Abstract
Mild traumatic brain injury (mTBI) is a risk factor for Alzheimer’s disease (AD), and evidence suggests cerebrovascular dysregulation initiates deleterious neurodegenerative cascades. We examined whether mTBI history alters cerebral blood flow (CBF) and cortical thickness in regions vulnerable to early AD-related changes. Seventy-four young to middle-aged Veterans (mean age = 34, range = 23–48) underwent brain scans. Participants were divided into: (1) Veteran Controls (n = 27), (2) 1–2 mTBIs (n = 26), and (2) 3+ mTBIs (n = 21) groups. Resting CBF was measured using MP-PCASL. T1 structural scans were processed with FreeSurfer. CBF and cortical thickness estimates were extracted from nine AD-vulnerable regions. Regression analyses examined whether mTBI moderated the association between age, CBF, and cortical thickness. Regressions adjusting for sex and posttraumatic stress revealed mTBI moderated the association between age and CBF of the precuneus as well as superior and inferior parietal cortices (p’s < .05); increasing age was associated with lower CBF in the 3+ mTBIs group, but not in the VCs or 1–2 mTBIs groups. mTBI did not moderate associations between age and cortical thickness (p’s >.05). Repetitive mTBI is associated with cerebrovascular dysfunction in AD-vulnerable regions and may accelerate pathological aging trajectories.
Introduction
As Veterans involved in the United States conflicts in Iraq and Afghanistan begin to age, there has been an increasing emphasis on understanding the chronic effects of mild traumatic brain injury (mTBI). While mTBI has been linked to greater rates of functional impairment and disability in the immediate years following initial injury,1,2 a growing body of literature has highlighted that negative neurobehavioral health outcomes are also evident in late-life. 3 For example, older Veterans with history of mTBI demonstrate more severe psychiatric symptomatology and poorer neurocognitive functioning relative to those with no history of head trauma.4,5 Moreover, recent epidemiological studies have revealed that, among civilian and military samples, mTBI is associated with an increased risk as well as earlier development of neurodegenerative diseases including Alzheimer’s disease (AD).6–8 Within samples of Veterans with mTBI, elevated rates of dementia diagnoses have even been observed in those without a history of loss of consciousness, and this risk is magnified within the context of increasing injury severity and repetitive head-trauma.6,9
Unfortunately, the precise pathophysiological mechanisms by which neurotrauma leads to adverse long-term neurobehavioral and health outcomes remain poorly understood. Nevertheless, recent research has provided compelling evidence that cerebrovascular pathology is a common consequence of TBI that likely represents a key mechanistic link between TBI history and the later development of neurodegenerative disorders such as AD.10,11 During TBI, linear and rotational forces that shift the brain within the cranial vault cause a host of primary injuries in the form of diffuse axonal injury, contused parenchyma, torn or broken vasculature, and microhemorrhages. 12 This structural damage initiates secondary metabolic, cellular, and inflammatory cascades that then have negative functional consequences. For example, altered cerebral blood flow (CBF), cerebrovascular reactivity, and blood–brain barrier permeability have been observed in both the acute and chronic phases of injury following all severities of TBI.13–16 Importantly, this prolonged cerebrovascular dysfunction not only directly negatively influences cognitive and behavioral functioning, but independently initiates degenerative processes within the brain.10,11 Indeed, we recently showed that lower CBF is associated with reduced axonal integrity in middle-aged Veterans with history of mild or moderate TBI. 17 Moreover, longitudinal neuroimaging studies have demonstrated that mTBI-related structural and functional brain changes do not appear to be static, but rather tend to evolve or worsen with time.18–20 Identifying the precise nature and temporal aspects of these pathologic processes is necessary for proper prevention of poor long-term outcomes.
Within the context of aging, cerebrovascular dysfunction has also been identified as an early marker and propagator of molecular AD and degenerative processes.21,22 Chronological age is known to be strongly associated with morphological changes to the vasculature (i.e. vascular stiffening, arterial wall thickening), as well as an increased presence of cerebrovascular risk factors (e.g. hypertension, hyperlipidemia). The vascular hypothesis of AD posits that the combination of these factors places strain on the cerebrovasculature and results in chronic vascular dysfunction in the form of increased blood–brain-barrier (BBB) permeability and CBF reductions.23–25 These changes in cerebrovascular dynamics, in conjunction with the accumulation of pathologic proteins such as amyloid-β and tau, likely jointly contribute to the subsequent neurodegenerative and behavioral changes that characterize AD. 26 However, the extent to which vascular changes represent a mechanism of adverse health outcomes in aging samples with history of mTBI remains incompletely characterized.
It is critical to recognize that post-traumatic neurodegenerative cascades may also contribute to progressive age-related dementia processes. Within the context of TBI, traumatic axonal injury is a primary and well-documented consequence of neurotrauma that initiates a number of pathological processes including Wallerian or retrograde degeneration (i.e. axonal or neuronal loss), prolonged microglial activation or inflammation, and increased amyloid accumulation and deposition (see Bigler and Maxwell 27 for review). Although the temporal course of these injury cascades remains poorly understood, human structural MRI studies have highlighted consequences that include white matter microstructural alterations,17,28 global and regional volumetric reductions,29,30 and cortical thinning following mTBI.31,32 Importantly, these TBI-initiated brain changes may serve to (1) place an individual at increased risk for dementia by reducing brain reserve or (2) exacerbate independent age-related pathological processes.
Within the context of AD risk, cortical thinning represents one of the earliest measurable markers of brain changes associated with pathological aging.33,34 This neurodegeneration is the consequence of (1) pathological protein accumulation that tends to occur in a regionally distinct manner in the earliest phases of the disease and/or (2) altered cerebral hemodynamics, and has been linked to both development and progression of AD. Although studies exploring the intersection of aging and TBI-related neurodegeneration are relatively limited, a recent study showed that older Vietnam-era Veterans with self-reported history of mTBI showed greater cortical thinning in AD-vulnerable regions relative to their non-injured counterparts, and that cortical thinning was associated with higher CSF tau accumulation. 35 While it is difficult to disentangle whether cortical thinning is a primary consequence of TBI or an age-related processes, clarifying the temporal course of structural changes in tandem with functional changes (i.e. cerebrovascular dysfunction) across the lifespan may provide insight into relevant mechanisms.
Further complicating our understanding of the chronic effects of mTBI is the observation that many individuals report sustaining more than one head injury throughout their lifetime,36,37 and studies have shown that the neural consequences of TBI may be more pronounced in those with repetitive TBI.38,39 For example, repetitive mTBI has also been linked to neurovascular changes (e.g. decreased vessel density, perivascular tau pathology, global reductions in CBF) in both animal and human studies.38,40 Additionally, perivascular tau accumulation has been implicated in BBB breakdown and cerebrovascular dysfunction following TBI in mice.22,41 Although human studies that directly examine brain changes resulting from repetitive mTBI relative to single mTBI events are limited, one study showed that those with repetitive TBI demonstrate changes in CBF, an increased prevalence of microhemorrhages, and altered brain connectivity relative to those with a single mTBI. 39 Additionally, structural changes in both white and gray matter have also been noted in individuals with repetitive mTBI.42,43 In other words, repetitive head trauma may represent a previously unexplored factor in the link between aging, cerebrovascular dysfunction, and cortical thinning when examining poor long-term outcomes.
The current study sought to clarify potential mechanisms underlying dementia risk in a sample of young to middle-aged Veterans with and without history of mTBI. We focused on AD-vulnerable regions in an effort to understand the extent to which age-related structural and functional brain changes may be altered by mTBI. We hypothesized that greater age-related changes in CBF and cortical thickness would occur in those with history of mTBI and that these changes would be especially pronounced in those with repetitive mTBI. Importantly, identifying the nature and the extent to which age-related neural processes may be altered following mTBI may represent a critical point of prevention and intervention for dementia risk in late-life.
Materials and methods
Participants and procedures
Study participants were recruited directly from paper and television advertisements that are located in the waiting room areas of medical and mental health outpatient clinics within the VA San Diego Healthcare System (VASDHS). Participants were 74 Operation Enduring Freedom, Operation Iraqi Freedom, or Operation New Dawn Veterans. Veterans were subdivided in Veteran Controls with no history of mTBI (VCs; n = 27), those with history of 1–2 mTBIs (n = 26), and those with a history of 3 or more mTBIs (3+ mTBIs; n = 21). Veterans with a history of mTBI were dichotomized via a median split of the total number of lifetime mTBIs (Median = 2). These groupings were also based on similar studies exploring the negative influence of repetitive head-trauma on behavior in other Veteran samples.44,45
All procedures involved in this study were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975. All participants gave written informed consent upon enrollment in the study, and the protocol for the study was approved by the Institutional Review Boards of both the VASDHS and University of California, San Diego. Study participation involved completion of TBI and substance use clinical interviews, self-report symptom rating scales, and magnetic resonance imaging (MRI) brain scans. Clinical interviews and questionnaires were completed at the Veterans Medical Research Foundation located at the La Jolla VASDHS campus. MRI brain scans occurred on the medical school campus of UCSD at the center for functional MRI.
Inclusion and exclusion criteria
Participants were included in the study if they (1) were between the ages of 18–50, (2) VCs who denied a history of head-trauma; (3) TBI participants who endorsed at least one injury that met clinical criteria for mTBI as defined by a loss of consciousness (LOC) of < 30 min, alteration of consciousness (AOC) of ≤ 24 h, and/or posttraumatic amnesia (PTA) ≤ 24 h, 46 (4) demonstrated no structural abnormalities, gross lesions, or visible contusions on T1 scans, and (5) had both T1 and arterial spin labeling (ASL) sequences collected during their MRI visit.
Participants were excluded from the study if they had a history of moderate or severe TBI as defined by a LOC >30, AOC > 24 h, or PTA >24 h; 46 endorsed a history of neurological disorders including epilepsy, multiple sclerosis, stroke, chronic fatigue syndrome, or Parkinson’s disease; had previously been diagnosed with schizophrenia, bipolar, or psychotic disorder; met criteria for current substance or alcohol abuse in accordance with the Diagnostic and Statistical Manual of Mental Disorders—Fourth Edition, Text Revision (DSM-IV-TR) 47 met criteria for current or past substance/alcohol dependence as per DSM-IV-TR criteria; or had a positive result on the Rapid Response 10-drug Test Panel.
Assessment of TBI history and diagnostic procedures
Trained study staff at the post-baccalaureate or graduate school level conducted TBI clinical interviews. Each participant was queried about any head injuries that occurred (1) prior to their enlistment in the armed forces, (2) during their military service (including any deployments), and (3) after their discharge from the military. This interview is a lab-based assessment tool that was adapted from the VA Semi-Structured Clinical Interview for TBI 48 and allows for the assessment of up to 10 lifetime head injuries. The interviewer captures data about the presence and duration of LOC, AOC, or PTA, as well as the details surrounding the setting (e.g. military vs. non-military event), mechanism of injury (blast-related vs. blunt/mechanical force), and date of injury.
Diagnosis and classification (i.e. mild, moderate, or severe) of TBI were largely based upon retrospective self-report of LOC, AOC, and PTA durations as per the VA/DoD Clinical Practice Guideline for Management of Concussion. 46 For individuals who had been previously assessed by the VASDHS TBI or Polytrauma Clinics, VA medical charts were reviewed for consistency of injury reporting. Data from the TBI interview were used to quantify the total number of lifetime injuries that met clinical criteria for mTBI for each participant, as well as to calculate how much time had passed since the date of each participant’s last mTBI. Finally, the presence and duration of LOC and/or AOC for each reported injury were compared and used to identify an individual’s most significant mTBI; injuries in which participants endorsed a LOC were considered more severe than those in which only AOC was endorsed.
Psychological and post-concussive symptom inventories
Current symptoms of posttraumatic stress were captured using the PTSD Checklist (PCL-M). 49 The PCL-M is a 17-item self-report questionnaire that assesses reexperiencing, avoidance, and hypervigilance symptoms in accordance with DSM-IV-TR criteria for PTSD. Participants use a 5-point scale of 1 (not at all) to 5 (extremely) to rate the extent to which each symptom has “bothered them over the past month,” and the sum of each item can be generated into a total score. The total score ranges from 17 to 85, with higher scores indicating more severe PTSD symptomatology.
Current symptoms of depression were captured using the Beck Depression Inventory-II (BDI-II). 50 The BDI-II is a 21-item self-report questionnaire that assesses the severity of cognitive (e.g. concentration or attention difficulties), affective (e.g. anhedonia, apathy), and somatic (e.g. fatigue or sleep changes) depressive symptoms that overlap with the DSM-IV-TR criteria for Major Depressive Disorder. Participants use a 4-point scale of 0 (not present or absent) to 3 (severe or present most days) to characterize the average severity of each depressive symptom during the past two weeks. Each item can be summed into a total score ranges that from 0 to 63, with higher scores indicating more severe depressive symptomatology.
Current post-concussive symptoms were captured using the Neurobehavioral Symptom Inventory (NSI), 51 a 22-item self-report questionnaire that assesses a variety of cognitive (e.g. forgetfulness, concentration difficulties), emotional (e.g. sadness, irritability), and physical (e.g. dizziness, light sensitivity) symptoms that frequently present after head-trauma. Participants use a 5-point scale of 0 (not at all) to 4 (very severe) to rate the extent to which each symptom has disturbed them in the past two weeks and the sum of each item can be generated into a total score. The total score ranges from 0 to 88, with higher scores indicating more severe post-concussive symptomatology.
Neuroimaging data acquisition
Participants were scanned using an 8-channel head coil on a 3.0 Tesla General Electric MR750 whole body systems scanner at the UCSD Center for Functional Imaging on the UCSD La Jolla campus. Scans took place in a dark room with no visual stimulation, and participants were instructed to remain motionless for the duration of the scan.
A three-dimensional fast spoiled gradient recalled echo T1-weighted anatomic scan was acquired with the following parameters: field of view = 24 cm, 256 × 192 matrix, repetition time = 8.1 ms, echo time = 3.192 ms, flip angle = 12°, inversion time = 550 ms, bandwidth = 31.25 kHZ, and 1.2 mm 172 contiguous sagittal slices.
Whole-brain resting CBF was acquired using a multiphase pseudocontinuous ASL (PCASL) sequence with the following parameters: 20 5 mm axial slices (1 mm gap), field of view = 24, PCASL labeling duration = 2000 ms, post labeling delay = 1600 ms, repetition time = 4200 ms, echo time = 3.3 ms, volumes = 60. Additionally, a scan with the inversion pulses turned off to obtain an estimate of the magnetization of cerebrospinal fluid (CSF) was acquired under the following parameters: repetition time = 4000 ms, echo time = 3.3 ms, NEX = 9, 90° pulse which is turned off for first eight repetitions to create PDW image contrast. The CSF scan signal was utilized to estimate the equilibrium magnetization of blood and convert the perfusion signal into CBF units of mL/(100 g-min). To adjust for coil inhomogeneities during the CBF quantification step, a minimum contrast scan was acquired with the follow parameters: repetition time = 4000 ms, echo time = 3.3 ms, NEX = 9. Finally, to correct for distortion and signal drop, a field map was acquired with the following parameters: repetition time = 500 ms, echo time 1 = 6.5 ms, echo time 2 = 8.5 ms, flip angle 45°.
Neuroimaging data processing
Participants’ MRI data underwent pre- and post- processing utilizing Analysis of Functional NeuroImages, 52 FMRIB Software Library (FSL), 53 Freesurfer,54,55 and in-house Matlab scripts.
T1-weighted anatomic scans were processed using FreeSurfer version 5.1’s recon-all processing pipeline 55 and additional details surrounding these technical procedures are described in prior publications.54–57 This pipeline allows for automatic cortical reconstruction and volumetric segmentation of neural structures of interest. Briefly, these procedures include motion correction of T1-weighted images, skull-stripping, automated Talairach transformation, intensity normalization, tessellation of the gray matter and white matter tissue boundaries, automated topology correction, surface deformation and inflation, and registration to a spherical atlas to ensure cortical geometry is matched across subjects.54–57 All images were visually inspected and, when necessary, manual edits were performed. FreeSurfer output of interest included volume and thickness estimates. Cortical thickness was calculated as the measure of the distance of the gray/white matter boundary and the gray matter/CSF boundary at each vertex on the cortical surface.
Processing of the raw resting ASL data was completed through the Cerebral Blood Flow Biomedical Informatics Research Network established at the UCSD Center for Functional MRI.58,59 ASL images were field map and motion corrected, skull-stripped, and segmented using FSL’s Automated Segmentation Tool. 53 A high resolution T1-anatomic scan and partial volume segmentations were then registered to ASL space, and partial volume segmentations were down-sampled to the resolution of the ASL scans. The mean ASL image was then converted to absolute units of CBF (mL/100 g tissue/min), and CBF estimates were partial volume corrected, blurred at 4.0 mm full-width, filtered at half-maximum Gaussian, and thresholded to only include voxels with at least 90% probability of containing gray matter. Quantified CBF maps were then downloaded to a local server, and the data for each participant were further thresholded by removing physiologically improbable values (voxels with CBF values < 10 or > 150).
Regions of interest
FreeSurfer neuroanatomical labels from the Desikan-Killiany atlas 60 were used to extract cortical thickness and CBF estimates of nine a priori regions of interest (ROIs) in the frontal, parietal, and temporal lobes. Specifically, we examined CBF and cortical thickness estimates of regions that have been previously implicated in AD58,61: the superior and inferior parietal cortices, precuneus, posterior cingulate, inferior temporal cortex, entorhinal cortex, parahippocampal cortex, medial orbitofrontal cortex, and rostral middle frontal gyrus. Averages of bilateral CBF and cortical thickness values for each of these nine ROIs were used as outcome variables within the current study. See Figure 1 for a depiction of the ROIs used in the study. We focused on AD-vulnerable regions in an effort to understand the extent to which age-related structural and functional brain changes may be altered by mTBI.
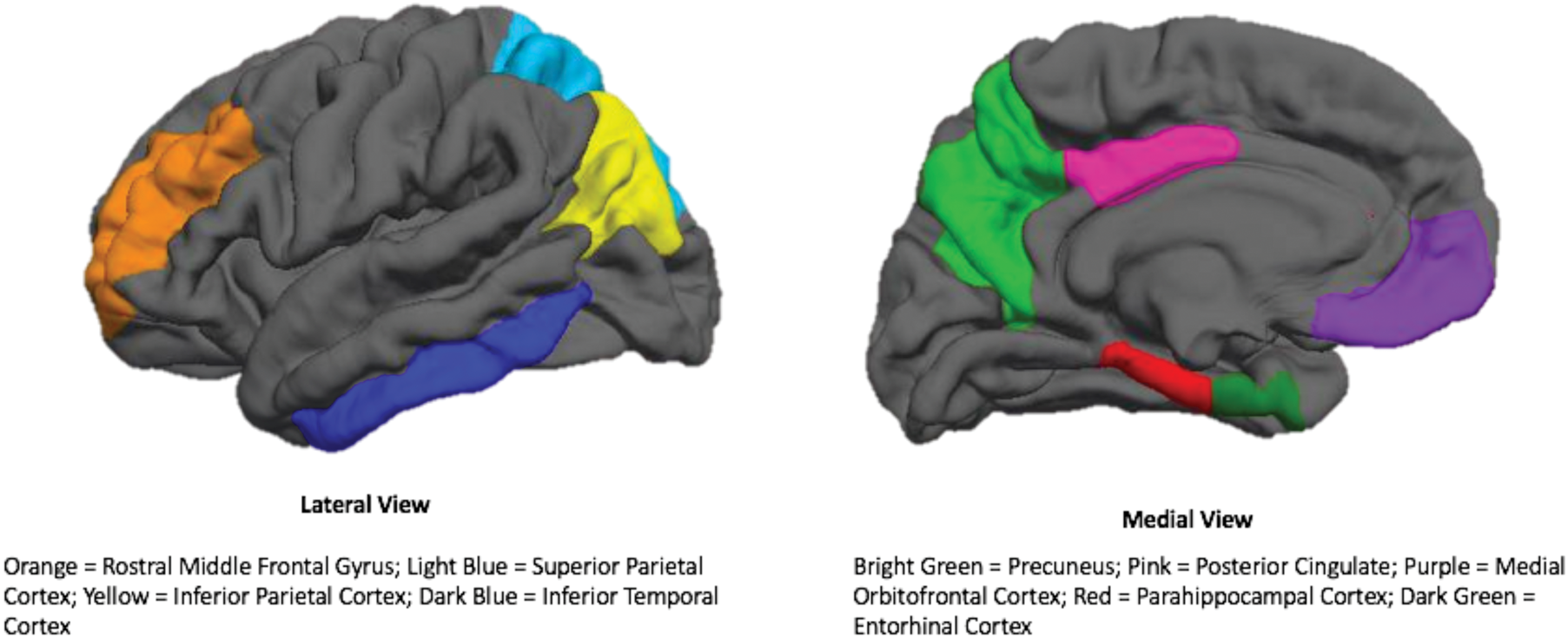
AD-vulnerable regions of interest utilized in the current study. Legend: Depictions of ROIs are of one hemisphere only. Left side of the figure is the lateral view of the brain. Orange: rostral middle frontal gyrus; Light Blue: superior parietal cortex; Yellow: inferior parietal cortex; Dark Blue: inferior temporal cortex. Right side of the figure is the medial view of the brain. Bight Green: precuneus; Pink: posterior cingulate; Purple: medial orbitofrontal cortex; Red: parahippocampal cortex; Dark Green: entorhinal cortex.
Statistical analyses
Analyses of variance (ANOVAs) were performed to determine whether the study groups (VCs, 1–2 mTBIs, 3+ mTBIs) differed on basic demographic variables, quantitative TBI injury characteristics, and self-report questionnaire data. Chi-squared analyses (or Likelihood ratios when cell counts were less than 5) were utilized to examine group differences on categorical demographic (e.g. sex, ethnicity) and TBI injury variables. Multiple linear regressions were used to determine whether group moderated the association between age and brain variables of interest (i.e. CBF and cortical thickness). All predictor variables were mean centered to reduce multicollinearity and analyses were conducted with these centered variables. All statistical analyses were conducted with Statistical Package for the Social Sciences (SPSS) version 25 62 and R version 3.5.0. 63
Results
Sample demographic and TBI injury characteristics
Participant demographics and TBI injury characteristics are presented in Table 1. The VCs, 1–2 mTBIs, and 3+ mTBIs groups did not differ on any demographic variables, including age and sex (all p’s > .05). With respect to clinical variables, results revealed the TBI groups did not differ on severity of post-concussive symptoms, but the three groups significantly differed in terms of psychiatric symptoms (p’s <.001); both TBI groups displayed worse depressive and PTSD symptoms relative to the VC groups (p’s <.001), but the 1–2 mTBIs and 3+ mTBIs did not significantly differ from one another (p’s >.08). As expected, the 3+ mTBIs group had a greater number of self-reported injuries that met clinical criteria for mTBI relative to the 1–2 mTBIs group (p <.001). Finally, there were no significant differences in the proportion of individuals that endorsed a LOC versus AOC for their most significant injury (p = .579) or time since injury intervals between the groups (p = .076), and blunt-force trauma was the primary mechanism by which these injuries occurred within each group.
Participant demographics and TBI injury characteristics.
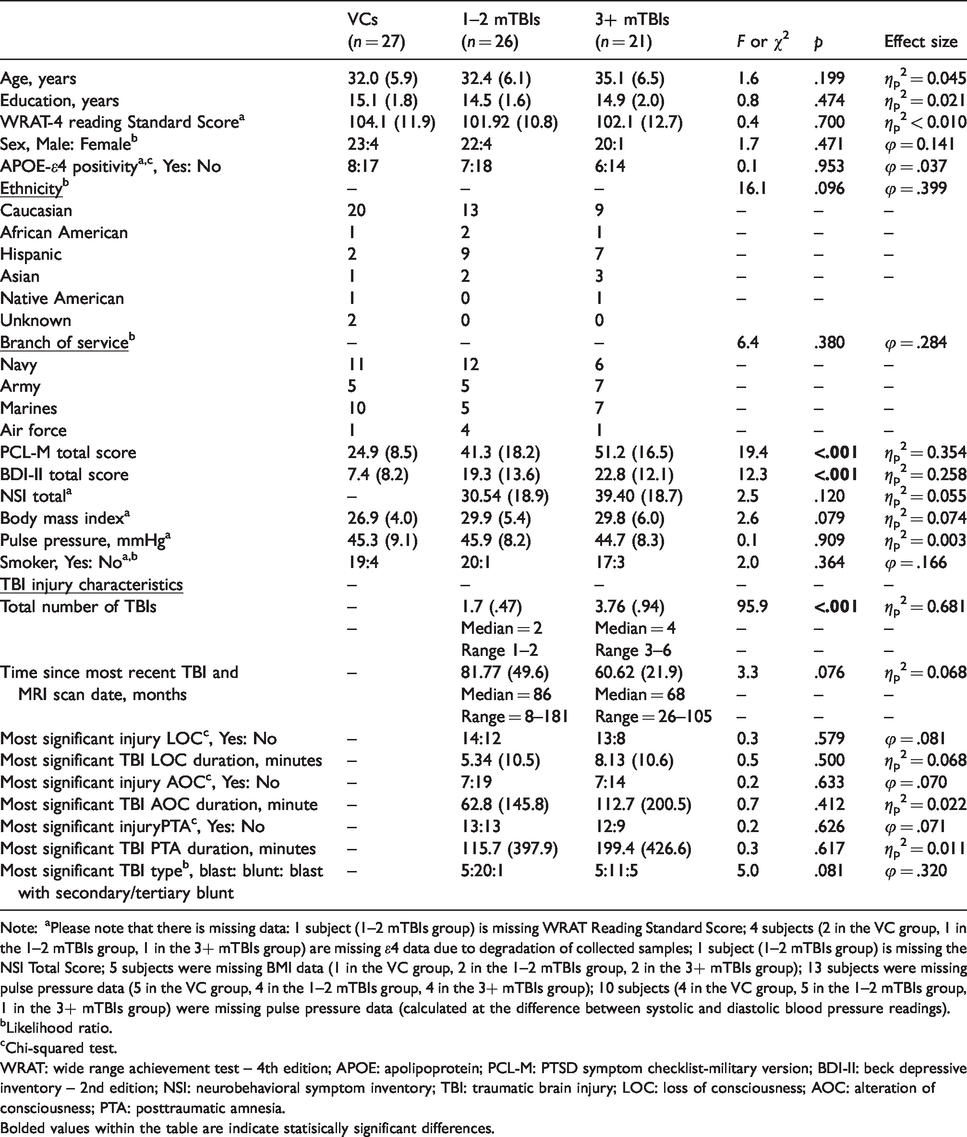
Note: aPlease note that there is missing data: 1 subject (1–2 mTBIs group) is missing WRAT Reading Standard Score; 4 subjects (2 in the VC group, 1 in the 1–2 mTBIs group, 1 in the 3+ mTBIs group) are missing ε4 data due to degradation of collected samples; 1 subject (1–2 mTBIs group) is missing the NSI Total Score; 5 subjects were missing BMI data (1 in the VC group, 2 in the 1–2 mTBIs group, 2 in the 3+ mTBIs group); 13 subjects were missing pulse pressure data (5 in the VC group, 4 in the 1–2 mTBIs group, 4 in the 3+ mTBIs group); 10 subjects (4 in the VC group, 5 in the 1–2 mTBIs group, 1 in the 3+ mTBIs group) were missing pulse pressure data (calculated at the difference between systolic and diastolic blood pressure readings).
bLikelihood ratio.
cChi-squared test.
WRAT: wide range achievement test – 4th edition; APOE: apolipoprotein; PCL-M: PTSD symptom checklist-military version; BDI-II: beck depressive inventory – 2nd edition; NSI: neurobehavioral symptom inventory; TBI: traumatic brain injury; LOC: loss of consciousness; AOC: alteration of consciousness; PTA: posttraumatic amnesia.Bolded values within the table are indicate statisically significant differences.
Group × age on CBF of AD vulnerable regions
A series of multiple linear regressions were performed in order to determine whether group (VCs, 1–2 mTBIs, 3+ mTBIs) moderated the association between age and CBF for each ROI. The dependent variable of interest in each model was CBF of the relevant ROI. The main independent variable of interest was the group × age interaction term, and age, group, sex, and PCL-M total score were also included in the model.
Results revealed that, independent of sex and PCL-M total score, there were significant group × age interactions for resting CBF of several parietal ROIs including the superior parietal cortex (unstandardized B = −0.33, β = −0.45, t = −3.62, p = .011, partial correlation = −0.28), inferior parietal cortex (unstandardized B = −0.27, β = −0.39, t = −2.20, p = .031, partial correlation = −0.25), and precuneus (unstandardized B = −0.36, β = −0.41, t = −2.47, p = .016, partial correlation = −0.26). Examination of simple main effects revealed that, within the 3+ mTBIs group, increasing age was strongly associated with lower CBF in the superior parietal cortex (r = −.76, p < . 001), inferior parietal cortex (r = −.67, p = .001), and precuneus (r = −.78, p < .001; see Figure 2). No such associations between age and CBF were observed in the VCs (r’s = −.14 to −.25, p’s = .21 to .47) or 1–2 mTBIs group (r’s = −.08 to −.03, p’s = .68 to .88).
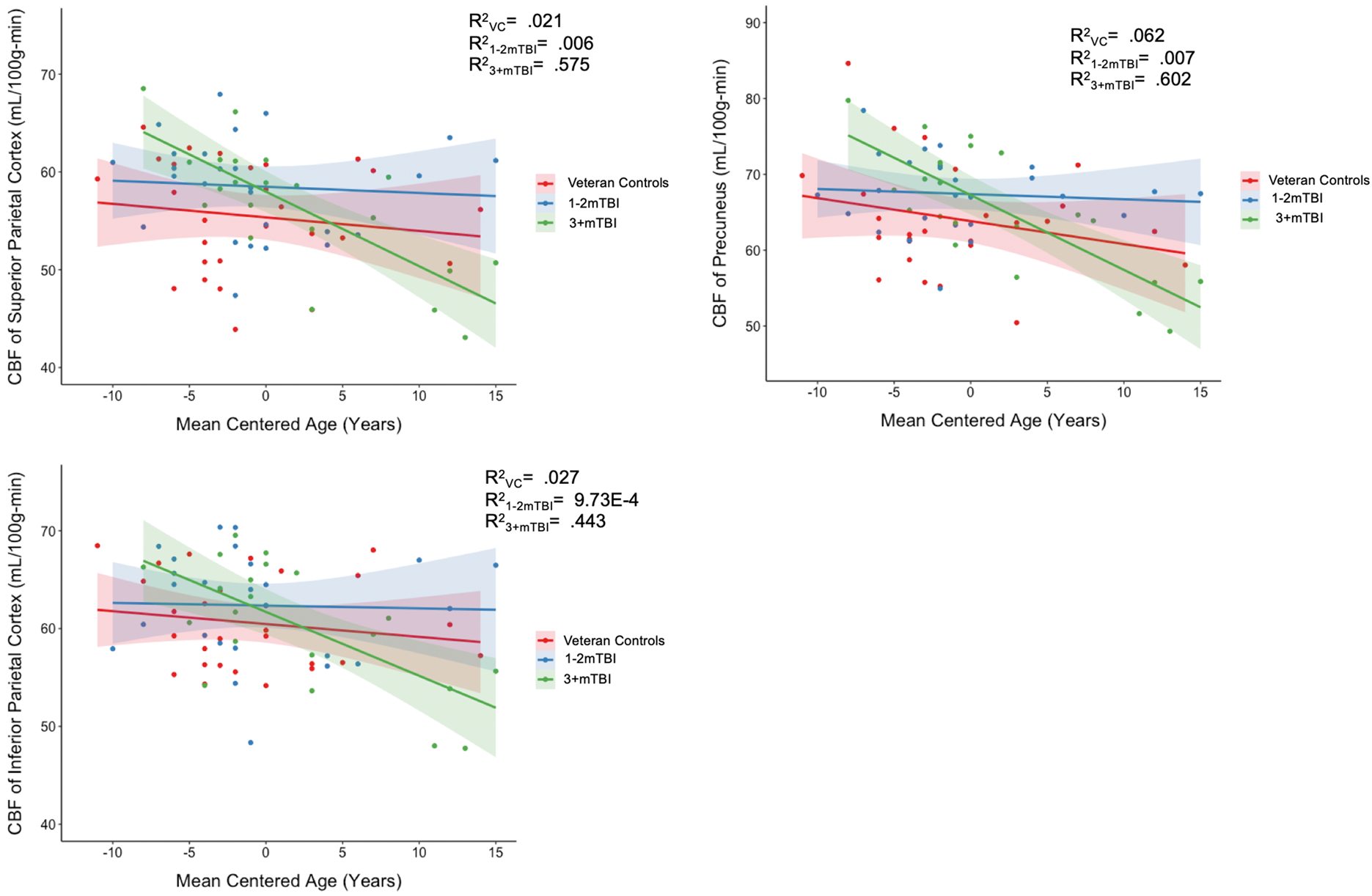
Plots of significant TBI × age interactions on CBF of AD vulnerable regions. Legend: Left upper part of the figure depicts CBF of superior parietal cortex in mL/100 g-min on the Y axis and mean age centered in years on the X axis. Red dots and line represent the VCs group. Blue dots and line represent the 1–2 mTBIs group. Green lines and dots represent the 3+ mTBIs group. Left bottom part of the figure depicts CBF of inferior parietal cortex in mL/100 g-min on the Y axis and mean age centered in years on the X axis. Red dots and line represent the VCs group. Blue dots and line represent the 1–2 mTBIs group. Green lines and dots represent the 3+ mTBIs group. Right part of the figure depicts CBF of Precuneus in mL/100 g-min on the Y axis and mean age centered in years on the X axis. Red dots and line represent the VCs group. Blue dots and line represent the 1–2 mTBIs group. Green lines and dots represent the 3+ mTBIs group.
Additional analyses revealed that there were no significant group − age interactions for the posterior cingulate (unstandardized B = −0.23, β = −0.19, t = −1.10, p = . 273, partial correlation = −0.12), inferior temporal cortex (unstandardized B = −0.29, β = −0.35, t = −1.89, p = .064, partial correlation = −0.22), entorhinal cortex (unstandardized B = −0.25, β = −0.22, t = −1.16, p = .249, partial correlation = −0.14), parahippocampal cortex (unstandardized B = −0.13, β = −0.13, t = −0.72, p = .472, partial correlation = 0.08), medial orbitofrontal cortex (unstandardized B = −0.14, β = −0.17, t = −0.92, p = .361, partial correlation = −0.10), or rostral middle frontal gyrus (unstandardized B = −0.19, β = −0.22, t = −1.24, p = .221, partial correlation = −0.14).
Group × age on cortical thickness of AD vulnerable regions
A series of multiple linear regressions were performed in order to determine whether group (VCs vs. 1–2 mTBIs vs. 3+ mTBIs) moderated the association between age and cortical thickness for each ROI. The dependent variable of interest in each model was cortical thickness of the relevant ROI. The main independent variable of interest was the group × age interaction term, and age, group, sex, and PCL-M total score were also included in the model.
Results revealed that, independent of sex and PCL-M total score, there were no significant group × age interactions for any of the ROIs (superior frontal gyrus [unstandardized B = −0.004, β = −0.20, t = −1.09, p = .277, partial correlation = −0.13], inferior parietal cortex [unstandardized B = 0.001, β = 0.06, t = −0.30, p = .763, partial correlation = 0.04], precuneus [unstandardized B = 0.002, β = 0.10, t = 0.57, p = .574, partial correlation = 0.06], posterior cingulate [unstandardized B = 0.004, β = −0.24, t = −1.32, p = .191, partial correlation = 0.15], inferior temporal cortex [unstandardized B = −0.000, β = 0.02, t = 0.09, p = .927, partial correlation = 0.01], entorhinal cortex [unstandardized B = 0.01, β = 0.27, t = 1.47, p = .146, partial correlation = 0.17], parahippocampal cortex [unstandardized B = 0.002, β = 0.045, t = 0.23, p = .815, partial correlation = 0.03], medial orbitofrontal cortex [unstandardized B = −0.002, β = −0.12, t = −0.69, p = .495, partial correlation = −0.08], or rostral middle frontal gyrus [unstandardized B = 0.001, β = 0.05, t = 0.26, p = .793, partial correlation = 0.03]).
Discussion
The current study sought to clarify (1) neural mechanisms underlying poor long-term outcomes and (2) factors that may relate to increased risk for late-life cognitive impairment in those with history of mTBI. Specifically, we explored whether repetitive mTBI was associated with greater age-related changes in CBF and cortical thickness of AD-vulnerable regions. Results showed that, independent of sex and posttraumatic symptoms, increasing age was associated with decreased CBF of several parietal regions (i.e. inferior and superior parietal cortex, precuneus) in the 3+mTBI group, but not in the VC or 1-2 mTBIs group. However, in contrast, repetitive mTBI was not associated with age-related changes in cortical thickness in any a priori region explored in this study. These findings suggest that, among Veterans with history of mTBI, those with repetitive (i.e. 3+) injuries are at increased risk for age-related CBF reductions. Importantly, these functional changes occur in AD-vulnerable regions and appear to precede age-related neurodegenerative or structural alterations.
The primary finding of this study was that the 3+ mTBIs group displayed greater age-related decreases in CBF relative to the VCs or 1–2 mTBIs group. As noted by Tagge et al., 64 a single impact injury has been demonstrated to initiate microvascular pathology in mice, but multiple concussive events conferred both traumatic microvascular injury and BBB breakdown in post-mortem brains of athletes. Importantly, sustaining one mTBI places an individual at an increased risk for another, 65 and research from studies of sports concussion suggests that repetitive head trauma may have detrimental effects on cerebrovascular function.39,66 For example, a study of amateur athletes revealed that after two months of training and competing, BBB pathology was evident on dynamic contrast enhancing MRI in nearly 40% of football players relative to control athletes with no history of head-trauma. 67 Moreover, a head impact monitoring study of female soccer players revealed that athletes with a higher cumulative load of concussive impacts (i.e. impacts accelerations >20 g) experienced pre- and post-season changes in cerebrovascular reactivity. 66 Thus, our results align with existing literature demonstrating links between repetitive mTBI and vascular pathology, which likely contributes to cerebrovascular dysfunction.
Within the 3+ mTBIs group, age-related decreases in CBF were largely posterior in distribution and limited to the parietal lobe. Although frontal and temporal regions are generally considered most vulnerable to injury in mTBI,68,69 CBF changes in these posterior parietal regions have repeatedly been implicated in the aging/AD literature.58,70–72 For example, decreased CBF in the lateral temporo-parietal cortex has been observed in adults at risk for AD, and more specifically, CBF reductions in the precuneus, cuneus, and inferior parietal lobe have been observed at least three years prior to conversion from mild cognitive impairment to AD. 73 Moreover, Lee et al. 74 showed that greater parietal white matter hyperintensity burden—a common consequence of reduced CBF—has been observed more than two decades prior to symptom onset in individuals with autosomal dominant AD. These vascular changes may represent the earliest markers of age-related neural inefficiencies that contribute to dementia risk and, importantly, may have an accelerated onset in those with repetitive mTBI.
Although we observed age-related decreases in CBF within the 3+ mTBIs group, there were no group differences in the association between age and cortical thickness within the sample. These results demonstrate that in young to middle-aged Veterans with repetitive mTBI, functional changes may precede age-related structural changes. Importantly, these findings align with the vascular hypothesis of AD which suggests that cerebrovascular dysfunction occurs prior to—and is even responsible for initiating—neurodegenerative processes in late-life. 23 Both cerebrovascular damage and dysfunction have been linked to increased amyloid-β and tau pathology, and increased accumulation of these pathological proteins has negative consequences on neuronal health.21,22 However, it is important to note that our null structural findings are in direct contrast to a study which showed greater age-related cortical thinning in frontal, temporal, and parietal regions that have also been implicated in AD within a similarly aged sample of active duty service members with mTBI. 32 Notably, only individuals reporting persisting post-concussive symptoms were included in the mTBI group, and thus discrepancies with our study may be the consequence of differences in sample characteristics, as we included mTBI individuals both with and without persisting complaints.
Although our mTBI sample did not demonstrate age-related cortical thinning, it is important to recognize that there is a complex relationship between CBF changes and brain structure that warrants additional exploration. Within the aging literature, vascular risk factors (i.e. hypertension, diabetes) and CBF changes have been directly linked to white or gray matter structural changes. Moreover, several theories suggest that mTBI-related CBF changes may drive neurodegenerative changes in late-life.7,10,75In an effort to understand this potential link, we conducted a series of partial correlations controlling for age, which demonstrated no significant associations between CBF and cortical thickness within the groups. It is possible that mTBI-related CBF changes may need to persist for a prolonged period of time before structural alterations are observed, and that these may only become apparent with increased age. Additional prospective and longitudinal work is needed to more fully examine the interplay between structural and functional brain changes in individuals with mTBI.
Interestingly, several recent studies have demonstrated that mTBI is also associated with an earlier age of dementia onset.7,8 In an effort to clarify mechanisms underlying this risk following mTBI, we chose to focus on how mTBI might initiate CBF and cortical thickness in regions that have been linked to onset and progression of AD processes. Our goal was to identify, in an a priori targeted approach, brain changes within vulnerable regions that may predispose an individual to dementia risk. However, TBI is a highly heterogeneous injury that can cause both focal and diffuse brain changes, and mTBI-related vascular and structural changes in other regions may be playing an important role. We conducted a series of exploratory analyses which yielded no other significant group × age interactions across averaged bilateral CBF and cortical thickness estimates of any other FreeSurfer-derived ROI. Thus, our findings illustrate that cerebrovascular dysfunction within specific AD-vulnerable regions represents an important mechanism for dementia risk following mTBI.
Within the extant literature, repetitive mTBI is most commonly associated with chronic traumatic encephalopathy—a primary tauopathy—that is thought to be fundamentally distinct from AD. Currently, there is considerable debate about the clinical and pathological features of CTE, and therefore its prevalence in individuals that have sustained a single or multiple mTBIs is largely unknown.76,77 Nevertheless, recent research has demonstrated that CTE-mediated tau pathology has negative effects on cerebrovascular functioning.41,64 As such, it is also possible that an independent tau-mediated process confers age-related CBF reductions in other neural regions. Alternatively, polypathology associated with unique or independent neurodegenerative processes is common following mTBI, 76 and multiple processes may be at play—as they may also be in AD. 78
The epidemiology of repetitive mTBI in military samples remains poorly understood, as most Veteran mTBI studies primarily focus on injuries sustained during deployment or enlistment in the military and do not assess injuries that may have occurred at other epochs in time.19,79 In contrast, we conducted comprehensive interviews surrounding potential head-injuries that Veterans may have sustained prior to, during, or after discharge from the military. Results revealed that, on average, the sample met criteria for more than one mTBI throughout their lifetime. Interestingly, our findings raise the possibility of a potential threshold effect in which Veterans with three or more injuries may be more likely to experience CBF reductions as they age. Notably, a similar threshold has been observed in other behaviorally and psychiatrically focused civilian and military studies.44,45 Preliminary work from our lab also suggests that compensatory neurovascular processes occur even in those with fewer than two injuries, but are not evident in those three or more injuries. 80 Relatedly, although Table 1 focuses on mechanisms of injury related to their “most significant TBI,” many individuals reported experiencing injuries of other or mixed mechanisms as well. While it is difficult to tease apart potential brain effects across mechanisms of injury within the framework of the current study, some human mTBI studies have shown that brain changes differ by mechanism,81–84 whereas others have not.85,86 Additional research is needed in order to disentangle the extent to which chronic pathological and behavioral consequences may differ between those within single versus multiple mTBIs, as well as to delineate effects that emerge at certain injury thresholds and injury mechanism subtypes.
There are several limitations to our study that warrant careful consideration. First, our sample consisted of predominantly young and middle-aged Veterans, and findings need to be replicated in an older sample. It is possible that cortical thinning occurs at a later time point (i.e. after the 5th decade of life) and our null findings are a product of the relative limited age range of the sample. Nevertheless, these findings are important as they show mTBI modifies age-related CBF associations in relatively young participants, that functional changes precede structural changes, and that CBF dysfunction may represent a potential point of intervention in reducing risk for poor long-term outcomes. Second, there was some sample variability in time since injury between our mTBI groups, as our 3+ mTBI group was slightly closer to their last reported injury. However, we conducted a series of sensitivity analyses within the TBI groups to ensure findings were independent of injury interval discrepancies and all observed findings remained the same. Please note significant results for this two group (1–2 mTBIs vs. 3+ mTBIs) × age interaction on CBF are presented in a regression table included in the supplemental material. Third, while our groups were comparable on APOE-ε4 positivity ratios, previous work in animals and humans has highlighted that individuals at genetic risk may be at increased risk for brain changes following TBI.87,88 As such, we conducted a series of exploratory analyses within the mTBI group, but did find any significant APOE-ε4 × age interactions within the TBI group. However, we did note some large effect sizes for the interaction between APOE-ε4 × age on CBF and cortical thickness in the posterior cingulate, precuneus, and inferior temporal cortex (partial eta 2 > .025), and future efforts from our laboratory will focus on examining how aging trajectories in the aftermath of TBI may be altered within the context of APOE-ε4. Finally, we explored how mTBI altered age-related CBF and cortical thickness estimates, and future work in older samples should directly relate brain variables to functional outcome measures in an effort to clarify posited relationships. Nevertheless, our results provide unique insights into a distinct pattern of age-related CBF changes that emerge in those with histories of repetitive head trauma.
Conclusion
To our knowledge, this is the first known study to demonstrate repetitive mTBIs in Veterans is associated with greater age-related reductions in CBF but not cortical thickness. These results provide critical insight into potential mechanisms of poor outcomes observed in individuals diagnosed with progressive cognitive impairment in late-life. Moreover, they align with recent research highlighting that mTBI can be conceptualized as a vascular insult and that cerebrovascular dysfunction may underlie chronic behavioral dysfunction and poor functional outcomes seen in many individuals with chronic TBI. Future research should focus on replicating these findings in larger samples with inclusion of older participants, and further investigate relationships to cognitive, behavioral, and health outcomes observed in at-risk older adults with history of mTBI. Finally, future longitudinal studies are needed to clarify the temporal course and neurobehavioral consequences of TBI-related pathological processes in order to enhance potential intervention and prevention opportunities.
Supplemental Material
JCB897443 Supplemental Material - Supplemental material for Repetitive mTBI is associated with age-related reductions in cerebral blood flow but not cortical thickness
Supplemental material, JCB897443 Supplemental Material for Repetitive mTBI is associated with age-related reductions in cerebral blood flow but not cortical thickness by Alexandra L Clark, Alexandra J Weigand, Katherine J Bangen, Victoria C Merritt, Mark W Bondi and Lisa Delano-Wood in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Veterans Affairs and Department of Defense grants awarded to Dr. Delano-Wood (829-MR-NB-25860; W81XWH-10-2-0169). This work was further supported by The National Institutes of Health (National Institute of Neurological Disorders and Stroke Ruth-Kirschstein Fellowship: F31NS09870) and a Veterans Affairs Advanced Polytrauma and TBI Rehabilitation Research Fellowship awarded to Dr. Clark.
Acknowledgements
The authors would like to thank all of the OEF/OIF/OND Veterans for their service and are extremely grateful to those who agreed to participate in this study. The authors also appreciate the Veterans Affairs San Diego Healthcare System (VASDHS) Center for Stress and Mental Health (CESAMH) for their organizational assistance, as well as the dedicated research assistants and volunteers who devote their time to our research laboratory.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
AC contributed to manuscript conception. AC performed some of the data collection, processed all of the neuroimaging data, and conducted the statistical analyses. AC, AW, and KB provided feedback on analyses and assisted with interpretation of results. AC drafted the paper with intellection contributions from AW and VM. Paper comments and revisions were provided by KB, AW, VM, MB, and LD-W. All authors declare no conflicts of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
