Abstract
We examined the neural mechanisms for increases in regional cerebral blood flow (rCBF) in the neocortex associated with mastication, focusing on the cortical vasodilative system derived from the nucleus basalis of Meynert (NBM). In pentobarbital-anesthetized rats, parietal cortical rCBF was recorded simultaneously with electromyogram (EMG) of jaw muscles, local field potentials of frontal cortex, multi-unit activity of NBM neurons, and systemic mean arterial pressure (MAP). When spontaneous rhythmic EMG activity was observed with cortical desynchronization, an increase in NBM activity and a marked rCBF increase independent of MAP changes were observed. A similar rCBF increase was elicited by repetitive electrical stimulation of unilateral cortical masticatory areas. The magnitude of rCBF increase was partially attenuated by administration of the GABAergic agonist muscimol into the NBM. The rCBF increase persisted after immobilization with systemic muscle relaxant (vecuronium). rCBF did not change when jaw muscle activity was induced by electrical stimulation of the pyramidal tract. The results suggest that activation of NBM vasodilator neurons contributes at least in part to the rCBF increase associated with masticatory muscle activity, and that the NBM activation is induced by central commands from the motor cortex, independently of feedback from brainstem central pattern generator or contracting muscles.
Introduction
In addition to its role in feeding and digestion, mastication has been suggested to have a beneficial role in maintaining higher brain functions such as enhancing alertness and preventing cognitive decline.1–5 It has been shown by various methods (PET, fMRI, Xe-CT, ultrasound Doppler, etc.) in humans that regional cerebral blood flow (rCBF) in widespread cortices is increased during chewing.6–9 For example, by using positron emission tomography, it has been shown that the rCBF increased in the sensorimotor cortex, supplementary motor area, insular cortex, cerebellum and striatum during mastication in humans and the maximum increase of 25–28% was observed in primary sensory motor areas. 6 Using ultrasound Doppler flowmetry, the middle cerebral artery blood flow increased by nearly 20% during the mastication. 10 In conscious cats, it has been shown by the laser Doppler method that the rCBF of the frontal lobe increased during mastication. 11 However, the neural mechanisms involved in such rCBF increases associated with mastication remain undetermined.
The basal nucleus of Meynert (NBM) and septal complex in the basal forebrain that send cholinergic fibers widely to the cerebral cortices and hippocampus are significantly degenerated in Alzheimer's disease and have an essential role in cognitive function.12–16 Stimulation of NBM and septal nuclei leads to a marked increase in rCBF17,18 and changes in neuronal activity patterns19–23 in the cerebral cortex and hippocampus. It has been hypothesized that basal forebrain cholinergic vasodilative systems may be activated during walking and contribute to an increase rCBF in the cerebral cortices and hippocampus. This assumption is based on the fact that, during treadmill walking in conscious rats, the extracellular acetylcholine is increased in the cerebral cortex and hippocampus and rCBF increases independently of blood pressure change, and that the rCBF change is partially attenuated by acetylcholine receptor blockers.24–26 A recent in vivo calcium imaging study in mice suggested that cholinergic neurons in the basal forebrain were consistently excited during movement, including running and licking. 27 However, direct evidence of involvement of NBM activation on rCBF changes associated with movement, by simultaneous recording nor inactivating the NBM is lacking.
The increase in rCBF during walking or mastication can be due to reflex activation of basal forebrain vasodilative fibers via excitation of somatic afferents during limb or jaw movements. Because basal forebrain neurons are excited during noxious28,29 or non-noxious 30 somatic stimulation which increases rCBF,31–33 and the rCBF response is attenuated by inactivating the NBM using local muscimol injection. 33
Another possible trigger to increase rCBF during walking or mastication is the central motor command. The central command from the motor cortex produces cardiovascular responses via changing autonomic efferent nerve activities during exercise.34,35 A similar mechanism initiated by the central command may act to increase rCBF during walking or masticatory movement, but this possibility has not been clarified yet.
Based on the above findings, we hypothesized that the basal forebrain cholinergic vasodilative system is activated to increase the cerebral cortical rCBF during the masticatory movement, which is a major physical activity of daily living like walking. In this study, we aimed to investigate the neural mechanism of rCBF increase associated with masticatory movement, focusing on NBM that is important for cognitive function and cortical rCBF regulation.
Masticatory movement can be observed spontaneously or induced by electrical stimulation of the cortical masticatory areas or axonal tract from them in anesthetized animals.36–41 The model is beneficial to record NBM neuronal activity simultaneously with rCBF, and then to examine effects of inhibiting the NBM activation on rCBF response. Further, importantly, we can apply a muscle relaxant to clarify contribution of peripheral somatosensory inputs. Therefore, using anesthetized rats, we examined whether local field potential (LFP) and rCBF in the cerebral cortex, blood pressure and NBM neuronal activity changes in association with the jaw muscle electomyographic (EMG) activity observed spontaneously or induced by focal cerebral stimulation. Furthermore, we examined the effect of inhibition of NBM neuronal activity on rCBF responses. Also, using a muscle relaxant, we examined whether removal of peripheral somatosensory input affects the rCBF response.
Materials and methods
All animal experiments were performed at the Tokyo Metropolitan Institute of Gerontology (TMIG) and were approved by the TMIG Animal Care and Use Committee. This study was conducted in accordance with Guidelines for Proper Conduct of Animal Experiments (established by Science Council of Japan in 2006), and the experiments reported are in compliance with the ARRIVE guidelines. The experiments were performed using adult male Wistar rats (n = 24, 3 to 6 months old). The weight of the animals was in the range of 300–415 g. Animals were anesthetized with pentobarbital in combination with a local anesthetic. The initial dose of pentobarbital was 50 mg/kg i.p. For local anesthesia 1% procaine hydrochloride or 2% xylocaine jelly was applied around the incision area before and after surgery. Pentobarbital supplements of 15 mg/kg were given i.v. or s.c. every 30 minutes during surgery, and then decreased to 4–5 mg/kg (s.c. as needed) after surgery to maintain constant anesthesia and sedation levels. During the experiment, the depth of anesthesia was confirmed by observing respiration, blood pressure, and cortical electrical activity. When these conditions were unstable, extra pentobarbital was given. We selected pentobarbital for anesthesia because we have observed spontaneous rhythmic jaw movements in rats with this anesthetic.
Body temperature was maintained at 37–38°C with an automatically controlled heating pad and lamp (ATB-1100, Nihon Kohden, Tokyo, Japan). In all experiments, the trachea was intubated with a catheter to secure the airway, and the femoral venous and arterial catheters were used for drug administration and systemic arterial pressure recording, respectively. Experiments were performed under spontaneous respiration. In four rats in which the effect of immobilization with a muscle relaxant (vecuronium bromide, 2 mg/kg, i.v.) was examined, artificial ventilation was performed before immobilization. During artificial ventilation, end-tidal CO2 levels monitored with a gas analyzer (Microcap, Oridion Medical, Jerusalem, Israel) were adjusted to maintain at 4–5%, by changing the tidal volume and rate of respirator.
The rats were placed on a brain stereotaxic apparatus (SR-5, Narishige, Tokyo, Japan). EMG activity of the jaw muscles (Figure 1(a)), local cerebral cortical activity, and rCBF (open circles in Figure 1(b)) were recorded simultaneously in all 24 rats. Responses during spontaneous jaw movements were monitored in six rats and effects of electrical stimulation of cerebral masticatory areas or their axonal tract (closed circles in Figure 1(b)) were examined in the other 18 rats (supplementary Table 1).
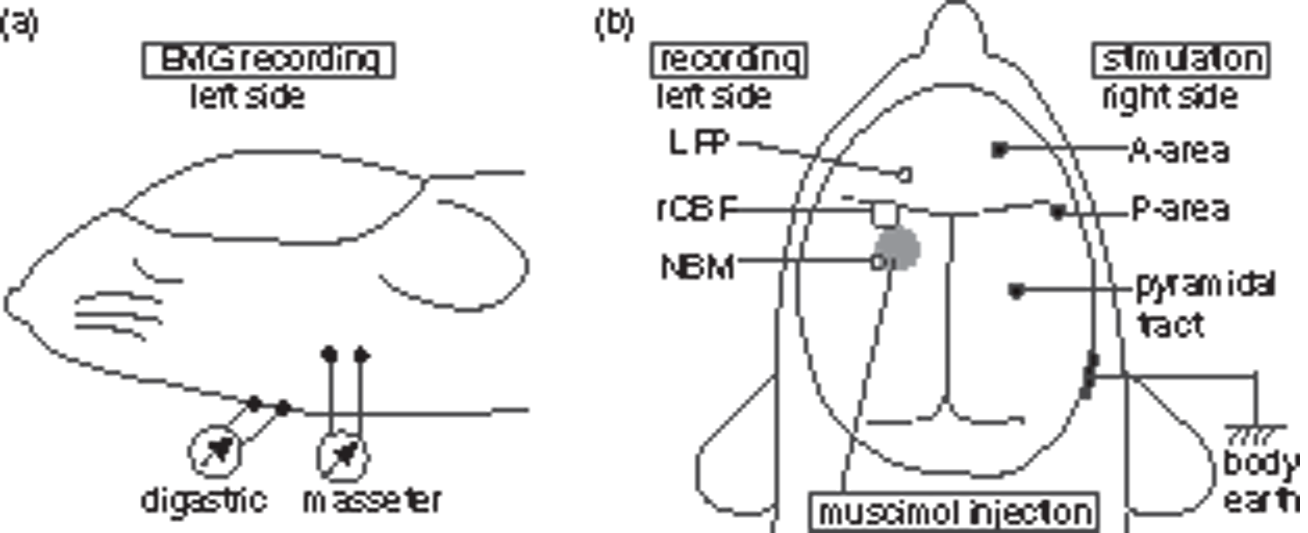
Schematic diagram showing the location of the EMG recording (a) and cerebral manipulation (b). (a) EMG recording from digastric and/or masseter muscles on the left side. (b) Recording of LFP, rCBF (by laser Doppler flowmetry), and NBM neuronal activity on the left side (open circles). Muscimol injection into left NBM (a gray circle). Stimulation of A-area, P-area, or pyramidal tract on the right side (closed circles).
Rats were euthanized by injecting an overdose of pentobarbital at the end of the experiment.
Measurement of EMG
In order to monitor the EMG activity of the jaw muscles, a pair of polyethylene-coated silver wires (approximately 5 mm between electrodes) with bare ends (1 mm) were inserted into the digastric (jaw opener muscle) and/or masseter (jaw closer muscle) muscles on the left side (contralateral to the stimulation site). EMG was amplified with an AC preamplifier (MEG-2100, Nihon Kohden, low cut 50 Hz, high cut 3 kHz), and digitized (see below).
Recording of local field potential (LFP)
After partial craniotomy of the frontal bone, coaxial metal microelectrodes (outer diameter 0.2 mm, Unique Medical, Tokyo, Japan) was inserted on the left frontal lobe (AP = +2 mm from bregma, L = 2 mm to the midline, 1 mm vertical under the brain surface) to monitor local cerebral cortical activity in primary motor area. The local field potential (LFP) was recorded and amplified with an AC preamp (MEG-2100, Nihon Kohden, low cut 0.5 Hz, high cut 10 kHz), and extracted offline from digitized records with band pass for 1–100 Hz. 20
Measurement of rCBF
rCBF was measured by laser Doppler flowmetry (in all 24 rats) or laser speckle contrast imaging (in five rats). Laser Doppler flowmetry: After partial craniotomy, the probe (diameter 0.8 mm) of laser Doppler flowmeter (ALF21D, Advance, Tokyo, Japan) was placed on the surface of the left parietal lobe (AP = +1 to −1 mm from bregma, L = 3 to 4 mm to the midline) avoiding large blood vessels and fixed with a balancing holder (ALF-B, Advance).
Laser speckle contrast imaging: The frontal and parietal bones were bilaterally drilled with a dental drill to thin the bones until the blood vessels on the surface of the brain were visible.33,42 The laser speckle contrast device was then fixed, the zoom was adjusted to cover the dorsal surface of the brain, and the polarizing lens was carefully adjusted to minimize specular reflection. Laser speckle contrast imaging was performed using a Moor full-field blood flow imaging device (Moor Instruments, Devon, UK) consisting of an infrared laser diode (785 nm wavelength) and a CCD camera. The field of view is approximately 300 mm2 (20 × 15 mm) and the matrix is 760 × 568 pixels, exhibiting a resolution of approximately 26.4 µm per pixel. Images were sampled at 1 Hz with 4 ms exposure time. For quantification, six regions of interest (ROI) were set in the frontal (AP = +3.5 to +2.5 mm from bregma, L = 0.5 to 1.5 mm to the midline), parietal (AP = −0.5 to −1.5 mm, L = 3 to 4 mm), and occipital (AP = −4.5 to −5.5 mm, L = 3 to 4 mm) cortices bilaterally.
Recording of multi unitary action potential of the NBM
After partial craniotomy, a metal multi-channel microelectrode (300–400 kΩ resistance, Unique Medical) was inserted to the left NBM (2.3 mm posterior to the bregma, 3.7 mm lateral to the midline, 7.0 mm vertically under the bregma; AP: −2.3, L: 3.7, V: 7.0), and extracellular neuronal action potentials were recorded in six rats. Multiunit activity was amplified using AC preamplifiers (MEG-2100, Nihon Kohden, high cut 1 kHz, low cut 150 Hz) and digitized. The analog signal output was connected to an oscilloscope and a speaker, to confirm unitary activity from the waveform and sound of the action potential. NBM neurons projecting to the ipsilateral cortex were confirmed as projection neurons by antidromic stimulation of their axons according to a previous paper. 29 The electrode inserted into the frontal cortex for LFP recording (see above) was temporarily used as a stimulator for this purpose.
At the end of each experiment, 250 µA direct current was applied for 30 s to localize the NBM recording site. The rats were then euthanized by an overdose of pentobarbital and the brain was removed. Histological verification of the position of the electrode tip was performed using a 30 µm thick frozen cross sections.
Electrical stimulation of cerebral masticatory areas
After partial craniotomy, coaxial metal microelectrode (outer diameter 0.2 mm, Unique Medical) was inserted to the masticatory cortex or its axonal tract, i.e., pyramidal tract, unilaterally on the right side. The position of pyramidal tract for electrical stimulation was set at internal capsule caudal to the posterior end of the NBM.
According to the previous studies,36,37,41 repetitive stimuli with a pulse width of 0.2 ms and a frequency of 50 Hz were applied for 20 s at various intensities to produce rhythmic jaw movements similar to masticatory movements. The most effective stimulation site was determined by changing the position of the stimulus electrode rostro-caudally and mediolaterally.
After the experiment, a direct current of 250 µA was applied for 30 s to localize the cortical or pyramidal tract stimulation site, and the electrode position was confirmed histologically on a frozen coronal brain sections cut to a thickness of 30 µm.
Pharmacological inactivation of the NBM
Pharmacological inactivation of the NBM (a gray circle in Figure 1(b)) was performed in five rats using the method previously described, 33 with some modifications. A small hole was made in the occipital bone with a dental drill to inject solution into the left NBM (AP: −1.4 mm, L: 2.5 mm, V: 7.6 mm). 43 A microsyringe needle (Hamilton 65458-02, 32 gauge, Reno, NV, USA) was inserted into the NBM at an inclination of 30° off the coronal plane to move the needle caudally away from the rCBF imaging site. Muscimol (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in phosphate buffered saline (PBS), at a concentration of 0.2 mg/ml (1.75 mM). The solution was injected at a volume of 300 nl. Therefore, 60 ng of muscimol was administered in each animal. Control (vehicle) injections were 300 nl of PBS, which were tested in the same animals prior to muscimol. Injections were performed at a rate of 60 nl/min to minimize compression of brain tissue using a micro syringe pump (UMP 3 T-4, World Precision Instruments, Sarasota, FL, USA). After waiting at least for 30 min from the onset of infusion, we started stimulation. After the experiment, 300 nl of Evans blue was administered via the microsyringe to localize the injection site. The position of the needle was confirmed histologically on frozen coronal brain sections (thickness of 30 µm).
Data collection and analysis
All obtained analog signals (including systemic arterial pressure, rCBF, NBM neuronal activity, LFP and EMG) were digitized (Micro 1401, Cambridge Electronic Design, UK) and displayed on a computer monitor for online and offline analysis using Spike 2 software (Cambridge Electronic Design). LFP was expressed as RMS amplitude (time constant: 0.2 s) to summarize time course of desynchronization. Spike counts were performed for EMG and NBM neuronal activity, and an artifact was excluded if exist within latency of 0.5 ms from each stimulus pulse.
Values are expressed as the median and interquartile range (25%–75%). The time course of the changes in each parameter was assessed by one-way repeated measures analysis of variance (ANOVA) followed by Dunnett’s test. Paired t-tests were performed to compare difference of peak response between vehicle (PBS) and muscimol injection. Statistical significance was set at the 5% level. One or two trials in each rat were used for statistical analysis.
Results
Changes in rCBF and NBM activity associated with cortical desynchronization and spontaneous jaw muscle activity
After completion of the surgery, six rats were left unstimulated at least for 60 min. In each rat, the rCBF was stable for about 50 min. However, after that period, rCBF suddenly increased. The abrupt rCBF change was always associated with LFP desynchronization and also with rhythmic jaw movement represented as an increase in jaw muscle EMG (as shown in Figure 2(c)). A slight increase in MAP was also observed.
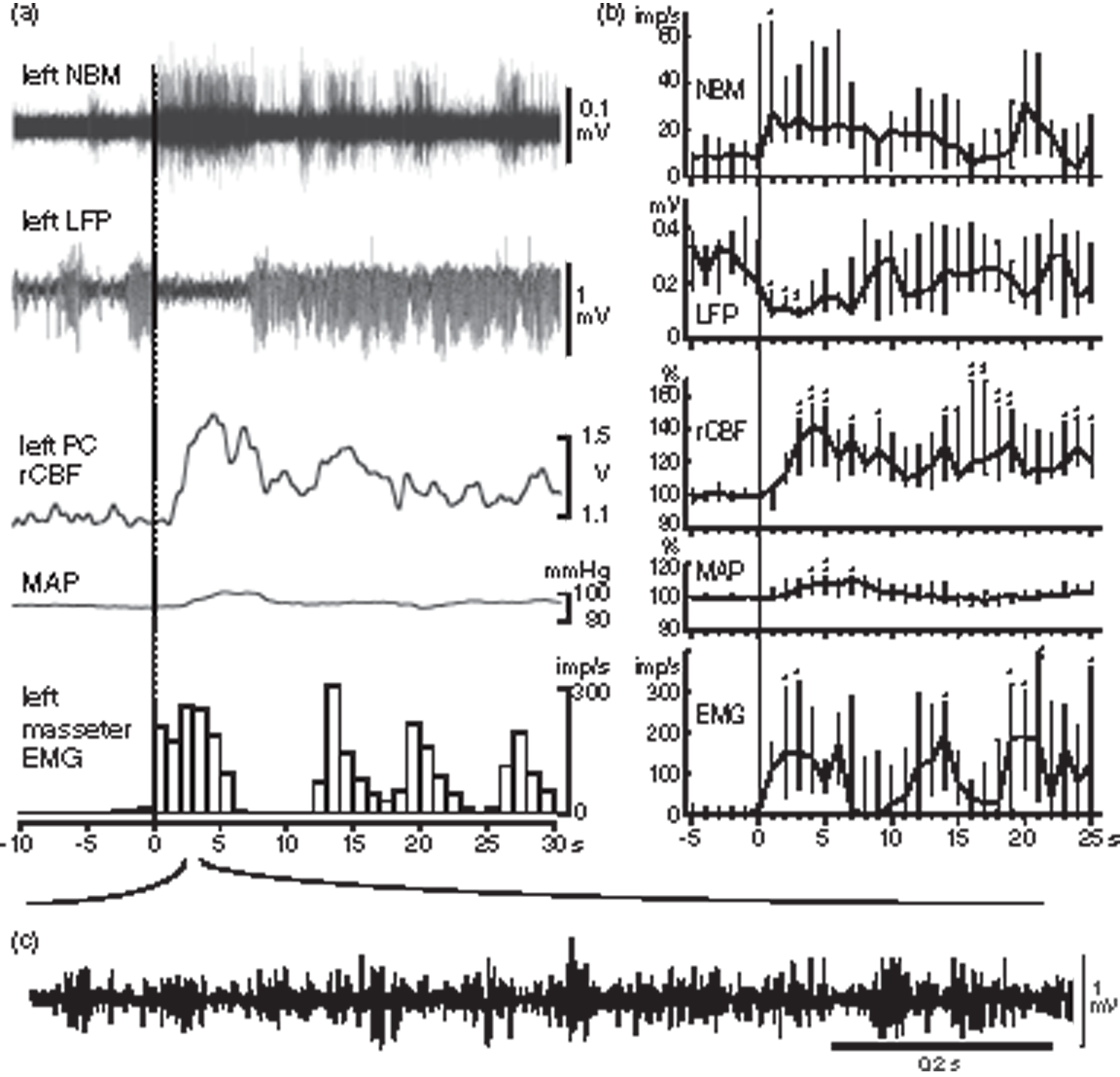
Spontaneous increases in rCBF and NBM neuronal activity associated with cortical desynchronization and jaw muscle activity. (a) A representative simultaneous recording of NBM activity, LFP in left frontal cortex, rCBF in left parietal cortex, MAP and EMG in left masseter muscle (from top to bottom) in one rat. Onset of an increase in EMG is indicated as time zero on the abscissa. EMG activity is shown by the number of action potentials counted every second. (b) Graphs summarizing time course of changes in each parameter every 1 s. Changes in rCBF and MAP are expressed as the percentages of corresponding control values (mean value for 10 s before time zero). Each point and vertical bar shows the median and interquartile range (25%–75%) (n = 5 from three rats). *p < 0.05, **p < 0.01; significantly different from control values by one-way repeated ANOVA followed by Dunnett’s multiple comparisons test. (c) Original recording of rhythmic EMG activity by expanding the time scale.
In three of the six rats, neuronal activity of NBM was simultaneously recorded with LFP, CBF, MAP, and EMG. As shown in a representative example (Figure 2(a)), the NBM neuronal activity always increased prior to an increase in rCBF. Figure 2(b) summarizes the time course of spontaneous changes in NBM activity, LFP, rCBF, MAP, and EMG simultaneously recorded in five cases in the three rats. The onset of increase in EMG activity was set as zero. The rCBF started to increase at 1.5 s (median) (range: 1.3 s–1.8 s) after the onset of EMG increase, reached a peak at 4 s (range: 3.6–4.5 s), and returned to the resting level within 2 min. In summary, rCBF increased by 42% (range: 18%–56%) of basal control level at 4 s, whereas MAP increased by 11% (range: 3%–12%) at 7 s. The NBM activity increased to 27 imp/s (range: 15 imp/s–66 imp/s) at 1 s after the onset of EMG increase from 9.3 imp/s (range: 0 imp/s–14 imp/s) of basal activity.
In half of the six rats examined, at 5–30 min prior to the remarkable rCBF change associated with LFP desynchronization and rhythmic jaw muscle EMG activity, similar rhythmic EMG activity was observed without any apparent changes in LFP, rCBF, nor NBM neuronal activity (data not shown).
Changes in rCBF induced by electrical stimulation of cortical masticatory areas
Two cortical areas that are known as cortical masticatory areas in rats were stimulated: (1) the anterior rhythmic jaw movement area (A-area), located in the deep part of the primary motor area (AP: +3.2 mm, L: 2.4 mm, V: 2.8 mm); and (2) the posterior rhythmic jaw movement area (P-area), located in the insular cortex (AP: +0.6 to 0 mm, L: 5.0 to 5.5 mm, V: 6.0 to 7.0 mm). Small amounts of muscle activity were induced by A-area stimulation and large amounts of muscle activity were induced by P-area stimulation, as reported previously.37,41 With either stimulation, induction of jaw muscle activity was always accompanied by both cortical desynchronization and rCBF increase (a typical example is shown in Figure 3(a) for A-area stimulation and supplementary Figure 1(A) for P-area stimulation). MAP increased slightly during P-area stimulation but did not change during A-area stimulation. Similar responses were reproduced by the same stimulation when applied after recovery of the response.
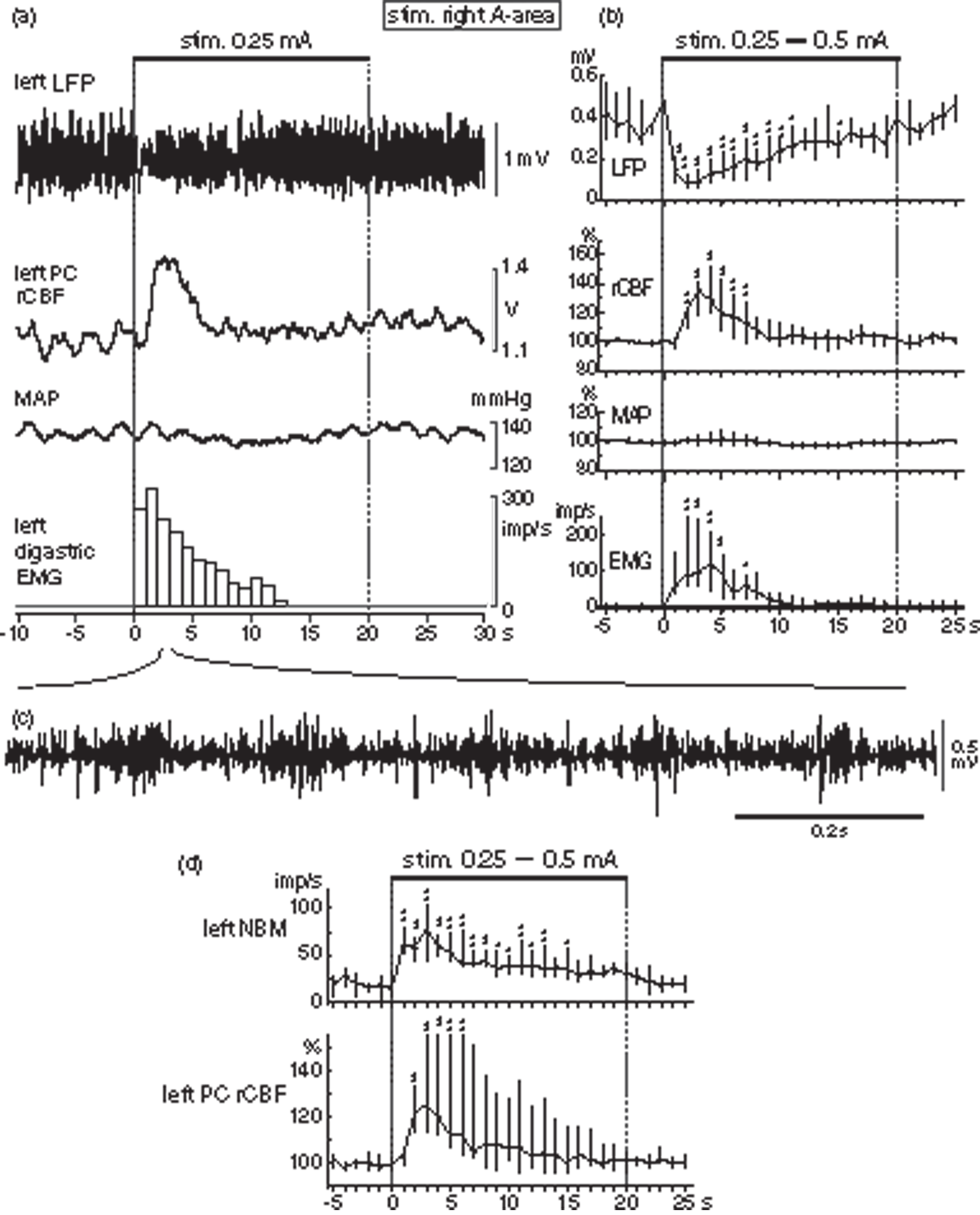
Increases in rCBF and NBM neuronal activity associated with cortical desynchronization and jaw muscle activity induced by electrical stimulation of A-area. (a) A representative simultaneous recording of LFP in left frontal cortex, rCBF in left parietal cortex, MAP and EMG in left digastric muscle (from top to bottom) in one rat. EMG activity is shown by the number of action potentials counted every second excluding stimulus artifacts. (b) Graphs summarizing time course of changes in each parameter every 1 s. Upper horizontal bar shows stimulation applied to right A-area with 0.25 or 0.5 mA pulse of 0.2 ms duration at 50 Hz for 20 s. Changes in rCBF and MAP are expressed as the percentages of corresponding control values at the onset of stimulation. Each point and vertical bar shows the median and interquartile range (25%–75%) (n = 6 from 6 rats). *p < 0.05, **p < 0.01; significantly different from control values by one-way repeated ANOVA followed by Dunnett’s multiple comparisons test. (c) Original recording of rhythmic EMG activity during A-area stimulation expanding the time scale. (d) Changes in NBM activity and rCBF simultaneously recorded (n = 6 from 3 rats). Other details are same as for (b).
The cortical stimulation started at an intensity of 0.25 or 0.5 mA and increased or decreased by two times or a half, and the threshold of rCBF increase was examined. The threshold intensity to increase rCBF or EMG activity for A-area stimulation tested in six rats was 0.25–0.5 mA, and the threshold for P-area stimulation tested in six rats was 0.1–0.25 mA. In both stimulations, the rCBF (supplementary Figure 2(A) and (B) upper graphs) and EMG activity (supplementary Figure 2(A) and (B) lower graphs) increased in a stimulus intensity dependent manner. Usually, the threshold and the stimulus intensity dependency for EMG response corresponded to those of rCBF response, but there were cases where the threshold for rCBF response was lower than that for EMG (in one rat for A-area stimulation, in another rat for P-area stimulation).
Figure 3(b) summarizes the time course of changes in LFP and rCBF of the cerebral cortex, MAP and jaw muscle EMG induced by A-area stimulation (0.25–0.5 mA) in six rats. The rCBF started to increase at 0.9 s (range: 0.8 s–1.4 s) after stimulus onset, peaked at 3.3 s (range: 2.7 s–3.9 s), reaching 36% (range: 19%–42% at 3 s), and returned to its original level within 10 s. The LFP amplitude reached its minimum at 1.9 s (range: 1.4 s–3.2 s) after the onset of stimulation, when frequency of EMG activity reached nearly its maximum. Since MAP did not change, rCBF increases induced by A-area stimulation appear to be due to active vasodilation in the cortex.
The time course of rCBF changes induced by P-area stimulation (supplementary Figure 1(B), n = 6) was similar to that of the A-area stimulation, and the rCBF increased by 56% (range: 38%–67%) at 4 s after the onset of stimulation. The increase in rCBF induced by P-area stimulation may, in part, due to a passive increase due to an increase in MAP, but the increase in MAP was only up to 11% (range: 9%–18%) at 4 s, which was much smaller than the increase in rCBF. The change in LFP amplitude was transient, reaching a minimum at 1 s after the onset of stimulation. In contrast, EMG activity persisted throughout a 20 s stimulation period.
Effects of A-area stimulation on neuronal activity of NBM
Since NBM causes dilation of cerebral cortical arteries independent of MAP change, NBM may contribute to an increase in rCBF induced by A-area stimulation. The effect of A-area stimulation on NBM neuronal activity was examined in three rats by simultaneous recording with rCBF and jaw EMG activity. The activity of NBM neurons increased prior to an increase in rCBF (Figure 3(d)), as seen during spontaneous jaw movement (Figure 2). The activity reached its peak at 1.8 s (range: 0.7 s–3.0 s), and then gradually decreased during stimulation toward the pre-stimulus level.
Effects of A-area stimulation on rCBF in widespread cortical areas
To examine whether unilateral A-area stimulation increases rCBF not only in the contralateral parietal lobe, but also in other cortical areas bilaterally, rCBF in the entire dorsal surface of the cerebral cortex was measured by means of laser speckle contrast imaging in five rats. rCBF increased during A-area stimulation bilaterally in widespread areas of the frontal, parietal and occipital lobes (Figure 4). The rCBF change in the left parietal lobe (contralateral to the stimulus) was similar in magnitude and time course that measured by laser Doppler flowmetry. Peak changes in the left frontal, parietal, and occipital lobes were 68% (range: 35%–76%), 44% (range: 26%–48%), and 9% (range: 2%–17%), showing larger increases in the frontal and parietal lobes, and a smaller increase in the occipital lobe. Furthermore, the peak changes on the right frontal, parietal and occipital lobes were equivalent to those changes in corresponding areas on the left side.
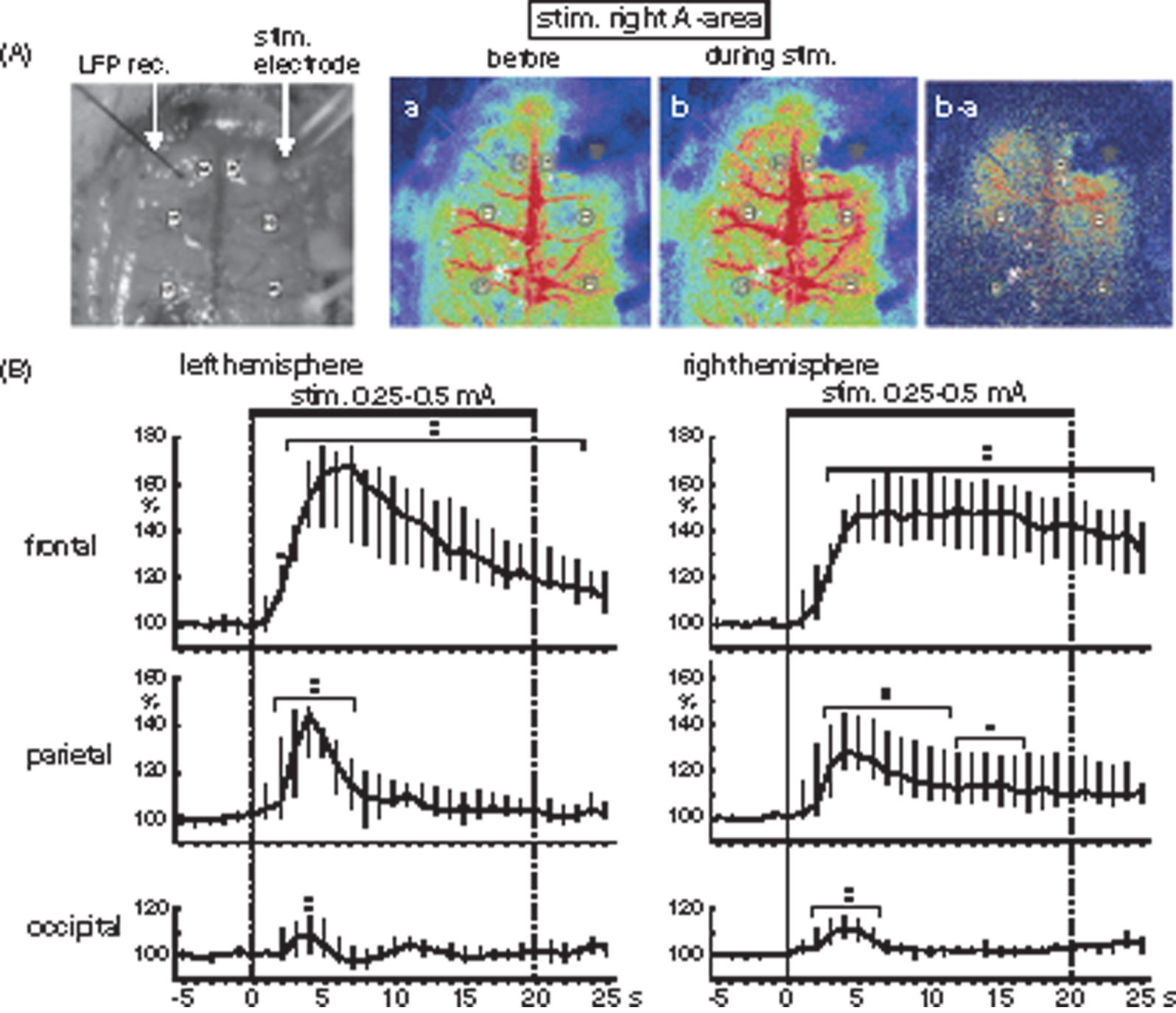
Effects of A-area stimulation on rCBF in widespread cortical areas. (A) Sample recordings of rCBF by laser speckle contrast imaging device in one rat. Monochrome photograph is a brain showing the stimulus electrode inserted into A-area and the recording electrode for LFP. Color photographs are laser speckle contrast imaging before stimulation (a), during stimulation (b), and subtract imaging of b minus a. (B) Graphs summarizing the time course of rCBF responses in the frontal, parietal and occipital cortices of left (contralateral to the stimulation) and right (ipsilateral to the stimulation) hemisphere (n=6 from 5 rats), extracted from the six ROIs indicated by the open circles in A. Other details are same as Figure 2(b).
Effects of inactivating the NBM on rCBF response induced by A-area stimulation
NBM neurons project unilaterally to the ipsilateral cortices. A small amount of muscimol was administered to the left NBM to inhibit its activation and examine the contribution of NBM excitation on the rCBF response to A-area stimulation in five rats. After muscimol was administered to the left NBM, parietal rCBF responses during A-area stimulation were attenuated selectively on the left side which was ipsilateral to the muscimol administration (Figure 5(a)). On the left side, the magnitude of rCBF response was 20% (range: 14%–27%) after muscimol, which was significantly smaller than that 32% (range: 25%–40%) after PBS (p = 0.0167 by two-tailed paired t-test). However, parietal rCBF response on the right side, which was contralateral to the injection, was equivalent after PBS and muscimol. When the vehicle (PBS) was administered, the rCBF responses were equivalent to those without PBS administration in both left and right side.
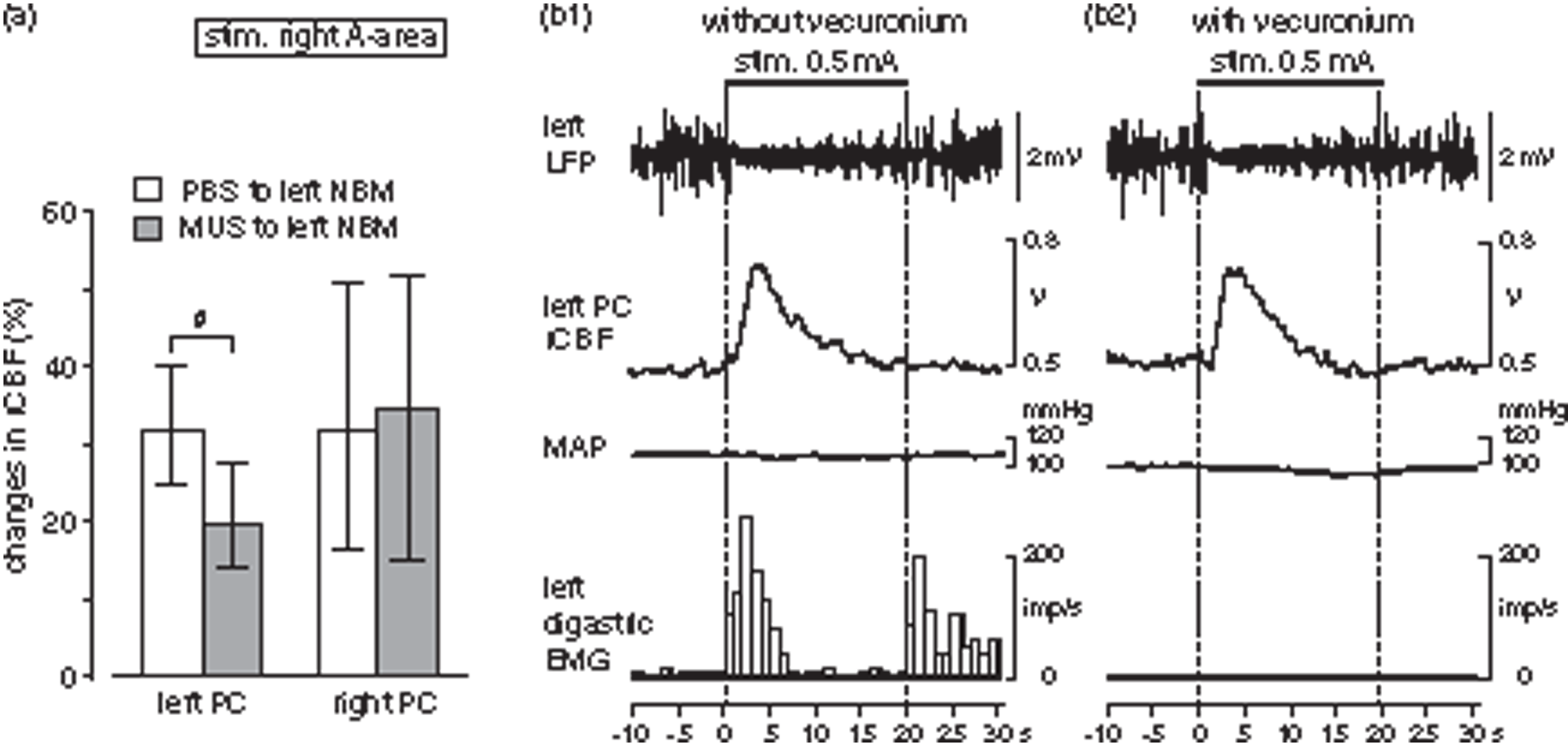
Effects of drugs on rCBF response in parietal cortex induced by A-area stimulation. (a) Effects of inactivating the NBM on rCBF response in parietal cortex induced by A-area stimulation. Changes in left (ipsilateral to administration) and right (contralateral to administration) rCBF after PBS or muscimol administration to the left NBM. Each column and vertical bar shows the median and interquartile range (25%–75%) (n=6 from 5 rats). # <0.05; significantly different between PBS (white column) and muscimol (dark column) administration by paired t-test. (b) Effect of muscle relaxation on rCBF response induced by A-area stimulation. Typical example of LFP, parietal rCBF, MAP and digastric EMG without (b1) and with (b2) vecuronium in one rat under artificial ventilation. EMG activity is shown by the number of action potentials counted every second excluding stimulus artifacts.
Effect of muscle relaxation on rCBF response induced by A-area stimulation
In order to investigate the possibility that peripheral somatosensory inputs due to muscle contraction might be involved in the increase in rCBF induced by A-area stimulation, responses were compared before and after immobilization using a muscle relaxant under artificial respiration. The control responses of LFP, rCBF, MAP and EMG without vecuronium obtained under artificial respiration (Figure 5(b1)) were all similar to those obtained under spontaneous respiration (see Figure 3). After vecuronium administration, the EMG activity observed during A-area stimulation almost completely disappeared (Figure 5(b2)). However, changes in LFP and rCBF in the cerebral cortex were equivalent before and after muscle relaxation (Figure 5(b1) and (b2)). Similar results were obtained in all four rats tested for A-area stimulation and also in all three rats tested for P-area stimulation.
Effect of pyramidal tract stimulation on rCBF
It is known that motor commands from cortical masticatory areas induce rhythmic jaw movement by activating a central pattern generator in the brainstem through the pyramidal tracts. We examined in three rats whether rCBF changes similar to those induced by stimulating the cortical masticatory areas could be observed when jaw muscle activity was induced by stimulating the pyramidal tract. In accordance with previous studies, 38 electrical stimulation of the pyramidal tract (AP: −3.4 mm, L: 3.0 to 3.5 mm, V: 7.5 to 8.0 mm) induced masticatory muscle EMG activity equivalent to those during cortical stimulation. However, neither rCBF nor LFP scarcely changed during stimulation of the pyramidal tract (Figure 6). A slight increase in MAP by 10–20% was sometimes observed during stimulation, depending on stimulation site. In such cases, rCBF was increased by 10–20% in parallel with MAP changes. However, an increase in rCBF independent of MAP changes as observed during cortical stimulation was never observed during pyramidal tract stimulation.
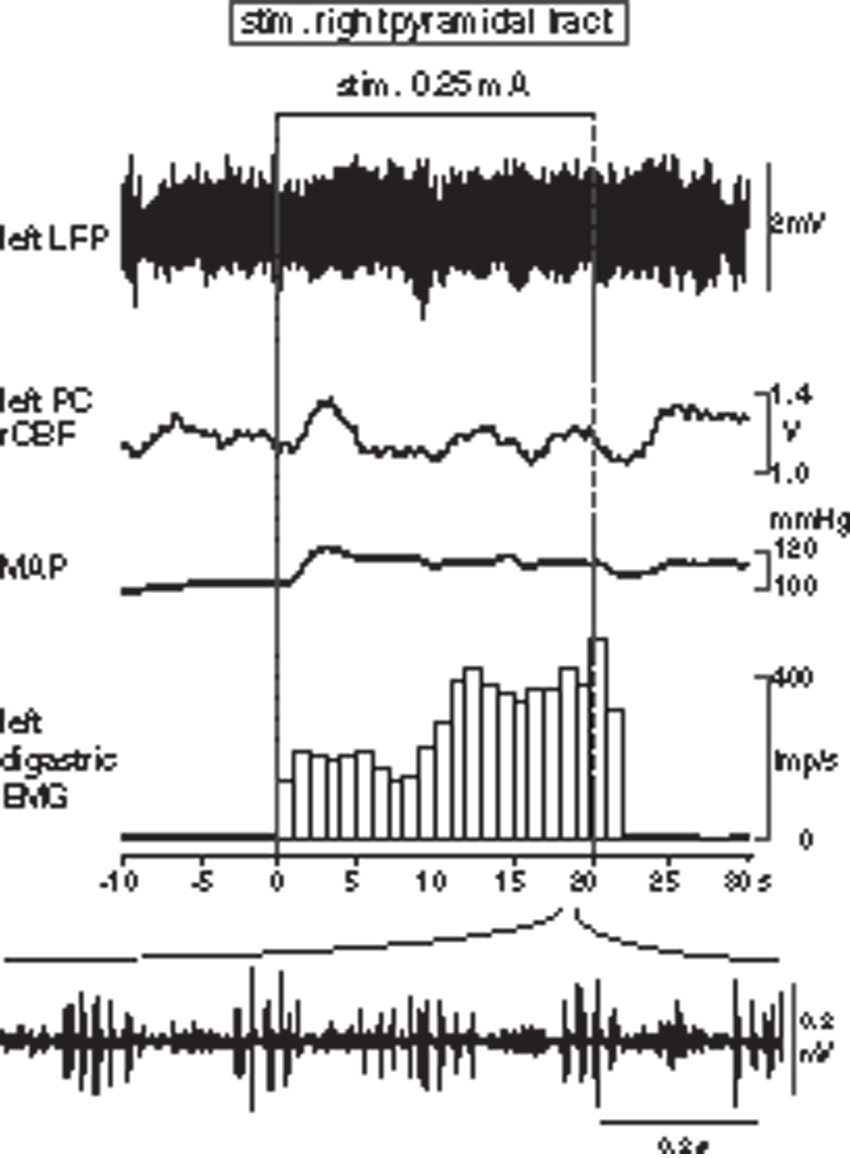
Responses in LFP, parietal rCBF, MAP and digastric EMG induced by electrical stimulation of pyramidal tract for 20 s. EMG activity is shown by the number of action potentials counted every second excluding stimulus artifacts. Bottom: Original recording of rhythmic EMG activity during pyramidal tract stimulation expanding the time scale.
Discussion
In the present study, we showed for the first time that cortical rCBF increased during stimulation of cortical masticatory areas, and that activation of NBM vasodilator neurons is an important component of this rCBF response (Figure 7). Furthermore, we found that, to produce this rCBF increase response, peripheral input from the jaw muscle is not essential, and the central command from the cortical masticatory area activates NBM by intracerebral mechanisms independent of motor command to the brainstem central pattern generator for masticatory movement.
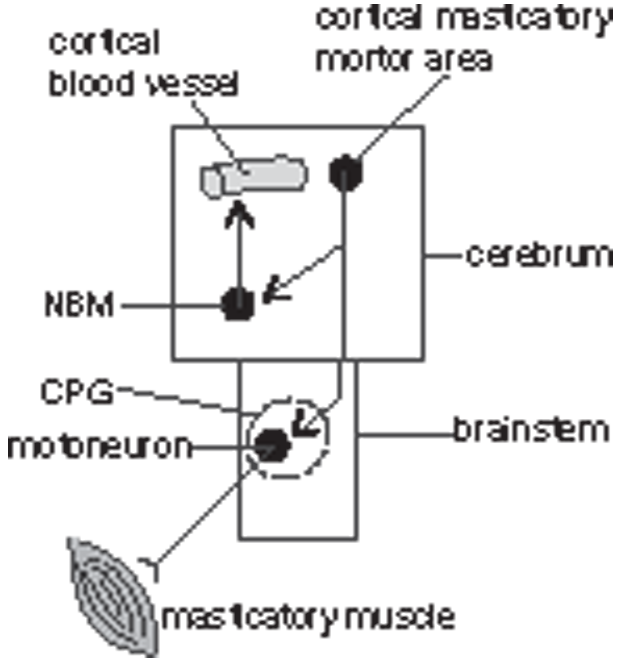
Schematic diagram showing that motor commands from cortical masticatory areas lead to an increase in cortical rCBF, possibly via a pathway within the forebrain, independent of activating the central pattern generator (CPG) in the brainstem.
Properties of rCBF increase associated with jaw muscle activity
In anesthetized rats, a robust increase in rCBF by about 40–60% was observed in association with jaw muscle activity induced spontaneously or by electrical stimulation of the cortical masticatory areas. These rCBF changes are similar to those reported during voluntary chewing or clenching in conscious humans and cats.6–8,10,11,44 MAP increased during P-area stimulation but unchanged during A-area stimulation, rCBF increased similarly regardless with or without changes in MAP. The latency and peak time of the rCBF responses induced by A- and P-area stimulation were similar. The amplitude of rCBF response induced by P-area stimulation (by 56%) was slightly larger than that induced by A-area stimulation (by 36%). This difference may be due to the secondary effects of MAP increase (by 11%) during P-area stimulation, but not during A-area stimulation. Therefore, we focused on the mechanisms of the rCBF response induced by A-area stimulation of which responses were due purely to active vasodilation. The rCBF increased during unilateral A-area stimulation in a wide cortical area including frontal, parietal, and occipital lobes bilaterally, similarly to those reported during walking. 25
Involvement of NBM on the rCBF changes associated with jaw muscle activity
The present results showed that (1) NBM activity increased prior to rCBF increase induced spontaneously or by A-area stimulation, and (2) increase in rCBF during A-area stimulation was attenuated by inhibiting the NBM activity using a GABAergic agonist (muscimol). These results indicate that the excitation of NBM is involved in the increase in rCBF induced by stimulation of A-area. The latency of rCBF increase induced by A-area stimulation was 1–2 s, which was similar to that induced by NBM stimulation.17,45 Topographical changes in rCBF in wide cortical areas particularly remarkable in the frontal and parietal lobes during A-area stimulation was also similar to that induced by NBM stimulation.45,46
It has been shown that NBM neurons are excited by noxious skin stimuli in anesthetized rats, 29 or excited with voluntary limb or face movements in conscious monkeys 47 and mice.48,49 A recent retrograde tracing study showed that the basal forebrain cholinergic system receives monosynaptic inputs from ipsilateral anterior cortices (e.g., orbital frontal cortex, motor cortex, and insular cortex) and from several subcortical nuclei (e.g., lateral septum, central amygdala, paraventricular nucleus of hypothalamus, dorsal raphe, and parabrachial nucleus). 50 However, the functional neuronal circuits to activate the NBM by somatosensory stimulation or associated with movements remain poorly understood.
The projection from the NBM to the cerebral cortex is ipsilateral, and unilateral NBM stimulation increases ipsilateral rCBF, but the rCBF response in this study was seen bilaterally with unilateral A-area stimulation. However, since the unilateral A-area connects to the contralateral A-area, 51 unilateral stimulation of the A-area may activate bilateral NBM. The details of the neural circuit connecting primary motor cortex and NBM need to be clarified in the future.
Involvement of feedback from the contracting muscles and the brainstem masticatory central pattern generator
It has been suggested that peripheral inputs from skeletal muscle cause a pressor response via somato-autonomic reflexes because muscle contraction during electrical stimulation of the jaw muscles induced a pressor response, and the response disappeared after immobilization using a muscle relaxant. 52 However, in our study, the rCBF increase by A-area stimulation was not affected by muscle relaxation. Our result indicates that there is a mechanism in which central commands from the cortical masticatory areas directly induce cortical rCBF response without peripheral sensory input from contracting muscles. In a human study measured jaw muscle activity simultaneously with middle cerebral artery blood flow, the magnitude and time course of blood flow response during voluntary chewing was constant irrespective of variations in muscle activity. 53 This finding is consistent with our conclusion on the independence of rCBF response from muscle activity.
The central pattern generator for masticatory jaw movements is located in the brainstem.54,55 Similar patterns of jaw movements can occur even after the cortical masticatory areas were ablated. 55 In our study, rCBF did not change when jaw movement were induced by stimulation of the pyramidal tract. These results indicate that activation of the central pattern generator for masticatory jaw movements is not responsible for increase in rCBF. Motor commands from cortical masticatory area appear to produce an increase in cortical rCBF via a pathway within the forebrain (Figure 7).
Physiological significance
The activation of the NBM induced by central motor command from the primary motor cortex may act when an organism voluntarily decides to eat. The NBM activation and robust increase in cortical rCBF may cause alertness and help plasticity, such as aversion or preference taste memory. We have previously shown that activation of NBM enhances secretion of nerve growth factor in the cerebral cortex,30,56,57 and that vasodilation during NBM stimulation helps to prevent delayed neuronal death following ischemia. 58 Therefore, the NBM activation associated with masticatory motor command revealed in this study would explain the association between chewing and higher brain functions.
Supplemental Material
JCB895244 Supplemental Material - Supplemental material for Involvement of the basal nucleus of Meynert on regional cerebral cortical vasodilation associated with masticatory muscle activity in rats
Supplemental material, JCB895244 Supplemental Material for Involvement of the basal nucleus of Meynert on regional cerebral cortical vasodilation associated with masticatory muscle activity in rats by Harumi Hotta, Harue Suzuki, Tomio Inoue and Mark Stewart: on behalf of the PROMISE Study Investigators in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI (grant number 24500635 and 17K01550 to HH).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
HH, TI and MS contributed to conception and design of research; HH and HS performed experiments, analyzed data and interpreted the results of experiments; HH and HS drafted the manuscript; all the authors edited, revised the manuscript and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
