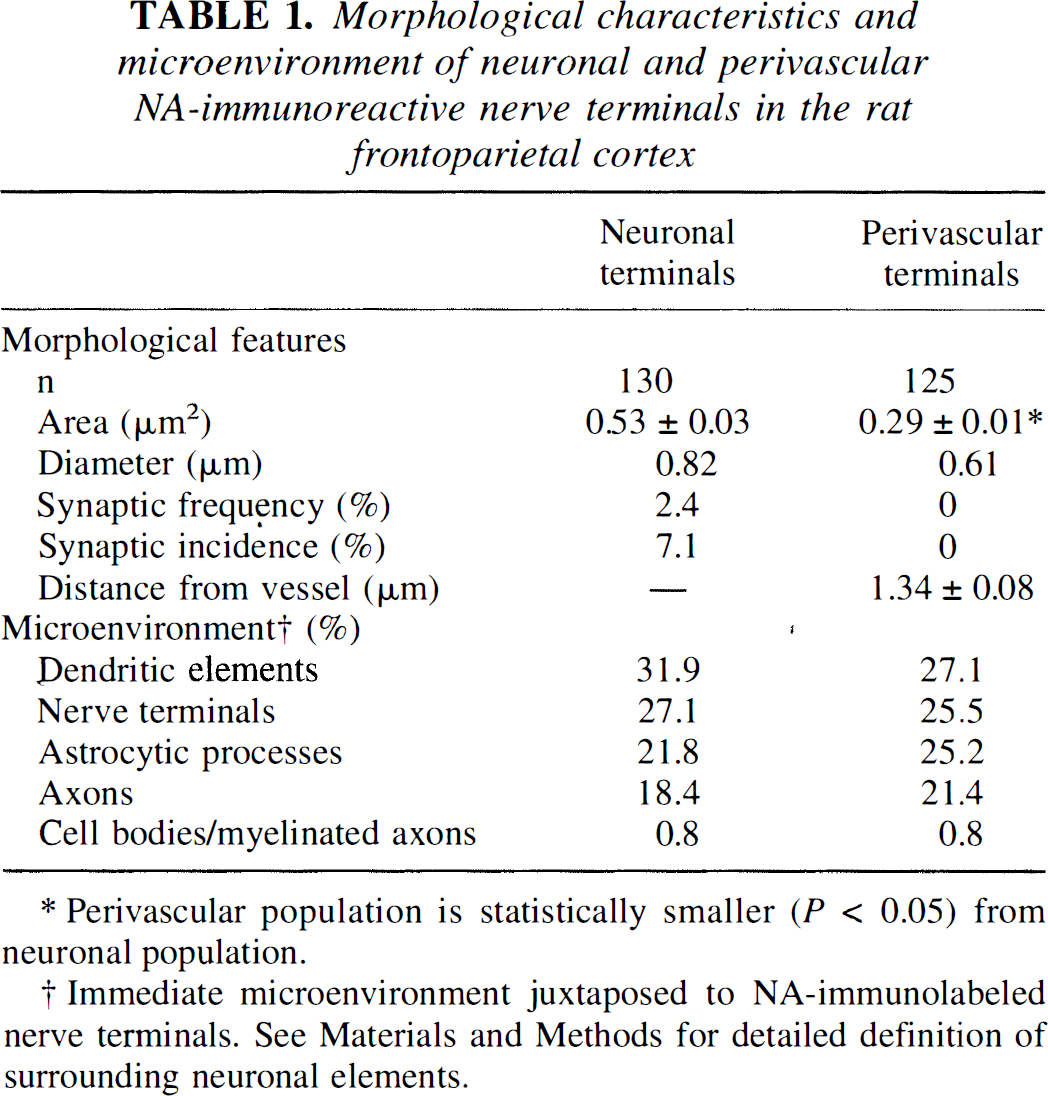
Perivascular population is statistically smaller (P < 0.05) from neuronal population.
Immediate microenvironment juxtaposed to NA-immunolabeled nerve terminals. See Materials and Methods for detailed definition of surrounding neuronal elements.
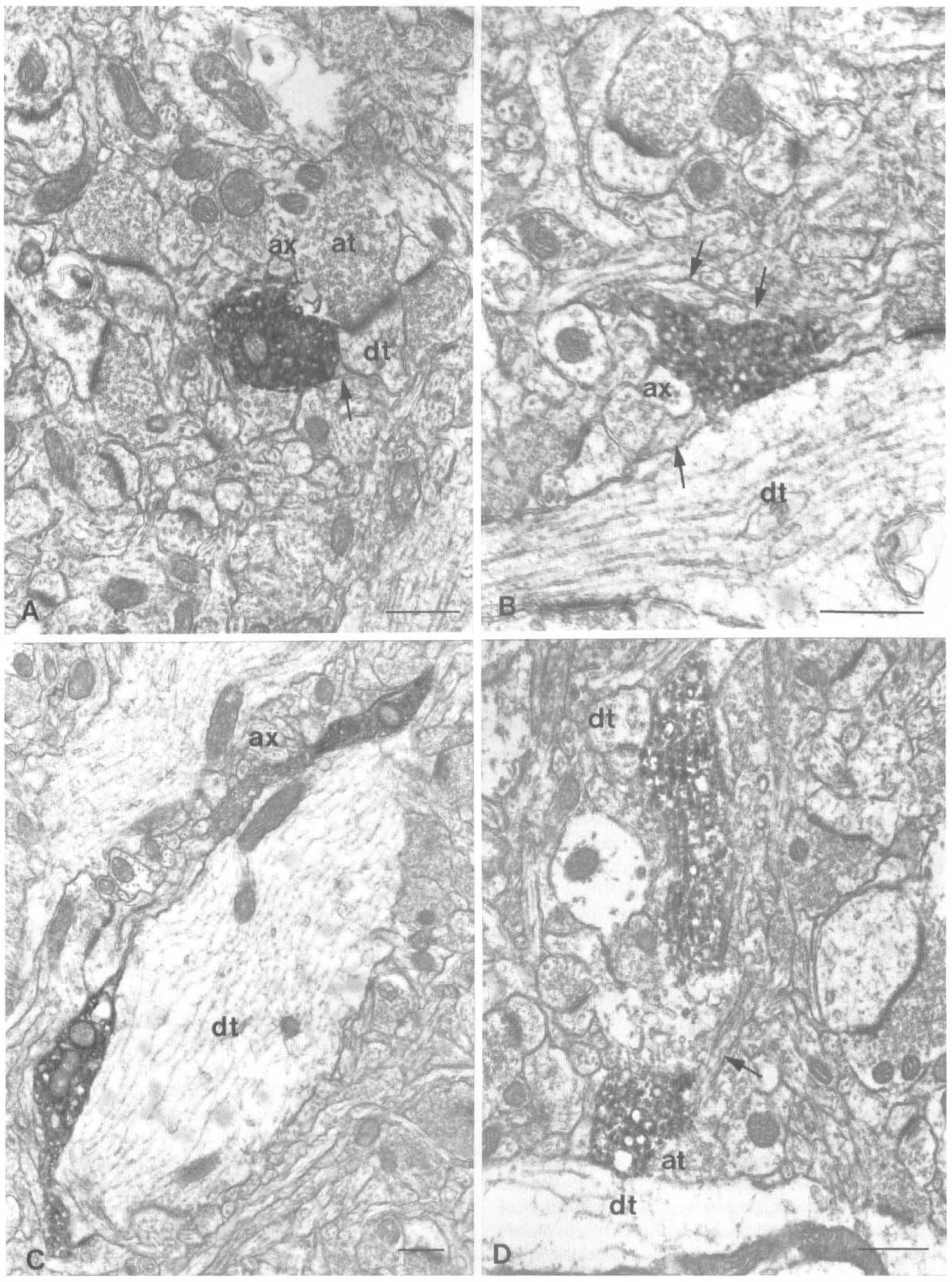
Electron micrographs illustrating the general morphologic features of neuronal NA-immunostained axon terminals in the frontoparietal cortex and their immediate microenvironment. Terminals occasionally contain dense-core vesicles and are primarily apposed to dendrites (dt), axon terminals (at), axons (ax), and astrocytes (arrows). Scale bars = 0.5 µm.
Perivascular terminals.
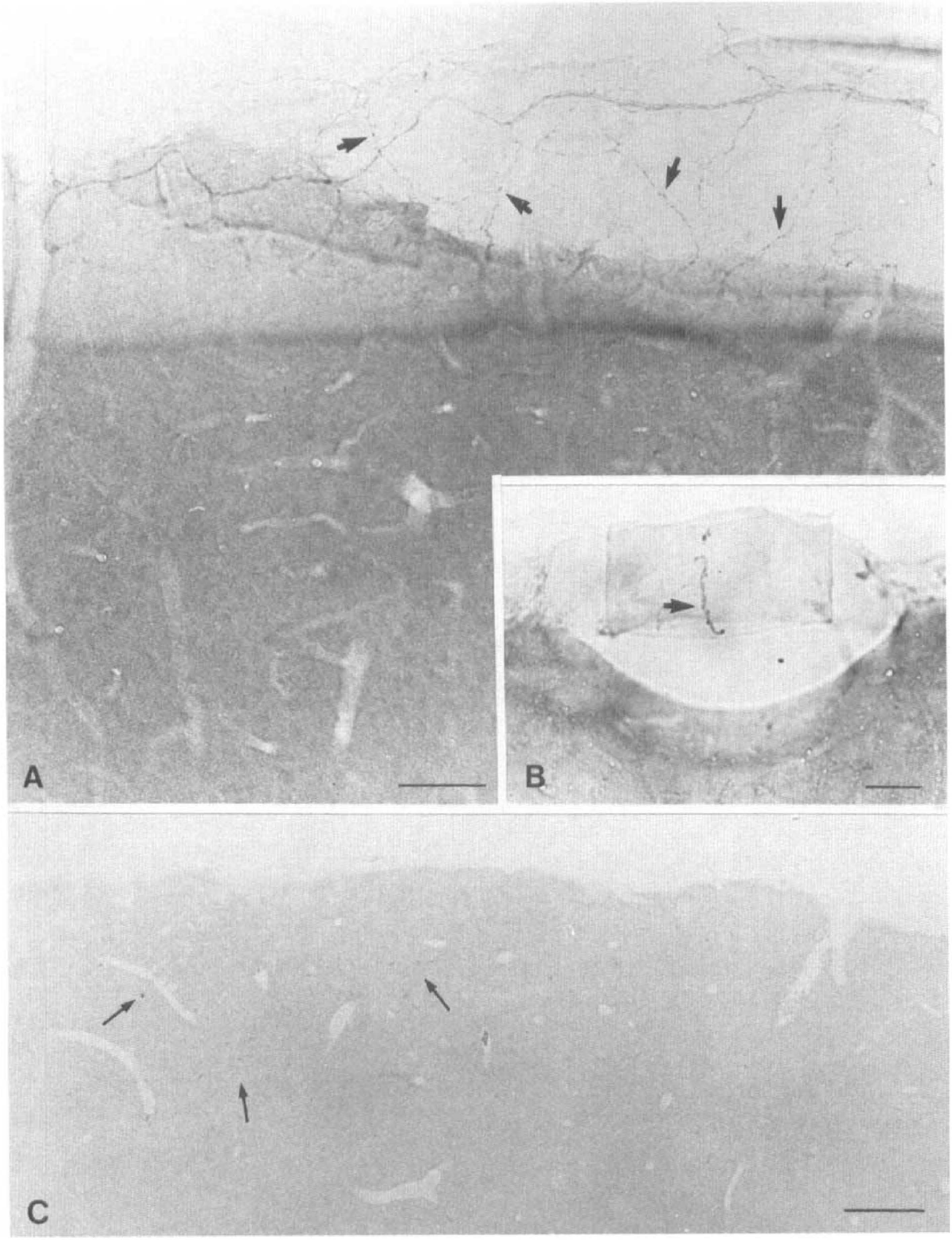
A total of 458 NA-immunoreactive terminals were counted in the frontoparietal cortex, and 8.8% (n = 44) of them were found to be located within 3 µm from microvessels (Fig. 4). When studied in more detail on a population of 125 perivascular NA terminals, 23 terminals were located in the immediate vicinity (≤0.25 µm) of the vessel basement membrane (Fig. 4 through Fig. 6). The remaining terminals were distributed uniformly throughout the 3-µm perimeter around vessel walls (Fig. 6). Perivascular terminals were similar in appearance and shape to their neuronal congeners, except that they corresponded almost exclusively to axonal varicosities (Figs. 4 and 5) and seldom to axon fibers. The microenvironment of the perivascular terminals, like that of the neuronal population, was composed mostly of dendrites, nerve terminals, and astrocytes (Table 1). In the closest interval (0.25 µm or less) around the vessel wall, however, 75% of the terminals (17 of 23) abutted on a perivascular astrocyte, whereas of all targets, the astrocytes (either perivascular or in the neuropil) represented the primary cellular elements of apposition (32.7%) followed by nerve terminals (25.2%), axons (21.5%), and dendrites (19.6%). Most of perivascular associations were on capillaries (∼65%) compared with small arterioles (∼35%). Interruptions in the perivascular astrocytic leaflets allowing NA terminals to directly touch the vascular basal lamina were rarely observed. None of the perivascular terminals were seen to engage in synaptic contact with any of the apposed cellular elements within the perivascular perimeter. The perivascular terminals were located on the average 1.34 ± 0.08 µm from intracortical blood vessel walls and were significantly smaller (mean surface area of 0.29 ± 0.01 µm2, P < 0.05) than their neuronal counterparts (Table 1).
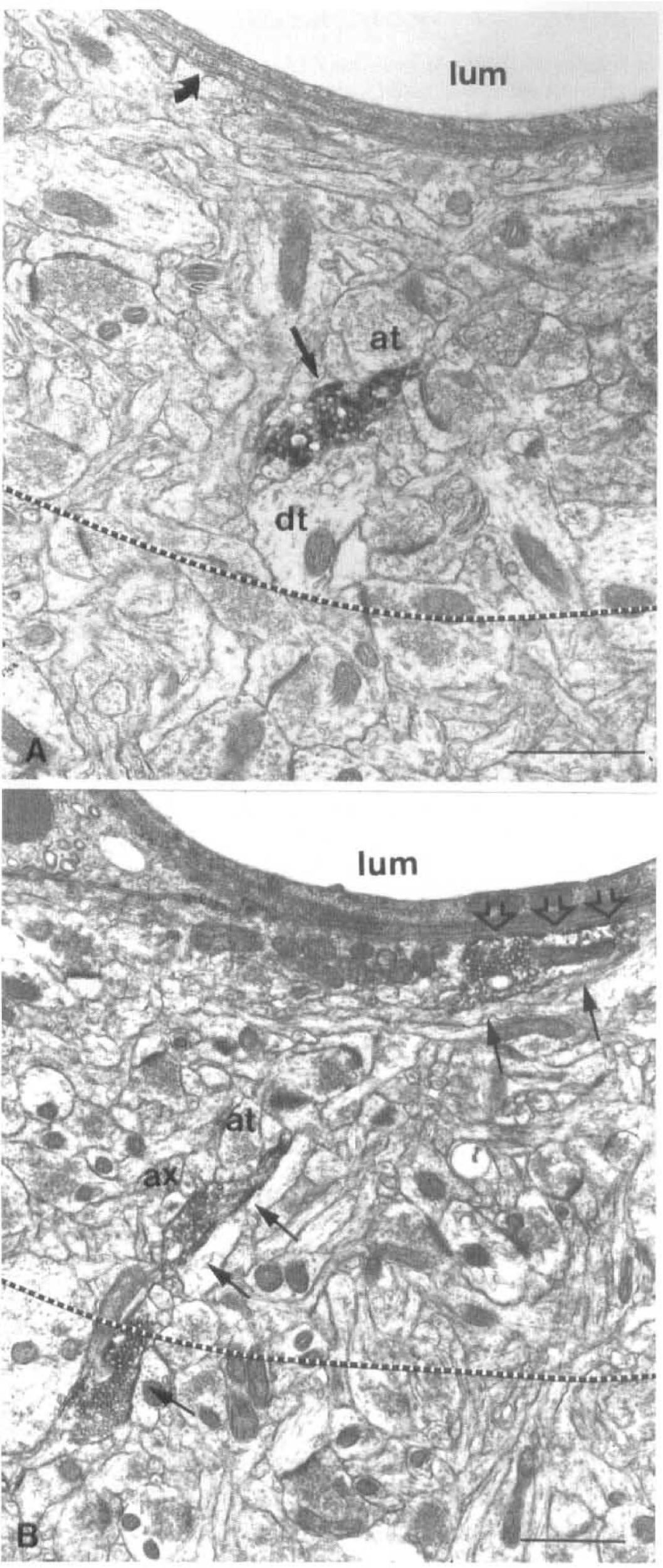
Electron micrographs of NA perivascular terminals in the rat frontoparietal cortex. The 3-µm perimeter around the vessel basal lamina is delineated by the dotted line.
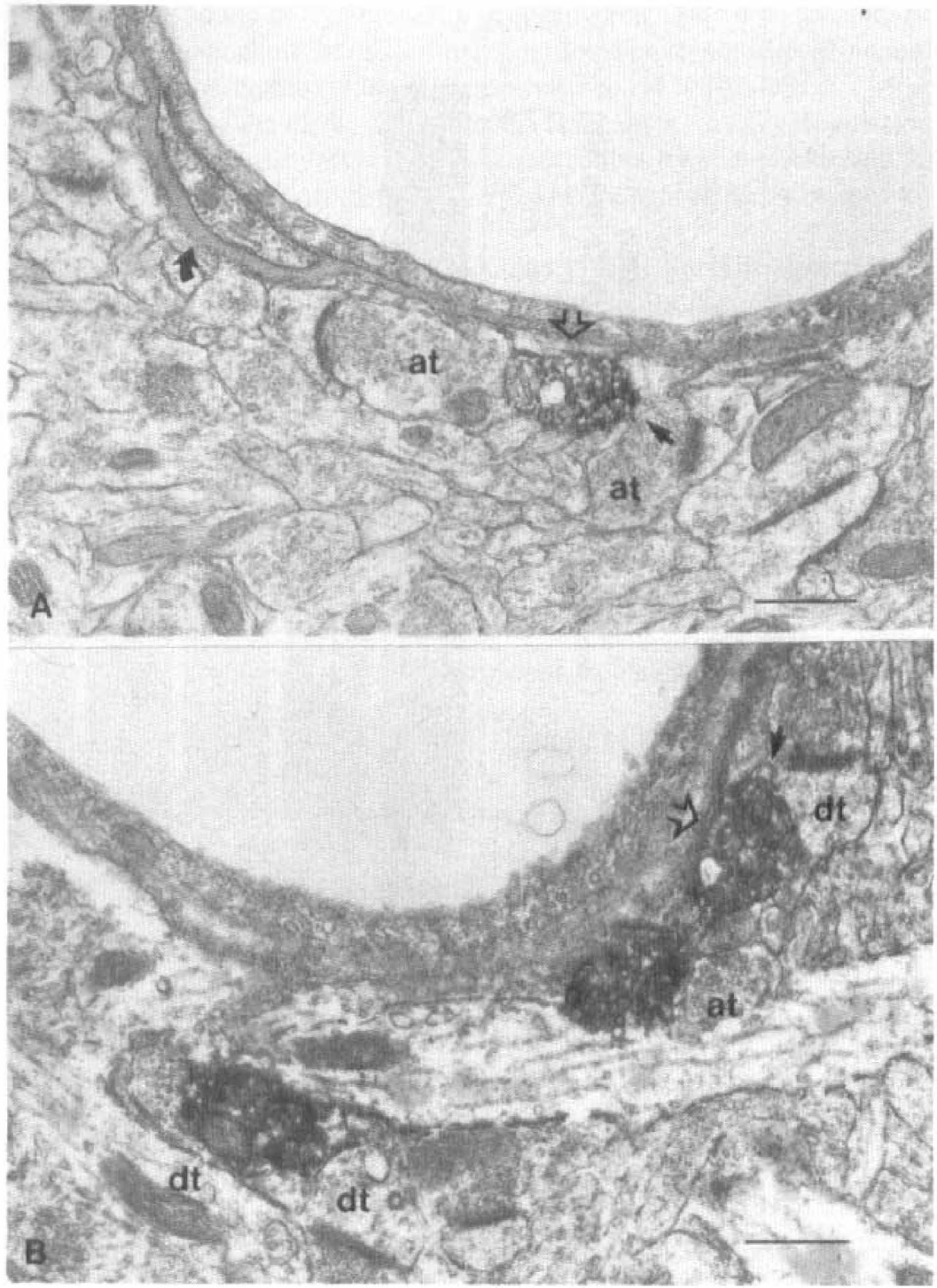
Electron micrographs of NA terminals associated with cortical microvessels. NA-labeled terminals are separated from the basal lamina of a capillary (A) or microarteriole (B) only by the perivascular astrocytic leaflet (open arrows). The curved arrow in (A) points to a small pericyte embedded within the basal lamina. at, axon terminal; dt, dendrite; solid arrows, astrocyte. Scale Bar = 0.5 µm.
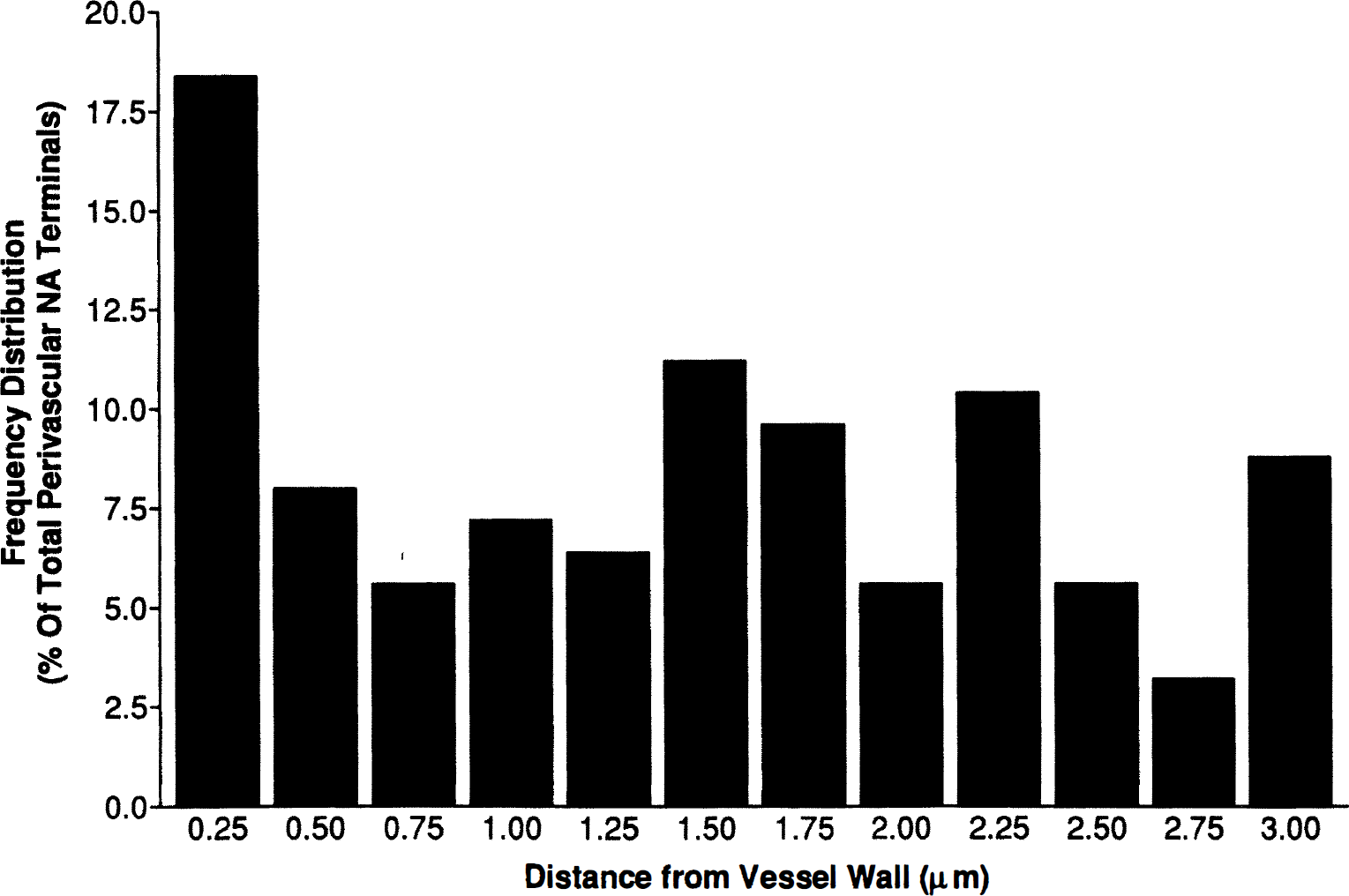
Histogram of the distribution of NA-immunoreactive terminals within the 3-µm perimeter around intracortical microvessels. The distance from the blood vessels is separated into 0.25-µm intervals, and percentages correspond to the number of terminals found within each interval, compared with the total number (n = 125) of perivascular terminals.
DISCUSSION
The current study examines the relations between NA-containing neurons and intracortical astrocytes and microvessels. Our results show that the LC is the major, if not exclusive, source of NA fibers to the cortical neuropil but also to the local microvascular bed. Furthermore, the results show that NA terminals in the cerebral cortex frequently target glial processes and, more frequently so, the perivascular astroglial leaflets.
General features
The LM and ultrastructural analyses of NA immunostained elements in the frontoparietal cortex yielded results consistent with those of previous radioautographic and immunocytochemical studies on cortical NA innervation (Olschowka et al., 1981; Audet et al., 1988; Papadopoulos et al., 1989; Séguéla et al., 1990). The NA innervation of the cerebral cortex was predominantly nonjunctional, and perivascular terminals never established synaptic junctions with vascular, glial, or other neuronal elements. They were smaller compared with neuronal terminals, perhaps because of the fact that the perivascular elements or blood vessels themselves were their final targets. The frequent incidence of axo-axonic appositions for both neuronal and perivascular cortical terminals is fully compatible with the capacity of NA to modulate other neurotransmitters release within the cerebral cortex (Beani et al., 1986; Pittaluga et al., 1990; Raiteri et al., 1990), an aspect that also may be highly relevant to local regulation of brain perfusion (Raszkiewicz et al., 1992).
Perivascular NA terminals
Selective destruction of NA neurons from the LC with DSP-4 (Grzanna et al., 1989; Fritschy et al., 1990) resulted in a massive denervation of not only the cortical neuropil but also the penetrating arteries and intracortical microvessels. This observation suggests that the noradrenergic input to the cortical microcirculation, like that of the hypothalamus (Swanson et al., 1977), is of central origin. This conclusion concurs with the reported “denervation supersensitivity” of cortical microvascular adrenoceptors in rats after chemical lesions of the LC (Kalaria et al., 1989) and in patients with Alzheimer's disease, a condition known to be accompanied by degeneration of the NA system (Kalaria and Harik, 1989). Together with the presence of functional high-affinity α- and β-adrenoceptors in brain microvessels, smooth muscle, and endothelial cells in culture (Nathanson et al., 1979; Wroblewska et al., 1984; Bacic et al., 1992), these observations strongly suggest that intracerebral vessels can respond to neurally released NA. Interestingly, DSP-4 treatment did not affect the NA innervation of the pial vessels, which is consistent with the superior cervical ganglion origin of their noradrenergic innervation (Duverger et al., 1987). These results suggest that distinct neurogenic mechanisms with sympathetic and central origins provide a NA input to extracerebral and intracerebral blood vessels, respectively.
However, the neurovascular associations between NA terminals and the cortical microvessels were not as frequent and/or as intimate as those reported previously for basalocortical acetylcholine and brain stem serotonin (5-hydroxytryptamine) cortical projections (Chédotal et al., 1994; Vaucher and Hamel, 1995; Cohen et al., 1995), two systems believed to affect local CBF partly through a direct action on the microvascular bed (Sato and Sato, 1992; Cohen et al., 1996). Cortical neurovascular NA associations compared much better with those of acetylcholine (Vaucher and Hamel, 1995) and 5-hydroxytryptamine (Cohen et al., 1995) in brain areas where only weak or no changes in CBF have been found. This may indicate that NA regulation of CBF, at least in the cerebral cortex, is not the primary role of these neurovascular or neuronal-glial-vascular interactions. Such contention is supported by the relatively minor changes that have consistently been observed in cortical perfusion after manipulations of the NA system (Adachi et al., 1991; Kobayashi et al., 1991) and may point to a role of perivascular NA terminals in BBB and astrocytic functions.
The NA terminals preferentially targeted capillaries compared with microarterioles. In this respect, LC stimulation has been shown to produce significant changes in the transport of water and solutes across the BBB (Raichle et al., 1975; Sarmento et al., 1994; Borges et al., 1994), an effect consistent with the presence of functional adrenoceptors on brain endothelial cells (Bacic et al., 1992). The possibility that the primary role of the perivascular NA terminals is related to regulation of the BBB permeability is of interest since dysfunctions at this level, including alterations in microvascular adrenoceptors (Kalaria and Harik, 1989), are thought to contribute to the overall pathology of Alzheimer's disease (see Kalaria, 1992; De la Torre and Mussivand, 1993).
Association of NA terminals with astrocytes
The astroglial cells constituted the third most abundant targets for cortical NA-immunolabeled terminals (Table 1; Séguéla et. al., 1990), a percentage that we found to be slightly higher for the perivascular NA nerve terminals and even more so for those located close to vessels. Indeed, for NA terminals located in the immediate vicinity (0.25 µm or less) of blood vessels, the astrocytic processes, whether perivascular or in the surrounding neuropil, represented their major cellular target (Table 1; see Results). These observations strongly suggest that the astrocytic processes are likely to be the direct cellular target of perivascular NA terminals. Such a statement is supported by a recent, although nonquantitative, ultra-structural study on the relations of NA terminals with nonneuronal elements in the rat visual cortex (Paspalas and Papadopoulos, 1996). Together, our studies and theirs strongly underline the postulated contribution of brain astrocytes as integral components of brain neuronal-glial or vascular interactions within the cerebral cortex (Barres, 1991; Hertz, 1992; Cancilla et al., 1993). The reported ability of NA to modulate production and secretion of nerve growth factor from astroglial cells (Furukawa et al., 1987; Schwartz and Mishler, 1990) and its possible intrinsic growth properties on brain microvessels, similar to those reported on smooth muscle cells of peripheral origin (Blaes and Boissel, 1983; Chen et al., 1995), also are in line with a localization of NA terminals in the vicinity of cortical microvessels and astrocytes.
Functional adrenergic receptors of various types are widely expressed in brain astrocytes (Salm and McCarty, 1992; Hösli and Hösli, 1993), and, in the rat visual cortex, astrocytic processes exhibiting immunoreactivity to the β-adrenergic receptor subtype often were interposed between NA nerve endings and the vascular basal lamina (Aoki, 1992). It is thus likely that NA, through parasynaptic interactions with astroglial cellular elements, could regulate astrocytic functions whether related to BBB properties, intercellular communication along the astrocytic syncytium, and metabolic or ionic homeostasis (Giaume et al., 1991; Hertz, 1992; Laterra et al., 1994; Montgomery, 1994; Tsacopoulos and Magistretti, 1996). More specifically, the frequent associations between NA terminals and astrocytes may provide the morphologic basis for both the β-adrenoceptor-mediated, rapid, and long-term control of glycogen levels induced by NA in cerebral cortical astrocytes (Sorg and Magistretti, 1991, 1992). However, astrocytes also have been shown to reorganize their syncytial network, a mechanism regulated by NA (Giaume et al., 1991) to promote neuronal survival after ischemic injury (Hossain et al., 1994). In this regard, lesions of the LC increase neuronal necrosis after ischemia (Blomqvist et al., 1985), whereas stimulation of NA neurons decreases the detrimental effects of ischemia (Gustafson et al., 1990). It is thus tempting to speculate that NA neurotransmission would not only regulate astrocytic functions under normal conditions, but that NA neuronal-glial interactions could be of primary importance in protecting the brain from damage caused by ischemic insults. In conclusion, these results emphasize the current view that neurotransmitters are important mediators of astrocytic functions, whether related to their actions in neurotransmission, homeostasis, or vascular regulation.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Dr. Elena Galea for helpful discussions, Helmut Bernhard and Markus Arts for expert photographic assistance, and Linda Michel for preparation of the manuscript. The authors also Dr. Michel Geffard, Laboratoire d'Immunologie et Pathologie, Université de Bordeaux II, France, for the generous supply of the NA-antiserum.
