Abstract
The recent advances of endovascular procedures to treat stroke due to large cerebral vessel occlusion have made it possible to analyze the retrieved thrombus material. Analysis of cerebral thrombi is emerging as a relevant opportunity to complement the diagnostic workup of etiology, to develop new lytic approaches and to optimize the acute treatment of stroke due to large vessel occlusion. Nonetheless, retrieved thrombi are frequently discarded since their informative potential is often neglected and standards are missing. This review provides an overview of the current knowledge and expanding research relating to thrombus composition analysis in large vessel occlusions. We first discuss the heterogeneity of thrombogenic factors that underlie the thrombotic formation in stroke and its implications to identify stroke etiology and thrombus age. Further, we show that understanding structural characteristics of thrombus is pivotal for the development of new-targeted lytic therapies as well as to improve, through thrombus modeling, the development of thrombectomy devices. Finally, we discuss the on-going attempts to identify a signature of thrombus composition indirectly through imaging and peripheral blood biomarkers, which might in future assist treatment decision-making as well as secondary prevention. Thrombus analysis might contribute to the advancement and optimization of personalized stroke treatments.
Introduction
Ischemic stroke is the consequence of occlusion of one or multiple arteries in the brain. Blockade of blood flow by an occlusive thrombus or embolus leads to rapid and irreversible damage of the non-perfused cerebral tissue. A mainstay of acute ischemic stroke treatment is to rapidly remove the occlusion to allow recanalization and tissue reperfusion to minimize tissue injury. Afterward, it is pivotal to understand the etiology of the stroke so to set up an effective prevention strategy to avoid recurrent strokes that may occur in up to one every four-stroke patient. 1
Before the diffusion in clinical practice of endovascular thrombectomy for large vessel occlusions, the key causative elements of ischemic stroke – i.e., the arterial occlusive thrombi – were inaccessible for histological analysis, with the rare exceptions of post-mortem and experimental studies. 2 Consequently, up to most recent years, little was known about the composition of cerebral thrombi. The issue is not of secondary importance, since, differently from coronary thrombi that share mostly an atherosclerotic etiology, the diverse etiologies of brain stroke likely underpin variable thrombus compositions, which may present a different response to pharmacological and to mechanical recanalization therapies as well as may require a different secondary prevention strategy. More recently, the growing number of available thrombus samples has stimulated research to evaluate thrombus features. While initial studies on thrombi were mainly focused on morphology and categorizing thrombus components, such as fibrin, platelets, red and white blood cells, more recent studies are trying to correlate thrombus characteristics to stroke etiology, to lysis susceptibility, and to neuroradiological imaging. Moreover, findings on thrombus composition are helping in engineering ex-vivo analogs of thrombi for the development of even more effective thrombectomy devices. Thus, new insights into the pathogenesis of large vessel occlusion stroke are emerging.
Emerging applications of cerebral thrombus analysis to improve stroke care.

Heterogeneity of source of embolism and thrombogenic factors in stroke
Thrombi retrieved from a large vessel occlusion causing ischemic stroke can derive from a wide range of different sources: (i) Cardiac embolism (e.g. intra-cardiac thrombosis due to ventricular dysfunction or atrial fibrillation, embolization from endocarditic vegetations, thrombosis on non-native valves); (ii) Atheroembolism (e.g. migration of thrombi detached from unstable atherosclerotic plaques on cervical arteries or aortic arch); (iii) Local cerebral arterial thrombosis (e.g. in situ thrombosis on atherosclerotic plaques, or on diseased endothelium such as in inflammatory artery diseases); (iv) Venous embolism (paradox embolism from a venous thrombosis source associated with a right-to-left shunt) and (v) Embolization from other sources (e.g. neoplasms, fragments of vessel walls, detachment of devices like catheters, biologic valves) (Figure 1).
Résumé of possible sources of embolism and involved thrombogenic factors in large vessel occlusion stroke.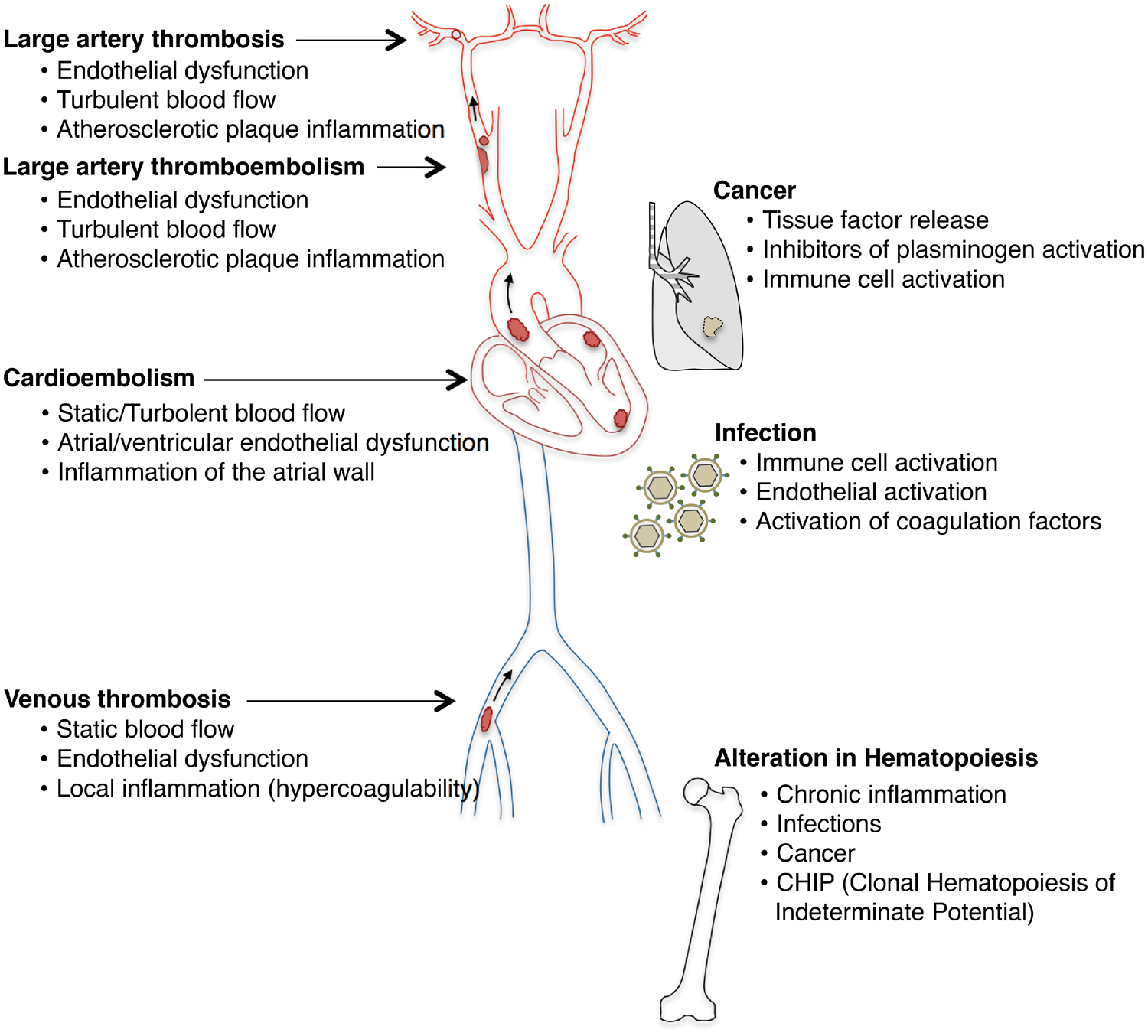
Despite intensive diagnostic workup in up to 35% of acute ischemic strokes, the etiology cannot be clarified and are therefore classified as cryptogenic. 3 Cryptogenic strokes pose multiple problems: repeated blood tests, cardiologic examinations and neuroimaging studies are performed to unravel the etiology with high costs for the health care system. Nonetheless, often the cause of stroke cannot be established timely. Consequently, since in cryptogenic stroke secondary prevention therapy cannot be tailored on the etiology, the risk of recurrence (up to 30% in the first five years 4 ) dominates.
The origin and formation of thrombus recognize indeed a complex underlying pathophysiological mechanism that characterizes its composition. For many years, thrombi have been broadly distinguished into venous and arterial. 5 Venous thrombi have been traditionally described with a predominant component of entrapped red blood cells in the fibrin structure that confers the appearance of ‘red clots’ 6 ; the low shear stress and the slow blood flow in venous vessels would allow the accumulation of erythrocytes within the thrombus structure. On the other side, arterial thrombosis usually superimposes on atherosclerotic lesions: here platelets and abnormalities of the vessel wall play a key role in thrombus formation, that occurs after an acute rupture or erosion of an atherosclerotic plaque, with exposure of the subendothelial matrix and/or activation of endothelial cells. In a series of sequential steps, platelets adhere to the injured vessel wall, additional platelets are recruited, and thrombin is activated. Arterial thrombi complicating atherosclerotic lesions have thus been described as rich in platelets, appearing as ‘white clots’. 7 This simplistic, categorical distinction between ‘red’ and ‘white’ clots has also been adopted for therapeutic purposes: ‘red clots’, venous, have been traditionally treated with anticoagulants, while ‘white clots’, arterial, have been treated with antiplatelets. 8 However, the composition of thrombi in stroke is far more interspersed due to the multiple possible etiologies. One of the reasons for the shortcomings of the categorical thrombus classification into “red” and “white”, “arterial” and “venous” is that thrombosis in stroke, depending on the source, might share common causes and similar systemic pathological conditions in a discrete continuum. Indeed ‘Virchow's triad’, namely alterations in the (i) vessel wall (endothelium), (ii) blood flow (i.e. both blood stasis and turbulence), and (iii) in blood coagulability, originally described in venous thrombosis, has been proven to be valid for arterial thrombosis as well, although with some differences. 7
Alterations in the vessel wall
One of the better-characterized factor determining endothelial dysfunction and thrombosis is the atherosclerotic plaque. The on-going lipid accumulation and inflammatory process within the plaque can lead over time to disruption of the overlying endothelial layer (unstable plaque) causing local platelet accumulation, platelet-monocyte aggregation and coagulation cascade activation leading to thrombosis.9,10 Recent findings suggest that in addition to atherosclerosis, even hypertension, atrial fibrillation, 11 cancer, venous thrombosis 12 and artery dissection play a pivotal role in causing endothelial activation and dysfunction, predisposing to thrombosis. Indeed, edema and fibrinous transformation, with areas of endocardial denudation, myocytic hypertrophy, necrosis and mononuclear cell infiltrate are recognized in atrial fibrillation as predisposing to local thrombosis. 13
Alterations in blood flow
Modifications of the physiological laminar flow of blood increase thrombotic risk. 14 In arterial stenosis, turbulent flow can cause injury, dysfunction and activation of the endothelium, as well as can promote the interaction between the endothelium and platelets and other blood cells, thus increasing the possibility of thrombosis initiation, but also opposite flows resulting in regional blood stasis can occur. Even static blood flow is detrimental: pro-thrombotic proteins (i.e. thrombin) accumulate, shifting the equilibrium toward hemostasis rather than fibrinolysis; the reduction of blood flow triggers, probably through reduced oxygen tension, 15 modifications in the endothelium, which releases P-selectin and vWF and initiates coagulation.16,17 In atrial fibrillation, abnormal blood stasis is favored by the left atrial dilatation promoted by the arrhythmia itself, and further increased in case of valvular atrial fibrillation due to mitral stenosis. Abnormal stasis in the left atrium can be visualized on trans-esophageal echocardiography as spontaneous echo contrast, which has been shown to be an independent predictor of the risk of thromboembolism. 18
Alterations in blood coagulability
Genetic or acquired abnormalities in coagulation factors or co-factors, although quite rare in adult stroke, can determine increased risk of thrombosis. In recent years, other acquired triggers for blood coagulation, such as inflammation, infection and cancer, are emerging, traditionally less considered, but likely epidemiologically more relevant.
Inflammatory cells are thought to be one of the main triggers of thrombosis both in infectious and sterile conditions, such as in immunologic and autoimmune diseases (e.g. Behçet disease (BD), Takayasu arteritis, rheumatoid arthritis, systemic lupus erythematosus, antiphospholipid syndrome). In other cases, primary dysfunction of immune cells that occur in disorders of hematopoiesis (e.g. clonal hematopoiesis of indeterminate potential (CHIP), 19 myeloproliferative diseases) can also facilitate the occurrence of thrombosis.
Neutrophils, in particular, have been in the spotlight, since they can promote thrombus formation through different mechanisms, including the release of proteases, 20 direct interactions with platelets 21 and the release of neutrophil extracellular traps (NETs).12,22,23 Neutrophils and more in general inflammation also damage endothelium reducing its physiologic anticoagulant, antiaggregant and vasodilatatory properties.24,25 Infections, as trigger of inflammation, have been recognized as possible initiators of both venous and arterial thrombosis. Infections of any severity, ranging from sepsis to more localized infections such as pneumonia and urinary tract infections, trigger the production of inflammatory cytokines, endothelial activation, leukocyte migration, and ultimately lead to a subtle prothrombotic state with altered coagulation/fibrinolysis balance and platelet activation. Inflammation and/or infections are, however, not sufficient to cause thrombosis alone, but concur, in particular in the elderly, with other abnormalities either in the blood flow (e.g. stenosis, atrial fibrillation, venous insufficiency) or in the endothelium (e.g. atherosclerotic plaque), to destabilize the underlying disease and ultimately to thrombosis.
Hypercoagulability and thrombotic manifestation, first observed by Trousseau in 1865 (also known as Trousseau Syndrome), 26 are also often found in cancer and represent an important element to be investigated in cryptogenic stroke. Cancer hypercoagulability is due to various pro-angiogenic factors, which act as potent stimulants for tissue factor expression. Moreover, activated oncogenes or inactivated tumor suppressors may increase the risk of thrombosis by inducing the expression of tissue factor and plasminogen activator inhibitor-1, a fibrinolysis inhibitor. 27 These effectors may act locally but also circulate in blood changing the composition and state of the entire coagulation system.
Although heterogeneous mechanisms co-operate in thrombus formation, it might be inferred that the final composition of the thrombus directly depends on the mechanisms of formation, 28 on the hemodynamic properties (blood flow speed, shear stress) of the vessel district in which it has formed as well as on its age. 29 This conception thus has opened new research areas that aim at identifying thrombus composition signatures from which the origin and age can be inferred.
Characteristics of cerebral thrombi and implications for determining stroke etiology and age
Thrombi are highly heterogeneous in gross appearance. 30 When analyzing cerebral thrombi, the first issue is to describe their macroscopic features possibly on the freshly extracted thrombus, before storage, to avoid artifacts. Given the great variety of stroke causes and the absence of an established and shared terminology, the histo-morphological descriptions widely vary among the studies.
Studies describing the composition and histo-morphology of cerebral thrombi retrieved in acute ischemic stroke.
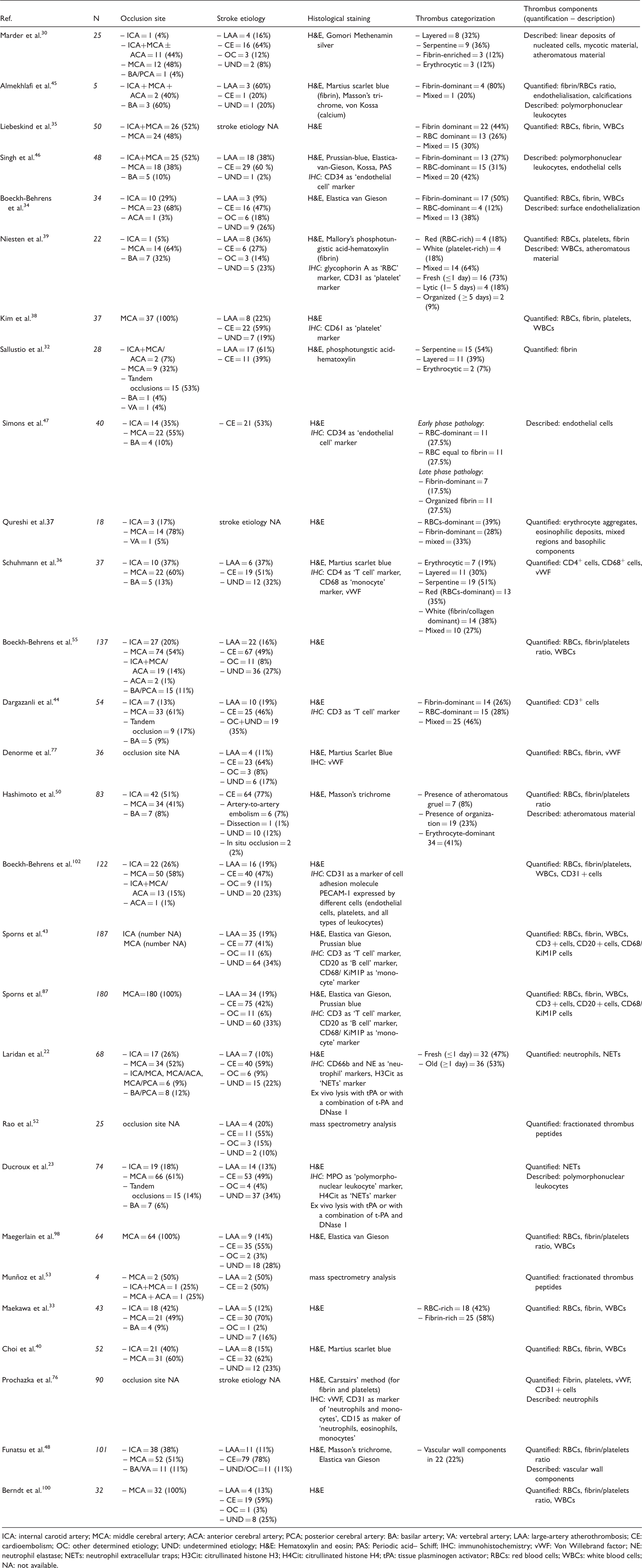
ICA: internal carotid artery; MCA: middle cerebral artery; ACA: anterior cerebral artery; PCA; posterior cerebral artery: BA: basilar artery; VA: vertebral artery; LAA: large-artery atherothrombosis; CE: cardioembolism; OC: other determined etiology; UND: undetermined etiology; H&E: Hematoxylin and eosin; PAS: Periodic acid– Schiff; IHC: immunohistochemistry; vWF: Von Willebrand factor; NE: neutrophil elastase; NETs: neutrophil extracellular traps; H3Cit: citrullinated histone H3; H4Cit: citrullinated histone H4; tPA: tissue plasminogen activator; RBCs: red blood cells; WBCs: white blood cells; NA: not available.
Although a mainstay for the comprehensive thrombus assessment, the morphological macroscopic evaluation alone gives only rough indications and loosely correlates with etiology.30,32 Therefore, studies also focused on the evaluation of the microscopic components of thrombi. Common structural elements are indeed shared by thrombi of different origin with a large variability: red blood cells (RBCs) constituting 10–70% of the thrombus are tied together with platelets (5–80% of thrombus area) into a scaffold of coagulation proteins, mainly represented by fibrin (40–80% of thrombus area) and Von Willebrand Factor (vWF, 5–50% of thrombus area). According to the predominant composition based on quantitative analyses, cerebral thrombi have been categorized into red blood cell (RBC)-dominant, fibrin-dominant, platelet-dominant or mixed, with most studies agreeing on overall prevalence of fibrin-dominant thrombi33–35 (Table 2).
Histological patterns have been also described for their organized structure as layered (platelet bands arranged in layers), serpentine (platelet bands arranged in a serpentine way) or erythrocytic (with erythrocytes and nucleated cells interspersed).30,36 Other authors identified distinct regions within the thrombus: dense erythrocytes aggregates; mixed region of dense eosinophilic amorphous material deposits with intermingled erythrocytes; eosinophilic amorphous material deposits; and basophilic staining cells in linear aggregates. 37
Importantly, populations of white blood cells (WBCs) are often found to infiltrate the thrombus structure at different proportions.34,35,38–40 Polymorphonucleates (PMNs), in particular, have been described as the most representative cellular components22,23, ranging from about 1500 to 20,000 cells/mm2. Neutrophils have been demonstrated to play a key role in experimental deep vein thrombosis,
12
coronary artery disease
41
and ischemic stroke for their ability to promote thrombus formation through different mechanisms, including the release of proteases, direct interactions with platelets and the release of NETs.12,41 Moreover, early blood neutrophil counts have been shown to correlate with outcome and stroke complications.
42
For their direct involvement in the thrombotic process in inflammatory as well as in autoimmune diseases, the presence of NETs has been recently investigated also in cerebral thrombi.22,23 Regarding other inflammatory cells, only few studies have described the presence of monocytes, T and B cells36,43,44: among these cells, despite the different methods of quantification, the prevalence of CD4+ T cells within the thrombi, ranges from 4% to 20% of the stained area.
43
Monocytes, CD68+ cells, are less represented and detectable in variable proportions within thrombi, from 0.5% to 18%, while only a small fraction of B cells could be observed36,43 (Figure 2).
Schematic drawing of the main components constituting the thrombus.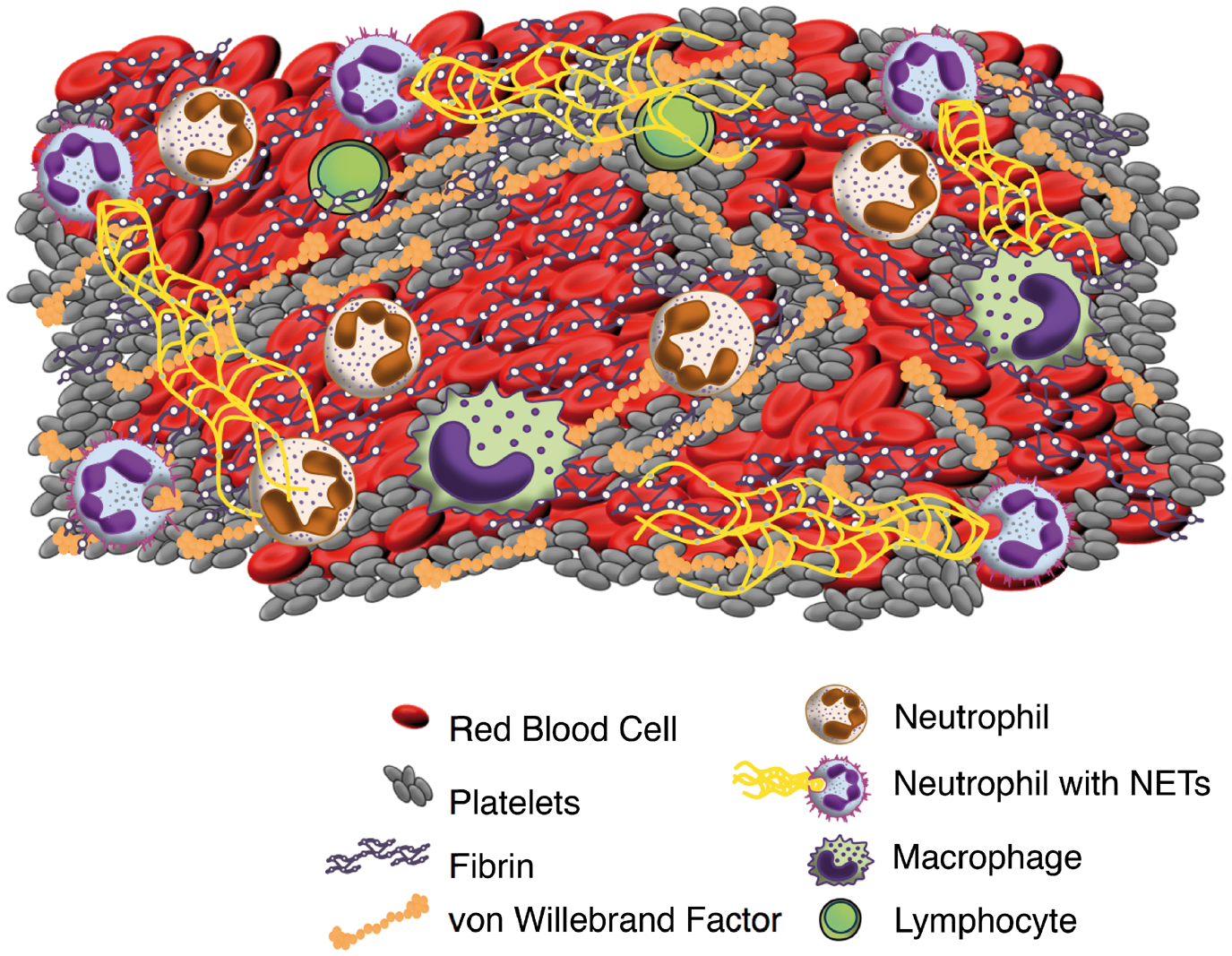
Interestingly, also the presence of endothelial cells and vascular wall components (banded collagen fibers, mostly located at thrombus periphery), has been found in some cases34,45–48 with the unresolved question about their significance, potentially deriving from the retrieval procedure or representing a degree of thrombus organization.
Cerebral thrombi might also contain other components such as those found in atherosclerotic plaques and in particular, calcifications, small atheroma with attached arterial intima and subintima, and cholesterol crystals, although only few descriptions exist.30,39,45,49,50
In the existing literature, different methods for the analysis of thrombus composition have been used; earlier reports evaluated semi-quantitatively the proportions, while more recent studies proposed the quantification in percentage of the total area, or as cells count per area. Even for the histological description, the standardization of the stains and quantification method is of utmost importance for sharing and comparing data, as suggested by De Meyer et al. 31 In particular, basic analysis of thrombi should comprehend the determination of the percentage of thrombus area positive for red blood cells, fibrin, platelets and leukocytes. 31 While various staining can be employed, it would be advisable to stain for hematoxylin and eosin, for Martius scarlet blue (to determine red blood cell and fibrin areas), as well as to perform specific immunostainings to quantify areas of platelets, von Willebrand factor and other immune cell subtypes (e.g. neutrophils, T-cells, macrophages). 31 From a technical point of view, thrombi should be stored immediately after retrieval either in a fixative (paraformaldehyde or formalin for subsequent inclusion in paraffin) or directly by freezing. The choice of the storage method should be tailored on the analysis to be performed (e.g. histological staining, RNA analysis, proteomic analysis). Most of histological stainings can be performed in paraffin-embedded specimens but some membrane antigens (in particular of leukocytes) might suffer from prolonged fixation. Other technical challenges in performing quantifications encompass the thrombus heterogeneity. 31 Ideally serial sections of the whole thrombus, to capture a fully representative sampling, should be analysed by stereological methods. 51
With the purpose of standardizing thrombus processing and analysis for better data comparison on large samples, two multicenter studies (STRIP, Stroke Thromboembolism Registry of Imaging and Pathology, led by the Mayo Clinic and COMPO-CLOT, NCT03268668, led by Fondation Ophtalmologique Adolphe de Rothschild) are ongoing, aiming to couple clot histopathology, clinical data, imaging characterization and procedural outcome measures.
Besides histological feature analysis, pilot studies have shown the feasibility of proteomic analysis by mass spectrometry on thrombi,52,53 as well as studies on RNA sequencing (RNA Sequencing Analysis in Large Vessel Occlusion Stroke DATA Bank, RNASA-LVOSB, NCT03490552) might prospect additional information to differentiate thrombi from patients with varying clinical characteristics. Even bacterial DNA retrieval, as found in coronary thrombi of patients with myocardial infarction undergoing primary percutaneous coronary intervention 41 or peripheral thrombi, 54 might be valuable, although up to now no published study on cerebral thrombi has explored the diagnostic role of bacterial signatures in collected thrombi.
A certain intrinsic bias should also be considered in thrombus studies: histopathology cannot be assessed for thrombi with refractoriness to mechanical thrombectomy as well as for thrombi completely dissolved either spontaneously or after rtPA administration; further, thrombus material cannot always reflect the whole occlusive thrombus since fragments might be lost during the retrieval procedure and thrombus morphology/shape might be altered by the retrieval technique.
Analysis of histo-morphological characteristics of cerebral thrombi for determining stroke etiology
Thrombus composition according to etiology classification.
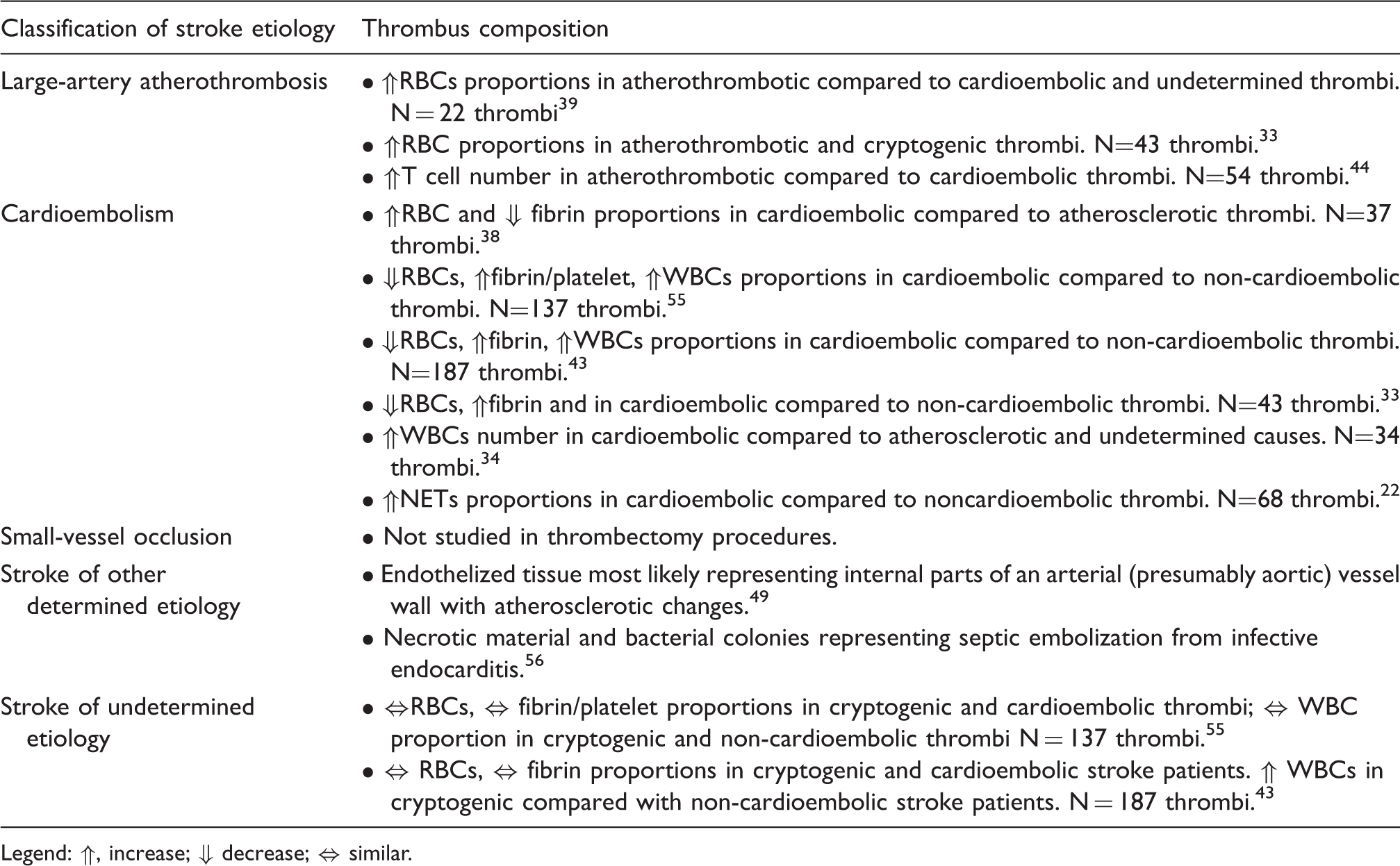
Legend: ⇑, increase; ⇓ decrease; ⇔ similar.
Early in 2014, a first report on the analysis of 22 thrombotic samples reported higher RBCs percentages in atherothrombotic thrombi than in cardioembolic and undetermined thrombi, 39 while soon thereafter an opposite finding was described. 38 Subsequent studies on larger series of thrombi33,43,55 confirmed the early findings of Niesten et al.; cardioembolic thrombi seem to encompass higher proportions of fibrin and smaller fractions of RBCs than non-cardioembolic thrombi (including stroke from large artery atherosclerosis and stroke from other determined causes in the same group). These recent studies,33,43,55 supported by a large sample size, thus question the traditional concept of cardioembolic thrombi being mostly red, erythrocyte-rich clots. Interestingly, cohorts of thrombi from cryptogenic and cardioembolic stroke patients43,55 were described to be similar in terms of the fibrin/platelet ratio and in the percentage of RBCs. The similar characteristics of these thrombus cohorts lead the authors hypothesize that a large proportion of cryptogenic strokes might have a cardioembolic origin43,55 although a prediction on single thrombi cannot still be made. Beside the differences found in the quantitative composition on cohorts of thrombi of different etiology, up to now no clear thrombus signature has been established that enables to univocally determine the single thrombus origin. In the direction of unveiling a more specific ‘signature’ of the etiology, the most recent studies have thus focused on the role of inflammatory cells in the organization of cerebral thrombi.
Indeed differences in underlying inflammatory cell infiltration of the thrombus have also been used to classify the heterogeneity of thrombi. Higher percentages of white blood cells (WBCs) in the thrombus were associated with a cardioembolic origin34,43,55 compared to non-cardioembolic thrombi. Interestingly, while neutrophil counts were not found to be associated with stroke etiology, NETs released by neutrophils were found to be in higher proportions in cardioembolic compared to non-cardioembolic thrombi. 22 A non-significant trend of higher levels of CD68/KiM1P monocytes in cardioembolic compared with non-cardioembolic thrombi was also described. 43 Atherothrombotic thrombi instead, seem to contain more CD3+ T cells. 44 However, further studies are needed to validate the data on immune cells and clarify whether other immune cell patterns might add valuable information to unravel thrombus origin.
Besides quantitative analysis of thrombus components, also histological analysis of the thrombus qualitative structure and components is pivotal since in some cases it might unequivocally be of help in clarifying stroke etiology. A recent clinical case analyzing the embolic material retrieved from a patient with middle cerebral artery stroke, who had previously undergone a trans catheter aortic valve implantation, found endothelized tissue representing internal parts of an arterial (presumably aortic) vessel wall. 49 In another case, the analysis of a cerebral thrombus, showing necrotic material and bacterial colonies consistent with septic emboli, allowed to suggest stroke caused by infective endocarditis. 56 These case reports highlight how in some cases histological thrombus analysis might be already straightforward in the diagnostic work-up on stroke etiology.
Analysis of histo-morphological characteristics for determining thrombus age
The histological composition of the thrombus might also be informative on its age. Indeed the formation of a thrombus, either occurring in territories with low speed flow or superimposed to an atherosclerotic plaque, is highly dynamic and may require relatively long periods of time before becoming symptomatic. Thus, dating thrombus formation might provide important insights into stroke etiology and in understanding the possible failure of antithrombotic therapies. Moreover, important insights also for thrombolytic therapies can be extrapolated, since in older thrombi fibrin is masked by other connective and inflammatory components thus making it less accessible to fibrinolysis processes.
Thrombus maturation has been particularly studied in venous thrombosis (VT), in which the incidence of relapses and chronic complications is elevated.57,58 Venous thrombi undergo a process of maturation and remodeling steps resembling wound healing59,60 in which leukocytes actively participate. Discrete maturation phases have been described. The acute thrombus is described as fibrin dominant. At this early stage, the structure is highly dynamic: alternate layers of fibrin (laminations) and cells (mainly red blood cells but also platelets) are deposited for the continuous blood flow forming the so-called ‘Zahn's lines’ (after Friedrich Wilhelm Zahn). Depositing platelets attract neutrophils that become the dominant leukocyte subtype; moreover, neutrophils release a network of histone and extracellular DNA (NETs) that is often visible. The fibrin-dominant thrombus evolves in approximately five to seven days 61 in the intermediate thrombus. Poorly organized collagen deposits by incoming fibroblasts characterize the intermediate thrombus that also bears initial signs of neovascularization. As the thrombus becomes more structurally organized, the leukocyte infiltration shifts from neutrophil toward monocyte/macrophages invasion; importantly, hemosiderin-stained macrophages (so called hemosiderophages) can be identified. 59 The thrombus evolves towards organization in 7 to 10 days usually in a periphery towards center declination. In the chronic organized thrombus (one to three months), macrophages dominate among leukocytes, microvessel network become more and more defined, acellular connective tissue forms the prevalent structure and often the thrombus is incorporated into the vessel wall and becomes endothelized.
Composition and age evaluations have been made also for coronary thrombi,62–64 in which a pattern of structural and cellular evolution steps very similar to venous thrombi was described.
64
Current age categorization for coronary thrombi in the literature, referring to the work of Rittersma et al.
62
distinguishes fresh (less than one days old), lytic (1–5 days old) and organized (more than five days old) thrombi.
Although in previous studies the estimated thrombus age did not differ between the different stroke etiology,22,39 its evaluation can give important indications on thrombus remodeling, thus revealing possible thrombotic on-going processes as well as plaque disturbances before the acute clinical symptoms. 62 It is therefore of utmost importance to refine methods to objectively stage thrombus age using both microscopic features and possibly additional markers such as hemoglobin degradation steps.65,66
Thrombus characteristics and potential implications for reperfusion therapies
Thrombus characteristics and susceptibility to lytic treatments
Knowledge of the structure and composition of the thrombus is essential to envisage and design pharmacological treatments able to lyse the thrombi. In the 1980s, studies on activators of the fibrinolytic system led to the synthesis of tissue plasminogen activator that could be administered systemically to reduce the clot burden in ischemic disorders. 67 While recombinant tissue plasminogen activator (r-tPA) remains the main pharmacological treatment of acute stroke, new therapeutic approaches to tackle other thrombus components are being developed. The extended use of stent retrievers enabling thrombus studies in patients with stroke is providing further evidence in this field, together with imaging techniques that provide a definition of thrombolysis responsiveness or resistance.
Lysis of the fibrin component
Within the thrombus, tPA binds to fibrin and converts the entrapped plasminogen to plasmin leading to fibrinolysis. Rates of arterial recanalization after intravenous r-tPA treatment are relatively low and estimated to be around 20–46% depending on the study,68,69 on the site of occlusion, delay of treatment and ascertainment of revascularization. The causes of the failure of recanalization despite thrombolysis, the so-called “r-tPA resistance,” are still partially unexplained but may depend on the location of the occlusion, the dimensions, composition and age of the thrombus. 70 Thrombus architecture and composition is an important aspect since r-tPA tackles the fibrin component. 71 Previous in vitro studies showed that response to r-tPA is associated with fibrin structure, 70 that is influenced by the concentration of RBCs within the clot.72,73 In experimental studies, some reports highlight higher r-tPA resistance in thrombi enriched in RBCs entrapped within fibrin, 74 as well as others report more vulnerability of erythrocyte-rich thrombi compared to platelet and fibrin-rich clots. 71 The efficacy of thrombolysis has been observed also to depend on platelet enrichment. 37
Lysis of the von Willebrand factor meshwork
It has been shown that platelet crosslinking to fibrin during arterial thrombosis/embolism involves von Willebrand factor (vWF) multimers. 75 This latter has been recently demonstrated to be present in cerebral thrombi extracted during mechanical thrombectomy.76,77 Proteolysis of vWF might thus be a promising target to disaggregate platelet-rich thrombi. In this regards, N-Acetylcysteine (NAC, a clinically approved mucolytic drug) has been described to reduce intrachain disulfide bonds in large polymeric proteins. 78 In three different models of experimental stroke, it has been shown that intravenous injection of NAC in particular when combined with antagonist of platelet GpIIb/IIIa, induced stable arterial recanalization and improved ischemic lesion size and neurological deficits. 79 Accordingly, administration of the vWF-cleaving enzyme ADAMTS13 in experimental stroke was able to dissolve r-tPA-resistant vWF rich thrombi resulting in fast recanalization and reduced cerebral infarct sizes. 77 Indeed, also in acute coronary syndrome, a strong correlation between elevated vWF and the incidence and outcome of heart ischemia has been observed, further underscoring the importance to target vWF in ischemia. 80 Interestingly, first generation inhibitors of the vWF-GPIb have been studied in clinical studies in diverse thrombotic diseases (clinical gov trial, NCT02553317, NCT00742612).
Lysis of the neutrophil extracellular traps
Another stabilizing structural component under investigation for targeted lytic approaches are NETs. It has been shown that the abundant presence of stabilizing NETs in coronary thrombi could be counteracted by deoxyribonuclease (DNAse) I treatment that accelerates their lysis. 41 Interestingly in experimental stroke, DNase I treatment resulted in a protective in vivo effect in murine models of ischemic stroke 81 as well as in vein thrombosis. 12 The presence of NETs was confirmed even in human cerebral thrombi and DNase treatment combined to standard r-tPA, was shown, at least ex vivo, to increase thrombus lyisis, providing proof of concept of the utility to tackle NETs.22,23
In conclusion, study of thrombus composition can unravel new targets for the development of innovative lytic approaches. While r-tPA remains mainly active on fibrin, other constituents of the thrombus might be directly targeted by other specific treatments. In this direction, studying the role of the other structural elements in thrombus formation or stabilization, for example vWF, inflammatory cells and their by-products (e.g. NETs), is an important on-going research field that likely will soon deliver new therapeutic options.
Thrombus composition to optimize retrieval techniques
Effective recanalization of occlusive thrombi in large cerebral arteries is influenced by a number of underlying factors, including retrieval technique, operator experience, anatomy and occluded vessel type, but also by the clot composition. 82 Studying the thrombus composition, its physical properties (as stiffness or softness) and the device-clot interactions may unravel possible associations between thrombus characteristics and procedure outcome (speed and rate of recanalization) or complications (for example, rates of fragmentation and distal embolization). To this purpose, studies on human retrieved cerebral thrombi should include the assessment of procedure times (onset to recanalization time, groin puncture to recanalization time), number of procedure maneuvers, measures of the success of recanalization (e.g. the modified or extended Treatment In Cerebral Ischemia, TICI score), assessment of procedure complications (i.e. distal embolization), besides morphological and histological characterization of the retrieved thrombus. The knowledge of such associations is becoming more and more important in clinical practice to guide device selection, optimize the procedures and to develop new retrieval devices.
Recently published studies have suggested that clot composition can affect the ideal choice of technique used during clot retrieval since it might play a role in device-clot interaction. 83 In experimental models, blood vessels occluded with fibrin-rich thrombi needed a greater number of thrombectomy attempts, and thus longer recanalization time, compared to thrombi rich in red blood cells, since RBC-rich thrombi were easily mobilized and demonstrated more fragmentation. 84 In parallel, ex vivo observations on experimental clot analogs with different compositions of RBCs showed that RBC-rich clots began to slide earlier on a consecutively more angulated surface, indicating that the friction in non-RBC-rich clots was higher than in RBC-rich thrombi. 85 This might contribute to their relative resistance to clot removal during thrombectomy. Indeed, the mechanical characteristics of a thrombus (e.g. friction, softness or stiffness, and their change over time and repeated attempts of retrieval) might also affect the interaction ability between thrombus and stent-retriever device or suction catheter, thus influencing the capability to remove it and potentially requiring adapted devices. Similarly, the fragmentation potential of a cerebral thrombus could lead to clinical repercussions by distal embolization, potentially preventable with the additional use of protection devices (e.g. proximal balloon catheters for flow arrest 86 ) or further technical modifications.
In human stroke, analogously to experimental studies, cerebral thrombi composed of organized fibrin were found to necessitate longer recanalization time when subjected to thrombectomy procedures.47,87 Instead, strokes with erythrocyte-rich thrombi necessitated a smaller number of recanalization maneuvers and shorter procedure times,33,87 and were associated to better reperfusion scores. 50 As for the inflammatory component, higher proportion of leukocytes has been shown to be associated with extended mechanical recanalization time, less favorable recanalization and clinical outcome. 34
Data about the potential for fragmentation, migration and/or embolization of cerebral thrombi in relationship to their composition are still controversial. In a series of cases, a significant association was observed between higher amounts of fibrin, lower percentage of red blood cells and secondary embolism. 87 Partially in contrast, a study observed that a higher portion of RBC-rich thrombi showed the phenomenon of thrombus migration, defined by the distal location of the thrombus by the first digital subtraction angiography (DSA) compared to the pre-interventional computed tomography angiography (CTA). 88
Further, refinements in thrombus synthesis, by the preparation of clot analogs made on the bench,83,89 will facilitate the development of new and refined techniques in the thrombectomy procedure of acute ischemic stroke; in vitro occlusion models might also be used for training in interventional techniques. Moreover imaging biomarkers of thrombus composition would be highly desirable as aid to predict recanalization success in intravenous thrombolysis and mechanical thrombectomy and to guide device selection, thus advancing the possibility of efficacious treatment personalization.
Thrombus features and biomarkers on imaging and blood
Thrombus composition is emerging as an important indicator not only of stroke etiology, but also of stroke treatment procedural options. It thus becomes critical to develop correlations between thrombus characteristics and imaging features, in order to possibly predict thrombus composition in the acute stroke. Imaging biomarkers could become an important tool to optimize pharmacological or endovascular treatments. The presence of a Hyperdense Artery Sign (HAS) on the basal CT scan or a Susceptibility Vessel Sign (SVS) in the MRI scan is commonly encountered in the acute phase of stroke and indicates the presence of a large vessel occlusion on that site. However, these neuroradiological signs are absent in about 50% of thrombotic occlusion 90 exemplifying how thrombi of different composition might have diverse imaging features. These imaging characteristics might thus be exploited to unravel thrombus composition. The presence of HAS on CT scan has been indeed shown to be associated with higher red blood cell content within the retrieved thrombus.33,35,39,47 By measuring the vascular density in Hounsfield Units (HU) adjusting for the density of the contralateral MCA (rHU = HUThrombus/HUContralateralMCA), a correlation was found between high rHU and high amounts of red blood cells38,87 and between low rHU and high fibrin content. 87 This is consistent with previous data from experimental studies and investigations upon venous sinus thrombosis, showing a linear correlation between the concentration of hemoglobin and the attenuation of the thrombus on CT, while platelets and atheromatous or cellular debris are known to decrease this attenuation.91,92 Similarly, studies investigating the MRI brain scans in the stroke due to large vessel occlusion observed a higher proportion of RBC in cerebral thrombi characterized by a positive SVS.35,38,40 The prediction of the RBC-dominance of an intravascular cerebral thrombus could have important implications: higher proportions of RBCs have been shown to be associated to higher response to thrombolysis, shorter procedure times, higher recanalization rates and likely higher risk of distal embolization. Interestingly and in line with these results, some studies have suggested that the presence of HAS or SVS might be associated with successful recanalization,93,94 while other have shown no association. 95 Controversial results still exist regarding stroke etiology: some CT studies found an association between the HAS and a cardioembolic etiology, whereas others found no association 96 ; a meta-analysis examining the association between etiology and HAS found no association between imaging characteristics and stroke etiology. 97 Recently, it has been observed that thrombus perviousness (defined as a change in thrombus attenuation between basal CT and CTA) shows a correlation with lower fractions of red blood cells counts and more fibrin/platelets conglomerations, concurrent with an association with cardioembolic origin. 98 The identification of a specific pattern or “signature” on the retrieved thrombus of stroke etiology is still needed before being able to combine imaging biomarkers to reveal its identity.
Besides imaging as valuable tool to indirectly unravel thrombus composition, also blood biomarkers are being investigated. In heart ischemia circulating platelet activation marker sCD40 ligand was found to correlate with thrombus composition in terms of platelet content and negatively with its fibrin content. 99 Another study identified Profilin-1 levels in plasma to inversely correlate with Profilin-1 content in coronary thrombi and to directly correlate with thrombus age. 29 Regarding cerebral thrombi, in a single series of cases, a significant relationship was found between plasma vWF and the proportions of vWF, fibrin and platelets in the thromboembolus. 76 Moreover, an on-going trial is exploring the correlation between circulating plasma levels of diverse proteins (eg. vWF, fibrin) and its presence in the thrombus (clinicaltrials.gov NCT03268668). To this purpose, both imaging and peripheral blood biomarkers could be useful indicators and should be explored while studying thrombus composition.
Conclusion
The diverse causes and pathogenic mechanisms underlying thrombus formation and large cerebral artery occlusion in stroke are very peculiar and open up the possibility to exploit thrombus composition analysis to ameliorate stroke care and understanding of stroke pathogenesis. On-going research is projected in particular towards analyzing retrieved cerebral thrombi, in order to improve stroke care, comprehending assessment of: (i) age of thrombus formation; (ii) thrombus origin and stroke etiology; (iii) susceptibility/resistance to thrombolytic treatments; (iv) procedural thrombectomy clues (e.g. more adapted devices and optimization of the retrieval possibility). Since time is an important factor in stroke care, histological analysis of the thrombus might be only indicated for secondary stroke prevention strategies and for deepening understanding of pathophysiology. Importantly, imaging and peripheral blood biomarkers, by inferring thrombus composition cues, will be a valid tool in the acute phase to predict recanalization feasibility using either pharmacological and/or mechanical thrombectomy and to guide device selection. Circulating thrombus biomarkers could further contribute to the choice for the best secondary prevention, even when the thrombus is not retrieved or not available for histopathological analysis. From the reported literature studies, it is clear that thrombus analysis is complex and cannot rely simply on gross morphological descriptions, but deserves sophisticated analysis. While initial studies used mainly haematoxylin and eosin staining to evaluate thrombi composition, the spectrum of histological staining to describe thrombus characteristics is becoming more complex and a plethora of thrombus composition features is needed to properly depict the thrombus. While comparing the same feature over the spectrum of stroke etiologies revealed for some components (e.g. red cell blood areas, number of neutrophils, NETs) a statistical significance, unfortunately these measures are still poor in identifying on the single patient stroke etiology; specificity and sensitivity are quite low and a definite “composition signatures” is still lacking. Further studies are required to identify a sensitive and specific thrombus signature while linking imaging and blood biomarkers. Nonetheless, in some specific cases, histological analysis of the thrombus is already informative, and should be attempted whenever possible.
Keypoints
Histo-morphological characteristics of the thrombus can indicate etiology. Age of the thrombus can be estimated by histological analysis. Analysis of thrombus composition might open to new lytic treatment investigations to tackle thrombus scaffold. Establishing biomarkers might support in predicting the thrombus composition. Unraveling thrombus composition can lead to optimize endovascular strategies.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' contributions
All authors contributed to the work presented in this paper. All authors made critical revision of the manuscript.
