Abstract
Clinical studies report that low circulating angiopoietin-1 concentration at presentation predicts worse outcomes after ischaemic stroke. Upregulating angiopoietin-1 may therefore have therapeutic benefit for ischaemic stroke. This systematic review assessed whether upregulating angiopoietin-1 improved outcomes in rodent models of ischaemic stroke. Random-effects models quantified the effect of angiopoietin-1 upregulation on stroke severity in terms of the size of cerebral infarction and the extent of blood–brain barrier permeability. Eleven studies utilising rat and mouse models of ischaemic stroke fulfilled the inclusion criteria. Meta-analyses demonstrated that angiopoietin-1 upregulation significantly reduced cerebral infarction size (standardised mean difference: –3.02; 95% confidence intervals: –4.41, –1.63;
Keywords
Introduction
Stroke is the world's third leading cause of death, and primary cause of adult disability. 1 Approximately 80% of all strokes result from acute cerebral ischaemia. 2 An important treatment for ischaemic stroke is rapid restoration of the cerebral blood supply by mechanical or chemical thrombolysis. 2 Disadvantages of this therapy include a risk of substantial complications, need for specialist expertise and resources not available in many regions of the world, limited windows of effectiveness and lack of suitability for many patients.3,4 Identifying novel therapies for ischaemic stroke is a key research priority. 5
The angiopoietins (angpts) are growth factors which play an important role in maintaining vascular function.6–9 Angpts bind to the Tie-2 receptor which is predominantly expressed on the vascular endothelium.6–9 An observational study previously reported that patients who suffered a recent ischaemic stroke had significantly lower serum angpt-1 concentrations than healthy controls. 10 Patients with serum angpt-1 concentration in the lowest tertile were more likely to have died or have severe disability three months after the stroke than those in the highest tertile. 10 Experimental studies suggest that angpt-1 may improve the outcome of ischaemic stroke in several ways (please see Eklund et al. 7 ; Moss 8 ; and van Meurs et al. 11 for detailed reviews). First, Tie-2 activation has been reported to activate PI3/Akt and inhibit nuclear factor kappa-light-chain-enhancer of activated B cells to induce pro-survivor and anti-inflammatory pathways in endothelial cells.7,8,11 Angpt-1 activation of the Tie-2 receptor is also important in regulating vascular integrity by maintaining the contacts between neighbouring endothelial cells and the extracellular matrix.7,8,11 Angpt-1 has also been demonstrated to induce the migration of endothelial cells.7,8,11 Overall, these studies suggest that angpt-1 inhibits inflammation, promotes angiogenesis and improves endothelial integrity, which may act to limit the severity of cerebral infarction following stroke. Previous studies have examined the effect of upregulating or administering angpt-1 on the outcome of ischaemic stroke in rodent models, but findings have been inconsistent.12–22 The current study systematically reviewed the efficacy of upregulating or administering angpt-1 in improving recovery from experimentally induced ischaemic stroke in rodent models.
Methods
This systematic review was performed in line with the guidelines of PRISMA and the Systematic Review Centre for Laboratory animal Experimentation statement. A protocol was developed following these guidelines 23 and was registered in the CAMARADES database (date of registration: 21 August 2017).
Search criteria
A systematic literature search was conducted to identify studies investigating the effects of upregulating angpt-1 in rodent ischaemic stroke models (Supplement 1). Eligible studies reported original experiments involving angpt-1 administration or upregulation in a rodent model of acute ischaemic stroke (regardless of dose, method, timing or frequency of administration). Studies also had to provide data on at least one of the following outcome measures: (i) cerebral infarction size; (ii) blood–brain barrier (BBB) permeability; (iii) neurobehavioural outcomes as assessed by a neurological deficit score. Review articles, editorials and publications in languages other than English were excluded. Studies which investigated the effect of angpt-1 in combination with other interventions were excluded unless data on the effects of angpt-1 upregulation alone could be extracted. In cases of ambiguity, authors were contacted to provide additional information. The final literature search was completed in January 2019.
Data extraction and quality assessment
Data extraction and quality assessment was independently conducted by three authors (JVM, AFT and DRN). Information extracted from each study included the species, strain, age and/or body weight of rodents used, duration of cerebral ischaemia, the mechanisms by which angpt-1 was up-regulated, the interventions administered to control animals and the timing of administration relative to stroke induction. Information extracted for each outcome measure included the methods used to assess the outcome, timing of the assessment relative to stroke induction and where possible detailed numerical data to quantify the results, in addition to reported
The quality and potential bias of the included studies was assessed using a modified CAMARADES score (Supplement 2 and Macleod et al. 24 ). Individual scores were calculated based on the number of ‘yes’ answers. Studies were allocated a maximum score of 11, a score ≤ 3 was considered to denote a poor-quality study, those with a score of 4–7 were considered to be of moderate quality and high-quality studies achieved scores ≥ 8. No articles were excluded based on their quality assessment score. Discrepancies in the extracted data were resolved at a consensus meeting.
Data analysis
The primary outcome measure for the meta-analysis was the difference in cerebral infarction size between rodents in which angpt-1 was up-regulated compared to controls. This was chosen since it is a widely employed outcome measure used in rodent models and human studies. Secondary outcome measures were the differences in BBB permeability and neurological function (as assessed using a defined scoring system) between experimental groups. Data were extracted from publications, and, where necessary, were converted to provide common units for comparison between studies. If studies reported findings as mean and standard error, standard deviations were calculated by multiplying the standard error by the square root of the sample size. Where necessary, data were extrapolated from graphs using the analysis function of Photoshop CC 2018 (Adobe Inc, San Jose, CA).
Meta-analysis was conducted when comparable outcome data were available from at least three studies. As high inter-study variation was anticipated, random-effects models were constructed (RevMan V5, Cochrane Collaboration). Effect sizes were calculated as standardised mean difference (SMD) and 95% confidence intervals (CI). Sub-analyses assessing the impact of angpt-1 upregulation on the outcome of stroke were performed in (a) permanent versus temporary cerebral ischaemia models and (b) studies which up-regulated angpt-1 before or after stroke induction. Inter-study variation was assessed using the I2 statistic. Leave-one-out sensitivity analyses excluding data from individual studies were conducted to assess the robustness of findings. Significance for all analyses was accepted if
Results
Characteristics of included studies
The initial database search identified 642 potentially eligible studies. After duplicate removal and title and abstract screening, the full-text of 18 studies was reviewed. After excluding seven studies, 11 were included in the current systematic review (Figure 1; Table 1).12–22 Eight studies used rats and three employed mice. Four studies induced stroke by occluding the middle cerebral artery for two hours,12,14,17,21 seven induced permanent cerebral ischaemia by inserting a filament into the middle cerebral artery,15,16,18,19 or intravenous thrombus injection.13,25 Venkat et al. induced embolic cerebral ischaemia but did not specify their methodologies.
20
PRISMA diagram outlining the literature search. Characteristics of included studies. COMP: cartilage oligomeric matrix protein. tPA: tissue plasminogen activator; angpt-1: angiopoietin 1.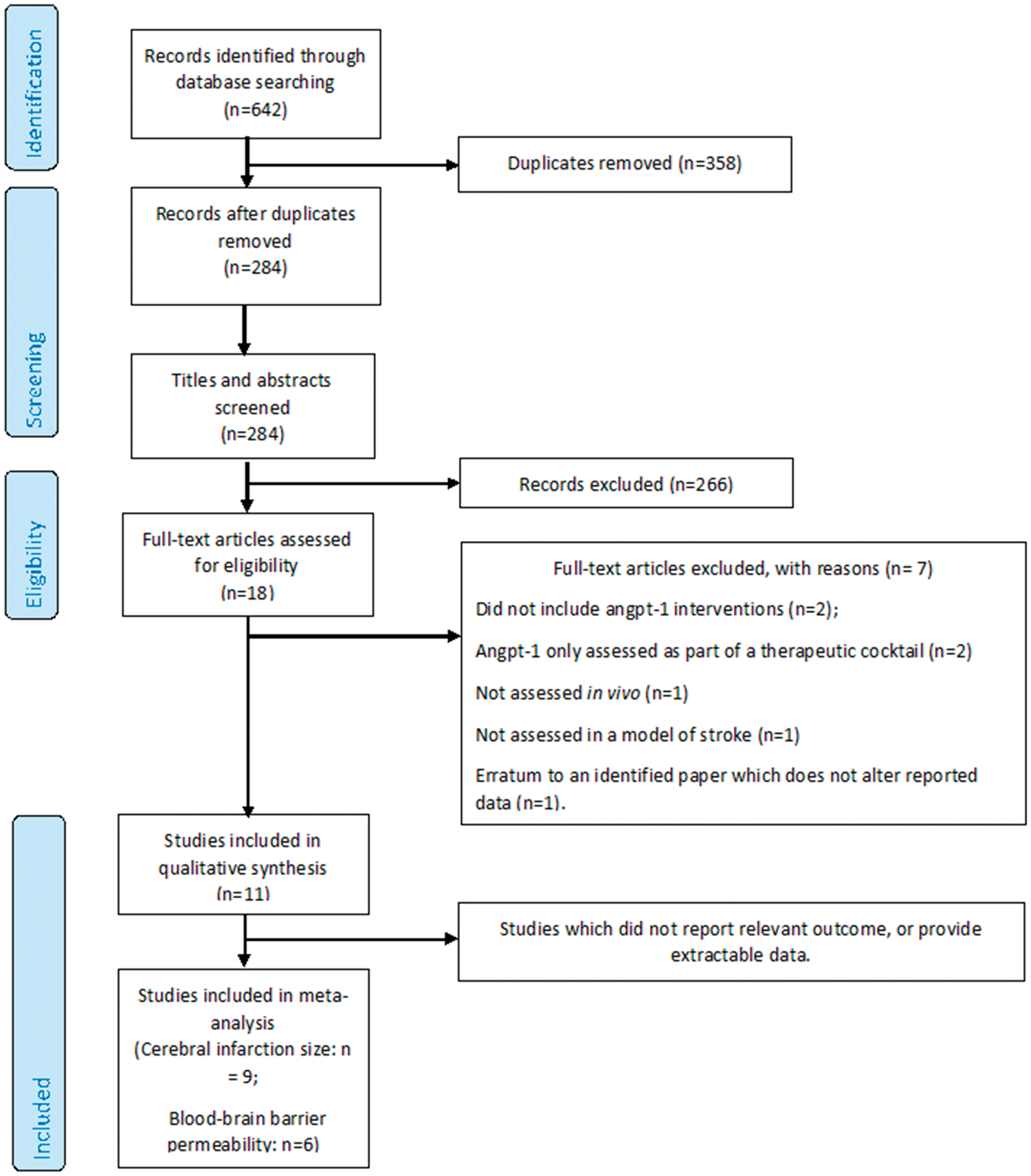
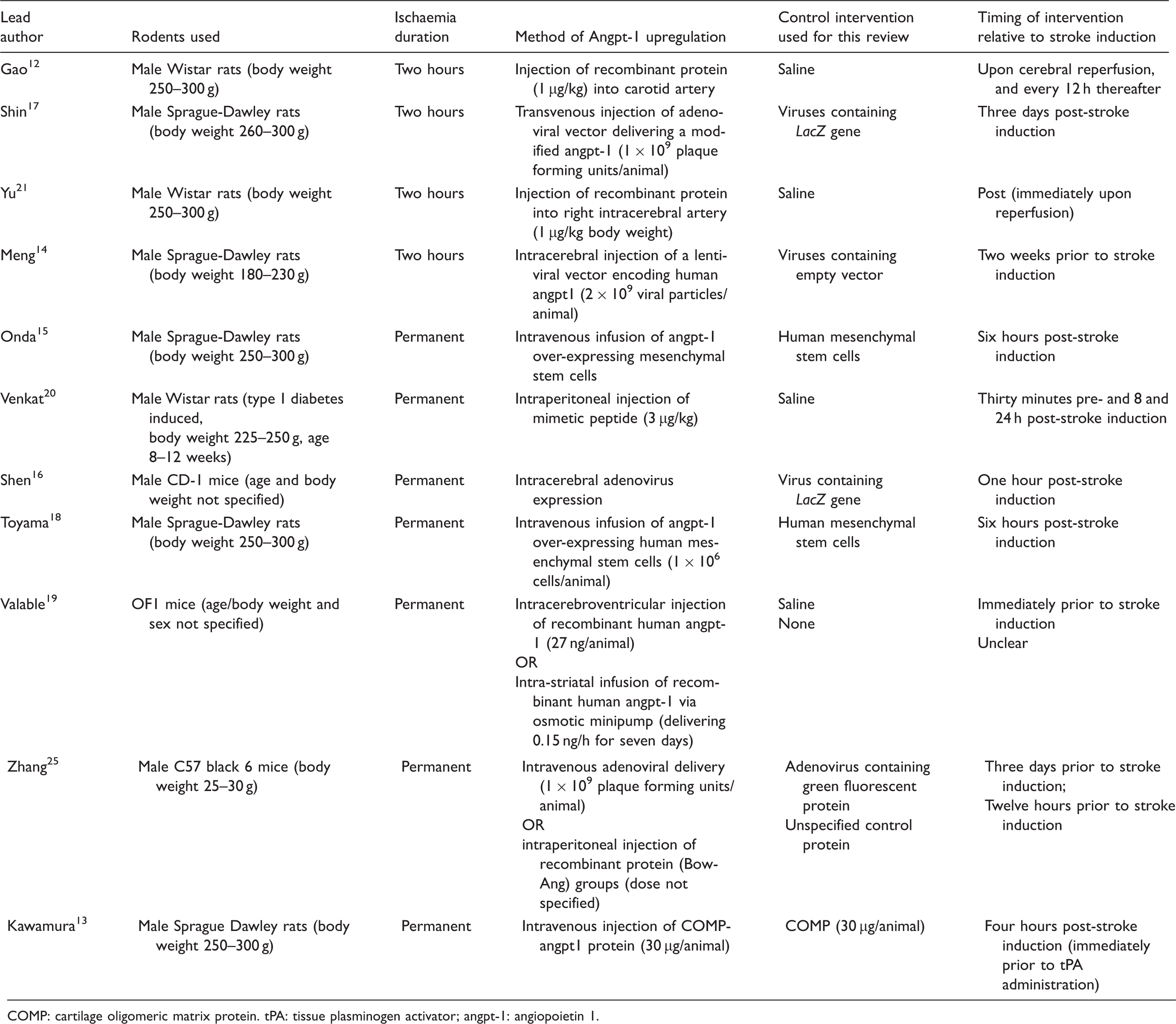
Nine studies tested a single angpt-1 intervention, two19,25 tested two angpt-1 upregulation approaches (Table 1; total of 13 interventions). Three of the interventions were virus-based, two were stem-cell-based and eight utilised proteins or peptides. Variations in the dosage, methods of delivery and timing of the angpt-1 interventions were observed. Five interventions were administered prior to stroke induction, seven were administered following cerebral ischaemia induction and the timing of one was unclear (Table 1). Recombinant angpt-1 was co-administered with tissue plasminogen activator in one study, 13 and animals receiving tissue plasminogen activator plus a control protein were used as controls in the current meta-analysis.
Quality assessment
Three studies were of low quality, six were of moderate quality and two were of high quality (Supplement 2). Only two studies reported randomising rodents to experimental groups,20,21 only one provided a sample-size calculation 13 and only two reported blinding observers. 20 No studies provided data regarding the inter/intra-rater reproducibility of the reported outcome measures.
Primary outcome assessment: The effect of angpt-1 upregulation on cerebral infarction size
Summary of outcome assessments reported by each study.
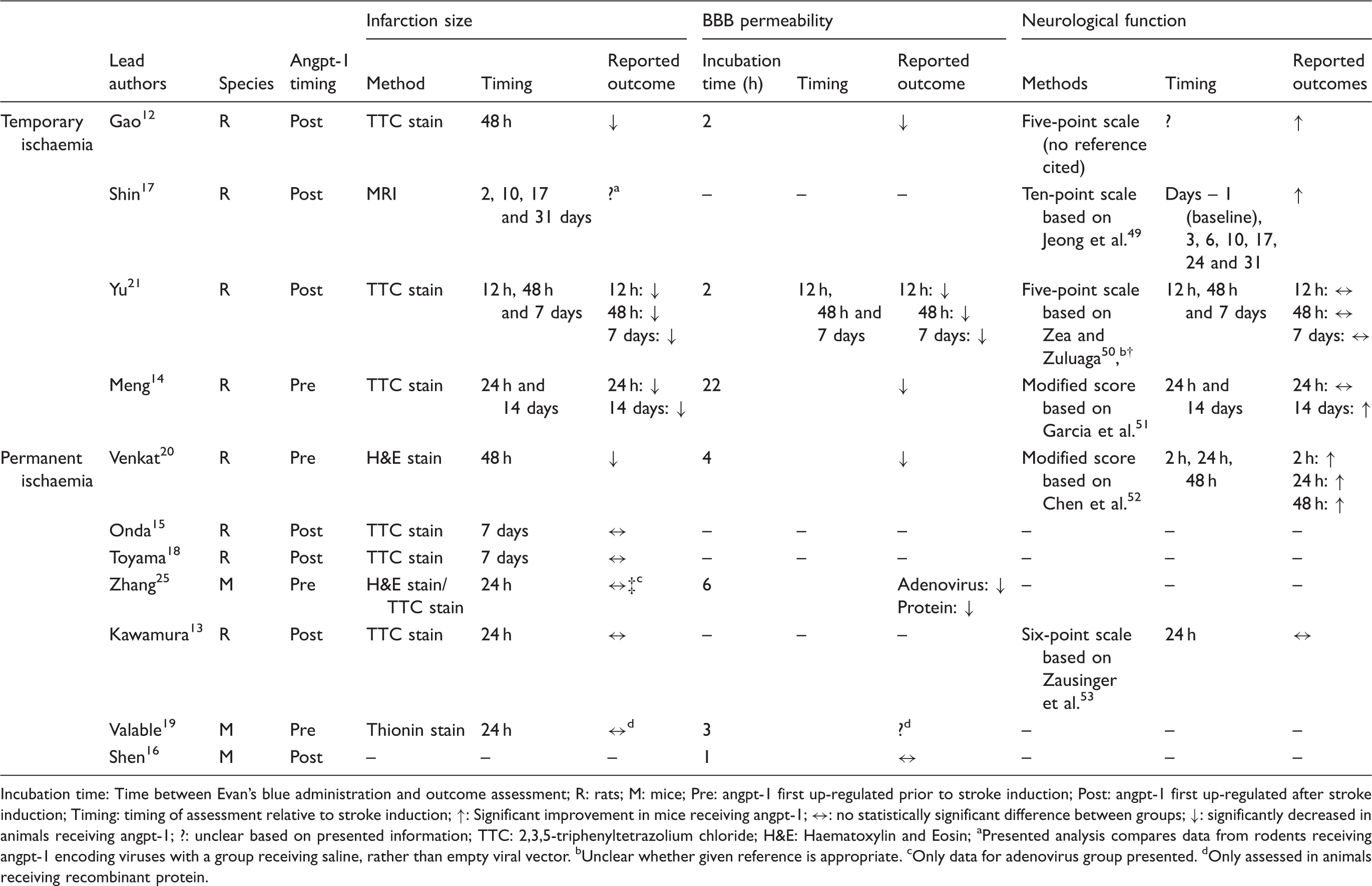
Incubation time: Time between Evan's blue administration and outcome assessment; R: rats; M: mice; Pre: angpt-1 first up-regulated prior to stroke induction; Post: angpt-1 first up-regulated after stroke induction; Timing: timing of assessment relative to stroke induction; ↑: Significant improvement in mice receiving angpt-1; ↔: no statistically significant difference between groups; ↓: significantly decreased in animals receiving angpt-1; ?: unclear based on presented information; TTC: 2,3,5-triphenyltetrazolium chloride; H&E: Haematoxylin and Eosin;
Presented analysis compares data from rodents receiving angpt-1 encoding viruses with a group receiving saline, rather than empty viral vector.
Unclear whether given reference is appropriate.
Only data for adenovirus group presented.
Only assessed in animals receiving recombinant protein.
Meta-analysis suggested that upregulating angpt-1 significantly reduced cerebral infarction size (SMD for overall effect: –3.02; 95% CI: –4.41, –1.63; Meta-analysis comparing cerebral infarction size in rodents receiving angpt-1 interventions compared to controls split by (a) duration of ischaemia and (b) timing of intervention relative to stroke induction.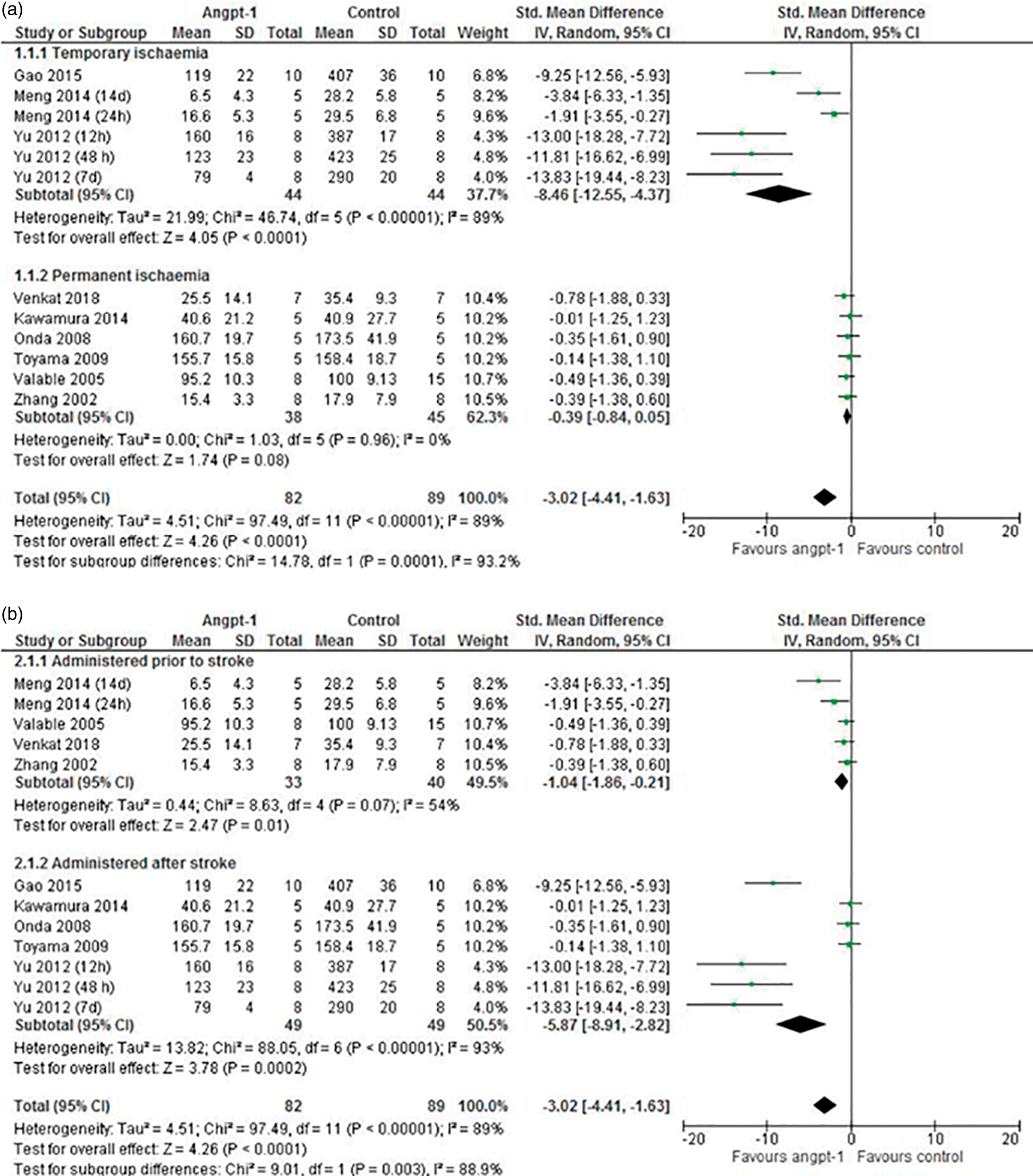
Secondary outcome assessment: The effect of angpt-1 upregulation on BBB permeability
Seven of the 11 eligible studies assessed the impact of angpt-1 administration on BBB permeability (Supplement 6).12,14,16,19–21,25 Four studies used experimental rats, and three utilised mice. Temporary cerebral ischaemia was induced in three studies,12,14,21 while permanent cerebral ischaemia was induced in four studies.16,19,20,25 All studies assessed BBB permeability using Evan's blue staining, although the timing between stain administration and tissue collection varied. Data reported by Valable et al. could not be extracted from the original paper; no response to correspondence was received and data from this study was excluded from the meta-analysis. 19 All other studies reported data as the amount of Evan's blue dye recovered per unit weight of cerebral tissue; however, results differed between studies by several orders of magnitude (Supplement 6).
Meta-analysis of findings from a total of 129 rodents from six studies demonstrated that BBB permeability was significantly lower in animals over-expressing angpt-1 compared to controls (overall SMD: –2.02; 95% CI: –3.27, –0.77; Meta-analysis comparing BBB permeability in rodents receiving angpt-1 interventions compared to controls split by (a) duration of ischaemia and (b) timing of intervention relative to stroke induction.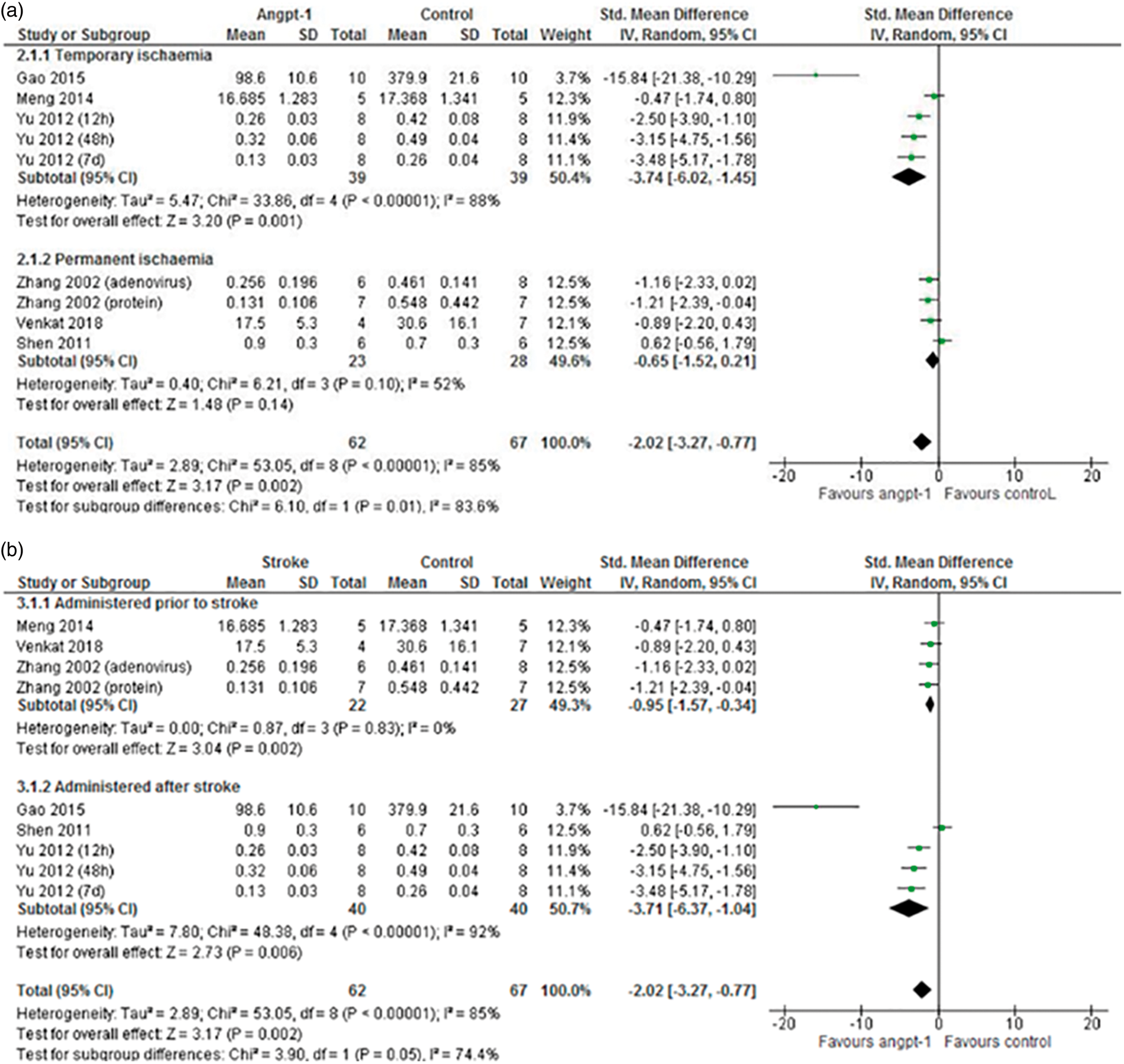
Secondary outcome assessment: The effect of angpt-1 upregulation on neurological function
Six studies (96 animals) assessed the effect of angpt-1 upregulation on neurological function in the stroke models (Table 2; Supplement 9) using standardised scoring systems to measure neurological deficit.12–14,17,20,21 All studies were performed in rats. Cerebral ischaemia was induced temporarily in four studies,12,14,17,21 and permanently in two studies.13,20 The timing of outcome assessment relative to cerebral ischaemia varied between studies. Four studies assessed neurological function repeatedly during follow-up.14,17,20,21 All studies utilised different methods to assess the severity of the neurological deficit, thereby prohibiting meta-analysis. Four studies reported that the severity of neurological deficit was significantly less in rats receiving angpt-1 upregulation during at least one of the assessed time points.12,14,17,20 The remaining two studies reported no statistically significant differences in neurological function between groups13,21 (Table 2; Supplement 9).
Discussion
This meta-analysis suggests that angpt-1 upregulation improves outcomes following temporary cerebral ischaemia, evidenced by significant reductions in cerebral infarction size and BBB permeability. Four studies also reported that angpt-1 up-regulation improved post-stroke neurological function; however, this outcome could not be meta-analysed. Care must be taken when interpreting these findings as inter-study heterogeneity in many aspects of study design including the choice of rodent model, methods used to induce cerebral ischaemia and mechanisms to up-regulate angpt-1 was observed. Only two of the 11 included studies were considered to be of high quality.13,21 Funnel plots suggested potential publication bias for the primary outcome (cerebral infarction size), however, this was difficult to objectively assess as relatively few studies were included. Finally, only male rodents were included in the identified studies. It is therefore unclear whether findings of the current meta-analysis can be generalised to females.
Cerebral infarction size was the primary outcome measure for this meta-analysis as it was the most widely reported outcome measure, and has translational potential since cerebral infarction volume has been linked with stroke severity in clinical investigations.
26
Preserving hypo-perfused brain tissue following ischaemic stroke relies on rapid restoration of the cerebral blood supply before irreversible damage occurs
27
; clinically, this can be achieved through chemical and/or mechanical thrombolysis.2,28 Data from the current meta-analysis suggest that angpt-1 upregulation may augment endogenous physiological responses. Neovascularisation, through angiogenesis or arteriogenesis, occurs after stroke in order to restore blood supply to the ischaemic tissue.
29
Angpt-1 is a key regulator of neovascularisation and stabilises blood vessels during formation and remodelling.7,30 Genetic data suggest a role for angpt-1 in the response to cerebral ischaemia as single nucleotide polymorphisms in the
Ischaemic stroke also increases BBB permeability, and this has been suggested to promote inflammation and oedema formation.36,37 Clinical studies suggest that patients with the most severe BBB disruption after ischaemic stroke experience poorer functional outcomes, than those with greater BBB integrity.36,38 Importantly, cerebral reperfusion by mechanical or chemical thrombolysis promotes cerebral oedema.4,13 The ability of angpt-1 to reduce BBB leakiness after temporary cerebral ischaemia may therefore have clinical importance.
The BBB comprises vascular endothelial cells, neurons and pericytes supported by an extracellular matrix. 39 Following ischaemia, BBB integrity is lost, although the mechanisms causing this are incompletely understood. Traditionally, stroke-related increases in vascular permeability are thought to result from disassembly of tight- and adherens-junctions between neighbouring endothelial cells, and angpt-1 has been demonstrated to play an important role in maintaining these junctions.7,8,37,39–41 Two studies included in the current review16,21 reported that cerebral expression of tight junction proteins such as ZO-1 and occludin was significantly higher in animals receiving angpt-1 than controls. Thus, the observed reduction in BBB permeability may be due to preservation of tight- and adherens-junctions between endothelial cells.
The severity of neurological dysfunction immediately following ischaemic stroke is strongly predictive of outcome at one year. 42 It has been recommended that animal stroke model research should assess neurological function.43,44 Thus, it is surprising that the effect of angpt-1 upregulation on the severity of neurological deficits was the least widely studied outcome. Moreover, there was marked heterogeneity in the methods used to measure neurological dysfunction, complicating overall interpretation of the findings; and similar heterogeneity has been noted in clinical studies.45,46 Assessing other aspects of physical function such as behaviour or exercise capacity may also further contextualise neurological function outcomes; however, only four of the included studies presented such data.14,15,17,18 As with the neurological data, inter-study heterogeneity in the manner by which behavioural assessments were conducted and reported prevented detailed analysis of these outcomes. Further studies which directly assess the impact of angpt-1 upregulation on neurological and behavioural capacity are needed to provide greater insight into the translational potential of this intervention.
In conclusion, this meta-analysis suggests that angpt-1 upregulation significantly reduces cerebral infarction size and BBB leakiness following temporary cerebral ischaemia. This systematic review highlights the need for high-quality rodent model research that includes standardised assessment of neurological function and incorporates key clinical aspects, such as upregulating angpt-1 after (not before) stroke induction and including patient co-morbidities, within the experimental design. 47 Adoption of the Animal Research: Reporting of In Vivo Experiments and Stroke Therapy Academic Industry Roundtable guidelines will also improve the quality of evidence produced from future rodent studies.44,48
Supplemental Material
JCB876876 Supplemental Material - Supplemental material for The effect of angiopoietin-1 upregulation on the outcome of acute ischaemic stroke in rodent models: A meta-analysis
Supplemental material, JCB876876 Supplemental Material for The effect of angiopoietin-1 upregulation on the outcome of acute ischaemic stroke in rodent models: A meta-analysis by Joseph V Moxon, Alexandra F Trollope, Brittany Dewdney, Catherine de Hollander, Domenico R Nastasi, Jane M Maguire and Jonathan Golledge in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JVM holds an Advance Queensland Fellowship from the Queensland Government. This research was supported by grants from the National Health and Medical Research Council (1098717, 1079369 and 1022752), Townsville Hospital and Health Service and Queensland Government. JG holds a Practitioner Fellowship from the National Health and Medical Research Council (1117601) and a Senior Clinical Research Fellowship from the Queensland Government.
Acknowledgements
We thank Jieli Chen and Poornima Venkat for providing additional information, and Georgina Anderson, Stephen Anderson and Sam Rannard for assistance with the literature search strategy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
