Abstract
We earlier reported that electrical stimulation of the rat nucleus basalis of Meynert (NBM) induces large cerebral blood flow increases, particularly in frontal cortical areas but also in some subcortical regions. The present study was designed to address the issue of blood flow control exerted by NBM projections. To this aim, we have determined whether these flow increases were associated with proportionate changes in metabolic activity as evaluated by cerebral glucose utilization (CGU) strictly under the same experimental conditions in the conscious rat. An electrode was chronically implanted in a reactive site of the NBM as determined by laser-Doppler flowmetry (LDF) of the cortical circulation. One to two weeks later, while the cortical blood flow was monitored by LDF, we measured CGU using the [14C]2-deoxyglucose autoradiographic technique during unilateral electrical stimulation of the NBM, and analyzed the local flow-metabolism relationship. The large increases in cortical blood flow induced by NBM stimulation, exceeding 300% in various frontal areas, were associated with at most 24% increases in CGU (as compared with the control group) in one frontal area. By contrast, strong increases in CGU exceeding 150% were observed in subcortical regions ipsilateral to the stimulation, especially in extrapyramidal structures, associated with proportionate CBF changes. Thus, none of the blood flow changes observed in the cortex can be ascribed to an increased metabolic activity, whereas CBF and CGU were coupled in many subcortical areas. This result indicates that different mechanisms, which do not necessarily involve any metabolic factor, contribute to the regulation of the cerebral circulation at the cortical and subcortical level. Because the distribution of the uncoupling is coincident with that of cholinergic NBM projections directly reaching cortical microvessels, these data strongly support the hypothesis that NBM neurons are capable of exerting a neurogenic control of the cortical microcirculation.
Keywords
The basalocortical system originating in the nucleus basalis magnocellularis, or nucleus of Meynert (NBM), which constitutes the main source of the cortical cholinergic innervation in mammals (Woolf 1991, Alheid et al., 1995), is classically associated with cortical activation, attention, memory and learning processes (for review see Voytko et al., 1996). Conversely, NBM dysfunction has been related to the cognitive declines of Alzheimer's disease (Bierer et al., 1995) because this nucleus consistently undergoes degeneration in this pathology.
Recent studies have shown that NBM activation induces intense blood flow increases in the cerebral cortex of conscious rats, mostly through cholinergic mechanisms (Vaucher et al., 1995, and see Sato and Sato, 1995 for review). Concurrent morphological investigations have shown intimate associations between cholinergic axon terminals and cortical microvessels (Chédotal et al., 1994), predominantly originating in the NBM (Vaucher and Hamel, 1995). In agreement with the early hypothesis of a neurogenic control of brain circulation (Reis 1984), both type of experimental data suggest that basal forebrain nerve fibers are able to control the cortical perfusion. Previous data also showed that the cortical hemodynamic response to NBM stimulation could hardly be accounted for by a metabolic activation (Lacombe et al., 1989; Dauphin et al., 1991). Likewise, in the anesthetized rat, NBM stimulation did not change cerebral glucose utilization (CGU) (Kimura et al., 1990). These results suggest that local metabolism does not contribute to the flow changes induced by NBM activation. However, no functional approach has been performed yet to investigate in parallel the local circulatory and metabolic responses to NBM activation using the autoradiographic technique. Only such an approach can provide suitable data to test the hypothesis of a neurogenic control.
To this aim, we have evaluated the flow-metabolism relationship throughout the brain during NBM electrical stimulation in the conscious rat, by complementing our previous autoradiographic study on CBF (Vaucher et al., 1995). Cerebral glucose utilization was measured using the [14C]2-deoxyglucose technique under strictly the same experimental conditions as for the CBF study. Moreover, cortical blood flow was monitored by laser-Doppler flowmetry during the whole CGU measurement period, to ascertain that the blood flow changes were comparable in the CGU and CBF experiments.
MATERIAL AND METHODS
One difficulty in the interpretation of data on coupling is the unavoidable difference in the durations of the CBF and CGU measurements (30 seconds and 45 minutes respectively), which hampers strict quantification of the relationship between these two variables. To circumvent this limitation, we have (1) ascertained the steadiness of the cortical circulatory response during CGU measurement by laser-Doppler flowmetry (LDF) monitoring, and (2) synchronized the two measurement periods with respect to the NBM stimulation period. The animals were prepared in a two-step procedure, electrode implantation and CGU measurement, both involving LDF evaluation of the cortical perfusion.
The experimental protocols were approved by the Bureau de la Protection Animale of the Ministère de l'Agriculture, (authorization No. 03001 for P.L.).
Electrode implantation under laser-Doppler flowmetry monitoring
Thirteen male OFA/Sprague Dawley rats (Iffa-Credo, France) weighing 300 to 320 g were used for CGU measurement to complete the 14 rats previously used for CBF measurement (Vaucher et al., 1995). The rats were anesthetized with 0.6 to 0.8% halothane after induction at 4% and a-chloralose (40 mg/kg, subcutaneously) positioned in a Kopf stereotaxic frame (model 1400). Their skull surface was skimmed down unilaterally over the frontoparietal cortex until translucency (so that the pial vessels were visible). The LDF probe (tip diameter 0.8 mm, with three optical fibers, one light emitter, and two collectors, interaxis distance 0.5 mm) of the LDF apparatus (Moor Instruments, MBF3D, Axminster, United Kingdom; 1.5 mW maximal power, wavelength 810 nm) was carefully positioned using a micromanipulator to avoid major cerebral vessels and to obtain low and stable values.
A monopolar tungsten electrode (350 m; outer diameter) was stereotaxically lowered into the ipsilateral NBM at the following coordinates in millimeters (incisor bar at −5.0): interaural axis +7.0 and lateral 3.0. Electrical pulses (100 Hz, 0.5 ms, 50 μA) were delivered using two stimulation paradigms. First, exploratory stimulations (continuous trains of 10 seconds) were applied at different levels from 5.0 to 7.0 mm below the pial surface, to determine a responsive site within the NBM as evidenced by cortical blood flow increase monitored by LDF. Second, stimulation trains (1 second on/1 second off) were repeated at the most responsive site to determine whether the blood flow increases were sustained for a period covering the CGU measurement. The stimulation site in the NBM was considered appropriate when both stimulations induced flow increases that exceeded 100%. The electrode was left in place (usually at 5.5 mm from the pial surface) and chronically sealed, together with the polyethylene tubing guide of the LDF probe. The latter was removed and could be precisely repositioned during the second stage of the experiment.
Procedure for combined parallel cerebral glucose utilization and cerebral blood flow investigations
After 11 to 14 days of recovery from implantation, the rats weighed 354 ± 22 g (mean ± SD). They were catheterized in both femoral arteries and in one vein under 1% halothane, then comfortably installed in a hammock and allowed to recover from anesthesia for 2.5 hours until CGU measurement. The rectal temperature was maintained at 37.5°C using a thermostatically controlled heating pad. All variables, arterial blood pressure, heart rate, and LDF signal and stimulations were recorded both on paper (Gould 3400) and computer. The arterial blood gases and pH were periodically measured. In four animals, the cortical blood flow was monitored through the LDF probe repositioned in the implanted guide. The stimulation (1 second on/1 second off paradigm) started 5 minutes before the CGU measurement and its intensity was increased progressively up to 50 μA during 2 minutes. The precise location of the stimulated site was reconstructed from cresyl violet-stained serial sections using a vertical microprojector. The distribution of sites in the CGU group was virtually identical to that in the CBF group.
Cerebral glucose utilization measurement started with intravenous infusion of 0.8 mL of a solution containing 125 μCi/kg of body weight of [14C]2-deoxyglucose (55 mCi/mmol, ARC-Isotopchim, France) for a period of 40 seconds. During the subsequent 45 minutes, 20 arterial blood samples were collected in microtubes and centrifuged for determination of the time-concentration curve of the plasma tracer (in 20 μL aliquots) and glycemia (10 samples of 10 μL). Plasma glucose levels were determined using the glucose oxidase assay and a spectrophotometer (Beckman DU-640). After 45 minutes, the rat was killed and its brain was rapidly removed and frozen for later autoradiography processing. Local CGU was calculated using the integrated equation of Sokoloff and colleagues (1977).
The protocols for CBF and CGU measurements were matched. Cerebral blood flow measurement was initiated after 20 minutes of NBM stimulation, that is, 15 minutes after the deoxyglucose infusion, so that it coincided with the median point of integrated deoxyglucose specific activity in brain tissue. A saline solution (0.9 mL containing 100 μCi/kg of body weight) of [14C]iodoantipyrine (54 mCi/mmol, ARC-Isotopchim) were infused using a programmable syringe pump (Harvard 44) so that the arterial tracer input function was a 30 second ramp. This infusion protocol precluded the finding of apparently excessive flow values (above 700 ml, 100g−1 min−1) which might be found in some stimulated, conscious rats.
For autoradiogram preparation, coronal 20-μm sections of the brain were cut sequentially at −20°C as follows: 1 for histological examination, 4 for autoradiography, and the next 7 were discarded. The sections for autoradiography were exposed for 5 days to Kodak SB-5 X-ray film with 14C standards (ARC, 146C). The autoradiograms were analyzed using a computerized image analysis system (Biocom 2000, France, with RAG software). The regions of interest were closely outlined on the basis of the corresponding cresyl violet-stained sections and the mean optical density of each region was determined bilaterally on three consecutive sections.
Data and statistical analysis
Glucose utilization was measured either in sham conditions (n = 6, electrode implanted but no current) or during unilateral stimulation (n = 7, stimulated group). A one-way analysis of variance was used to compare the four series of CGU values obtained for each region of both hemibrains of the two groups. The analysis of variance was followed by post hoc Tukey's multiple range tests. Comparisons between homotopic regions of the hemibrains ipsilateral and contralateral to the stimulation were made with paired t-tests.
Some of the CBF values used in this study are derived from a previous article (Vaucher et al., 1995). However, because the spatial resolution of CGU autoradiograms is much better than that of CBF, we delimited more precise regions of interest, based on the cresyl violet-stained sections (particularly in the cortical layers, the substantia nigra, the subthalamic region), in order to recalculate the corresponding CBF values.
A two-step procedure was used for analysis of the CBF-CGU relationship. First, the classical linear regression was calculated to evaluate the correlation coefficient between mean regional CBF and mean regional CGU for each hemibrain of the group of stimulated rats, so that to characterize the global hemispheric effect of the stimulation. However, because this procedure was justifiably criticized by McCulloch et al. (1982), we have followed the more appropriate procedure defined by these authors, which is applied on the ratios of the blood flow to glucose utilization for each region using the model: Ln (CBF) = Ln (CGU) + constant. Accordingly, the second step was to compare globally the stimulated and unstimulated hemibrains using a modified t-test, and then test for the homogeneity of the regional ratios (CBF/CGU) using a repeated measure analysis of variance (for which the region is the repetition factor), followed by the Greenhouse-Geisser test using a modified F distribution. Hence, the confidence limits of the mean ratios were evaluated to identify the regions in which the coupling was selectively disturbed at P < 0.01 (McCulloch et al., 1982).
RESULTS
Effects of nucleus basalis of Meynert stimulation on continuously investigated physiological variables
Nucleus basalis of Meynert stimulation in the conscious rat induced a slight hypertension (less than 5% during the first 10 to 20 minutes of stimulation), but did not significantly alter arterial blood gases and pH (data not shown). As the intensity of stimulation was progressively increased, the rats showed increasing stereotyped movements: vibrissal vibrations, chewing, sometimes head rotations, which varied from one animal to another. When stimulation intensity was stabilized (after 2 minutes), these behavioral phenomena faded (less so for the vibrissal vibrations and vacuous chewing) in the CGU group as in the CBF group.
Continuous blood flow monitoring by LDF in parallel with CGU measurement showed that cortical blood flow increased by up to 200% of baseline when NBM stimulation was applied and remained elevated during 50 minutes (Fig. 1).
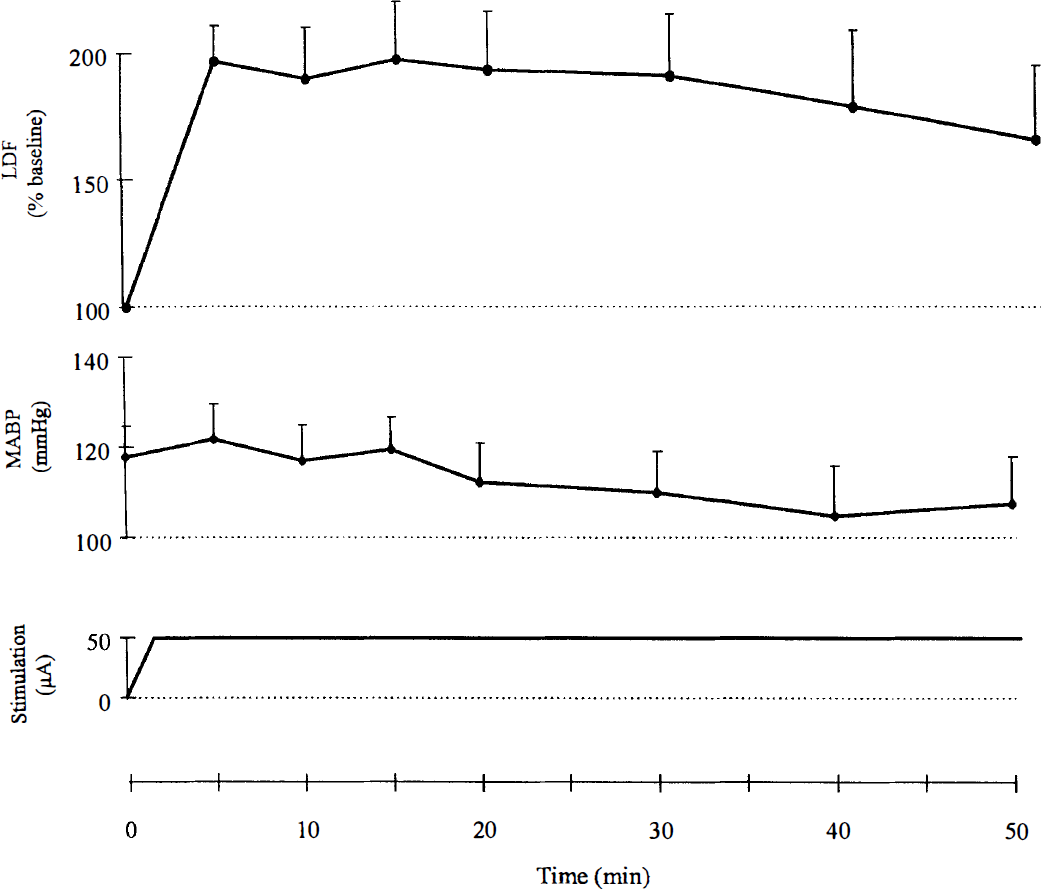
Graph of cortical blood flow as measured by laser-Doppler flowmetry
Effects of nucleus basalis of Meynert stimulation on the flow-metabolism relationship in cortical areas
Fig. 2 clearly shows that the effects of NBM stimulation on glucose utilization in the cortex were modest compared with the effects on blood flow. Significant interhemispheric differences in metabolic activity were few and of limited magnitude (up to 15%): increases in frontal and entorhinal areas and decreases in parietal areas, ipsilateral to the stimulation (Fig. 3A; Table 1). These changes were restricted to layer V. Comparison with the CGU values of the ipsilateral hemisphere of the control group showed a greater difference (24%) in the frontal area 10 (Table 1). This disparity is to be related to the interhemispheric differences observed in the control group (CGU decreases of up to 10% in the frontal area of the implanted hemisphere).
Effects of unilateral stimulation of the nucleus basalis of Meynert on local cerebral glucose utilization
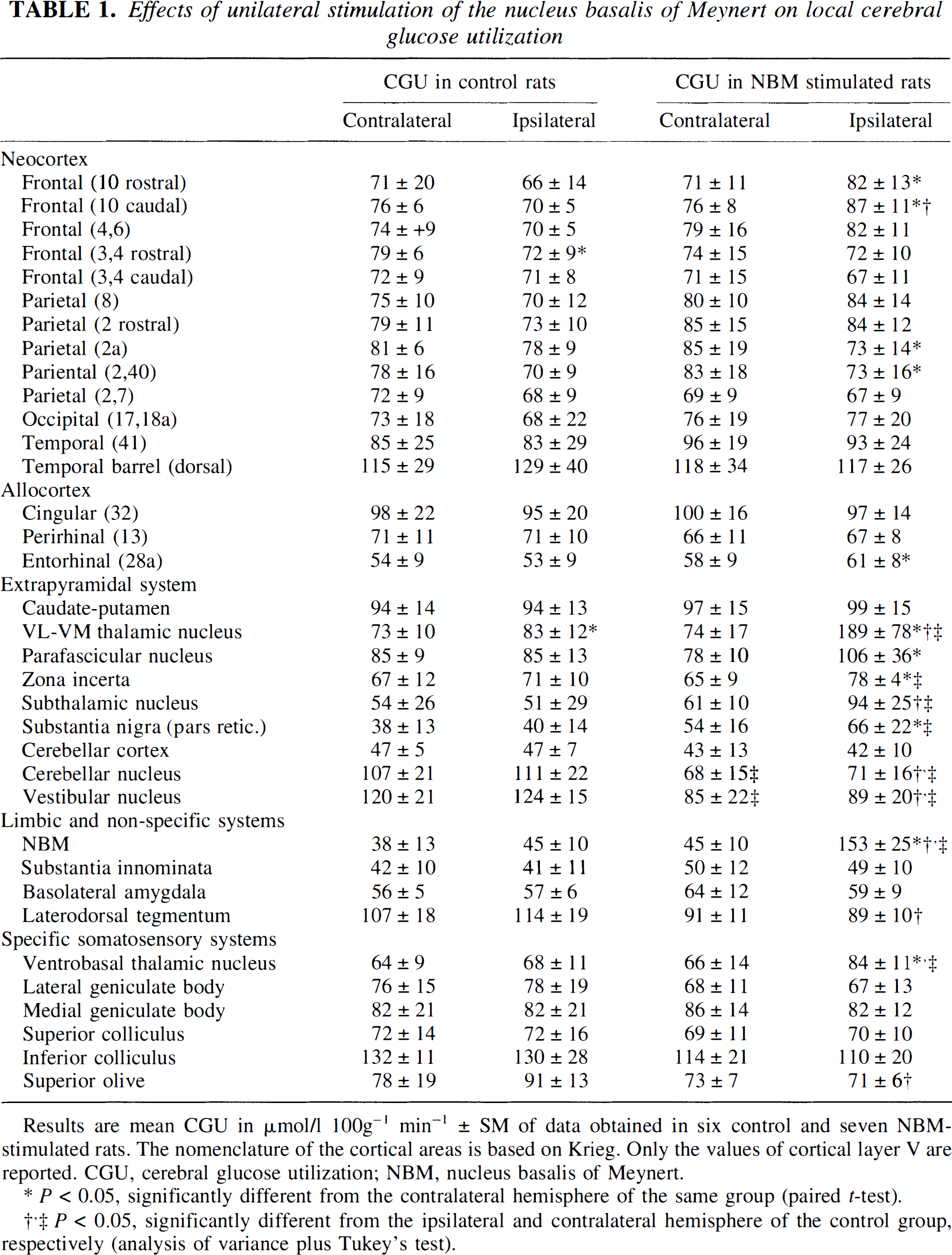
Results are mean CGU in μmol/1 100g−1 min−1 ± SM of data obtained in six control and seven NBM-stimulated rats. The nomenclature of the cortical areas is based on Krieg. Only the values of cortical layer V are reported. CGU, cerebral glucose utilization; NBM, nucleus basalis of Meynert.
P < 0.05, significantly different from the contralateral hemisphere of the same group (paired t-test).
P < 0.05, significantly different from the ipsilateral and contralateral hemisphere of the control group, respectively (analysis of variance plus Tukey's test).
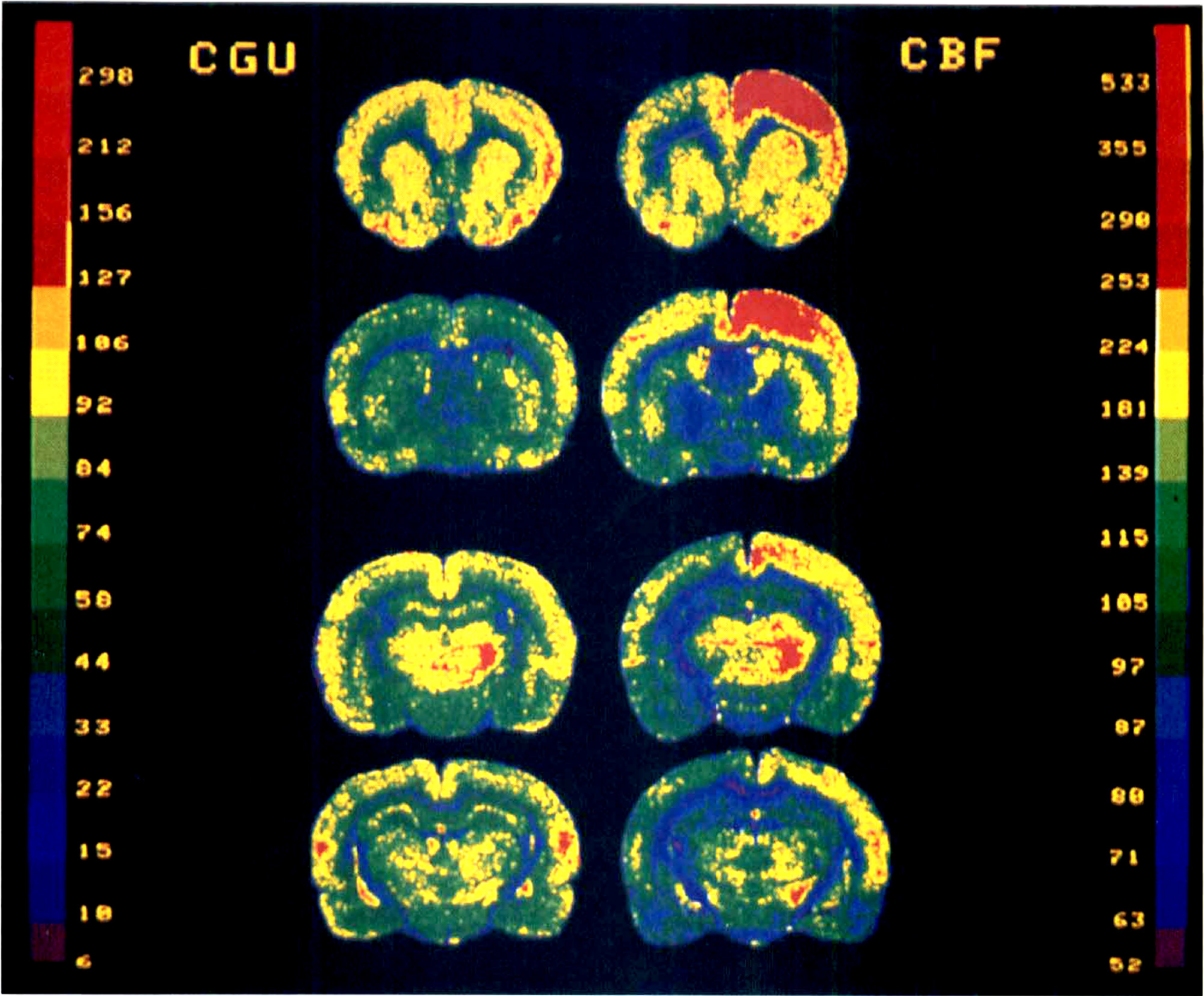
Representative color-coded autoradiograms from rats infused with [14C]2-deoxyglucose for CGU measurement (left panels and color scale expressed in μmol 100 g−1 min−1), and [14C]iodoantipyrine for CBF measurement (right panels and color scale expressed in mL 100 g−1 min−1) during unilateral stimulation of the NBM (right side of the section). The four coronal levels include from top to bottom: frontal (4, 6) and parietal (2) cortical areas, caudate-putamen and accumbens nucleus; frontal (3, 4) and parietal (2, 2a) cortical areas, globus pallidus; parietal (7, 2, 40, from dorsal to ventral) cortical areas, VL-VM and ventrobasal thalamus nuclei, parietal (17, 18a) cortical areas, parafascicular nucleus, and zona incerta. The frontal and frontoparietal cortical areas denote an obvious uncoupling between CGU and CBF. The CGU changes are limited to cortical layer V.
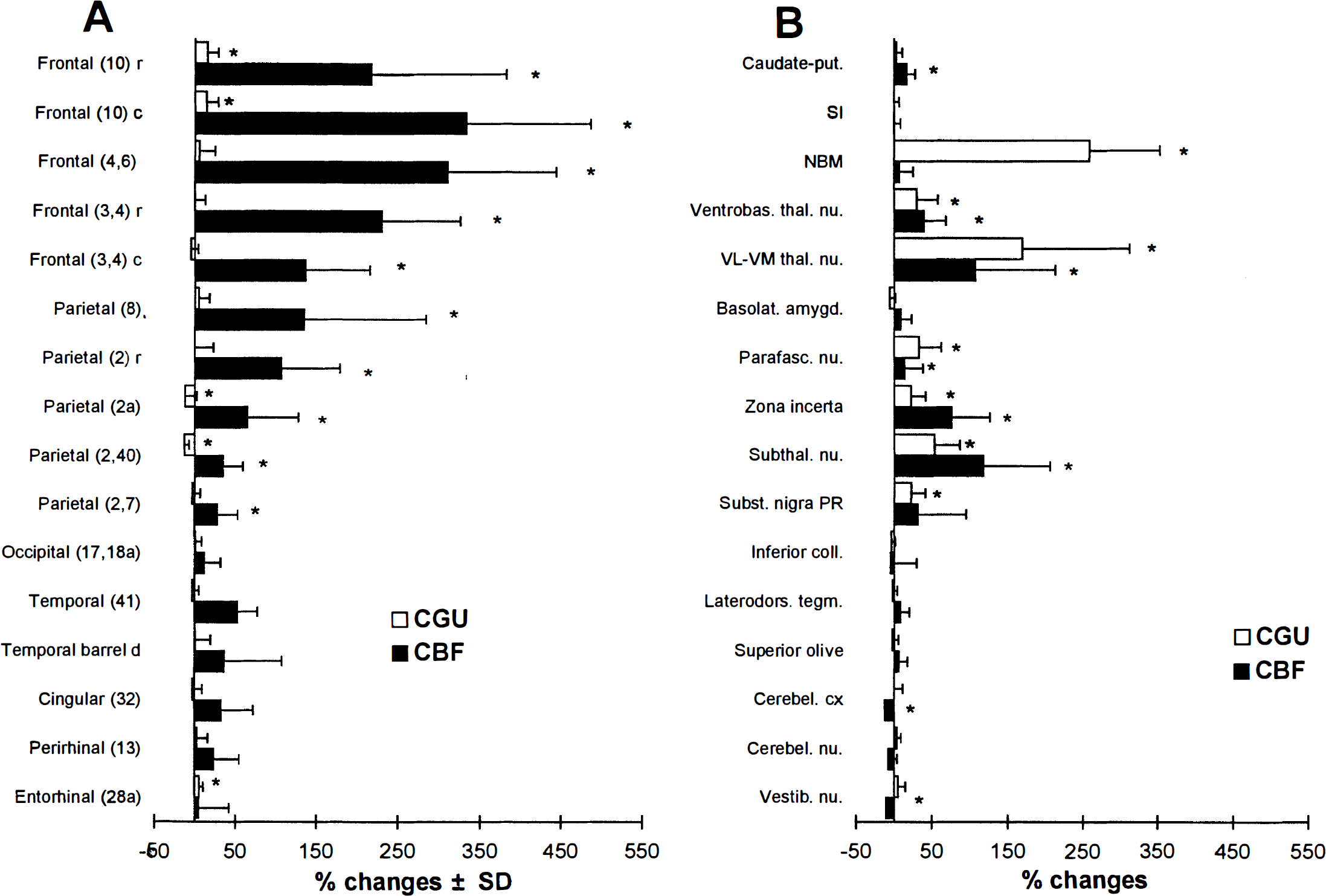
Percent changes (ipsilateral versus contralateral) in CBF and CGU (mean ± SD) during NBM stimulation in some cortical
Thus, the strongest blood flow increases observed in the frontal and parietal cortex were associated with non-significant changes in glucose utilization. The regression analysis (Fig. 4) shows that CBF and CGU values of stimulated rats are correlated in the regions contralateral, but not ipsilateral to the stimulation. Analysis of the flow-metabolism ratios indicates a significant uncoupling in all five areas of the frontal cortex examined and the two rostral areas of the parietal cortex. These ratios ranged from 3.5 to 8.2 in the stimulated hemisphere and were considerably greater than in the contralateral hemisphere, by 2.2 to 3.9 fold, the lower values being found caudally and ventrally.
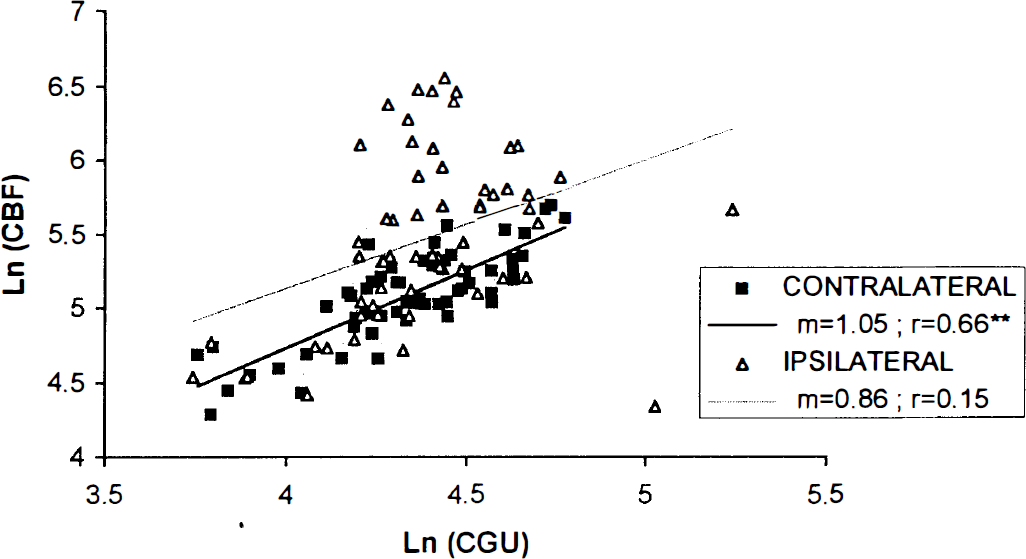
Relationship between mean local CBF and mean local CGU as expressed in neperian logarithm in contralateral (filled diamond) and ipsilateral (empty triangle) hemibrains of NBM-stimulated rats. The slope (m) and coefficient of correlation (r) derived from the two linear regression analyses are indicated. Only the correlation in the hemibrain contralateral to the stimulation is significant (**P < 0.01).
Effects of nucleus basalis of Meynert stimulation on the flow-metabolism relationship in subcortical regions
In contrast to the cortex, glucose utilization was much more affected in subcortical regions ipsilateral to the stimulation (Fig. 2; Table 1). Comparison with the contralateral hemisphere revealed significant increases (Fig. 3B), which ranged from 23 to 170% (excluding the NBM), especially in structures of the extrapyramidal system. The changes in glucose utilization in the thalamic and subthalamic areas were proportionate to the blood flow changes (Fig. 3B). The flow-metabolism ratios of these areas were around 2 and decreased under NBM stimulation (especially in the VL-VM thalamic nuclei), except for the subthalamic nucleus and the zona incerta.
Comparison with the control group (Table 1) showed clear-cut bilateral decreases in CGU in the vestibular and the cerebellar nucleus (26 to 36%). These decreases were associated with increases in CBF (+ 63 to 71%). A similar tendency was observed in the laterodorsal tegmentum and in some brainstem (auditory) structures. Though not detected by our analysis, uncoupling can be postulated in these regions because they are the site of significant opposite changes in glucose utilization and blood flow. These regions show the highest ratios in resting conditions (range 2.6 to 3.3, unstimulated hemibrain).
The stimulation site displayed a huge CGU change (+ 259%, Fig 3B) seen by a black spot of 300 to 500 mm diameter on the autoradiogram delimiting the area directly activated by the electrode tip. This spot, strictly localized in the NBM, indicates that the stimulation was spatially selective and did not spread to adjacent area (unchanged CGU value in the underlying substantia innominata, Fig. 3B). On CBF autoradiograms, the stimulated site corresponded to unchanged blood flow.
DISCUSSION
The most remarkable point revealed by the comparison between the quantified distributions of the metabolic and circulatory effects of NBM stimulation is that stimulation of sites from which sizable cortical blood flow increases were elicited induced only small and scarce increases in CGU in the various cortical areas investigated. In contrast, strong metabolic increases associated with proportionate circulatory responses occurred simultaneously in some subcortical regions. We shall discuss successively (1) the relationship between CBF and CGU, its considerable heterogeneity and its functional significance under NBM activation, and (2) the possible mechanisms that may account for the uncoupled flow-metabolism responses.
Relationship between cerebral blood flow and cerebral glucose utilization
Stimulation of the basal forebrain brought about a marked dissociation in the regional circulatory and metabolic responses in the cortical mantel. In the present study, quantification of the relationship between CBF and CGU measurements has been optimized by LDF monitoring during NBM stimulation, which confirmed that the time constants of the CGU measurement and the circulatory cortical events were compatible. Flow-metabolism dissociation is consistent with the changes observed in brain tissue gases during NBM stimulation in the conscious rat. These showed increase in PO2 and decrease in PCO2 that indicate an absence of metabolic activation in the frontal cortex (Lacombe et al., 1989). Likewise, nonsignificant CGU increases were obtained in the halothane-anesthetized rats (Kimura et al., 1990). Therefore, NBM stimulation cannot be held responsible for widespread 'metabolic cortical arousal,' or seizures (see Ingvar and Siesjö, 1983 for comparison), as we previously suggested on the basis of the absence of EEG desynchronization during stimulation (Lacombe et al., 1989). Such a result is in agreement with the limited behavioral changes evoked by the stimulation that consist mainly of stereotyped movements probably of extra-pyramidal origin. Thus, this study confirms that the circulatory effects of NBM activation in the cortex do not necessarily result from metabolic changes and could be independent.
One unexpected feature of our data is the considerable heterogeneity of the flow-metabolism relationship under NBM stimulation. In addition to the significant uncoupling found in the frontal cortex, a second situation of uncoupling can be postulated, such as that in the cerebellar and vestibular nuclei. It comprises significant blood flow increases associated with significant decreases in glucose utilization. These changes were not detected as uncoupled because our analysis was applied to the CBF/CGU ratios, and therefore did not take into consideration the sign of the combined changes in CBF and CGU. They indicate a situation of inverse coupling which has been rarely described. Somewhat proportionate changes in glucose utilization and blood flow were found in several thalamic and subthalamic nuclei. (It is notable that in the VL-VM thalamic nuclei, metabolic increases were proportionately greater than circulatory increases, which indicates that CGU measurement is not less sensitive than CBF one.) This is the situation usually encountered during physiological stimulation in the conscious (Ginsberg et al., 1987) or anesthetized rat (Ueki et al., 1988).
Thus, NBM activation brings about a variety of changes in the flow-metabolism relationship, a prominent feature of which is regional-dependency. Observation of a true dissociation between blood flow and metabolic activity in the conscious rat under physiological conditions demonstrates that uncoupling is not restricted to either extreme or pathophysiological conditions. Further, the present study shows that this situation can be sustained during several tens of minutes, and hence is not limited to short-term or transient cerebrovascular events.
Possible mechanisms of flow-metabolism relationship
The complete dissociation between changes in blood flow and glucose utilization induced by NBM stimulation clearly shows that first, the mechanisms that control cerebral circulation and metabolism are different and not necessarily linked. Second, the heterogeneity of the regional distribution and types of flow-metabolism relationship suggests the existence of mechanisms which vary from one region to another. These results do not seem compatible with the widely prevailing contention of mechanisms in series between neuronal activation, metabolism and blood flow. They suggest the involvement of parallel mechanisms.
Measurements at the stimulation site show that electrical activation by itself results in no effect on local blood flow whereas it strongly increases local CGU. Comparison with the effects in distant regions indicates that structural tissue elements are needed for eliciting a circulatory response. The cortical vasodilations are mediated by predominantly cholinergic mechanisms (Dauphin et al., 1991; Linville et al., 1993; Sato and Sato, 1995), and cholinergic mediation has frequently been associated with uncoupling (Scremin et al., 1993). The muscarinic agonist arecoline induced uncoupling in the frontal and parietal but not the sensory cortex of the conscious rat (Maiese et al., 1994). In complete agreement with our results, Ogawa et al. (1994) have shown that scopolamine abolishes the circulatory but not the metabolic response to somatosensory stimulation. These data strongly support the notion that the cortical vasodilations unaccompanied by metabolic changes can be ascribed to a direct cholinergic, largely nitric oxide-dependent mechanism (Zhang et al, 1995). The NBM cholinergic projections reaching cortical microvessels constitute a structural basis for this function (Vaucher and Hamel, 1995). The frontal predominance of the cortical uncoupled responses is compatible with the distribution of the decrease in choline acetyltransferase activity after NBM lesion (Peruzzi et al., 1993). Conversely, the other type of flow-metabolism relationship might be related to the paucity and/or indirectness of cholinergic NBM projections (including their proportion and mode of transmission, synaptic or volumic). The differing local regulation of blood flow could result from mechanisms depending on the equilibrium between cholinergic and noncholinergic influences.
Our results also relate to the debated issue of the involvement of intracortical neurons in the response to NBM stimulation. The absence of increased glucose utilization both in cortical areas where blood flow was considerably increased and in the corresponding subcortical efferent projection areas, makes it unlikely that intracortical neurons are activated targets or the origin of an efferent inhibition or activation. Hence, intracortical neurons could be facultative mediators of the circulatory response. This is quite consistent with (1), the result of Linville and colleagues (1993) who showed that the circulatory NBM response was not altered by lesion of all cortical neurons, and (2) the results of Zhang et al. (1995) who showed that the nitric oxide synthase-dependent component of the response to NBM stimulation is of endothelial rather than neuronal origin. Altogether, these data support a predominant involvement of the direct basalocortical neurovascular connections. However, they seem incompatible with both the biochemical results of Galea et al. (1991) who showed that choline acetyltransferase activity of isolated microvessels was not affected by NBM lesion, and anatomical observations of some intracortical neurons that contact the microvascular environment (Chédotal et al., 1994) and receive terminals from NBM neurons, in particular nitrergic and GABAergic (Vaucher et al., unpublished observations). There is as yet no definite explanation for these discrepant data, but it is possible that intracortical neurons are inhibited during electrical stimulation (see below), whereas they regulate the circulatory response to physiological activation.
This last point raises the question as to how activation of NBM projections can have effects so powerful on the cortical circulation and simultaneously so weak on the metabolic activity. The answer might lie in the particular targets of these projections. The ultrastructural features of the neurovascular relationship support the functional capacity of such an innervation (Chédotal et al., 1994). These authors pointed out that a large majority of the cholinergic terminals found close to capillary abuts on perivascular astrocytic endfeet. Astrocytes are thought to occupy a key position between neuronal activity and microcirculation (see Kuschinsky 1996). They possess muscarinic receptors (Moro et al., 1995) and can be activated by several neurotransmitters, so that they could mediate the relationship between neuronal activity and glucose metabolism (Magistretti and Pellerin, 1996). During NBM activation, astrocytes may drive the micro-circulation more efficiently than activate the neuropil. Vascular mechanisms might complement these neurogenic mechanisms. In muscle, a myogenic cholinergic mechanism originating in capillaries and propagating upstream to the feeding arteriole contribute to vasodilation (Song and Tyml, 1993). In the brain, such a mechanism might account for some diffusion of the circulatory response, that is, to adjacent cortical layers in a single barrel (Cox et al., 1993). A complementary explanation is that acetylcholine is not merely excitatory on the neuropil under these experimental conditions. This hypothesis is supported by the study of Metherate and Ashe (1995) who showed that acetylcholine does not increase glutamate release in cortical brain slices, and by Magistretti and Pellerin (1996) who did not observe any activatory effect of acetylcholine on cultured astrocytes. Moreover, NBM projections are functionally heterogeneous. Inhibitory GABAergic intracortical neurons or basalocortical fibers may interact with cortical cholinergic elements (Vaucher and Hamel. 1995) and abolish the metabolic effects of basal forebrain stimulation.
In conclusion, we provide evidence that a central neuronal system belonging to the basal forebrain is able to induce remote, selective (without systemic changes), primary (without CGU changes) blood flow increases. That vasodilation can occur without significant changes in glucose utilization indicates that these two variables might evolve independently, thereby questioning the principle of coupling during activation. We ascribe this property to the particular cholinergic innervation of microvessel of some cortical areas. This system presents the potential to exert a direct, fast control of the intraparenchymal microcirculation through a locally differentiated action. In physiological conditions, it could provide adequate and early blood supply simultaneously to an activation through parallel mechanisms, which could anticipate an increase in metabolism. This mechanism thus represents an intrinsic neurogenic control of local CBF. Furthermore, our results extend the significance of uncoupling by showing that it can be sustained during tens of minutes, so that it may correspond to a long-term component of a dynamic flow-metabolism relationship (Kuschinsky 1996). It might also account for some luxury perfusion (see Villringer and Dirnagl, 1995) and the enhanced attention-modulated CBF response to somatosensory stimulation in man (Meyer et al., 1991). These mechanisms that allow blood flow to change independently of glucose-derived energy production argue against glucose oxidation as a unique regulator of CBF, as suggested for physiological activations (Fox et al., 1988; Ueki et al., 1988; for review see Villringer and Dirnagl, 1995).
Because aging in the rat has been shown to reduce the cerebrovascular response to NBM activation, to halve the increased oxygen availability to brain tissue (Sercombe et al., 1994) and to locally decrease cerebral metabolic activity (Bassant et al., 1994), the issue of the consequences of NBM dysfunction, such as occurs in dementia of the Alzheimer type becomes relevant. Indeed, micro-vascular abnormalities (see for review Kalaria, 1992; Buée et al., 1994) and denervation microangiopathy (Wallace et al, 1993; de la Torre 1994), both demonstrated in Alzheimer's disease, argue in favor of a causative involvement of microcirculatory dysfunctioning in the cascade of events leading to the neurodegenerative disorders.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Dominique Von Euw for valuable help with the illustration.
