Abstract
Our previous studies demonstrated that axonal remodeling of the corticospinal tract (CST) contributes to neurological recovery after stroke in rodents. The present study employed a novel non-invasive peripheral approach, to over-express tPA in denervated spinal motor neurons via recombinant adeno-associated virus (AAV) intramuscular injection in transgenic mice subjected to permanent middle cerebral artery occlusion (MCAo), in which the CST axons are specifically and completely labeled with yellow fluorescent protein (YFP). One day after surgery, mice were randomly selected to receive saline, AAV5-RFP, or tPA (1 × 1010 viral particles) injected into the stroke-impaired forelimb muscles (n = 10/group). Functional deficits and recovery were monitored with foot-fault and single pellet reaching tests. At day 28 after MCAo, mice received intramuscular injection of PRV-614-mRFP (1.52 × 107 pfu) as above, and were euthanized four days later. Compared with saline or AAV-RFP-treated mice, AAV-tPA significantly enhanced behavioral recovery (p < 0.01, both tests), as well as increased CST axonal density in the denervated gray matter of the cervical cord (p < 0.001), and RFP-positive pyramidal neurons in both ipsilesional and contralesional cortices (p < 0.001). Behavioral outcomes were significantly correlated to neural remodeling (p < 0.05). Our results provide a fundamental basis for the development of therapeutic approaches aimed at promoting corticospinal innervation for stroke treatment.
Keywords
Introduction
One of the most common impairments after stroke is hemiparesis of the contralesional side to the affected cerebral hemisphere. Although spontaneous and rehabilitation-improved functional recovery is often present after stroke, the recovery is generally incomplete. Of stroke survivors, 50% have some hemiparesis, 30% are unable to walk without assistance, 26% are dependent in activities of daily living at six months after stroke, and approximately 15% to 30% are left permanently disabled. 1 Unfortunately, other than thrombolysis by recombinant tissue plasminogen activator (tPA) and mechanical thrombectomy for acute ischemic stroke patients, there are no efficacious therapies available to maximize neurological recovery during their convalescence. Thus, there is a compelling need to develop therapeutics to be employed for the vast majority of patients during the subacute and chronic phases after stroke.
Because hemiparesis after stroke is a consequence of the loss or interruption of motor signals from the motor cortex to the spinal motoneurons, reestablishment of corticospinal innervation provides a physical substrate for motor functional recovery. The corticospinal tract (CST), the long axons of the cortical pyramidal neurons extending to the spinal cord, is the only direct descending pathway and the primary transmission tract innervating the spinal motoneurons from the sensorimotor cortex, and thus forms the neuroanatomical basis for brain controlled voluntary movements of the peripheral muscles. 2 Clinical studies demonstrated that the extent of functional disability and the potential for functional recovery is dependent on the CST integrity in stroke patients.3,4 Our previous studies have demonstrated that axonal remodeling of the CST in the spinal cord contributes to neurological recovery after stroke in rodents.5–7 Therefore, we hypothesized that enhancing CST axonal remodeling within the stroke-impaired side of the spinal cord will promote neurological recovery.
In the present study, we designed a non-invasive peripheral approach, intramuscular injection of recombinant adeno-associated virus (AAV)-mediated tPA gene delivery into the stroke-impaired muscles. Here, tPA is not used as a fibrinolytic agent in the circulation system, but as a neurorestorative agent in the central nervous system to stimulate axonal remodeling. With this procedure, the AAV vectors were retrogradely transported into the spinal motor neurons in the denervated spinal gray matter along their axons innervating these muscles. We employed a transgenic mouse line, in which the CST axons are specifically and completely labeled with yellow fluorescent protein (YFP), 8 combined with a retrograde trans-synaptic tracer, pseudorabies virus (PRV)-monomeric red fluorescent protein (RFP), to investigate the therapeutic effects of tPA in adult mice subjected to permanent unilateral middle cerebral artery occlusion (MCAo).
Materials and methods
All experimental procedures were approved by the Institutional Animal Care and Use Committee (No. 1636) and the Institutional Recombinant DNA and Biosafety Committee (No. 2007.02 and 2017.03) of Henry Ford Hospital and complied with guidelines set forth in the National Institute of Health’s Guide for the Care and Use of Laboratory Animals. The facilities at the Henry Ford Hospital are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) and in compliance with the Standards for Humane Care and Use of Laboratory Animals of the Office of Laboratory Animal Welfare (HFHS policy No. 39 and No. 40). The manuscript is in compliance with the Animal Research Reporting In Vivo Experiments (ARRIVE) guidelines. The treatment assignment was blinded to investigators who participated in behavioral tests and endpoint data collections.
Animals
CST-YFP mice were generated by our in-house breeding colony using two transgenic mouse strains of B6.Cg-Tg(Thy1-EYFP)15Jrs/J and B6.129-Emx1™1(cre)Krj/J obtained from Jackson Laboratories (Bar Harbor, ME). In the Thy1-STOP-YFP mice, YFP expression is driven by neuron-specific regulatory elements of the Thy1 promoter after Cre-mediated excision of STOP sequences. In the Emx-Cre mice, Cre recombinase is specifically expressed in the embryonic forebrain, the area of origin of the CST. Therefore, in CST-YFP mice generated by mating Thy1-STOP-YFP with Emx-Cre strain, YFP expression is limited to the forebrain and CST. 8
Ischemic stroke model
For the experimental stroke model, 30 adult male CST-YFP mice (two to three months-old, body weight 25–30 g) were subjected to permanent right MCAo using a method of intraluminal suture vascular occlusion 9 with some modifications. Briefly, under isoflurane anesthesia, the right external carotid artery (ECA) was ligated at the distal part, and an 8–0 surgical nylon suture with an expanded silicone tip (1.5 mm in length, 0.20–0.25 mm in diameter) tip was advanced through an incision at the proximal part of the right external carotid artery into the lumen of the internal carotid artery, to block the origin of the MCA. During the MCAo surgery and for the first 72 h after MCAo, the mice were placed on a 37°C heating pad. All mice survived until the endpoint and were included for data collection.
Intramuscular AAV injection
To validate the efficiency of AAV5 mediating transduction into the spinal cord after intramuscular delivery, 1 × 1010 viral particles in 5 µl of AAV5 carrying RFP per mouse (n = 6; 1.78 × 1013 VG/ml, purchased from SignaGen Laboratories, Gaithersburg, MD) or tPA (n = 3; 1.06 × 1013 VG/ml, SignaGen) under the control of the neuronal specific Synapsin promoter or 5 µl of saline (n = 3), divided into multiple injections of 1 µl, were injected with a 10-µl Hamilton syringe through a skin incision into the wrist extensor and flexor muscles of the left forelimb in WT male C57BL/6 mice (two month-old, body weight 20–25 g; purchased from the Jackson Laboratory) under isoflurane anesthesia. WT mice were euthanized at day 21 after AAV intramuscular injection. Three mice received AAV-RFP and were perfused with saline, followed by 4% paraformaldehyde (PFA). The cervical cord was removed and immersed in 4% PFA overnight. The cervical enlargement (C4–C7) was processed for longitudinal consecutive vibratome sections, which were scanned with an Olympus laser-scanning confocal imaging system mounted onto an Olympus Bax 40 microscope (FV1200; Center Valley, PA) to digitize RFP-positive spinal motor neurons. The cervical cord from the remaining WT mice that received AAV-RFP or AAV-tPA was removed and processed for analysis of mRNA. Total RNAs were extracted and DNase digested using an RNeasy mini kit (Qiagen, Santa Clarita, CA). RNA was reverse transcribed with SuperScript® III Reverse Transcriptase (Life Technologies, Carlsbad, CA), and amplified by QuantiTect SYBR Green PCR kit (Qiagen) using custom primers (Sequence of primers for tPA: forward 5ʹ- GAGGCCTTGTCTCCTTTCTATTC-3ʹ, reverse 5ʹ- GTCGGTGACTGTTCTGTTAAGT-3ʹ; RFP: forward 5ʹ- CCACTACACCTGCGACTTCA-3ʹ, reverse 5ʹ- CTCCACCACGGTGTAGTCCT-3ʹ; Invitrogen, Carlsbad, CA). Quantitative RT-PCR was performed on an ABI ViiA7 Real-Time PCR system (Applied Biosystems, Foster City, CA). Analysis of gene expression was carried out by the 2−ΔΔCt method. Additional WT mice subjected to MCAo were treated with saline, AAV-RFP or AAV-tPA administered intramuscularly as above, respectively (n = 3/group). The cervical (C4–7) and lumbar (L1–5) cord were collected at 21 days after injection. tPA expression was examined using Western blot with antibody against tPA (SC515562, Santa Cruz Biotechnology; Santa Cruz, CA) following the standard protocol (Molecular Clone, Edition II). β-Actin was used as the internal control. The densities of bands were analyzed using Image J.
CST-YFP mice subjected to MCAo were randomly assigned to three groups based on body weight using a random number table generated by Microsoft Excel 2016 at one day after surgery, to receive intramuscular injections of AAV5-RFP, or AAV-tPA as above, or 5 µl of saline, respectively (n = 10/group). The treatment assignment was blinded to investigators who participated in behavioral tests and endpoint data collections.
Behavioral tests
To evaluate the motor functional deficits and recovery, a series of behavioral tests were performed at three days after MCAo and weekly thereafter. For non-skilled motor performance, a foot-fault test was used to assess the accuracy of forepaw placement on a non-equidistant grid as the percentage of foot faults of the stroke-impaired forepaw to total steps. 10 For skilled voluntary control of the stroke-impaired forepaw, a single-pellet reaching test was employed to characterize the ability of reaching and retrieving 20-mg food pellets (Bio-Serv Inc., Frenchtown, NJ) through a vertical slot on the front wall of a plexiglas box.7,11 Animals were pre-trained for two weeks before surgery. The outcome was evaluated with success score (A score of 1: extracts the pellet and brings it to the mouth; 0.5: drops the pellet inside the box before eating; 0: knocks the pellet off the shelf).
Retrograde PRV tracing
To confirm the synaptic innervation between the motor cortex and the stroke-impaired peripheral target tissues, a 10 µl total volume of PRV-614-mRFP (1.52 × 109 pfu/ml; Gift from Dr. Lynn Enquist, Princeton University, Princeton, NJ), a retrograde trans-synaptic neural tracer, 12 was injected into the left stroke-impaired forelimb muscles at day 28 after MCAo when the behavioral tests were done. Then the animals survived for an additional 94–96 h in a Biosafety Level-2 room to allow for virus transport from the axonal terminals in the muscles to the pyramidal neurons in the cortex. 6
Tissue preparation and data collection
CST-YFP mice were euthanized by transcardial perfusion with saline, followed by 4% PFA at 32 days after MCAo. The entire brain and the cervical cord were immersed in 4% PFA overnight. The forebrain, the medulla of the hindbrain, and the cervical enlargement (C4–C7) were cut into 100-µm thick coronal sections using a vibratome machine. A series of forebrain sections in 900-µm intervals were stained with hematoxylin and eosin (H&E) for infarct volume measurement. Lesion volume was measured using NIH imaging software (ImageJ) and presented as a volume percentage of the lesion area compared with the contralesional hemisphere. An Olympus laser-scanning confocal imaging system was used to digitize the RFP-positive pyramidal neurons on another series of forebrain sections with 400-µm intervals and the YFP-positive CST axons on the medulla hindbrain sections and the cervical cord sections. The ImageJ software was used to measure the number of RFP-positive pyramidal neurons in the bilateral cerebral cortices on each one of five forebrain sections (total six sections for each animal), number of YFP-positive CST axons in the bilateral pyramidal tracts on three medulla sections, and density of YFP-positive CST axons in the bilateral sides of the gray matter. Numbers of the RFP-positive neurons from a series forebrain sections in 0.5-mm interval are presented as a sum for each hemisphere. Numbers of the YFP-positive CST axons in each side of the pyramidal tracts were counted as the average of three medulla sections. Density of the YFP-positive CST axons in each side of the gray matter was measured by fluorescent area on 30 consecutive cervical cord sections. To avoid potential inter-section variation in fluorescent measurements, the percentage of CST density in the stroke-impaired side to the contralesional side on the same sections was calculated, and the average number in each animal was used as an index for CST axonal remodeling after stroke subjected to different treatment.
Statistics
Data represent mean ± SD. The Global test using Generalize Estimating Equation (GEE) 13 was performed to test the treatment effect on functional recovery measured from both skilled (pellet reaching test) and non-skilled (foot-fault test) behavioral task at each time point with a focus of functional recovery at day 28 (as primary endpoint). The analysis started testing the overall group effect, followed the subgroup comparisons using CONTRAST statement in Software of Analytics Software & Solutions (SAS). Any sub-group analysis was considered as exploratory if there is no overall group effect at the 0.05 level. The Global test of multiple correlated outcomes is more efficient test than a single outcome, when the group effects are consistent on all the outcomes (e.g. the positive correlation). The one-way analysis of variance (ANOVA) was used to test the treatment effect on neuronal remodeling in the spinal cord and brain. To test the relationship between behavioral outcome and neuronal reorganization, the correlation coefficients between the performance scores of the stroke-impaired forepaw and the index of axonal density in the denervated cervical gray matter or RFP-positive cell numbers in the bilateral cortices were calculated by Pearson’s correlation coefficients after MCAo.
Results
Verification of retrograde transport of AAV vectors after intramuscular injection
To confirm whether AAV5 is capable of mediating transduction of the spinal neurons after intramuscular delivery in adult mice, AAV5 carrying RFP or tPA was injected into left forelimb muscles in normal wild-type mice. Three weeks after AAV injection, the mice were euthanized. As shown in Figure 1, 12–15 spinal motoneurons were visualized in the left side of the ventral horn of the cervical cord with RFP expression on longitudinal spinal cord sections (a; n = 3), and transcription of RFP mRNA was analyzed by quantitative RT-PCR (b; n = 3, p < 0.001). In addition, intramuscular AAV-tPA injection induced a 66% increase of tPA mRNA level in the cervical cord (c; n = 3, p < 0.001). tPA protein expression was also examined using Western blot in mice subjected to MCAo. Compared to saline or AAV-RFP-treated mice, tPA level was significantly increased by 249% in the cervical cord, and not in the lumbar cord (d; n = 3, p < 0.001), suggesting that AAV vectors were efficiently transported and successfully expressed transferred genes to the targeting area of the spinal cord. There was no RFP labelling in the muscles where AAV was injected.
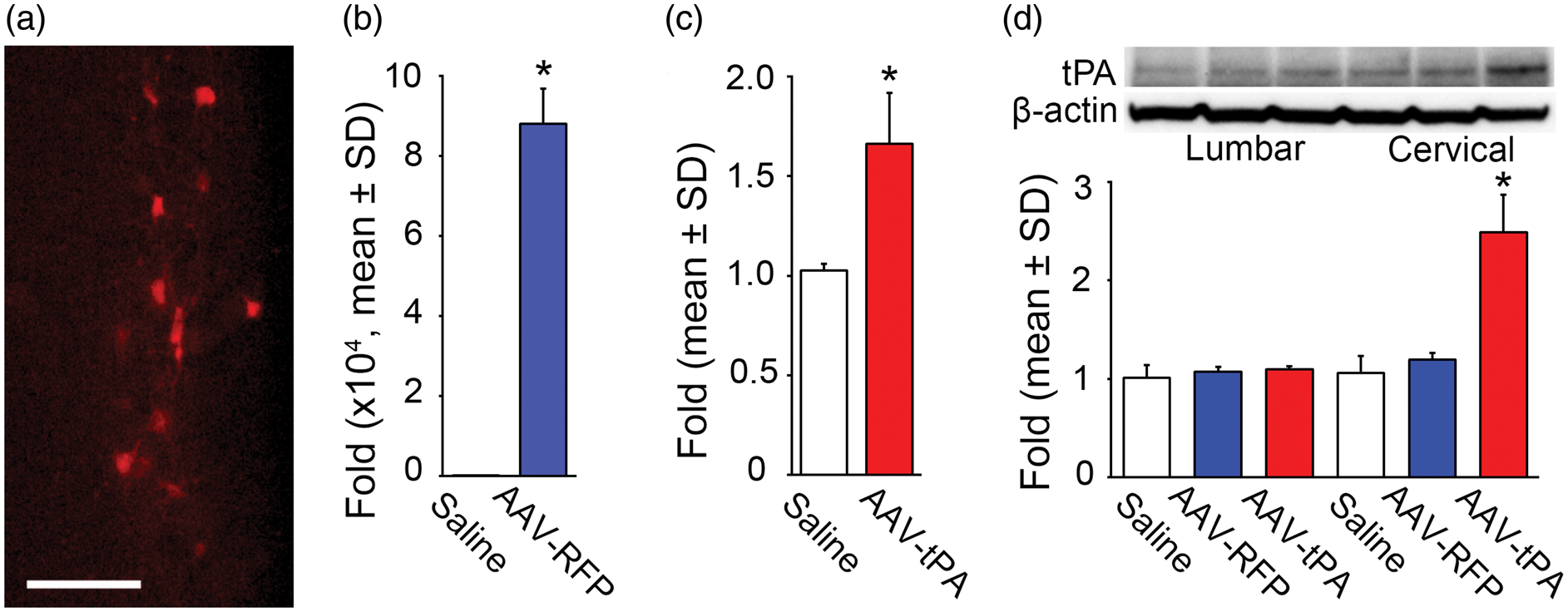
Retrograde transport of AAV injected into forelimb muscles to the spinal cord in normal mice. RFP expression observed in the spinal motor neurons on a representative longitudinal section (a), and mRNA transcription levels of RFP (b) and tPA (c) are increased in the cervical cord as assessed by qRT-PCR at 21 days after intramuscular injection (n = 3/group, p < 0.001, t-test). In mice subjected to MCAo, intramuscular injection of AAV-tPA significantly increased tPA protein level in the cervical cord, but not in the lumbar cord, as measured using Western Blot at 21 days after injection (d; n = 3/group, p < 0.001, one-way ANOVA). Scale bar = 100 µm.
Intramuscular AAV-tPA administration improves neurological recovery after MCAo
For the ischemic stroke model, mice were subjected to permanent unilateral MCAo, 9 and modified here by using an 8–0 monofilament suture which yielded highly consistent infarct volume without any mortality among animal groups (Figure 2).
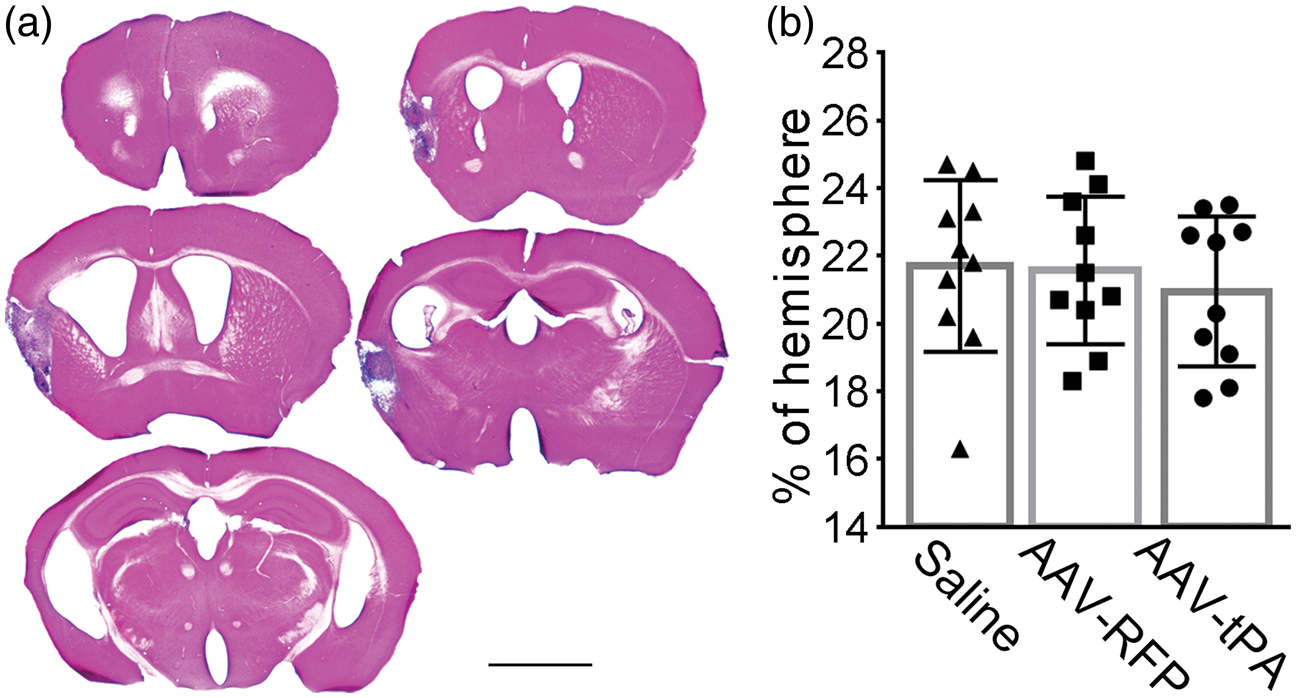
Permanent suture MCAo induced ischemic infarct in mice. (a) A series of Hematoxylin and Eosin stained brain vibratome sections cut at 1 mm intervals from a representative mouse shows a typical lesion 28 days after ischemia. (b) There was no significant difference in quantification of the infarct volume among animal groups treated with saline, AAV-RFP or AAV-tPA (n = 10/group, p > 0.05, one-way ANOVA). Scale bar = 2 mm.
To evaluate neurologic functional deficit and recovery after stroke, a single pellet reaching test for skilled task and a foot-fault test for non-skilled task were performed. As shown in Figure 3, in all animals, severe behavioral deficits were induced by MCAo at day 1 compared to the baseline scores prior to MCAo, and were comparable among treatment groups. Although the motor performance of the stroke-impaired forelimb in all mice gradual improved with time, the recovery was incomplete. The treatment effects were analyzed by a Global test, followed by individual test and pair-wise comparisons. A significant overall treatment effect was observed (p < 0.001) on functional improvement during days 14 to 28 after MCAo. Compared to animals treated with saline or AAV-RFP, mice in AAV-tPA-treated group had significantly improved functional recovery on both pellet reaching and foot fault tests, while no effect was observed for AAV-RFP treatment compared to saline (p > 0.05).
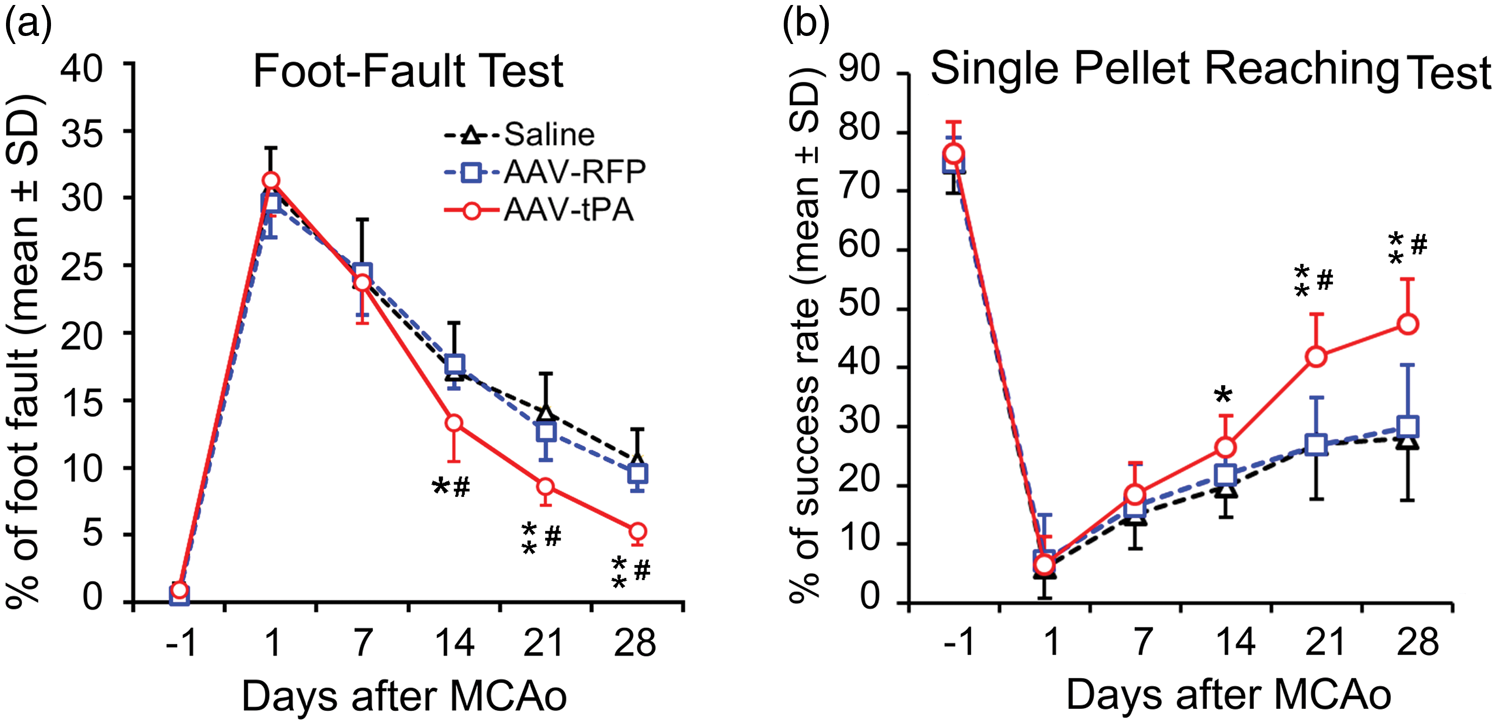
Profile of behavioral deficit and recovery after MCAo. Motor performance of the stroke-impaired left forepaw was assessed with foot-fault test (a) for non-skilled motor performance with accuracy of forepaw placement on a non-equidistant grid as the percentage of foot-faults of the stroke-impaired forepaw to total steps; and single pellet reaching test (b) for skilled voluntary control of the stroke-impaired forepaw with success rate of reaching and retrieving food pellets through a vertical slot of a plexiglas box. Note that significant behavioral deficits were evident one day post stroke, and were then followed by continuous, gradual, however, incomplete recovery as assessed using both tests. Intramuscular AAV-tPA treatment significantly enhanced functional improvements compared with saline and AAV-RFP (n = 10/group; *p < 0.01, **p < 0.001 vs. saline, #p < 0.001 vs. AAV-RFP; Global test).
Intramuscular AAV-tPA administration promotes CST axonal remodeling in the denervated side of the cervical cord
The present study employed a transgenic CST-YFP mouse strain, in which the CST axons are specifically and completely labeled with YFP, to measure the CST axonal density in the gray matter of the cervical cord 32 days after MCAo. The ratio of CST axonal density in the stroke-impaired side to the opposite side on same sections was used to assess axonal remodeling in the cervical cord. Compared to saline (Figure 4(a)) or AAV-RFP- (b) treated mice, CST axonal density in the stroke-impaired cervical gray matter was significantly increased in mice treated with AAV-tPA (c and d, p < 0.001).
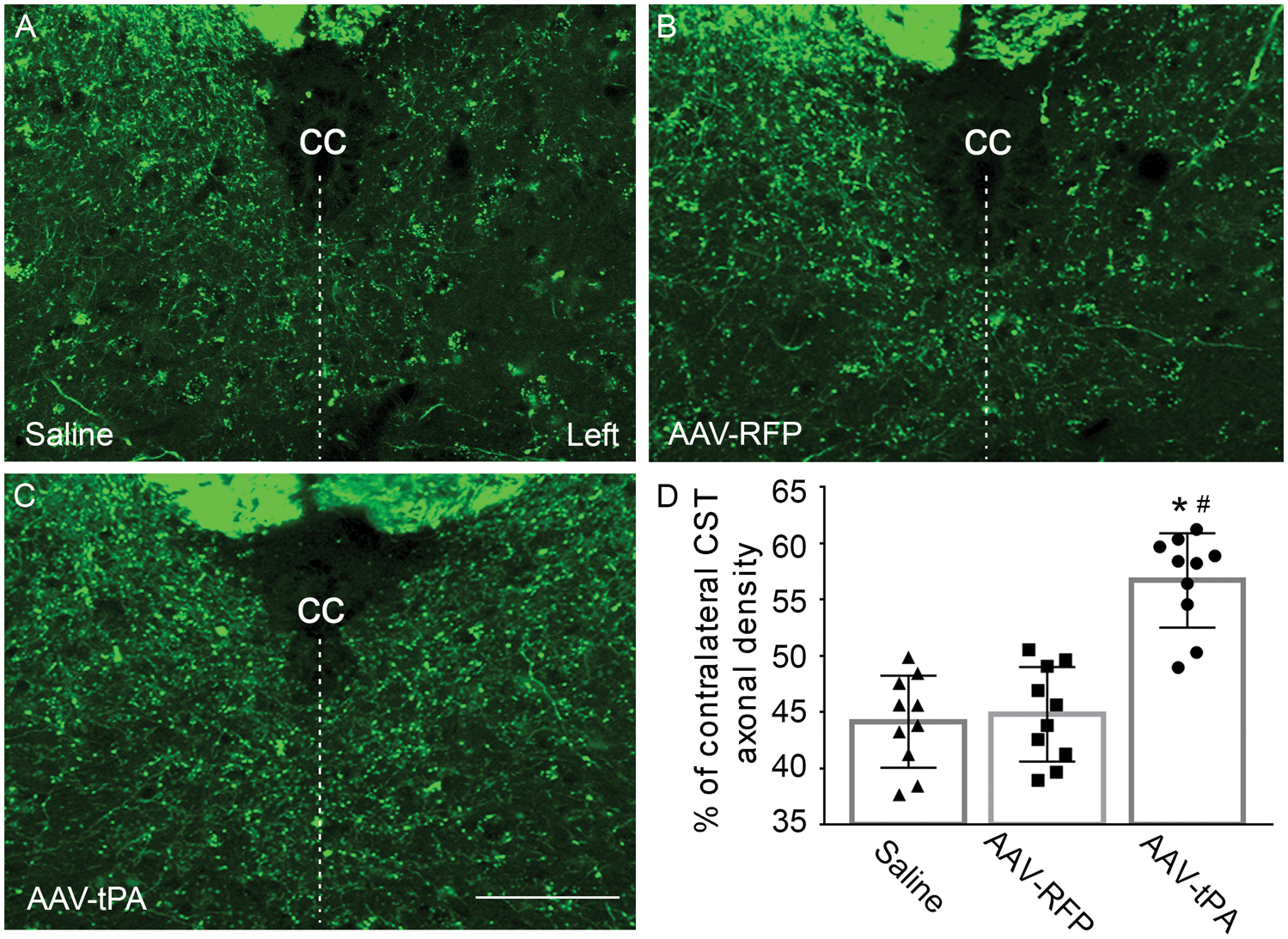
Single layer confocal images showing CST axons in the gray matter of cervical cord. CST axons were specifically and completely labeled with YFP in the transgenic CST-YFP mice. Density of YFP-positive CST axons in both sides of the gray matter was measured on transverse sections of the cervical cord. Ratio of CST axonal density in the stroke-impaired side (Left) to the contralesional side was calculated as an index for axonal remodeling. Compared to animals treated with saline (a) or AAV-RFP (b), intramuscular AAV-tPA treatment significantly increased density of CST axons in the denervated side of the cervical gray matter at 28 days after MCAo (c and d; n = 10/group; *p < 0.001 vs. saline, #p < 0.001 vs. AAV-RFP; one-way ANOVA). cc: central canal, indicating midline of the spinal cord. Scale bar = 50 µm.
Intramuscular AAV-tPA administration does not alter CST axonal number in the pyramidal tracts
To clarify the source of AAV-tPA treatment induced increase of CST axons in the denervated side of the cervical gray matter, we measured the number of CST axons in bilateral pyramidal tracts at the medulla level rostral to the CST decussation, where all CST axons including crossed and non-crossed CST axons are involved. Our data showed that the CST axonal numbers were comparable among all three animal groups treated with saline, AAV-RFP, or AAV-tPA (Figure 5, p > 0.05), respectively, indicating that the CST axonal increase in the cervical gray matter induced by intramuscular AAV-tPA treatment was attributed to local axonal outgrowth within the gray matter, and not to long-range axonal regeneration from the motor cortex throughout the CST (white matter), or rescue from the ischemic damage.
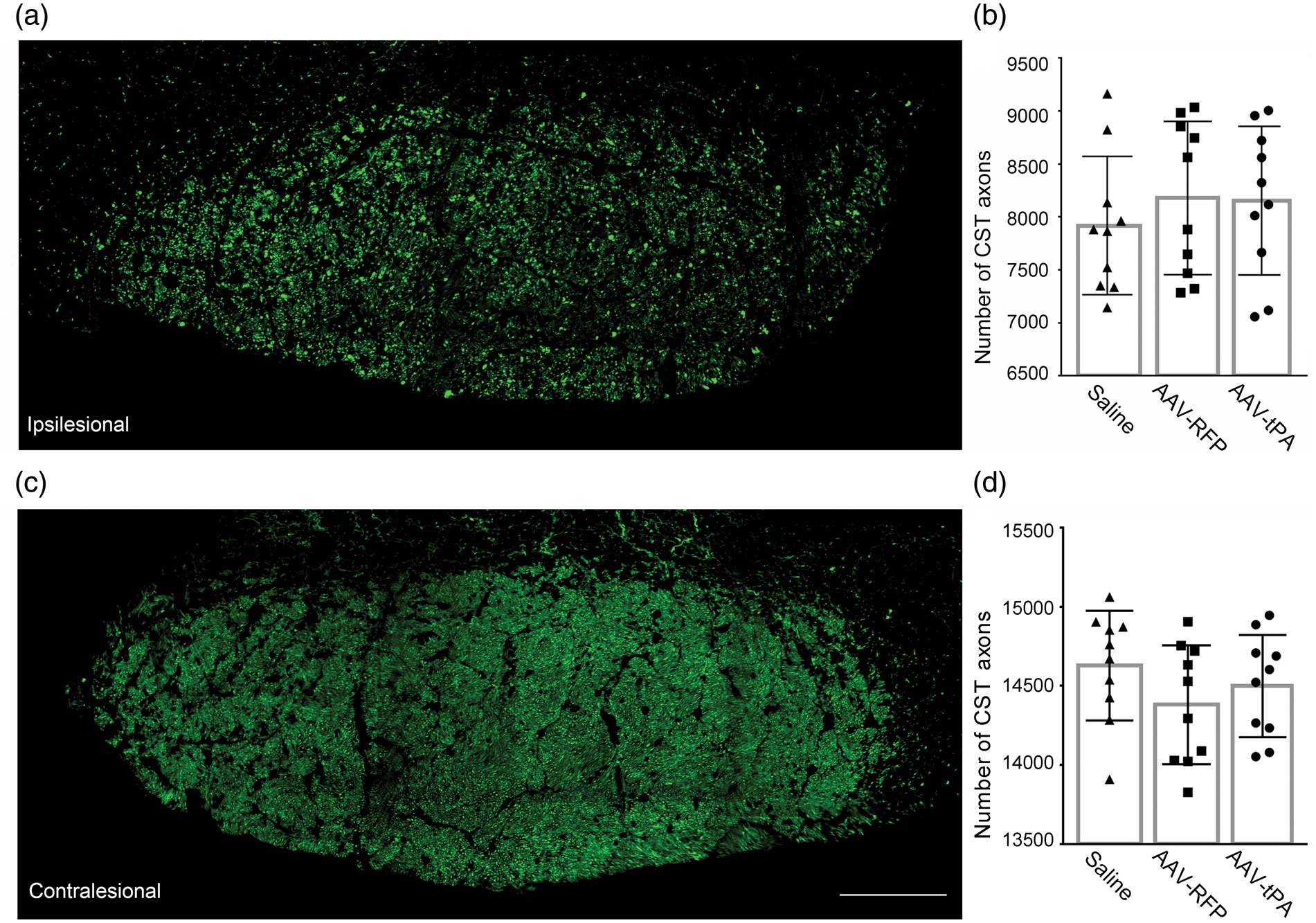
Single layer confocal images showing CST axons in the pyramidal tract at the medulla level. Representative images of YFP-labeled CST axons in ipsilesional (a) and contralesional (c) pyramidal tract were digitalized on a coronal section of a CST-YFP mouse subjected to MCAo treated with saline. Axon numbers were counted using ImageJ. Data showed that the numbers of CST axons in both ipsilesional (b) and contralesional (d) pyramidal tracts were not altered by intramuscular treatment of saline, AAV-RFP or AAV-tPA (n = 10/group; p > 0.05; one-way ANOVA), indicating that the AAV-tPA induced increase of CST axons in the cervical gray matter was attributed to local axonal growth, and not to long-range axonal regeneration in the white matter. Scale bar = 100 µm.
Intramuscular AAV-tPA administration enhances corticospinal innervation originating from bilateral cortices
In order to verify the functional innervation established by CST axonal remodeling in the denervated spinal gray matter, we injected trans-synaptic tracer PRV-614-RFP into the stroke-impaired forelimb muscles to retrogradely label the neural pathways from the peripheral tissue to the motor cortices. Thus, the RFP labeling in the pyramidal neurons represents that these neurons physically and functionally connect to the peripheral tissue where the PRV tracer was injected. Consistent with our previous data,6,14 RFP labeling was found in the layer V pyramidal neurons of both rostral and caudal forelimb motor areas, primarily in the ipsilesional cortex (Figure 6(a) to (c)), while scattered RFP-positive neurons were detected in the contralesional hemisphere (e–g) in stroke mice treated with saline (a and e), AAV-RFP (b and f), or AAV-tPA (c and g). The data showed that AAV-tPA treatment significantly increased the number of RFP-positive neurons in both ipsilesional (d, p < 0.01 vs. saline, p < 0.001 vs. AAV-RFP) and contralesional (h, p < 0.05 vs. saline, p < 0.01 vs. AAV-RFP) cortices.
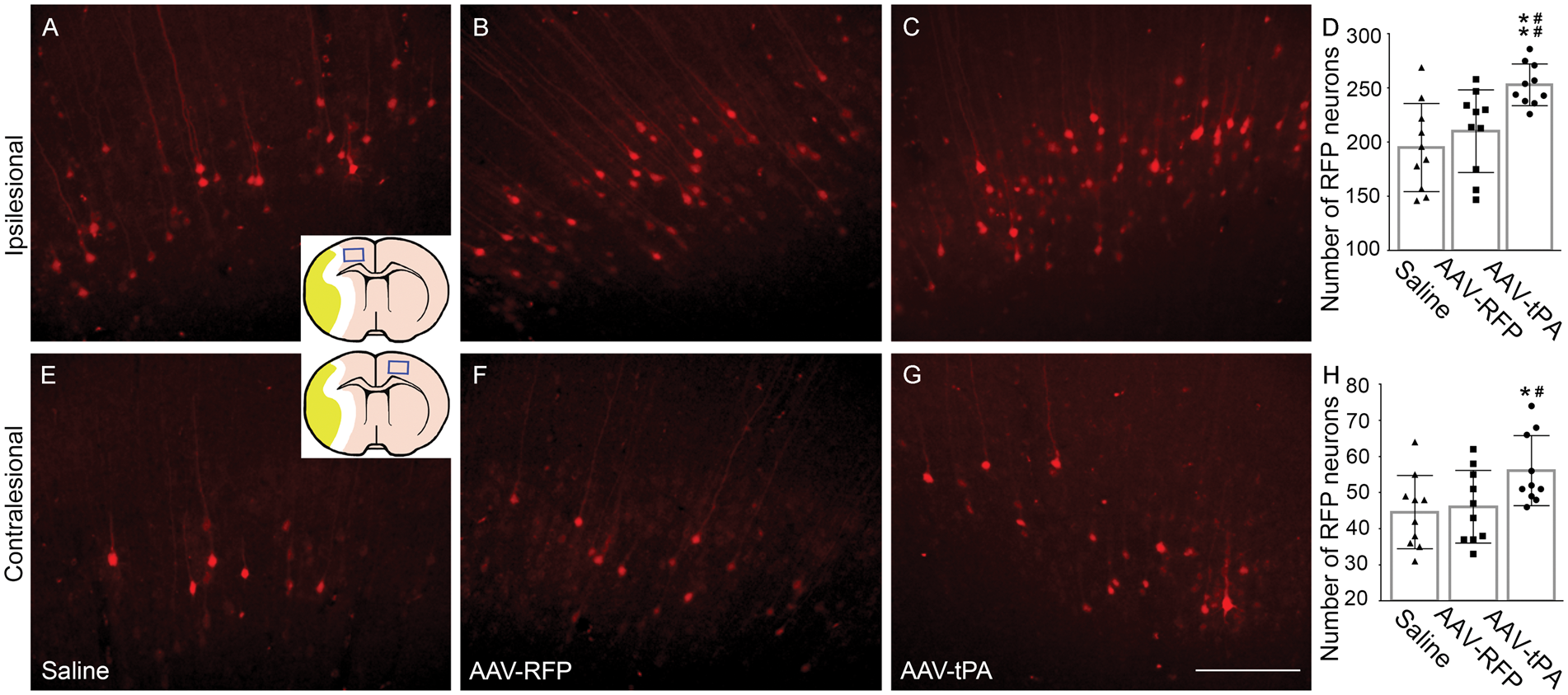
Confocal images showing RFP-positive cortical neurons. A retrograde trans-synaptic tracer PRV-614-mRFP was injected into the stroke-impaired forelimb muscles at 28 days after MCAo. Four days later, the brain samples were processed for coronal vibratome sectioning. RFP-positive neurons in bilateral cortices, that indicate synaptic innervation with the stroke-impaired forelimb muscles, were counted on a series of sections in 0.5 mm distances. In all animals treated with saline (a and e), AAV-RFP (b and f) or AAV-tPA (c and g), the majority of RFP labeled cortical neurons were found within the ipsilesional cortex (a–c), while only a few scattered neurons in the contralesional cortex (e–g) were labeled. Compared to saline and AAV-RFP-treated animals, AAV-tPA significantly increased the numbers of RFP-positive cortical neurons in both ipsilesional (d, **p < 0.01 vs. saline, ##p < 0.001 vs. AAV-RFP) and contralesional (h) cortices (n = 10/group; *p < 0.05 vs. saline, #p < 0.01 vs. AAV-RFP; one-way ANOVA). Scale bar = 200 µm.
Behavioral recovery significantly correlates to neuronal remodeling
Pearson’s correlation test was used to evaluate the correlations between individual behavioral scores and neuronal remodeling in the denervated cervical gray matter and bilateral cortices (Figure 7). The results showed that both skilled and non-skilled performance were strongly correlated with CST axonal density (p < 0.001) in the cervical gray matter and with the number of RFP-positive cortical neurons in the ipsilesional hemisphere (p < 0.01). While significant, however, weaker correlations were found with RFP-positive neurons in the contralesional cortex (p < 0.05), indicating that neuronal remodeling in the spinal cord and bilateral cortices may contribute to the motor recovery after stroke.
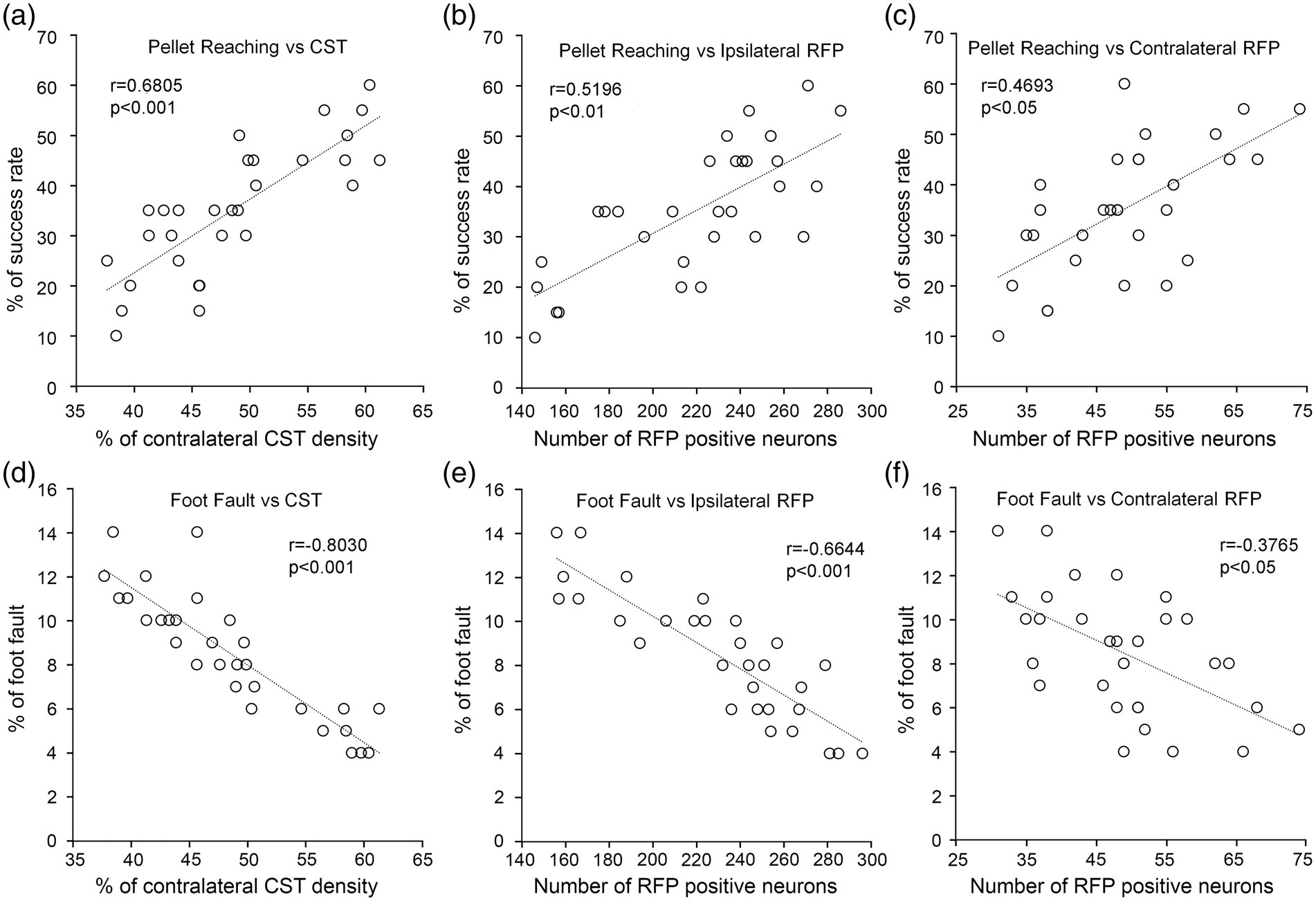
Correlations between neural remodeling and behavioral recovery. Pearson correlation analysis showed significant positive correlations between performance in the skilled pellet reaching task and the index of CST axonal remodeling in the denervated cervical gray matter (a), innervation originating from the ipsilesional cortex (b) or contralesional cortex (c), as well as negative correlations between non-skilled foot fault task to CST axonal remodeling (d), ipsilesional cortical innervation (e), and contralesional cortical innervation (f) at 28 days after MCAo.
Discussion
In the present study, we propose a novel strategy to promote motor functional recovery after stroke by specifically rewiring the denervated spinal motor neurons via enhancing the CST axonal remodeling in the spinal gray matter. We employ a non-invasive peripheral approach, intramuscular injection of recombinant AAV encoding tPA gene into the stroke-impaired muscles, to overexpress tPA in the spinal motor neurons that innervate these muscles. tPA released into the surrounding area in the spinal gray matter stimulates CST axonal outgrowth toward these neurons to re-establish the cortical innervation of the stroke-impaired muscles.
To avoid tPA expression in the muscles of the injection side, tPA expression was regulated under a neuron specific Synapsin promoter. Intramuscular delivery provides significant advantages over other delivery methods. Intravenous injection of AAV vectors requires transmission across the blood–brain barrier and has low specificity for the CNS. Direct infusion into the brain and spinal parenchyma is limited by poor diffusion 15 and surgical invasion. Injection into the cerebrospinal fluid via either the lateral ventricles or thecal space may be beneficial for neuronal degenerative diseases to achieve widespread gene transfer into the entire CNS, but is not effective for targeted local delivery. Non-targeted widespread delivery may induce aberrant axonal sprouting and outgrowth in the dorsal horn of the spinal cord leading to hyperalgesia and allodynia. 16 Additionally, this targeted delivery requires less volume of the AAV vector, with substantially reduced off-target expression, to reduce the exposure of immune system, hence reducing the potential deleterious inflammatory response. 17
Due to its properties of non-pathogenicity in humans and animals, low immunogenicity and long-lasting, stable gene expression, 18 AAV-mediated gene transfer approaches have been widely used for the treatment of many neurological disorders, safely and efficaciously in both experimental animal studies19,20 and in phase I/II/III clinical trials.21–25 A single intramuscular injection of AAV vectors significantly elevates gene expression in the spinal cord at two weeks after injection 26 and the expression level peaks approximately three months after injection. 27 In addition, intramuscular AAV injection and expression do not alter neuromuscular junction innervation. 28 Therefore, the efficient and persistent AAV-mediated gene delivery is appropriate for restorative agent delivery to enhance neurological recovery during subacute and chronic phases after stroke. Numerous AAV serotypes have been identified with variable tropism by different structures of the capsids, and unique membrane-associated receptors and intracellular trafficking. 29 Thus, each serotype has a distinct pattern of natural tropism, which affects transduction efficiency for gene transfer in certain tissue and/or cell type, 30 and has different potentials for retrograde transport 31 through a given route of administration. In our preliminary study to identify the optimal AAV serotype to deliver genes from the peripheral muscles to the spinal cord, we have injected 1 × 1010 AAV serotypes of 1, 2, 5, 6, 8 and 9 carrying RFP gene into the wrist extensor and flexor muscles in mice. We found that mice injected with serotype 5, which was employed in this study, showed the highest number of RFP-labeled spinal motoneurons detected with fluorescent microscopy (unpublished data). Our results are consistent with a recent study that AAV5 is particularly efficient for selective retrograde axonal transport in mouse brain. 32 As the safest viral vector system, with the first approval of AAV-mediated gene therapy by the European Medicines Agency in 2012 (Glybera®), and by the US Food and Drug Administration in 2018 (Spark Therapeutics), 33 there will likely be increased efforts to optimize and maximize clinical manufacturing technologies for AAV-based gene therapies, and a rapid movement of this AAV technology into the clinic.
To verify our strategy of promoting stroke recovery by enhancing CST axonal remodeling in the denervated spinal cord, we overexpressed tPA as an example in the spinal motor neurons delivered by intramuscular injection. Other than vascular endothelium-derived tPA in the circulation, 34 tPA synthesized in neuroendocrine tissue including neurons and glial cells,35,36 is widely distributed in the brain, and is involved in neurite outgrowth,37–39 axonal path finding,38,40 synaptic plasticity,41–43 and dendritic remodeling 44 during development, and after stroke or brain injury. During the onset of ischemia, there were reports that tPA may be neuronal excitotoxic in the brain parenchymal tissue; 45 however, recent studies also demonstrated that tPA protects neurons from excitotoxic-induced cell death.46,47 Overexpression of tPA specifically in neurons leads to decreasing the ischemic lesion volume and improving neurological outcome following ischemic stroke in mice. 48 Importantly, tPA-treated patients continue to improve faster and to a larger extent during the rehabilitation period beyond the acute phase than non-tPA-treated patients. 49 Given the very large number of acute stroke patients worldwide who have been treated with tPA, to our knowledge, there are no clinical reports indicating a neurotoxic effect of tPA treatment. Thus, the preponderance of experimental and clinical stroke data supports a neuroprotective and not a neurotoxic role for tPA. Therefore, in the present study, tPA overexpressed in the spinal motor neurons is not a means to lyse a clot as a fibrinolytic agent but acts as a neurorestorative agent in the denervated side of the spinal cord, and thereby promotes motor functional recovery during the subacute phase after stroke.
In the adult mammal, the injured CNS is a highly inhibitory environment for axonal regeneration, severely limiting functional recovery after damage. Although adult CNS neurons retain the capacity for axon outgrowth, however, this capacity is insufficient to extend long axons crossing over the lesion barrier in the white matter to restore the lost functions. 50 To verify whether neurological recovery after stroke is attributed to CST axonal regeneration, we measured the CST axonal numbers in the pyramidal tract at the medulla level among animal groups subjected to different treatments. Consistent with our previous study of CST tracing using biotinylated dextran amines in rats, 5 we found that the CST axonal numbers of the pyramidal tract originating from either ipsilesional or contralesional cortical hemisphere at the upper cervical level were not affected by AAV-tPA treatment compared with saline or AAV-RFP treatment. In contrast, the CST axonal density in the denervated gray matter was significantly restored in the AAV-tPA-treated mice, which was highly correlated with motor performance of the stroke-impaired forelimb. Our observations further supported that the spinal gray matter is permissive for axonal outgrowth. 51 Thus, instead of long-distance axonal regeneration of the injured CST in the white matter of the CNS, our approach specifically targets the neural remodeling in the denervated spinal gray matter by inducing local short-range axonal sprouting and outgrowth of the neighboring uninjured fibers, to increase their arborization and form new synaptic connections with denervated spinal neurons, which thereby restores innervation from the motor cortex. This strategy can circumvent the limitation of axonal outgrowth in the white matter, which may be important for the development of efficient therapies for stroke and other CNS diseases. Although neuronal plasticity in the brain and spinal cord has been increasingly appreciated as required for neurological improvement post stroke, ours is the first demonstration of wiring of the stroke-impaired spinal motor neurons via a non-invasive peripheral approach.
In the present study, we employed a retrograde trans-synaptic neural tracer, PRV-RFP, to identify the neuronal origins in the cortex for the innervation of the stroke-impaired forelimb muscles. We found that intramuscular AAV-tPA treatment significantly increased numbers of RFP-positive cortical neurons in both ipsilesional and contralesional hemispheres; however, considering that the extent of labeled neurons in the ipsilesional cortex was much greater than in the contralesional cortex, we suggest that behavioral recovery after stroke, at least in our model, is mostly attributed to neuronal reorganization originating from the area surrounding the ischemic lesion. Since the transport of attenuated Bartha strain of PRV occurs only at the points of synaptic contact and proceeds in retrograde direction (i.e. from postsynaptic to presynaptic neuron), thus the virus spread along neuronal hierarchical chains with substantial synaptic connections. 52 Therefore, our data of increased neuronal PRV labeling in the AAV-tPA-treated animals demonstrated that intramuscular injection of AAV-tPA into the stroke-impaired forelimb muscles not only enhanced CST axonal outgrowth in the denervated spinal gray matter measured by YFP density, but also promoted these axons forming synaptic innervation with the denervated spinal motor neurons, and thereby enhanced the motor control of the stroke-impaired forelimb.
Although the CST anatomy differs between rodents and humans in several fundamental ways, for example, in humans, the main CST is located in the lateral white matter, whereas in the rodents it is located in the dorsal funicular, basically, the anatomic distribution of its origin sites in motor cortices that control the opposite side of the body indicates homology of the CST between rodents and primates. At the medullary pyramid level, in which essentially all CST axons pass through, the number of CST axons is approximately 1,000,000 in humans, 70,000 in rats, and 30,000 in mice. 53 This provides anatomical bases for the well-developed fine skilled movement control of the upper limbs, especially the fingers in humans. For stroke survivors with long-term disability, regaining upper extremity function may lead to greater independence, thereby improving quality of life. 54 It has been demonstrated that skilled limb movements, such as reach-to-grasp movement, show very similar motor components in humans and in rodents. 55 In the present study, we employed two motor behavioral tests, the foot-fault test and the single pellet reaching test, to estimate the motor behavioral outcome of the impaired forepaw in mice subjected to unilateral MCAo. In both behavioral tests, the mice need to voluntarily control the paw movement. When mice walk on the non-equidistant grids, each step requires adjustment in stride length and distribution of body weight to place the limb appropriately on the rung and then grasp it. 56 For the skilled reaching task, mice advance the forelimb aimed to the pellet, pronate the paw on it, grasp it, extract it, and release the food into the mouth. 11 As a proof-of-principle study, we only delivered AAV-tPA into the wrist extensor and flexor muscles, which are the most important for grasp motion needed for daily living and significantly impaired after stroke. Since stroke impairs both upper and lower limbs in most patients, multiple injections in the lower limbs may be tested in subsequent studies. In addition, further dose-response and time-window studies are needed to develop a complete therapeutic approach for stroke patients. It is expected that the strategy of promoting corticospinal innervation in the denervated spinal gray matter via our noninvasive peripheral approach to enhance neurological recovery after stroke may be more effective and important in primates, due to their much better developed corticospinal innervation than in rodents.
In summary, we propose an innovative strategy to facilitate axonal rewiring in the denervated spinal gray matter in order to improve neurological recovery after stroke. Compared to animals treated with saline or AAV-RFP, AAV-tPA treatment significantly improves motor behavioral recovery after stroke, as well as enhances CST axonal remodeling in the denervated spinal gray matter and restores cortical innervation of the stroke-impaired forelimb muscles. Our results provide a powerful impetus for further pharmacokinetic and mechanistic studies in non-human primates, and possibly, clinical trials for stroke patients.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors greatly appreciate Dr. Lynn Enquist, Princeton University, for the gift of PRV-614-mRFP, who is supported by NIH Virus Center grant no. P40RR018604.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Experimental design: ZL, MC and LH.
Experimental performance: ZL, XG, HX and WG.
Data Acquisition: XG and FW.
Data analysis: ML.
Manuscript writing: ZL and MC.
